Comparative Neurodevelopment Effects of Bisphenol A and Bisphenol F on Rat Fetal Neural Stem Cell Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Culture
Propagation of rNSCs
2.3. rNSC Monolayer Model for Directed-Differentiation to Generate Astrocytes, Neurons, and Oligodendrocytes
2.3.1. Coating Chamber Slides for 3-Types of Directed-Differentiation Processes
2.3.2. Differentiating Media
2.3.3. rNSC Culturing for Differentiation
2.4. Cell Proliferation or Cytotoxicity Assays
2.5. Effects of BPA or BPF on the Differentiation Rate of rNSCs into Astrocytes, Oligodendrocytes, and Neurons
2.6. Immunochemistry and Image Analysis
2.7. Morphometric Analysis of Neurons, Astrocyte and Oligodendrocyte Differentiated from rNSC in the Presence and Absence of BPA or BPF
2.8. Statistical Analysis
3. Results
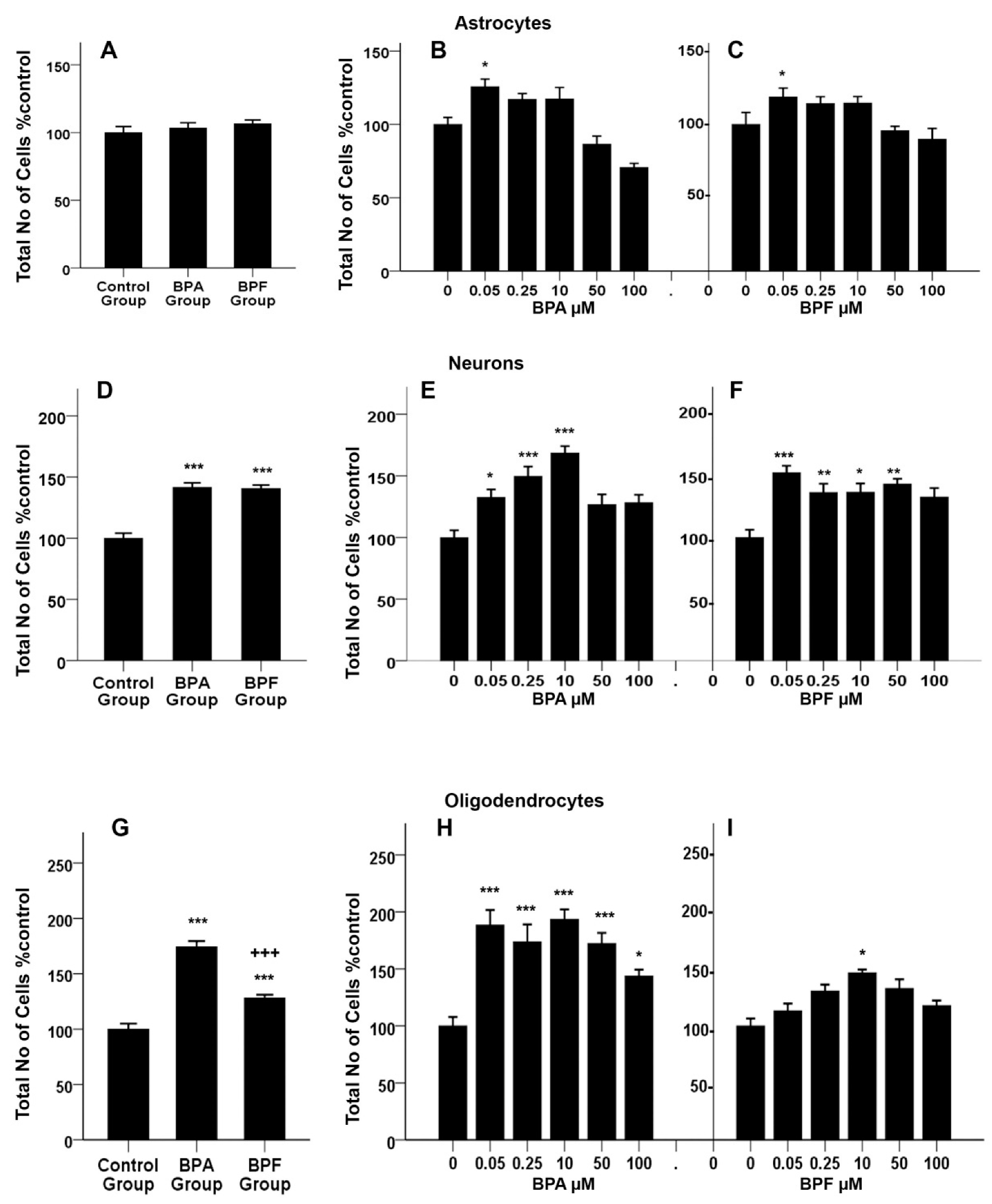
3.1. Effects of BPA and BPF on rNSC Differentiation Directed into Astrocytes
3.2. Effects of BPA or BPF on Stem Cell Properties of rNSCs Cultured in rNSC Growth Medium
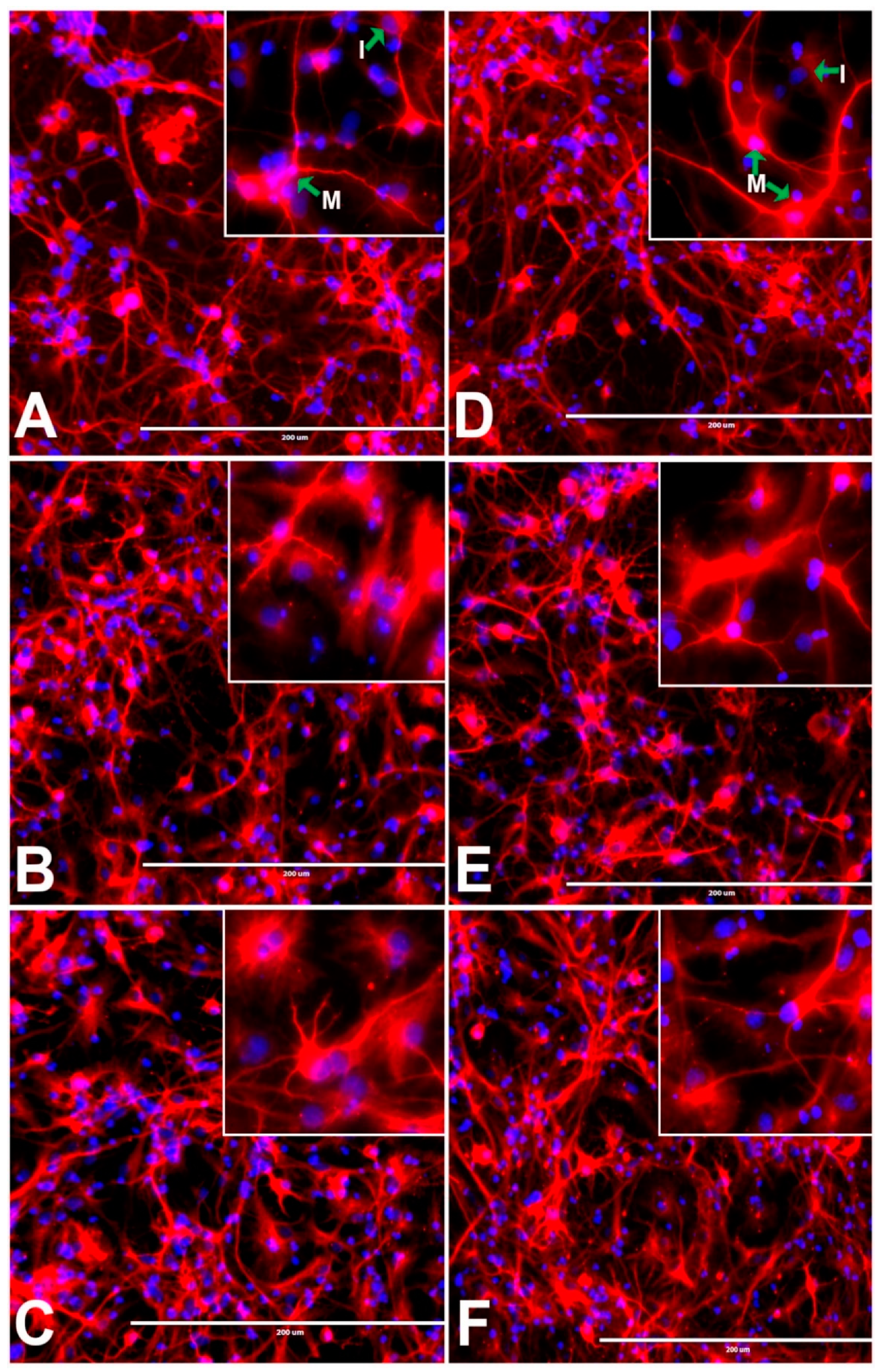
3.3. Effects of BPA or BPF on rNSC Differentiation Directed into Neurons
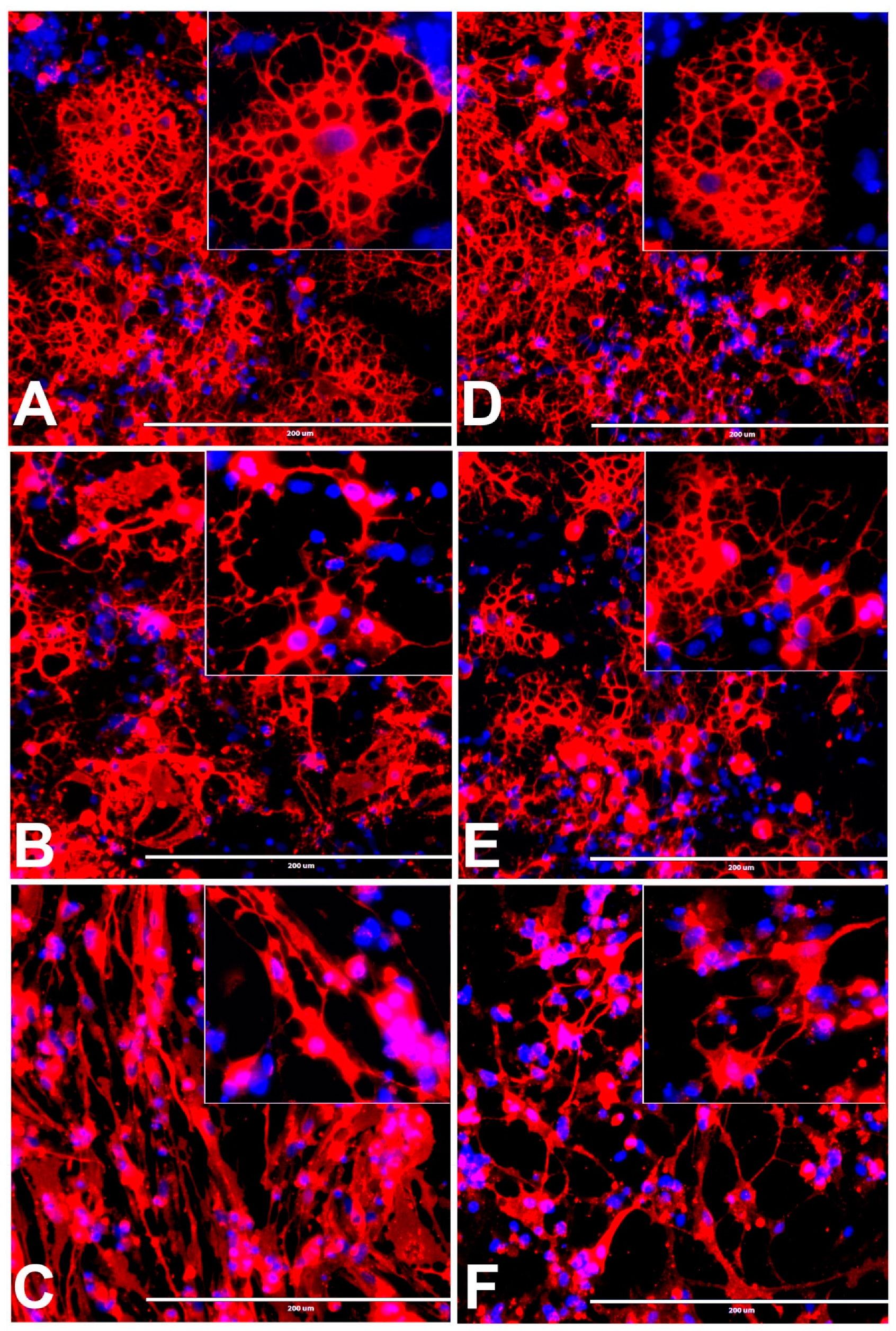
3.4. Effects of BPA or BPF on rNSC Differentiation Directed into Oligodendrocytes
3.5. Concentration-Response Effects of BPA and BPF on the Degree of % Differentiation of rNSCs Directed into Astrocytes
3.6. Concentration-Response Effects of BPA and BPF on the % Differentiation of rNSCs Directed into Neurons
3.7. Concentration-Response Effects of BPA and BPF on the % Differentiation of rNSCs into Oligodendrocytes
3.8. Effects of BPA or BPF on Cell Proliferation of Differentiating rNSCs into Astrocytes
3.9. Effects of BPA or BPF on Cell Proliferation of Differentiating rNSCs into Neurons
3.10. Effects of BPA or BPF on Cell-Proliferation of Differentiating rNSCs into Oligodendrocytes
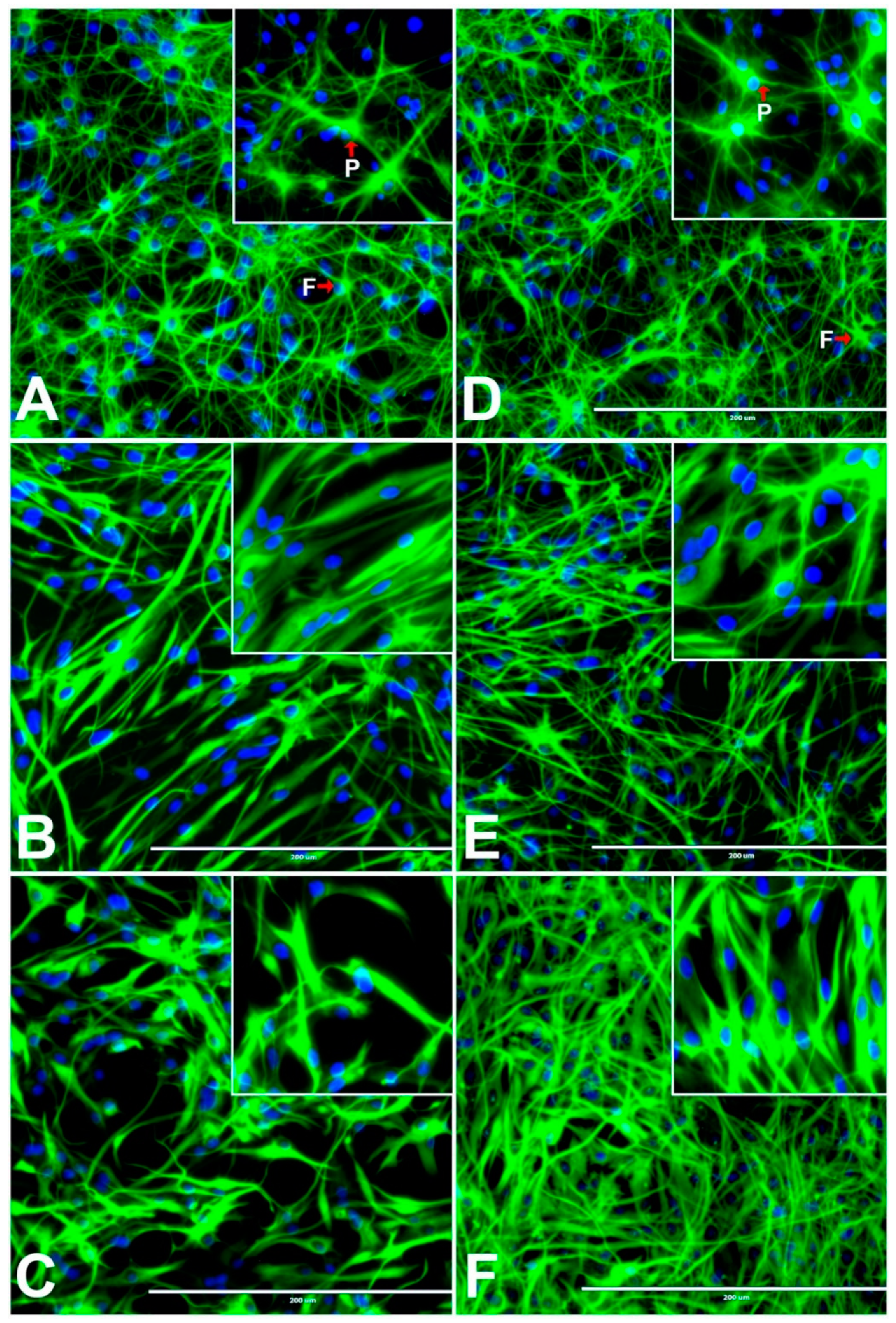
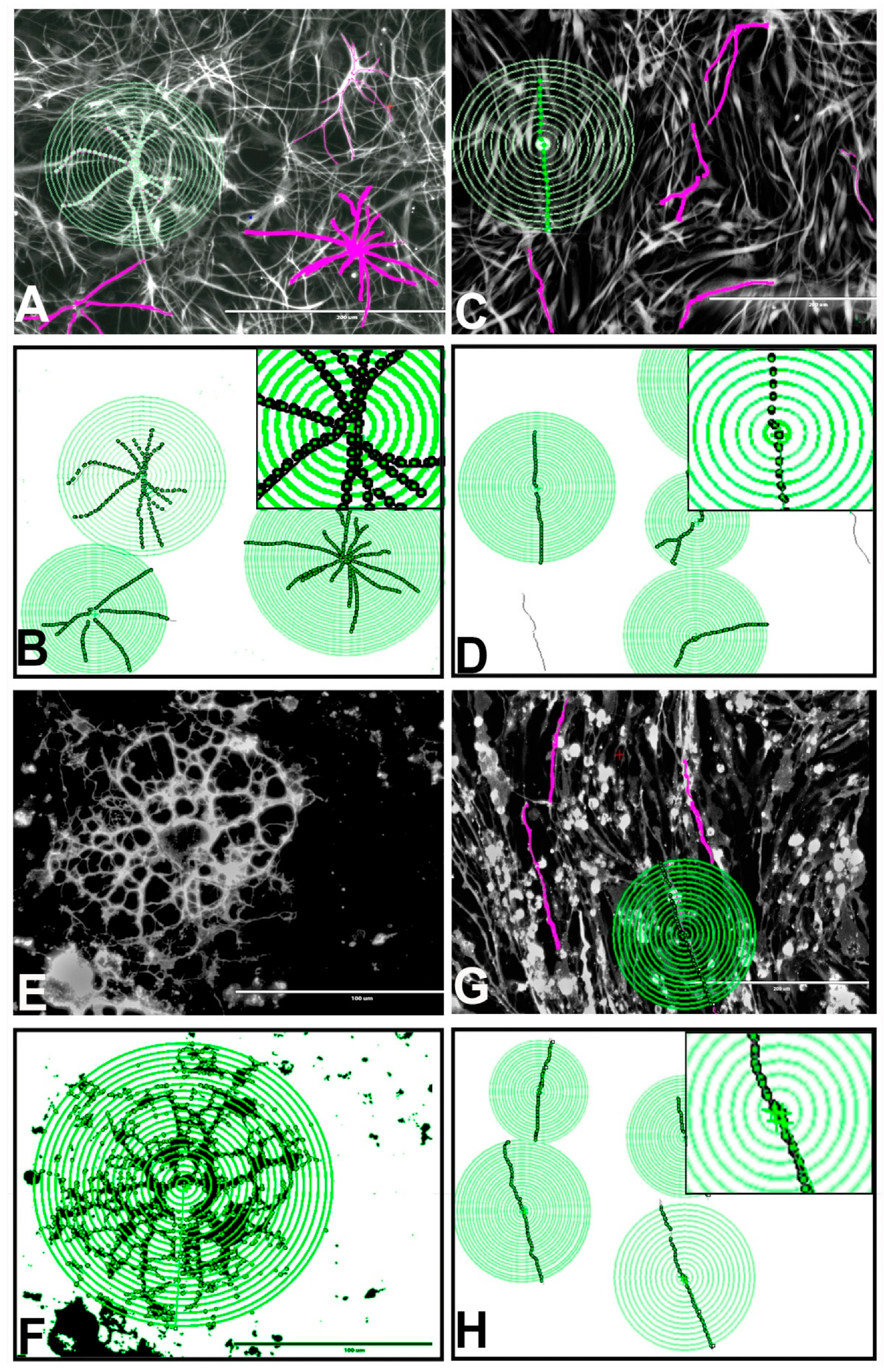
3.11. Effects of BPA or BPF on the Morphologies of Astrocytes, Oligodendrocytes, and Neurons Differentiated from rNSCs
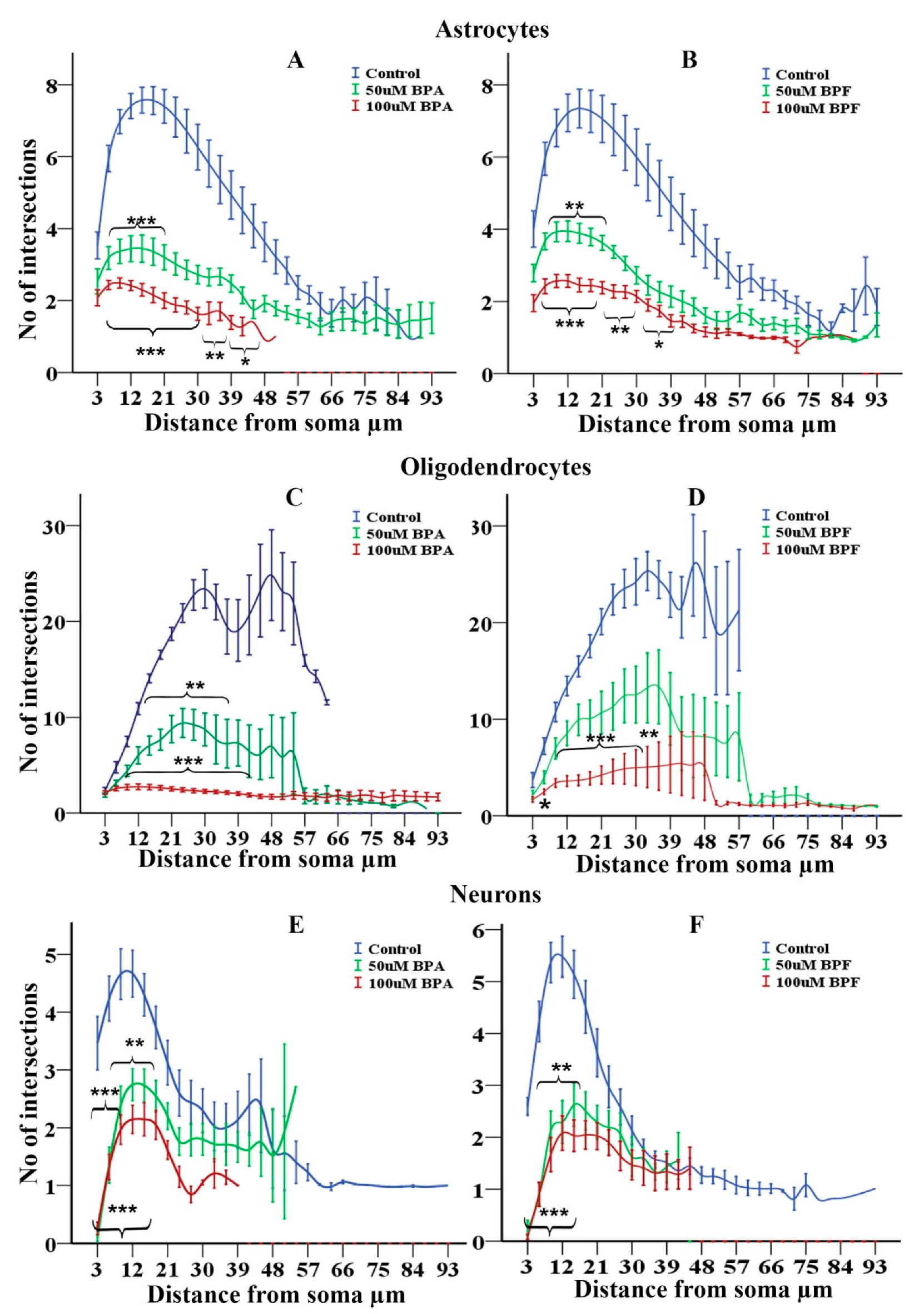
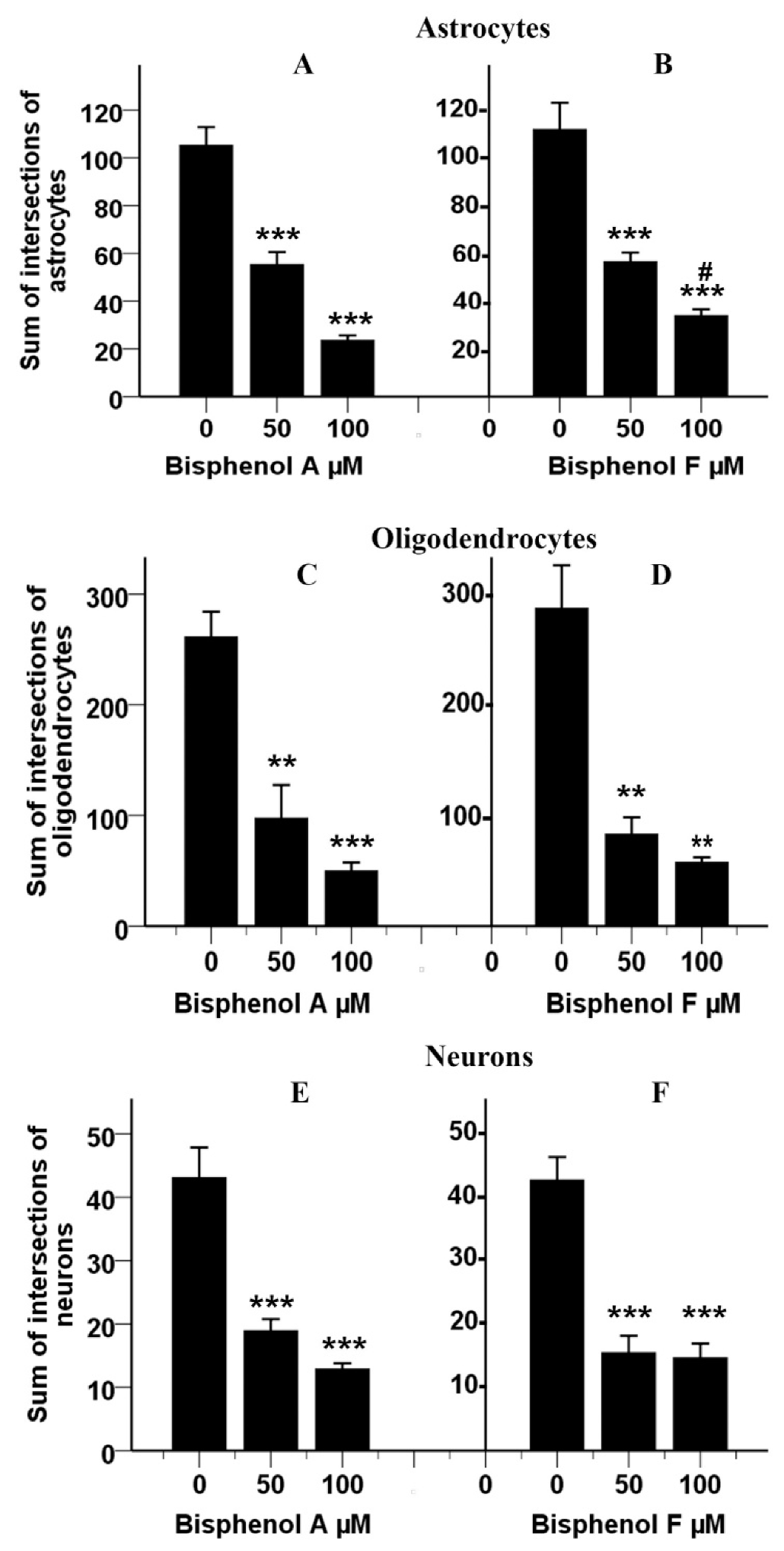
3.12. Effects of BPA and BPF on Sholl Analysis on Astrocytes
3.13. Effects of BPA and BPF on Sholl Analysis on Oligodendrocytes
3.14. Effects of BPA and BPF on Sholl Analysis on Neurons
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bergman, A.; Heindel, J.J.; Kasten, T.; Kidd, K.A.; Jobling, S.; Neira, M.; Zoeller, R.T.; Becher, G.; Bjerregaard, P.; Bornman, R.; et al. The Impact of Endocrine Disruption: A Consensus Statement on the State of the Science. Environ. Health Perspect. 2013, 121, A104–A106. [Google Scholar] [CrossRef]
- Dogan, S.; Simsek, T. Possible Relationship between Endocrine Disrupting Chemicals and Hormone Dependent Gynecologic Cancers. Med. Hypotheses 2016, 92, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Usman, A.; Ahmad, M. From BPA to Its Analogues: Is It a Safe Journey? Chemosphere 2016, 158, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Michałowicz, J. Bisphenol A—Sources, Toxicity and Biotransformation. Environ. Toxicol. Pharmacol. 2014, 37, 738–758. [Google Scholar] [CrossRef] [PubMed]
- Stogsdill, J.A.; Ramirez, J.; Liu, D.; Kim, Y.-H.; Baldwin, K.T.; Enustun, E.; Ejikeme, T.; Ji, R.-R.; Eroglu, C. Astrocytic Neuroligins Control Astrocyte Morphogenesis and Synaptogenesis. Nature 2017, 551, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Almeida, S.; Raposo, A.; Almeida-González, M.; Carrascosa, C. Bisphenol A: Food Exposure and Impact on Human Health. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1503–1517. [Google Scholar] [CrossRef]
- Lehmler, H.-J.; Liu, B.; Gadogbe, M.; Bao, W. Exposure to Bisphenol A, Bisphenol F, and Bisphenol S in U.S. Adults and Children: The National Health and Nutrition Examination Survey 2013–2014. ACS Omega 2018, 3, 6523–6532. [Google Scholar] [CrossRef]
- Geens, T.; Aerts, D.; Berthot, C.; Bourguignon, J.-P.; Goeyens, L.; Lecomte, P.; Maghuin-Rogister, G.; Pironnet, A.-M.; Pussemier, L.; Scippo, M.-L.; et al. A Review of Dietary and Non-Dietary Exposure to Bisphenol-A. Food Chem. Toxicol. 2012, 50, 3725–3740. [Google Scholar] [CrossRef]
- Ribeiro, E.; Ladeira, C.; Viegas, S. Occupational Exposure to Bisphenol A (BPA): A Reality That Still Needs to Be Unveiled. Toxics 2017, 5, 22. [Google Scholar] [CrossRef]
- Konieczna, A.; Rutkowska, A.; Rachoń, D. Health Risk of Exposure to Bisphenol A (BPA). Rocz. Panstw. Zakl. Hig. 2015, 66, 5–11. [Google Scholar]
- Vandenberg, L.N.; Chahoud, I.; Heindel, J.J.; Padmanabhan, V.; Paumgartten, F.J.R.; Schoenfelder, G. Urinary, Circulating, and Tissue Biomonitoring Studies Indicate Widespread Exposure to Bisphenol A. Environ. Health Perspect. 2010, 118, 1055–1070. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R. Bisphenol A and Human Health: A Review of the Literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Schug, T.T.; Birnbaum, L.S. Human Health Effects of Bisphenol A. In Toxicants in Food Packaging and Household Plastics: Exposure and Health Risks to Consumers; Snedeker, S.M., Ed.; Molecular and Integrative Toxicology; Springer: London, UK, 2014; pp. 1–29. ISBN 978-1-4471-6500-2. [Google Scholar]
