Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (–)-Epicatechin, and Betaine
Abstract
1. Introduction
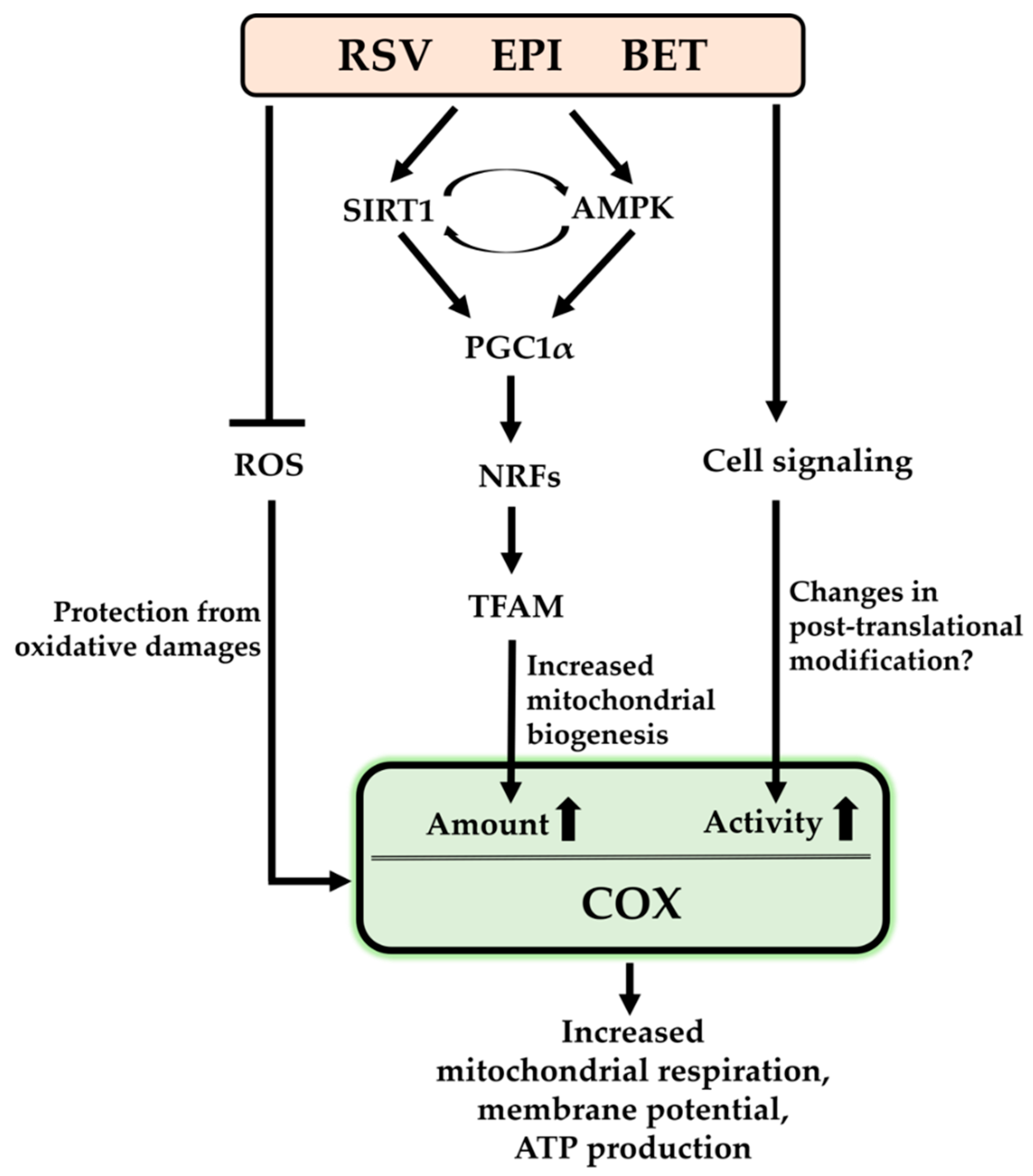
2. Resveratrol
3. (–)-Epicatechin
4. Betaine
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RSV | resveratrol |
| EPI | (–)-epicatechin |
| BET | betaine |
| COX | cytochrome c oxidase |
| ETC | electron transport chain |
| OXPHOS | oxidative phosphorylation |
| ROS | reactive oxygen species |
References
- Cheng, Z.; Ristow, M. Mitochondria and metabolic homeostasis. Antioxid. Redox Signal. 2013, 19, 240–242. [Google Scholar] [CrossRef]
- Wu, M.; Gu, J.; Zong, S.; Guo, R.; Liu, T.; Yang, M. Research journey of respirasome. Protein Cell 2020, 11, 318–338. [Google Scholar] [CrossRef]
- Genova, M.L.; Lenaz, G. Functional role of mitochondrial respiratory supercomplexes. Biochim. Biophys. Acta 2014, 1837, 427–443. [Google Scholar] [CrossRef] [PubMed]
- Milenkovic, D.; Blaza, J.N.; Larsson, N.G.; Hirst, J. The enigma of the respiratory chain supercomplex. Cell Metab. 2017, 25, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.M.; Williams, J.A.; Ding, W.X. Mitochondrial dynamics and mitochondrial quality control. Redox Biol. 2015, 4, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Westermann, B. Bioenergetic role of mitochondrial fusion and fission. Biochim. Biophys. Acta 2012, 1817, 1833–1838. [Google Scholar] [CrossRef]
- Silva Ramos, E.; Larsson, N.G.; Mourier, A. Bioenergetic roles of mitochondrial fusion. Biochim. Biophys. Acta 2016, 1857, 1277–1283. [Google Scholar] [CrossRef]
- Tsukihara, T.; Aoyama, H.; Yamashita, E.; Tomizaki, T.; Yamaguchi, H.; Shinzawa-Itoh, K.; Nakashima, R.; Yaono, R.; Yoshikawa, S. The whole structure of the 13-subunit oxidized cytochrome c oxidase at 2.8. Science 1996, 272, 1136–1144. [Google Scholar] [CrossRef]
- Kadenbachand, B.; Hüttemann, M. The subunit composition and function of mammalian cytochrome c oxidase. Mitochondrion 2015, 24, 64–76. [Google Scholar] [CrossRef]
- Watson, S.A.; McStay, G.P. Functions of cytochrome c oxidase assembly factors. Int. J. Mol. Sci. 2020, 21, 7254. [Google Scholar] [CrossRef] [PubMed]
- Zong, S.; Wu, M.; Gu, J.; Liu, T.; Guo, R.; Yang, M. Structure of the intact 14-subunit human cytochrome c oxidase. Cell Res. 2018, 28, 1026–1034. [Google Scholar] [CrossRef]
- Hüttemann, M.; Helling, S.; Sanderson, T.H.; Sinkler, C.; Samavati, L.; Mahapatra, G.; Varughese, A.; Lu, G.; Liu, J.; Ramzan, R.; et al. Regulation of mitochondrial respiration and apoptosis through cell signaling: Cytochrome c oxidase and cytochrome c in ischemia/reperfusion injury and inflammation. Biochim. Biophys. Acta 2012, 1817, 598–609. [Google Scholar] [CrossRef]
- Sinkler, C.A.; Kalpage, H.; Shay, J.; Lee, I.; Malek, M.H.; Grossman, L.I.; Hüttemann, M. Tissue- and condition-specific isoforms of mammalian cytochrome c oxidase subunits: From function to human disease. Oxid. Med. Cell. Longev. 2017, 2017, 1534056. [Google Scholar] [CrossRef]
- Howitzand, K.T.; Sinclair, D.A. Xenohormesis: Sensing the chemical cues of other species. Cell 2008, 133, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Gibellini, L.; Bianchini, E.; de Biasi, S.; Nasi, M.; Cossarizza, A.; Pinti, M. Natural compounds modulating mitochondrial functions. Evid. Based Complement. Alternat. Med. 2015, 2015, 527209. [Google Scholar] [CrossRef] [PubMed]
- Sandoval-Acuna, C.; Ferreira, J.; Speisky, H. Polyphenols and mitochondria: An update on their increasingly emerging ros-scavenging independent actions. Arch. Biochem. Biophys. 2014, 559, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Revel, J.S.; Maier, C.S. Mitochondria-centric review of polyphenol bioactivity in cancer models. Antioxid. Redox Signal. 2018, 29, 1589–1611. [Google Scholar] [CrossRef]
- Kicinska, A.; Jarmuszkiewicz, W. Flavonoids and mitochondria: Activation of cytoprotective pathways? Molecules 2020, 25, 3060. [Google Scholar] [CrossRef]
- Raza, H.; John, A.; Brown, E.M.; Benedict, S.; Kambal, A. Alterations in mitochondrial respiratory functions, redox metabolism and apoptosis by oxidant 4-hydroxynonenal and antioxidants curcumin and melatonin in pc12 cells. Toxicol. Appl. Pharmacol. 2008, 226, 161–168. [Google Scholar] [CrossRef]
- Rastogi, M.; Ojha, R.P.; Sagar, C.; Agrawal, A.; Dubey, G.P. Protective effect of curcuminoids on age-related mitochondrial impairment in female wistar rat brain. Biogerontology 2014, 15, 21–31. [Google Scholar] [CrossRef]
- Ray Hamidie, R.D.; Yamada, T.; Ishizawa, R.; Saito, Y.; Masuda, K. Curcumin treatment enhances the effect of exercise on mitochondrial biogenesis in skeletal muscle by increasing camp levels. Metabolism 2015, 64, 1334–1347. [Google Scholar] [CrossRef] [PubMed]
- Jia, N.; Sun, Q.; Su, Q.; Chen, G. Sirt1-mediated deacetylation of pgc1alpha attributes to the protection of curcumin against glutamate excitotoxicity in cortical neurons. Biochem. Biophys. Res. Commun. 2016, 478, 1376–1381. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Tang, J.; Li, Y.; Xie, Y.; Shan, H.; Chen, M.; Zhang, J.; Yang, X.; Zhang, Q.; Yang, X. Curcumin attenuates skeletal muscle mitochondrial impairment in copd rats: Pgc-1alpha/sirt3 pathway involved. Chem. Biol. Interact. 2017, 277, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, A.; Plumitallo, C.; de Nuccio, C.; Visentin, S.; Minghetti, L. Curcumin promotes oligodendrocyte differentiation and their protection against tnf-alpha through the activation of the nuclear receptor ppar-gamma. Sci. Rep. 2021, 11, 4952. [Google Scholar] [CrossRef] [PubMed]
- Rayamajhi, N.; Kim, S.K.; Go, H.; Joe, Y.; Callaway, Z.; Kang, J.G.; Ryter, S.W.; Chung, H.T. Quercetin induces mitochondrial biogenesis through activation of ho-1 in hepg2 cells. Oxid. Med. Cell Longev. 2013, 2013, 154279. [Google Scholar] [CrossRef] [PubMed]
- Ballmann, C.; Denney, T.S.; Beyers, R.J.; Quindry, T.; Romero, M.; Amin, R.; Selsby, J.T.; Quindry, J.C. Lifelong quercetin enrichment and cardioprotection in mdx/utrn+/- mice. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H128–H140. [Google Scholar] [CrossRef] [PubMed]
- Dave, A.; Shukla, F.; Wala, H.; Pillai, P. Mitochondrial electron transport chain complex dysfunction in mecp2 knock-down astrocytes: Protective effects of quercetin hydrate. J. Mol. Neurosci. 2019, 67, 16–27. [Google Scholar] [CrossRef]
- Sharma, S.; Raj, K.; Singh, S. Neuroprotective effect of quercetin in combination with piperine against rotenone- and iron supplement-induced parkinson’s disease in experimental rats. Neurotox. Res. 2020, 37, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, G.; Dong, Y.; Li, M.; Yang, L.; Lu, W. Quercetin protects against lipopolysaccharide-induced intestinal oxidative stress in broiler chickens through activation of nrf2 pathway. Molecules 2020, 25, 1053. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Bae, H. An overview of stress-induced resveratrol synthesis in grapes: Perspectives for resveratrol-enriched grape products. Molecules 2017, 22, 294. [Google Scholar] [CrossRef]
