Mice Lacking the Calcitonin Receptor Do Not Display Improved Bone Healing
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Surgical Procedure
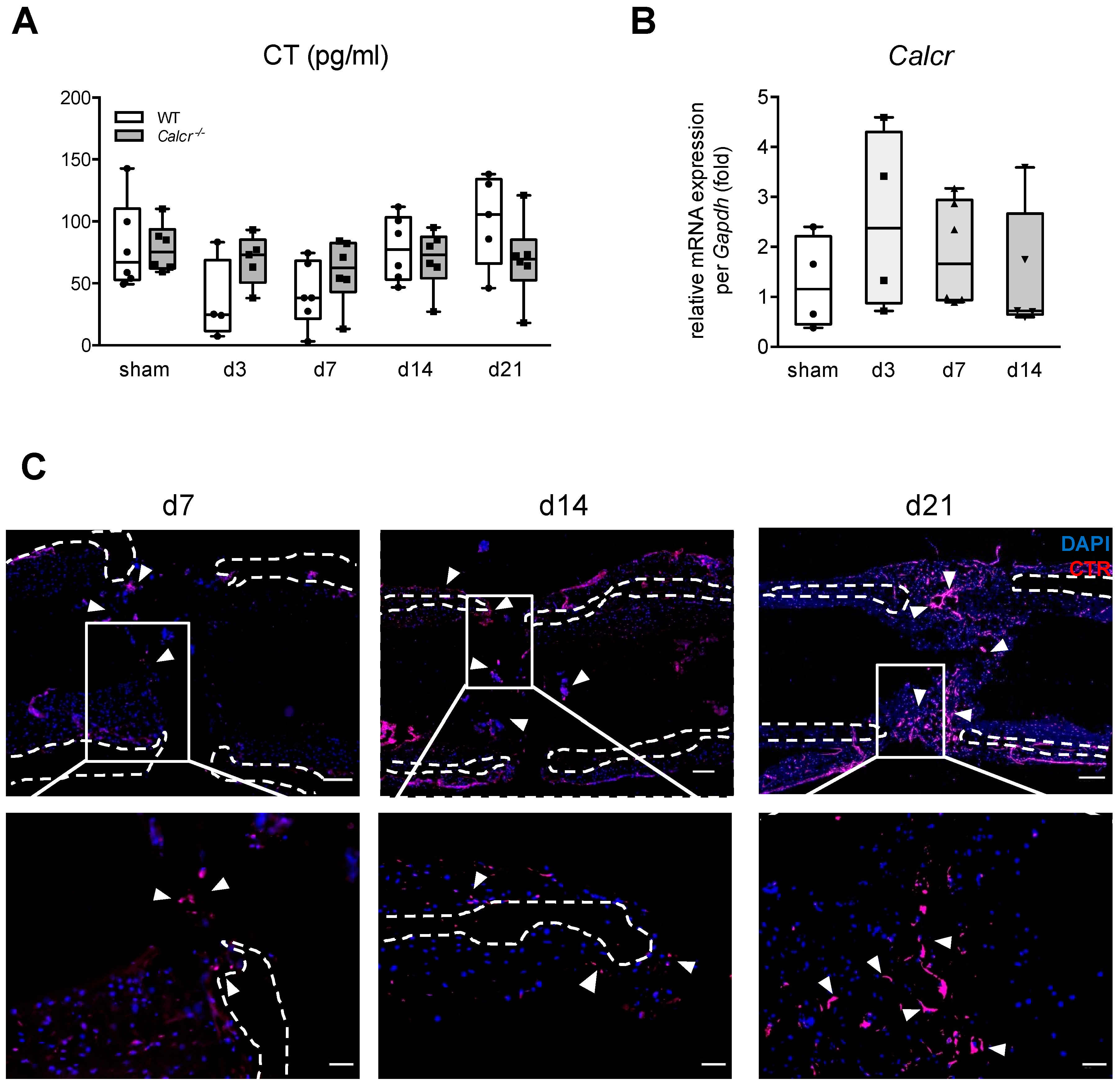
2.3. Serum Analysis
2.4. Gene Expression Analysis
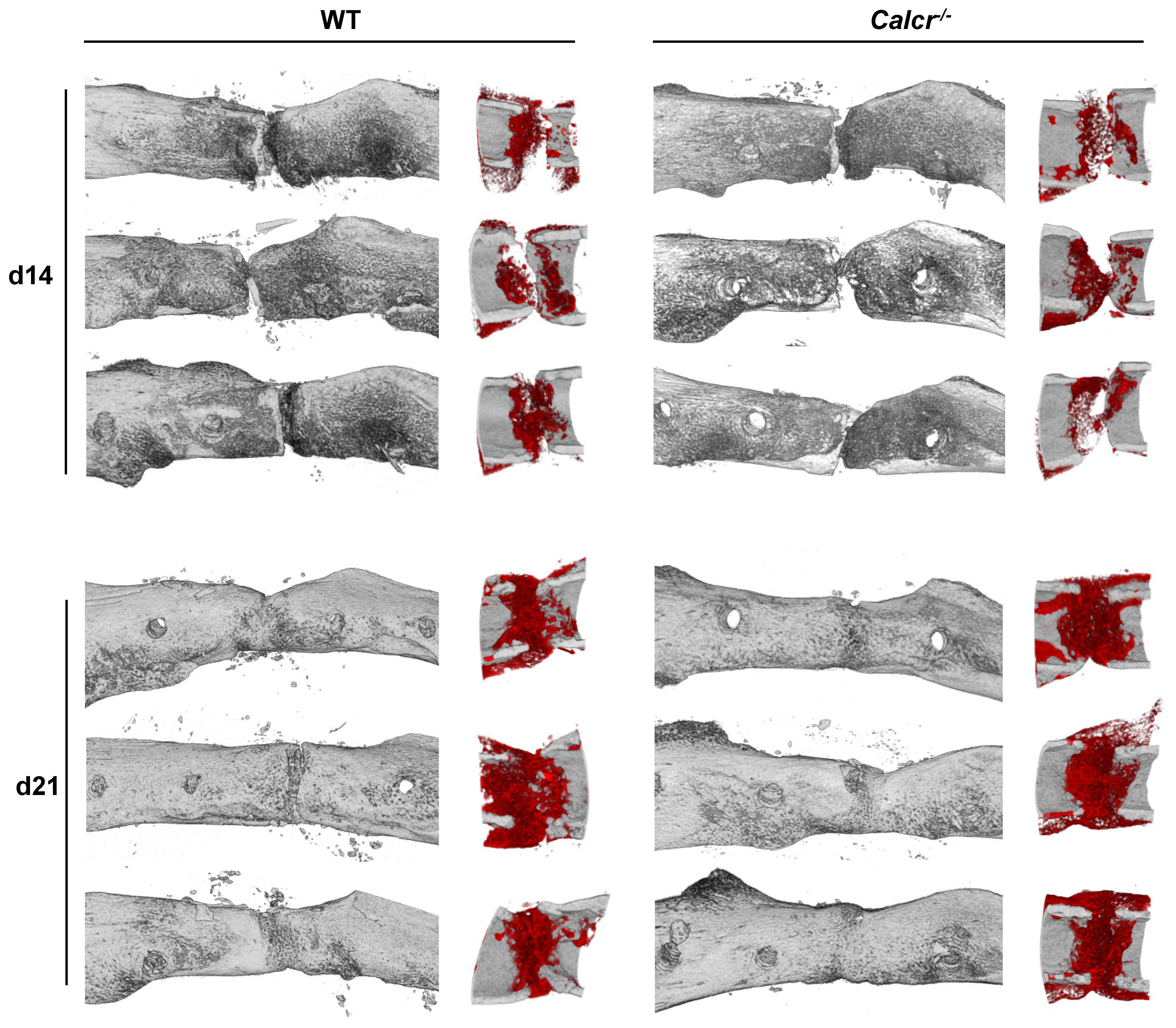
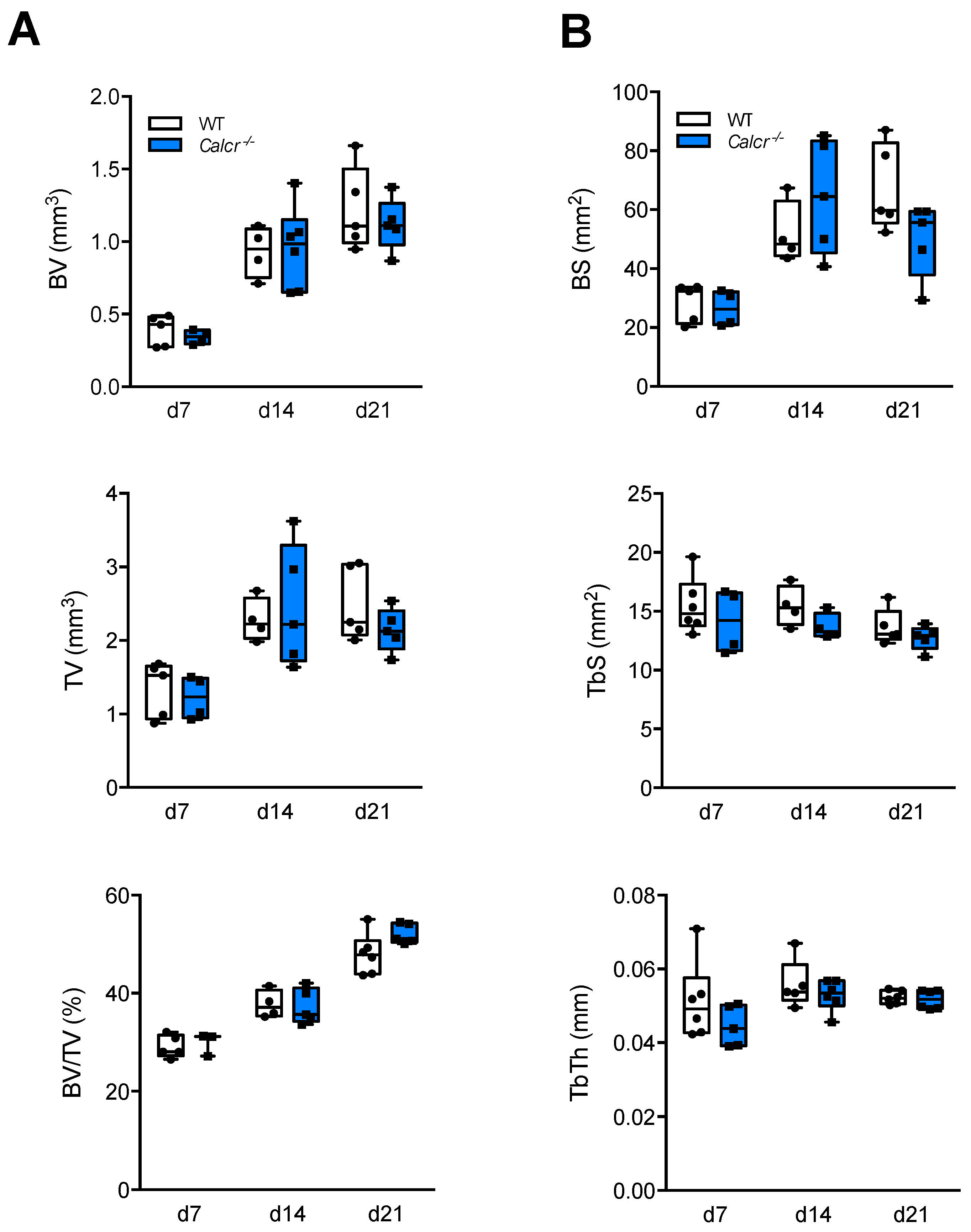
2.5. µCT Analysis
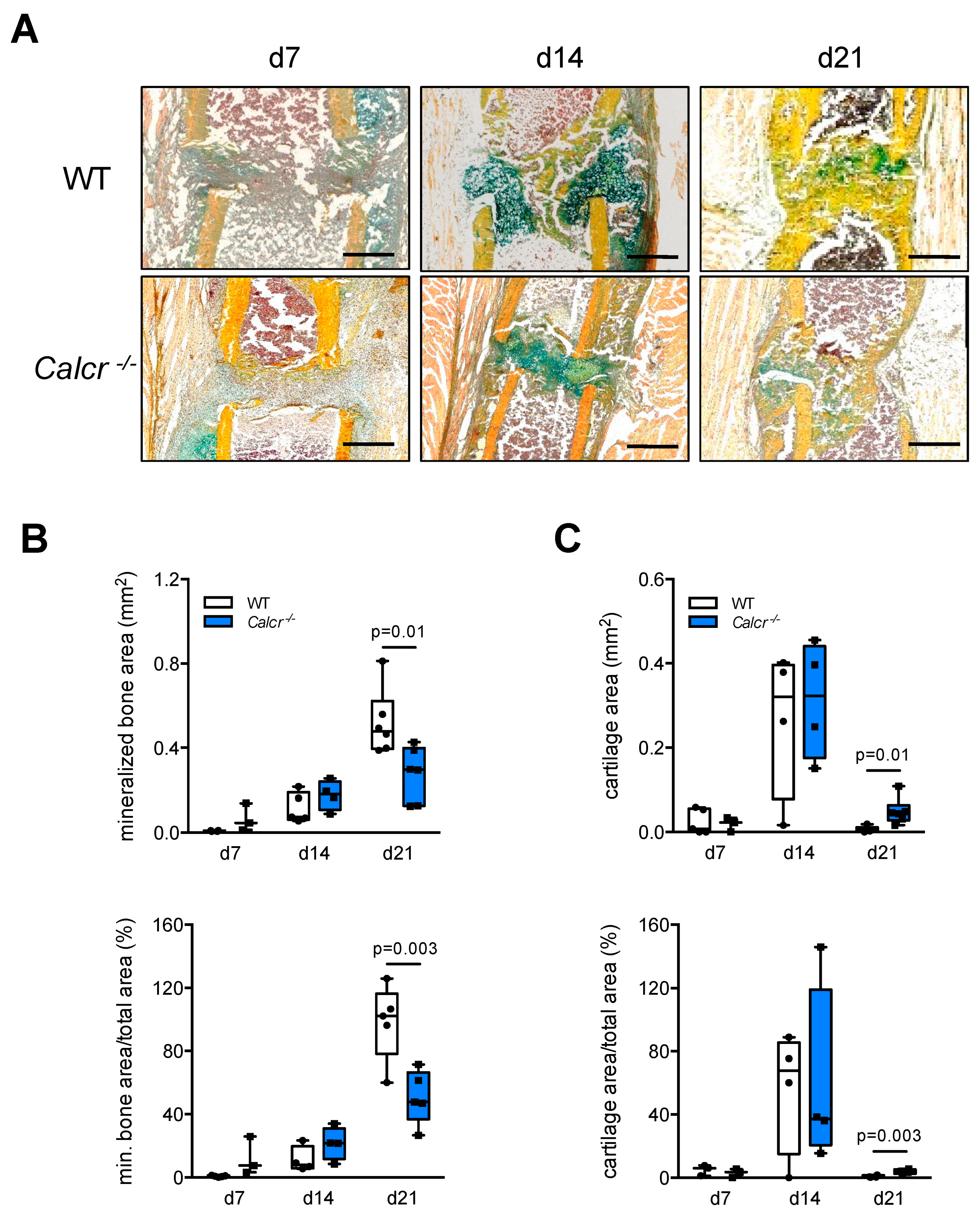
2.6. Histomorphometric Analysis
2.7. Histology
2.8. Fluorescent Immunohistochemical Staining
2.9. Statistical Analysis
3. Results
3.1. CTR Is Expressed in Regenerating Bone
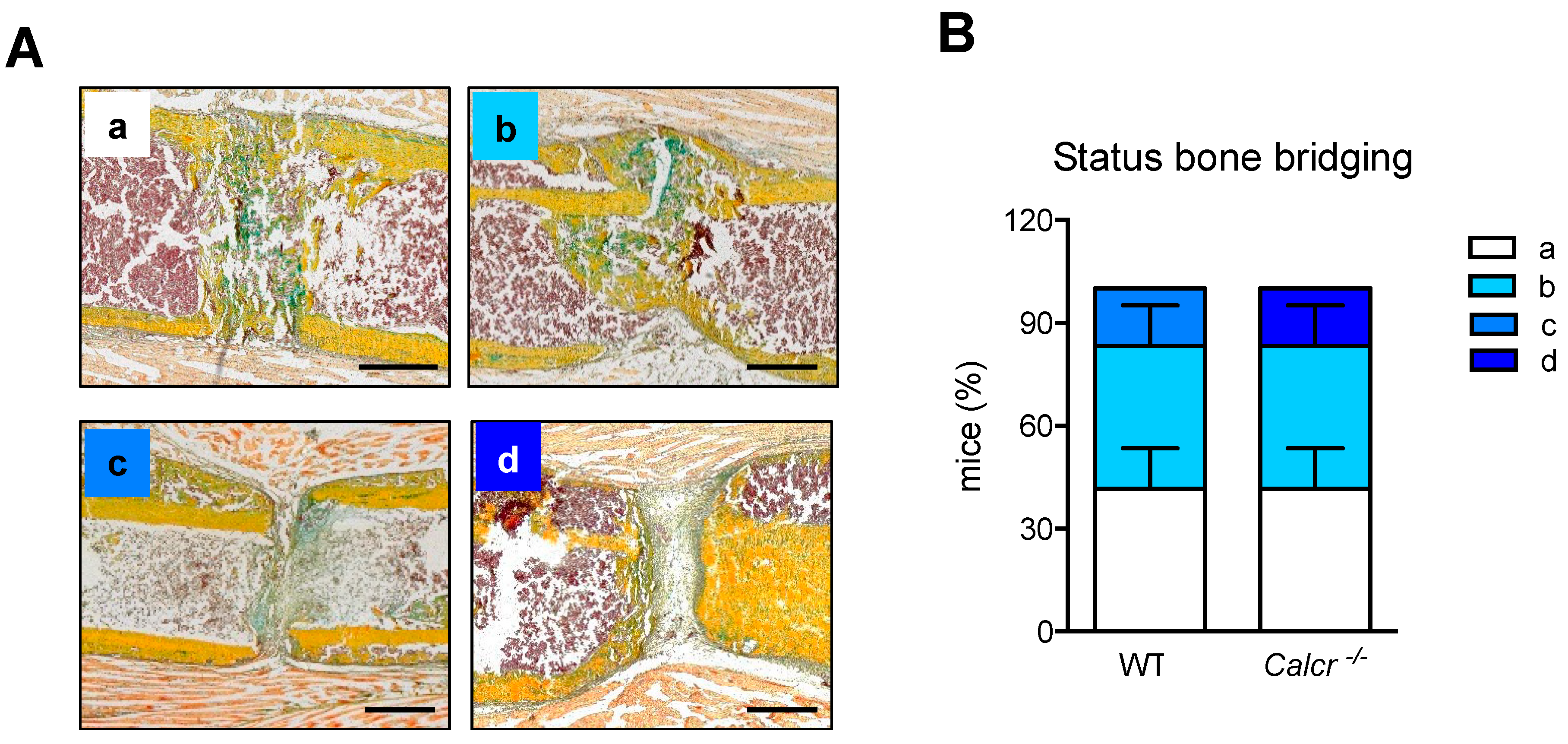
3.2. Assessment of Bone Regeneration in CTR-Deficient Mice
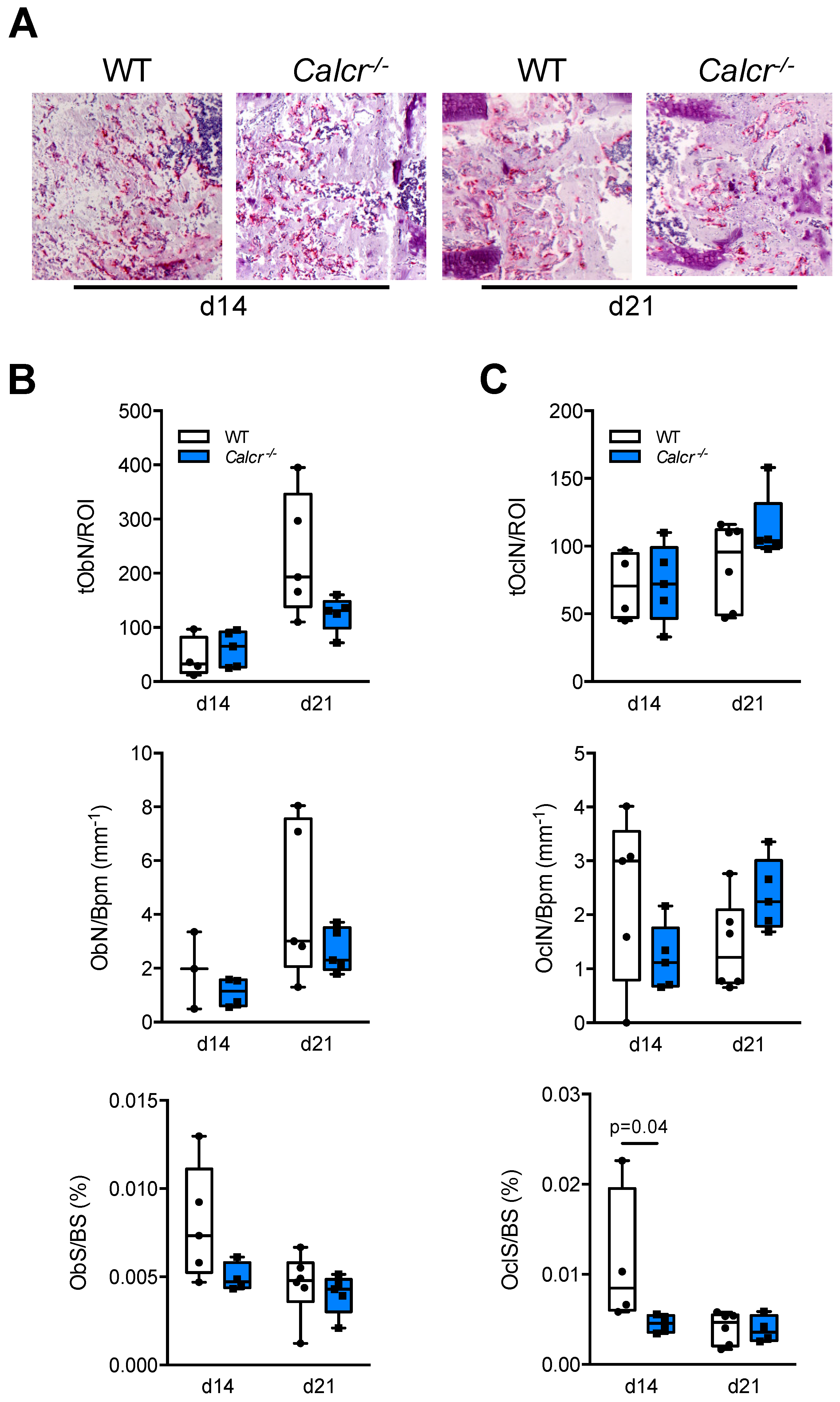
3.3. Assessment of Cellular Callus Remodeling in CTR-Deficient Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tzioupis, C.; Giannoudis, P.V. Prevalence of long-bone non-unions. Injury 2007, 38 (Suppl. 2), S3–S9. [Google Scholar] [CrossRef]
- Hak, D.J.; Fitzpatrick, D.; Bishop, J.A.; Marsh, J.L.; Tilp, S.; Schnettler, R.; Simpson, H.; Alt, V. Delayed union and nonunions: Epidemiology, clinical issues, and financial aspects. Injury 2014, 45 (Suppl. 2), S3–S7. [Google Scholar] [CrossRef]
