Oncofetal Protein CRIPTO Is Involved in Wound Healing and Fibrogenesis in the Regenerating Liver and Is Associated with the Initial Stages of Cardiac Fibrosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Controls
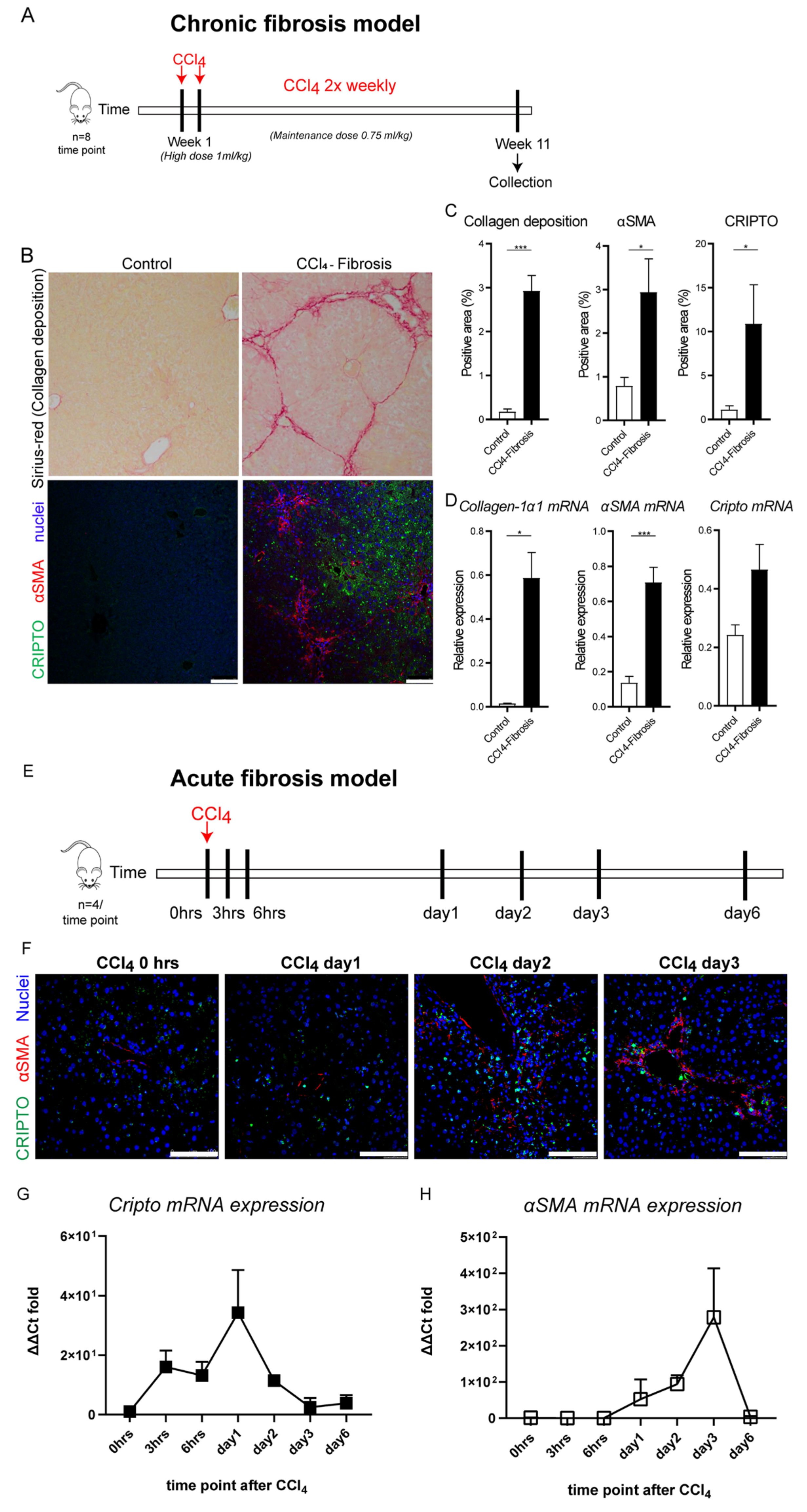
2.2. Mouse Models of Fibrosis
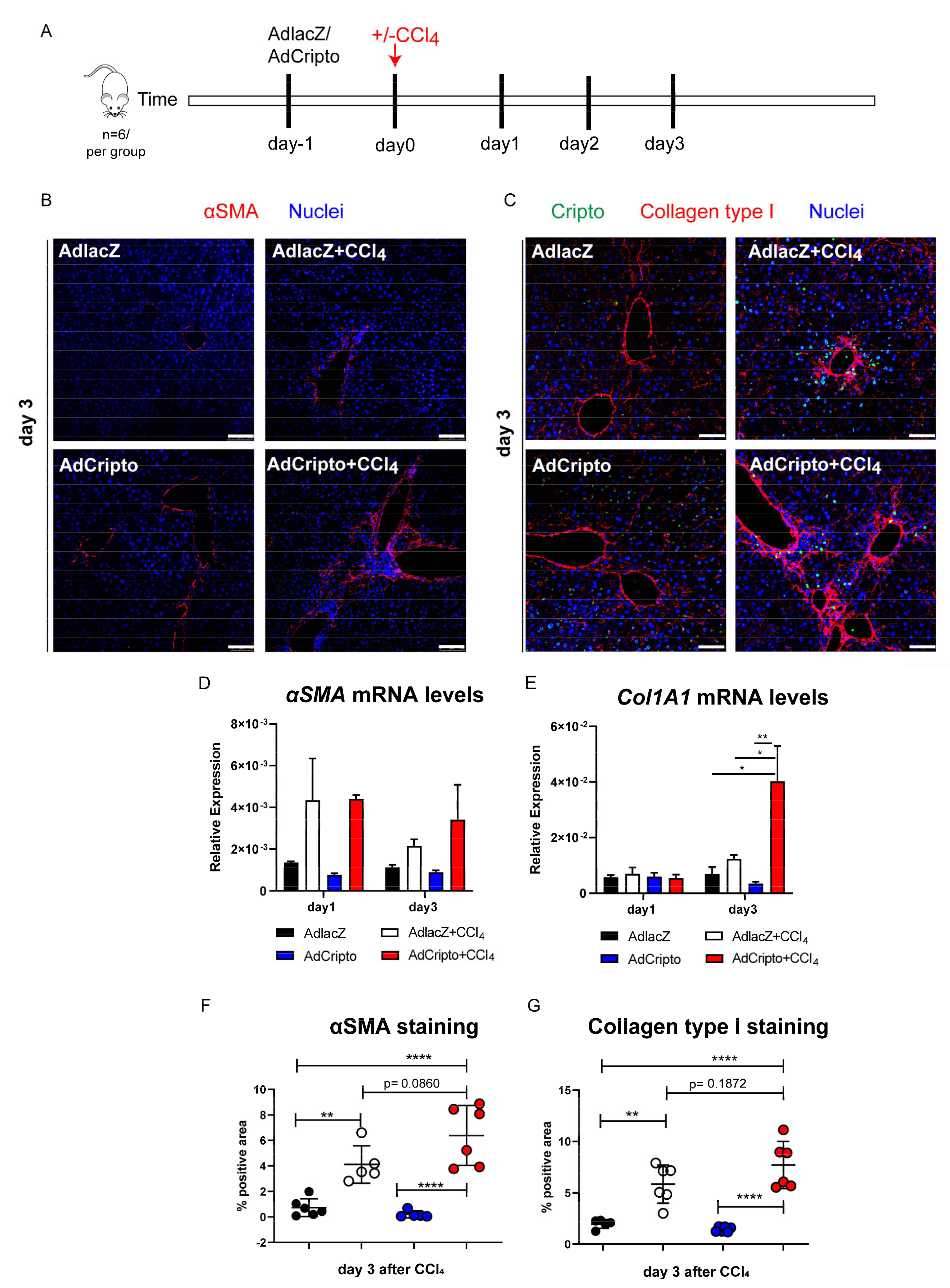
2.3. In Vivo Administration of Adenovirus
2.4. Preparation of ALK4Fc
2.5. Histological Examination of Fibrosis
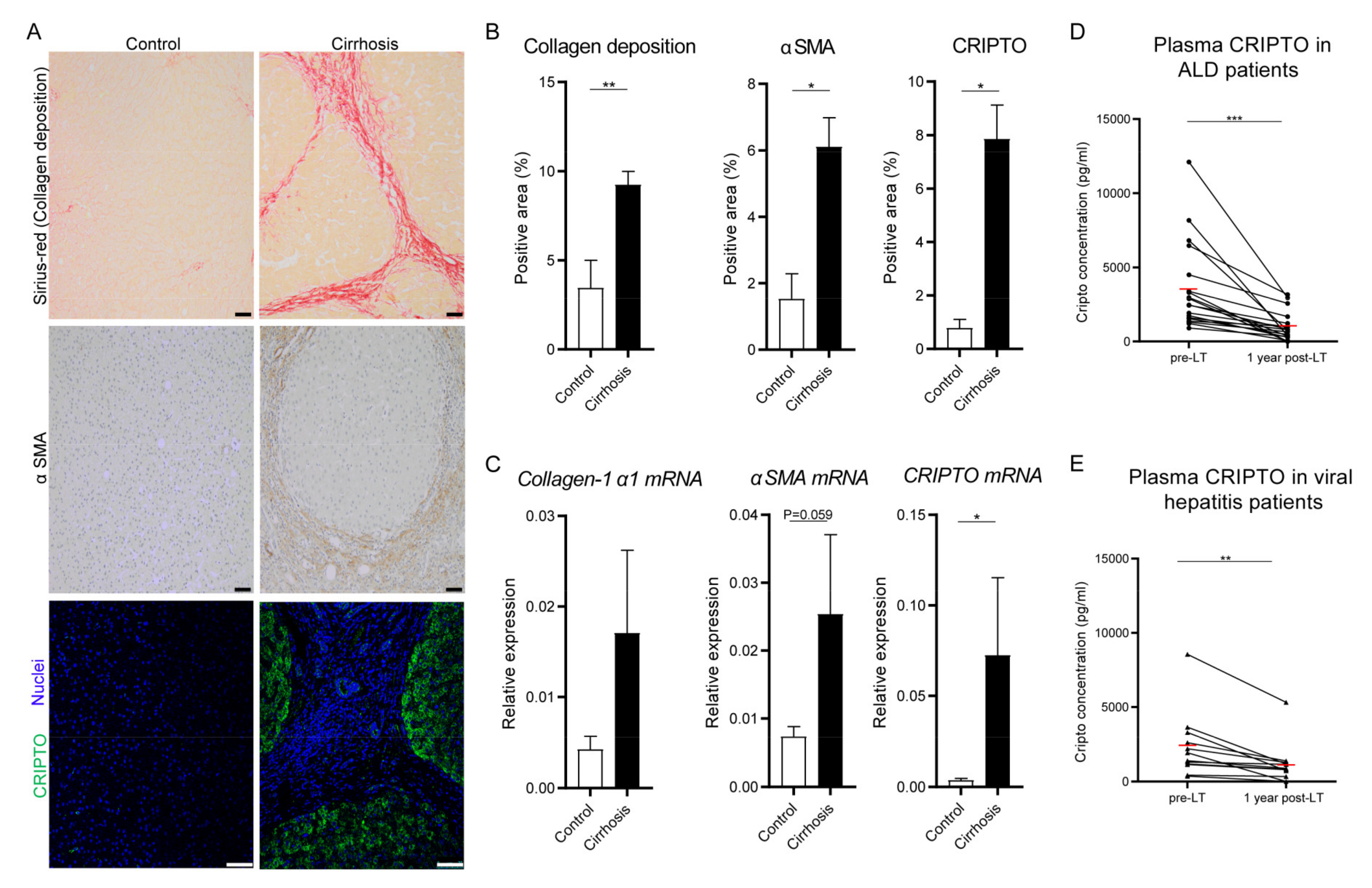
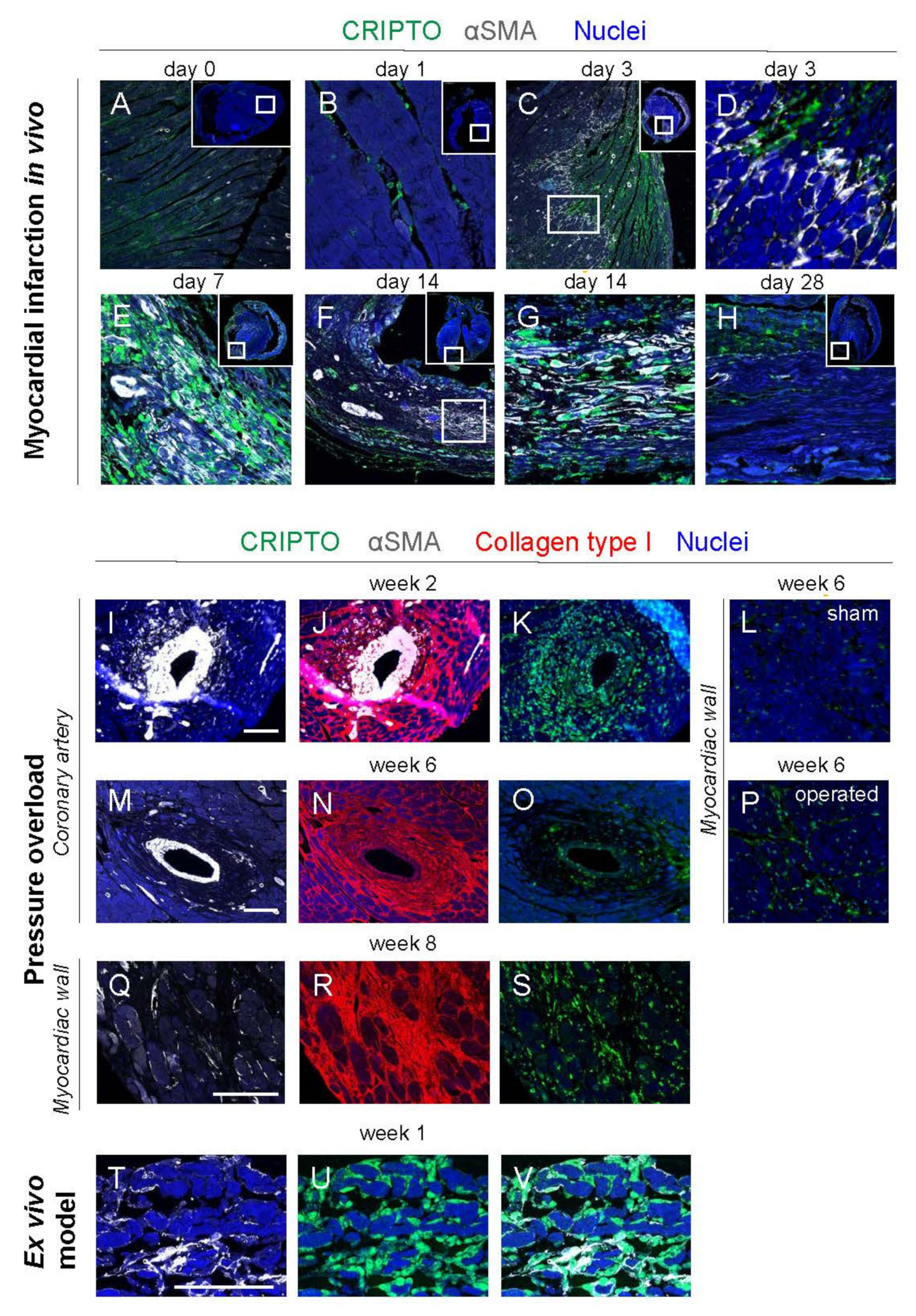
2.6. Immunohistochemistry, Immunofluorescence Imaging, and Quantification
2.7. RNA Isolation, cDNA Synthesis, and Quantitative Polymerase Chain Reaction (qPCR)
2.8. Cell Culture and Reporter Assay
2.9. Western Immunoblotting
2.10. Human Plasma CRIPTO Measurements
2.11. Statistical Analysis
3. Results
3.1. Cripto Expression in Patients with End-Stage Liver Cirrhosis
3.2. CRIPTO Expression in Patients with End-Stage Heart Failure
3.3. CRIPTO Expression in Mouse Models of Liver Fibrosis
3.4. CRIPTO Expression in Mouse Models of Cardiac Fibrosis
3.5. Adenoviral-Mediated in Vivo Overexpression of CRIPTO Leads to Increased Fibrogenesis
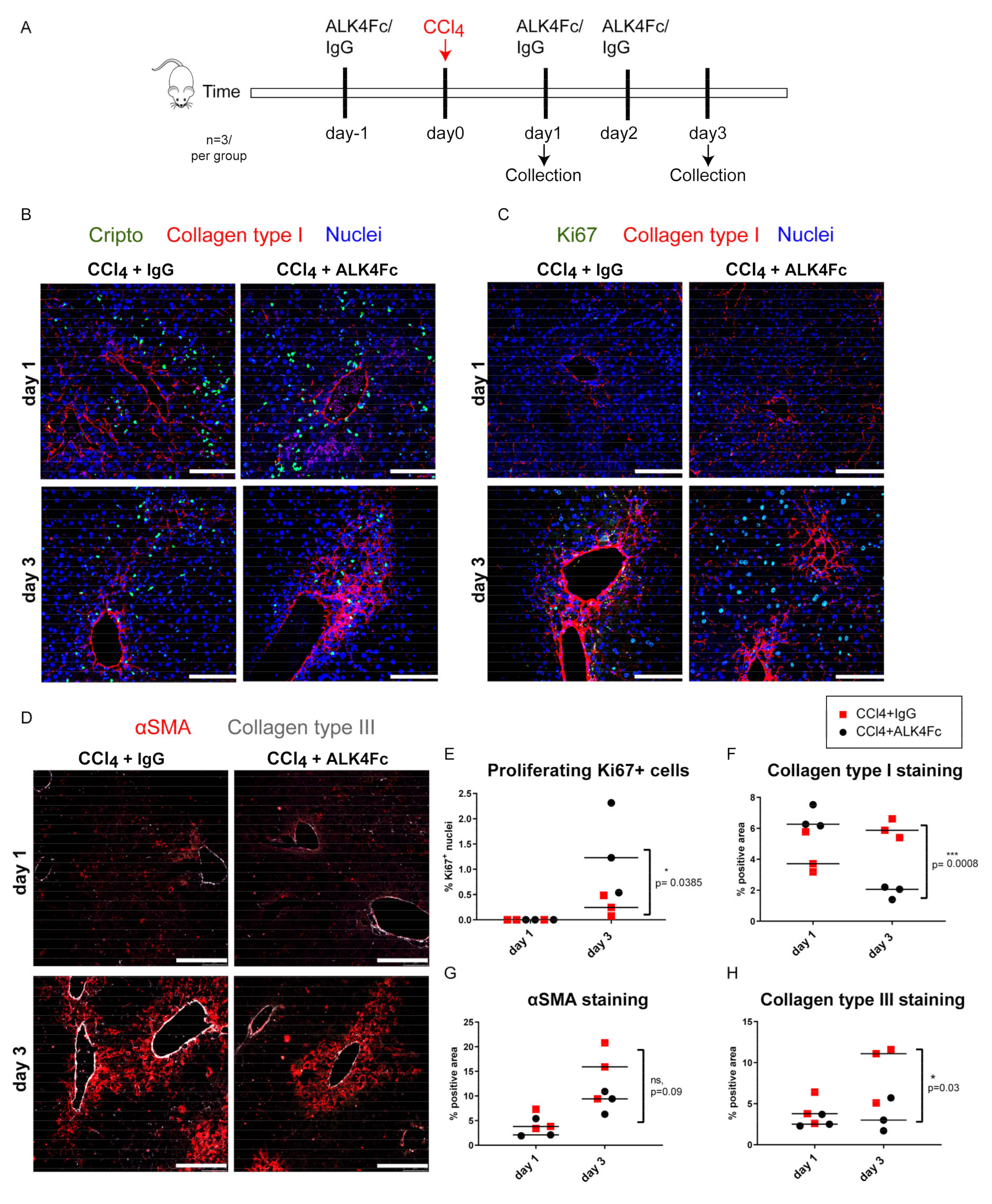
3.6. Functional Inhibition of CRIPTO by ALK4Fc Ligand Trap Reduces Collagen Deposition and Stimulates Hepatocyte Proliferation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nat. Med. 2012, 18, 1028–1040. [Google Scholar] [CrossRef]
- Karkampouna, S.; Ten Dijke, P.; Dooley, S.; Julio, M.K. Tgfbeta signaling in liver regeneration. Curr. Pharm. Des. 2012, 18, 4103–4113. [Google Scholar] [CrossRef]
- Travers, J.G.; Kamal, F.A.; Robbins, J.; Yutzey, K.E.; Blaxall, B.C. Cardiac fibrosis: The fibroblast awakens. Circ. Res. 2016, 118, 1021–1040. [Google Scholar] [CrossRef] [PubMed]
- Cox, T.R.; Erler, J.T. Remodeling and homeostasis of the extracellular matrix: Implications for fibrotic diseases and cancer. Dis. Models Mech. 2011, 4, 165–178. [Google Scholar] [CrossRef]
- Klingberg, F.; Hinz, B.; White, E.S. The myofibroblast matrix: Implications for tissue repair and fibrosis. J. Pathol. 2013, 229, 298–309. [Google Scholar] [CrossRef]
