A New Player in Jasmonate-Mediated Stomatal Closure: The Arabidopsis thaliana Copper Amine Oxidase β
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials, Growth Conditions and Treatments
2.2. Identification of the T-DNA Insertional Loss-of-Function Atcuaoβ.3 Mutant
2.3. RNA Extraction, RT-PCR and RT-Quantitative PCR (RT-qPCR)
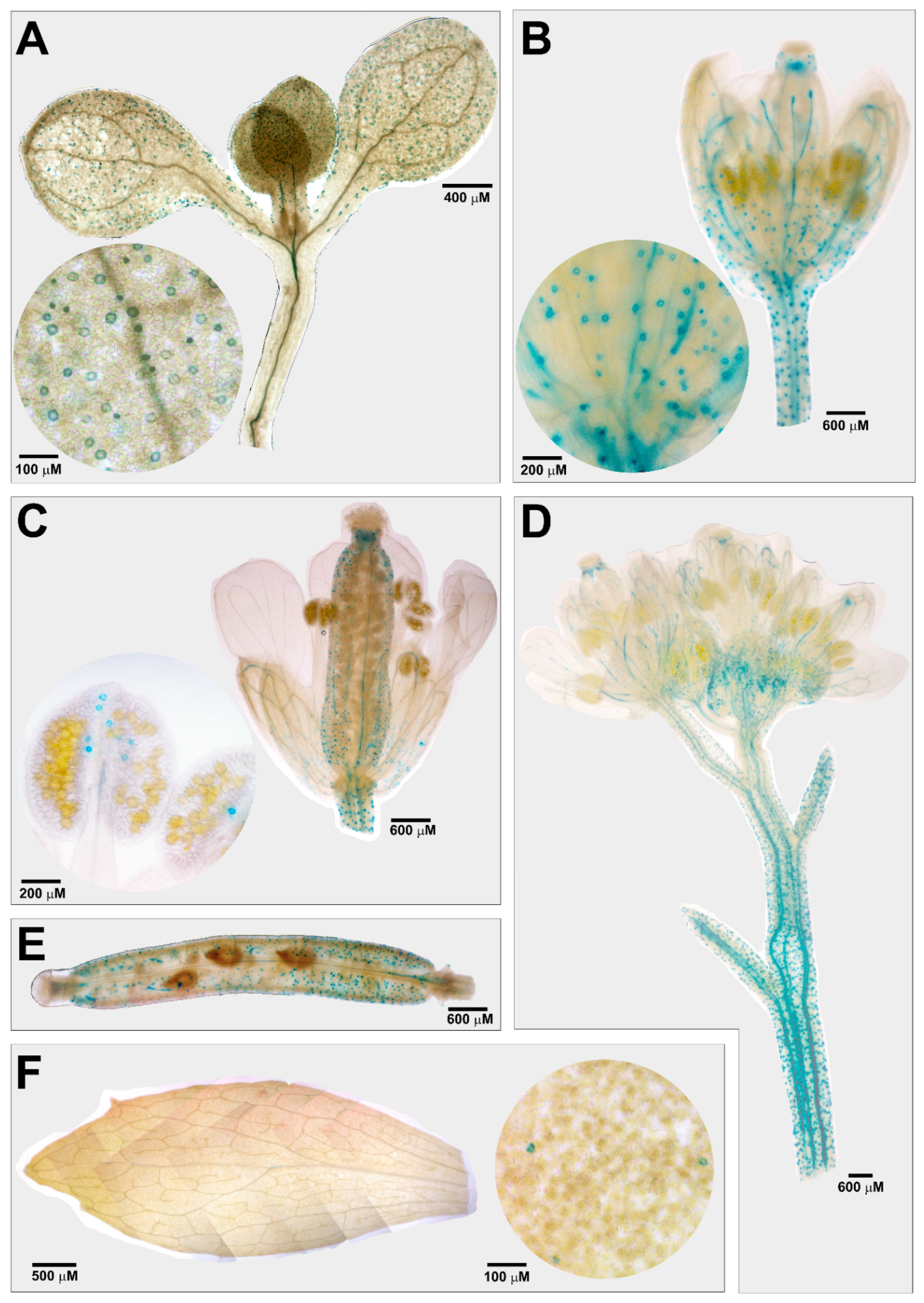
2.4. Histochemical Analysis of GUS Assay
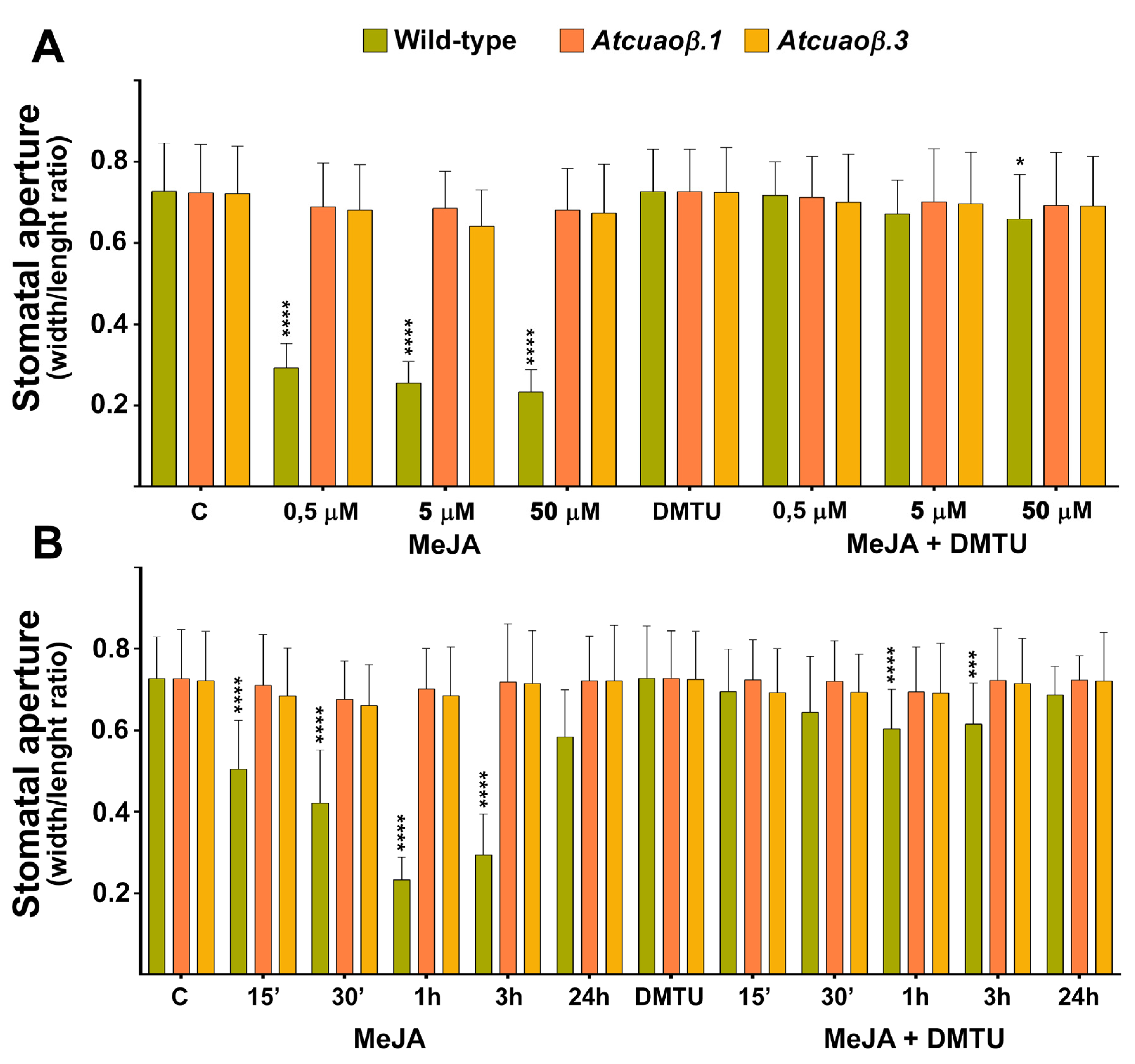
2.5. Measurement of Stomatal Aperture
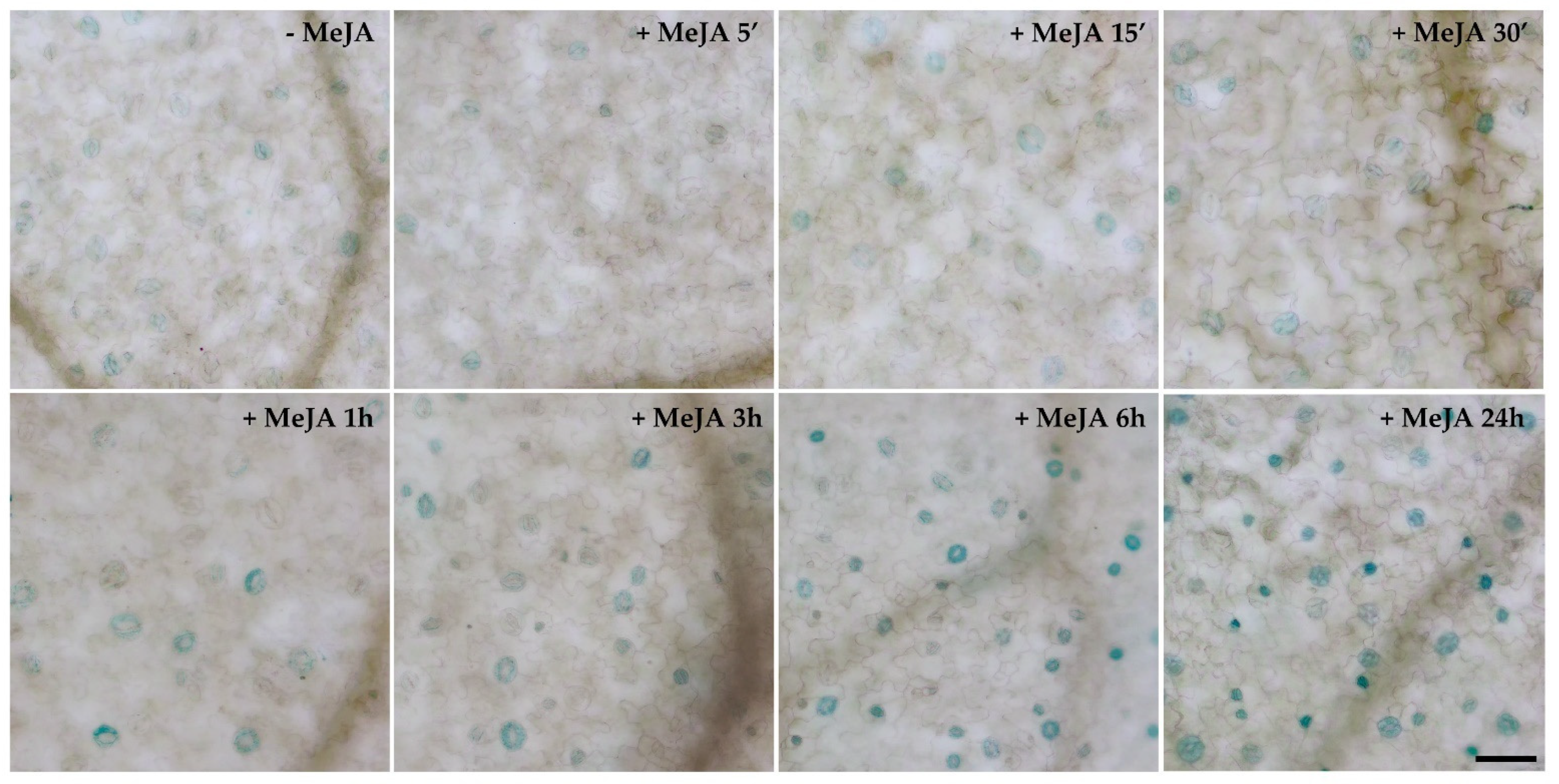
2.6. In Situ Detection of Reactive Oxygen Species (ROS) in Guard Cells
2.7. Statistics
3. Results
3.1. Tissue-Specific Expression Profile of AtCuAOβ in Stomata Guard Cells
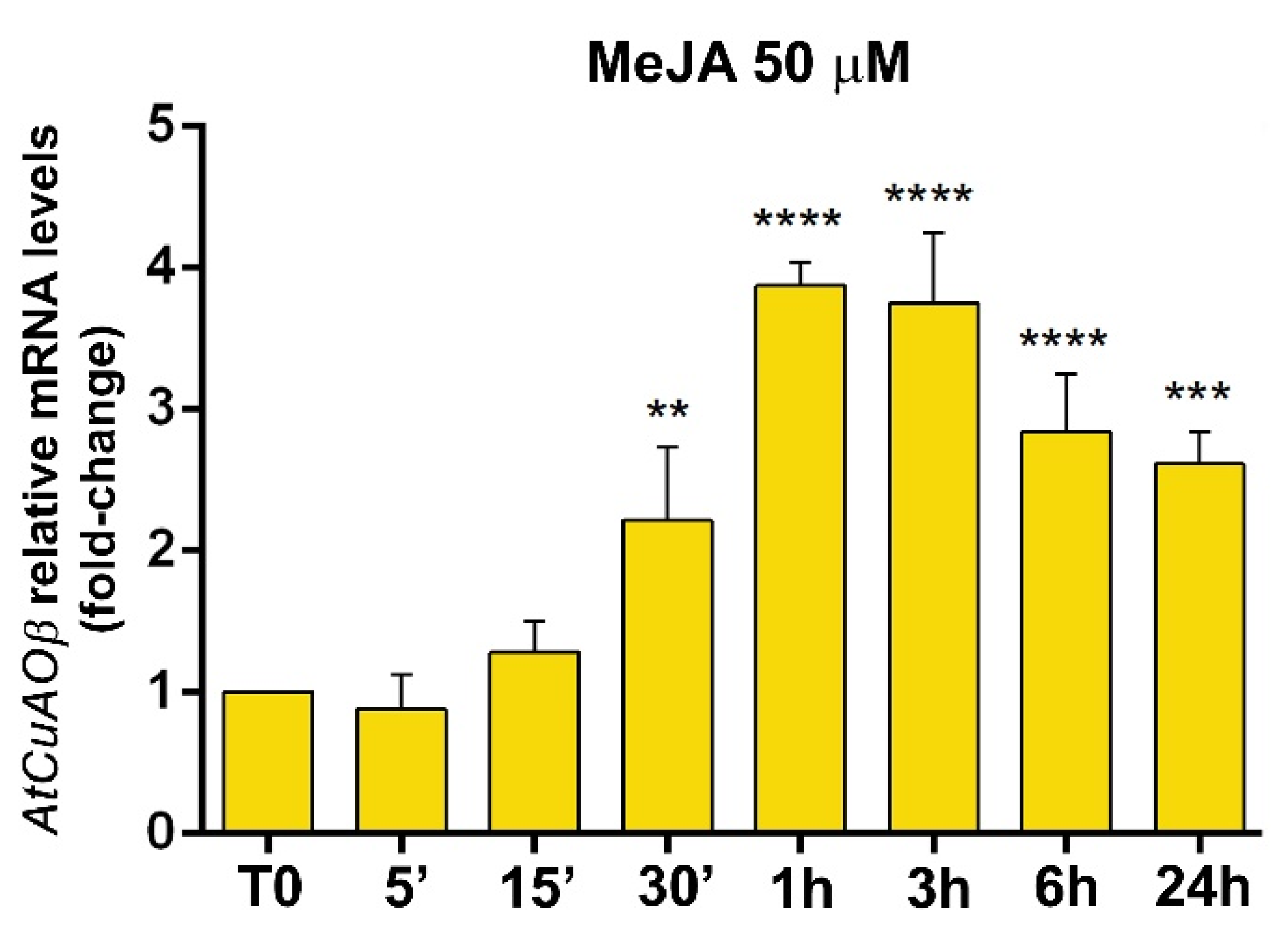
3.2. Expression Profile of AtCuAOβ after MeJA Treatment
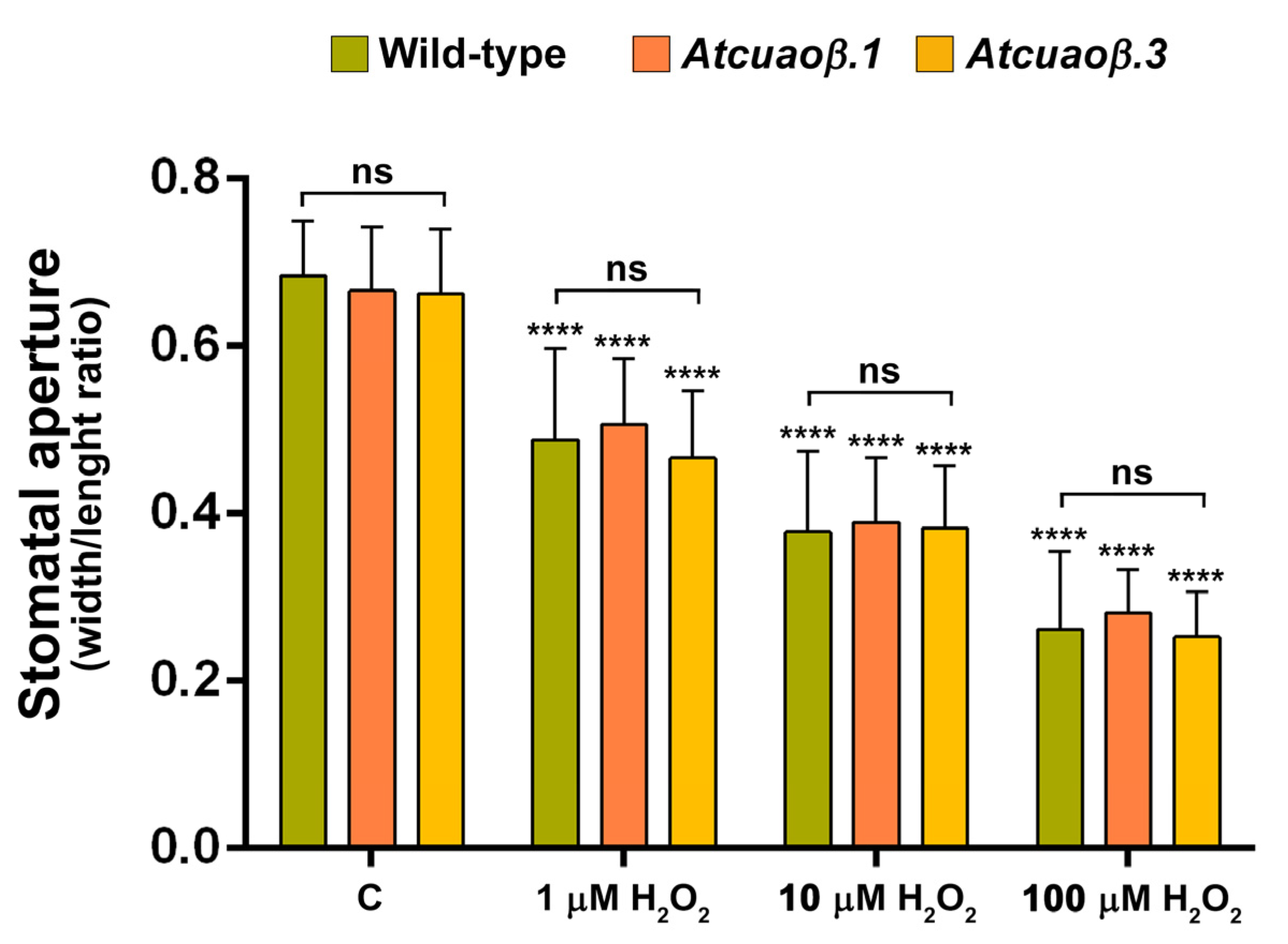
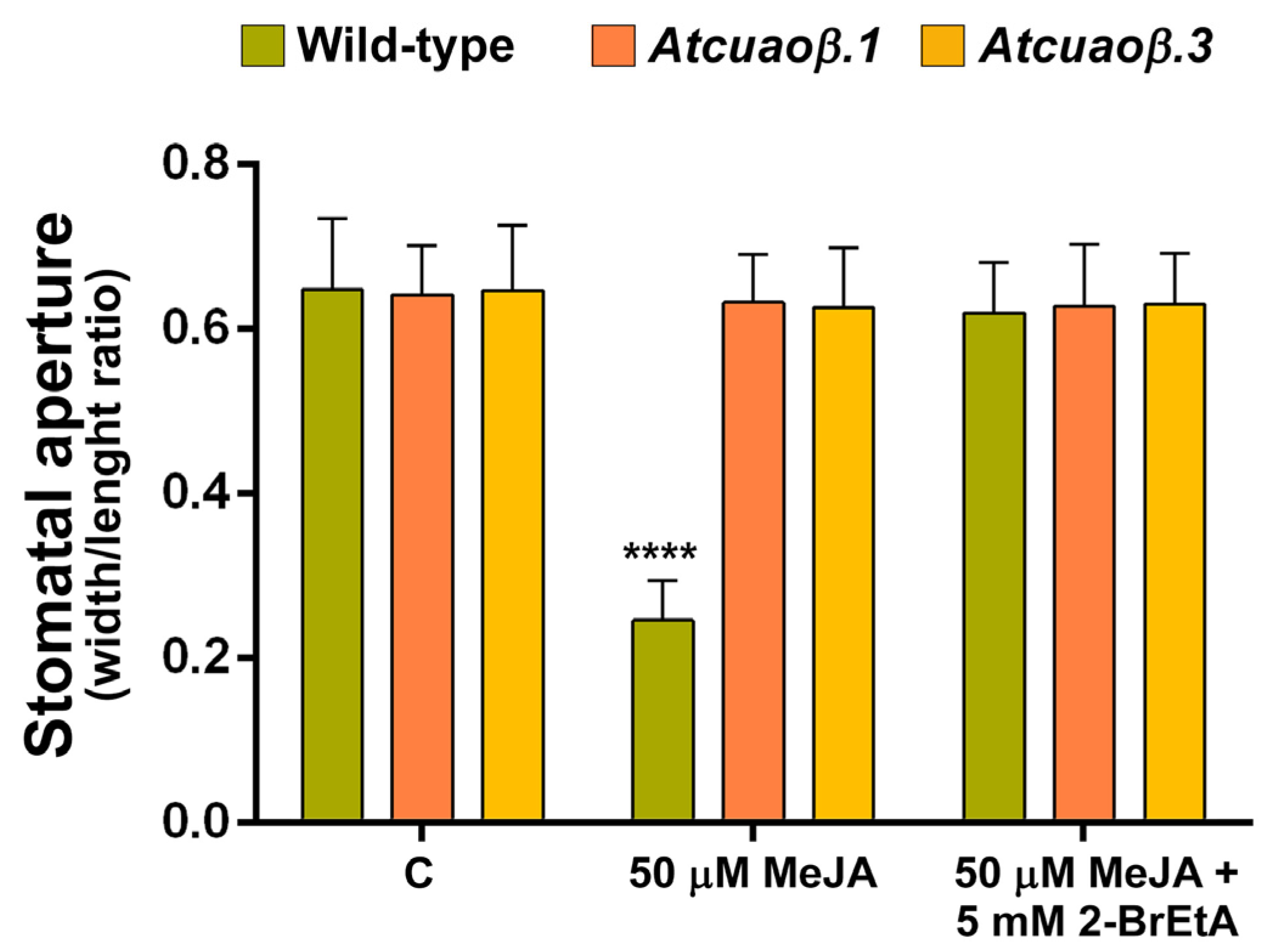
3.3. AtCuAOβ-Dependent H2O2 Production Is Involved in MeJA-Induced Stomatal Closure
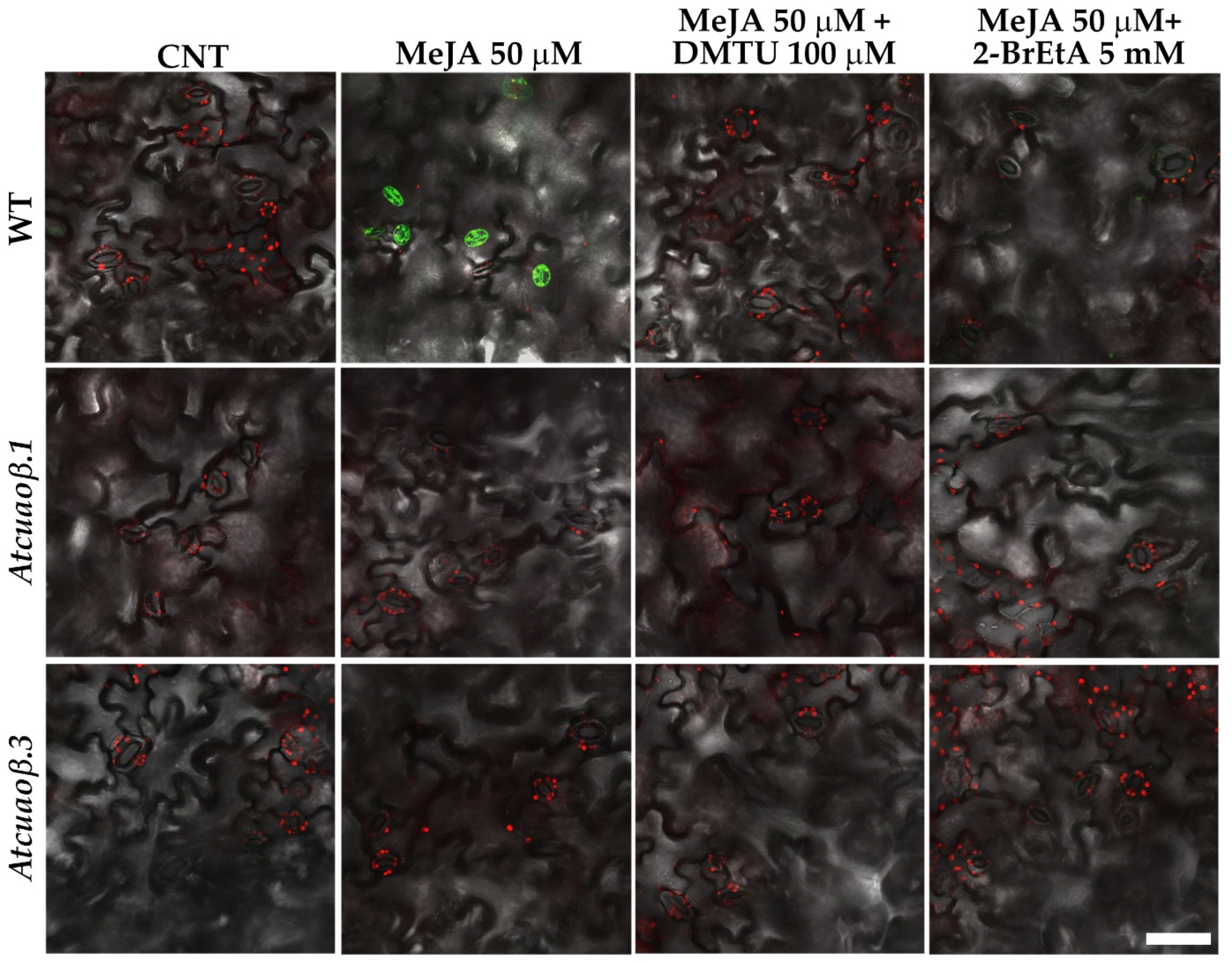
3.4. Stomatal Closure Induced by MeJA Is Associated with a ROS Level Increase in Guard Cells Mediated by AtCuAOβ
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Ollas, C.; Dodd, I.C. Physiological impacts of ABA-JA interactions under water-limitation. Plant Mol. Biol. 2016, 91, 641–650. [Google Scholar] [CrossRef]
