The Effects of Ionising and Non-Ionising Electromagnetic Radiation on Extracellular Matrix Proteins
Abstract
1. Introduction
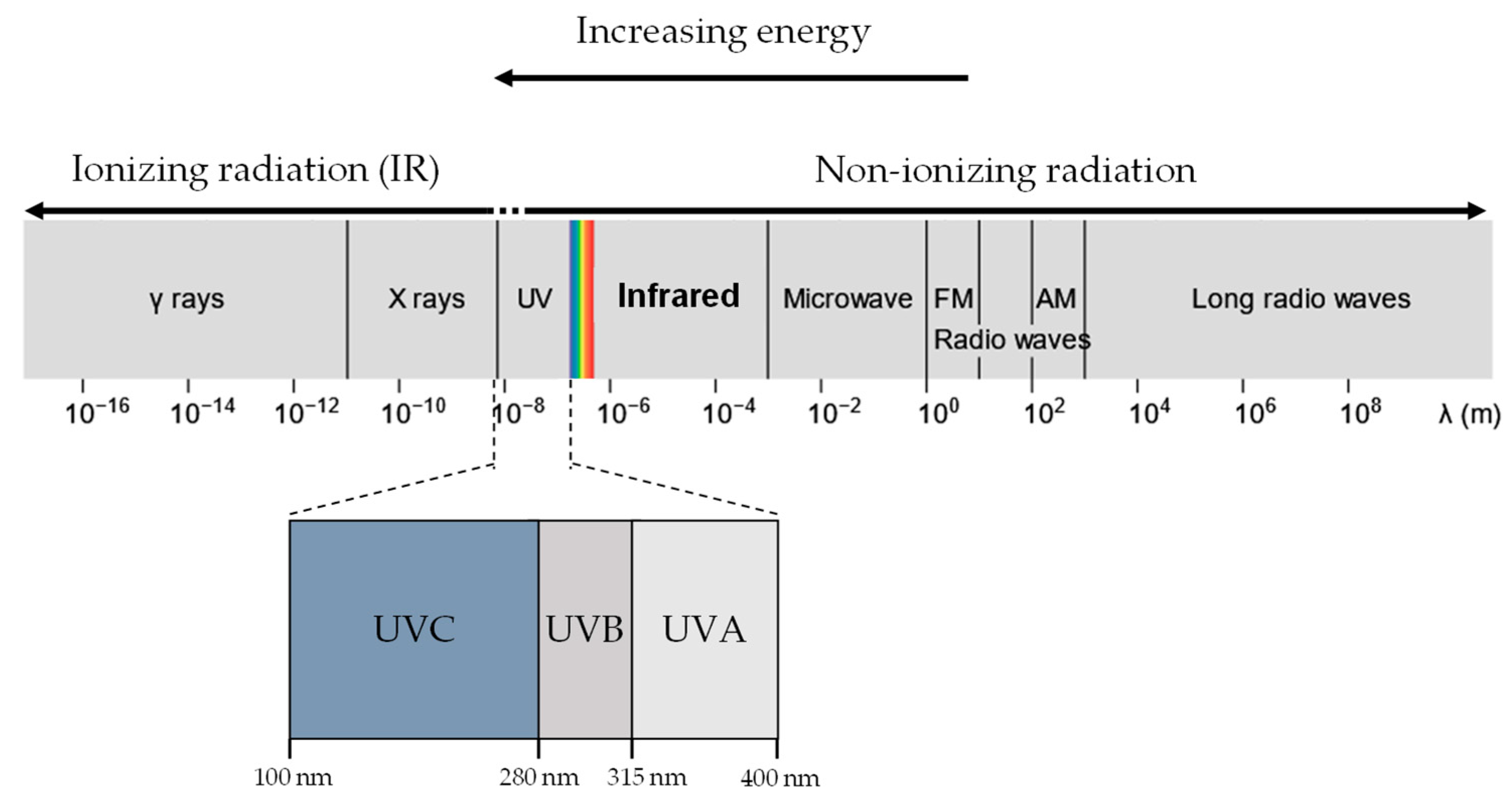
Electromagnetic Radiation
2. Non-Ionising Radiation (UVR)
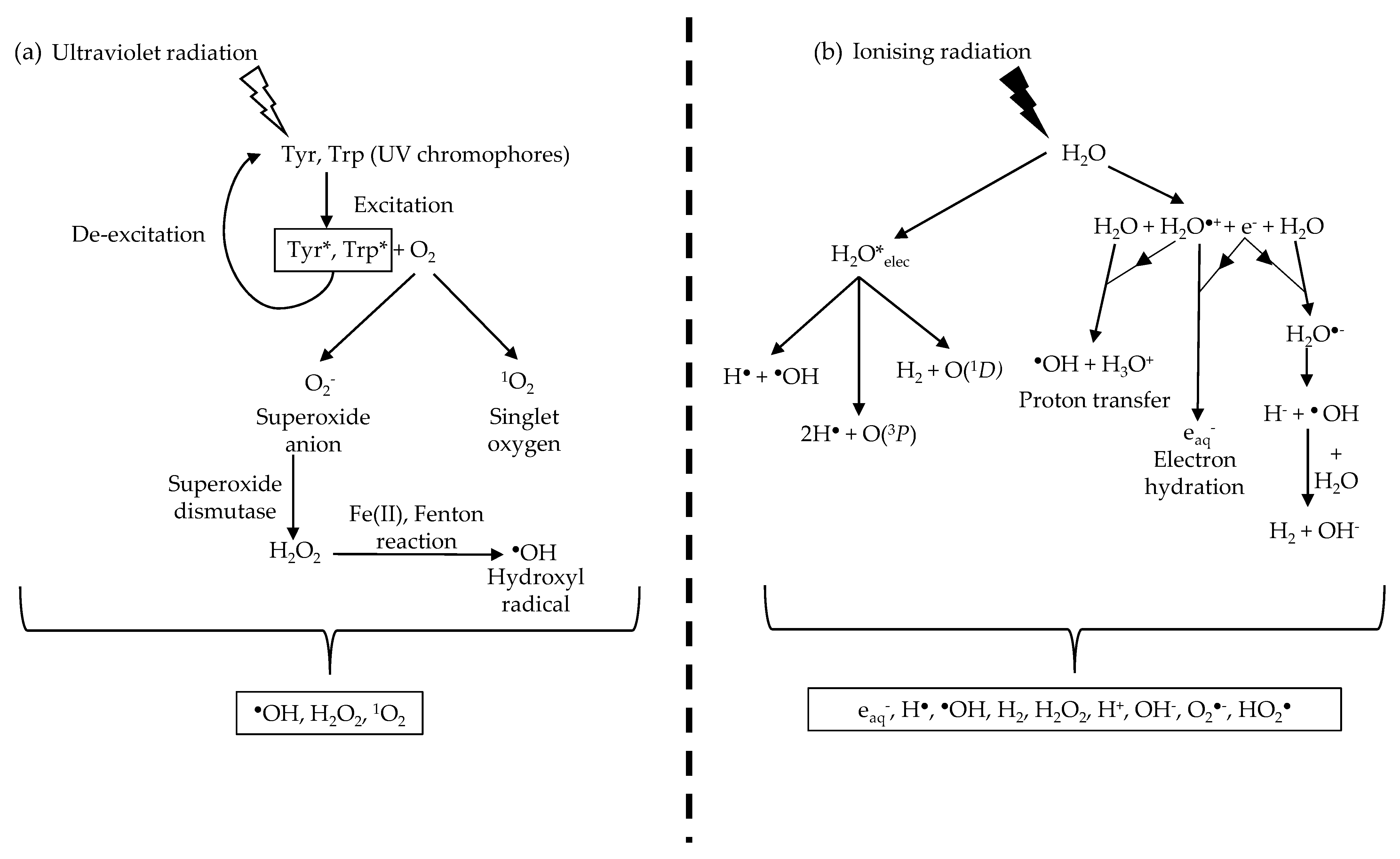
2.1. Absorption of Non-Ionising Radiation (UVR)
2.2. Biological Consequences of UVR Exposure
2.3. Repair and Prevention of UVR Damage
3. Ionising Radiation (X-rays/Gamma Rays)
3.1. Absorption of Ionising Radiation (X-rays/Gamma Rays)
3.2. Biological Consequences of Exposure to Ionising Radiation
3.3. Repair and Prevention of Ionising Radiation Damage
4. Model Tissue Systems for Radiation Studies
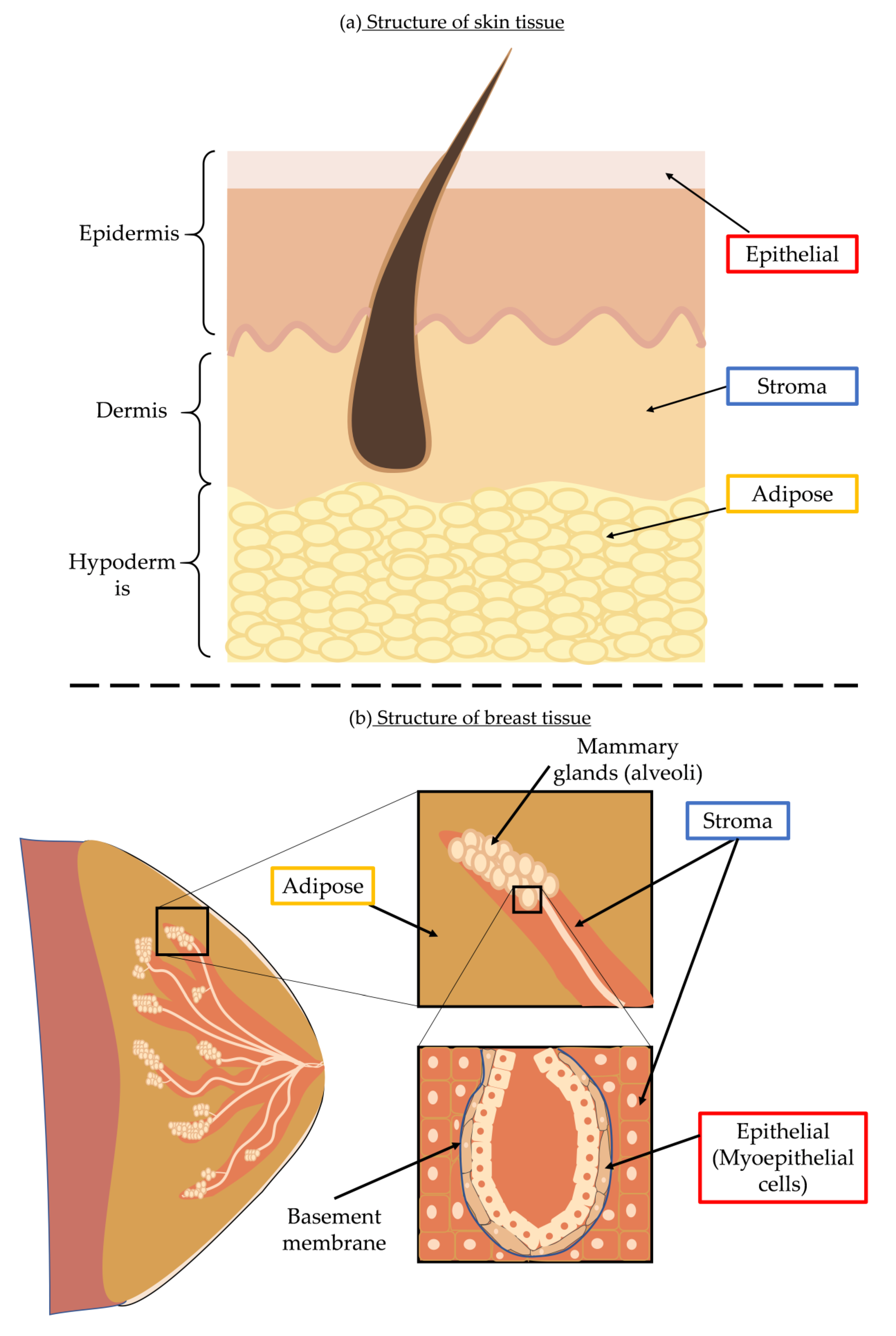
4.1. Cellular and Acellular Responses of Skin to UVR
4.2. Outcomes of UVR Exposure on Skin
4.3. Structure of Breast Tissue
4.4. Cellular and Acellular Responses of Breast Tissue to Ionising Radiation
4.5. Clinical Outcomes of Ionising Radiation Exposure on Breast Tissue
5. The Extracellular Matrix as a Target of Radiation Damage
5.1. Radiation and ECM Mechanical Properties
5.2. Radiation and ECM Biochemistry
5.3. Challenges of Studying the ECM and the Current State of Knowledge
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Lopez Payares, G.M.; Ali, F.A. Vitamin D deficiency. In 5-Minute Clin Consult Stand 2016, 24th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2015; pp. 266–281. [Google Scholar]
- Wlaschek, M.; Tantcheva-Poór, I.; Naderi, L.; Ma, W.; Schneider, L.A.; Razi-Wolf, Z.; Schüller, J.; Scharffetter-Kochanek, K. Solar UV irradiation and dermal photoaging. J. Photochem. Photobiol. B Biol. 2001, 63, 41–51. [Google Scholar] [CrossRef]
- Martens, M.C.; Seebode, C.; Lehmann, J.; Emmert, S. Photocarcinogenesis and skin cancer prevention strategies: An update. Anticancer Res. 2018, 38, 1153–1158. [Google Scholar] [CrossRef]
- Ballabriga, R.; Alozy, J.; Bandi, F.N.; Campbell, M.; Egidos, N.; Fernandez-Tenllado, J.M.; Heijne, E.H.M.; Kremastiotis, I.; Llopart, X.; Madsen, B.J.; et al. Photon Counting Detectors for X-ray Imaging with Emphasis on CT. IEEE Trans. Radiat. Plasma Med. Sci. 2021, 5, 422–440. [Google Scholar] [CrossRef]
- Allen, A.M.; Pawlicki, T.; Dong, L.; Fourkal, E.; Buyyounouski, M.; Cengel, K.; Plastaras, J.; Bucci, M.K.; Yock, T.I.; Bonilla, L.; et al. An evidence based review of proton beam therapy: The report of ASTRO ’ s emerging technology committee. Radiother. Oncol. 2012, 103, 8–11. [Google Scholar] [CrossRef]
- Olaciregui-Ruiz, I.; Beddar, S.; Greer, P.; Jornet, N.; McCurdy, B.; Paiva-Fonseca, G.; Mijnheer, B.; Verhaegen, F. In vivo dosimetry in external beam photon radiotherapy: Requirements and future directions for research, development, and clinical practice. Phys. Imaging Radiat. Oncol. 2020, 15, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Purkayastha, A.; Sharma, N.; Sarin, A.; Bhatnagar, S.; Chakravarty, N. Radiation Fibrosis Syndrome: The Evergreen Menace of Radiation Therapy. Asia Pac. J. Oncol. Nurs. 2019, 6, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Zelefsky, M.J.; Housman, D.M.; Pei, X.; Alicikus, Z.; Magsanoc, J.M.; Dauer, L.T.; St. Germain, J.; Yamada, Y.; Kollmeier, M.; Cox, B.; et al. Incidence of secondary cancer development after high-dose intensity-modulated radiotherapy and image-guided brachytherapy for the treatment of localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 953–959. [Google Scholar] [CrossRef]
- Helm, A.; Tinganelli, W.; Durante, M. Advances in Radiation Biology of Particle Irradiation. Prog. Tumor Res. 2018, 44, 105–121. [Google Scholar] [CrossRef]
- Bagshaw, M. Cosmic radiation in commercial aviation. Travel Med. Infect. Dis. 2008, 6, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Ball, T.K.; Miles, J.C.H. Geological and geochemical factors affecting the radon concentration in homes in Cornwall and Devon, UK. Environ. Geochem. Health 1993, 15, 27–36. [Google Scholar] [CrossRef]
- Withers, P.J.; Grimaldi, D.; Hagen, C.K.; Maire, E.; Manley, M.; Plessis, A. Du X-ray computed tomography. Nat. Rev. Methods Prim. 2021, 1, 18. [Google Scholar] [CrossRef]
