Cytogenetic Evidence Clarifies the Phylogeny of the Family Rhynchocyclidae (Aves: Passeriformes)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Specimens and Chromosome Preparation
2.2. Giemsa Staining
2.3. Fluorescence In Situ Hybridization (FISH)
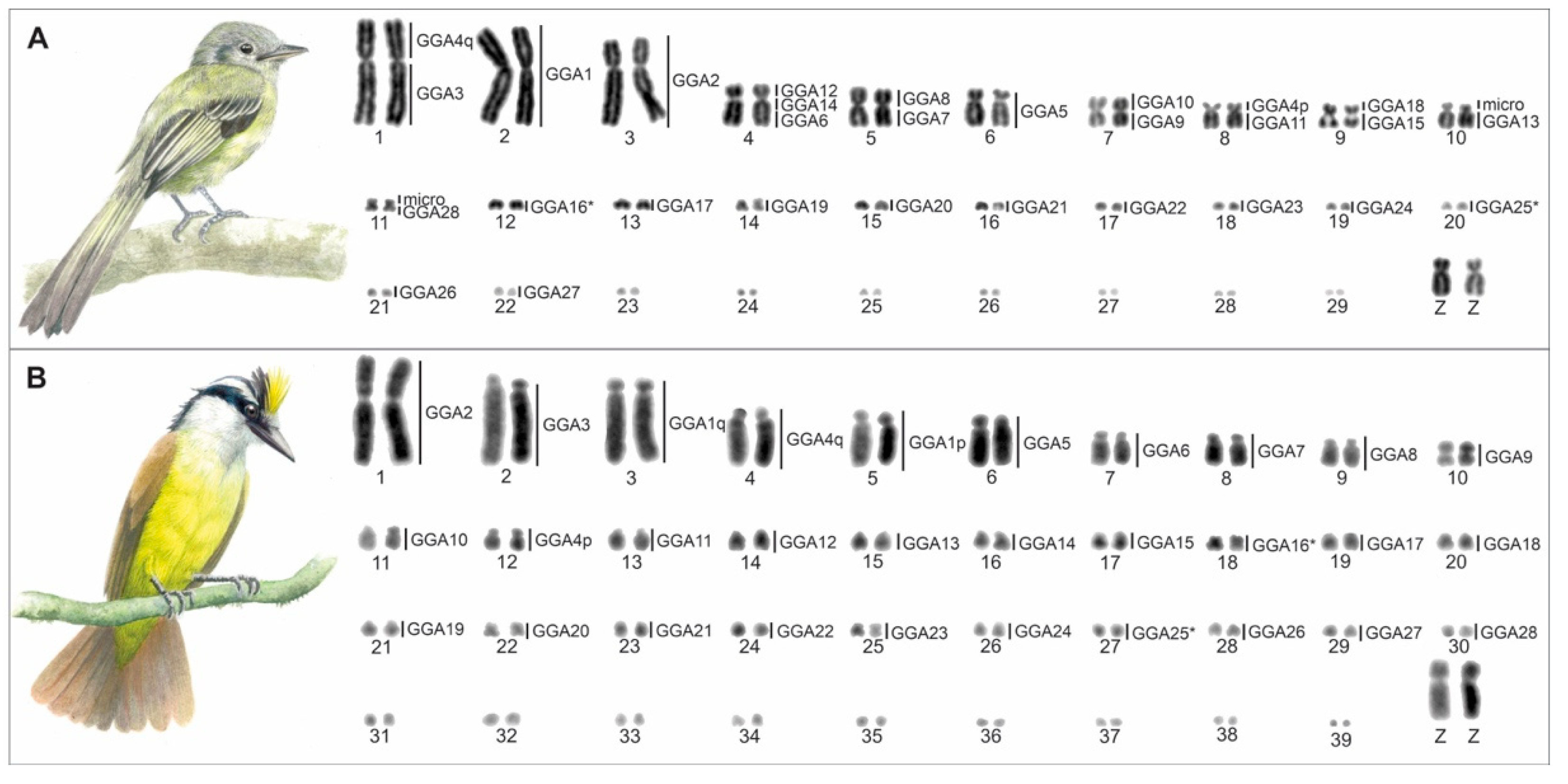
3. Results
3.1. Karyotype Description
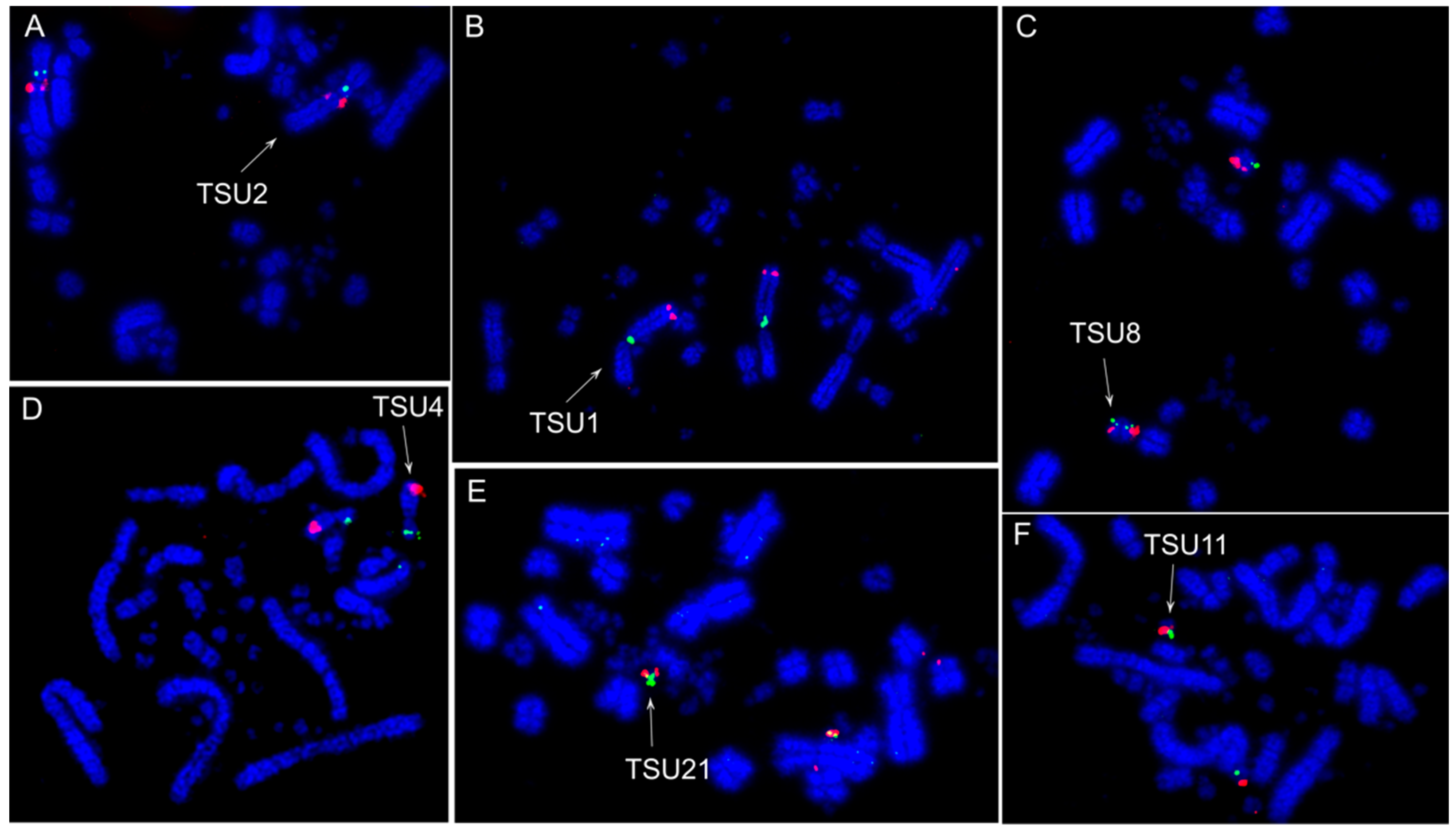
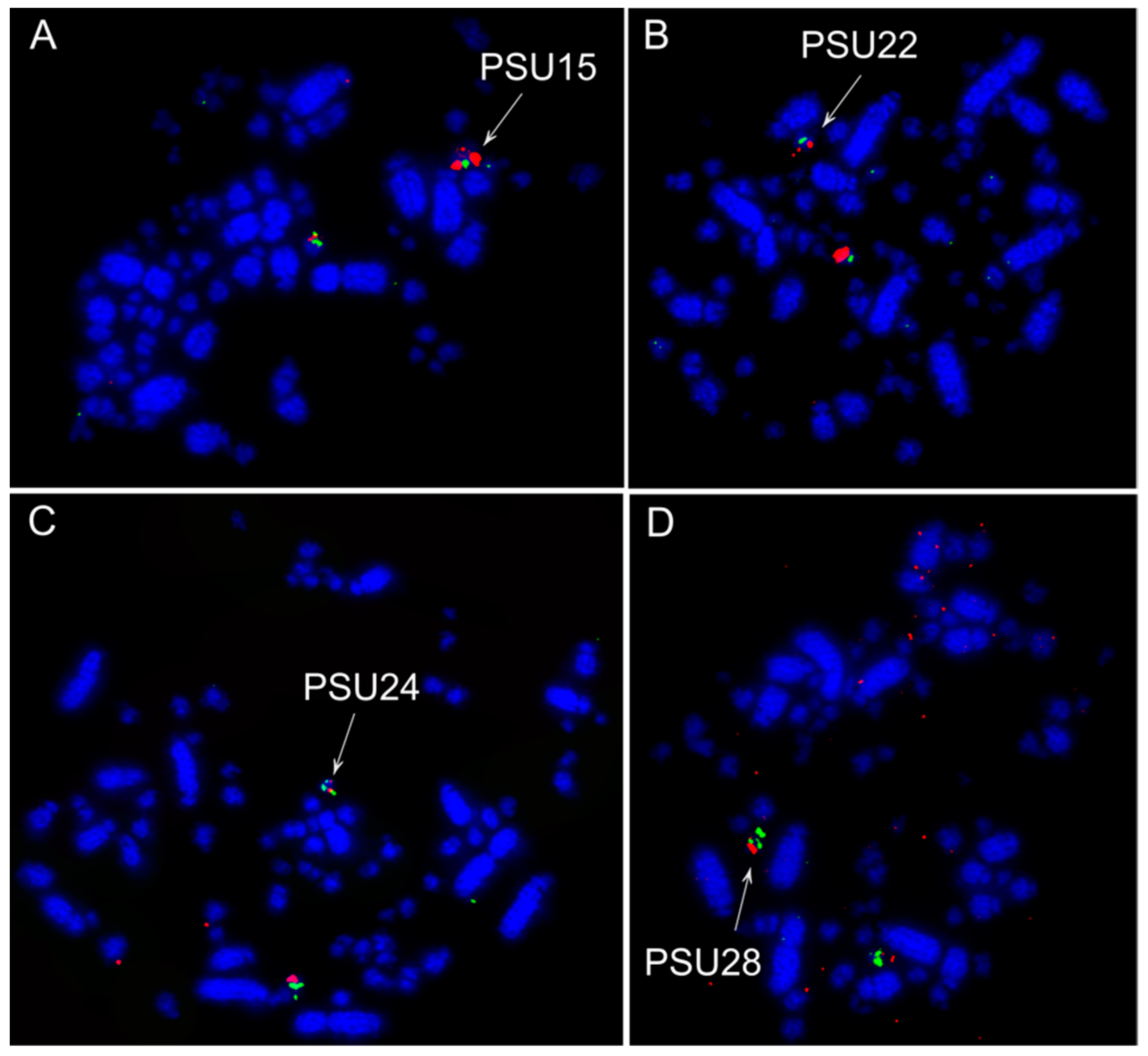
3.2. Fluorescence In Situ Hybridization (FISH) Experiments
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fitzpatrick, J.W. Family Tyrannidae (Tyrant-Flycatchers). In Handbook of the Birds of the World, Cotingas to Pipits and Wagtails; del Hoyo, J., Elliott, A., Christie, D.A., Eds.; Lynx Edicions: Barcelona, Spain, 2004; Volume 9, pp. 170–462. [Google Scholar]
- Tello, J.G.; Bates, J.M. Molecular phylogenetics of the tody-tyrant and flatbill assemblage of tyrant flycatchers (Tyrannidae). Auk 2007, 124, 134–154. [Google Scholar] [CrossRef]
- Rheindt, F.E.; Norman, J.A.; Christidis, L. Phylogenetic relationships of tyrant-flycatchers (Aves: Tyrannidae), with an emphasis on the elaeniine assemblage. Mol. Phylogenetics Evol. 2008, 46, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Tello, J.G.; Moyle, R.G.; Marchese, D.J.; Cracraft, J. Phylogeny and phylogenetic classification of the tyrant flycatchers, cotingas, manakins, and their allies (Aves: Tyrannides). Cladistics 2009, 25, 429–467. [Google Scholar] [CrossRef]
- Ohlson, J.I.; Irestedt, M.; Ericson, P.G.P.; Fjeldsa, J. Phylogeny and classification of the New World suboscines (Aves, Passeriformes). Zootaxa 2013, 3613, 1–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harvey, M.G.; Bravo, G.A.; Claramunt, S.; Cuervo, A.M.; Derryberry, G.E.; Battilana, J.; Seeholzer, G.F.; McKay, J.S.; O’Meara, B.C.; Faircloth, B.C.; et al. The evolution of a tropical biodiversity hotspot. Science 2020, 370, 1343–1348. [Google Scholar] [CrossRef]