- Tavladoraki, P.; Cona, A.; Angelini, R. Copper-containing amine oxidases and FAD-dependent polyamine oxidases are key players in plant tissue differentiation and organ development. Front. Plant Sci. 2016, 7, 824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qu, Y.; An, Z.; Zhuang, B.; Jing, W.; Zhang, Q.; Zhang, W. Copper amine oxidase and phospholipase D act independently in abscisic acid (ABA)-induced stomatal closure in Vicia faba and Arabidopsis. J. Plant Res. 2014, 127, 533–544. [Google Scholar] [CrossRef]
- Fraudentali, I.; Ghuge, S.A.; Carucci, A.; Tavladoraki, P.; Angelini, R.; Cona, A.; Rodrigues-Pousada, R.A. The Copper Amine Oxidase Atcuaoδ participates in Abscisic Acid-Induced Stomatal Closure in Arabidopsis. Plants 2019, 8, 183. [Google Scholar] [CrossRef] [Green Version]
- An, Z.; Jing, W.; Liu, Y.; Zhang, W. Hydrogen peroxide generated by copper amine oxidase is involved in abscisic acid-induced stomatal closure in Vicia faba. J. Exp. Bot. 2008, 59, 815–825. [Google Scholar] [CrossRef] [Green Version]
- Paschalidis, K.A.; Toumi, I.; Moschou, P.N.; Roubelakis-Angelakis, K.A. ABA-dependent amine oxidases-derived H2O2 affects stomata conductance. Plant Signal Behav. 2010, 5, 1153–1156. [Google Scholar]