- Marín, A.; Martín, M.; Liñán, O.; Alvarenga, F.; López, M.; Fernández, L.; Büchser, D.; Cerezo, L. Bystander effects and radiotherapy. Reports Pract. Oncol. Radiother. 2015, 20, 12–21. [Google Scholar] [CrossRef]
- Nguyen, H.Q.; To, N.H.; Zadigue, P.; Kerbrat, S.; De La Taille, A.; Le Gouvello, S.; Belkacemi, Y. Ionizing radiation-induced cellular senescence promotes tissue fibrosis after radiotherapy. A review. Crit. Rev. Oncol. Hematol. 2018, 129, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Bostrom, P.J.; Soloway, M.S. Secondary Cancer After Radiotherapy for Prostate Cancer: Should We Be More Aware of the Risk? Eur. Urol. 2007, 52, 973–982. [Google Scholar] [CrossRef]
- Mullenders, L.H.F. Solar UV damage to cellular DNA: From mechanisms to biological effects. Photochem. Photobiol. Sci. 2018, 17, 1842–1852. [Google Scholar] [CrossRef]
- Green, C.; Diffey, B.L.; Hawk, J.L.M. Ultraviolet radiation in the treatment of skin disease. Phys. Med. Biol. 1992, 37, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, Z.W.; German, G.K. Ultraviolet light degrades the mechanical and structural properties of human stratum corneum. J. Mech. Behav. Biomed. Mater. 2019, 100, 103391. [Google Scholar] [CrossRef]
- Miyauchi, M.; Nakajima, H. Determining an Effective UV Radiation Exposure Time for Vitamin D Synthesis in the Skin Without Risk to Health: Simplified Estimations from UV Observations. Photochem. Photobiol. 2016, 92, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.E. Ocular photoxicity. J. Photochem. Photobiol. B Biol. 2001, 64, 136–143. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Andreev, S.N.; Barmina, E.V.; Bunkin, N.F.; Kartabaeva, B.B.; Nesvat, A.P.; Stepanov, E.V.; Taranda, N.I.; Khramov, R.N.; Glinushkin, A.P. Effect of visible light on biological objects: Physiological and pathophysiological aspects. Phys. Wave Phenom. 2017, 25, 207–213. [Google Scholar] [CrossRef]
- Tsai, S.R.; Hamblin, M.R. Biological effects and medical applications of infrared radiation. J. Photochem. Photobiol. B Biol. 2017, 170, 197–207. [Google Scholar] [CrossRef]
- Darvin, M.E.; Zastrov, L.; Gonchukov, S.A.; Lademann, J. Influence of IR radiation on the carotenoid content in human skin. Opt. Spectrosc. (Engl. Transl. Opt. i Spektrosk.) 2009, 107, 917–920. [Google Scholar] [CrossRef]
- Siingh, D.; Singh, A.K.; Patel, R.P.; Singh, R.; Singh, R.P.; Veenadhari, B.; Mukherjee, M. Thunderstorms, lightning, sprites and magnetospheric whistler-mode radio waves. Surv. Geophys. 2008, 29, 499–551. [Google Scholar] [CrossRef]
- Behrouzkia, Z.; Joveini, Z.; Keshavarzi, B.; Eyvazzadeh, N.; Aghdam, R.Z. Hyperthermia: How can it be used? Oman Med. J. 2016, 31, 89–97. [Google Scholar] [CrossRef]
- Van Rongen, E.; Croft, R.; Juutilainen, J.; Lagroye, I.; Miyakoshi, J.; Saunders, R.; De Seze, R.; Tenforde, T.; Verschaeve, L.; Veyret, B.; et al. Effects of radiofrequency electromagnetic fields on the human nervous system. J. Toxicol. Environ. Health-Part B Crit. Rev. 2009, 12, 572–597. [Google Scholar] [CrossRef]
- Lomax, M.E.; Folkes, L.K.; O’Neill, P. Biological consequences of radiation-induced DNA damage: Relevance to radiotherapy. Clin. Oncol. 2013, 25, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Yokoya, A.; Shikazono, N.; Fujii, K.; Urushibara, A.; Akamatsu, K.; Watanabe, R. DNA damage induced by the direct effect of radiation. Radiat. Phys. Chem. 2008, 77, 1280–1285. [Google Scholar] [CrossRef]
- Lyons, A.; Ghazali, N. Osteoradionecrosis of the jaws: Current understanding of its pathophysiology and treatment. Br. J. Oral Maxillofac. Surg. 2008, 46, 653–660. [Google Scholar] [CrossRef]
- Weihermann, A.C.; Lorencini, M.; Brohem, C.A.; de Carvalho, C.M. Elastin structure and its involvement in skin photoageing. Int. J. Cosmet. Sci. 2017, 39, 241–247. [Google Scholar] [CrossRef]
- Herzberg, G. Molecular Spectra and Molecular Structure I: Spectra of Diatomic Molecules, 2nd ed.; D. Van Nostrand Company: Toronto, ON, Canada, 1963. [Google Scholar]
- Hertel, I.V.; Schulz, C.-P. Molecular Spectroscopy. In Purinergic Signalling; Springer: Heidelberg, Germany, 2015; pp. 289–381. Available online: http://link.springer.com/10.1007/978-3-642-54313-5 (accessed on 24 April 2021).