- Cano-Nicolau, J.; Garoche, C.; Hinfray, N.; Pellegrini, E.; Boujrad, N.; Pakdel, F.; Kah, O.; Brion, F. Several Synthetic Progestins Disrupt the Glial Cell Specific-Brain Aromatase Expression in Developing Zebra Fish. Toxicol. Appl. Pharmacol. 2016, 305, 12–21. [Google Scholar] [CrossRef]
- Liu, B.; Lehmler, H.J.; Sun, Y.; Xu, G.; Sun, Q.; Snetselaar, L.G.; Wallace, R.B.; Bao, W. Association of Bisphenol A and Its Substitutes, Bisphenol F and Bisphenol S, with Obesity in United States Children and Adolescents. Diabetes Metab. J. 2019, 43, 59–75. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R.; Lee, D.-H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef] [PubMed]
- Arambula, S.E.; Fuchs, J.; Cao, J.; Patisaul, H.B. Effects of Perinatal Bisphenol A Exposure on the Volume of Sexually-Dimorphic Nuclei of Juvenile Rats: A CLARITY-BPA Consortium Study. Neurotoxicology 2017, 63, 33–42. [Google Scholar] [CrossRef]
- Maragou, N.C.; Makri, A.; Lampi, E.N.; Thomaidis, N.S.; Koupparis, M.A. Migration of Bisphenol A from Polycarbonate Baby Bottles under Real Use Conditions. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2008, 25, 373–383. [Google Scholar] [CrossRef] [PubMed]
- WHO; FAO. Joint FAO/WHO Expert Meeting to Review Toxicological and Health Aspects of Bisphenol A: Final Report, Including Report of Stakeholder Meeting on Bisphenol A, 1–5 November 2010, Ottawa, Canada; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Siddque, S.; Zhang, G.; Coleman, K.; Kubwabo, C. Migration of Bisphenol A Analogues in Baby Bottles and Sippy Cups. In Proceedings of the 2020 Health Canada Science Forum, Ottawa, ON, Canada, 20–21 January 2020; p. 142. [Google Scholar]
- Andújar, N.; Gálvez-Ontiveros, Y.; Zafra-Gómez, A.; Rodrigo, L.; Álvarez-Cubero, M.J.; Aguilera, M.; Monteagudo, C.; Rivas, A.A. Bisphenol A Analogues in Food and Their Hormonal and Obesogenic Effects: A Review. Nutrients 2019, 11, 2136. [Google Scholar] [CrossRef] [PubMed]
- Cunha, S.C.; Fernandes, J.O. Quantification of Free and Total Bisphenol A and Bisphenol B in Human Urine by Dispersive Liquid-Liquid Microextraction (DLLME) and Heart-Cutting Multidimensional Gas Chromatography-Mass Spectrometry (MD-GC/MS). Talanta 2010, 83, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Molina-Molina, J.-M.; Amaya, E.; Grimaldi, M.; Sáenz, J.-M.; Real, M.; Fernández, M.F.; Balaguer, P.; Olea, N. In Vitro Study on the Agonistic and Antagonistic Activities of Bisphenol-S and Other Bisphenol-A Congeners and Derivatives via Nuclear Receptors. Toxicol. Appl. Pharmacol. 2013, 272, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Kannan, K. A Survey of Bisphenol A and Other Bisphenol Analogues in Foodstuffs from Nine Cities in China. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Gallart-Ayala, H.; Moyano, E.; Galceran, M.T. Analysis of Bisphenols in Soft Drinks by On-Line Solid Phase Extraction Fast Liquid Chromatography-Tandem Mass Spectrometry. Anal. Chim. Acta 2011, 683, 227–233. [Google Scholar] [CrossRef]
- Gallart-Ayala, H.; Moyano, E.; Galceran, M.T. Fast Liquid Chromatography-Tandem Mass Spectrometry for the Analysis of Bisphenol A-Diglycidyl Ether, Bisphenol F-Diglycidyl Ether and Their Derivatives in Canned Food and Beverages. J. Chromatogr. A 2011, 1218, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Fromme, H.; Küchler, T.; Otto, T.; Pilz, K.; Müller, J.; Wenzel, A. Occurrence of Phthalates and Bisphenol A and F in the Environment. Water Res. 2002, 36, 1429–1438. [Google Scholar] [CrossRef]
- Wong, Y.M.; Li, R.; Lee, C.K.F.; Wan, H.T.; Wong, C.K.C. The Measurement of Bisphenol A and Its Analogues, Perfluorinated Compounds in Twenty Species of Freshwater and Marine Fishes, a Time-Trend Comparison and Human Health Based Assessment. Mar. Pollut. Bull. 2017, 124, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Charisiadis, P.; Andrianou, X.D.; van der Meer, T.P.; den Dunnen, W.F.A.; Swaab, D.F.; Wolffenbuttel, B.H.R.; Makris, K.C.; van Vliet-Ostaptchouk, J.V. Possible Obesogenic Effects of Bisphenols Accumulation in the Human Brain. Sci. Rep. 2018, 8, 8186. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.-P.; Liu, Z.-H.; Yin, H.; Dang, Z.; Wu, P.-X.; Zhu, N.-W.; Lin, Z. Bisphenol A Concentrations in Human Urine, Human Intakes across Six Continents, and Annual Trends of Average Intakes in Adult and Child Populations Worldwide: A Thorough Literature Review. Sci. Total Environ. 2018, 626, 971–981. [Google Scholar] [CrossRef]
- Fan, X.; Kubwabo, C.; Katuri, G.P.; Caza, A.; Rasmussen, P.E. Analysis of Bisphenol A Analogues in Canadian Indoor House Dust by Gas Chromatography-Tandem Mass Spectrometry. In Proceedings of the 2020 Health Canada Science Forum, Ottawa, ON, Canada, 20–21 January 2020; p. 98. [Google Scholar]
- Liu, J.; Li, J.; Wu, Y.; Zhao, Y.; Luo, F.; Li, S.; Yang, L.; Moez, E.K.; Dinu, I.; Martin, J.W. Bisphenol A Metabolites and Bisphenol S in Paired Maternal and Cord Serum. Environ. Sci. Technol. 2017, 51, 2456–2463. [Google Scholar] [CrossRef]
- Asimakopoulos, A.G.; Xue, J.; DeCarvalho, B.P.; Iyer, A.; Abualnaja, K.O.; Yaghmoor, S.S.; Kumosani, T.A.; Kannan, K. Corrigendum to “Urinary Biomarkers of Exposure to 57 Xenobiotics and Its Association with Oxidative Stress in a Population in Jeddah, Saudi Arabia”. Environ. Res. 2017, 153, 205. [Google Scholar] [CrossRef]
