ABHD4-Regulating RNA Panel: Novel Biomarkers in Acute Coronary Syndrome Diagnosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. In Silico Data Analysis
2.3. Purification of Total RNA Including miRNAs from Sera Samples and Quantitaive Real Time PCR (RT-qPCR)
2.4. Statistical Analysis
3. Results
3.1. Biochemical and Clinical Markers in the Investigated Groups
3.2. Expression of the Serum RNAs Molecular Network
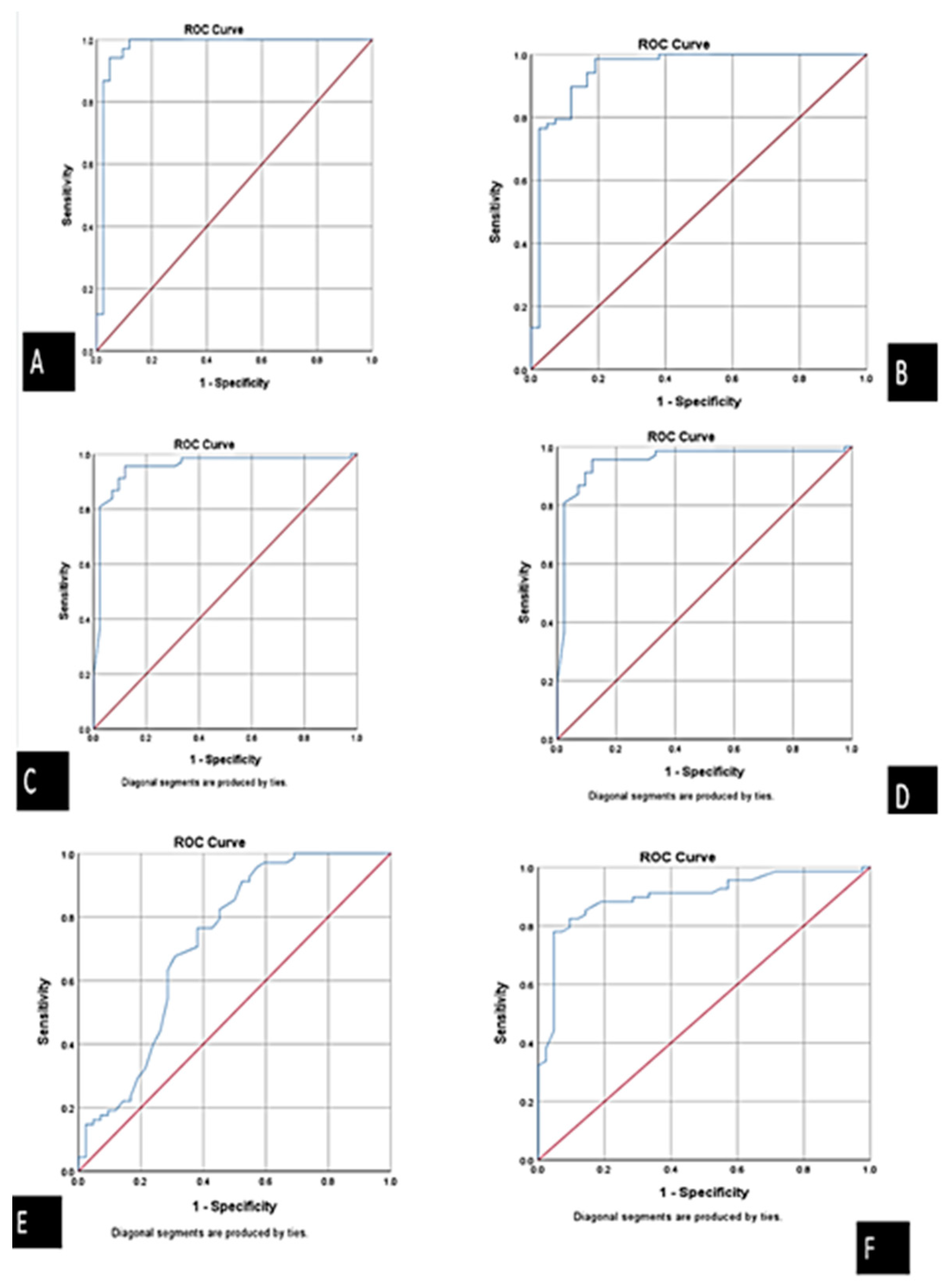
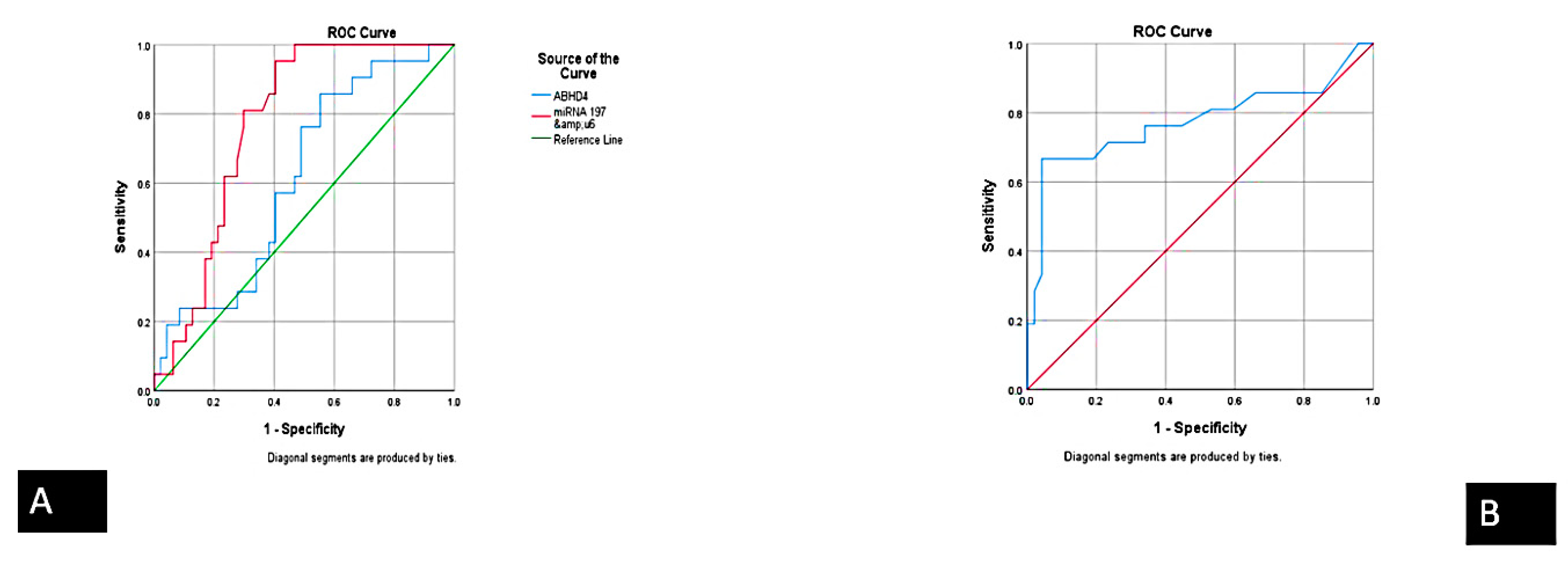
3.3. Correlation between Serum ABHD4 Regulating RNAs and Cardiac Troponin among the Study Groups
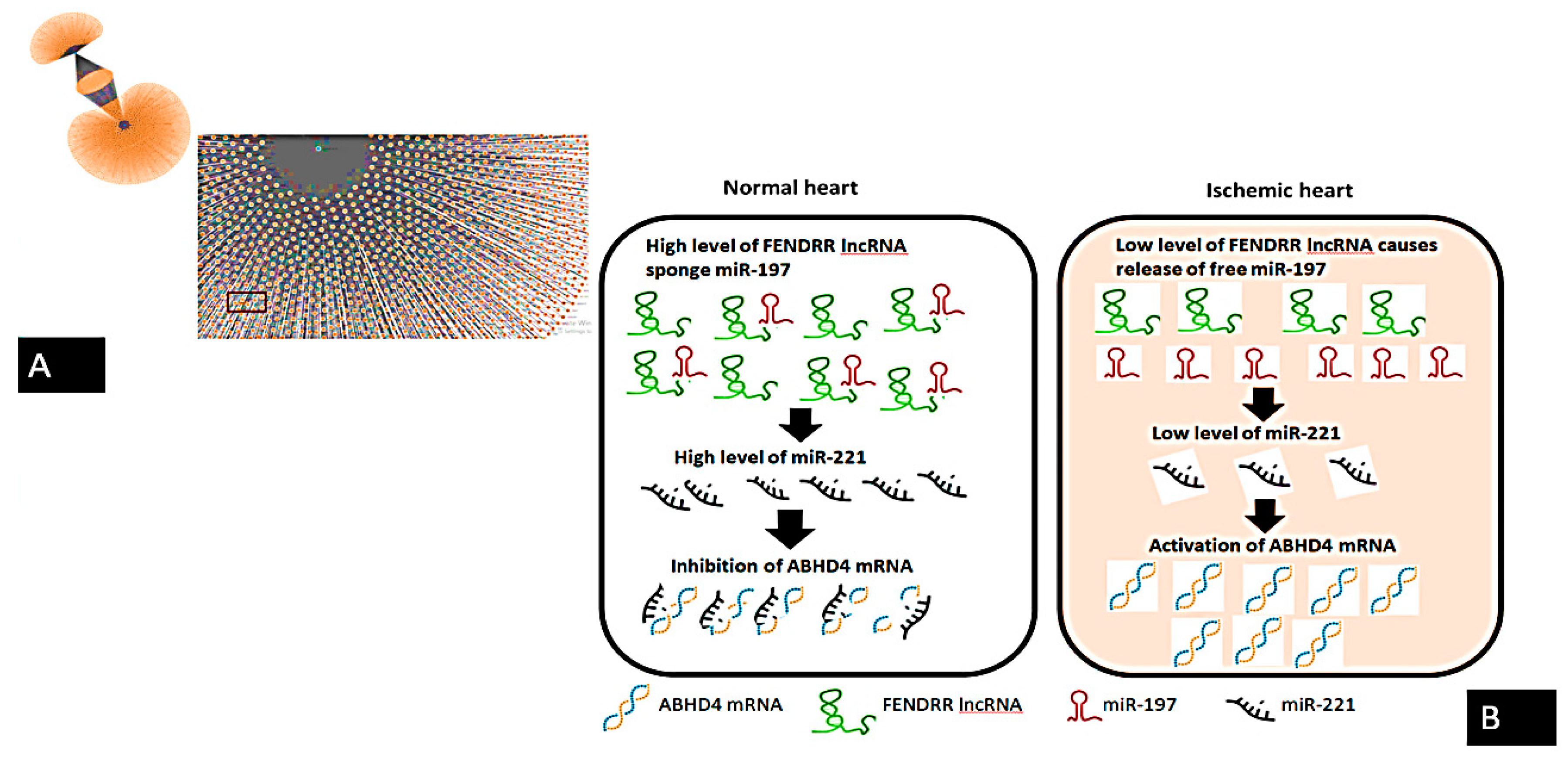
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moran, A.E.; Tzong, K.Y.; Forouzanfar, M.H.; Roth, G.A.; Mensah, G.A.; Ezzati, M.; Murray, C.J.; Naghavi, M. Variations in ischemic heart disease burden by age, country, and income: The Global Burden of Diseases, Injuries, and Risk Factors 2010 study. Glob. Heart 2014, 9, 91–99. [Google Scholar] [CrossRef]
- Windecker, S. ESC Committee for Practice Guidelines: Providing knowledge to everyday clinical practice. Cardiovasc. Res. 2020, 116, e146–e148. [Google Scholar] [CrossRef]
- Barnett, K.; Mercer, S.W.; Norbury, M.; Watt, G.; Wyke, S.; Guthrie, B. Epidemiology of multimorbidity and implications for health care, research, and medical education: A cross-sectional study. Lancet 2012, 380, 37–43. [Google Scholar] [CrossRef]
- White, H.D.; Chew, D.P. Acute myocardial infarction. Lancet 2008, 372, 570–584. [Google Scholar] [CrossRef]
- Thygesen, K.; Alpert, J.S.; White, H.D.; Joint ESC/ACCF/AHA/WHF. Task Force for the Redefinition of Myocardial Infarction. Universal definition of myocardial infarction. Eur. Heart J. 2007, 2525–2538. [Google Scholar] [CrossRef]
- Suzuki, K.; Komukai, K.; Nakata, K.; Kang, R.; Oi, Y.; Muto, E.; Kashiwagi, Y.; Tominaga, M.; Miyanaga, S.; Ishikawa, T.; et al. The Usefulness and Limitations of Point-of-care Cardiac Troponin Measurement in the Emergency Department. Intern. Med. 2018, 57, 1673–1680. [Google Scholar] [CrossRef]