- Armentrout, P.B. Chemistry of Excited Electronic States. Science 1991, 251, 175–179. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. ISO 21348 Definitions of Solar Irradiance Spectral Categories. 2007. Available online: https://web.archive.org/web/20131029233428/http://www.spacewx.com/pdf/SET_21348_2004.pdf (accessed on 24 April 2021).
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef]
- Frederick, J.E.; Snell, H.E.; Haywood, E.K. Solar Ultraviolet Radiation At the Earth’S Surface. Photochem. Photobiol. 1989, 50, 443–450. [Google Scholar] [CrossRef]
- Watanabe, K. Ultraviolet Absorption Processes in the Upper Atmosphere. In Advances in Geophysics; Elsevier: Amsterdam, The Netherlands, 1958; pp. 153–221. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0065268708600783 (accessed on 10 January 2021).
- Bernath, P.F.; Mérienne, M.F.; Colin, R. The near ultraviolet rotation-vibration spectrum of water. J. Chem. Phys. 2000, 113, 1546–1552. [Google Scholar] [CrossRef]
- Mitchell, D.L. the Relative Cytotoxicity of(6–4) Photoproducts and Cyclobutane Dimers in Mammalian Cells. Photochem. Photobiol. 1988, 48, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Schreier, W.J.; Kubon, J.; Regner, N.; Haiser, K.; Schrader, T.E.; Zinth, W.; Clivio, P.; Gilch, P. Thymine dimerization in DNA model systems: Cyclobutane photolesion is predominantly formed via the singlet channel. J. Am. Chem. Soc. 2009, 131, 5038–5039. [Google Scholar] [CrossRef] [PubMed]
- BEnsasson, R.V.; Land, E.J.; Salet, C. Triplet excited state of furocoumarins: Reaction with nucleic acid bases and amino acids. Photochem. Photobiol. 1978, 27, 273–280. [Google Scholar] [CrossRef]
- Pattison, D.I.; Rahmanto, A.S.; Davies, M.J. Photo-oxidation of proteins. Photochem. Photobiol. Sci. 2012, 11, 38–53. [Google Scholar] [CrossRef] [PubMed]
- Aitken, A.; Learmonth, M.P. Protein Determination by UV Absorption. Protein Protoc. Handb. 2009, 205, 3–6. [Google Scholar] [CrossRef]
- Jhappan, C.; Noonan, F.P.; Merlino, G. Ultraviolet radiation and cutaneous malignant melanoma. Oncogene 2003, 22, 3099–3112. [Google Scholar] [CrossRef]
- Petruska, J. Changes in the electronic transitions of aromatic hydrocarbons on chemical substitution. I. Perturbation theory for substituted cyclic polyenes. J. Chem. Phys. 1961, 34, 1111–1120. [Google Scholar] [CrossRef]
- Saik, V.O.; Lipsky, S. Absorption spectrum of neat liquid benzene and its concentrated solutions in n-hexane from 220 to 170 nm. J. Phys. Chem. 1995, 99, 4406–4413. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Ravanat, J.L. Oxidatively generated damage to the guanine moiety of DNA: Mechanistic aspects and formation in cells. Acc. Chem. Res. 2008, 41, 1075–1083. [Google Scholar] [CrossRef]
- Cheng, K.C.; Cahill, D.S.; Kasai, H.; Nishimura, S.; Loeb, L.A. 8-Hydroxyguanine, an abundant form of oxidative DNA damage, causes G → T and A → C substitutions. J. Biol. Chem. 1992, 267, 166–172. [Google Scholar] [CrossRef]
- Cadet, J.; Richard Wagner, J. DNA base damage by reactive oxygen species, oxidizing agents, and UV radiation. Cold Spring Harb. Perspect. Biol. 2013, 5, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Häder, D.P. UV-induced DNA damage and repair: A review. Photochem. Photobiol. Sci. 2002, 1, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Weisiger, R.A.; Fridovich, I. Superoxide dismutase. Organelle specificity. J. Biol. Chem. 1973, 248, 3582–3592. [Google Scholar] [CrossRef]
- Folz, R.J.; Crapo, J.D. Extracellular superoxide dismutase (sod3): Tissue-specific expression, genomic characterization, and computer-assisted sequence analysis of the human ec sod gene. Genomics 1994, 22, 162–171. [Google Scholar] [CrossRef]
- Thomas, C.; Mackey, M.M.; Diaz, A.A.; Cox, D.P. Hydroxyl radical is produced via the Fenton reaction in submitochondrial particles under oxidative stress: Implications for diseases associated with iron accumulation. Redox Rep. 2009, 14, 102–108. [Google Scholar] [CrossRef]
- van Loon, B.; Markkanen, E.; Hübscher, U. Oxygen as a friend and enemy: How to combat the mutational potential of 8-oxo-guanine. DNA Repair 2010, 9, 604–616. [Google Scholar] [CrossRef]
- Zhao, Z.; Poojary, M.M.; Skibsted, L.H.; Lund, M.N.; Lund, M.N. Cleavage of Disulfide Bonds in Cystine by UV-B Illumination Mediated by Tryptophan or Tyrosine as Photosensitizers. J. Agric. Food Chem. 2020, 68, 6900–6909. [Google Scholar] [CrossRef]
- Meesungnoen, J.; Jay-Gerin, J.-P. Radiation Chemistry of Liquid Water with Heavy Ions: Monte Carlo Simulation Studies. In Charged Particle and Photon Interactions with Matter Recent Advances, Applications, and Interfaces; Hatano, Y., Katsumura, Y., Mozumder, A., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2010; pp. 355–392. Available online: https://www.taylorfrancis.com/books/9781439811801 (accessed on 16 March 2021).
- Douki, T.; Sage, E. Dewar valence isomers, the third type of environmentally relevant DNA photoproducts induced by solar radiation. Photochem. Photobiol. Sci. 2016, 15, 24–30. [Google Scholar] [CrossRef]
- Breen, A.P.; Murphy, J.A. Reactions of oxyl radicals with DNA. [Review] [280 refs]. Free Radic. Biol. Med. 1995, 18, 1033–1077. [Google Scholar] [CrossRef]
- Nakagawa, M.; Watanabe, H.; Kodato, S. A valid model for the mechanism of oxidation of tryptophan to formylkynurenine—25 Years later. Proc. Natl. Acad. Sci. USA 1977, 74, 4730–4733. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.; Bubb, W.A.; Hawkins, C.L.; Davies, M.J. Singlet Oxygen–mediated Protein Oxidation: Evidence for the Formation of Reactive Side Chain Peroxides on Tyrosine Residues. Photochem. Photobiol. 2002, 76, 35. [Google Scholar] [CrossRef]
- Agon, V.V.; Bubb, W.A.; Wright, A.; Hawkins, C.L.; Davies, M.J. Sensitizer-mediated photooxidation of histidine residues: Evidence for the formation of reactive side-chain peroxides. Free Radic. Biol. Med. 2006, 40, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Murray, R.W.; Jindal, S.L. The Photosensitized Oxidation of Disulfides Related To Cystine. Photochem. Photobiol. 1972, 16, 147–151. [Google Scholar] [CrossRef]
- Rauk, A.; Yu, D.; Armstrong, D.A. Oxidative damage to and by cysteine in proteins: An ab initio study of the radical structures, C-H, S-H, and C-C bond dissociation energies, and transition structures for H abstraction by thiyl radicals. J. Am. Chem. Soc. 1998, 120, 8848–8855. [Google Scholar] [CrossRef]
- Schöneich, C. Methionine oxidation by reactive oxygen species: Reaction mechanisms and relevance to Alzheimer’s disease. Biochim. Biophys. Acta-Proteins Proteom. 2005, 1703, 111–119. [Google Scholar] [CrossRef]
- Xu, P.; Zheng, Y.; Zhu, X.; Li, S.; Zhou, C. L-lysine and L-arginine inhibit the oxidation of lipids and proteins of emulsion sausage by chelating iron ion and scavenging radical. Asian-Australas. J. Anim. Sci. 2018, 31, 905–913. [Google Scholar] [CrossRef]
- Liu, F.; Lai, S.; Tong, H.; Lakey, P.S.J.; Shiraiwa, M.; Weller, M.G.; Pöschl, U.; Kampf, C.J. Release of free amino acids upon oxidation of peptides and proteins by hydroxyl radicals. Anal. Bioanal. Chem. 2017, 409, 2411–2420. [Google Scholar] [CrossRef] [PubMed]
- Sclavi, B.; Sullivan, M.; Chance, M.R.; Brenowitz, M.; Woodson, S.A. RNA folding at millisecond intervals by synchrotron hydroxyl radical footprinting. Science 1998, 279, 1940–1943. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R.; Levine, R.L. Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 2003, 25, 207–218. [Google Scholar] [CrossRef]
- Rabotyagova, O.S.; Cebe, P.; Kaplan, D.L. Collagen structural hierarchy and susceptibility to degradation by ultraviolet radiation. Mater. Sci. Eng. C 2008, 28, 1420–1429. [Google Scholar] [CrossRef]
- Sionkowska, A. Thermal stability of UV-irradiated collagen in bovine lens capsules and in bovine cornea. J. Photochem. Photobiol. B Biol. 2005, 80, 87–92. [Google Scholar] [CrossRef]
- Sionkowska, A. Thermal denaturation of UV-irradiated wet rat tail tendon collagen. Int. J. Biol. Macromol. 2005, 35, 145–149. [Google Scholar] [CrossRef]
- Yin, H.; Xu, L.; Porter, N.A. Free radical lipid peroxidation: Mechanisms and analysis. Chem. Rev. 2011, 111, 5944–5972. [Google Scholar] [CrossRef]
- Eckersley, A.; Ozols, M.; O’Cualain, R.; Keevill, E.J.; Foster, A.; Pilkington, S.; Knight, D.; Griffiths, C.E.M.; Watson, R.E.B.; Sherratt, M.J. Proteomic fingerprints of damage in extracellular matrix assemblies. Matrix Biol. Plus 2020, 5, 100027. [Google Scholar] [CrossRef]
- Kennett, E.C.; Chuang, C.Y.; Degendorfer, G.; Whitelock, J.M.; Davies, M.J. Mechanisms and consequences of oxidative damage to extracellular matrix. Biochem. Soc. Trans. 2011, 39, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Hibbert, S.A.; Watson, R.E.B.; Gibbs, N.K.; Costello, P.; Baldock, C.; Weiss, A.S.; Griffiths, C.E.M.; Sherratt, M.J. A potential role for endogenous proteins as sacrificial sunscreens and antioxidants in human tissues. Redox Biol. 2015, 5, 101–113. [Google Scholar] [CrossRef]
- Schärer, O.D. Nucleotide excision repair in Eukaryotes. Cold Spring Harb. Perspect. Biol. 2013, 5, 1–19. [Google Scholar] [CrossRef]
- Holmgren, A.; Johansson, C.; Berndt, C.; Lönn, M.E.; Hudemann, C.; Lillig, C.H. Thiol redox control via thioredoxin and glutaredoxin systems. Biochem. Soc. Trans. 2005, 33, 1375–1377. [Google Scholar] [CrossRef]
- Davies, K.J.A. Degradation of oxidized proteins by the 20S proteasome. Biochimie 2001, 83, 301–310. [Google Scholar] [CrossRef]
- Fedorova, M.; Bollineni, R.C.; Hoffmann, R. Protein carbonylation as a major hallmark of oxidative damage: Update of analytical strategies. Mass Spectrom. Rev. 2014, 33, 79–97. [Google Scholar] [CrossRef]
- Yao, H.; Arunachalam, G.; Hwang, J.W.; Chung, S.; Sundar, I.K.; Kinnula, V.L.; Crapo, J.D.; Rahman, I. Extracellular superoxide dismutase protects against pulmonary emphysema by attenuating oxidative fragmentation of ECM. Proc. Natl. Acad. Sci. USA 2010, 107, 15571–15576. [Google Scholar] [CrossRef] [PubMed]
- Schamberger, A.C.; Schiller, H.B.; Fernandez, I.E.; Sterclova, M.; Heinzelmann, K.; Hennen, E.; Hatz, R.; Behr, J.; Vašáková, M.; Mann, M.; et al. Glutathione peroxidase 3 localizes to the epithelial lining fluid and the extracellular matrix in interstitial lung disease. Sci. Rep. 2016, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). Sources and Effects of Ionizing Radiation UNSCEAR 2008 Report to the General Assembly with Scientific Annexes Volume 1; United Nations Publication: New York, NY, USA, 2008; Volume I, 156p. [Google Scholar]
- ICRP. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Volume 37, Annals of the ICRP. April 2007. Available online: http://journals.sagepub.com/doi/10.1016/j.icrp.2007.10.003 (accessed on 15 February 2021).