- Claes, L.; Recknagel, S.; Ignatius, A. Fracture healing under healthy and inflammatory conditions. Nat. Rev. Rheumatol. 2012, 8, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Petersen, A.; Dienelt, A.; Schwarz, C.; Duda, G.N. Initiation and early control of tissue regeneration—Bone healing as a model system for tissue regeneration. Expert Opin. Biol. Ther. 2014, 14, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Hoff, A.O.; Catala-Lehnen, P.; Thomas, P.M.; Priemel, M.; Rueger, J.M.; Nasonkin, I.; Bradley, A.; Hughes, M.R.; Ordonez, N.; Cote, G.J.; et al. Increased bone mass is an unexpected phenotype associated with deletion of the calcitonin gene. J. Clin. Investig. 2002, 110, 1849–1857. [Google Scholar] [CrossRef]
- Huebner, A.K.; Keller, J.; Catala-Lehnen, P.; Perkovic, S.; Streichert, T.; Emeson, R.B.; Amling, M.; Schinke, T. The role of calcitonin and alpha-calcitonin gene-related peptide in bone formation. Arch. Biochem. Biophys. 2008, 473, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Catala-Lehnen, P.; Huebner, A.K.; Jeschke, A.; Heckt, T.; Lueth, A.; Krause, M.; Koehne, T.; Albers, J.; Schulze, J.; et al. Calcitonin controls bone formation by inhibiting the release of sphingosine 1-phosphate from osteoclasts. Nat. Commun. 2014, 5, 5215. [Google Scholar] [CrossRef]