- Lu, S.-Y.; Li, Y.-X.; Zhang, J.-Q.; Zhang, T.; Liu, G.-H.; Huang, M.-Z.; Li, X.; Ruan, J.-J.; Kannan, K.; Qiu, R.-L. Associations between Polycyclic Aromatic Hydrocarbon (PAH) Exposure and Oxidative Stress in People Living near e-Waste Recycling Facilities in China. Environ. Int. 2016, 94, 161–169. [Google Scholar] [CrossRef]
- Heffernan, A.L.; Thompson, K.; Eaglesham, G.; Vijayasarathy, S.; Mueller, J.F.; Sly, P.D.; Gomez, M.J. Rapid, Automated Online SPE-LC-QTRAP-MS/MS Method for the Simultaneous Analysis of 14 Phthalate Metabolites and 5 Bisphenol Analogues in Human Urine. Talanta 2016, 151, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Liu, F.; Alomirah, H.; Loi, V.D.; Mohd, M.A.; Moon, H.-B.; Nakata, H.; Kannan, K. Bisphenol S in Urine from the United States and Seven Asian Countries: Occurrence and Human Exposures. Environ. Sci. Technol. 2012, 46, 6860–6866. [Google Scholar] [CrossRef]
- Eladak, S.; Grisin, T.; Moison, D.; Guerquin, M.-J.; N’Tumba-Byn, T.; Pozzi-Gaudin, S.; Benachi, A.; Livera, G.; Rouiller-Fabre, V.; Habert, R. A New Chapter in the Bisphenol A Story: Bisphenol S and Bisphenol F Are Not Safe Alternatives to This Compound. Fertil. Steril. 2015, 103, 11–21. [Google Scholar] [CrossRef]
- Maćczak, A.; Cyrkler, M.; Bukowska, B.; Michałowicz, J. Bisphenol A, Bisphenol S, Bisphenol F and Bisphenol AF Induce Different Oxidative Stress and Damage in Human Red Blood Cells (in Vitro Study). Toxicology 2017, 41, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Kannan, K.; Tan, H.; Zheng, Z.; Feng, Y.-L.; Wu, Y.; Widelka, M. Bisphenol Analogues Other Than BPA: Environmental Occurrence, Human Exposure, and Toxicity-A Review. Environ. Sci. Technol. 2016, 50, 5438–5453. [Google Scholar] [CrossRef] [PubMed]
- Cabaton, N.; Zalko, D.; Rathahao, E.; Canlet, C.; Delous, G.; Chagnon, M.-C.; Cravedi, J.-P.; Perdu, E. Biotransformation of Bisphenol F by Human and Rat Liver Subcellular Fractions. Toxicology 2008, 22, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef]
- Andra, S.S.; Charisiadis, P.; Arora, M.; van Vliet-Ostaptchouk, J.V.; Makris, K.C. Biomonitoring of Human Exposures to Chlorinated Derivatives and Structural Analogs of Bisphenol A. Environ. Int. 2015, 85, 352–379. [Google Scholar] [CrossRef]
- Usman, A.; Ahmad, M. Binding of Bisphenol-F, a Bisphenol Analogue, to Calf Thymus DNA by Multi-Spectroscopic and Molecular Docking Studies. Chemosphere 2017, 181, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Moreman, J.; Lee, O.; Trznadel, M.; David, A.; Kudoh, T.; Tyler, C.R. Acute Toxicity, Teratogenic, and Estrogenic Effects of Bisphenol A and Its Alternative Replacements Bisphenol S, Bisphenol F, and Bisphenol AF in Zebrafish Embryo-Larvae. Environ. Sci. Technol. 2017, 51, 12796–12805. [Google Scholar] [CrossRef] [PubMed]
- Pelch, K.; Wignall, J.A.; Goldstone, A.E.; Ross, P.K.; Blain, R.B.; Shapiro, A.J.; Holmgren, S.D.; Hsieh, J.-H.; Svoboda, D.; Auerbach, S.S.; et al. A Scoping Review of the Health and Toxicological Activity of Bisphenol A (BPA) Structural Analogues and Functional Alternatives. Toxicology 2019, 424, 152235. [Google Scholar] [CrossRef]
- Gill, S.; Kumara, V.M.R. Detecting Neurodevelopmental Toxicity of Domoic Acid and Ochratoxin A Using Rat Fetal Neural Stem Cells. Mar. Drugs 2019, 17, 566. [Google Scholar] [CrossRef]
- Sholl, D.A. Dendritic Organization in the Neurons of the Visual and Motor Cortices of the Cat. J. Anat. 1953, 87, 387–406. [Google Scholar]
- Gensel, J.C.; Schonberg, D.L.; Alexander, J.K.; McTigue, D.M.; Popovich, P.G. Semi-Automated Sholl Analysis for Quantifying Changes in Growth and Differentiation of Neurons and Glia. J. Neurosci. Methods 2010, 190, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Langhammer, C.G.; Previtera, M.L.; Sweet, E.S.; Sran, S.S.; Chen, M.; Firestein, B.L. Automated Sholl Analysis of Digitized Neuronal Morphology at Multiple Scales: Whole Cell Sholl Analysis versus Sholl Analysis of Arbor Subregions. Cytom. A 2010, 77, 1160–1168. [Google Scholar] [CrossRef]
- Binley, K.E.; Ng, W.S.; Tribble, J.R.; Song, B.; Morgan, J.E. Sholl Analysis: A Quantitative Comparison of Semi-Automated Methods. J. Neurosci. Methods 2014, 225, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Bird, A.D.; Cuntz, H. Dissecting Sholl Analysis into Its Functional Components. Cell Rep. 2019, 27, 3081–3096.e5. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, A.; Srinivasan, A.; Ferland, R.J. AutoSholl Allows for Automation of Sholl Analysis Independent of User Tracing. J. Neurosci. Methods 2020, 331, 108529. [Google Scholar] [CrossRef] [PubMed]
- Mehder, R.H.; Bennett, B.M.; Andrew, R.D. Morphometric Analysis of Hippocampal and Neocortical Pyramidal Neurons in a Mouse Model of Late Onset Alzheimer’s Disease. J. Alzheimers Dis. 2020, 74, 1069–1083. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Won, S.-Y.; Yoon, B.-J. CRMP2 Mediates GSK3β Actions in the Striatum on Regulating Neuronal Structure and Mania-like Behavior. J. Affect. Disord. 2019, 245, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Gobran, J.; Rawlyk, B.; Henderson, D.C.; Hooper, M.L.; Farrell, S.R.M.; Chauhan, B.C. Effects of 3D Stratification of Retinal Ganglion Cells in Sholl Analysis. J. Neurosci. Methods 2020, 346, 108907. [Google Scholar] [CrossRef] [PubMed]