- Hou, Z.H.; Liu, G.H.; Wang, L.X.; Liu, X. Regulatory function of polyamine oxidase-generated hydrogen peroxide in ethylene-induced stomatal closure in Arabidopsis thaliana. J. Integr. Agricult. 2013, 12, 251–262. [Google Scholar] [CrossRef]
- Ghuge, S.A.; Carucci, A.; Rodrigues-Pousada, R.A.; Tisi, A.; Franchi, S.; Tavladoraki, P.; Angelini, R.; Cona, A. The MeJA-inducible copper amine oxidase AtAO1 is expressed in xylem tissue and guard cells. Plant Signal. Behav. 2015, 10, e1073872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghuge, S.A.; Carucci, A.; Rodrigues-Pousada, R.A.; Tisi, A.; Franchi, S.; Tavladoraki, P.; Angelini, R.; Cona, A. The apoplastic copper AMINE OXIDASE1 mediates jasmonic acid-induced protoxylem differentiation in Arabidopsis roots. Plant Physiol. 2015, 168, 690–707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fraudentali, I.; Rodrigues-Pousada, R.A.; Angelini, R.; Ghuge, S.A.; Cona, A. Plant Copper Amine Oxidases: Key Players in Hormone Signaling Leading to Stress-Induced Phenotypic Plasticity. Int. J. Mol. Sci. 2021, 22, 5136. [Google Scholar] [CrossRef]
- Valvekens, D.; Van Montagu, M.; Van Lijsebettens, M. Agrobacterium tumefaciens-mediated transformation of Arabidopsis thaliana root explants by using kanamycin selection. Proc. Natl. Acad. Sci. USA 1988, 85, 5536–5540. [Google Scholar] [CrossRef] [Green Version]