- Van den Borne, S.W.; Diez, J.; Blankesteijn, W.M.; Verjans, J.; Hofstra, L.; Narula, J. Myocardial remodeling after infarction: The role of myofibroblasts. Nat. Rev. Cardiol. 2010, 7, 30–37. [Google Scholar] [CrossRef]
- Distler, J.H.W.; Györfi, A.-H.; Ramanujam, M.; Whitfield, M.L.; Königshoff, M.; Lafyatis, R. Shared and distinct mechanisms of fibrosis. Nat. Rev. Rheumatol. 2019, 15, 705–730. [Google Scholar] [CrossRef]
- Kanisicak, O.; Khalil, H.; Ivey, M.J.; Karch, J.; Maliken, B.D.; Correll, R.N.; Brody, M.J.; SC, J.L.; Aronow, B.J.; Tallquist, M.D.; et al. Genetic lineage tracing defines myofibroblast origin and function in the injured heart. Nat. Commun 2016, 7, 12260. [Google Scholar] [CrossRef]
- Moore-Morris, T.; Guimaraes-Camboa, N.; Banerjee, I.; Zambon, A.C.; Kisseleva, T.; Velayoudon, A.; Stallcup, W.B.; Gu, Y.; Dalton, N.D.; Cedenilla, M.; et al. Resident fibroblast lineages mediate pressure overload-induced cardiac fibrosis. J. Clin. Investig. 2014, 124, 2921–2934. [Google Scholar] [CrossRef]
- El–Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.M.; Kim, W.R. Epidemiology and healthcare burden of acute-on-chronic liver failure. Semin. Liver Dis. 2016, 36, 123–126. [Google Scholar] [CrossRef]
- Friedman, S.L. Mechanisms of hepatic fibrogenesis. Gastroenterology 2008, 134, 1655–1669. [Google Scholar] [CrossRef] [PubMed]
- Bataller, R.; Brenner, D.A. Liver fibrosis. J. Clin. Investig. 2005, 115, 209–218. [Google Scholar] [CrossRef]
- Lee, Y.A.; Wallace, M.C.; Friedman, S.L. Pathobiology of liver fibrosis—A translational success story. Gut 2015, 64, 1337. [Google Scholar] [CrossRef] [PubMed]
- Frangogiannis, N.G. The extracellular matrix in ischemic and nonischemic heart failure. Circ. Res. 2019, 125, 117–146. [Google Scholar] [CrossRef]
- Byass, P. The global burden of liver disease: A challenge for methods and for public health. BMC Med. 2014, 12, 159. [Google Scholar] [CrossRef]
- Liver, E.A.S. Easl recommendations on treatment of hepatitis c 2016. J. Hepatol. 2017, 66, 153–194. [Google Scholar]
- Mathurin, P.; Hadengue, A.; Bataller, R.; Addolorato, G.; Burra, P.; Burt, A.; Caballeria, J.; Cortez-Pinto, H.; Day, C.P.; Forrest, E.H.; et al. Easl clinical practical guidelines: Management of alcoholic liver disease. J. Hepatol. 2012, 57, 399–420. [Google Scholar] [CrossRef]