- Degrandi, T.M.; Barcelos, S.A.; Costa, A.L.; Garnero, A.d.V.; Hass, I.; Gunski, R.J. Introducing the Bird Chromosome Database: An overview of cytogenetic studies on birds. Cytogenet. Genome Res. 2020, 160, 199–205. [Google Scholar] [CrossRef]
- Griffin, D.K.; Robertson, L.B.W.; Tempest, H.G.; Skinner, B.M. The evolution of the avian genome as revealed by comparative molecular cytogenetic. Cytogenet. Genome Res. 2007, 117, 64–77. [Google Scholar] [CrossRef]
- White, M.J.D. Animal Cytology and Evolution, 3rd ed.; Cambridge University Press: Cambridge, UK, 1973. [Google Scholar]
- Kretschmer, R.; Furo, I.O.; Cioffi, M.B.; Gunski, R.J.; Garnero, A.d.V.; O’Brien, P.C.M.; Ferguson-Smith, M.A.; de Freitas, T.R.O.; de Oliveira, E.H.C. Extensive chromosomal fissions and repetitive DNA accumulation shaped the atypical karyotypes of two Ramphastidae (Aves: Piciformes) species. Biol. J. Linn. Soc. 2020, 130, 839–849. [Google Scholar] [CrossRef]
- Furo, I.O.; Kretschmer, R.; dos Santos, M.S.; de Lima, C.A.C.; Gunski, R.J.; O’Brien, P.C.M.; Ferguson-Smith, M.A.; Cioffi, M.B.; de Oliveira, E.H.C. Chromosomal mapping of repetitive DNAs in Myiopsitta monachus and Amazona aestiva (Psittaciformes, Psittacidae) with emphasis on the sex chromosomes. Cytogenet. Genome Res. 2017, 151, 151–160. [Google Scholar] [CrossRef]
- Furo, I.O.; Kretschmer, R.; O’Brien, P.C.M.; Pereira, J.; Garnero, A.d.V.; Gunski, R.J.; O’Connor, R.E.; Griffin, D.K.; Gomes, A.J.B.; Ferguson-Smith, M.A.; et al. Chromosomal evolution in the phylogenetic context in Neotropical Psittacidae with emphasis on a species with high karyotypic reorganization (Myiopsitta monachus). Front. Genet. 2020, 11, 721. [Google Scholar] [CrossRef]
- Gunski, R.J.; Cabanne, G.S.; Ledesma, M.A.; Garnero, A.d.V. Analisis cariotipico de siete especies de Tiranidos (Tyrannidae). El Hornero 2000, 15, 103–109. [Google Scholar]