- Fraudentali, I.; Ghuge, S.A.; Carucci, A.; Tavladoraki, P.; Angelini, R.; Rodrigues-Pousada, R.A.; Cona, A. Developmental, hormone- and stress-modulated expression profiles of four members of the Arabidopsis copper-amine oxidase gene family. Plant Physiol and Biochem. 2020, 147, 141–160. [Google Scholar] [CrossRef]
- Klimyuk, V.I.; Carroll, B.J.; Thomas, C.M.; Jones, J.D. Alkali treatment for rapid preparation of plant material for reliable PCR analysis. Plant J. 1993, 3, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Czechowski, T.; Stitt, M.; Altmann, T.; Udvardi, M.K. Genome-Wide Identification and Testing of Superior Reference Genes for Transcript Normalization. Plant Physiol. 2005, 139, 5–17. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Jefferson, R.A. Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol. Biol. 1987, 5, 387–405. [Google Scholar] [CrossRef]
- Jung, C.; Seo, J.S.; Han, S.W.; Koo, Y.J.; Kim, C.H.; Song, S.I.; Nahm, B.H.; Choi, Y.D.; Cheong, J.J. Overexpression of AtMYB44 enhances stomatal closure to confer abiotic stress tolerance in transgenic Arabidopsis. Plant Physiol. 