- Young, K.C.; Oduko, J.M. Radiation doses received in the United Kingdom breast screening programme in 2010 to 2012. Br. J. Radiol. 2016, 89. [Google Scholar] [CrossRef]
- Podgorsak, E.B. External photon beams: Physical aspects. In Radiation Oncology Physics: A Handbook for Teachers and Students; IAEA (International Atomic Energy Agency): Vienna, Austria, 2005; Chapter 6; pp. 161–217. Available online: https://www-pub.iaea.org/mtcd/publications/pdf/pub1196_web.pdf (accessed on 25 December 2020).
- Koulis, T.A.; Phan, T.; Olivotto, I.A. Hypofractionated whole breast radiotherapy: Current perspectives. Breast Cancer Targets Ther. 2015, 7, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Rogers, L.; Demanes, D.J.; Morton, G.; Prestidge, B.R.; Pouliot, J.; Cohen, G.N.; Zaider, M.; Ghilezan, M.; Hsu, I.C. American Brachytherapy Society consensus guidelines for high-dose-rate prostate brachytherapy. Brachytherapy 2012, 11, 20–32. [Google Scholar] [CrossRef]
- Yahyapour, R.; Salajegheh, A.; Safari, A.; Amini, P.; Rezaeyan, A.; Amraee, A.; Najafi, M. Radiation-induced Non-targeted Effect and Carcinogenesis; Implications in Clinical Radiotherapy. J. Biomed. Phys. Eng. 2018, 8, 435–446. [Google Scholar] [CrossRef]
- Tommasino, F.; Durante, M. Proton radiobiology. Cancers 2015, 7, 353–381. [Google Scholar] [CrossRef]
- Mohan, R.; Grosshans, D. Proton therapy—Present and future. Adv. Drug Deliv. Rev. 2017, 109, 26–44. [Google Scholar] [CrossRef] [PubMed]
- Reisz, J.A.; Bansal, N.; Qian, J.; Zhao, W.; Furdui, C.M. Effects of ionizing radiation on biological molecules—Mechanisms of damage and emerging methods of detection. Antioxidants Redox Signal. 2014, 21, 260–292. [Google Scholar] [CrossRef] [PubMed]
- Lalande, M.; Schwob, L.; Vizcaino, V.; Chirot, F.; Dugourd, P.; Schlathölter, T.; Poully, J.C. Direct Radiation Effects on the Structure and Stability of Collagen and Other Proteins. ChemBioChem 2019, 20, 2972–2980. [Google Scholar] [CrossRef]
- Feldberg, R.S.; Carew, J.A. Water radiolysis products and nucleotide damage in γ-irradiated DNA. Int. J. Radiat. Biol. 1981, 40, 11–17. [Google Scholar] [CrossRef]
- Misawa, M.; Takahashi, J. Generation of reactive oxygen species induced by gold nanoparticles under x-ray and UV Irradiations. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 604–614. [Google Scholar] [CrossRef]
- Shtarkman, I.N.; Gudkov, S.V.; Chernikov, A.V.; Bruskov, V.I. Effect of amino acids on X-ray-induced hydrogen peroxide and hydroxyl radical formation in water and 8-oxoguanine in DNA. Biochemistry 2008, 73, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Fulford, J.; Nikjoo, H.; Goodhead, D.T.; O’Neill, P. Yields of SSB and DSB induced in DNA by AlK ultrasoft X-rays and α-particles: Comparison of experimental and simulated yields. Int. J. Radiat. Biol. 2001, 77, 1053–1066. [Google Scholar] [CrossRef]
- Ward, J.F. Some biochemical consequences of the spatial distribution of ionizing radiation-produced free radicals. Radiat. Res. 1981, 86, 185–195. [Google Scholar] [CrossRef]
- Khanna, K.K.; Jackson, S.P. DNA double-strand breaks: Signaling, repair and the cancer connection. Nat. Genet. 2001, 27, 247–254. [Google Scholar] [CrossRef]
- Burma, S.; Chen, B.P.C.; Chen, D.J. Role of non-homologous end joining (NHEJ) in maintaining genomic integrity. DNA Repair 2006, 5, 1042–1048. [Google Scholar] [CrossRef]
- Rothkamm, K.; Kühne, M.; Jeggo, P.A.; Löbrich, M. Radiation-induced genomic rearrangements formed by nonhomologous end-joining of DNA double-strand breaks. Cancer Res. 2001, 61, 3886–3893. [Google Scholar]
- Morgan, W.F. Non-targeted and delayed effects of exposure to ionizing radiation: I. Radiation-induced genomic instability and bystander effects in Vitro. Radiat. Res. 2012, 178, 581–596. [Google Scholar] [CrossRef] [PubMed]
- De Santis, M.; Di Gianantonio, E.; Straface, G.; Cavaliere, A.F.; Caruso, A.; Schiavon, F.; Berletti, R.; Clementi, M. Ionizing radiations in pregnancy and teratogenesis: A review of literature. Reprod. Toxicol. 2005, 20, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Guven, M.; Brem, R.; Macpherson, P.; Peacock, M.; Karran, P. Oxidative Damage to RPA Limits the Nucleotide Excision Repair Capacity of Human Cells. J. Investig. Dermatol. 2015, 135, 2834–2841. [Google Scholar] [CrossRef]
- Schwob, L.; Lalande, M.; Rangama, J.; Egorov, D.; Hoekstra, R.; Pandey, R.; Eden, S.; Schlathölter, T.; Vizcaino, V.; Poully, J.C. Single-photon absorption of isolated collagen mimetic peptides and triple-helix models in the VUV-X energy range. Phys. Chem. Chem. Phys. 2017, 19, 18321–18329. [Google Scholar] [CrossRef]
- Pietraforte, D.; Paulicelli, E.; Patrono, C.; Gambardella, L.; Scorza, G.; Testa, A.; Fattibene, P. Protein oxidative damage and redox imbalance induced by ionising radiation in CHO cells. Free Radic. Res. 2018, 52, 465–479. [Google Scholar] [CrossRef]
- Prasad, N.R.; Menon, V.P.; Vasudev, V.; Pugalendi, K.V. Radioprotective effect of sesamol on γ-radiation induced DNA damage, lipid peroxidation and antioxidants levels in cultured human lymphocytes. Toxicology 2005, 209, 225–235. [Google Scholar] [CrossRef]
- Henderson, R. Cryo-protection of protein crystals against radiation damage in electron and X-ray diffraction. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1990, 241, 6–8. [Google Scholar] [CrossRef]
- Ravelli, R.B.G.; McSweeney, S.M. The “fingerprint” that X-rays can leave on structures. Structure 2000, 8, 315–328. [Google Scholar] [CrossRef]
- Weik, M.; Ravelli, R.B.G.; Kryger, G.; McSweeney, S.; Raves, M.L.; Harel, M.; Gros, P.; Silman, I.; Kroon, J.; Sussman, J.L. Specific chemical and structural damage to proteins produced by synchrotron radiation. Proc. Natl. Acad. Sci. USA 2000, 97, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Burmeister, W.P. Structural changes in a cryo-cooled protein crystal owing to radiation damage. Acta Crystallogr. Sect. D Biol. Crystallogr. 2000, 56, 328–341. [Google Scholar] [CrossRef]
- Dubnovitsky, A.P.; Ravelli, R.B.G.; Popov, A.N.; Papageorgiou, A.C. Strain relief at the active site of phosphoserine aminotransferase induced by radiation damage. Protein Sci. 2009, 14, 1498–1507. [Google Scholar] [CrossRef] [PubMed]
- Garman, E.F.; Weik, M. Radiation damage in macromolecular crystallography. Methods Mol. Biol. 2017, 1607, 467–489. [Google Scholar] [CrossRef]
- Matsui, Y.; Sakai, K.; Murakami, M.; Shiro, Y.; Adachi, S.I.; Okumura, H.; Kouyama, T. Specific damage induced by X-ray radiation and structural changes in the primary photoreaction of bacteriorhodopsin. J. Mol. Biol. 2002, 324, 469–481. [Google Scholar] [CrossRef]
- Mees, A.; Klar, T.; Gnau, P.; Hennecke, U.; Eker, A.P.M.; Carell, T.; Essen, L.O. Crystal structure of a photolyase bound to a CPD-like DNA lesion after in situ repair. Science 2004, 306, 1789–1793. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, E.; Vellieux, F.M.D.; Amara, P.; Madern, D.; Weik, M. Specific radiation damage to acidic residues and its relation to their chemical and structural environment. J. Synchrotron Radiat. 2007, 14, 84–91. [Google Scholar] [CrossRef]
- Sjöblom, B.; Polentarutti, M.; Djinović-Carugo, K. Structural study of X-ray induced activation of carbonic anhydrase. Proc. Natl. Acad. Sci. USA 2009, 106, 10609–10613. [Google Scholar] [CrossRef]
- Nass, K.; Foucar, L.; Barends, T.R.M.; Hartmann, E.; Botha, S.; Shoeman, R.L.; Doak, R.B.; Alonso-Mori, R.; Aquila, A.; Bajt, S.; et al. Indications of radiation damage in ferredoxin microcrystals using high-intensity X-FEL beams. J. Synchrotron Radiat. 2015, 22, 225–238. [Google Scholar] [CrossRef]
- Caldecott, K.W. Single-strand break repair and genetic disease. Nat. Rev. Genet. 2008, 9, 619–631. [Google Scholar] [CrossRef]
- David, S.S.; O’Shea, V.L.; Kundu, S. Base-excision repair of oxidative DNA damage. Nature 2007, 447, 941–950. [Google Scholar] [CrossRef]
- Lieber, M.R. The mechanism of DSB repair by the NHEJ. Annu. Rev. Biochem. 2011, 79, 181–211. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, A.T.; Palitti, F. DNA repair and chromosomal alterations. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2008, 657, 3–7. [Google Scholar] [CrossRef]
- Takata, M.; Sasaki, M.S.; Sonoda, E.; Morrison, C.; Hashimoto, M.; Utsumi, H.; Yamaguchi-Iwai, Y.; Shinohara, A.; Takeda, S. Homologous recombination and non-homologous end-joining pathways of DNA double-strand break repair have overlapping roles in the maintenance of chromosomal integrity in vertebrate cells. EMBO J. 1998, 17, 5497–5508. [Google Scholar] [CrossRef]