- Jaffe, A.S.; Ravkilde, J.; Roberts, R.; Naslund, U.; Apple, F.S.; Galvani, M.; Katus, H. It’s time for a change to a troponin standard. Circulation 2000, 102, 1216–1220. [Google Scholar] [CrossRef]
- Piccioni, A.; Valletta, F.; Zanza, C.; Esperide, A.; Franceschi, F. Novel biomarkers to assess the risk for acute coronary syndrome: Beyond troponins. Intern. Emerg. Med. 2020, 15, 1193–1199. [Google Scholar] [CrossRef]
- Piccioni, A.; de Cunzo, T.; Valletta, F.; Covino, M.; Rinninella, E.; Raoul, P.; Zanza, C.; Mele, M.C.; Franceschi, F. Gut Microbiota and Environment in Coronary Artery Disease. Int. J. Environ. Res. Public Health 2021, 18, 4242. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, L.; Harvey-White, J.; Huang, B.X.; Kim, H.Y.; Luquet, S.; Palmiter, R.D.; Krystal, G.; Rai, R.; Mahadevan, A.; et al. Multiple pathways involved in the biosynthesis of anandamide. Neuropharmacology 2008, 54, 1–7. [Google Scholar] [CrossRef]
- Nardini, M.; Dijkstra, B.W. Alpha/beta hydrolase fold enzymes: The family keeps growing. Curr. Opin. Struct. Biol. 1999, 9, 732–737. [Google Scholar] [CrossRef]
- Lord, C.C.; Thomas, G.; Brown, J.M. Mammalian alpha beta hydrolase domain (ABHD) proteins: Lipid metabolizing enzymes at the interface of cell signaling and energy metabolism. Biochim. Biophys. Acta 2013, 1831, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Emirkan, A.; van Duijn, C.M.; Ugocsai, P.; Isaacs, A.; Pramstaller, P.P.; Liebisch, G.; Wilson, J.F.; Johansson, A.; Rudan, I.; Aulchenko, Y.S.; et al. Genome-wide association study identifies novel loci associated with circulating phospho- and sphingolipid concentrations. PLoS Genet. 2012, 8, e1002490. [Google Scholar]
- Jensen, M.K.; Rimm, E.B.; Rader, D.; Schmidt, E.B.; Sørensen, T.I.; Vogel, U.; Overvad, K.; Mukamal, K.J. S447X variant of the lipoprotein lipase gene, lipids, and risk of coronary heart disease in 3 prospective cohort studies. Am. Heart J. 2009, 157, 384–390. [Google Scholar] [CrossRef]
