Natural Compounds of Marine Origin as Inducers of Immunogenic Cell Death (ICD): Potential Role for Cancer Interception and Therapy
Abstract
1. Introduction
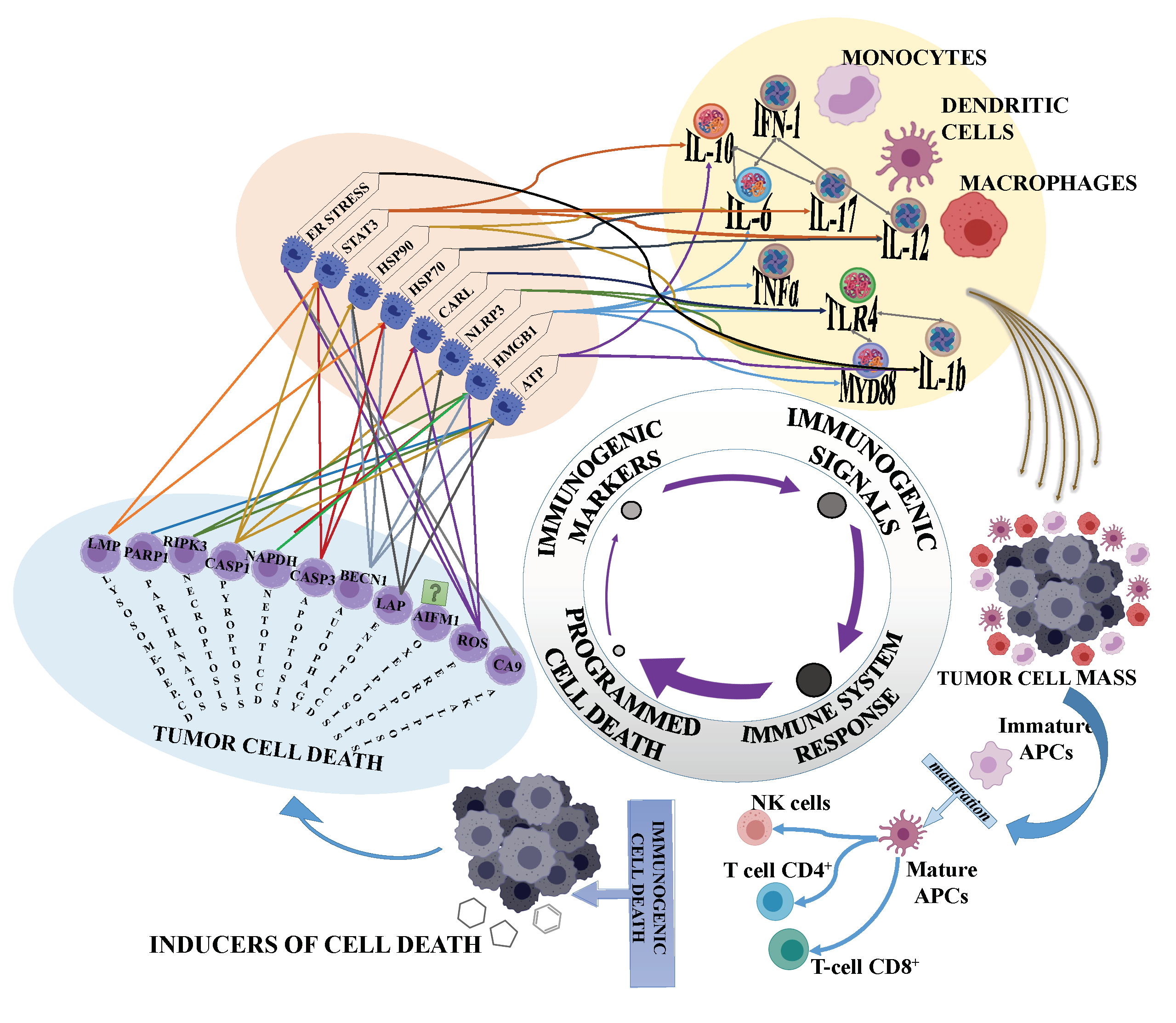
2. Immunogenic Cell Death and Cancer
3. Anticancer Drugs and Terrestrial Natural Compounds Inducing ICD
4. Marine Microalgal Compounds Inducing ICD in Cancer Cells
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Albini, A.; Pennesi, G.; Donatelli, F.; Cammarota, R.; De Flora, S.; Noonan, D.M. Cardiotoxicity of anticancer drugs: The need for cardio-oncology and cardio-oncological prevention. J. Natl. Cancer Inst. 2010, 102, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Sa, G.; Saha, B.; Das, K. Multifocal signal modulation therapy of cancer: Ancient weapon, modern targets. Mol. Cell Biochem. 2010, 336, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Panda, A.K.; Chakraborty, D.; Sarkar, I.; Khan, T.; Sa, G. New insights into therapeutic activity and anticancer properties of curcumin. J. Exp. Pharmacol. 2017, 9, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Warren, S.; Adjemian, S.; Agostinis, P.; Martinez, A.B.; Chan, T.A.; Coukos, G.; Demaria, S.; Deutsch, E.; et al. Consensus guidelines for the definition, detection and interpretation of immunogenic cell death. J. Immunother. Cancer 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Montico, B.; Nigro, A.; Casolaro, V.; Dal Col, J. Immunogenic Apoptosis as a Novel Tool for Anticancer Vaccine Development. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Fuchs, Y.; Steller, H. Live to die another way: Modes of programmed cell death and the signals emanating from dying cells. Nat. Rev. Mol. Cell Biol. 2015, 16, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Buque, A.; Kepp, O.; Zitvogel, L.; Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 2017, 17, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Bravo-San Pedro, J.M.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Alnemri, E.S.; Altucci, L.; Andrews, D.; Annicchiarico-Petruzzelli, M.; et al. Essential versus accessory aspects of cell death: Recommendations of the NCCD 2015. Cell Death Differ. 2015, 22, 58–73. [Google Scholar] [CrossRef]
- Teng, X.; Hardwick, J.M. Cell death in genome evolution. Semin. Cell Dev. Biol. 2015, 39, 3–11. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cirone, M.; Gilardini Montani, M.S.; Granato, M.; Garufi, A.; Faggioni, A.; D’Orazi, G. Autophagy manipulation as a strategy for efficient anticancer therapies: Possible consequences. J. Exp. Clin. Cancer Res. 2019, 38, 262. [Google Scholar] [CrossRef] [PubMed]
- Nirmala, J.G.; Lopus, M. Cell death mechanisms in eukaryotes. Cell Biol. Toxicol. 2020, 36, 145–164. [Google Scholar] [CrossRef]
- Smith, A.G.; Macleod, K.F. Autophagy, cancer stem cells and drug resistance. J. Pathol. 2019, 247, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Kang, R.; Berghe, T.V.; Vandenabeele, P.; Kroemer, G. The molecular machinery of regulated cell death. Cell Res. 2019, 29, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Maltese, W.A.; Overmeyer, J.H. Methuosis: Nonapoptotic cell death associated with vacuolization of macropinosome and endosome compartments. Am. J. Pathol. 2014, 184, 1630–1642. [Google Scholar] [CrossRef]
- Maltese, W.A.; Overmeyer, J.H. Non-apoptotic cell death associated with perturbations of macropinocytosis. Front. Physiol. 2015, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.L.; Lai, J.J.; Kono, H. Innate and adaptive immune responses to cell death. Immunol. Rev. 2011, 243, 191–205. [Google Scholar] [CrossRef]
- Matzinger, P. The danger model: A renewed sense of self. Science 2002, 296, 301–305. [Google Scholar] [CrossRef]
- Pai, S.I.; Cesano, A.; Marincola, F.M. The Paradox of Cancer Immune Exclusion: Immune Oncology Next Frontier. Cancer Treat. Res. 2020, 180, 173–195. [Google Scholar] [CrossRef]
- Garg, A.D.; More, S.; Rufo, N.; Mece, O.; Sassano, M.L.; Agostinis, P.; Zitvogel, L.; Kroemer, G.; Galluzzi, L. Trial watch: Immunogenic cell death induction by anticancer chemotherapeutics. Oncoimmunology 2017, 6, e1386829. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Kepp, O.; Zitvogel, L. Immunogenic cell death in cancer therapy. Annu. Rev. Immunol. 2013, 31, 51–72. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Fletcher, R.; Yu, J.; Zhang, L. Immunogenic effects of chemotherapy-induced tumor cell death. Genes. Dis. 2018, 5, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Probst, H.C.; McCoy, K.; Okazaki, T.; Honjo, T.; van den Broek, M. Resting dendritic cells induce peripheral CD8+ T cell tolerance through PD-1 and CTLA-4. Nat. Immunol. 2005, 6, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, G.; Chen, Y.; Wang, H.; Hua, Y.; Cai, Z. Immunogenic cell death in cancer therapy: Present and emerging inducers. J. Cell Mol. Med. 2019, 23, 4854–4865. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Del Valle, A.; Anel, A.; Naval, J.; Marzo, I. Immunogenic Cell Death and Immunotherapy of Multiple Myeloma. Front. Cell Dev. Biol. 2019, 7, 50. [Google Scholar] [CrossRef]