- Watson, R.E.B.; Gibbs, N.K.; Griffiths, C.E.M.; Sherratt, M.J. Damage to skin extracellular matrix induced by UV exposure. Antioxidants Redox Signal. 2014, 21, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Abe, O.; Abe, R.; Enomoto, K.; Kikuchi, K.; Koyama, H.; Masuda, H.; Nomura, Y.; Sakai, K.; Sugimachi, K.; Tominaga, T.; et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: An overview of the randomised trials. Lancet 2005, 366, 2087–2106. [Google Scholar] [CrossRef]
- Hamdy, F.C.; Donovan, J.L.; Lane, J.A.; Mason, M.; Metcalfe, C.; Holding, P.; Davis, M.; Peters, T.J.; Turner, E.L.; Martin, R.M.; et al. 10-Year Outcomes after Monitoring, Surgery, or Radiotherapy for Localized Prostate Cancer. N. Engl. J. Med. 2016, 375, 1415–1424. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.K.; Shen, D.; Sheikine, Y.; Ahsen, O.O.; Wang, H.H.; Schmolze, D.B.; Johnson, N.B.; Brooker, J.S.; Cable, A.E.; Connolly, J.L.; et al. Assessment of breast pathologies using nonlinear microscopy. Proc. Natl. Acad. Sci. USA 2014, 111, 15304–15309. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, K.B.; Son, Y.H.; Shin, J.; Lee, J.H.; Kim, H.J.; Hong, A.Y.; Bae, H.W.; Kwon, M.A.; Lee, W.J.; et al. Transglutaminase 2 mediates UV-induced skin inflammation by enhancing inflammatory cytokine production. Cell Death Dis. 2017, 8, e3148. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, A.; Walterova, D.; Vostalova, J. Ultraviolet light induced alteration to the skin. Biomed. Pap. Med. Fac. Univ. Palacky. Olomouc. Czech. Repub. 2006, 150, 25–38. [Google Scholar] [CrossRef]
- Fagot, D.; Asselineau, D.; Bernerd, F. Direct role of human dermal fibroblasts and indirect participation of epidermal keratinocytes in MMP-1 production after UV-B irradiation. Arch. Dermatol. Res. 2002, 293, 576–583. [Google Scholar] [CrossRef]
- Tewari, A.; Grys, K.; Kollet, J.; Sarkany, R.; Young, A.R. Upregulation of MMP12 and its activity by UVA1 in human skin: Potential implications for photoaging. J. Investig. Dermatol. 2014, 134, 2598–2609. [Google Scholar] [CrossRef]
- Seo, J.Y.; Lee, S.H.; Youn, C.S.; Choi, H.R.; Rhie, G.E.; Cho, K.H.; Kim, K.H.; Park, K.C.; Eun, H.C.; Chung, J.H. Ultraviolet radiation increases tropoelastin mRNA expression in the epidermis of human skin in vivo. J. Investig. Dermatol. 2001, 116, 915–919. [Google Scholar] [CrossRef]
- Juhl, P.; Bondesen, S.; Hawkins, C.L.; Karsdal, M.A.; Bay-Jensen, A.C.; Davies, M.J.; Siebuhr, A.S. Dermal fibroblasts have different extracellular matrix profiles induced by TGF-β, PDGF and IL-6 in a model for skin fibrosis. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Verrecchia, F.; Mauviel, A. Transforming growth factor-β signaling through the Smad pathway: Role in extracellular matrix gene expression and regulation. J. Investig. Dermatol. 2002, 118, 211–215. [Google Scholar] [CrossRef]
- Qin, Z.; Robichaud, P.; He, T.; Fisher, G.J.; Voorhees, J.J.; Quan, T.; Zhou, Z. Oxidant exposure induces cysteine-rich protein 61 (CCN1) via c-Jun/AP-1 to reduce collagen expression in human dermal fibroblasts. PLoS ONE 2014, 9, e0115402. [Google Scholar] [CrossRef] [PubMed]
- Borkham-Kamphorst, E.; Schaffrath, C.; Van de Leur, E.; Haas, U.; Tihaa, L.; Meurer, S.K.; Nevzorova, Y.A.; Liedtke, C.; Weiskirchen, R. The anti-fibrotic effects of CCN1/CYR61 in primary portal myofibroblasts are mediated through induction of reactive oxygen species resulting in cellular senescence, apoptosis and attenuated TGF-β signaling. Biochim. Biophys. Acta-Mol. Cell Res. 2014, 1843, 902–914. [Google Scholar] [CrossRef]
- Lee, C.H.; Shin, H.J.; Cho, I.H.; Kang, Y.M.; Kim, I.A.; Park, K.D.; Shin, J.W. Nanofiber alignment and direction of mechanical strain affect the ECM production of human ACL fibroblast. Biomaterials 2005, 26, 1261–1270. [Google Scholar] [CrossRef]
- Qin, Z.; Fisher, G.J.; Voorhees, J.J.; Quan, T. Actin cytoskeleton assembly regulates collagen production via TGF-β type II receptor in human skin fibroblasts. J. Cell. Mol. Med. 2018, 22, 4085–4096. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Shao, Y.; He, T.; Qin, Z.; Perry, D.; Voorhees, J.J.; Quan, T. Reduction of fibroblast size/mechanical force down-regulates TGF-β type II receptor: Implications for human skin aging. Aging Cell 2016, 15, 67–76. [Google Scholar] [CrossRef]
- Tamura, I.; Rosenbloom, J.; Macarak, E.; Chaqour, B. Regulation of Cyr61 gene expression by mechanical stretch through multiple signaling pathways. Am. J. Physiol.-Cell Physiol. 2001, 281, 1524–1532. [Google Scholar] [CrossRef]
- Cenizo, V.; André, V.; Reymermier, C.; Sommer, P.; Damour, O.; Perrier, E. LOXL as a target to increase the elastin content in adult skin: A dill extract induces the LOXL gene expression. Exp. Dermatol. 2006, 15, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Noblesse, E.; Cenizo, V.; Bouez, C.; Borel, A.; Gleyzal, C.; Peyrol, S.; Jacob, M.P.; Sommer, P.; Damour, O. Lysyl oxidase-like and lysyl oxidase are present in the dermis and epidermis of a skin equivalent and in human skin and are associated to elastic fibers. J. Investig. Dermatol. 2004, 122, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.; Jonason, A.S.; Leffellt, D.J.; Simon, J.A.; Sharma, H.W.; Kimmelman, J.; Remington, L.; Jacks, T.; Brash, D.E. Sunburn and p53 in the onset of skin cancer. Nature 1994, 372, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Bellono, N.W.; Oancea, E. UV light phototransduction depolarizes human melanocytes. Channels 2013, 7, 243–248. [Google Scholar] [CrossRef]
- Cui, R.; Widlund, H.R.; Feige, E.; Lin, J.Y.; Wilensky, D.L.; Igras, V.E.; D’Orazio, J.; Fung, C.Y.; Schanbacher, C.F.; Granter, S.R.; et al. Central Role of p53 in the Suntan Response and Pathologic Hyperpigmentation. Cell 2007, 128, 853–864. [Google Scholar] [CrossRef]
- Sherratt, M.J.; Bayley, C.P.; Reilly, S.M.; Gibbs, N.K.; Griffiths, C.E.M.; Watson, R.E.B. Low-dose ultraviolet radiation selectively degrades chromophore-rich extracellular matrix components. J. Pathol. 2010, 222, 32–40. [Google Scholar] [CrossRef]
- Dhital, B.; Durlik, P.; Rathod, P.; Gul-E-Noor, F.; Wang, Z.; Sun, C.; Chang, E.J.; Itin, B.; Boutis, G.S. Ultraviolet radiation reduces desmosine cross-links in elastin. Biochem. Biophys. Rep. 2017, 10, 172–177. [Google Scholar] [CrossRef]
- Besaratinia, A.; Pfeifer, G.P. Sunlight ultraviolet irradiation and BRAF V600 mutagenesis in human melanoma. Hum. Mutat. 2008, 29, 983–991. [Google Scholar] [CrossRef]
- Wisdom, K.M.; Indana, D.; Chou, P.E.; Desai, R.; Kim, T.; Chaudhuri, O. Covalent cross-linking of basement membrane-like matrices physically restricts invasive protrusions in breast cancer cells. Matrix Biol. 2020, 85–86, 94–111. [Google Scholar] [CrossRef]
- Kothari, C.; Diorio, C.; Durocher, F. The importance of breast adipose tissue in breast cancer. Int. J. Mol. Sci. 2020, 21, 5760. [Google Scholar] [CrossRef] [PubMed]
- Liakou, E.; Mavrogonatou, E.; Pratsinis, H.; Rizou, S.; Evangelou, K.; Panagiotou, P.N.; Karamanos, N.K.; Gorgoulis, V.G.; Kletsas, D. Ionizing radiation-mediated premature senescence and paracrine interactions with cancer cells enhance the expression of syndecan 1 in human breast stromal fibroblasts: The role of TGF-β. Aging 2016, 8, 1650–1669. [Google Scholar] [CrossRef]
- Tsai, K.K.C.; Chuang, E.Y.Y.; Little, J.B.; Yuan, Z.M. Cellular mechanisms for low-dose ionizing radiation-induced perturbation of the breast tissue microenvironment. Cancer Res. 2005, 65, 6734–6744. [Google Scholar] [CrossRef]
- Giannelli, G.; Falk-Marzillier, J.; Schiraldi, O.; Stetler-Stevenson, W.G.; Quaranta, V. Induction of cell migration by matrix metalloprotease-2 cleavage of laminin-5. Science 1997, 277, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Koshikawa, N.; Giannelli, G.; Cirulli, V.; Miyazaki, K.; Quaranta, V. Role of cell surface metalloprotease MT1-MMP in epithelial cell migration over laminin-5. J. Cell Biol. 2000, 148, 615–624. [Google Scholar] [CrossRef]
- Ehrhart, E.J.; Segarini, P.; Tsang, M.L.-S.; Carroll, A.G.; Barcellos-Hoff, M.H. Latent transforming growth factor β1 activation in situ: Quantitative and functional evidence after low-dose γ-irradiation 1. FASEB J. 1997, 11, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- Herskind, C.; Rodemann, H.P. Spontaneous and radiation-induced differentiationof fibroblasts. Exp. Gerontol. 2000, 35, 747–755. [Google Scholar] [CrossRef]
- Paquette, B.; Baptiste, C.; Therriault, H.; Arguin, G.; Plouffe, B.; Lemay, R. In vitro irradiation of basement membrane enhances the invasiveness of breast cancer cells. Br. J. Cancer 2007, 97, 1505–1512. [Google Scholar] [CrossRef]