- López-Cabrera, J.D.; Hernández-Pérez, L.A.; Orozco-Morales, R.; Lorenzo-Ginori, J.V. New Morphological Features Based on the Sholl Analysis for Automatic Classification of Traced Neurons. J. Neurosci. Methods 2020, 343, 108835. [Google Scholar] [CrossRef]
- Reeves, A.M.B.; Shigetomi, E.; Khakh, B.S. Bulk Loading of Calcium Indicator Dyes to Study Astrocyte Physiology: Key Limitations and Improvements Using Morphological Maps. J. Neurosci. 2011, 31, 9353–9358. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, M.; Chauhan, G.; Roy, K.; Sahu, S.; Deep, S.; Jain, V.; Kishore, K.; Ray, K.; Thakur, L.; Panjwani, U. Caffeine and Modafinil Ameliorate the Neuroinflammation and Anxious Behavior in Rats during Sleep Deprivation by Inhibiting the Microglia Activation. Front. Cell Neurosci. 2018, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Diene, L.D.; Costa-Ferro, Z.S.M.; Barbosa, S.; Milanesi, B.B.; Lazzari, G.Z.; Neves, L.T.; Paz, L.V.; Neves, P.F.R.; Battisti, V.; Martins, L.A.; et al. Selective Brain Neuronal and Glial Losses without Changes in GFAP Immunoreactivity: Young versus Mature Adult Wistar Rats. Mech Ageing Dev. 2019, 182, 111128. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Lohr, C.; Droste, D. Age-Dependent Heterogeneity of Murine Olfactory Bulb Astrocytes. Front. Aging Neurosci. 2020, 12, 172. [Google Scholar] [CrossRef]
- Gómez, G.I.; Falcon, R.V.; Maturana, C.J.; Labra, V.C.; Salgado, N.; Rojas, C.A.; Oyarzun, J.E.; Cerpa, W.; Quintanilla, R.A.; Orellana, J.A. Heavy Alcohol Exposure Activates Astroglial Hemichannels and Pannexons in the Hippocampus of Adolescent Rats: Effects on Neuroinflammation and Astrocyte Arborization. Front. Cell Neurosci. 2018, 12, 472. [Google Scholar] [CrossRef]
- Miguel-Hidalgo, J.J.; Carter, K.; Deloach, P.H.; Sanders, L.; Pang, Y. Glucocorticoid-Induced Reductions of Myelination and Connexin 43 in Mixed Central Nervous System Cell Cultures Are Prevented by Mifepristone. Neuroscience 2019, 411, 255–269. [Google Scholar] [CrossRef]
- Murtie, J.C.; Macklin, W.B.; Corfas, G. Morphometric Analysis of Oligodendrocytes in the Adult Mouse Frontal Cortex. J. Neurosci. Res. 2007, 85, 2080–2086. [Google Scholar] [CrossRef]
- Flygt, J.; Ruscher, K.; Norberg, A.; Mir, A.; Gram, H.; Clausen, F.; Marklund, N. Neutralization of Interleukin-1β Following Diffuse Traumatic Brain Injury in the Mouse Attenuates the Loss of Mature Oligodendrocytes. J. Neurotrauma 2018, 35, 2837–2849. [Google Scholar] [CrossRef]
- Janowska, J.; Gargas, J.; Ziemka-Nalecz, M.; Zalewska, T.; Sypecka, J. Oligodendrocyte Response to Pathophysiological Conditions Triggered by Episode of Perinatal Hypoxia-Ischemia: Role of IGF-1 Secretion by Glial Cells. Mol. Neurobiol. 2020, 57, 4250–4268. [Google Scholar] [CrossRef] [PubMed]
- Chklovskii, D.B. Synaptic Connectivity and Neuronal Morphology: Two Sides of the Same Coin. Neuron 2004, 43, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Stepanyants, A.; Tamás, G.; Chklovskii, D.B. Class-Specific Features of Neuronal Wiring. Neuron 2004, 43, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.A.; Blackman, A.V.; Oyrer, J.; Jayabal, S.; Chung, A.J.; Watt, A.J.; Sjöström, P.J.; van Meyel, D.J. Neuronal Morphometry Directly from Bitmap Images. Nat. Methods 2014, 11, 982–984. [Google Scholar] [CrossRef]
- Brewer, G.J.; Cotman, C.W. Survival and Growth of Hippocampal Neurons in Defined Medium at Low Density: Advantages of a Sandwich Culture Technique or Low Oxygen. Brain Res. 1989, 494, 65–74. [Google Scholar] [CrossRef]
- Chen, Y.; Stevens, B.; Chang, J.; Milbrandt, J.; Barres, B.A.; Hell, J.W. NS21: Re-Defined and Modified Supplement B27 for Neuronal Cultures. J. Neurosci. Methods 2008, 171, 239–247. [Google Scholar] [CrossRef]
- Shi, J.; Springer, S.; Escobar, P. Coupling Cytotoxicity Biomarkers with DNA Damage Assessment in TK6 Human Lymphoblast Cells. Mutat. Res. 2010, 696, 167–178. [Google Scholar] [CrossRef]
- O’Donovan, M. A Critique of Methods to Measure Cytotoxicity in Mammalian Cell Genotoxicity Assays. Mutagenesis 2012, 27, 615–621. [Google Scholar] [CrossRef][Green Version]
- Shaltouki, A.; Peng, J.; Liu, Q.; Rao, M.S.; Zeng, X. Efficient Generation of Astrocytes from Human Pluripotent Stem Cells in Defined Conditions. Stem. Cells 2013, 31, 941–952. [Google Scholar] [CrossRef]