- Peterkova, L.; Kmonickova, E.; Ruml, T.; Rimpelova, S. Sarco/Endoplasmic Reticulum Calcium ATPase Inhibitors: Beyond Anticancer Perspective. J. Med. Chem. 2020, 63, 1937–1963. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Davison, E.K.; Brimble, M.A. Natural product derived privileged scaffolds in drug discovery. Curr. Opin. Chem. Biol. 2019, 52, 1–8. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Galluzzi, L.; Zitvogel, L.; Kroemer, G. Immunological Mechanisms Underneath the Efficacy of Cancer Therapy. Cancer Immunol. Res. 2016, 4, 895–902. [Google Scholar] [CrossRef]
- Cragg, G.M.; Pezzuto, J.M. Natural Products as a Vital Source for the Discovery of Cancer Chemotherapeutic and Chemopreventive Agents. Med. Princ. Pract. 2016, 25 (Suppl. S2), 41–59. [Google Scholar] [CrossRef]
- Demain, A.L.; Vaishnav, P. Natural products for cancer chemotherapy. Microb. Biotechnol. 2011, 4, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801. [Google Scholar] [CrossRef]
- Darvin, P.; Toor, S.M.; Sasidharan Nair, V.; Elkord, E. Immune checkpoint inhibitors: Recent progress and potential biomarkers. Exp. Mol. Med. 2018, 50, 1–11. [Google Scholar] [CrossRef]
- Lo Russo, G.; Moro, M.; Sommariva, M.; Cancila, V.; Boeri, M.; Centonze, G.; Ferro, S.; Ganzinelli, M.; Gasparini, P.; Huber, V.; et al. Antibody-Fc/FcR Interaction on Macrophages as a Mechanism for Hyperprogressive Disease in Non-small Cell Lung Cancer Subsequent to PD-1/PD-L1 Blockade. Clin. Cancer Res. 2019, 25, 989–999. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, P.C.; Wilke, D.V.; Branco, P.C.; Bauermeister, A.; Rezende-Teixeira, P.; Gaudencio, S.P.; Costa-Lotufo, L.V. Enriching cancer pharmacology with drugs of marine origin. Br. J. Pharmacol. 2020, 177, 3–27. [Google Scholar] [CrossRef]
- Liang, X.; Luo, D.; Luesch, H. Advances in exploring the therapeutic potential of marine natural products. Pharmacol. Res. 2019, 147, 104373. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Sorolla, M.A.; Krishnan, P.D.G.; Sorolla, A. From Seabed to Bedside: A Review on Promising Marine Anticancer Compounds. Biomolecules 2020, 10. [Google Scholar] [CrossRef]
- Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2020, 37, 175–223. [Google Scholar] [CrossRef]
- Galasso, C.; Gentile, A.; Orefice, I.; Ianora, A.; Bruno, A.; Noonan, D.M.; Sansone, C.; Albini, A.; Brunet, C. Microalgal Derivatives as Potential Nutraceutical and Food Supplements for Human Health: A Focus on Cancer Prevention and Interception. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Del Mondo, A.; Smerilli, A.; Sane, E.; Sansone, C.; Brunet, C. Challenging microalgal vitamins for human health. Microb. Cell Fact. 2020, 19, 201. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hack, M.E.; Abdelnour, S.; Alagawany, M.; Abdo, M.; Sakr, M.A.; Khafaga, A.F.; Mahgoub, S.A.; Elnesr, S.S.; Gebriel, M.G. Microalgae in modern cancer therapy: Current knowledge. Biomed. Pharmacother. 2019, 111, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Sansone, C.; Brunet, C. Promises and Challenges of Microalgal Antioxidant Production. Antioxidants 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Martinez Andrade, K.A.; Lauritano, C.; Romano, G.; Ianora, A. Marine Microalgae with Anti-Cancer Properties. Mar. Drugs. 2018, 16. [Google Scholar] [CrossRef]
- Galasso, C.; Nuzzo, G.; Brunet, C.; Ianora, A.; Sardo, A.; Fontana, A.; Sansone, C. The Marine Dinoflagellate Alexandrium minutum Activates a Mitophagic Pathway in Human Lung Cancer Cells. Mar. Drugs. 2018, 16. [Google Scholar] [CrossRef]
- Riccio, G.; Lauritano, C. Microalgae with Immunomodulatory Activities. Mar. Drugs. 2019, 18. [Google Scholar] [CrossRef]
- Wolf, M.T.; Ganguly, S.; Wang, T.L.; Anderson, C.W.; Sadtler, K.; Narain, R.; Cherry, C.; Parrillo, A.J.; Park, B.V.; Wang, G.; et al. A biologic scaffold-associated type 2 immune microenvironment inhibits tumor formation and synergizes with checkpoint immunotherapy. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef]
- Garg, A.D.; Agostinis, P. Editorial: Immunogenic Cell Death in Cancer: From Benchside Research to Bedside Reality. Front. Immunol. 2016, 7, 110. [Google Scholar] [CrossRef]
- Hernandez, C.; Huebener, P.; Schwabe, R.F. Damage-associated molecular patterns in cancer: A double-edged sword. Oncogene 2016, 35, 5931–5941. [Google Scholar] [CrossRef]
- Sharma, D.; Kanneganti, T.D. The cell biology of inflammasomes: Mechanisms of inflammasome activation and regulation. J. Cell Biol. 2016, 213, 617–629. [Google Scholar] [CrossRef]
- Galluzzi, L.; Bravo-San Pedro, J.M.; Kepp, O.; Kroemer, G. Regulated cell death and adaptive stress responses. Cell Mol. Life. Sci. 2016, 73, 2405–2410. [Google Scholar] [CrossRef] [PubMed]