- Jones, J.I.; Gockerman, A.; Busby, W.H.; Camacho-Hubner, C.; Clemmons, D.R. Extracellular matrix contains insulin-like growth factor binding protein-5: Potentiation of the effects of IGF-I. J. Cell Biol. 1993, 121, 679–687. [Google Scholar] [CrossRef]
- Hinz, B. The extracellular matrix and transforming growth factor-β1: Tale of a strained relationship. Matrix Biol. 2015, 47, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Mettler, F.A. Medical effects and risks of exposure to ionising radiation. J. Radiol. Prot. 2012, 32, N9–N13. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D.; Salomaa, S.; Bouffler, S.; Ottolenghi, A.; Smyth, V.; Sabatier, L. Progress in low dose health risk research: Novel effects and new concepts in low dose radiobiology. Mutat. Res.-Rev. Mutat. Res. 2018, 776, 46–69. [Google Scholar] [CrossRef] [PubMed]
- Kole, A.J.; Kole, L.; Moran, M.S. Acute radiation dermatitis in breast cancer patients: Challenges and solutions. Breast Cancer Targets Ther. 2017, 9, 313–323. [Google Scholar] [CrossRef]
- Dewey, W.C.; Ling, C.C.; Meyn, R.E. Radiation-induced apoptosis: Relevance to radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1995, 33, 781–796. [Google Scholar] [CrossRef]
- Martin, A.M.; Cagney, D.N.; Catalano, P.J.; Alexander, B.M.; Redig, A.J.; Schoenfeld, J.D.; Aizer, A.A. Immunotherapy and symptomatic radiation necrosis in patients with brain metastases treated with stereotactic radiation. JAMA Oncol. 2018, 4, 1123–1124. [Google Scholar] [CrossRef]
- LaRiviere, M.J.; Chao, H.H.; Doucette, A.; Kegelman, T.P.; Taunk, N.K.; Freedman, G.M.; Vapiwala, N. Factors Associated With Fatigue in Patients with Breast Cancer Undergoing External Beam Radiation Therapy. Pract. Radiat. Oncol. 2020, 10, 409–422. [Google Scholar] [CrossRef]
- Hille-Betz, U.; Vaske, B.; Bremer, M.; Soergel, P.; Kundu, S.; Klapdor, R.; Hillemanns, P.; Henkenberens, C. Late radiation side effects, cosmetic outcomes and pain in breast cancer patients after breast-conserving surgery and three-dimensional conformal radiotherapy. Strahlentherapie und Onkol. 2016, 192, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Baverstock, K.; Karotki, A.V. Towards a unifying theory of late stochastic effects of ionizing radiation. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2011, 718, 1–9. [Google Scholar] [CrossRef]
- Abbotts, R.; Wilson, D.M. Coordination of DNA single strand break repair. Free Radic. Biol. Med. 2017, 107, 228–244. [Google Scholar] [CrossRef]
- Patel, A.; Jackson, B. Low-dose radiation use in diagnostic imaging and cancer therapy settings. Radiol. Medica 2018, 123, 618–619. [Google Scholar] [CrossRef]
- Liu, D.P.; Song, H.; Xu, Y. A common gain of function of p53 cancer mutants in inducing genetic instability. Oncogene 2010, 29, 949–956. [Google Scholar] [CrossRef]
- Hainaut, P.; Hollstein, M. p53 and Human Cancer: The First Ten Thousand Mutations. Adv. Cancer Res. 1999, 77, 81–86. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.A.; Quan, T.; Voorhees, J.J.; Fisher, G.J. Extracellular matrix regulation of fibroblast function: Redefining our perspective on skin aging. J. Cell Commun. Signal. 2018, 12, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular Matrix Degradation and Remodeling in Development and Disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, S.D.; Endicott, S.K.; Province, M.A.; Pierce, J.A.; Campbell, E.J. Marked longevity of human lung parenchymal elastic fibers deduced from prevalence of D-aspartate and nuclear weapons-related radiocarbon. J. Clin. Investig. 1991, 87, 1828–1834. [Google Scholar] [CrossRef] [PubMed]
- Kielty, C.M.; Sherratt, M.J.; Marson, A.; Baldock, C. Fibrillin microfibrils. Adv. Protein Chem. 2005, 70, 405–436. [Google Scholar] [CrossRef]
- Sherratt, M.J. Tissue elasticity and the ageing elastic fibre. Age 2009, 31, 305–325. [Google Scholar] [CrossRef]
- Verzijl, N.; DeGroot, J.; Thorpe, S.R.; Bank, R.A.; Shaw, J.N.; Lyons, T.J.; Bijlsma, J.W.J.; Lafeber, F.P.J.G.; Baynes, J.W.; TeKoppele, J.M. Effect of collagen turnover on the accumulation of advanced glycation end products. J. Biol. Chem. 2000, 275, 39027–39031. [Google Scholar] [CrossRef]
- Darby, S.C.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Brønnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of Ischemic Heart Disease in Women after Radiotherapy for Breast Cancer. N. Engl. J. Med. 2013, 368, 987–998. [Google Scholar] [CrossRef]
- Takeuchi, H.; Kimura, T.; Okamoto, K.; Aoyagi, E.; Miyamoto, H.; Kaji, M.; Takenaka, H.; Okamura, S.; Sato, Y.; Kato, J.; et al. A mechanism for abnormal angiogenesis in human radiation proctitis: Analysis of expression profile for angiogenic factors. J. Gastroenterol. 2012, 47, 56–64. [Google Scholar] [CrossRef]
- Tsoutsou, P.G.; Koukourakis, M.I. Radiation pneumonitis and fibrosis: Mechanisms underlying its pathogenesis and implications for future research. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 1281–1293. [Google Scholar] [CrossRef]
- Yoo, K.C.; Suh, Y.; An, Y.; Lee, H.J.; Jeong, Y.J.; Uddin, N.; Cui, Y.H.; Roh, T.H.; Shim, J.K.; Chang, J.H.; et al. Proinvasive extracellular matrix remodeling in tumor microenvironment in response to radiation. Oncogene 2018, 37, 3317–3328. [Google Scholar] [CrossRef]
- Miller, J.P.; Borde, B.H.; Bordeleau, F.; Zanotelli, M.R.; LaValley, D.J.; Parker, D.J.; Bonassar, L.J.; Pannullo, S.C.; Reinhart-King, C.A. Clinical doses of radiation reduce collagen matrix stiffness. APL Bioeng. 2018, 2, 031901. [Google Scholar] [CrossRef]
- Hubbard, B.; Buczek-Thomas, J.A.; Nugent, M.A.; Smith, M.L. Fibronectin Fiber Extension Decreases Cell Spreading and Migration. J. Cell. Physiol. 2016, 231, 1728–1736. [Google Scholar] [CrossRef]
- Dong, Y.; Zheng, Q.; Wang, Z.; Lin, X.; You, Y.; Wu, S.; Wang, Y.; Hu, C.; Xie, X.; Chen, J.; et al. Higher matrix stiffness as an independent initiator triggers epithelial-mesenchymal transition and facilitates HCC metastasis. J. Hematol. Oncol. 2019, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.C.; Fattet, L.; Tsai, J.H.; Guo, Y.; Pai, V.H.; Majeski, H.E.; Chen, A.C.; Sah, R.L.; Taylor, S.S.; Engler, A.J.; et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway. Nat. Cell Biol. 2015, 17, 678–688. [Google Scholar] [CrossRef]
- Junker, J.P.E.; Kratz, C.; Tollbäck, A.; Kratz, G. Mechanical tension stimulates the transdifferentiation of fibroblasts into myofibroblasts in human burn scars. Burns 2008, 34, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Chu, J.S.; Tsou, A.D.; Diop, R.; Tang, Z.; Wang, A.; Li, S. The effect of matrix stiffness on the differentiation of mesenchymal stem cells in response to TGF-β. Biomaterials 2011, 32, 3921–3930. [Google Scholar] [CrossRef]
- Ulrich, T.A.; De Juan Pardo, E.M.; Kumar, S. The mechanical rigidity of the extracellular matrix regulates the structure, motility, and proliferation of glioma cells. Cancer Res. 2009, 69, 4167–4174. [Google Scholar] [CrossRef] [PubMed]
- Hibbert, S.A.; Watson, R.E.B.; Griffiths, C.E.M.; Gibbs, N.K.; Sherratt, M.J. Selective proteolysis by matrix metalloproteinases of photo-oxidised dermal extracellular matrix proteins. Cell. Signal. 2019, 54, 191–199. [Google Scholar] [CrossRef]
- Taipale, J.; Keski-Oja, J. Growth factors in the extracellular matrix. FASEB J. 1997, 11, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Annes, J.P.; Chen, Y.; Munger, J.S.; Rifkin, D.B. Integrin αvβ6-mediated activation of latent TGF-β requires the latent TGF-β binding protein-1. J. Cell Biol. 2004, 165, 723–734. [Google Scholar] [CrossRef] [PubMed]
- Hawinkels, L.J.A.C.; Zuidwijk, K.; Verspaget, H.W.; de Jonge-Muller, E.S.M.; van Duijn, W.; Ferreira, V.; Fontijn, R.D.; David, G.; Hommes, D.W.; Lamers, C.B.H.W.; et al. VEGF release by MMP-9 mediated heparan sulphate cleavage induces colorectal cancer angiogenesis. Eur. J. Cancer 2008, 44, 1904–1913. [Google Scholar] [CrossRef]
- Schultz, G.S.; Wysocki, A. Interactions between extracellular matrix and growth factors in wound healing. Wound Repair Regen. 2009, 17, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.; Grose, R. Fibroblast growth factor signalling: From development to cancer. Nat. Rev. Cancer 2010, 10, 116–129. [Google Scholar] [CrossRef]
- Walton, K.L.; Johnson, K.E.; Harrison, C.A. Targeting TGF-β mediated SMAD signaling for the prevention of fibrosis. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Kisling, A.; Lust, R.M.; Katwa, L.C. What is the role of peptide fragments of collagen I and IV in health and disease? Life Sci. 2019, 228, 30–34. [Google Scholar] [CrossRef]