- Sadowski, R.N.; Wise, L.M.; Park, P.Y.; Schantz, S.L.; Juraska, J.M. Early Exposure to Bisphenol A Alters Neuron and Glia Number in the Rat Prefrontal Cortex of Adult Males, but Not Females. Neuroscience 2014, 279, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.M.; Sadowski, R.N.; Kim, T.; Willing, J.; Juraska, J.M. Long-Term Effects of Adolescent Exposure to Bisphenol A on Neuron and Glia Number in the Rat Prefrontal Cortex: Differences between the Sexes and Cell Type. Neurotoxicology 2016, 53, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Miyatake, M.; Miyagawa, K.; Mizuo, K.; Narita, M.; Suzuki, T. Dynamic Changes in Dopaminergic Neurotransmission Induced by a Low Concentration of Bisphenol-A in Neurones and Astrocytes. J. Neuroendocrinol. 2006, 18, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Janowska, J.; Ziemka-Nalecz, M.; Sypecka, J. The Differentiation of Rat Oligodendroglial Cells Is Highly Influenced by the Oxygen Tension: In Vitro Model Mimicking Physiologically Normoxic Conditions. Int. J. Mol. Sci. 2018, 19, 331. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Suk, K.; Kim, I.K.; Jang, I.-S.; Park, J.-W.; Johnson, V.J.; Kwon, T.K.; Choi, B.-J.; Kim, S.-H. Signaling Pathways of Bisphenol A-Induced Apoptosis in Hippocampal Neuronal Cells: Role of Calcium-Induced Reactive Oxygen Species, Mitogen-Activated Protein Kinases, and Nuclear Factor-KappaB. J. Neurosci. Res. 2008, 86, 2932–2942. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Agarwal, S.; Chauhan, L.K.S.; Mishra, V.N.; Chaturvedi, R.K. Bisphenol-A Impairs Myelination Potential during Development in the Hippocampus of the Rat Brain. Mol. Neurobiol. 2015, 51, 1395–1416. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.K.; Agarwal, S.; Seth, B.; Yadav, A.; Ray, R.S.; Mishra, V.N.; Chaturvedi, R.K. Inhibitory Effects of Bisphenol-A on Neural Stem Cells Proliferation and Differentiation in the Rat Brain Are Dependent on Wnt/β-Catenin Pathway. Mol. Neurobiol. 2015, 52, 1735–1757. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Agarwal, S.; Tripathi, A.; Chaturvedi, R.K. Bisphenol-A Mediated Inhibition of Hippocampal Neurogenesis Attenuated by Curcumin via Canonical Wnt Pathway. Mol. Neurobiol. 2016, 53, 3010–3029. [Google Scholar] [CrossRef]
- Verbanck, M.; Canouil, M.; Leloire, A.; Dhennin, V.; Coumoul, X.; Yengo, L.; Froguel, P.; Poulain-Godefroy, O. Low-Dose Exposure to Bisphenols A, F and S of Human Primary Adipocyte Impacts Coding and Non-Coding RNA Profiles. PLoS ONE 2017, 12, e0179583. [Google Scholar] [CrossRef]
- Cano-Nicolau, J.; Vaillant, C.; Pellegrini, E.; Charlier, T.D.; Kah, O.; Coumailleau, P. Estrogenic Effects of Several BPA Analogs in the Developing Zebrafish Brain. Front. Neurosci. 2016, 10, 112. [Google Scholar] [CrossRef]
- Pellegrini, E.; Mouriec, K.; Anglade, I.; Menuet, A.; Le Page, Y.; Gueguen, M.-M.; Marmignon, M.-H.; Brion, F.; Pakdel, F.; Kah, O. Identification of Aromatase-Positive Radial Glial Cells as Progenitor Cells in the Ventricular Layer of the Forebrain in Zebrafish. J. Comp. Neurol. 2007, 501, 150–167. [Google Scholar] [CrossRef]
- Coumailleau, P.; Kah, O. Expression of the Cyp19a1 Gene in the Adult Brain of Xenopus Is Neuronal and Not Sexually Dimorphic. Gen. Comp. Endocrinol. 2015, 221, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Diotel, N.; Vaillant, C.; Kah, O.; Pellegrini, E. Mapping of Brain Lipid Binding Protein (Blbp) in the Brain of Adult Zebrafish, Co-Expression with Aromatase B and Links with Proliferation. Gene Expr. Patterns 2016, 20, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Castro, B.; Sánchez, P.; Torres, J.M.; Preda, O.; del Moral, R.G.; Ortega, E. Bisphenol A Exposure during Adulthood Alters Expression of Aromatase and 5α-Reductase Isozymes in Rat Prostate. PLoS ONE 2013, 8, e55905. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Tian, X.; Fang, X.; Ji, F. Waterborne Exposure to Bisphenol F Causes Thyroid Endocrine Disruption in Zebrafish Larvae. Chemosphere 2016, 147, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Castro, B.; Sánchez, P.; Torres, J.M.; Ortega, E. Bisphenol A, Bisphenol F and Bisphenol S Affect Differently 5α-Reductase Expression and Dopamine-Serotonin Systems in the Prefrontal Cortex of Juvenile Female Rats. Environ. Res. 2015, 142, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Le Fol, V.; Aït-Aïssa, S.; Sonavane, M.; Porcher, J.-M.; Balaguer, P.; Cravedi, J.-P.; Zalko, D.; Brion, F. In Vitro and in Vivo Estrogenic Activity of BPA, BPF and BPS in Zebrafish-Specific Assays. Ecotoxicol. Environ. Saf. 2017, 142, 150–156. [Google Scholar] [CrossRef]
- Ohtani, N.; Iwano, H.; Suda, K.; Tsuji, E.; Tanemura, K.; Inoue, H.; Yokota, H. Adverse Effects of Maternal Exposure to Bisphenol F on the Anxiety- and Depression-like Behavior of Offspring. J. Vet. Med. Sci. 2017, 79, 432–439. [Google Scholar] [CrossRef]
- Molina-Molina, J.M.; Jiménez-Díaz, I.; Fernández, M.F.; Rodriguez-Carrillo, A.; Peinado, F.M.; Mustieles, V.; Barouki, R.; Piccoli, C.; Olea, N.; Freire, C. Determination of Bisphenol A and Bisphenol S Concentrations and Assessment of Estrogen- and Anti-Androgen-like Activities in Thermal Paper Receipts from Brazil, France, and Spain. Environ. Res. 2019, 170, 406–415. [Google Scholar] [CrossRef]
- Wang, H.; Chang, L.; Aguilar, J.S.; Dong, S.; Hong, Y. Bisphenol-A Exposure Induced Neurotoxicity in Glutamatergic Neurons Derived from Human Embryonic Stem Cells. Environ. Int. 2019, 127, 324–332. [Google Scholar] [CrossRef]
- vom Saal, F.S.; Welshons, W.V. Evidence That Bisphenol A (BPA) Can Be Accurately Measured without Contamination in Human Serum and Urine, and That BPA Causes Numerous Hazards from Multiple Routes of Exposure. Mol. Cell. Endocrinol. 2014, 398, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Miao, M.; Zhou, Z.; Gao, E.; Chen, J.; Wang, J.; Sun, F.; Yuan, W.; Li, D.-K. Exposure to Bisphenol-A and Reproductive Hormones among Male Adults. Environ. Toxicol. Pharmacol. 2015, 39, 934–941. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Miao, M.; Wu, C.; Yuan, W.; Gao, E.; Zhou, Z.; Li, D.-K. Occupational Exposure Levels of Bisphenol A among Chinese Workers. J. Occup. Health 2009, 51, 432–436. [Google Scholar] [CrossRef]