2008, 146, 623–635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cona, A.; Rea, G.; Angelini, R.; Federico, R.; Tavladoraki, P. Functions of amine oxidases in plant development and defence. Trends Plant Sci. 2006, 11, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Angelini, R.; Tisi, A.; Rea, G.; Chen, M.M.; Botta, M.; Federico, R.; Cona, A. Involvement of polyamine oxidase in wound healing. Plant Physiol. 2008, 146, 162–177. [Google Scholar] [CrossRef] [Green Version]
- Devireddy, A.R.; Zandalinas, S.I.; Fichman, Y.; Mittler, R. Integration of reactive oxygen species and hormone signaling during abiotic stress. Plant J. 2021, 105, 459–476. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.S. Ultrastructure of bacterial penetration in plants. Ann. Rev. Phytopathol. 1986, 24, 141157. [Google Scholar] [CrossRef]
- Melotto, M.; Underwood, W.; He, S.Y. Role of stomata in plant innate immunity and foliar bacterial diseases. Annu. Rev. Phytopathol. 2008, 46, 101–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haworth, M.; Marino, G.; Cosentino, S.L.; Brunetti, C.; De Carlo, A.; Avola, G. Increased free abscisic acid during drought enhances stomatal sensitivity and modifies stomatal behaviour in fast growing giant reed (Arundodonax L.). Environ. Exp. Bot. 2018, 147, 116–124. [Google Scholar] [CrossRef]