- Kretschmer, R.; de Oliveira, E.H.C.; dos Santos, M.S.; Furo, I.O.; O’Brien, P.C.M.; Ferguson-Smith, M.A.; Garnero, A.d.V.; Gunski, R.J. Chromosome mapping of the large elaenia (Elaenia spectabilis): Evidence for a cytogenetic signature for passeriform birds? Biol. J. Linn. Soc 2015, 115, 391–398. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, B.S.; Kretschmer, R.; Gunski, R.J.; Garnero, A.d.V.; O’Brien, P.C.M.; Ferguson-Smith, M.; de Oliveira, E.H.C. Chromosome Painting in Tyrant Flycatchers Confirms a Set of Inversions Shared by Oscines and Suboscines (Aves, Passeriformes). Cytogenet. Genome Res. 2017, 153, 205–212. [Google Scholar] [CrossRef] [PubMed]
- de Lucca, E.J.; Chamma, L. Estudo do complemento cromossômico de 11 espécies de aves das ordens Columbiformes, Passeriformes e Tinamiformes. Braz. J. Med. Biol. Res. 1977, 10, 97–105. [Google Scholar]
- Shields, G.F.; Barlow, J.C.; James, R.D. Karyotypes of five species of Empidonax flycatchers. Wilson Bull. 1987, 99, 169–174. [Google Scholar]
- Kretschmer, R.; Ferguson-Smith, M.A.; de Oliveira, E.H.C. Karyotype evolution in birds: From conventional staining to chromosome painting. Genes 2018, 9, 181. [Google Scholar] [CrossRef] [Green Version]
- de Oliveira, E.H.C.; Tagliarini, M.M.; Rissino, J.D.; Pieczarka, J.C.; Nagamachi, C.Y.; O’Brien, P.C.M.; Ferguson-Smith, M.A. Reciprocal chromosome painting between white hawk (Leucopternis albicollis) and chicken reveals extensive fusions and fissions during karyotype evolution of Accipitridae (Aves, Falconiformes). Chromosome Res. 2010, 18, 349–355. [Google Scholar] [CrossRef]
- Smith, J.; Paton, I.R.; Bruley, C.K.; Windsor, D.; Burt, D.W.; Ponce de Leon, F.A.; Burke, D. Integration of the genetic and physical maps of the chicken macrochromosomes. Anim. Genet. 2000, 31, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Burt, D.W. Origin and evolution of avian microchromosomes. Cytogenet. Genome Res. 2002, 96, 97–112. [Google Scholar] [CrossRef]