- Kurano, M.; Kano, K.; Dohi, T.; Matsumoto, H.; Igarashi, K.; Nishikawa, M.; Ohkawa, R.; Ikeda, H.; Miyauchi, K.; Daida, H.; et al. Different origins of lysophospholipid mediators between coronary and peripheral arteries in acute coronary syndrome. J. Lipid Res. 2017, 58, 433–442. [Google Scholar] [CrossRef]
- Ohkawa, R.; Kurano, M.; Sakai, N.; Kishimoto, T.; Nojiri, T.; Igarashi, K.; Hosogaya, S.; Ozaki, Y.; Dohi, T.; Miyauchi, K.; et al. Measurement of plasma choline in acute coronary syndrome: Importance of suitable sampling conditions for this assay. Sci. Rep. 2018, 8, 4725. [Google Scholar] [CrossRef]
- Gaudet, P.; Livstone, M.S.; Lewis, S.E.; Thomas, P.D. Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief Bioinform. 2011, 12, 449–462. [Google Scholar] [CrossRef]
- Lee, H.C.; Simon, G.M.; Cravatt, B.F. ABHD4 regulates multiple classes of N-acyl phospholipids in the mammalian central nervous system. Biochemistry 2015, 54, 2539–2549. [Google Scholar] [CrossRef]
- Vickers, K.C.; Rye, K.A.; Tabet, F. MicroRNAs in the onset and development of cardiovascular disease. Clin. Sci. 2014, 126, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Zheng, S.; Zhou, C.; Yuan, W.; Wang, J.; Wang, T. Bone marrow mesenchymal stem cells for post-myocardial infarction cardiac repair: Micrornas as novel regulators. J. Cell. Mol. Med. 2012, 16, 657–671. [Google Scholar] [CrossRef]
- Meder, B.; Keller, A.; Vogel, B.; Haas, J.; Sedaghat-Hamedani, F.; Kayvanpour, E.; Just, S.; Borries, A.; Rudloff, J.; Leidinger, P.; et al. MicroRNA signatures in total peripheral blood as novel biomarkers for acute myocardial infarction. Basic Res. Cardiol. 2011, 106, 13–23. [Google Scholar] [CrossRef]
- Zhong, J.; He, Y.; Chen, W.; Shui, X.; Chen, C.; Lei, W. Circulating microRNA-19a as a potential novel biomarker for diagnosis of acute myocardial infarction. Int. J. Mol. Sci. 2014, 15, 20355–20364. [Google Scholar] [CrossRef] [PubMed]