- Bharath, P.; Gahir, S.; Raghavendra, A.S. Abscisic Acid-Induced Stomatal Closure: An Important Component of Plant Defense Against Abiotic and Biotic Stress. Front Plant Sci. 2021, 12, 615114. [Google Scholar] [CrossRef] [PubMed]
- Förster, S.; Schmidt, L.K.; Kopic, E.; Anschütz, U.; Huang, S.; Schlücking, K.; Köster, P.; Waadt, R.; Larrieu, A.; Batistič, O.; et al. Wounding-Induced Stomatal Closure Requires Jasmonate-Mediated Activation of GORK K+ Channels by a Ca2+ Sensor-Kinase CBL1-CIPK5 Complex. Dev. Cell 2019, 48, 87–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geng, S.; Misra, B.B.; de Armas, E.; Huhman, D.V.; Alborn, H.T.; Sumner, L.W.; Chen, S. Jasmonate-mediated stomatal closure under elevated CO2 revealed by time-resolved metabolomics. Plant J. 2016, 88, 947–962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, W.; Melotto, M.; He, S.Y. Plant stomata: A checkpoint of host immunity and pathogen virulence. Curr. Opin. Biotechnol. 2010, 21, 599–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, P.A.; Chen, Y.; Ponce, G.; Acevedo, F.E.; Lynch, J.P.; Anderson, C.T.; Ali, J.G.; Felton, G.W. Stomata-mediated interactions between plants, herbivores, and the environment. Trends Plant Sci. 2021, 24, 1360–1385. [Google Scholar] [CrossRef] [PubMed]