- Lo Nigro, C.; Macagno, M.; Sangiolo, D.; Bertolaccini, L.; Aglietta, M.; Merlano, M.C. NK-mediated antibody-dependent cell-mediated cytotoxicity in solid tumors: Biological evidence and clinical perspectives. Ann. Transl. Med. 2019, 7, 105. [Google Scholar] [CrossRef] [PubMed]
- Degli-Esposti, M.A.; Smyth, M.J. Close encounters of different kinds: Dendritic cells and NK cells take centre stage. Nat. Rev. Immunol. 2005, 5, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Hamerman, J.A.; Ogasawara, K.; Lanier, L.L. NK cells in innate immunity. Curr. Opin. Immunol. 2005, 17, 29–35. [Google Scholar] [CrossRef]
- Raulet, D.H. Interplay of natural killer cells and their receptors with the adaptive immune response. Nat. Immunol. 2004, 5, 996–1002. [Google Scholar] [CrossRef]
- Guo, C.; Manjili, M.H.; Subjeck, J.R.; Sarkar, D.; Fisher, P.B.; Wang, X.Y. Therapeutic cancer vaccines: Past, present, and future. Adv. Cancer Res. 2013, 119, 421–475. [Google Scholar] [CrossRef]
- Krysko, O.; Love Aaes, T.; Bachert, C.; Vandenabeele, P.; Krysko, D.V. Many faces of DAMPs in cancer therapy. Cell Death Dis. 2013, 4, e631. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Waxman, D.J. Metronomic cyclophosphamide eradicates large implanted GL261 gliomas by activating antitumor Cd8(+) T-cell responses and immune memory. Oncoimmunology 2015, 4, e1005521. [Google Scholar] [CrossRef]
- Schiavoni, G.; Sistigu, A.; Valentini, M.; Mattei, F.; Sestili, P.; Spadaro, F.; Sanchez, M.; Lorenzi, S.; D’Urso, M.T.; Belardelli, F.; et al. Cyclophosphamide synergizes with type I interferons through systemic dendritic cell reactivation and induction of immunogenic tumor apoptosis. Cancer Res. 2011, 71, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Iida, N.; Dzutsev, A.; Stewart, C.A.; Smith, L.; Bouladoux, N.; Weingarten, R.A.; Molina, D.A.; Salcedo, R.; Back, T.; Cramer, S.; et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science 2013, 342, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillere, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 2013, 342, 971–976. [Google Scholar] [CrossRef]
- Martins, I.; Kepp, O.; Schlemmer, F.; Adjemian, S.; Tailler, M.; Shen, S.; Michaud, M.; Menger, L.; Gdoura, A.; Tajeddine, N.; et al. Restoration of the immunogenicity of cisplatin-induced cancer cell death by endoplasmic reticulum stress. Oncogene 2011, 30, 1147–1158. [Google Scholar] [CrossRef]
- Shalapour, S.; Font-Burgada, J.; Di Caro, G.; Zhong, Z.; Sanchez-Lopez, E.; Dhar, D.; Willimsky, G.; Ammirante, M.; Strasner, A.; Hansel, D.E.; et al. Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy. Nature 2015, 521, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Emile, J.F.; Julie, C.; Le Malicot, K.; Lepage, C.; Tabernero, J.; Mini, E.; Folprecht, G.; Van Laethem, J.L.; Dimet, S.; Boulagnon-Rombi, C.; et al. Prospective validation of a lymphocyte infiltration prognostic test in stage III colon cancer patients treated with adjuvant FOLFOX. Eur. J. Cancer 2017, 82, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Chua, W.; Cheng, C.; Descallar, J.; Ng, W.; Solomon, M.; Bokey, L.; Wong, K.; Lee, M.T.; de Souza, P.; et al. Effect of neoadjuvant chemoradiation on tumor-infiltrating/associated lymphocytes in locally advanced rectal cancers. Anticancer Res. 2014, 34, 6505–6513. [Google Scholar] [PubMed]
- Zhu, X.; Messer, J.S.; Wang, Y.; Lin, F.; Cham, C.M.; Chang, J.; Billiar, T.R.; Lotze, M.T.; Boone, D.L.; Chang, E.B. Cytosolic HMGB1 controls the cellular autophagy/apoptosis checkpoint during inflammation. J. Clin. Invest. 2015, 125, 1098–1110. [Google Scholar] [CrossRef] [PubMed]
- Ghiringhelli, F.; Apetoh, L.; Tesniere, A.; Aymeric, L.; Ma, Y.; Ortiz, C.; Vermaelen, K.; Panaretakis, T.; Mignot, G.; Ullrich, E.; et al. Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors. Nat. Med. 2009, 15, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Antonioli, L.; Blandizzi, C.; Pacher, P.; Hasko, G. Immunity, inflammation and cancer: A leading role for adenosine. Nat. Rev. Cancer 2013, 13, 842–857. [Google Scholar] [CrossRef] [PubMed]
- Colangelo, T.; Polcaro, G.; Ziccardi, P.; Muccillo, L.; Galgani, M.; Pucci, B.; Milone, M.R.; Budillon, A.; Santopaolo, M.; Mazzoccoli, G.; et al. The miR-27a-calreticulin axis affects drug-induced immunogenic cell death in human colorectal cancer cells. Cell Death Dis. 2016, 7, e2108. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Agostinis, P. Cell death and immunity in cancer: From danger signals to mimicry of pathogen defense responses. Immunol. Rev. 2017, 280, 126–148. [Google Scholar] [CrossRef]
- Aguirre-Hernandez, C.; Maya-Pineda, H.; Millan, J.S.; Man, Y.K.S.; Lu, Y.J.; Hallden, G. Sensitisation to mitoxantrone-induced apoptosis by the oncolytic adenovirus Ad through Bcl-2-dependent attenuation of autophagy. Oncogenesis 2018, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, L.; Liang, R.; Luo, Z.; He, H.; Wu, Z.; Tian, H.; Zheng, M.; Ma, Y.; Cai, L. Bioinspired Hybrid Protein Oxygen Nanocarrier Amplified Photodynamic Therapy for Eliciting Anti-tumor Immunity and Abscopal Effect. ACS Nano 2018, 12, 8633–8645. [Google Scholar] [CrossRef] [PubMed]