- Hong, Q.; Li, O.; Zheng, W.; Xiao, W.Z.; Zhang, L.; Wu, D.; Cai, G.Y.; He, J.C.; Chen, X.M. LncRNA HOTAIR regulates HIF-1 α/AXL signaling through inhibition of miR-217 in renal cell carcinoma. Cell Death Dis. 2017, 8, e2772. [Google Scholar] [CrossRef]
- Li, X.; Zhou, J.; Huang, K. Inhibition of the lncRNA Mirt1 attenuates acute myocardial infarction by suppressing NF-κB activation. Cell. Physiol. Biochem. 2017, 42, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Dai, Y.; Yan, S.; Shi, Y.; Han, B.; Li, J.; Cha, L.; Mu, J. Down-regulation of lncRNA KCNQ1OT1 protects against myocardial ischemia/reperfusion injury following acute myocardial infarction. Biochem. Biophys. Res. Commun. 2017, 491, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Vausort, M.; Wagner, D.R.; Devaux, Y. Long noncoding RNAs in patients with acute myocardial infarction. Circ. Res. 2014, 115, 668–677. [Google Scholar] [CrossRef]
- Roger, V.L. Epidemiology of myocardial infarction. Med. Clin. N. Am. 2007, 91, 537–552. [Google Scholar] [CrossRef]
- Jenkins, C.M.; Mancuso, D.J.; Yan, W.; Sims, H.F.; Gibson, B.; Gross, R.W. Identification, cloning, expression, and purification of three novel human calcium-independent phospholipase A2 family members possessing triacylglycerol lipase and acylglycerol transacylase activities. J. Biol. Chem. 2004, 279, 48968–48975. [Google Scholar] [CrossRef]
- Yu, L.; Li, Y.; Grisé, A.; Wang, H. CGI-58: Versatile Regulator of Intracellular Lipid Droplet Homeostasis. In Lipid Transfer in Lipoprotein Metabolism and Cardiovascular Disease; Springer: Singapore, 2020; pp. 197–222. [Google Scholar]
- Simpson, C.D.; Hurren, R.; Kasimer, D.; MacLean, N.; Eberhard, Y.; Ketela, T.; Moffat, J.; Schimmer, A.D. A genome wide shRNA screen identifies α/β hydrolase domain containing 4 (ABHD4) as a novel regulator of anoikis resistance. Apoptosis 2012, 17, 666–678. [Google Scholar] [CrossRef]