- Yu, O.J.; Jez, M. Nature’s assembly line: Biosynthesis of simple phenylpropanoids and polyketides. Plant J. 2008, 54, 750–762. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, M. Resveratrol, a new phenolic compound, from veratrum grandiflorum. J. Chem. Soc. Jpn. 1939, 60, 1090–1100. [Google Scholar]
- Jasinski, M.; Jasinska, L.; Ogrodowczyk, M. Resveratrol in prostate diseases—A short review. Cent. Eur. J. Urol. 2013, 66, 144–149. [Google Scholar] [CrossRef]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef]
- Burkon, A.; Somoza, V. Quantification of free and protein-bound trans-resveratrol metabolites and identification of trans-resveratrol-c/o-conjugated diglucuronides—Two novel resveratrol metabolites in human plasma. Mol. Nutr. Food Res. 2008, 52, 549–557. [Google Scholar] [CrossRef]
- Kopp, P. Resveratrol, a phytoestrogen found in red wine. A possible explanation for the conundrum of the ‘french paradox’? Eur. J. Endocrinol. 1998, 138, 619–620. [Google Scholar] [CrossRef]
- Hsieh, T.C.; Wu, J.M. Resveratrol: Biological and pharmaceutical properties as anticancer molecule. Biofactors 2010, 36, 360–369. [Google Scholar] [CrossRef]
- Kalantari, H.; Das, D.K. Physiological effects of resveratrol. Biofactors 2010, 36, 401–406. [Google Scholar] [CrossRef]
- Schnekenburger, M.; Dicato, M.; Diederich, M. Plant-derived epigenetic modulators for cancer treatment and prevention. Biotechnol. Adv. 2014, 32, 1123–1132. [Google Scholar] [CrossRef]
- Chan, S.; Kantham, S.; Rao, V.M.; Palanivelu, M.K.; Pham, H.L.; Shaw, P.N.; McGeary, R.P.; Ross, B.P. Metal chelation, radical scavenging and inhibition of abeta(4)(2) fibrillation by food constituents in relation to alzheimer’s disease. Food Chem. 2016, 199, 185–194. [Google Scholar] [CrossRef]
- Blanquer-Rossello, M.D.; Hernandez-Lopez, R.; Roca, P.; Oliver, J.; Valle, A. Resveratrol induces mitochondrial respiration and apoptosis in sw620 colon cancer cells. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 431–440. [Google Scholar] [CrossRef]
- Singh, A.P.; Singh, R.; Verma, S.S.; Rai, V.; Kaschula, C.H.; Maiti, P.; Gupta, S.C. Health benefits of resveratrol: Evidence from clinical studies. Med. Res. Rev. 2019, 39, 1851–1891. [Google Scholar] [CrossRef]
- Zhou, J.; Liao, Z.; Jia, J.; Chen, J.L.; Xiao, Q. The effects of resveratrol feeding and exercise training on the skeletal muscle function and transcriptome of aged rats. PeerJ 2019, 7, e7199. [Google Scholar] [CrossRef]
- Sung, M.M.; Byrne, N.J.; Robertson, I.M.; Kim, T.T.; Samokhvalov, V.; Levasseur, J.; Soltys, C.L.; Fung, D.; Tyreman, N.; Denou, E.; et al. Resveratrol improves exercise performance and skeletal muscle oxidative capacity in heart failure. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H842–H853. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, K.; Fukumura, S.; Nikaido, K.; Tachi, N.; Kozuka, N.; Seino, T.; Hatakeyama, K.; Mori, M.; Ito, Y.M.; Takami, A.; et al. Resveratrol improves motor function in patients with muscular dystrophies: An open-label, single-arm, phase iia study. Sci. Rep. 2020, 10, 20585. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D. Resveratrol and cardiovascular diseases. Nutrients 2016, 8, 250. [Google Scholar] [CrossRef] [PubMed]
- Jardim, F.R.; de Rossi, F.T.; Nascimento, M.X.; da Silva Barros, R.G.; Borges, P.A.; Prescilio, I.C.; de Oliveira, M.R. Resveratrol and brain mitochondria: A review. Mol. Neurobiol. 2018, 55, 2085–2101. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an anti-cancer agent: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.Y.; Tain, Y.L.; Yu, H.R.; Huang, L.T. The effects of resveratrol in the treatment of metabolic syndrome. Int. J. Mol. Sci. 2019, 20, 535. [Google Scholar] [CrossRef] [PubMed]
- Brown, V.A.; Patel, K.R.; Viskaduraki, M.; Crowell, J.A.; Perloff, M.; Booth, T.D.; Vasilinin, G.; Sen, A.; Schinas, A.M.; Piccirilli, G.; et al. Repeat dose study of the cancer chemopreventive agent resveratrol in healthy volunteers: Safety, pharmacokinetics, and effect on the insulin-like growth factor axis. Cancer Res. 2010, 70, 9003–9011. [Google Scholar] [CrossRef]
- Ratz-Lyko, A.; Arct, J. Resveratrol as an active ingredient for cosmetic and dermatological applications: A review. J. Cosmet. Laser Ther. 2019, 21, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Schoonjans, K.; Auwerx, J. Sirtuin functions in health and disease. Mol. Endocrinol. 2007, 21, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: A dawn for evolutionary medicine. Annu. Rev. Genet. 2005, 39, 359–407. [Google Scholar] [CrossRef]
- Liang, H.; Ward, W.F. Pgc-1alpha: A key regulator of energy metabolism. Adv. Physiol. Educ. 2006, 30, 145–151. [Google Scholar] [CrossRef]
- Rowe, G.C.; Jiang, A.; Arany, Z. Pgc-1 coactivators in cardiac development and disease. Circ. Res. 2010, 107, 825–838. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating sirt1 and pgc-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Pearson, K.J.; Baur, J.A.; Lewis, K.N.; Peshkin, L.; Price, N.L.; Labinskyy, N.; Swindell, W.R.; Kamara, D.; Minor, R.K.; Perez, E.; et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab. 2008, 8, 157–168. [Google Scholar] [CrossRef]
- Higashida, K.; Kim, S.H.; Jung, S.R.; Asaka, M.; Holloszy, J.O.; Han, D.H. Effects of resveratrol and sirt1 on pgc-1alpha activity and mitochondrial biogenesis: A reevaluation. PLoS Biol. 2013, 11, e1001603. [Google Scholar] [CrossRef]
- Jager, S.; Handschin, C.; St-Pierre, J.; Spiegelman, B.M. Amp-activated protein kinase (ampk) action in skeletal muscle via direct phosphorylation of pgc-1alpha. Proc. Natl. Acad. Sci. USA 2007, 104, 12017–12022. [Google Scholar] [CrossRef] [PubMed]
- Fulco, M.; Sartorelli, V. Comparing and contrasting the roles of ampk and sirt1 in metabolic tissues. Cell Cycle 2008, 7, 3669–3679. [Google Scholar] [CrossRef] [PubMed]
- Price, N.L.; Gomes, A.P.; Ling, A.J.; Duarte, F.V.; Martin-Montalvo, A.; North, B.J.; Agarwal, B.; Ye, L.; Ramadori, G.; Teodoro, J.S.; et al. Sirt1 is required for ampk activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012, 15, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.J.; Xu, C.; Liu, M.Y.; Wang, B.K.; Liu, W.B.; Chen, D.H.; Zhang, L.; Xu, C.Y.; Li, X.F. Resveratrol improves the energy sensing and glycolipid metabolism of blunt snout bream megalobrama amblycephala fed high-carbohydrate diets by activating the ampk-sirt1-pgc-1alpha network. Front. Physiol. 2018, 9, 1258. [Google Scholar] [CrossRef] [PubMed]
- Qiang, L.; Wang, L.; Kon, N.; Zhao, W.; Lee, S.; Zhang, Y.; Rosenbaum, M.; Zhao, Y.; Gu, W.; Farmer, S.R.; et al. Brown remodeling of white adipose tissue by sirt1-dependent deacetylation of ppargamma. Cell 2012, 150, 620–632. [Google Scholar] [CrossRef]
- Zhao, Q.; Tian, Z.; Zhou, G.; Niu, Q.; Chen, J.; Li, P.; Dong, L.; Xia, T.; Zhang, S.; Wang, A. Sirt1-dependent mitochondrial biogenesis supports therapeutic effects of resveratrol against neurodevelopment damage by fluoride. Theranostics 2020, 10, 4822–4838. [Google Scholar] [CrossRef]