- Warren, W.C.; Hillier, L.W.; Tomlinson, C.; Minx, P.; Kremitzki, M.; Graves, T.; Markovic, C.; Bouk, N.; Pruitt, K.D.; Thibaud-Nissen, F.; et al. A new chicken genome assembly provides insight into avian genome structure. G3 Genes Genom. Genet. 2017, 7, 109–117. [Google Scholar] [CrossRef] [Green Version]
- Joseph, S.; O’Connor, R.E.; Al Mutery, A.F.; Watson, M.; Larkin, D.M.; Griffin, D.K. Chromosome level genome assembly and comparative genomics between three falcon species reveals an unusual pattern of genome organisation. Diversity 2018, 10, 113. [Google Scholar] [CrossRef] [Green Version]
- O’Connor, R.E.; Kiazim, L.; Skinner, B.; Fonseka, G.; Joseph, S.; Jennings, R.; Larkin, D.M.; Griffin, D.K. Patterns of microchromosome organization remain highly conserved throughout avian evolution. Chromosoma 2019, 128, 21–29. [Google Scholar] [CrossRef] [Green Version]
- Kretschmer, R.; de Souza, M.S.; Furo, I.O.; Gunski, R.J.; Garnero, A.d.V.; de Freitas, T.R.O.; de Oliveira, E.H.C.; O’Connor, R.E.; Griffin, D.K. Interspecies Chromosome Mapping in Caprimulgiformes, Piciformes, Suliformes, and Trogoniformes (Aves): Cytogenomic Insight into Microchromosome Organization and Karyotype Evolution in Birds. Cells 2021, 10, 826. [Google Scholar] [CrossRef]
- Kretschmer, R.; Gunski, R.J.; Garnero, A.d.V.; de Freitas, T.R.O.; Toma, G.A.; Cioffi, M.B.; de Oliveira, E.H.C.; O’Connor, R.E.; Griffin, D.K. Chromosomal analysis in Crotophaga ani (Aves, Cuculiformes) reveals extensive genomic reorganization and an unusual Z-autosome Robertsonian translocation. Cells 2021, 10, 4. [Google Scholar] [CrossRef]
