Hiding in Plain Sight: Modern Thiamine Deficiency
Abstract
:1. Introduction
2. Thiamine Deficiency Definitions and Testing
2.1. Symptoms
2.2. Testing
3. The RDA, Food Fortification and Thiamine Sufficiency
4. Thiamine Deficiency in the General Population
4.1. Obesity
4.2. Diabetes
4.3. Pregnancy
4.4. Psychiatry
4.5. Elderly
4.6. Neurocognitive and Neuromotor Diseases
4.7. In Hospitalized Patients
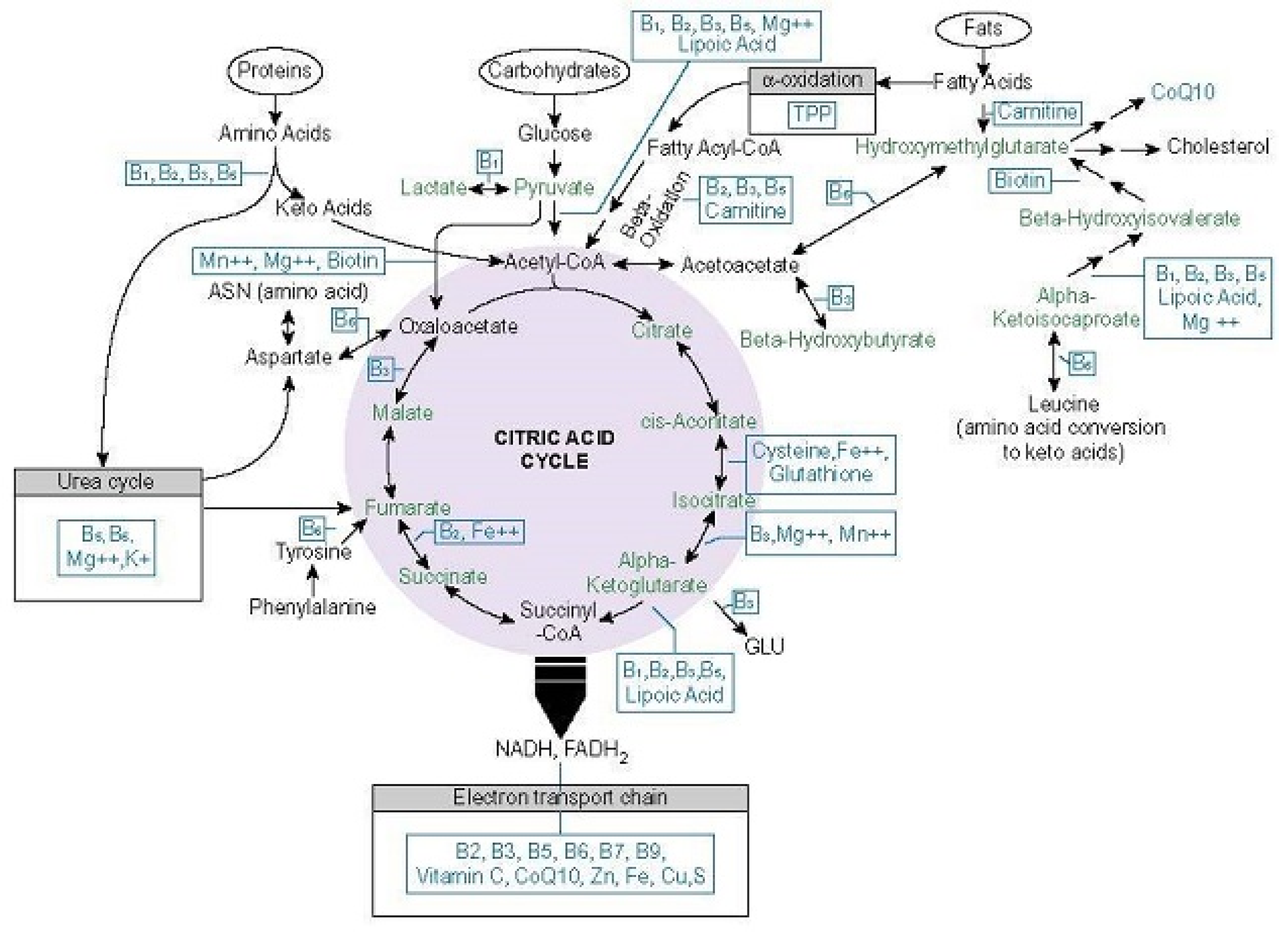
5. What Makes Thiamine So Important
5.1. Thiamine Dependent Enzymes
5.1.1. Transketolase
5.1.2. Pyruvate Dehydrogenase Complex
5.1.3. 2-Hydroxyacyl-CoA Lyase
5.1.4. Branched Chain Keto-Acid Dehydrogenase
5.1.5. Alpha-Ketoglutarate Dehydrogenase Complex
5.1.6. Thiamine-Influenced Enzymes
6. Thiamine Consumption, Uptake, Activation, and Excretion
6.1. Consumption
6.2. Uptake
- SLC19A1: folate transporter, but also, transports thiamine mono- and di- phospho derivatives [95].