- Xue, Y.; Wei, Z.; Ding, H.; Wang, Q.; Zhou, Z.; Zheng, S.; Zhang, Y.; Hou, D.; Liu, Y.; Zen, K.; et al. MicroRNA-19b/221/222 induces endothelial cell dysfunction via suppression of PGC-1α in the progression of atherosclerosis. Atherosclerosis 2015, 241, 671–681. [Google Scholar] [CrossRef]
- Coskunpinar, E.; Cakmak, H.A.; Kalkan, A.K.; Tiryakioglu, N.O.; Erturk, M.; Ongen, Z. Circulating miR-221-3p as a novel marker for early prediction of acute myocardial infarction. Gene 2016, 591, 90–96. [Google Scholar] [PubMed]
- Willeit, P.; Zampetaki, A.; Dudek, K.; Kaudewitz, D.; King, A.; Kirkby, N.S.; Crosby-Nwaobi, R.; Prokopi, M.; Drozdov, I.; Langley, S.R.; et al. Circulating microRNAs as novel biomarkers for platelet activation. Circ. Res. 2013, 112, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Karolina, D.S.; Tavintharan, S.; Armugam, A.; Sepramaniam, S.; Pek, S.L.; Wong, M.T.; Lim, S.C.; Sum, C.F.; Jeyaseelan, K. Circulating miRNA profiles in patients with metabolic syndrome. J. Clin. Endocrinol. Metab. 2012, 97, E2271–E2276. [Google Scholar] [CrossRef]
- Nichols, M.; Townsend, N.; Scarborough, P.; Rayner, M. Cardiovascular disease in Europe 2014: Epidemiological update. Eur. Heart J. 2014, 35, 2950–2959. [Google Scholar] [CrossRef] [PubMed]
- Gibler, W.B.; Lewis, L.M.; Erb, R.E.; Makens, P.K.; Kaplan, B.C.; Vaughn, R.H.; Biagini, A.V.; Blanton, J.D.; Campbell, W.B. Early detection of acute myocardial infarction in patients presenting with chest pain and nondiagnostic ECGs: Serial CK-MB sampling in the emergency department. Ann. Emerg. Med. 1990, 19, 1359–1366. [Google Scholar] [CrossRef]
- Pierce, G.F.; Jaffe, A.S. Increased creatine kinase MB in the absence of acute myocardial infarction. Clin. Chem. 1986, 32, 2044–2051. [Google Scholar]
- Katus, H.; Ziegler, A.; Ekinci, O.; Giannitsis, E.; Stough, W.G.; Achenbach, S.; Blankenberg, S.; Brueckmann, M.; Collinson, P.; Comaniciu, D.; et al. Early diagnosis of acute coronary syndrome. Eur. Heart J. 2017, 38, 3049–3055. [Google Scholar] [CrossRef]