- Li, X.; Zhu, L.; Wang, B.; Yuan, M.; Zhu, R. Drugs and targets in fibrosis. Front. Pharmacol. 2017, 8, 855. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.A.; Friedman, S.L. Reversal, maintenance or progression: What happens to the liver after a virologic cure of hepatitis c? Antivir. Res. 2014, 107, 23–30. [Google Scholar] [CrossRef]
- Angaswamy, N.; Tiriveedhi, V.; Sarma, N.J.; Subramanian, V.; Klein, C.; Wellen, J.; Shenoy, S.; Chapman, W.C.; Mohanakumar, T. Interplay between immune responses to hla and non-hla self-antigens in allograft rejection. Hum. Immunol. 2013, 74, 1478–1485. [Google Scholar] [CrossRef]
- Lucidi, V.; Gustot, T.; Moreno, C.; Donckier, V. Liver transplantation in the context of organ shortage: Toward extension and restriction of indications considering recent clinical data and ethical framework. Curr. Opin. Crit. Care 2015, 21, 163–170. [Google Scholar] [CrossRef]
- Hinderer, S.; Schenke-Layland, K. Cardiac fibrosis—A short review of causes and therapeutic strategies. Adv. Drug Deliv. Rev. 2019, 146, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Strizzi, L.; Bianco, C.; Normanno, N.; Salomon, D. Cripto-1: A multifunctional modulator during embryogenesis and oncogenesis. Oncogene 2005, 24, 5731–5741. [Google Scholar] [CrossRef]
- Sousa, E.R.; Zoni, E.; Karkampouna, S.; Manna, F.; Gray, P.C.; Menna, M.; Julio, M.K. A multidisciplinary review of the roles of cripto in the scientific literature through a bibliometric analysis of its biological roles. Cancers 2020, 12, 1480. [Google Scholar] [CrossRef]
- Spike, B.T.; Kelber, J.A.; Booker, E.; Kalathur, M.; Rodewald, R.; Lipianskaya, J.; La, J.; He, M.; Wright, T.; Klemke, R.; et al. Cripto/grp78 signaling maintains fetal and adult mammary stem cells ex vivo. Stem Cell Rep. 2014, 2, 427–439. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xu, C.-H.; Sheng, Z.-H.; Hu, H.-D.; Hao, K.-K.; Wang, Q.-B.; Yu, L.-K. Elevated expression of cripto-1 correlates with poor prognosis in non-small cell lung cancer. Tumor Biol. 2014, 35, 8673–8678. [Google Scholar] [CrossRef] [PubMed]