- Xiao, G.; Wang, R.; Cai, Y.; He, G.; Zhou, Z. Effect of bisphenol A on semen quality of exposed workers: A pilot study. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi 2009, 27, 741–743. [Google Scholar] [PubMed]
- Padmanabhan, V.; Siefert, K.; Ransom, S.; Johnson, T.; Pinkerton, J.; Anderson, L.; Tao, L.; Kannan, K. Maternal Bisphenol-A Levels at Delivery: A Looming Problem? J. Perinatol. 2008, 28, 258–263. [Google Scholar] [CrossRef]
- Schönfelder, G.; Wittfoht, W.; Hopp, H.; Talsness, C.E.; Paul, M.; Chahoud, I. Parent Bisphenol A Accumulation in the Human Maternal-Fetal-Placental Unit. Environ. Health Perspect. 2002, 110, A703–A707. [Google Scholar] [CrossRef] [PubMed]
- Ikezuki, Y.; Tsutsumi, O.; Takai, Y.; Kamei, Y.; Taketani, Y. Determination of Bisphenol A Concentrations in Human Biological Fluids Reveals Significant Early Prenatal Exposure. Hum. Reprod. 2002, 17, 2839–2841. [Google Scholar] [CrossRef]
- Grohs, M.N.; Reynolds, J.E.; Liu, J.; Martin, J.W.; Pollock, T.; Lebel, C.; Dewey, D. APrON Study Team Prenatal Maternal and Childhood Bisphenol a Exposure and Brain Structure and Behavior of Young Children. Environ. Health 2019, 18, 85. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Wong, E.W.; Lie, P.P.; Li, M.W.; Mruk, D.D.; Yan, H.H.; Mok, K.-W.; Mannu, J.; Mathur, P.P.; Lui, W.-Y.; et al. Regulation of Blood-Testis Barrier Dynamics by Desmosome, Gap Junction, Hemidesmosome and Polarity Proteins: An Unexpected Turn of Events. Spermatogenesis 2011, 1, 105–115. [Google Scholar] [CrossRef]
- Nishikawa, M.; Iwano, H.; Yanagisawa, R.; Koike, N.; Inoue, H.; Yokota, H. Placental Transfer of Conjugated Bisphenol A and Subsequent Reactivation in the Rat Fetus. Environ. Health Perspect. 2010, 118, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Negri-Cesi, P. Bisphenol A Interaction With Brain Development and Functions. Dose Response 2015, 13. [Google Scholar] [CrossRef]
- Li, H.; Ding, C.; Ding, Z.-L.; Ling, M.; Wang, T.; Wang, W.; Huang, B. 17β-Oestradiol Promotes Differentiation of Human Embryonic Stem Cells into Dopamine Neurons via Cross-Talk between Insulin-like Growth Factors-1 and Oestrogen Receptor β. J. Cell. Mol. Med. 2017, 21, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Miyazaki, W.; Koibuchi, N.; Katoh, T. The Effects of Low-Dose Bisphenol A and Bisphenol F on Neural Differentiation of a Fetal Brain-Derived Neural Progenitor Cell Line. Front. Endocrinol. 2018, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Varshney, M.K.; Inzunza, J.; Lupu, D.; Ganapathy, V.; Antonson, P.; Rüegg, J.; Nalvarte, I.; Gustafsson, J.-Å. Role of Estrogen Receptor Beta in Neural Differentiation of Mouse Embryonic Stem Cells. Proc. Natl. Acad. Sci. USA 2017, 114, E10428–E10437. [Google Scholar] [CrossRef]
- D’Errico, F.; Goverse, G.; Dai, Y.; Wu, W.; Stakenborg, M.; Labeeuw, E.; De Simone, V.; Verstockt, B.; Gomez-Pinilla, P.J.; Warner, M.; et al. Estrogen Receptor β Controls Proliferation of Enteric Glia and Differentiation of Neurons in the Myenteric Plexus after Damage. Proc. Natl. Acad. Sci. USA 2018, 115, 5798–5803. [Google Scholar] [CrossRef]
- Jensen, T.K.; Mustieles, V.; Bleses, D.; Frederiksen, H.; Trecca, F.; Schoeters, G.; Andersen, H.R.; Grandjean, P.; Kyhl, H.B.; Juul, A.; et al. Prenatal Bisphenol A Exposure Is Associated with Language Development but Not with ADHD-Related Behavior in Toddlers from the Odense Child Cohort. Environ. Res. 2019, 170, 398–405. [Google Scholar] [CrossRef]
- Bernstein, H.-G.; Steiner, J.; Guest, P.C.; Dobrowolny, H.; Bogerts, B. Glial Cells as Key Players in Schizophrenia Pathology: Recent Insights and Concepts of Therapy. Schizophr. Res. 2015, 161, 4–18. [Google Scholar] [CrossRef] [PubMed]
- Raabe, F.J.; Galinski, S.; Papiol, S.; Falkai, P.G.; Schmitt, A.; Rossner, M.J. Studying and Modulating Schizophrenia-Associated Dysfunctions of Oligodendrocytes with Patient-Specific Cell Systems. NPJ Schizophr. 2018, 4, 23. [Google Scholar] [CrossRef] [PubMed]
- Gandal, M.J.; Haney, J.R.; Parikshak, N.N.; Leppa, V.; Ramaswami, G.; Hartl, C.; Schork, A.J.; Appadurai, V.; Buil, A.; Werge, T.M.; et al. Shared Molecular Neuropathology across Major Psychiatric Disorders Parallels Polygenic Overlap. Science 2018, 359, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Harley, K.G.; Gunier, R.B.; Kogut, K.; Johnson, C.; Bradman, A.; Calafat, A.M.; Eskenazi, B. Prenatal and Early Childhood Bisphenol A Concentrations and Behavior in School-Aged Children. Environ. Res. 2013, 126, 43–50. [Google Scholar] [CrossRef]
- Mustieles, V.; Pérez-Lobato, R.; Olea, N.; Fernández, M.F. Bisphenol A: Human Exposure and Neurobehavior. Neurotoxicology 2015, 49, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Casas, M.; Forns, J.; Martínez, D.; Avella-García, C.; Valvi, D.; Ballesteros-Gómez, A.; Luque, N.; Rubio, S.; Julvez, J.; Sunyer, J.; et al. Exposure to Bisphenol A during Pregnancy and Child Neuropsychological Development in the INMA-Sabadell Cohort. Environ. Res. 2015, 142, 671–679. [Google Scholar] [CrossRef]