- Ribas, T.F.A.; Pieczarka, J.C.; Griffin, D.K.; Kiazim, L.G.; Nagamachi, C.Y.; O.´Brien, P.C.M.; Ferguson-Smith, M.A.; Yang, F.; Aleixo, A.; O’Connor, R.E. Analysis of multiple chromosomal rearrangements in the genome of Willisornis vidua using BAC-FISH and chromosome painting on a supposed conserved karyotype. BMC Evol. Biol. 2021, 21, 34. [Google Scholar] [CrossRef]
- Kretschmer, R.; Rodrigues, B.S.; Barcellos, A.S.; Costa, A.L.; Cioffi, M.B.; Garnero, A.d.V.; Gunski, R.J.; de Oliveira, E.H.C.; Griffin, D.K. Karyotype Evolution and Genomic Organization of Repetitive DNAs in the Saffron Finch, Sicalis flaveola (Passeriformes, Aves). Animals 2021, 11, 1456. [Google Scholar] [CrossRef] [PubMed]
- Guerra, M.S. Reviewing the chromosome nomenclature of Levan et al. Rev. Bras. Genética 1986, 4, 741–743. [Google Scholar]
- Romanov, M.N.; Farré, M.; Lithgow, P.E.; Fowler, K.E.; Skinner, B.M.; O’Connor, R.; Fonseka, G.; Backström, N.; Matsuda, Y.; Nishida, C.; et al. Reconstruction of gross avian genome structure, organization and evolution suggests that the chicken lineage most closely resembles the dinosaur avian ancestor. BMC Genom. 2014, 15, 1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lanyon, W.E. A phylogeny of the flatbill and tody-tyrant assemblage of tyrant flycatchers. Am. Mus. Novit. 1988, 2923, 1–41. [Google Scholar]
- Ericson, P.G.; Zuccon, D.; Ohlson, J.I.; Johansson, U.S.; Alvarenga, H.; Prum, R.O. Higher-level phylogeny and morphological evolution of tyrant flycatchers, cotingas, manakins, and their allies (Aves: Tyrannida). Mol. Phylogenetics Evol. 2006, 40, 471–483. [Google Scholar] [CrossRef]
- Ohlson, J.; Fjeldsa, J.; Ericson, P.G. Tyrant flycatchers coming out in the open: Phylogeny and ecological radiation of Tyrannidae (Aves, Passeriformes). Zool. Scr. 2008, 37, 315–335. [Google Scholar] [CrossRef]