- Poller, W.; Dimmeler, S.; Heymans, S.; Zeller, T.; Haas, J.; Karakas, M.; Leistner, D.M.; Jakob, P.; Nakagawa, S.; Blankenberg, S.; et al. Non-coding RNAs in cardiovascular diseases: Diagnostic and therapeutic perspectives. Eur. Heart J. 2018, 39, 2704–2716. [Google Scholar] [CrossRef]
- Vieyres, G.; Reichert, I.; Carpentier, A.; Vondran, F.W.; Pietschmann, T. The ATGL lipase cooperates with ABHD5 to mobilize lipids for hepatitis C virus assembly. PLoS Pathog. 2020, 16, e1008554. [Google Scholar]
- Schulte, C.; Molz, S.; Appelbaum, S.; Karakas, M.; Ojeda, F.; Lau, D.M.; Hartmann, T.; Lackner, K.J.; Westermann, D.; Schnabel, R.B.; et al. miRNA-197 and miRNA-223 predict cardiovascular death in a cohort of patients with symptomatic coronary artery disease. PLoS ONE 2015, 10, e0145930. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, J.; Dong, J.; Bai, R.; Song, D.; Ma, X.; Zhao, L.; Yao, Y.; Zhang, H.; Liu, T. Association of miR-197-5p, a circulating biomarker for heart failure, with myocardial fibrosis and adverse cardiovascular events among patients with stage C or D heart failure. Cardiology 2018, 141, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Grote, P.; Herrmann, B.G. The long non-coding RNA Fendrr links epigenetic control mechanisms to gene regulatory networks in mammalian embryogenesis. RNA Biol. 2013, 10, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Çekin, N.; Özcan, A.; Göksel, S.; Arslan, S.; Pınarbaşı, E.; Berkan, Ö. Decreased FENDRR and LincRNA-p21 expression in atherosclerotic plaque. Anatol. J. Cardiol. 2018, 19, 131. [Google Scholar] [CrossRef] [PubMed]
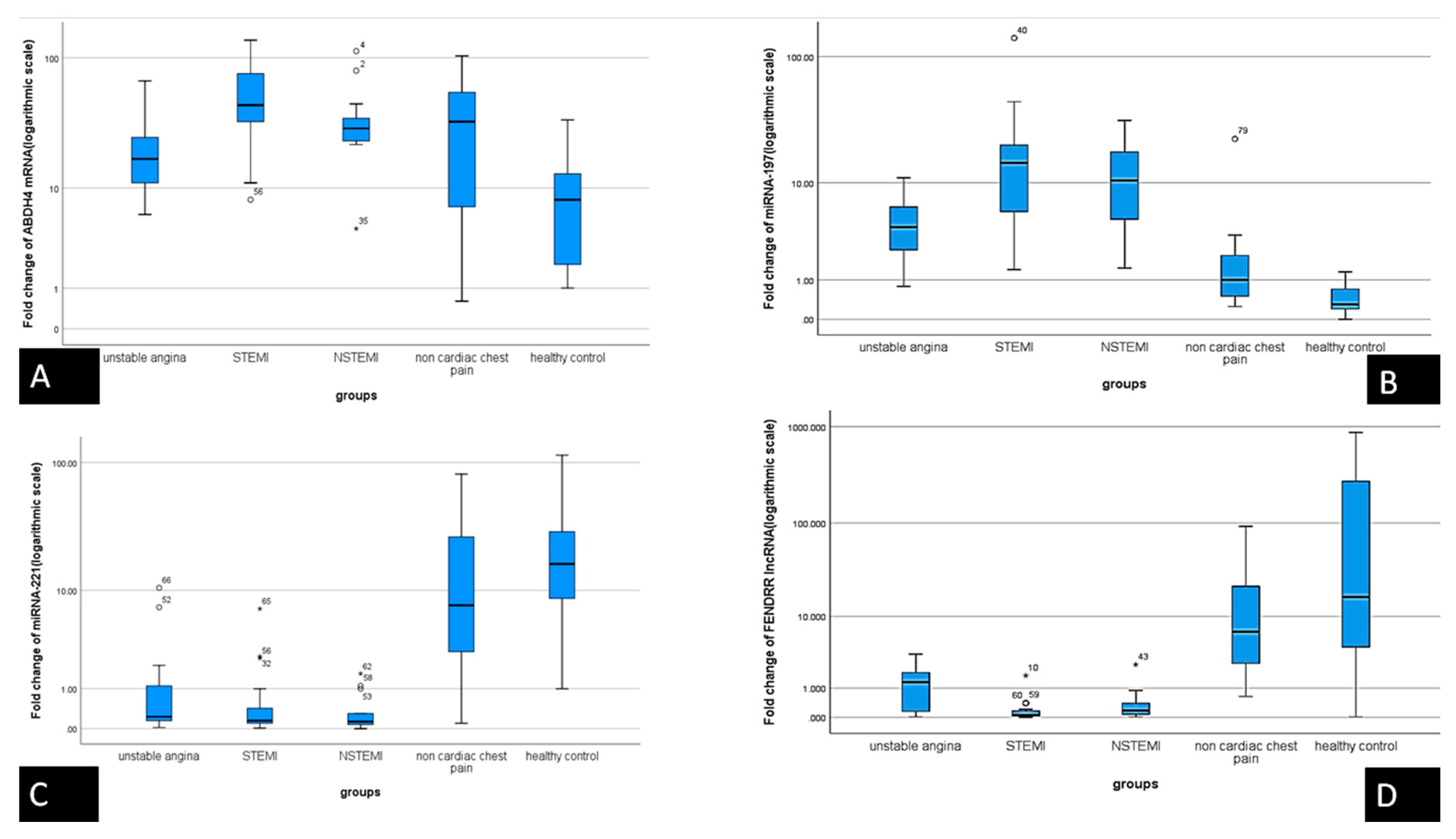

| i | Median | F | p | ||
|---|---|---|---|---|---|
| miRNA-221-3p | unstable angina | 21 | 0.2300 | a 13.053 | a 0.000 |
| STEMI | 31 | 0.1500 | b 0.00 | ||