- Roen, E.L.; Wang, Y.; Calafat, A.M.; Wang, S.; Margolis, A.; Herbstman, J.; Hoepner, L.A.; Rauh, V.; Perera, F.P. Bisphenol A Exposure and Behavioral Problems among Inner City Children at 7–9 Years of Age. Environ. Res. 2015, 142, 739–745. [Google Scholar] [CrossRef]
- Perera, F.; Nolte, E.L.R.; Wang, Y.; Margolis, A.E.; Calafat, A.M.; Wang, S.; Garcia, W.; Hoepner, L.A.; Peterson, B.S.; Rauh, V.; et al. Bisphenol A Exposure and Symptoms of Anxiety and Depression among Inner City Children at 10–12 Years of Age. Environ. Res. 2016, 151, 195–202. [Google Scholar] [CrossRef]
- de Cock, M.; Maas, Y.G.H.; van de Bor, M. Does Perinatal Exposure to Endocrine Disruptors Induce Autism Spectrum and Attention Deficit Hyperactivity Disorders? Review. Acta Paediatr. 2012, 101, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Stein, T.P.; Schluter, M.D.; Steer, R.A.; Guo, L.; Ming, X. Bisphenol A Exposure in Children With Autism Spectrum Disorders. Autism Res. 2015, 8, 272–283. [Google Scholar] [CrossRef]
- Johnson, S.A.; Javurek, A.B.; Painter, M.S.; Ellersieck, M.R.; Welsh, T.H.; Camacho, L.; Lewis, S.M.; Vanlandingham, M.M.; Ferguson, S.A.; Rosenfeld, C.S. Effects of Developmental Exposure to Bisphenol A on Spatial Navigational Learning and Memory in Rats: A CLARITY-BPA Study. Horm Behav. 2016, 80, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, M.H.; Swingle, H.M.; Christian, M.A.; Hessabi, M.; Lee, M.; Pitcher, M.R.; Campbell, S.; Mitchell, A.; Krone, R.; Loveland, K.A.; et al. Environmental Exposure to Dioxins, Dibenzofurans, Bisphenol A, and Phthalates in Children with and without Autism Spectrum Disorder Living near the Gulf of Mexico. Int. J. Environ. Res. Public Health 2017, 14, 1425. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.S. Neuroendocrine Disruption in Animal Models Due to Exposure to Bisphenol A Analogues. Front. Neuroendocrinol. 2017, 47, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Thongkorn, S.; Kanlayaprasit, S.; Jindatip, D.; Tencomnao, T.; Hu, V.W.; Sarachana, T. Sex Differences in the Effects of Prenatal Bisphenol A Exposure on Genes Associated with Autism Spectrum Disorder in the Hippocampus. Sci. Rep. 2019, 9, 3038. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Liu, S.; Yang, F.; Dong, P.; Yang, M.; Wong, M.; Zheng, C. Metabolism Disruption Analysis of Zebrafish Larvae in Response to BPA and BPA Analogs Based on RNA-Seq Technique. Ecotoxicol. Environ. Saf. 2019, 174, 181–188. [Google Scholar] [CrossRef]
- Mesnage, R.; Phedonos, A.; Arno, M.; Balu, S.; Corton, J.C.; Antoniou, M.N. Editor’s Highlight: Transcriptome Profiling Reveals Bisphenol A Alternatives Activate Estrogen Receptor Alpha in Human Breast Cancer Cells. Toxicol. Sci. 2017, 158, 431–443. [Google Scholar] [CrossRef]
- Kojima, H.; Takeuchi, S.; Sanoh, S.; Okuda, K.; Kitamura, S.; Uramaru, N.; Sugihara, K.; Yoshinari, K. Profiling of Bisphenol A and Eight Its Analogues on Transcriptional Activity via Human Nuclear Receptors. Toxicology 2019, 413, 48–55. [Google Scholar] [CrossRef]
- Li, R.; Liu, S.; Qiu, W.; Yang, F.; Zheng, Y.; Xiong, Y.; Li, G.; Zheng, C. Transcriptomic Analysis of Bisphenol AF on Early Growth and Development of Zebrafish (Danio Rerio) Larvae. Environ. Sci. Ecotechnol. 2020, 4, 100054. [Google Scholar] [CrossRef]
- Bruno, K.A.; Mathews, J.E.; Yang, A.L.; Frisancho, J.A.; Scott, A.J.; Greyner, H.D.; Molina, F.A.; Greenaway, M.S.; Cooper, G.M.; Bucek, A.; et al. BPA Alters Estrogen Receptor Expression in the Heart After Viral Infection Activating Cardiac Mast Cells and T Cells Leading to Perimyocarditis and Fibrosis. Front. Endocrinol. 2019, 10, 598. [Google Scholar] [CrossRef] [PubMed]
- Denley, M.C.S.; Gatford, N.J.F.; Sellers, K.J.; Srivastava, D.P. Estradiol and the Development of the Cerebral Cortex: An Unexpected Role? Front. Neurosci. 2018, 12, 245. [Google Scholar] [CrossRef]
- MacKay, H.; Abizaid, A. A Plurality of Molecular Targets: The Receptor Ecosystem for Bisphenol-A (BPA). Horm. Behav. 2018, 101, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Ho, D.M.; Artavanis-Tsakonas, S.; Louvi, A. The Notch Pathway in CNS Homeostasis and Neurodegeneration. Wiley Interdiscip. Rev. Dev. Biol. 2020, 9, e358. [Google Scholar] [CrossRef]
- Zhu, M.; Niu, Y.; Li, Y.; Dong, M.; Li, J.; Zeng, R.; Qin, Z. Low Concentrations of Tetrabromobisphenol A Disrupt Notch Signaling and Intestinal Development in in Vitro and in Vivo Models. Chem. Res. Toxicol. 2020, 33, 1418–1427. [Google Scholar] [CrossRef] [PubMed]
- Siebel, C.; Lendahl, U. Notch Signaling in Development, Tissue Homeostasis, and Disease. Physiol. Rev. 2017, 97, 1235–1294. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Murase, K.; Makino, A.; Nakajima, M.; Kaku, T.; Furukawa, S.; Furukawa, Y. Effects of Estrogens on Proliferation and Differentiation of Neural Stem/Progenitor Cells. Biomed. Res. 2008, 29, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Makino, A.; Nakajima, M.; Okuyama, S.; Furukawa, S.; Furukawa, Y. Estrogen Stimulates Proliferation and Differentiation of Neural Stem/Progenitor Cells through Different Signal Transduction Pathways. Int. J. Mol. Sci. 2010, 11, 4114–4123. [Google Scholar] [CrossRef]