- Diederich, M. Natural compound inducers of immunogenic cell death. Arch. Pharm. Res. 2019, 42, 629–645. [Google Scholar] [CrossRef] [PubMed]
- Casares, N.; Pequignot, M.O.; Tesniere, A.; Ghiringhelli, F.; Roux, S.; Chaput, N.; Schmitt, E.; Hamai, A.; Hervas-Stubbs, S.; Obeid, M.; et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J. Exp. Med. 2005, 202, 1691–1701. [Google Scholar] [CrossRef] [PubMed]
- Fredly, H.; Ersvaer, E.; Gjertsen, B.T.; Bruserud, O. Immunogenic apoptosis in human acute myeloid leukemia (AML): Primary human AML cells expose calreticulin and release heat shock protein (HSP) 70 and HSP90 during apoptosis. Oncol. Rep. 2011, 25, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cadena, A.; Uruena, C.; Prieto, K.; Martinez-Usatorre, A.; Donda, A.; Barreto, A.; Romero, P.; Fiorentino, S. Immune-system-dependent anti-tumor activity of a plant-derived polyphenol rich fraction in a melanoma mouse model. Cell Death Dis. 2016, 7, e2243. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.Y.; Shen, Y.L. Linalool exhibits cytotoxic effects by activating antitumor immunity. Molecules 2014, 19, 6694–6706. [Google Scholar] [CrossRef]
- Chen, H.M.; Wang, P.H.; Chen, S.S.; Wen, C.C.; Chen, Y.H.; Yang, W.C.; Yang, N.S. Shikonin induces immunogenic cell death in tumor cells and enhances dendritic cell-based cancer vaccine. Cancer Immunol. Immunother. 2012, 61, 1989–2002. [Google Scholar] [CrossRef]
- Iezzi, G.; Sonderegger, I.; Ampenberger, F.; Schmitz, N.; Marsland, B.J.; Kopf, M. CD40-CD40L cross-talk integrates strong antigenic signals and microbial stimuli to induce development of IL-17-producing CD4+ T cells. Proc. Natl. Acad. Sci. USA 2009, 106, 876–881. [Google Scholar] [CrossRef]
- Bley, K.; Boorman, G.; Mohammad, B.; McKenzie, D.; Babbar, S. A comprehensive review of the carcinogenic and anticarcinogenic potential of capsaicin. Toxicol. Pathol. 2012, 40, 847–873. [Google Scholar] [CrossRef]
- D’Eliseo, D.; Manzi, L.; Velotti, F. Capsaicin as an inducer of damage-associated molecular patterns (DAMPs) of immunogenic cell death (ICD) in human bladder cancer cells. Cell Stress Chaperones 2013, 18, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Humphreys, I.; Sahu, R.P.; Shi, Y.; Srivastava, S.K. In vitro and in vivo induction of apoptosis by capsaicin in pancreatic cancer cells is mediated through ROS generation and mitochondrial death pathway. Apoptosis 2008, 13, 1465–1478. [Google Scholar] [CrossRef] [PubMed]
- Diederich, M.; Muller, F.; Cerella, C. Cardiac glycosides: From molecular targets to immunogenic cell death. Biochem. Pharmacol. 2017, 125, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kepp, O.; Menger, L.; Vacchelli, E.; Adjemian, S.; Martins, I.; Ma, Y.; Sukkurwala, A.Q.; Michaud, M.; Galluzzi, L.; Zitvogel, L.; et al. Anticancer activity of cardiac glycosides: At the frontier between cell-autonomous and immunological effects. Oncoimmunology 2012, 1, 1640–1642. [Google Scholar] [CrossRef] [PubMed]
- Menger, L.; Vacchelli, E.; Adjemian, S.; Martins, I.; Ma, Y.; Shen, S.; Yamazaki, T.; Sukkurwala, A.Q.; Michaud, M.; Mignot, G.; et al. Cardiac glycosides exert anticancer effects by inducing immunogenic cell death. Sci. Transl. Med. 2012, 4, 143ra199. [Google Scholar] [CrossRef]
- Inoue, H.; Tani, K. Multimodal immunogenic cancer cell death as a consequence of anticancer cytotoxic treatments. Cell Death Differ. 2014, 21, 39–49. [Google Scholar] [CrossRef]
- Tewksbury, J.J.; Nabhan, G.P. Seed dispersal. Directed deterrence by capsaicin in chilies. Nature 2001, 412, 403–404. [Google Scholar] [CrossRef]
- Granato, M.; Gilardini Montani, M.S.; Filardi, M.; Faggioni, A.; Cirone, M. Capsaicin triggers immunogenic PEL cell death, stimulates DCs and reverts PEL-induced immune suppression. Oncotarget 2015, 6, 29543–29554. [Google Scholar] [CrossRef]
- Lin, K.H.; Lin, K.C.; Lu, W.J.; Thomas, P.A.; Jayakumar, T.; Sheu, J.R. Astaxanthin, a Carotenoid, Stimulates Immune Responses by Enhancing IFN-gamma and IL-2 Secretion in Primary Cultured Lymphocytes in Vitro and ex Vivo. Int. J. Mol. Sci. 2015, 17. [Google Scholar] [CrossRef]
- Hashimoto, H.; Arai, K.; Takahashi, J.; Chikuda, M. Effects of astaxanthin on VEGF level and antioxidation in human aqueous humor: Difference by sex. J. Clin. Biochem. Nutr. 2019, 65, 47–51. [Google Scholar] [CrossRef]
- Sansone, C.; Braca, A.; Ercolesi, E.; Romano, G.; Palumbo, A.; Casotti, R.; Francone, M.; Ianora, A. Diatom-derived polyunsaturated aldehydes activate cell death in human cancer cell lines but not normal cells. PLoS ONE 2014, 9, e101220. [Google Scholar] [CrossRef]
- Cutignano, A.; Nuzzo, G.; Ianora, A.; Luongo, E.; Romano, G.; Gallo, C.; Sansone, C.; Aprea, S.; Mancini, F.; D’Oro, U.; et al. Development and Application of a Novel SPE-Method for Bioassay-Guided Fractionation of Marine Extracts. Mar. Drugs. 2015, 13, 5736–5749. [Google Scholar] [CrossRef] [PubMed]