- Fraudentali, I.; Rodrigues-Pousada, R.A.; Tavladoraki, P.; Angelini, R.; Cona, A. Leaf-Wounding Long-Distance Signaling Targets AtCuAOβ Leading to Root Phenotypic Plasticity. Plants 2020, 9, 249. [Google Scholar] [CrossRef] [Green Version]
- Devireddy, A.R.; Arbogast, J.; Mittler, R. Coordinated and rapid whole-plant systemic stomatal responses. New Phytol. 2020, 225, 21–25. [Google Scholar] [CrossRef] [Green Version]
- Yin, Y.; Adachi, Y.; Nakamura, Y.; Munemasa, S.; Mori, I.C.; Murata, Y. Involvement of OST1 Protein Kinase and PYR/PYL/RCAR Receptors in Methyl Jasmonate-Induced Stomatal Closure in Arabidopsis Guard Cells. Plant Cell Physiol. 2016, 57, 1779–1790. [Google Scholar] [CrossRef] [PubMed]
- Andronis, E.A.; Moschou, P.N.; Toumi, I.; Roubelakis-Angelakis, K.A. Peroxisomal polyamine oxidase and NADPH-oxidase cross-talk for ROS homeostasis which affects respiration rate in Arabidopsis thaliana. Front. Plant Sci. 2014, 5, 132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Name of Primer | Sequence of Primer |
|---|---|
| LBa1 | 5′-GATGGTTCACGTAGTGGGCCATCGC-3′ |
| RP-Atcuaoβ.3 | 5′-ATCACTATAAAACCCACCGGC-3′ |
| LP-Atcuaoβ.3 | 5′-ACGTTCATGGACATTGGAGAG-3′ |
| Name of Primer | Sequence of Primer |