- Meshcheryakova, A.; Mechtcheriakova, D.; Pietschmann, P. Sphingosine 1-phosphate signaling in bone remodeling: Multifaceted roles and therapeutic potential. Expert Opin. Ther. Targets 2017, 21, 725–737. [Google Scholar] [CrossRef]
- Tian, J.; Ma, S.; Xie, W.Q.; Zhang, Y.M.; Tao, L.; Li, Y.S.; Xiao, W.F. Sphingosine 1-phosphate and osteoporosis: Pathophysiology and therapeutic aspects-a narrative review. Ann. Palliat. Med. 2021, 10, 4799–4805. [Google Scholar] [CrossRef]
- Teti, A. Mechanisms of osteoclast-dependent bone formation. Bonekey Rep. 2013, 2, 449. [Google Scholar] [CrossRef]
- Kim, J.M.; Lin, C.; Stavre, Z.; Greenblatt, M.B.; Shim, J.H. Osteoblast-Osteoclast Communication and Bone Homeostasis. Cells 2020, 9, 2073. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, P.F.; Lester, G.E.; Talmage, R.V. Calcitonin, an enigmatic hormone: Does it have a function? J. Musculoskelet. Neuronal Interact. 2001, 1, 299–305. [Google Scholar]
- Meller, Y.; Shainkin-Kestenbaum, R.; Shany, S.; Zuilli, I.; Yankowitz, N.; Giat, J.; Konforti, A.; Torok, G. Parathyroid hormone, calcitonin, and vitamin D metabolites during normal fracture healing in humans. A preliminary report. Clin. Orthop. Relat. Res. 1984, 183, 238–245. [Google Scholar]