- Csiszar, A.; Labinskyy, N.; Pinto, J.T.; Ballabh, P.; Zhang, H.; Losonczy, G.; Pearson, K.; de Cabo, R.; Pacher, P.; Zhang, C.; et al. Resveratrol induces mitochondrial biogenesis in endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H13–H20. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Chen, S.D.; Hsu, C.Y.; Chen, S.F.; Chen, N.C.; Jou, S.B. Resveratrol promotes mitochondrial biogenesis and protects against seizure-induced neuronal cell damage in the hippocampus following status epilepticus by activation of the pgc-1alpha signaling pathway. Int. J. Mol. Sci. 2019, 20, 998. [Google Scholar] [CrossRef]
- Song, C.; Zhang, Y.; Cheng, L.; Zhang, L.; Li, X.; Shi, M.; Zhao, H. Effects of resveratrol combined with soy isoflavones on apoptosis induced by oxidative stress in hippocampus of aging model rats. Wei Sheng Yan Jiu 2020, 49, 932–937. [Google Scholar] [CrossRef]
- Vincenzi, K.L.; Maia, T.P.; Delmonego, L.; Lima, A.B.; Pscheidt, L.C.; Delwing-Dal Magro, D.; Delwing-de Lima, D. Effects of resveratrol on alterations in cerebrum energy metabolism caused by metabolites accumulated in type i citrullinemia in rats. Naunyn. Schmiedebergs Arch. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Low, I.C.; Chen, Z.X.; Pervaiz, S. Bcl-2 modulates resveratrol-induced ros production by regulating mitochondrial respiration in tumor cells. Antioxid. Redox Signal. 2010, 13, 807–819. [Google Scholar] [CrossRef]
- Chen, K.G.; Kang, R.R.; Sun, Q.; Liu, C.; Ma, Z.; Liu, K.; Deng, Y.; Liu, W.; Xu, B. Resveratrol ameliorates disorders of mitochondrial biogenesis and mitophagy in rats continuously exposed to benzo(a)pyrene from embryonic development through adolescence. Toxicology 2020, 442, 152532. [Google Scholar] [CrossRef] [PubMed]
- Saracco, S.A.; Fox, T.D. Cox18p is required for export of the mitochondrially encoded saccharomyces cerevisiae cox2p c-tail and interacts with pnt1p and mss2p in the inner membrane. Mol. Biol. Cell 2002, 13, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Alway, S.E.; McCrory, J.L.; Kearcher, K.; Vickers, A.; Frear, B.; Gilleland, D.L.; Bonner, D.E.; Thomas, J.M.; Donley, D.A.; Lively, M.W.; et al. Resveratrol enhances exercise-induced cellular and functional adaptations of skeletal muscle in older men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1595–1606. [Google Scholar] [CrossRef] [PubMed]
- Desquiret-Dumas, V.; Gueguen, N.; Leman, G.; Baron, S.; Nivet-Antoine, V.; Chupin, S.; Chevrollier, A.; Vessieres, E.; Ayer, A.; Ferre, M.; et al. Resveratrol induces a mitochondrial complex i-dependent increase in nadh oxidation responsible for sirtuin activation in liver cells. J. Biol. Chem. 2013, 288, 36662–36675. [Google Scholar] [CrossRef] [PubMed]
- Zini, R.; Morin, C.; Bertelli, A.; Bertelli, A.A.; Tillement, J.P. Effects of resveratrol on the rat brain respiratory chain. Drugs Exp. Clin. Res. 1999, 25, 87–97. [Google Scholar]
- Gledhill, J.R.; Montgomery, M.G.; Leslie, A.G.; Walker, J.E. Mechanism of inhibition of bovine f1-atpase by resveratrol and related polyphenols. Proc. Natl. Acad. Sci. USA 2007, 104, 13632–13637. [Google Scholar] [CrossRef]
- Kipp, J.L.; Ramirez, V.D. Effect of estradiol, diethylstilbestrol, and resveratrol on f0f1-atpase activity from mitochondrial preparations of rat heart, liver, and brain. Endocrine 2001, 15, 165–175. [Google Scholar] [CrossRef]
- Zheng, J.; Ramirez, V.D. Inhibition of mitochondrial proton f0f1-atpase/atp synthase by polyphenolic phytochemicals. Br. J. Pharmacol. 2000, 130, 1115–1123. [Google Scholar] [CrossRef]
- Khanduja, K.L.; Bhardwaj, A. Stable free radical scavenging and antiperoxidative properties of resveratrol compared in vitro with some other bioflavonoids. Indian J. Biochem. Biophys. 2003, 40, 416–422. [Google Scholar]
- Juan, M.E.; Wenzel, U.; Daniel, H.; Planas, J.M. Resveratrol induces apoptosis through ros-dependent mitochondria pathway in ht-29 human colorectal carcinoma cells. J. Agric. Food Chem. 2008, 56, 4813–4818. [Google Scholar] [CrossRef]
- Rai, G.; Suman, S.; Mishra, S.; Shukla, Y. Evaluation of growth inhibitory response of resveratrol and salinomycin combinations against triple negative breast cancer cells. Biomed. Pharmacother. 2017, 89, 1142–1151. [Google Scholar] [CrossRef]
- Luo, H.; Yang, A.; Schulte, B.A.; Wargovich, M.J.; Wang, G.Y. Resveratrol induces premature senescence in lung cancer cells via ros-mediated DNA damage. PLoS ONE 2013, 8, e60065. [Google Scholar] [CrossRef]
- Tseng, S.H.; Lin, S.M.; Chen, J.C.; Su, Y.H.; Huang, H.Y.; Chen, C.K.; Lin, P.Y.; Chen, Y. Resveratrol suppresses the angiogenesis and tumor growth of gliomas in rats. Clin. Cancer Res. 2004, 10, 2190–2202. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stokes, J., 3rd; Singh, U.P.; Scissum-Gunn, K.; Singh, R.; Manne, U.; Mishra, M.K. Prolonged exposure of resveratrol induces reactive superoxide species-independent apoptosis in murine prostate cells. Tumour Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Chavez, J.; Fonseca-Sanchez, M.A.; Arechaga-Ocampo, E.; Flores-Perez, A.; Palacios-Rodriguez, Y.; Dominguez-Gomez, G.; Marchat, L.A.; Fuentes-Mera, L.; Mendoza-Hernandez, G.; Gariglio, P.; et al. Proteomic profiling reveals that resveratrol inhibits hsp27 expression and sensitizes breast cancer cells to doxorubicin therapy. PLoS ONE 2013, 8, e64378. [Google Scholar] [CrossRef]
- Osman, A.M.; Al-Harthi, S.E.; AlArabi, O.M.; Elshal, M.F.; Ramadan, W.S.; Alaama, M.N.; Al-Kreathy, H.M.; Damanhouri, Z.A.; Osman, O.H. Chemosensetizing and cardioprotective effects of resveratrol in doxorubicin- treated animals. Cancer Cell Int. 2013, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.M.; Telity, S.A.; Telity, S.A.; Damanhouri, Z.A.; Al-Harthy, S.E.; Al-Kreathy, H.M.; Ramadan, W.S.; Elshal, M.F.; Khan, L.M.; Kamel, F. Chemosensitizing and nephroprotective effect of resveratrol in cisplatin -treated animals. Cancer Cell Int. 2015, 15, 6. [Google Scholar] [CrossRef]
- Sprouse, A.A.; Herbert, B.S. Resveratrol augments paclitaxel treatment in mda-mb-231 and paclitaxel-resistant mda-mb-231 breast cancer cells. Anticancer. Res. 2014, 34, 5363–5374. [Google Scholar]
- Tu, L.F.; Cao, L.F.; Zhang, Y.H.; Guo, Y.L.; Zhou, Y.F.; Lu, W.Q.; Zhang, T.Z.; Zhang, T.; Zhang, G.X.; Kurihara, H.; et al. Sirt3-dependent deacetylation of cox-1 counteracts oxidative stress-induced cell apoptosis. FASEB J. 2019, 33, 14118–14128. [Google Scholar] [CrossRef] [PubMed]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef]
- Wood, J.G.; Rogina, B.; Lavu, S.; Howitz, K.; Helfand, S.L.; Tatar, M.; Sinclair, D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004, 430, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Rezaizadehnajafi, L.; Wink, M. Influence of resveratrol on oxidative stress resistance and life span in caenorhabditis elegans. J. Pharm. Pharmacol. 2013, 65, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, M.; Kim, S.K.; Berdichevsky, A.; Guarente, L. A role for sir-2.1 regulation of er stress response genes in determining c. Elegans life span. Dev. Cell 2005, 9, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.H.; Goupil, S.; Garber, G.B.; Helfand, S.L. An accelerated assay for the identification of lifespan-extending interventions in drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2004, 101, 12980–12985. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Hubbard, B.P. Lifespan and healthspan extension by resveratrol. Biochim. Biophys. Acta 2015, 1852, 1209–1218. [Google Scholar] [CrossRef]