- Lopes, L.E.; Chaves, A.V.; de Aquino, M.M.; Silveira, L.F.; dos Santos, F.R. The striking polyphyly of Suiriri: Convergent evolution and social mimicry in two cryptic Neotropical birds. J. Zool. Syst. Evol. Res. 2018, 56, 270–279. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, C.; Krishnaswamy, L.; Peterson, T. Transposable elements as catalysts for chromosome rearrangements. Methods Mol. Biol. 2011, 701, 315–326. [Google Scholar] [CrossRef]
- Farré, M.; Narayan, J.; Slavov, G.T.; Damas, J.; Auvil, L.; Li, C.; Jarvis, E.D.; Burt, D.W.; Griffin, D.K.; Larkin, D.M. Novel in-sights into chromosome evolution in birds, archosaurs, and reptiles. Genome Biol. Evol. 2016, 8, 2442–2451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansson, U.S.; Fjeldså, J.; Bowie, R.C.K. Phylogenetic relationships within Passerida (Aves: Passeriformes): A review and a new molecular phylogeny based on three nuclear intron markers. Mol. Phylogenetics Evol. 2008, 48, 858–876. [Google Scholar] [CrossRef]
- Gill, F.; Donsker, D.; Rasmussen, P. IOC World Bird List; Version 10.1; IOC: London, UK, 2020. [Google Scholar]
- Volker, M.; Backstrom, N.; Skinner, B.M.; Langley, E.J.; Bunzey, S.K.; Ellegren, H.; Griffin, D.K. Copy number variation, chromosome rearrangement, and their association with recombination during avian evolution. Genome Res. 2010, 20, 503–511. [Google Scholar] [CrossRef] [Green Version]
- Warren, W.C.; Clayton, D.F.; Ellegren, H.; Arnold, A.P.; Hillier, L.W.; Künstner, A.; Searle, S.; White, S.; Vilella, A.J.; Fairley, S.; et al. The genome of a songbird. Nature 2010, 464, 757–762. [Google Scholar] [CrossRef]

| Species | 2n | Family | Reference |
|---|---|---|---|
| Platyrinchus mystaceus | 2n = 60 | Platyrinchidae | [13] |
| Elaenia parvirostris | 2n = 78 | Tyrannidae | [13] |
| Elaenia spectabilis | 2n = 80 | Tyrannidae | [14] |
| Serpophaga subcristata | 2n = 82 | Tyrannidae | [15] |
| Pitangus sulphuratus | 2n = 80 | Tyrannidae | [15] |
| Tyrannus melancholicus | 2n = 78 | Tyrannidae | [13] |
| Tyrannus savana | 2n = 78 | Tyrannidae | [13] |