- Cocciadiferro, L.; Miceli, V.; Kang, K.-S.; Polito, L.M.; Trosko, J.E.; Carruba, G. Profiling cancer stem cells in androgen-responsive and refractory human prostate tumor cell lines. Ann. N. Y. Acad. Sci. 2009, 1155, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Terry, S.; El-Sayed, I.Y.; Destouches, D.; Maille, P.; Nicolaiew, N.; Ploussard, G.; Semprez, F.; Pimpie, C.; Beltran, H.; Londono-Vallejo, A.; et al. Cripto overexpression promotes mesenchymal differentiation in prostate carcinoma cells through parallel regulation of akt and fgfr activities. Oncotarget 2015, 6, 11994–12008. [Google Scholar] [CrossRef] [PubMed]
- D’Antonio, A.; Losito, S.; Pignata, S.; Grassi, M.; Perrone, F.; De Luca, A.; Tambaro, R.; Bianco, C.; Gullick, W.J.; Johnson, G.R.; et al. Transforming growth factor alpha, amphiregulin and cripto-1 are frequently expressed in advanced human ovarian carcinomas. Int. J. Oncol. 2002, 21, 941–948. [Google Scholar] [PubMed]
- Fujii, K.; Yasui, W.; Kuniyasu, H.; Itoh, M.; Hanada, K.; Kajiyama, G.; Tahara, E. Expression of cripto in human gall bladder lesions. J. Pathol. 1996, 180, 166–168. [Google Scholar] [CrossRef]
- Giorgio, E.; Liguoro, A.; D’Orsi, L.; Mancinelli, S.; Barbieri, A.; Palma, G.; Arra, C.; Liguori, G.L. Cripto haploinsufficiency affects in vivo colon tumor development. Int. J. Oncol. 2014, 45, 31–40. [Google Scholar] [CrossRef]
- Sun, C.; Sun, L.; Jiang, K.; Gao, D.-M.; Kang, X.-N.; Wang, C.; Zhang, S.; Huang, S.; Qin, X.; Li, Y.; et al. Nanog promotes liver cancer cell invasion by inducing epithelial–mesenchymal transition through nodal/smad3 signaling pathway. Int. J. Biochem. Cell Biol. 2013, 45, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Strizzi, L.; Margaryan, N.V.; Gilgur, A.; Hardy, K.M.; Normanno, N.; Salomon, D.S.; Hendrix, M.J.C. The significance of a cripto-1-positive subpopulation of human melanoma cells exhibiting stem cell-like characteristics. Cell Cycle 2013, 12, 1450–1456. [Google Scholar] [CrossRef]