- Hector, K.L.; Lagisz, M.; Nakagawa, S. The effect of resveratrol on longevity across species: A meta-analysis. Biol. Lett. 2012, 8, 790–793. [Google Scholar] [CrossRef]
- Semba, R.D.; Ferrucci, L.; Bartali, B.; Urpi-Sarda, M.; Zamora-Ros, R.; Sun, K.; Cherubini, A.; Bandinelli, S.; Andres-Lacueva, C. Resveratrol levels and all-cause mortality in older community-dwelling adults. JAMA Intern. Med. 2014, 174, 1077–1084. [Google Scholar] [CrossRef]
- De Paepe, B.; Van Coster, R. A critical assessment of the therapeutic potential of resveratrol supplements for treating mitochondrial disorders. Nutrients 2017, 9, 1017. [Google Scholar] [CrossRef]
- Lopes Costa, A.; le Bachelier, C.; Mathieu, L.; Rotig, A.; Boneh, A.; de Lonlay, P.; Tarnopolsky, M.A.; Thorburn, D.R.; Bastin, J.; Djouadi, F. Beneficial effects of resveratrol on respiratory chain defects in patients’ fibroblasts involve estrogen receptor and estrogen-related receptor alpha signaling. Hum. Mol. Genet. 2014, 23, 2106–2119. [Google Scholar] [CrossRef]
- Douiev, L.; Soiferman, D.; Alban, C.; Saada, A. The effects of ascorbate, n-acetylcysteine, and resveratrol on fibroblasts from patients with mitochondrial disorders. J. Clin. Med. 2016, 6, 1. [Google Scholar] [CrossRef]
- De Paepe, B.; Vandemeulebroecke, K.; Smet, J.; Vanlander, A.; Seneca, S.; Lissens, W.; van Hove, J.L.; Deschepper, E.; Briones, P.; van Coster, R. Effect of resveratrol on cultured skin fibroblasts from patients with oxidative phosphorylation defects. Phytother Res. 2014, 28, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, C.; Artacho, R.; Gimenez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef]
- Fraga, C.G.; Galleano, M.; Verstraeten, S.V.; Oteiza, P.I. Basic biochemical mechanisms behind the health benefits of polyphenols. Mol. Asp. Med. 2010, 31, 435–445. [Google Scholar] [CrossRef]
- Ottaviani, J.I.; Momma, T.Y.; Heiss, C.; Kwik-Uribe, C.; Schroeter, H.; Keen, C.L. The stereochemical configuration of flavanols influences the level and metabolism of flavanols in humans and their biological activity in vivo. Free Radic. Biol. Med. 2011, 50, 237–244. [Google Scholar] [CrossRef]
- Actis-Goretta, L.; Leveques, A.; Giuffrida, F.; Romanov-Michailidis, F.; Viton, F.; Barron, D.; Duenas-Paton, M.; Gonzalez-Manzano, S.; Santos-Buelga, C.; Williamson, G.; et al. Elucidation of (–)-epicatechin metabolites after ingestion of chocolate by healthy humans. Free Radic. Biol. Med. 2012, 53, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Bernatonieneand, J.D.; Kopustinskiene, M. The role of catechins in cellular responses to oxidative stress. Molecules 2018, 23, 965. [Google Scholar] [CrossRef]
- Mangels, D.R.; Mohler, E.R., 3rd. Catechins as potential mediators of cardiovascular health. Arter. Thromb. Vasc. Biol. 2017, 37, 757–763. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Yazdi, H.S.; Samarghandian, S. The protective effects of green tea catechins in the management of neurodegenerative diseases: A review. Curr. Drug Discov. Technol. 2019, 16, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.; Elbaz, H.A.; Lee, I.; Zielske, S.P.; Malek, M.H.; Hüttemann, M. Molecular mechanisms and therapeutic effects of (–)-epicatechin and other polyphenols in cancer, inflammation, diabetes, and neurodegeneration. Oxid. Med. Cell Longev. 2015, 2015, 181260. [Google Scholar] [CrossRef]
- Cremonini, E.; Iglesias, D.E.; Kang, J.; Lombardo, G.E.; Mostofinejad, Z.; Wang, Z.; Zhu, W.; Oteiza, P.I. (–)-epicatechin and the comorbidities of obesity. Arch. Biochem. Biophys. 2020, 690, 108505. [Google Scholar] [CrossRef]
- Rahman, T.; Hosen, I.; Islam, M.M.T.; Shekhar, H.U. Oxidative stress and human health. Adv. Biosci. Biotechnol. 2012, 3, 997–1019. [Google Scholar] [CrossRef]
- McCullough, M.L.; Chevaux, K.; Jackson, L.; Preston, M.; Martinez, G.; Schmitz, H.H.; Coletti, C.; Campos, H.; Hollenberg, N.K. Hypertension, the kuna, and the epidemiology of flavanols. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. 2), S103–S109. [Google Scholar] [CrossRef]
- Wollgast, J.; Anklam, E. Review on polyphenols in theobroma cacao: Changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Engler, M.B.; Engler, M.M. The emerging role of flavonoid-rich cocoa and chocolate in cardiovascular health and disease. Nutr. Rev. 2006, 64, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Shrime, M.G.; Bauer, S.R.; McDonald, A.C.; Chowdhury, N.H.; Coltart, C.E.; Ding, E.L. Flavonoid-rich cocoa consumption affects multiple cardiovascular risk factors in a meta-analysis of short-term studies. J. Nutr. 2011, 141, 1982–1988. [Google Scholar] [CrossRef]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and chocolate in human health and disease. Antioxid. Redox Signal. 2011, 15, 2779–2811. [Google Scholar] [CrossRef]
- Faridi, Z.; Njike, V.Y.; Dutta, S.; Ali, A.; Katz, D.L. Acute dark chocolate and cocoa ingestion and endothelial function: A randomized controlled crossover trial. Am. J. Clin. Nutr. 2008, 88, 58–63. [Google Scholar] [CrossRef]
- Oteiza, P.I.; Fraga, C.G.; Mills, D.A.; Taft, D.H. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Asp. Med. 2018, 61, 41–49. [Google Scholar] [CrossRef]
- Nehlig, A. The neuroprotective effects of cocoa flavanol and its influence on cognitive performance. Br. J. Clin. Pharmacol. 2013, 75, 716–727. [Google Scholar] [CrossRef]
- Taub, P.R.; Ramirez-Sanchez, I.; Ciaraldi, T.P.; Gonzalez-Basurto, S.; Coral-Vazquez, R.; Perkins, G.; Hogan, M.; Maisel, A.S.; Henry, R.R.; Ceballos, G.; et al. Perturbations in skeletal muscle sarcomere structure in patients with heart failure and type 2 diabetes: Restorative effects of (–)-epicatechin-rich cocoa. Clin. Sci. 2013, 125, 383–389. [Google Scholar] [CrossRef]
- Ramirez-Sanchez, I.; de los Santos, S.; Gonzalez-Basurto, S.; Canto, P.; Mendoza-Lorenzo, P.; Palma-Flores, C.; Ceballos-Reyes, G.; Villarreal, F.; Zentella-Dehesa, A.; Coral-Vazquez, R. (–)-epicatechin improves mitochondrial-related protein levels and ameliorates oxidative stress in dystrophic delta-sarcoglycan null mouse striated muscle. FEBS J. 2014, 281, 5567–5580. [Google Scholar] [CrossRef]
- Angel Garcia-Merino, J.; Moreno-Perezde, D.; Lucas, B.; Montalvo-Lominchar, M.G.; Munoz, E.; Sanchez, L.; Naclerio, F.; Herrera-Rocha, K.M.; Moreno-Jimenez, M.R.; Rocha-Guzman, N.E.; et al. Chronic flavanol-rich cocoa powder supplementation reduces body fat mass in endurance athletes by modifying the follistatin/myostatin ratio and leptin levels. Food Funct. 2020, 11, 3441–3450. [Google Scholar] [CrossRef]