- De Bastiani, G.; Mosconi, F.; Spagnol, G.; Nicolato, A.; Ferrari, S.; Aprili, F. High calcitonin levels in unconscious polytrauma patients. J. Bone Jt. Surg. Br. 1992, 74, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Appelt, J.; Baranowsky, A.; Jahn, D.; Yorgan, T.; Köhli, P.; Otto, E.; Farahani, S.K.; Graef, F.; Fuchs, M.; Herrera, A.; et al. The neuropeptide calcitonin gene-related peptide alpha is essential for bone healing. EBioMedicine 2020, 59, 102970. [Google Scholar] [CrossRef]
- Tsitsilonis, S.; Seemann, R.; Misch, M.; Wichlas, F.; Haas, N.P.; Schmidt-Bleek, K.; Kleber, C.; Schaser, K.D. The effect of traumatic brain injury on bone healing: An experimental study in a novel in vivo animal model. Injury 2015, 46, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Mehta, M.; Strube, P.; Peters, A.; Perka, C.; Hutmacher, D.; Fratzl, P.; Duda, G.N. Influences of age and mechanical stability on volume, microstructure, and mineralization of the fracture callus during bone healing: Is osteoclast activity the key to age-related impaired healing? Bone 2010, 47, 219–228. [Google Scholar] [CrossRef]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Müller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner. Res. 2010, 25, 1468–1486. [Google Scholar] [CrossRef]