- Tysnes, B.B.; Satran, H.A.; Mork, S.J.; Margaryan, N.V.; Eide, G.E.; Petersen, K.; Strizzi, L.; Hendrix, M.J.C. Age-dependent association between protein expression of the embryonic stem cell marker cripto-1 and survival of glioblastoma patients. Transl. Oncol. 2013, 6, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Karkampouna, S.; van der Helm, D.; Gray, P.C.; Chen, L.; Klima, I.; Grosjean, J.; Burgmans, M.C.; Farina-Sarasqueta, A.; Snaar-Jagalska, E.B.; Stroka, D.M.; et al. Cripto promotes an aggressive tumour phenotype and resistance to treatment in hepatocellular carcinoma. J. Pathol. 2018, 245, 297–310. [Google Scholar] [CrossRef]
- Van der Helm, D.; Barnhoorn, M.C.; de Jonge-Muller, E.S.M.; Molendijk, I.; Hawinkels, L.; Coenraad, M.J.; van Hoek, B.; Verspaget, H.W. Local but not systemic administration of mesenchymal stromal cells ameliorates fibrogenesis in regenerating livers. J. Cell. Mol. Med. 2019, 23, 6238–6250. [Google Scholar] [CrossRef]
- Duim, S.N.; Kurakula, K.; Goumans, M.J.; Kruithof, B.P. Cardiac endothelial cells express wilms’ tumor-1: Wt1 expression in the developing, adult and infarcted heart. J. Mol. Cell. Cardiol. 2015, 81, 127–135. [Google Scholar] [CrossRef]
- Van Deel, E.D.; de Boer, M.; Kuster, D.W.; Boontje, N.M.; Holemans, P.; Sipido, K.R.; van der Velden, J.; Duncker, D.J. Exercise training does not improve cardiac function in compensated or decompensated left ventricular hypertrophy induced by aortic stenosis. J. Mol. Cell. Cardiol. 2011, 50, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Kruithof, B.P.T.; Lieber, S.C.; Kruithof-de Julio, M.; Gaussin, V.; Goumans, M.J. Culturing mouse cardiac valves in the miniature tissue culture system. J. Vis. Exp. 2015, e52750. [Google Scholar] [CrossRef]
- Kruithof, B.P.T.; Paardekooper, L.; Hiemstra, Y.L.; Goumans, M.J.; Palmen, M.; Delgado, V.; Klautz, R.J.M.; Ajmone Marsan, N. Stress-induced remodelling of the mitral valve: A model for leaflet thickening and superimposed tissue formation in mitral valve disease. Cardiovasc. Res. 2020, 116, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Gray, P.C.; Shani, G.; Aung, K.; Kelber, J.; Vale, W. Cripto binds transforming growth factor beta (tgf-beta) and inhibits tgf-beta signaling. Mol. Cell. Biol. 2006, 26, 9268–9278. [Google Scholar] [CrossRef] [PubMed]