- Cremonini, E.; Fraga, C.G.; Oteiza, P.I. Epicatechin in the control of glucose homeostasis: Involvement of redox-regulated mechanisms. Free Radic. Biol. Med. 2019, 130, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Buitrago-Lopez, A.; Sanderson, J.; Johnson, L.; Warnakula, S.; Wood, A.; di Angelantonio, E.; Franco, O.H. Chocolate consumption and cardiometabolic disorders: Systematic review and meta-analysis. BMJ 2011, 343, d4488. [Google Scholar] [CrossRef] [PubMed]
- Daussin, F.N.; Heyman, E.; Burelle, Y. Effects of (–)-epicatechin on mitochondria. Nutr. Rev. 2021, 79, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Salmean, G.; Ortiz-Vilchis, P.; Vacaseydel, C.M.; Garduno-Siciliano, L.; Chamorro-Cevallos, G.; Meaney, E.; Villafana, S.; Villarreal, F.; Ceballos, G.; Ramirez-Sanchez, I. Effects of (–)-epicatechin on a diet-induced rat model of cardiometabolic risk factors. Eur. J. Pharmacol. 2014, 728, 24–30. [Google Scholar] [CrossRef]
- Panneerselvam, M.; Tsutsumi, Y.M.; Bonds, J.A.; Horikawa, Y.T.; Saldana, M.; Dalton, N.D.; Head, B.P.; Patel, P.M.; Roth, D.M.; Patel, H.H. Dark chocolate receptors: Epicatechin-induced cardiac protection is dependent on delta-opioid receptor stimulation. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H1604–H1609. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Sanchez, I.; Mansour, C.; Navarrete-Yanez, V.; Ayala-Hernandez, M.; Guevara, G.; Castillo, C.; Loredo, M.; Bustamante, M.; Ceballos, G.; Villarreal, F. Epicatechin induced reversal of endothelial cell aging and improved vascular function: Underlying mechanisms. Food Funct. 2018, 9, 4802–4813. [Google Scholar] [CrossRef]
- Rozza, A.L.; Hiruma-Lima, C.A.; Tanimoto, A.; Pellizzon, C.H. Morphologic and pharmacological investigations in the epicatechin gastroprotective effect. Evid. Based Complement. Alternat. Med. 2012, 2012, 708156. [Google Scholar] [CrossRef]
- Navarrete-Yanez, V.; Garate-Carrillo, A.; Rodriguez, A.; Mendoza-Lorenzo, P.; Ceballos, G.; Calzada-Mendoza, C.; Hogan, M.C.; Villarreal, F.; Ramirez-Sanchez, I. Effects of (–)-epicatechin on neuroinflammation and hyperphosphorylation of tau in the hippocampus of aged mice. Food Funct. 2020, 11, 10351–10361. [Google Scholar] [CrossRef]
- Nogueira, L.; Ramirez-Sanchez, I.; Perkins, G.A.; Murphy, A.; Taub, P.R.; Ceballos, G.; Villarreal, F.J.; Hogan, M.C.; Malek, M.H. Epicatechin enhances fatigue resistance and oxidative capacity in mouse muscle. J. Physiol. 2011, 589, 4615–4631. [Google Scholar] [CrossRef]
- Schwarz, N.A.; Blahnik, Z.J.; Prahadeeswaran, S.; McKinley-Barnard, S.K.; Holden, S.L.; Waldhelm, A. (–)-epicatechin supplementation inhibits aerobic adaptations to cycling exercise in humans. Front. Nutr. 2018, 5, 132. [Google Scholar] [CrossRef] [PubMed]
- Si, H.; Wang, X.; Zhang, L.; Parnell, L.D.; Admed, B.; LeRoith, T.; Ansah, T.A.; Zhang, L.; Li, J.; Ordovas, J.M.; et al. Dietary epicatechin improves survival and delays skeletal muscle degeneration in aged mice. FASEB J. 2019, 33, 965–977. [Google Scholar] [CrossRef]
- Gutierrez-Salmean, G.; Ortiz-Vilchis, P.; Vacaseydel, C.M.; Rubio-Gayosso, I.; Meaney, E.; Villarreal, F.; Ramirez-Sanchez, I.; Ceballos, G. Acute effects of an oral supplement of (–)-epicatechin on postprandial fat and carbohydrate metabolism in normal and overweight subjects. Food Funct. 2014, 5, 521–527. [Google Scholar] [CrossRef]
- Cremonini, E.; Bettaieb, A.; Haj, F.G.; Fraga, C.G.; Oteiza, P.I. (–)-epicatechin improves insulin sensitivity in high fat diet-fed mice. Arch. Biochem. Biophys. 2016, 599, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Nagayasu, S.; Suzuki, S.; Iwashita, M.; Yamashita, A.; Shinjo, T.; Sanui, T.; Kushiyama, A.; Kanematsu, T.; Asano, V.; et al. Epicatechin downregulates adipose tissue ccl19 expression and thereby ameliorates diet-induced obesity and insulin resistance. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 249–259. [Google Scholar] [CrossRef]
- Ramirez-Sanchez, I.; Rodriguez, A.; Moreno-Ulloa, A.; Ceballos, G.; Villarreal, F. (–)-epicatechin-induced recovery of mitochondria from simulated diabetes: Potential role of endothelial nitric oxide synthase. Diab. Vasc. Dis. Res. 2016, 13, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ulloa, A.; Nogueira, L.; Rodriguez, A.; Barboza, J.; Hogan, M.C.; Ceballos, G.; Villarreal, F.; Ramirez-Sanchez, I. Recovery of indicators of mitochondrial biogenesis, oxidative stress, and aging with (–)-epicatechin in senile mice. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 1370–1378. [Google Scholar] [CrossRef]
- Taub, P.R.; Ramirez-Sanchez, I.; Ciaraldi, T.P.; Perkins, G.; Murphy, A.N.; Naviaux, R.; Hogan, M.; Maisel, A.S.; Henry, R.R.; Ceballos, G.; et al. Alterations in skeletal muscle indicators of mitochondrial structure and biogenesis in patients with type 2 diabetes and heart failure: Effects of epicatechin rich cocoa. Clin. Transl. Sci. 2012, 5, 43–47. [Google Scholar] [CrossRef]
- Moreno-Ulloa, A.; Cid, A.; Rubio-Gayosso, I.; Ceballos, G.; Villarreal, F.; Ramirez-Sanchez, I. Effects of (–)-epicatechin and derivatives on nitric oxide mediated induction of mitochondrial proteins. Bioorg. Med. Chem. Lett. 2013, 23, 4441–4446. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Savickas, A.; Vetchy, D.; Masteikova, R.; Bernatoniene, A.K.J. Direct effects of (–)-epicatechin and procyanidin b2 on the respiration of rat heart mitochondria. Biomed. Res. Int. 2015, 2015, 232836. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.M.; Ramirez-Sanchez, I.; Oskarsson, B.; Joyce, N.; Aguilar, C.; Nicorici, A.; Dayan, J.; Goude, E.; Abresch, R.T.; Villarreal, F.; et al. (–)-epicatechin induces mitochondrial biogenesis and markers of muscle regeneration in adults with becker muscular dystrophy. Muscle Nerve 2021, 63, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ulloa, A.; Miranda-Cervantes, A.; Licea-Navarro, A.; Mansour, C.; Beltran-Partida, E.; Donis-Maturano, L.; de la Herran, H.C.D.; Villarreal, F.; Alvarez-Delgado, C. (–)-epicatechin stimulates mitochondrial biogenesis and cell growth in c2c12 myotubes via the g-protein coupled estrogen receptor. Eur. J. Pharmacol. 2018, 822, 95–107. [Google Scholar] [CrossRef]
- Hüttemann, M.; Lee, I.; Malek, M.H. (–)-epicatechin maintains endurance training adaptation in mice after 14 days of detraining. FASEB J. 2012, 26, 1413–1422. [Google Scholar] [CrossRef]
- Lee, I.; Hüttemann, M.; Malek, M.H. (–)-epicatechin attenuates degradation of mouse oxidative muscle following hindlimb suspension. J. Strength Cond Res. 2016, 30, 1–10. [Google Scholar] [CrossRef]
- Hüttemann, M.; Lee, I.; Perkins, G.A.; Britton, S.L.; Koch, L.G.; Malek, M.H. (–)-epicatechin is associated with increased angiogenic and mitochondrial signalling in the hindlimb of rats selectively bred for innate low running capacity. Clin. Sci. 2013, 124, 663–674. [Google Scholar] [CrossRef]
- Lee, I.; Hüttemann, M.; Kruger, A.; Bollig-Fischer, A.; Malek, M.H. (–)-epicatechin combined with 8 weeks of treadmill exercise is associated with increased angiogenic and mitochondrial signaling in mice. Front. Pharmacol. 2015, 6, 43. [Google Scholar] [CrossRef]