|---|---|
| UBC21-for | 5′-CTGCGACTCAGGGAATCTTCTAA-3′ |
| UBC21-rev | 5′-TTGTGCCATTGAATTGAACCC-3′ |
| AtCuAOβ-qPCR-for | 5′-CAAGTGGGGAAGCTGAAATAAGTTTAGTG-3′ |
| AtCuAOβ-qPCR-rev | 5′-TCCTCCGAGAAGACGTTTTGTTAACTTC-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fraudentali, I.; Pedalino, C.; Tavladoraki, P.; Angelini, R.; Cona, A. A New Player in Jasmonate-Mediated Stomatal Closure: The Arabidopsis thaliana Copper Amine Oxidase β. Cells 2021, 10, 3399. https://doi.org/10.3390/cells10123399
Fraudentali I, Pedalino C, Tavladoraki P, Angelini R, Cona A. A New Player in Jasmonate-Mediated Stomatal Closure: The Arabidopsis thaliana Copper Amine Oxidase β. Cells. 2021; 10(12):3399. https://doi.org/10.3390/cells10123399
Chicago/Turabian StyleFraudentali, Ilaria, Chiara Pedalino, Paraskevi Tavladoraki, Riccardo Angelini, and Alessandra Cona. 2021. "A New Player in Jasmonate-Mediated Stomatal Closure: The Arabidopsis thaliana Copper Amine Oxidase β" Cells 10, no. 12: 3399. https://doi.org/10.3390/cells10123399
APA StyleFraudentali, I., Pedalino, C., Tavladoraki, P., Angelini, R., & Cona, A. (2021). A New Player in Jasmonate-Mediated Stomatal Closure: The Arabidopsis thaliana Copper Amine Oxidase β. Cells, 10(12), 3399. https://doi.org/10.3390/cells10123399