- Shayakhmetov, D.M.; Li, Z.Y.; Ni, S.; Lieber, A. Analysis of adenovirus sequestration in the liver, transduction of hepatic cells, and innate toxicity after injection of fiber-modified vectors. J. Virol. 2004, 78, 5368–5381. [Google Scholar] [CrossRef]
- Balcioglu, O.; Heinz, R.E.; Freeman, D.W.; Gates, B.L.; Hagos, B.M.; Booker, E.; Mehrabad, E.M.; Diesen, H.T.; Bhakta, K.; Ranganathan, S.; et al. CRIPTO antagonist ALK4(L75A)-Fc inhibits breast cancer cell plasticity and adaptation to stress. Breast Cancer Res. 2020, 22, 125. [Google Scholar] [CrossRef]
- Kelber, J.A.; Panopoulos, A.D.; Shani, G.; Booker, E.C.; Belmonte, J.C.; Vale, W.W.; Gray, P.C. Blockade of cripto binding to cell surface grp78 inhibits oncogenic cripto signaling via mapk/pi3k and smad2/3 pathways. Oncogene 2009, 28, 2324–2336. [Google Scholar] [CrossRef]
- Jun, J.-I.; Lau, L.F. Resolution of organ fibrosis. J. Clin. Investig. 2018, 128, 97–107. [Google Scholar] [CrossRef]
- Zeisberg, M.; Kalluri, R. Cellular mechanisms of tissue fibrosis. 1. Common and organ-specific mechanisms associated with tissue fibrosis. Am. J. Physiol. Cell Physiol. 2013, 304, C216–C225. [Google Scholar] [CrossRef] [PubMed]
- Sebastiani, G.; Castera, L.; Halfon, P.; Pol, S.; Mangia, A.; Di Marco, V.; Pirisi, M.; Voiculescu, M.; Bourliere, M.; Alberti, A. The impact of liver disease aetiology and the stages of hepatic fibrosis on the performance of non-invasive fibrosis biomarkers: An international study of 2411 cases. Aliment. Pharmacol. 2011, 34, 1202–1216. [Google Scholar] [CrossRef]
- Afify, S.M.; Tabll, A.; Nawara, H.M.; Kassas, M.E.; Elfert, A.; Seno, M.; El-Kousy, S. Five fibrosis biomarkers together with serum ferritin level to diagnose liver fibrosis and cirrhosis. Clin. Lab. 2018, 64, 1685–1693. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.; Chi, X.; Fan, Y.; Shi, Y.; Niu, J. High level of serum cripto-1 in hepatocellular carcinoma, especially with hepatitis b virus infection. Medicine 2018, 97, e11781. [Google Scholar] [CrossRef]
- Ngo, D.; Sinha, S.; Shen, D.; Kuhn, E.W.; Keyes, M.J.; Shi, X.; Benson, M.D.; O’Sullivan, J.F.; Keshishian, H.; Farrell, L.A.; et al. Aptamer-based proteomic profiling reveals novel candidate biomarkers and pathways in cardiovascular disease. Circulation 2016, 134, 270–285. [Google Scholar] [CrossRef]