- Bitner, B.F.; Ray, J.D.; Kener, K.B.; Herring, J.A.; Tueller, J.A.; Johnson, D.K.; Freitas, C.M.T.; Fausnacht, D.W.; Allen, M.E.; Thomson, A.H.; et al. Common gut microbial metabolites of dietary flavonoids exert potent protective activities in beta-cells and skeletal muscle cells. J. Nutr. Biochem. 2018, 62, 95–107. [Google Scholar] [CrossRef]
- Varela, C.E.; Rodriguez, A.; Romero-Valdovinos, M.; Mendoza-Lorenzo, P.; Mansour, C.; Ceballos, G.; Villarreal, F.; Ramirez-Sanchez, I. Browning effects of (–)-epicatechin on adipocytes and white adipose tissue. Eur. J. Pharmacol. 2017, 811, 48–59. [Google Scholar] [CrossRef]
- Elbaz, H.A.; Lee, I.; Antwih, D.A.; Liu, J.; Hüttemann, M.; Zielske, S.P. Epicatechin stimulates mitochondrial activity and selectively sensitizes cancer cells to radiation. PLoS ONE 2014, 9, e88322. [Google Scholar] [CrossRef]
- Allgrove, J.; Farrell, E.; Gleeson, M.; Williamson, G.; Cooper, K. Regular dark chocolate consumption’s reduction of oxidative stress and increase of free-fatty-acid mobilization in response to prolonged cycling. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 113–123. [Google Scholar] [CrossRef]
- Patel, R.K.; Brouner, J.; Spendiff, O. Dark chocolate supplementation reduces the oxygen cost of moderate intensity cycling. J. Int. Soc. Sports Nutr. 2015, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, F.G.; Fisher, M.G.; Thornley, T.T.; Roemer, K.; Pritchett, R.; Freitas, E.C.; Pritchett, K. Cocoa flavanol effects on markers of oxidative stress and recovery after muscle damage protocol in elite rugby players. Nutrition 2019, 62, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.H.; Park, K.S.; Lee, K.U.; Lee, H.K. Mitochondrial metabolism and diabetes. J. Diabetes Investig. 2010, 1, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Castellano-Gonzalez, G.; Pichaud, N.; Ballard, J.W.; Bessede, A.; Marcal, H.; Guillemin, G.J. Epigallocatechin-3-gallate induces oxidative phosphorylation by activating cytochrome c oxidase in human cultured neurons and astrocytes. Oncotarget 2016, 7, 7426–7440. [Google Scholar] [CrossRef]
- Zhao, G.; He, F.; Wu, C.; Li, P.; Li, N.; Deng, J.; Zhu, G.; Ren, W.; Peng, Y. Betaine in inflammation: Mechanistic aspects and applications. Front. Immunol. 2018, 9, 1070. [Google Scholar] [CrossRef]
- Sakamoto, A.; Murata, N. Genetic engineering of glycinebetaine synthesis in plants: Current status and implications for enhancement of stress tolerance. J. Exp. Bot. 2000, 51, 81–88. [Google Scholar] [CrossRef]
- Day, C.R.; Kempson, S.A. Betaine chemistry, roles, and potential use in liver disease. Biochim. Biophys. Acta 2016, 1860, 1098–1106. [Google Scholar] [CrossRef]
- Craig, S.A. Betaine in human nutrition. Am. J. Clin. Nutr. 2004, 80, 539–549. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Mar, M.H.; Howe, J.C.; Holden, J.M. Concentrations of choline-containing compounds and betaine in common foods. J. Nutr. 2003, 133, 1302–1307. [Google Scholar] [CrossRef]
- Lever, M.; Sizeland, P.C.; Bason, L.M.; Hayman, C.M.; Chambers, S.T. Glycine betaine and proline betaine in human blood and urine. Biochim. Biophys. Acta 1994, 1200, 259–264. [Google Scholar] [CrossRef]
- Schwahn, B.C.; Hafner, D.; Hohlfeld, T.; Balkenhol, N.; Laryea, M.D.; Wendel, U. Pharmacokinetics of oral betaine in healthy subjects and patients with homocystinuria. Br. J. Clin. Pharmacol. 2003, 55, 6–13. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Li, H.; Lai, F.; Li, X.; Tang, Y.; Min, T.; Wu, H. Antioxidant mechanism of betaine without free radical scavenging ability. J. Agric. Food Chem. 2016, 64, 7921–7930. [Google Scholar] [CrossRef]
- Fernández, C.; Gallego, L.; Lopez-Bote, C.J. Effect of betaine on fat content in growing lambs. Anim. Feed Sci. Technol. 1998, 73, 329–338. [Google Scholar] [CrossRef]
- Eklund, M.; Bauer, E.; Wamatu, J.; Mosenthin, R. Potential nutritional and physiological functions of betaine in livestock. Nutr. Res. Rev. 2005, 18, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.Y.; Xu, Z.R.; Li, W.F. Effects of betaine on growth performance and carcass characteristics in growing pigs. Asian Australas. J. Anim. Sci. 2004, 17, 1700–1704. [Google Scholar] [CrossRef]
- Best, P. Designer nutrition for pigs: Feeding against backfat. Feed Int. 1994, 5, 38–39. [Google Scholar]
- Gudev, D.; Popova-Ralcheva, S.; Yanchev, I.; Moneva, P.; Petkov, E.; Ignatova, M. Effect of betaine on egg performance and some blood constituents in laying hens reared indoor under natural summer temperatures and varying levels of air ammonia. Bulg. J. Agric. Sci. 2011, 17, 859–866. [Google Scholar]
- Al-Shukri, A.Y.; Kaab, H.T.; Abdulwahab, H.M. Effect of betaine on some productive traits of turkey under heat stress. Kufa J. Vet. Med. Sci. 2012, 3, 12–20. [Google Scholar]
- Akhavan-Salamatand, H.H.; Ghasemi, A. Alleviation of chronic heat stress in broilers by dietary supplementation of betaine and turmeric rhizome powder: Dynamics of performance, leukocyte profile, humoral immunity, and antioxidant status. Trop Anim. Health Prod. 2016, 48, 181–188. [Google Scholar] [CrossRef]
- Mendoza, S.M.; Boyd, R.D.; Ferket, P.R.; van Heugten, E. Effects of dietary supplementation of the osmolyte betaine on growing pig performance and serological and hematological indices during thermoneutral and heat-stressed conditions. J. Anim. Sci. 2017, 95, 5040–5053. [Google Scholar] [CrossRef]
- Clarke, W.C.; Virtanen, E.; Blackburn, J.; Higgs, D.A. Effects of a dietary betaine/amino acid additive on growth and seawater adaptation in yearling chinook salmon. Aquaculture 1994, 121, 137–145. [Google Scholar] [CrossRef]
- Ali, S.; Abbas, Z.; Seleiman, M.F.; Rizwan, M.; Yava, S.I.; Alhammad, B.A.; Shami, A.; Hasanuzzaman, M.; Kalderis, D. Glycine betaine accumulation, significance and interests for heavy metal tolerance in plants. Plants 2020, 9, 896. [Google Scholar] [CrossRef]
- Allard, F.; Houde, M.; Krol, M.; Ivanov, A.; Huner, N.P.A.; Sarhan, F. Betaine improves freezing tolerance in wheat. Plant Cell Physiol. 1998, 39, 1194–1202. [Google Scholar] [CrossRef]
- Nomura, M.; Muramoto, Y.; Yasuda, S.; Takabe, T.; Kishitani, S. The accumulation of glycinebetaine during cold acclimation in early and late cultivars of barley. Euphytica 1995, 83, 247–250. [Google Scholar] [CrossRef]
- Zou, H.; Chen, N.; Shi, M.; Xian, M.; Song, Y.; Liu, J. The metabolism and biotechnological application of betaine in microorganism. Appl. Microbiol. Biotechnol. 2016, 100, 3865–3876. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Xu, P. Betaine and beet molasses enhance l-lactic acid production by bacillus coagulans. PLoS ONE 2014, 9, e100731. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.C.; Hynes, S.H.; Ingledew, W.M. Effects of particulate materials and osmoprotectants on very-high-gravity ethanolic fermentation by saccharomyces cerevisiae. Appl. Environ. Microbiol. 1994, 60, 1519–1524. [Google Scholar] [CrossRef]