- Showalter, A.; Limaye, A.; Oyer, J.L.; Igarashi, R.; Kittipatarin, C.; Copik, A.J.; Khaled, A.R. Cytokines in immunogenic cell death: Applications for cancer immunotherapy. Cytokine 2017, 97, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Barsanti, L.; Gualtieri, P. Paramylon, a Potent Immunomodulator from WZSL Mutant of Euglena gracilis. Molecules 2019, 24. [Google Scholar] [CrossRef]
- Yim, J.H.; Kim, S.J.; Ahn, S.H.; Lee, H.K. Characterization of a novel bioflocculant, p-KG03, from a marine dinoflagellate, Gyrodinium impudicum KG03. Bioresour. Technol. 2007, 98, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Chandrarathna, H.; Liyanage, T.D.; Edirisinghe, S.L.; Dananjaya, S.H.S.; Thulshan, E.H.T.; Nikapitiya, C.; Oh, C.; Kang, D.H.; De Zoysa, M. Marine Microalgae, Spirulina maxima-Derived Modified Pectin and Modified Pectin Nanoparticles Modulate the Gut Microbiota and Trigger Immune Responses in Mice. Mar. Drugs. 2020, 18. [Google Scholar] [CrossRef]
- Souza, F.P.; Lima, E.C.S.; Urrea-Rojas, A.M.; Suphoronski, S.A.; Facimoto, C.T.; Bezerra Junior, J.D.S.; Oliveira, T.E.S.; Pereira, U.P.; Santis, G.W.D.; Oliveira, C.A.L.; et al. Effects of dietary supplementation with a microalga (Schizochytrium sp.) on the hemato-immunological, and intestinal histological parameters and gut microbiota of Nile tilapia in net cages. PLoS ONE 2020, 15, e0226977. [Google Scholar] [CrossRef] [PubMed]
- Park, G.T.; Go, R.E.; Lee, H.M.; Lee, G.A.; Kim, C.W.; Seo, J.W.; Hong, W.K.; Choi, K.C.; Hwang, K.A. Potential Anti-proliferative and Immunomodulatory Effects of Marine Microalgal Exopolysaccharide on Various Human Cancer Cells and Lymphocytes In Vitro. Mar. Biotechnol. 2017, 19, 136–146. [Google Scholar] [CrossRef]
- Chen, X.; Song, L.; Wang, H.; Liu, S.; Yu, H.; Wang, X.; Li, R.; Liu, T.; Li, P. Partial Characterization, the Immune Modulation and Anticancer Activities of Sulfated Polysaccharides from Filamentous Microalgae Tribonema sp. Molecules 2019, 24. [Google Scholar] [CrossRef]
- Hau, A.M.; Greenwood, J.A.; Lohr, C.V.; Serrill, J.D.; Proteau, P.J.; Ganley, I.G.; McPhail, K.L.; Ishmael, J.E. Coibamide A induces mTOR-independent autophagy and cell death in human glioblastoma cells. PLoS ONE 2013, 8, e65250. [Google Scholar] [CrossRef]
- Wu, Q.; Liu, L.; Miron, A.; Klimova, B.; Wan, D.; Kuca, K. The antioxidant, immunomodulatory, and anti-inflammatory activities of Spirulina: An overview. Arch. Toxicol. 2016, 90, 1817–1840. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Cutignano, A.; Pagano, D.; Gallo, C.; Barra, G.; Nuzzo, G.; Sansone, C.; Ianora, A.; Urbanek, K.; Fenoglio, D.; et al. A new marine-derived sulfoglycolipid triggers dendritic cell activation and immune adjuvant response. Sci. Rep. 2017, 7, 6286. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Gallo, C.; Fioretto, L.; Nuzzo, G.; Barra, G.; Pagano, D.; Krauss, I.R.; Paduano, L.; Ziaco, M.; DellaGreca, M.; et al. Diasteroselective Colloidal Self-Assembly Affects the Immunological Response of the Molecular Adjuvant Sulfavant. ACS Omega 2019, 4, 7807–7814. [Google Scholar] [CrossRef]
- You, L.; Jin, S.; Zhu, L.; Qian, W. Autophagy, autophagy-associated adaptive immune responses and its role in hematologic malignancies. Oncotarget 2017, 8, 12374–12388. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, J.; Dasgupta, S.; Nagle, V.; Bhadra, B. COVID-19: Potential of Microalgae Derived Natural Astaxanthin As Adjunctive Supplement in Alleviating Cytokine Storm. SSRN Tomorrow Res. Today 2020. [Google Scholar] [CrossRef]
- Zhang, D.W.; Shao, J.; Lin, J.; Zhang, N.; Lu, B.J.; Lin, S.C.; Dong, M.Q.; Han, J. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 2009, 325, 332–336. [Google Scholar] [CrossRef]
- Kaiser, W.J.; Daley-Bauer, L.P.; Thapa, R.J.; Mandal, P.; Berger, S.B.; Huang, C.; Sundararajan, A.; Guo, H.; Roback, L.; Speck, S.H.; et al. RIP1 suppresses innate immune necrotic as well as apoptotic cell death during mammalian parturition. Proc. Natl. Acad. Sci. USA 2014, 111, 7753–7758. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Zhang, Y.; Han, J. RIP3 is an upregulator of aerobic metabolism and the enhanced respiration by necrosomal RIP3 feeds back on necrosome to promote necroptosis. Cell Death Differ. 2018, 25, 821–824. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Ma, Y.; Chen, G.; Zhou, H.; Yamazaki, T.; Klein, C.; Pietrocola, F.; Vacchelli, E.; Souquere, S.; Sauvat, A.; et al. Contribution of RIP3 and MLKL to immunogenic cell death signaling in cancer chemotherapy. Oncoimmunology 2016, 5, e1149673. [Google Scholar] [CrossRef]
- Florean, C.; Dicato, M.; Diederich, M. Immune-modulating and anti-inflammatory marine compounds against cancer. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef]