- Duca, L.; Floquet, N.; Alix, A.J.P.; Haye, B.; Debelle, L. Elastin as a matrikine. Crit. Rev. Oncol. Hematol. 2004, 49, 235–244. [Google Scholar] [CrossRef]
- Wells, J.M.; Gaggar, A.; Blalock, J.E. MMP generated matrikines. Matrix Biol. 2015, 44–46, 122–129. [Google Scholar] [CrossRef]
- Jariashvili, K.; Madhan, B.; Brodsky, B.; Kuchava, A.; Namicheishvili, L.; Metreveli, N. Uv damage of collagen: Insights from model collagen peptides. Biopolymers 2012, 97, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K. Analysis of ultraviolet radiation wavelengths causing hardening and reduced elasticity of collagen gels in vitro. Cosmetics 2018, 5, 14. [Google Scholar] [CrossRef]
- Mao, X.W.; Mekonnen, T.; Kennedy, A.R.; Gridley, D.S. Differential expression of oxidative stress and extracellular matrix remodeling genes in low- or high-dose-rate photon-irradiated skin. Radiat. Res. 2011, 176, 187–197. [Google Scholar] [CrossRef]
- Zhou, G. Mechanisms underlying FLASH radiotherapy, a novel way to enlarge the differential responses to ionizing radiation between normal and tumor tissues. Radiat. Med. Prot. 2020, 1, 35–40. [Google Scholar] [CrossRef]
- Svojtková, E.; Deyl, Z.; Adam, M. Aging of connective tissue. Age dependency of collagen depolymerization by ionizing radiation. Exp. Gerontol. 1973, 8, 157–164. [Google Scholar] [CrossRef]
- Bowes, J.H.; Moss, J.A. The Effect of Gamma Radiation on Collagen. Radiat. Res. 1962, 16, 211. [Google Scholar] [CrossRef]
- Alves, S.M.; Zhu, T.; Shostak, A.; Rossen, N.S.; Rafat, M. Studying normal tissue radiation effects using extracellular matrix hydrogels. J. Vis. Exp. 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Giobbe, G.G.; Crowley, C.; Luni, C.; Campinoti, S.; Khedr, M.; Kretzschmar, K.; De Santis, M.M.; Zambaiti, E.; Michielin, F.; Meran, L.; et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat. Commun. 2019, 10, 5658. [Google Scholar] [CrossRef]
- Gosztyla, C.; Ladd, M.R.; Werts, A.; Fulton, W. A Comparison of Sterilization Techniques for Production of Decellularized Intestine in Mice. Tissue Eng. Part C Methods 2020, 26, 67–79. [Google Scholar] [CrossRef]
- Poornejad, N.; Nielsen, J.J.; Morris, R.J.; Gassman, J.R.; Reynolds, P.R.; Roeder, B.L.; Cook, A.D. Comparison of four decontamination treatments on porcine renal decellularized extracellular matrix structure, composition, and support of renal tubular epithelium cells. J. Biomater. Appl. 2016, 30, 1154–1167. [Google Scholar] [CrossRef]
- Moradi, L.; Mohammadi Jobania, B.; Jafarnezhad-Ansariha, F.; Ghorbani, F.; Esmaeil-Pour, R.; Majidi Zolbina, M.; Kajbafzadeh, A.M. Evaluation of different sterilization methods for decellularized kidney tissue. Tissue Cell 2020, 66, 101396. [Google Scholar] [CrossRef] [PubMed]
- XIslam, M.M.; Sharifi, R.; Mamodaly, S.; Islam, R.; Nahra, D.; Abusamra, D.B.; Hui, P.C.; Adibnia, Y.; Goulamaly, M.; Paschalis, E.I.; et al. Effects of gamma radiation sterilization on the structural and biological properties of decellularized corneal xenografts. Acta Biomater. 2019, 96, 330–344. [Google Scholar] [CrossRef]
- Riekki, R.; Jukkola, A.; Oikarinen, A.; Kallioinen, M. Radiation Therapy Induces Tenascin Expression and Angiogenesis in Human Skin. Acta Derm. Venereol. 2001, 81, 329–333. [Google Scholar] [CrossRef]
- Pendleton, M.M.; Emerzian, S.R.; Liu, J.; Tang, S.Y.; O’Connell, G.D.; Alwood, J.S.; Keaveny, T.M. Effects of ex vivo ionizing radiation on collagen structure and whole-bone mechanical properties of mouse vertebrae. Bone 2019, 128, 115043. [Google Scholar] [CrossRef]
- Mohamed, F.; Bradley, D.A.; Winlove, C.P. Effects of ionizing radiation on extracellular matrix. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrom. Detect. Assoc. Equip. 2007, 580, 566–569. [Google Scholar] [CrossRef]
- Koontz, B.F.; Verhaegen, F.; De Ruysscher, D. Tumour and normal tissue radiobiology in mouse models: How close are mice to mini-humans? Br. J. Radiol. 2017, 90, 20160441. [Google Scholar] [CrossRef] [PubMed]
- Son, E.D.; Shim, J.H.; Choi, H.; Kim, H.; Lim, K.M.; Chung, J.H.; Byun, S.Y.; Lee, T.R. Cathepsin G inhibitor prevents ultraviolet B-induced photoaging in hairless mice via inhibition of fibronectin fragmentation. Dermatology 2012, 224, 352–360. [Google Scholar] [CrossRef]
- Myronchenko, S.; Zvyagintseva, T.; Ashukina, N. the Effect of Ultraviolet Radiation on the Organization and Structure of Collagen Fibers of Dermis. Georgian Med. News 2020, 302, 82–85. [Google Scholar]
- Rock, K.; Joosse, S.A.; Muller, J.; Heinisch, N.; Fuchs, N.; Meusch, M.; Zipper, P.; Reifenberger, J.; Pantel, K.; Fischer, J.W. Chronic UVB-irradiation actuates perpetuated dermal matrix remodeling in female mice: Protective role of estrogen. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Kotova, S.L.; Timashev, P.S.; Belkova, G.V.; Kochueva, M.V.; Babak, K.V.; Timofeeva, V.A.; Kiseleva, E.B.; Vasilieva, O.O.; Maslennikova, A.V.; Solovieva, A.B. Early Effects of Ionizing Radiation on the Collagen Hierarchical Structure of Bladder and Rectum Visualized by Atomic Force Microscopy. Microsc. Microanal. 2018, 24, 38–48. [Google Scholar] [CrossRef]
- Flanders, K.C.; Sullivan, C.D.; Fujii, M.; Sowers, A.; Anzano, M.A.; Arabshahi, A.; Major, C.; Deng, C.; Russo, A.; Mitchell, J.B.; et al. Mice lacking Smad3 are protected against cutaneous injury induced by ionizing radiation. Am. J. Pathol. 2002, 160, 1057–1068. [Google Scholar] [CrossRef]
- Maslennikova, A.; Kochueva, M.; Ignatieva, N.; Vitkin, A.; Zakharkina, O.; Kamensky, V.; Sergeeva, E.; Kiseleva, E.; Bagratashvili, V. Effects of gamma irradiation on collagen damage and remodeling. Int. J. Radiat. Biol. 2015, 91, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Eckersley, A.; Mellody, K.T.; Pilkington, S.; Griffiths, C.E.M.; Watson, R.E.B.; O’Cualain, R.; Baldock, C.; Knight, D.; Sherratt, M.J. Structural and compositional diversity of fibrillin microfibrils in human tissues. J. Biol. Chem. 2018, 293, 5117–5133. [Google Scholar] [CrossRef]
- Ozols, M.; Eckersley, A.; Mellody, K.T.; Mallikarjun, V.; Warwood, S.; O’Cualain, R.; Knight, D.; Watson, R.E.B.; Griffiths, C.E.M.; Swift, J.; et al. Peptide location fingerprinting reveals modification-associated biomarker candidates of ageing in human tissue proteomes. Aging Cell 2021, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Poobalarahi, F.; Baicu, C.F.; Bradshaw, A.D. Cardiac myofibroblasts differentiated in 3D culture exhibit distinct changes in collagen I production, processing, and matrix deposition. Am. J. Physiol.-Hear Circ. Physiol. 2006, 291, 2924–2932. [Google Scholar] [CrossRef] [PubMed]
- Swamydas, M.; Eddy, J.M.; Burg, K.J.L.; Dréau, D. Matrix compositions and the development of breast acini and ducts in 3D cultures. Vitr. Cell. Dev. Biol.-Anim. 2010, 46, 673–684. [Google Scholar] [CrossRef]
- Douki, T.; Perdiz, D.; Gróf, P.; Kuluncsics, Z.; Moustacchi, E.; Cadet, J.; Sage, E. Oxidation of guanine in cellular DNA by solar UV radiation: Biological role. Photochem. Photobiol. 1999, 70, 184–190. [Google Scholar] [CrossRef]
- Douki, T.; Reynaud-Angelin, A.; Cadet, J.; Sage, E. Bipyrimidine photoproducts rather than oxidative lesions are the main type of DNA damage involved in the genotoxic effect of solar UVA radiation. Biochemistry 2003, 42, 9221–9226. [Google Scholar] [CrossRef]
- Shin, J.W.; Kwon, S.H.; Choi, J.Y.; Na, J.I.; Huh, C.H.; Choi, H.R.; Park, K.C. Molecular mechanisms of dermal aging and antiaging approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef]
- Valentin, J. Relative biological effectiveness (RBE), quality factor (Q), and radiation weighting factor (wR). Ann ICRP. 2003, 33, 1–121. [Google Scholar] [CrossRef]
| Type | Environmental Exposure | Clinical Exposure | Biological Consequence | |
|---|---|---|---|---|
| Ionising | X-rays/Gamma rays | Cosmic radiation [10], Radon gas [11] | Diagnostic imaging [12], Radiotherapy [13] | Fibrosis [14], Carcinogenesis [15] |
| Non-ionising | UVR | Sunlight [16] | UVR Phototherapy [17] | Skin photoageing [18], Vitamin D synthesis [19] |
| Visible light | Sunlight [20] | Photodynamic therapy [21] | Ocular phototoxicity [20] | |
| Infrared | Sunlight [22] | Neural stimulation [22] | Skin photoageing [23] | |
| Radiowaves | Lightning [24] | Hyperthermia [25] | Brain activity [26] |
| Radiation | Dose | Method | Ref. | Results |
|---|---|---|---|---|