- Holtmann, G.; Bremer, E. Thermoprotection of bacillus subtilis by exogenously provided glycine betaine and structurally related compatible solutes: Involvement of opu transporters. J. Bacteriol. 2004, 186, 1683–1693. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, H.; Guo, X.F.; Li, K.; Li, S.; Li, D. Effect of betaine on reducing body fat-a systematic review and meta-analysis of randomized controlled trials. Nutrients 2019, 11, 2480. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; Slow, S. The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin. Biochem. 2010, 43, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.S.; Kumar, T.; Dar, T.A.; Singh, L.R. Protein n-homocysteinylation: From cellular toxicity to neurodegeneration. Biochim. Biophys. Acta 2015, 1850, 2239–2245. [Google Scholar] [CrossRef] [PubMed]
- Wilcken, D.E.; Wilcken, B.; Dudman, N.P.; Tyrrell, P.A. Homocystinuria--the effects of betaine in the treatment of patients not responsive to pyridoxine. N. Engl. J. Med. 1983, 309, 448–453. [Google Scholar] [CrossRef]
- Da Deminice, R.; Silva, R.P.; Lamarre, S.G.; Kelly, K.B.; Jacobs, R.L.; Brosnan, M.E.; Brosnan, J.T. Betaine supplementation prevents fatty liver induced by a high-fat diet: Effects on one-carbon metabolism. Amino Acids 2015, 47, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Huang, L.; Gao, J.; Wen, S.; Tai, Y.; Chen, M.; Huang, Z.; Liu, R.; Tang, C.; Li, J. Betaine attenuates chronic alcoholinduced fatty liver by broadly regulating hepatic lipid metabolism. Mol. Med. Rep. 2017, 16, 5225–5234. [Google Scholar] [CrossRef]
- Arroyave-Ospina, J.C.; Wu, Z.; Geng, Y.; Moshage, H. Role of oxidative stress in the pathogenesis of non-alcoholic fatty liver disease: Implications for prevention and therapy. Antioxidants 2021, 10, 174. [Google Scholar] [CrossRef]
- You, M.; Crabb, D.W. Recent advances in alcoholic liver disease ii. Minireview: Molecular mechanisms of alcoholic fatty liver. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 287, G1–G6. [Google Scholar] [CrossRef]
- Idalsoaga, F.; Kulkarni, A.V.; Mousa, O.Y.; Arrese, M.; Arab, J.P. Non-alcoholic fatty liver disease and alcohol-related liver disease: Two intertwined entities. Front. Med. 2020, 7, 448. [Google Scholar] [CrossRef]
- Kathirvel, E.; Morgan, K.; Nandgiri, G.; Sandoval, B.C.; Caudill, M.A.; Bottiglieri, T.; French, S.W.; Morgan, T.R. Betaine improves nonalcoholic fatty liver and associated hepatic insulin resistance: A potential mechanism for hepatoprotection by betaine. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G1068–G1077. [Google Scholar] [CrossRef]
- Song, Z.; Deaciuc, I.; Zhou, Z.; Song, M.; Chen, T.; Hill, D.; McClain, C.J. Involvement of amp-activated protein kinase in beneficial effects of betaine on high-sucrose diet-induced hepatic steatosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G894–G902. [Google Scholar] [CrossRef]
- Ji, C.; Kaplowitz, N. Betaine decreases hyperhomocysteinemia, endoplasmic reticulum stress, and liver injury in alcohol-fed mice. Gastroenterology 2003, 124, 1488–1499. [Google Scholar] [CrossRef]
- Bucheli, P.; Gao, Q.; Redgwell, R.; Vidal, K.; Wang, J.; Zhang, W. Biomolecular and clinical aspects of chinese wolfberry. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 289–314. [Google Scholar]
- Mukherjee, S. Betaine and nonalcoholic steatohepatitis: Back to the future? World J. Gastroenterol. 2011, 17, 3663–3664. [Google Scholar] [CrossRef]
- Mukherjee, S. Role of betaine in liver disease-worth revisiting or has the die been cast? World J. Gastroenterol. 2020, 26, 5745–5748. [Google Scholar] [CrossRef] [PubMed]
- Ueland, P.M. Choline and betaine in health and disease. J. Inherit. Metab. Dis. 2011, 34, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Henke, W.; Herdel, K.; Jung, K.; Schnorr, D.; Loening, S.A. Betaine improves the pcr amplification of gc-rich DNA sequences. Nucleic Acids Res. 1997, 25, 3957–3958. [Google Scholar] [CrossRef] [PubMed]
- Cleland, D.; Krader, P.; McCree, C.; Tang, J.; Emerson, D. Glycine betaine as a cryoprotectant for prokaryotes. J. Microbiol. Methods 2004, 58, 31–38. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Casa, D.J.; Roti, M.W.; Lee, E.C.; Craig, S.A.; Sutherland, J.W.; Fiala, K.A.; Maresh, C.M. Influence of betaine consumption on strenuous running and sprinting in a hot environment. J. Strength Cond Res. 2008, 22, 851–860. [Google Scholar] [CrossRef]
- Hoffman, J.R.; Ratamess, N.A.; Kang, J.; Gonzalez, A.M.; Beller, N.A.; Craig, S.A. Effect of 15 days of betaine ingestion on concentric and eccentric force outputs during isokinetic exercise. J. Strength Cond Res. 2011, 25, 2235–2241. [Google Scholar] [CrossRef]
- Trepanowski, J.F.; Farney, T.M.; McCarthy, C.G.; Schilling, B.K.; Craig, S.A.; Bloomer, R.J. The effects of chronic betaine supplementation on exercise performance, skeletal muscle oxygen saturation and associated biochemical parameters in resistance trained men. J. Strength Cond Res. 2011, 25, 3461–3471. [Google Scholar] [CrossRef]
- Cholewa, J.M.; Wyszczelska-Rokiel, M.; Glowacki, R.; Jakubowski, H.; Matthews, T.; Wood, R.; Craig, S.A.; Paolone, V. Effects of betaine on body composition, performance, and homocysteine thiolactone. J. Int. Soc. Sports Nutr. 2013, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Jung, G.Y.; Won, S.B.; Kim, J.; Jeon, S.; Han, A.; Kwon, Y.H. Betaine alleviates hypertriglycemia and tau hyperphosphorylation in db/db mice. Toxicol. Res. 2013, 29, 7–14. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Varatharajalu, R.; Garige, M.; Leckey, L.C.; Arellanes-Robledo, J.; Reyes-Gordillo, K.; Shah, R.; Lakshman, M.R. Adverse signaling of scavenger receptor class b1 and pgc1s in alcoholic hepatosteatosis and steatohepatitis and protection by betaine in rat. Am. J. Pathol. 2014, 184, 2035–2044. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ma, J.; Meng, X.; Kang, S.Y.; Zhang, J.; Jung, H.W.; Park, Y.K. Regulatory effects of the fruit extract of lycium chinense and its active compound, betaine, on muscle differentiation and mitochondrial biogenesis in c2c12 cells. Biomed. Pharm. 2019, 118, 109297. [Google Scholar] [CrossRef]
- Tan, G.Y.; Yang, L.; Fu, Y.Q.; Feng, J.H.; Zhang, M.H. Effects of different acute high ambient temperatures on function of hepatic mitochondrial respiration, antioxidative enzymes, and oxidative injury in broiler chickens. Poult. Sci. 2010, 89, 115–122. [Google Scholar] [CrossRef]
- Huang, C.; Jiao, H.; Song, Z.; Zhao, J.; Wang, X.; Lin, H. Heat stress impairs mitochondria functions and induces oxidative injury in broiler chickens. J. Anim. Sci. 2015, 93, 2144–2153. [Google Scholar] [CrossRef]
- Ala, F.S.; Ahmad, H.; Golian, A. Effects of dietary supplemental methionine source and betaine replacement on the growth performance and activity of mitochondrial respiratory chain enzymes in normal and heatstressed broiler chickens. J. Anim. Physiol. Anim. Nutr. 2018, 103, 87–99. [Google Scholar]