| NSTEMI | 16 | 0.1300 | c 0.874 | ||
| non cardiac chest pain | 21 | 7.5000 | |||
| healthy control | 21 | 16.4000 | |||
| miRNA 197-5p | unstable angina | 21 | 4.0395 | a 7.473 | a 0.000 |
| STEMI | 31 | 14.6000 | b 0.001 | ||
| NSTEMI | 16 | 10.4500 | c 0.001 | ||
| non cardiac chest pain | 21 | 1.0000 | |||
| healthy control | 21 | 0.3000 | |||
| ABHD4 mRNA | unstable angina | 21 | 8.80000 | a 9.142 | a 0.000 |
| STEMI | 31 | 7.80000 | b 0.01 | ||
| NSTEMI | 16 | 21.40000 | c 0.103 | ||
| non cardiac chest pain | 21 | 0.04000 | |||
| healthy control | 21 | 0.01000 | |||
| FENDRR lncRNA | unstable angina | 21 | 1.30000 | a 8.994 | a 0.000 |
| STEMI | 31 | 0.06000 | b 0.00 | ||
| NSTEMI | 16 | 0.17000 | c 0.968 | ||
| non cardiac chest pain | 21 | 6.65000 | |||
| healthy control | 21 | 16.50000 | |||
| Creatine kinase-MB (CK-MB) | unstable angina | 21 | 17.00 | a 10.947 | a 0.000 |
| STEMI | 31 | 44.00 | b 0.128 | ||
| NSTEMI | 16 | 29.50 | c 0.1 | ||
| non cardiac chest pain | 21 | 33.00 | |||
| healthy control | 21 | 8.00 | |||
| Cardiac Troponin | unstable angina | 21 | 17.00 | a 79.243 | a 0.000 |
| STEMI | 31 | 44.00 | b 0.154 | ||
| NSTEMI | 16 | 29.50 | c 0.1 | ||
| non cardiac chest pain | 21 | 33.00 | |||
| healthy control | 21 | 8.00 | |||
| Biomarker | Sensitivity | Specificity | PPV (Positive Predictive Value) | NPV (Negative Predictive Value) | Accuracy |
|---|---|---|---|---|---|
| ABHD4 mRNA | 94.4% | 97.4% | 98.5% | 90.5% | 95.4% |
| FENDRR lncRNA | 93.3% | 90.5% | 82.4% | 76% | 93.3% |
| miRNA-221-3p | 95.4% | 86.7% | 91.2% | 92.9% | 91.8% |
| miRNA-197-5p | 89.3% | 97.1% | 98.5% | 81% | 91.8% |
| Cardiac troponin | 93.1% | 73.1% | 79.4% | 90.5% | 83.6% |
| MiRNA-221 | MiRNA-197 | ABHD4 | FENDRR | Troponin | |||
|---|---|---|---|---|---|---|---|
| Spearman’s rho | miRNA-221-3p | Correlation Coefficient | 1.000 | –0.614 ** | –0.596 ** | 0.678 ** | –0.586 ** |
| Sig. (2-tailed) | 0.000 | 0.000 | 0.000 | 0.000 | |||