- Guardiola, O.; Lafuste, P.; Brunelli, S.; Iaconis, S.; Touvier, T.; Mourikis, P.; De Bock, K.; Lonardo, E.; Andolfi, G.; Bouche, A.; et al. Cripto regulates skeletal muscle regeneration and modulates satellite cell determination by antagonizing myostatin. Proc. Natl. Acad. Sci. USA 2012, 109, E3231–E3240. [Google Scholar] [CrossRef]
- Xu, C.; Liguori, G.; Adamson, E.D.; Persico, M.G. Specific arrest of cardiogenesis in cultured embryonic stem cells lacking cripto-1. Dev. Biol. 1998, 196, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Parisi, S.; D’Andrea, D.; Lago, C.T.; Adamson, E.D.; Persico, M.G.; Minchiotti, G. Nodal-dependent cripto signaling promotes cardiomyogenesis and redirects the neural fate of embryonic stem cells. J. Cell Biol. 2003, 163, 303–314. [Google Scholar] [CrossRef]
- Bianco, C.; Cotten, C.; Lonardo, E.; Strizzi, L.; Baraty, C.; Mancino, M.; Gonzales, M.; Watanabe, K.; Nagaoka, T.; Berry, C.; et al. Cripto-1 is required for hypoxia to induce cardiac differentiation of mouse embryonic stem cells. Am. J. Pathol. 2009, 175, 2146–2158. [Google Scholar] [CrossRef]
- Xu, C.; Liguori, G.; Persico, M.G.; Adamson, E.D. Abrogation of the cripto gene in mouse leads to failure of postgastrulation morphogenesis and lack of differentiation of cardiomyocytes. Development 1999, 126, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Toraih, E.A.; Fawzy, M.S.; El-Falouji, A.I.; Hamed, E.O.; Nemr, N.A.; Hussein, M.H.; Abd El Fadeal, N.M. Stemness-related transcriptional factors and homing gene expression profiles in hepatic differentiation and cancer. Mol. Med. 2016, 22, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.; Trial, J.; Soeller, C.; Entman, M.L. Cardiac mesenchymal stem cells contribute to scar formation after myocardial infarction. Cardiovasc. Res. 2011, 91, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.R.; Jin, C.X.; Xu, G.T.; Yang, C.Q. Bone morphogenetic protein-7 regulates snail signaling in carbon tetrachloride-induced fibrosis in the rat liver. Exp. Med. 2012, 4, 1022–1026. [Google Scholar] [CrossRef]