- Vazquez, M.I.; Catalan-Dibene, J.; Zlotnik, A. B cells responses and cytokine production are regulated by their immune microenvironment. Cytokine 2015, 74, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Albini, A.; Bruno, A.; Noonan, D.M.; Mortara, L. Contribution to Tumor Angiogenesis from Innate Immune Cells Within the Tumor Microenvironment: Implications for Immunotherapy. Front. Immunol. 2018, 9, 527. [Google Scholar] [CrossRef] [PubMed]
- Bassani, B.; Baci, D.; Gallazzi, M.; Poggi, A.; Bruno, A.; Mortara, L. Natural Killer Cells as Key Players of Tumor Progression and Angiogenesis: Old and Novel Tools to Divert Their Pro-Tumor Activities into Potent Anti-Tumor Effects. Cancers 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.L.; Gladney, W.L. Immune escape mechanisms as a guide for cancer immunotherapy. Clin. Cancer Res. 2015, 21, 687–692. [Google Scholar] [CrossRef]
- Bruno, A.; Mortara, L.; Baci, D.; Noonan, D.M.; Albini, A. Myeloid Derived Suppressor Cells Interactions with Natural Killer Cells and Pro-angiogenic Activities: Roles in Tumor Progression. Front. Immunol. 2019, 10, 771. [Google Scholar] [CrossRef]
- Noonan, D.M.; De Lerma Barbaro, A.; Vannini, N.; Mortara, L.; Albini, A. Inflammation, inflammatory cells and angiogenesis: Decisions and indecisions. Cancer Metastasis Rev. 2008, 27, 31–40. [Google Scholar] [CrossRef]
- Szegezdi, E.; Logue, S.E.; Gorman, A.M.; Samali, A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006, 7, 880–885. [Google Scholar] [CrossRef]
- Green, D.R.; Ferguson, T.; Zitvogel, L.; Kroemer, G. Immunogenic and tolerogenic cell death. Nat. Rev. Immunol. 2009, 9, 353–363. [Google Scholar] [CrossRef]
- Zitvogel, L.; Daillere, R.; Roberti, M.P.; Routy, B.; Kroemer, G. Anticancer effects of the microbiome and its products. Nat. Rev. Microbiol. 2017, 15, 465–478. [Google Scholar] [CrossRef]
- Zitvogel, L.; Pietrocola, F.; Kroemer, G. Nutrition, inflammation and cancer. Nat. Immunol. 2017, 18, 843–850. [Google Scholar] [CrossRef]
- Garrett, W.S. Cancer and the microbiota. Science 2015, 348, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Helmink, B.A.; Khan, M.A.W.; Hermann, A.; Gopalakrishnan, V.; Wargo, J.A. The microbiome, cancer, and cancer therapy. Nat. Med. 2019, 25, 377–388. [Google Scholar] [CrossRef]
- Kroemer, G.; Zitvogel, L. Cancer immunotherapy in 2017: The breakthrough of the microbiota. Nat. Rev. Immunol. 2018, 18, 87–88. [Google Scholar] [CrossRef] [PubMed]
- Routy, B.; Gopalakrishnan, V.; Daillere, R.; Zitvogel, L.; Wargo, J.A.; Kroemer, G. The gut microbiota influences anticancer immunosurveillance and general health. Nat. Rev. Clin. Oncol. 2018, 15, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillere, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.R.; Jolly, C.A.; Chapkin, R.S. n-3 Polyunsaturated fatty acids exert immunomodulatory effects on lymphocytes by targeting plasma membrane molecular organization. Mol. Aspects. Med. 2012, 33, 46–54. [Google Scholar] [CrossRef]
- Fuentes, N.R.; Salinas, M.L.; Kim, E.; Chapkin, R.S. Emerging role of chemoprotective agents in the dynamic shaping of plasma membrane organization. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1668–1678. [Google Scholar] [CrossRef] [PubMed]
- Bartoszek, A.; Makaro, A.; Bartoszek, A.; Kordek, R.; Fichna, J.; Salaga, M. Walnut Oil Alleviates Intestinal Inflammation and Restores Intestinal Barrier Function in Mice. Nutrients 2020, 12. [Google Scholar] [CrossRef]
| Source | Compound or Fraction | Tumour Cell Type | ICD Pathway | Refs |
|---|---|---|---|---|
| Streptomyces peucetius (bacteria) | Doxorubicin | CT26 colon carcinoma cell line Pro-B melanoma murine cell line B16-F10 melanoma cell line | -DC maturation -Involvement of CD8+ T-cells | [74] |
| Streptomyces peucetius (bacteria) | Daunorubicin | AML acute myeloid leukemia cells | -CARL exposure -HSP70/HSP90 release -IFNγ release | [75] |
| Caesalpinia spinose (higher plant) | Gallotannin-rich Fraction | B16-F10 melanoma cell line A-375 melanoma cell line | -Activation of caspases 3 and 9 -ATP and HMGB1 | [76] |
| Plantago sp. (higher plant) | Linalool and p-coumaric | A549 lung carcinoma cell line T-47D breast cancer cell line SW620 colon adenocarcinoma cell line Hep G2 liver cancer c cell line | -Release of pro-inflammatory cytokines: IFN-γ, IL-13, IL-2, IL-21, IL-21R, IL-4, IL-6sR and TNF-α | [77] |
| Lithospermum erythrorhizon (higher plant) | Shikonin | B16-F10 melanoma cell line | -DC maturation -Differentiation stimuli for Th1 and Th17 cells | [78,79] |
| Capsicum sp. (higher plant) | Capsaicin | MDA-MB-231 breast cancer cell line MCF-7 breast cancer cell line 5637 urothelial bladder cancer cell line T24 bladder cancer cell line SD48 bladder cancer cell line BxPC-3 pancreatic cancer cell line AsPC-1 pancreatic cancer cell line SNU-1 gastric cancer cell line TMC-1 gastric cancer cell line SW480 colorectal cell line cancer cell line HCT-116 colorectal cancer cell line Primary effusion lymphoma cells | -ROS generation -Endoplasmic reticulum stress -CARL exposure -HSP70/HSP90 release -HMGB1 release -ATP | [80,81,82] |