- Kinch, C.D.; Ibhazehiebo, K.; Jeong, J.-H.; Habibi, H.R.; Kurrasch, D.M. Low-Dose Exposure to Bisphenol A and Replacement Bisphenol S Induces Precocious Hypothalamic Neurogenesis in Embryonic Zebrafish. Proc. Natl. Acad. Sci. USA 2015, 112, 1475–1480. [Google Scholar] [CrossRef] [PubMed]
- Baba, K.; Okada, K.; Kinoshita, T.; Imaoka, S. Bisphenol A Disrupts Notch Signaling by Inhibiting Gamma-Secretase Activity and Causes Eye Dysplasia of Xenopus Laevis. Toxicol. Sci. 2009, 108, 344–355. [Google Scholar] [CrossRef]
- Yang, C.-W.; Chou, W.-C.; Chen, K.-H.; Cheng, A.-L.; Mao, I.-F.; Chao, H.-R.; Chuang, C.-Y. Visualized Gene Network Reveals the Novel Target Transcripts Sox2 and Pax6 of Neuronal Development in Trans-Placental Exposure to Bisphenol A. PLoS ONE 2014, 9, e100576. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, S.; Mori, T.; Kinoshita, T. Bisphenol A Causes Malformation of the Head Region in Embryos of Xenopus Laevis and Decreases the Expression of the ESR-1 Gene Mediated by Notch Signaling. Biol. Pharm. Bull. 2007, 30, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Yin, N.; Liang, S.; Yang, R.; Liu, S.; Lu, Y.; Jiang, L.; Zhou, Q.; Jiang, G.; Faiola, F. Bisphenol A and Several Derivatives Exert Neural Toxicity in Human Neuron-like Cells by Decreasing Neurite Length. Food Chem. Toxicol. 2020, 135, 111015. [Google Scholar] [CrossRef] [PubMed]
- Demitrack, E.S.; Samuelson, L.C. Notch Regulation of Gastrointestinal Stem Cells. J. Physiol. 2016, 594, 4791–4803. [Google Scholar] [CrossRef]
- Engler, A.; Zhang, R.; Taylor, V. Notch and Neurogenesis. Adv. Exp. Med. Biol. 2018, 1066, 223–234. [Google Scholar] [CrossRef]
- Basak, O.; Giachino, C.; Fiorini, E.; Macdonald, H.R.; Taylor, V. Neurogenic Subventricular Zone Stem/Progenitor Cells Are Notch1-Dependent in Their Active but Not Quiescent State. J. Neurosci. 2012, 32, 5654–5666. [Google Scholar] [CrossRef]
- Breunig, J.J.; Silbereis, J.; Vaccarino, F.M.; Sestan, N.; Rakic, P. Notch Regulates Cell Fate and Dendrite Morphology of Newborn Neurons in the Postnatal Dentate Gyrus. Proc. Natl. Acad. Sci. USA 2007, 104, 20558–20563. [Google Scholar] [CrossRef] [PubMed]
- Kopan, R.; Ilagan, M.X.G. The Canonical Notch Signaling Pathway: Unfolding the Activation Mechanism. Cell 2009, 137, 216–233. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, K.; Yoon, K.; Dang, L.; Tokunaga, A.; Gaiano, N. Differential Notch Signalling Distinguishes Neural Stem Cells from Intermediate Progenitors. Nature 2007, 449, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, R.; Ohtsuka, T.; Kobayashi, T. Roles of Hes Genes in Neural Development. Dev. Growth Differ. 2008, 50 (Suppl. 1), S97–S103. [Google Scholar] [CrossRef] [PubMed]
- Tandon, A.; Singh, S.J.; Gupta, M.; Singh, N.; Shankar, J.; Arjaria, N.; Goyal, S.; Chaturvedi, R.K. Notch Pathway Up-Regulation via Curcumin Mitigates Bisphenol-A (BPA) Induced Alterations in Hippocampal Oligodendrogenesis. J. Hazard. Mater. 2020, 392, 122052. [Google Scholar] [CrossRef] [PubMed]
- Shimojo, H.; Ohtsuka, T.; Kageyama, R. Dynamic Expression of Notch Signaling Genes in Neural Stem/Progenitor Cells. Front. Neurosci. 2011, 5, 78. [Google Scholar] [CrossRef]
- Artavanis-Tsakonas, S.; Rand, M.D.; Lake, R.J. Notch Signaling: Cell Fate Control and Signal Integration in Development. Science 1999, 284, 770–776. [Google Scholar] [CrossRef]
- Murata, M.; Kang, J.-H. Bisphenol A (BPA) and Cell Signaling Pathways. Biotechnol. Adv. 2018, 36, 311–327. [Google Scholar] [CrossRef]
- Xu, X.; Lu, Y.; Zhang, G.; Chen, L.; Tian, D.; Shen, X.; Yang, Y.; Dong, F. Bisphenol A Promotes Dendritic Morphogenesis of Hippocampal Neurons through Estrogen Receptor-Mediated ERK1/2 Signal Pathway. Chemosphere 2014, 96, 129–137. [Google Scholar] [CrossRef]
- Alberi, L.; Liu, S.; Wang, Y.; Badie, R.; Smith-Hicks, C.; Wu, J.; Pierfelice, T.J.; Abazyan, B.; Mattson, M.P.; Kuhl, D.; et al. Activity-Induced Notch Signaling in Neurons Requires Arc/Arg3.1 and Is Essential for Synaptic Plasticity in Hippocampal Networks. Neuron 2011, 69, 437–444. [Google Scholar] [CrossRef]
- Ding, X.-F.; Gao, X.; Ding, X.-C.; Fan, M.; Chen, J. Postnatal Dysregulation of Notch Signal Disrupts Dendrite Development of Adult-Born Neurons in the Hippocampus and Contributes to Memory Impairment. Sci. Rep. 2016, 6, 25780. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gill, S.; Kumara, V.M.R. Comparative Neurodevelopment Effects of Bisphenol A and Bisphenol F on Rat Fetal Neural Stem Cell Models. Cells 2021, 10, 793. https://doi.org/10.3390/cells10040793
Gill S, Kumara VMR. Comparative Neurodevelopment Effects of Bisphenol A and Bisphenol F on Rat Fetal Neural Stem Cell Models. Cells. 2021; 10(4):793. https://doi.org/10.3390/cells10040793
Chicago/Turabian StyleGill, Santokh, and V. M. Ruvin Kumara. 2021. "Comparative Neurodevelopment Effects of Bisphenol A and Bisphenol F on Rat Fetal Neural Stem Cell Models" Cells 10, no. 4: 793. https://doi.org/10.3390/cells10040793
APA StyleGill, S., & Kumara, V. M. R. (2021). Comparative Neurodevelopment Effects of Bisphenol A and Bisphenol F on Rat Fetal Neural Stem Cell Models. Cells, 10(4), 793. https://doi.org/10.3390/cells10040793