- Dempster, D.W.; Compston, J.E.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R.; Parfitt, A.M. Standardized nomenclature, symbols, and units for bone histomorphometry: A 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2013, 28, 2–17. [Google Scholar] [CrossRef]
- Marongiu, G.; Dolci, A.; Verona, M.; Capone, A. The biology and treatment of acute long-bones diaphyseal fractures: Overview of the current options for bone healing enhancement. Bone Rep. 2020, 12, 100249. [Google Scholar] [CrossRef]
- Hankenson, K.D.; Gagne, K.; Shaughnessy, M. Extracellular signaling molecules to promote fracture healing and bone regeneration. Adv. Drug Deliv. Rev. 2015, 94, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Bessa, P.C.; Casal, M.; Reis, R.L. Bone morphogenetic proteins in tissue engineering: The road from laboratory to clinic, part II (BMP delivery). J. Tissue Eng. Regen. Med. 2008, 2, 81–96. [Google Scholar] [CrossRef]
- Russow, G.; Jahn, D.; Appelt, J.; Märdian, S.; Tsitsilonis, S.; Keller, J. Anabolic Therapies in Osteoporosis and Bone Regeneration. Int. J. Mol. Sci 2018, 20, 83. [Google Scholar] [CrossRef]
- Davey, R.A.; Findlay, D.M. Calcitonin: Physiology or fantasy? J. Bone Miner. Res. 2013, 28, 973–979. [Google Scholar] [CrossRef]