| UV | UV (254 nm), 24.0 J/cm2, 102.0 J/cm2, 396.0 J/cm2 | Collagen model peptides and rat tail tendon collagen I | [200] | Rat tail collagen exhibited stable intermediate after irradiation. Gly-Pro-Hyp mimetic collagen was more stable than Gly-Pro-Pro, while Gly-Ala-Hyp was more stable than Gly-Pro-Hyp. |
| UV (254 nm), 5–187 J/cm2 | Sterile rat tail collagen I | [70] | Collagen denatures with loss of hydrogen bonds with water molecules, followed by the loss of triple helix and peptide bond cleavage. | |
| Broadband UVB (270–380 nm) 0.1 J/cm2, Solar radiation (SSR), 30 J/cm2 | Fibrillin/Collagen VI microfibrils derived and purified from human dermal fibroblasts. Peptide mass fingerprinting | [74] | No changes for collagen IV. UVB/SSR increased protease susceptibility for fibrillin, possibly from ultrastructural changes. | |
| Broadband UVB (290–320 nm) 3.2–9.6 J/cm2, BL/DMR lamps (320–400 nm) 49–147 J/cm2 | Bovine dermis native collagen, made into collagen gels using sodium bicarbonate | [201] | UVR at 300–340 nm caused hardening and reduced elasticity of collagen gels, and 330 nm gave the greatest effect. Increase in tyrosine cross-linking was found. | |
| UVB (280–315 nm), 20–500 mJ/cm2 | Purified collagen-1, fibrillin microfibrils from biopsy/COS-1 cells, fibronectin from bovine plasma | [146] | UVB dose required to damage ultrastructure decreases with greater chromophore composition. Collagen I was the most UVB-resistant, followed by fibronectin and then fibrillin. | |
| UVA (365 nm). 9330 J/cm2 | Isolated bovine nuchal ligament elastic fibres | [147] | No 13C NMR shifts detected, 11% reduction of desmosine from cross-link cleavage. | |
| Ionising radiation | Co-60 γ-ray at 1.289k Gy/h, 5k–50k Gy | Lyophilised collagen from rat tail tendon irradiated and tested for solubility and melting temperature | [204] | Irradiated samples were, in general, more than twice as soluble as non-irradiated in 0.02 M acetic acid, 6 M lithium chloride and 6 M urea. Melting temperature reduces with increasing dose. |
| γ-ray (1 MeV), 50k–500k Gy | Grounded collagen irradiated in dry/wet (5%/80% moisture) state in the presence and absence of oxygen/nitrogen | [205] | Solubility unchanged when irradiated wet due to cross-linking, and solubility increased when irradiated dry. Significant molecular changes likely due to the breakage of peptide bonds. Degradation of Tyr; Hyp/Pro; Asp sensitive to oxygen/nitrogen. | |
| Near X-ray (13.8–22.1 eV) | Isolated collagen mimetic peptides, photon absorption in gas phase + mass spectrometry | [105] | Gly-Pro peptide bonds are more susceptible to cleavage, collagen triple helix stabilised by hydroxyproline. |
| Radiation | Dose | Method | Ref. | Results |
|---|---|---|---|---|
| UV | UV (254 nm) using UV cross-linker, 2 cycles (90 s each) | Decellularised Lewis rat intestines | [208] | No significant change in collagen/GAG content. Loss of villous ECM projections. |
| γ-ray (wavelength unspecified, 5000 Gy) | Rabbit kidney decellularised | [210] | Reduced tensile strength and young’s modulus with gamma ray. | |
| Ionising Radiation | Co-60 γ-ray, 25k Gy | Gamma irradiation of decellularised cornea | [211] | Increased stiffness/tensile strength, reduced elongation at break after irradiation, due to fragmented collagen cross-linking. |
| Cs-137 γ-ray, 1k–10k Gy | Decellularised whole porcine kidney | [209] | 3k Gy resulted in more than 50% loss in collagen content. Human renal cortical tubular epithelium (RCTE) cells reseeded and resulted in poor adhesion/growth. | |
| Cs-137 γ-ray, 20 Gy | Murine mammary fat pads decellularised and made into hydrogels. | [206] | Increased proliferation for murine TNBC reseeded on irradiated hydrogel. |
| Radiation | Dose | Method | Ref. | Results |
|---|---|---|---|---|
| UV | UVA (365 nm, 1.5 mw/cm2), UV-B (302 nm, 1.6 mw/cm2), UV-C (265 nm, 1.8 mw/cm2), dosage: 10–4000 J/cm2 | Stratum corneum from breast skin tissue extracted | [18] | Reduced stiffness, fracture stress/strain, at >4000 J/cm2 UVA and >400 J/cm2 UVB. The energy required to fracture decreases in a dose-dependent manner. |
| Cs-137 γ-ray, 10–63 Gy | Mammary tumours (MMTV-PyMT transgenic mice) immediately irradiated and frozen before tested for compression | [183] | Significantly reduced tensile and compression modulus after 60 Gy irradiation (fractionated and single dose). | |
| Ionising Radiation | 6–10 MeV X-rays, 30–56 Gy | Biopsy from radiation therapy treated breast cancer patients. Irradiated/non irradiated samples from the same patient 10-96 months after treatment | [212] | No observable change in elastic fibres/collagen, but stiffness is higher for irradiated regions. |
| 21 KeV X-rays, 50–35,000 Gy | Lumbar vertebrae excised and removed of soft tissue. Wrapped in saline-soaked gauze | [213] | Monotonic strength (one direction) decreased at 17,000 Gy and above. Increase in non-enzymatic cross-links at a lower dose (50–1000 Gy) by analysing AGEs. Crosslinks do not have a significant impact on vertebral strength. | |
| 6 MeV X-rays, 10–100 Gy | Bovine pericardial tissue (collagen), Bovine ligamentum nuchae (elastin) | [214] | For pericardial tissue, elastic modulus increased for small strain and decreased at larger strain after irradiation. Elastin has significantly reduced elastic modulus after irradiation. |
| Radiation | Dose | Method | Ref | Results |
|---|---|---|---|---|
| UV | UVB (285–350 nm, peak: 310 nm). 0.12 J/cm2, (MED) ×3/×6/×9 per week for 13 weeks | Skh1/Hr female mice irradiated with UVR over 13 weeks with increasing dose | [216] | KPA inhibited cathepsin G, which mediates MMP-1 upregulation through Fn fragmentation/activating pro-MMP-1. |
| UVA/B (240–320 nm), 1 MED | Albino guinea pigs (400–500 g) irradiated on shaved skin and decapitated 2/4/72/192 h after irradiation | [217] | Disorganisation of collagen I/II fibres worsen over time. Increase in collagen III detected. | |
| UVB (280–320 nm, peak: 313 nm). 0.08 J/cm2 (1 MED), 3 times per week for 20 weeks | Skh1/Hr female mice 8 weeks old irradiated with UVR over 20 weeks and allowed to recover for 10 weeks. Dorsal skin biopsies were taken at week 28 and 38 | [218] | After 20 weeks of irradiation, there was a 35% reduction in collagen content. Collagen further declined during recovery by ~70%. mRNA levels of MMP-3 and 9 were not regulated, while mRNA of MMP-13 decreased. Possible degradation of collagen by the activation of latent MMP rather than increased expression. | |
| Co-60 γ-ray, 2–22 Gy in fractions of 2 Gy/day | White, outbred rats, irradiated in bladder and rectum. For 2 Gy, rats were harvested 1 day/1 week/1 month after irradiation. Higher doses harvested after 1 day. | [219] | One-month post-2 Gy irradiation showed thickening of collagen fibres and tight, parallel packing for the bladder and rectum. One day post-irradiation for higher dose observed the same effects with the severity dependent on dose. Skin most sensitive showing similar damage at 8 Gy. | |
| Ionising Radiation | 300 kVp X-rays (30–60 Gy) for local, Cs-137 γ-ray (6–10 Gy) for whole body | C57BL/6 mice with smad3 gene knockout | [220] | Smad3 knockout mice have less TGF-β1 expression, less inflammation, less myofibroblasts after radiation |
| Co-60 γ-ray, 2–40 Gy, 1.7 Gy/min | 2-month-old, white wild type outbred rats, ~ 200 g, harvested 1 day/1 week/1,2,3 months after irradiation for rat’s tail tendon | [221] | Differential scanning calorimetry showed a dose-dependant increase in denaturing temperature 24 h after irradiation, but dose-independent after 1 week. Negligible change was observed for tertiary/secondary structures using second harmonic generation/cross-polarisation optical coherence tomography |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuieng, R.J.; Cartmell, S.H.; Kirwan, C.C.; Sherratt, M.J. The Effects of Ionising and Non-Ionising Electromagnetic Radiation on Extracellular Matrix Proteins. Cells 2021, 10, 3041. https://doi.org/10.3390/cells10113041
Tuieng RJ, Cartmell SH, Kirwan CC, Sherratt MJ. The Effects of Ionising and Non-Ionising Electromagnetic Radiation on Extracellular Matrix Proteins. Cells. 2021; 10(11):3041. https://doi.org/10.3390/cells10113041
Chicago/Turabian StyleTuieng, Ren Jie, Sarah H. Cartmell, Cliona C. Kirwan, and Michael J. Sherratt. 2021. "The Effects of Ionising and Non-Ionising Electromagnetic Radiation on Extracellular Matrix Proteins" Cells 10, no. 11: 3041. https://doi.org/10.3390/cells10113041
APA StyleTuieng, R. J., Cartmell, S. H., Kirwan, C. C., & Sherratt, M. J. (2021). The Effects of Ionising and Non-Ionising Electromagnetic Radiation on Extracellular Matrix Proteins. Cells, 10(11), 3041. https://doi.org/10.3390/cells10113041