| Myiarchus ferox | 2n = 76 | Tyrannidae | [13] |
| Knipolegus cyanirostris | 2n = 78 | Tyrannidae | [16] |
| Satrapa icterophrys | 2n = 82 | Tyrannidae | [15] |
| Cnemotriccus fuscatus | 2n = 84 | Tyrannidae | [13] |
| Empidonax alnorum | 2n = 82 | Tyrannidae | [17] |
| Empidonax flaviventris | 2n = 82 | Tyrannidae | [17] |
| Empidonax hammondii | 2n = 82 | Tyrannidae | [17] |
| Empidonax minimus | 2n = 82 | Tyrannidae | [17] |
| Empidonax traillii | 2n = 82 | Tyrannidae | [17] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kretschmer, R.; Franz, I.; de Souza, M.S.; Garnero, A.D.V.; Gunski, R.J.; de Oliveira, E.H.C.; O’Connor, R.E.; Griffin, D.K.; de Freitas, T.R.O. Cytogenetic Evidence Clarifies the Phylogeny of the Family Rhynchocyclidae (Aves: Passeriformes). Cells 2021, 10, 2650. https://doi.org/10.3390/cells10102650
Kretschmer R, Franz I, de Souza MS, Garnero ADV, Gunski RJ, de Oliveira EHC, O’Connor RE, Griffin DK, de Freitas TRO. Cytogenetic Evidence Clarifies the Phylogeny of the Family Rhynchocyclidae (Aves: Passeriformes). Cells. 2021; 10(10):2650. https://doi.org/10.3390/cells10102650
Chicago/Turabian StyleKretschmer, Rafael, Ismael Franz, Marcelo Santos de Souza, Analía Del Valle Garnero, Ricardo José Gunski, Edivaldo Herculano Corrêa de Oliveira, Rebecca E. O’Connor, Darren K. Griffin, and Thales Renato Ochotorena de Freitas. 2021. "Cytogenetic Evidence Clarifies the Phylogeny of the Family Rhynchocyclidae (Aves: Passeriformes)" Cells 10, no. 10: 2650. https://doi.org/10.3390/cells10102650
APA StyleKretschmer, R., Franz, I., de Souza, M. S., Garnero, A. D. V., Gunski, R. J., de Oliveira, E. H. C., O’Connor, R. E., Griffin, D. K., & de Freitas, T. R. O. (2021). Cytogenetic Evidence Clarifies the Phylogeny of the Family Rhynchocyclidae (Aves: Passeriformes). Cells, 10(10), 2650. https://doi.org/10.3390/cells10102650