- Huebner, A.K.; Schinke, T.; Priemel, M.; Schilling, S.; Schilling, A.F.; Emeson, R.B.; Rueger, J.M.; Amling, M. Calcitonin deficiency in mice progressively results in high bone turnover. J. Bone Miner. Res. 2006, 21, 1924–1934. [Google Scholar] [CrossRef]
- Dacquin, R.; Davey, R.A.; Laplace, C.; Levasseur, R.; Morris, H.A.; Goldring, S.R.; Gebre-Medhin, S.; Galson, D.L.; Zajac, J.D.; Karsenty, G. Amylin inhibits bone resorption while the calcitonin receptor controls bone formation in vivo. J. Cell Biol. 2004, 164, 509–514. [Google Scholar] [CrossRef]
- Hankenson, K.D.; Zimmerman, G.; Marcucio, R. Biological perspectives of delayed fracture healing. Injury 2014, 45 (Suppl. 2), S8–S15. [Google Scholar] [CrossRef]
- Schlickewei, C.W.; Kleinertz, H.; Thiesen, D.M.; Mader, K.; Priemel, M.; Frosch, K.H.; Keller, J. Current and Future Concepts for the Treatment of Impaired Fracture Healing. Int. J. Mol. Sci. 2019, 20, 5805. [Google Scholar] [CrossRef]
- Beil, F.T.; Barvencik, F.; Gebauer, M.; Beil, B.; Pogoda, P.; Rueger, J.M.; Ignatius, A.; Schinke, T.; Amling, M. Effects of increased bone formation on fracture healing in mice. J. Trauma 2011, 70, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.J.; Sims, N.A. Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol. Med. 2005, 11, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.C.; McGregor, N.E.; Poulton, I.J.; Pompolo, S.; Allan, E.H.; Quinn, J.M.; Gillespie, M.T.; Martin, T.J.; Sims, N.A. Cardiotrophin-1 is an osteoclast-derived stimulus of bone formation required for normal bone remodeling. J. Bone Miner. Res. 2008, 23, 2025–2032. [Google Scholar] [CrossRef] [PubMed]
- Pederson, L.; Ruan, M.; Westendorf, J.J.; Khosla, S.; Oursler, M.J. Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. Proc. Natl. Acad. Sci. USA 2008, 105, 20764–20769. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, S.; Fumoto, T.; Matsuoka, K.; Park, K.A.; Aburatani, H.; Kato, S.; Ito, M.; Ikeda, K. Osteoclast-secreted CTHRC1 in the coupling of bone resorption to formation. J. Clin. Investig. 2013, 123, 3914–3924. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, K.; Park, K.A.; Ito, M.; Ikeda, K.; Takeshita, S. Osteoclast-derived complement component 3a stimulates osteoblast differentiation. J. Bone Miner. Res. 2014, 29, 1522–1530. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.H.; Shin, W.C.; Kim, J.W. Effect of Osteoporosis Medication on Fracture Healing: An Evidence Based Review. J. Bone Metab. 