| Digitalis sp. (higher plant) | Digoxin Digitoxin Lanatoside C Ouabain | U-2 OS osteosarcoma cell line and other tumour cells lines | -CARL exposure -ATP and HMGB1 release | [83,84,85] |
| Source | Compound or Fraction | Cell Type Target | ICD and Immune Activation | Refs |
|---|---|---|---|---|
| Alexandrium minutum (dinophyceae) | Glycopeptide | A549 Lung adenocarcinoma cell line | Mitophagy | [45] |
| Hematococcus pluvialis (green alga) | Astaxanthin | Primary lymphocytes | IFN-γ and IL-2 release | [89,90] |
| Thalassiosira rotula, Skeletonema costatum and Pseudonitzschia delicatissima (diatoms) | Polyunsaturated aldehydes (PUAs) | Caco-2Colon adenocarcinoma cell line A569 Lung adenocarcinoma cell line COLO 205 Colon adenocarcinoma cell line | Extrinsic apoptosis Necroptosis | [44,91] |
| Alexandrium tamarense (dinophyceae) | Acetonitrile/aqueous fraction (Glycolipids/phospholipids) | Human Peripheral Blood Mononuclear Cell (PBMC) | IL-6 release | [92,93] |
| Chaetoceros calcitrans (diatoms) | Aqueous fraction (amino acids/saccharides) | Human Peripheral Mononuclear Blood Cell (PBMC) | IL-6 release | [92,93] |
| Skeletonema costatum (diatoms) | Methanolic extract (Apolar compounds) Dichloromethane/ethanol fraction (Triglycerides) | Human Peripheral Mononuclear Blood Cell (PBMC) | IL-6 release | [92,93] |
| Dunaliella salina (green alga) | Methanol/aqueous fraction (nucleosides) | Human Peripheral Mononuclear Blood Cell (PBMC) | IL-6 release | [92,93] |
| Euglena gracilis (euglenophyceae) | Paramylon | Human Peripheral Blood Mononuclear Cell (PBMC) | IL-6 and TNF-α release | [94] |
| Gyrodinium impudicum (dinophyceae) | Exopolysaccharide, p-KG03 | Lymphocytes Natural killer (NK) | IFN-γ and IL-2 release | [95] |
| Skeletonema marinoi (diatoms) | Methanolic extract (Apolar compounds) | Human Peripheral Blood Mononuclear Cell (PBMC) | IL-6 release | [5] |
| Thalassiosira weissflogii (diatoms) | Glycolipids and Phospholipids | Human Peripheral Blood Mononuclear Cell (PBMC) | IL-6 release upregulation of MHC II, CD83, CD86, CD54 | [92,93] |
| Spirulina maxima (cyanophyceae) | Modified pectin (SmP) | Modulation of gut microbiota | Mucin, IFN-α, IL-6 release | [96] |
| Schizochytrium sp. (Labyrinthulea) | Polyunsaturated fatty acids | Modulation of gut microbiota | Lymphocytes target | [97] |
| Thraustochytriidae sp. (Labyrinthulea) | Exopolysaccharides | Antibodies production stimulation | B-cell proliferation | [98] |
| Tribonema sp. (Xanthophyceae) | Sulphated polysaccharides | Macrophages | Cytokines release (IL-6, IL-12) | [99] |
| Skeletonema dohrni (diatoms) | Methanol/aqueous fraction (nucleosides) | Human Peripheral Blood Mononuclear Cell (PBMC) | IL-6 release | [92,93] |
| Leptolyngbya (cyanophyceae) | Coibamide | U87-MG Human glioblastoma cells cell line | ULK phosphorylation, autophagy activation | [100] |
| Status Clinical Trials. Gov Identifier: | Study Title | Conditions | Interventions | Locations | |
|---|---|---|---|---|---|
| Peptidoglycan | Recruiting NCT04183478 | The Efficacy and Safety of K-001 in the Treatment of Advanced Pancreatic Cancer | Pancreatic Cancer | Drug: K-001 (K001 is peptidoglycan, prepared from the fermentation of the marine microorganism Spirulina) Other: Placebo | RenJiH Shanghai, Shanghai, China |
| Fucoidan | Recruiting NCT04066660 | Study of Oligo-Fucoidan in Advanced Hepatocellular Carcinoma (HCC) | Advanced Hepatocellular Carcinoma | Dietary Supplement: Oligo Fucoidan Dietary Supplement: Placebo | Fudan University Zhongshan Hospital Shanghai, China |
| Recruiting NCT04597476 | A Randomized, Double-blind Study to Evaluate the Clinical Effect and Safety of Fucoidan in Patients with Squamous Cell Carcinomas of the Head and Neck | Squamous Cell Carcinomas of the Head and Neck | Dietary Supplement: Fucoidan Other: Placebo (Potato starch) | National Taiwan University Hospital Taipei county, Taiwan | |
| Recruiting NCT04342949 | The Auxiliary Effects of Fucoidan for Locally Advanced Rectal Cancer Patients | To Observe Whether the Fucoidan Can Improve the Quality of Life of Such Patients Receiving the Neoadjuvant CCRT | Behavioural: Quality of life | Chung-Ho Memorial Hospital, Kaohsiung Medical University: Kaohsiung, Taiwan | |
| Product from Red Marine Algae | Recruiting NCT03869905 | Aquamin® as an Adjuvant Intervention for Ulcerative Colitis | Ulcerative Colitis | Drug: Aquamin® Drug: Placebo first then Aquamin® Aquamin®, a Multi-mineral Natural Product from Red Marine Algae, as an Adjuvant Intervention for Mild Ulcerative Colitis and Ulcerative Colitis in Remission | The University of Michigan Ann Arbor, Michigan, United States |
| AMR101 Marine oil | Active, not recruiting NCT04216251 | PRevention Using EPA Against coloREctal Cancer | Colorectal Adenoma Colorectal Cancer | Drug: AMR101 (VASCEPA, icosapent ethyl) | Massachusetts General Hospital Boston, Massachusetts, United States |