- Kharbanda, K.K.; Todero, S.L.; King, A.L.; Osna, N.A.; McVicker, B.L.; Tuma, D.J.; Wisecarver, J.L.; Bailey, S.M. Betaine treatment attenuates chronic ethanol-induced hepatic steatosis and alterations to the mitochondrial respiratory chain proteome. Int. J. Hepatol. 2012, 2012, 962183. [Google Scholar] [CrossRef]
- Ganesan, B.; Rajesh, R.; Anandan, R.; Dhandapani, N. Biochemical studies on the protective effect of betaine on mitochondrial function in experimentally induced myocardial infarction in rats. J. Health Sci. 2007, 53, 671–681. [Google Scholar] [CrossRef]
- Hu, Y.; Sun, Q.; Liu, J.; Jia, Y.; Cai, D.; Idriss, A.A.; Omer, N.A.; Zhao, R. In ovo injection of betaine alleviates corticosterone-induced fatty liver in chickens through epigenetic modifications. Sci. Rep. 2017, 7, 40251. [Google Scholar] [CrossRef]
- Lee, I. Betaine is a positive regulator of mitochondrial respiration. Biochem. Biophys. Res. Commun. 2015, 456, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Apicella, J.M.; Lee, E.C.; Bailey, B.L.; Saenz, C.; Anderson, J.M.; Craig, S.A.; Kraemer, W.J.; Volek, J.S.; Maresh, C.M. Betaine supplementation enhances anabolic endocrine and akt signaling in response to acute bouts of exercise. Eur. J. Appl. Physiol. 2013, 113, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Song, H.P.; Chu, Z.G.; Zhang, D.X.; Dang, Y.M.; Zhang, Q. Pi3k-akt pathway protects cardiomyocytes against hypoxia-induced apoptosis by mitokatp-mediated mitochondrial translocation of pakt. Cell Physiol. Biochem. 2018, 49, 717–727. [Google Scholar] [CrossRef] [PubMed]
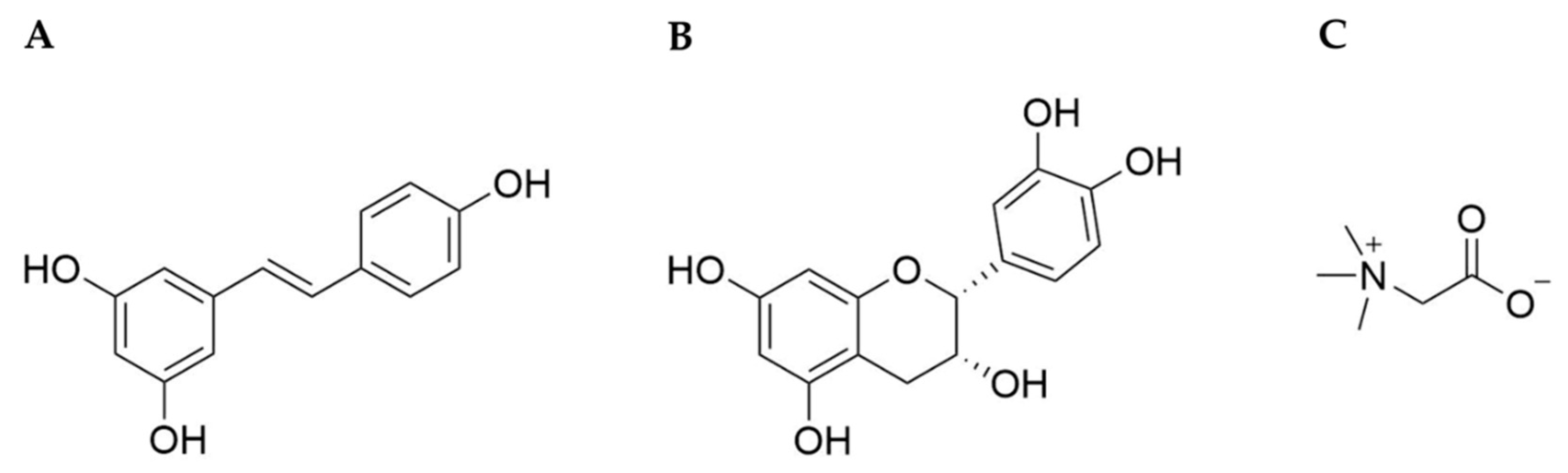
| Reference | Experimental Subject | Experimental Condition | Result |
|---|---|---|---|
| [57] | Gastrocnemius muscle from 8-week-old male C57BL/6J mice with high fat diet | Supplementation of 400 mg/kg/day RSV to high fat diet for 15 weeks | Increased mRNA levels of COX Va, COX IV-1, COX Vb, COX VIIaL |
| [62] | Cultured C2C12 mouse myoblast cells | Incubation with 25 μM RSV for 24 h | Increased mRNA level of COX Vb |
| [62] | Gastrocnemius muscle and hepatocytes from C57BL/6J mice with high fat diet; WT mouse for SIRT1-KO model | Supplementation of 25~30 mg/kg/day RSV with high fat diet for 8 months | Increased mRNA level of COX Vb |
| [67] | Hippocampus from male Sprague Dawley rats with status epilepticus induced by kainic acid | Microinjection of 100 μmol RSV into hippocampus prior to kainic acid treatment | Increased COX I amount |
| [68] | Hippocampus from aged female Sprague Dawley rats induced by bilateral ovariectomy combined with intraperitoneal injection of D-galactose | Intragastric administration of 80 mg/kg/day RSV alone and in combination with isoflavones for 12 weeks | Increased COX I amount by RSV alone and in combination with isoflavones |
| [69] | Hippocampus, cerebral cortex, and cerebellum from 60-day-old male Wistar rats | Incubation with 0.1, 0.5, 5 mM RSV for 1 h in combination with citrulline or ammonia in brain homogenates | Increased COX activity in a dose-dependent manner; increased COX activity in the presence of citrulline by 0.1, 0.5 mM RSV in cerebral cortex and 5 mM RSV in hippocampus & increased COX activity in the presence of ammonia by 0.1, 0.5 mM RSV in cerebral cortex and cerebellum |
| [70] | Cultured human lymphoblastic leukemia cells overexpressed with Bcl-2; CEM/Bcl-2 | Incubation with 10, 30, 50 μM RSV for 6 h | Decreased COX activity in a dose-dependent manner, while no change in COX I amount (50 μM RSV for 18 h) |
| [41] | Cultured human colon cancer cells; SW620 | Incubation with 10 μM RSV for 48 h | Increased COX activity (~45%) and COX I amount |
| Reference | Experimental Subject | Experimental Condition | Result |
|---|---|---|---|
| [144] | Cultured C2C12 mouse myoblast cells | Incubation with 10 μM EPI for 48 h | Increased COX I amount |
| [145] | Quadriceps femoris from detrained 5-month-old C57BL/6 male mice | Intragastric administration of 1 mg/kg of EPI twice a day during 14 days of detraining after 5 weeks of training | Increased COX activity (~108%) and COX II amount |
| [146] | Gastrocnemius muscle from hindlimb suspended 6-month-old male C57BL/6N mice | Intragastric administration of 1 mg/kg of EPI twice a day during 14 days of hindlimb suspension | Increased COX I amount |
| [147] | Plantaris muscle from 5-month-old male rats with congenital low running capacity | Intragastric administration of 1 mg/kg of EPI twice a day for 30 days | Increased COX II amount |
| [149] | Cultured rat β cells; INS-1 derived 832/13 cells | Incubation with 10 μM EPI for 24 h | Increased COX I amount |
| [150] | Cultured adipocytes excised from human subcutaneous adipose tissue | Incubation with 100 nM EPI for 72 h | Increased COX I amount |
| [129] | Cultured cow coronary artery endothelial cells with low passage number (young) and high passage number (aged) | Incubation with 1 μM EPI for 48 h | Increased COX I amount in both young and aged cells |
| [151] | Cultured Panc-1 pancreatic cancer cells | Incubation with 20, 50, 100, 200 μM EPI for 1 h | Increased COX activity in a dose-dependent manner (~59% by 200 μM EPI) |
| [147] | Plantaris muscle from healthy Sprague Dawley rats | Incubation with 20 μM EPI for 25 min | Increased COX activity |
| [148] | Quadriceps femoris muscle from 14-month-old male C57BL/6N mice | Incubation with 20 μM EPI for 25 min | Increased COX activity |
| Reference | Experimental Subject | Experimental Condition | Result |
|---|---|---|---|
| [209] | Livers from 45–48-day-old male Wister rats | Supplementation of 1% (w/v) BET to ethanol diet for 4–5 weeks | Increased amount of both COX holoenzyme and subunit I by BET in alcoholic liver, while no changes in controls |
| [210] | Hearts from Wistar male rats with myocardial infarction induced by isoprenaline | Intragastric administration of 250 mg/kg/day BET for 30 days prior to isoprenaline injection | Increased COX activity by BET in myocardial infarction, while no changes in controls |
| [211] | Livers from 8-week-old chickens injected with corticosterone | In ovo injection of 2.5 mg BET | Increased mRNA levels of COX I, II, and III and COX activity in corticosterone treated liver, while no changes in controls |
| [212] | Cultured H.2.35 mouse hepatocytes | Incubation of 0.5, 1, 2, 5, 10 mM BET for 30 min | Increased COX activity in a dose-dependent manner up to 2mM BET, while no change in activity of purified COX by incubation with EPI |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, I. Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (–)-Epicatechin, and Betaine. Cells 2021, 10, 1346. https://doi.org/10.3390/cells10061346
Lee I. Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (–)-Epicatechin, and Betaine. Cells. 2021; 10(6):1346. https://doi.org/10.3390/cells10061346
Chicago/Turabian StyleLee, Icksoo. 2021. "Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (–)-Epicatechin, and Betaine" Cells 10, no. 6: 1346. https://doi.org/10.3390/cells10061346
APA StyleLee, I. (2021). Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (–)-Epicatechin, and Betaine. Cells, 10(6), 1346. https://doi.org/10.3390/cells10061346