| N | 110 | 110 | 110 | 110 | 110 | ||
| miRNA 197-5p | Correlation Coefficient | –0.614 ** | 1.000 | 0.589 ** | –0.586 ** | 0.641 ** | |
| Sig. (2-tailed) | 0.000 | 0.000 | 0.000 | 0.000 | |||
| N | 110 | 110 | 110 | 110 | 110 | ||
| ABHD4 mRNA | Correlation Coefficient | –0.596 ** | 0.589 ** | 1.000 | –0.587 ** | 0.596 ** | |
| Sig. (2-tailed) | 0.000 | 0.000 | 0.000 | 0.000 | |||
| N | 110 | 110 | 110 | 110 | 110 | ||
| FENDRR lncRNA | Correlation Coefficient | 0.678 ** | –0.586 ** | –0.587 ** | 1.000 | –0.643 ** | |
| Sig. (2-tailed) | 0.000 | 0.000 | 0.000 | 0.000 | |||
| N | 110 | 110 | 110 | 110 | 110 | ||
| Cardiac Troponin | Correlation Coefficient | –0.586 ** | 0.641 ** | 0.596 ** | –0.643 ** | 1.000 | |
| Sig. (2-tailed) | 0.000 | 0.000 | 0.000 | 0.000 | |||
| N | 110 | 110 | 110 | 110 | 110 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agwa, S.H.A.; Elzahwy, S.S.; El Meteini, M.S.; Elghazaly, H.; Saad, M.; Abd Elsamee, A.M.; Shamekh, R.; Matboli, M. ABHD4-Regulating RNA Panel: Novel Biomarkers in Acute Coronary Syndrome Diagnosis. Cells 2021, 10, 1512. https://doi.org/10.3390/cells10061512
Agwa SHA, Elzahwy SS, El Meteini MS, Elghazaly H, Saad M, Abd Elsamee AM, Shamekh R, Matboli M. ABHD4-Regulating RNA Panel: Novel Biomarkers in Acute Coronary Syndrome Diagnosis. Cells. 2021; 10(6):1512. https://doi.org/10.3390/cells10061512
Chicago/Turabian StyleAgwa, Sara H. A., Sherif Samir Elzahwy, Mahmoud Shawky El Meteini, Hesham Elghazaly, Maha Saad, Aya M. Abd Elsamee, Rania Shamekh, and Marwa Matboli. 2021. "ABHD4-Regulating RNA Panel: Novel Biomarkers in Acute Coronary Syndrome Diagnosis" Cells 10, no. 6: 1512. https://doi.org/10.3390/cells10061512
APA StyleAgwa, S. H. A., Elzahwy, S. S., El Meteini, M. S., Elghazaly, H., Saad, M., Abd Elsamee, A. M., Shamekh, R., & Matboli, M. (2021). ABHD4-Regulating RNA Panel: Novel Biomarkers in Acute Coronary Syndrome Diagnosis. Cells, 10(6), 1512. https://doi.org/10.3390/cells10061512