- Gray, P.C.; Vale, W. Cripto/grp78 modulation of the tgf-beta pathway in development and oncogenesis. FEBS Lett. 2012, 586, 1836–1845. [Google Scholar] [CrossRef] [PubMed]
- Khalil, H.; Kanisicak, O.; Prasad, V.; Correll, R.N.; Fu, X.; Schips, T.; Vagnozzi, R.J.; Liu, R.; Huynh, T.; Lee, S.J.; et al. Fibroblast-specific tgf-beta-smad2/3 signaling underlies cardiac fibrosis. J. Clin. Investig. 2017, 127, 3770–3783. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mi, X.; Song, Z.; Li, Y.; YingShi; Niu, J. Cripto-1 promotes resistance to drug-induced apoptosis by activating the tak-1/nf-kappab/survivin signaling pathway. Biomed. Pharm. 2018, 104, 729–737. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karkampouna, S.; van der Helm, D.; Scarpa, M.; van Hoek, B.; Verspaget, H.W.; Goumans, M.-J.; Coenraad, M.J.; Kruithof, B.P.T.; Kruithof-de Julio, M. Oncofetal Protein CRIPTO Is Involved in Wound Healing and Fibrogenesis in the Regenerating Liver and Is Associated with the Initial Stages of Cardiac Fibrosis. Cells 2021, 10, 3325. https://doi.org/10.3390/cells10123325
Karkampouna S, van der Helm D, Scarpa M, van Hoek B, Verspaget HW, Goumans M-J, Coenraad MJ, Kruithof BPT, Kruithof-de Julio M. Oncofetal Protein CRIPTO Is Involved in Wound Healing and Fibrogenesis in the Regenerating Liver and Is Associated with the Initial Stages of Cardiac Fibrosis. Cells. 2021; 10(12):3325. https://doi.org/10.3390/cells10123325
Chicago/Turabian StyleKarkampouna, Sofia, Danny van der Helm, Mario Scarpa, Bart van Hoek, Hein W. Verspaget, Marie-Jose Goumans, Minneke J. Coenraad, Boudewijn P.T. Kruithof, and Marianna Kruithof-de Julio. 2021. "Oncofetal Protein CRIPTO Is Involved in Wound Healing and Fibrogenesis in the Regenerating Liver and Is Associated with the Initial Stages of Cardiac Fibrosis" Cells 10, no. 12: 3325. https://doi.org/10.3390/cells10123325
APA StyleKarkampouna, S., van der Helm, D., Scarpa, M., van Hoek, B., Verspaget, H. W., Goumans, M.-J., Coenraad, M. J., Kruithof, B. P. T., & Kruithof-de Julio, M. (2021). Oncofetal Protein CRIPTO Is Involved in Wound Healing and Fibrogenesis in the Regenerating Liver and Is Associated with the Initial Stages of Cardiac Fibrosis. Cells, 10(12), 3325. https://doi.org/10.3390/cells10123325