2020, 27, 15–26. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Appelt, J.; Tsitsilonis, S.; Otto, E.; Jahn, D.; Köhli, P.; Baranowsky, A.; Jiang, S.; Fuchs, M.; Bucher, C.H.; Duda, G.N.; et al. Mice Lacking the Calcitonin Receptor Do Not Display Improved Bone Healing. Cells 2021, 10, 2304. https://doi.org/10.3390/cells10092304
Appelt J, Tsitsilonis S, Otto E, Jahn D, Köhli P, Baranowsky A, Jiang S, Fuchs M, Bucher CH, Duda GN, et al. Mice Lacking the Calcitonin Receptor Do Not Display Improved Bone Healing. Cells. 2021; 10(9):2304. https://doi.org/10.3390/cells10092304
Chicago/Turabian StyleAppelt, Jessika, Serafeim Tsitsilonis, Ellen Otto, Denise Jahn, Paul Köhli, Anke Baranowsky, Shan Jiang, Melanie Fuchs, Christian H. Bucher, Georg N. Duda, and et al. 2021. "Mice Lacking the Calcitonin Receptor Do Not Display Improved Bone Healing" Cells 10, no. 9: 2304. https://doi.org/10.3390/cells10092304
APA StyleAppelt, J., Tsitsilonis, S., Otto, E., Jahn, D., Köhli, P., Baranowsky, A., Jiang, S., Fuchs, M., Bucher, C. H., Duda, G. N., Frosch, K.-H., & Keller, J. (2021). Mice Lacking the Calcitonin Receptor Do Not Display Improved Bone Healing. Cells, 10(9), 2304. https://doi.org/10.3390/cells10092304