| Recruiting NCT03661047 | OMega-3 Fatty Acid for the Immune Modulation of Colorectal Cancer | Colon Cancer | Drug: AMR101 (VASCEPA, icosapent ethyl) | Massachusetts General Hospital Boston, Massachusetts, United States | |
| Marine oil | Active, not recruiting NCT04269876 | A Study to Evaluate the Effects of a Marine Lipid Oil Concentrate Formulation on Inflammation | Inflammation Inflammatory Response | Dietary Supplement: Marine Lipid Oil Concentrate Dietary Supplement: Dietary Supplement Dietary Supplement: Placebo | Lfie Extension Clinical Research, Inc. Fort Lauderdale, Florida, United States |
| Recruiting NCT04209244 | Effect of Fish Oil on Hyperlipidemia and Toxicities in Children and Young Adults with Acute Lymphoblastic Leukemia | Leukaemia, Acute Lymphoblastic | Dietary Supplement: Eskimo-3 Pure Fish Oil Dietary Supplement: Rapeseed Oil | Aalborg University Hospital Aarhus University Hospital Aarhus, Rigshospitalet Copenhagen, Odense University Hospital Denmark | |
| Completed Has Results NCT01661764 | Fish Oil Supplementation, Nutrigenomics and Colorectal Cancer Prevention | Colorectal Adenomatous Polyps | Drug: Eicosapentanoic acid and docosahexanoic acid Drug: Oleic Acid | Vanderbilt University Medical Center Nashville, Tennessee, United States | |
| Completed Has Results NCT01813110 | Effects of a Prescription Omega-3 Fatty Acid Concentrate on Induced Inflammation | Inflammatory Responses | Drug: 4 g prescription omega-3 concentrate Drug: Placebo | Penn State University Pennsylvania United States | |
| VITAL: Marine oil and Vitamin D | Unknown † NCT02239874 † Study has passed its completion date and status has not been verified in more than two years | VITamin D and OmegA-3 TriaL: Effects on Mammographic Density and Breast Tissue | Benign Breast Disease | Dietary Supplement: Vitamin D and fish oil placebo Dietary Supplement: Fish oil and vitamin D placebo Dietary Supplement: Vitamin D placebo and fish oil placebo Dietary Supplement: Vitamin D and fish oil | Brigham and Women’s Hospital Boston, Massachusetts, United States |
| Active, not recruiting Has Results NCT01169259 | Vitamin D and Omega-3 Trial (VITAL) | Cancer Cardiovascular Disease | Dietary Supplement: Vitamin D3 Drug: Omega-3 fatty acids (fish oil) Dietary Supplement: Vitamin D3 placebo Dietary Supplement: Fish oil placebo | Brigham and Women’s Hospital Boston, Massachusetts, United States | |
| Active, not recruiting NCT04386577 | Effects of Vitamin D and Omega-3 Supplementation on Telomeres in VITAL | Aging | Dietary Supplement: Vitamin D3 (cholecalciferol) Drug: Fish oil | Georgia Prevention Institute Augusta, Georgia, United States | |
| Trabectedin | Recruiting NCT03886311 | Talimogene Laherparepvec, Nivolumab and Trabectedin for Sarcoma | Sarcoma | Drug: Talimogene Laherparepvec 100000000 PFU/1 ML Injection Suspension [IMLYGIC] Drug: Nivolumab IV Soln 100 MG/10 ML Drug: Trabectedin 0.25 MG/1 VIAL Intravenous Powder for Solution | Sarcoma Oncology Center Santa Monica, California, United States |
| Completed NCT02249702 | Activity of Trabectedin or Gemcitabine + Docetaxel in Uterine Leiomyosarcoma | Leiomyo sarcoma | Drug: Gemcitabine + docetaxel Drug: Trabectedin | Centro di Riferimento Oncologico Aviano, Pordenone, Italy 33 institutes | |
| Eribulin mesylate | Terminated NCT01534455 | Efficacy and Tolerability of Eribulin Plus Lapatinib in Patients with Metastatic Breast Cancer (E-VITA) | Metastatic Breast Cancer | Drug: Lapatinib + 1.23 mg Eribulin Drug: Lapatinib + 1.76 mg Eribulin | Klinikum der Otto-v.-Guericke-Universität Frauenklinik Magdeburg, Germany |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sansone, C.; Bruno, A.; Piscitelli, C.; Baci, D.; Fontana, A.; Brunet, C.; Noonan, D.M.; Albini, A. Natural Compounds of Marine Origin as Inducers of Immunogenic Cell Death (ICD): Potential Role for Cancer Interception and Therapy. Cells 2021, 10, 231. https://doi.org/10.3390/cells10020231
Sansone C, Bruno A, Piscitelli C, Baci D, Fontana A, Brunet C, Noonan DM, Albini A. Natural Compounds of Marine Origin as Inducers of Immunogenic Cell Death (ICD): Potential Role for Cancer Interception and Therapy. Cells. 2021; 10(2):231. https://doi.org/10.3390/cells10020231
Chicago/Turabian StyleSansone, Clementina, Antonino Bruno, Concetta Piscitelli, Denisa Baci, Angelo Fontana, Christophe Brunet, Douglas M. Noonan, and Adriana Albini. 2021. "Natural Compounds of Marine Origin as Inducers of Immunogenic Cell Death (ICD): Potential Role for Cancer Interception and Therapy" Cells 10, no. 2: 231. https://doi.org/10.3390/cells10020231
APA StyleSansone, C., Bruno, A., Piscitelli, C., Baci, D., Fontana, A., Brunet, C., Noonan, D. M., & Albini, A. (2021). Natural Compounds of Marine Origin as Inducers of Immunogenic Cell Death (ICD): Potential Role for Cancer Interception and Therapy. Cells, 10(2), 231. https://doi.org/10.3390/cells10020231