- SLC22A1 (OCT1): organic cation transporter 1, primary hepatic thiamine transporter [97].
- SLC25A19 (MTPP-1): mitochondrial thiamine pyrophosphate carrier [98].
- SLC44A4 (hTPPT/TPPT-1): absorption of microbiota-generated thiamine pyrophosphate in the large intestine [101].
6.3. Activation
6.4. Storage and Elimination
7. Factors Affecting Thiamine Availability and Demand
7.1. High Carbohydrate Diets
7.2. Food Chemicals
7.3. Alcohol, Tobacco, Coffee and Tea Consumption
7.4. Medications and Environmental Exposures
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Devisch, I.; Murray, S.J. ‘We hold these truths to be self-evident’: Deconstructing ‘evidence-based’medical practice. J. Eval. Clin. Pract. 2009, 15, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Kohnke, S.; Meek, C.L. Don’t seek, don’t find: The diagnostic challenge of Wernicke’s encephalopathy. Ann. Clin. Biochem. 2021, 58, 38–46. [Google Scholar] [CrossRef]
- Whitfield, K.C.; Bourassa, M.W.; Adamolekun, B.; Bergeron, G.; Bettendorff, L.; Brown, K.H.; Cox, L.; Fattal-Valevski, A.; Fischer, P.R.; Frank, E.L.; et al. Thiamine deficiency disorders: Diagnosis, prevalence, and a roadmap for global control programs. Ann. N. Y. Acad. Sci. 2018, 1430, 3. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.D.; Mason, H.L.; Wilder, R.M.; Smith, B.F. Observations on induced thiamine (vitamin B1) deficiency in man. Arch. Intern. Med. 1940, 66, 785–799. [Google Scholar] [CrossRef]
- Williams, R.D.; Mason, H.L.; Smith, B.F.; Wilder, R.M. Induced thiamine (vitamin B1) deficiency and the thiamine requirement of man: Further observations. Arch. Intern. Med. 1942, 69, 721–738. [Google Scholar] [CrossRef]
- Prakash, S. Gastrointestinal beriberi: A forme fruste of Wernicke’s encephalopathy? Case Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Donnino, M. Gastrointestinal beriberi: A previously unrecognized syndrome. Ann. Intern. Med. 2004, 141, 898–899. [Google Scholar] [CrossRef]
- Harper, C.G.; Giles, M.; Finlay-Jones, R. Clinical signs in the Wernicke-Korsakoff complex: A retrospective analysis of 131 cases diagnosed at necropsy. J. Neurol. Neurosurg. Psychiatry 1986, 49, 341–345. [Google Scholar] [CrossRef] [Green Version]
- Rifai, N. Tietz Fundamentals of Clinical Chemistry and Molecular Diagnostics 8 E; South Asia Edition; e-Book; Elsevier: Chennai, India, 2019. [Google Scholar]
- Lynch, P.L.M.; Young, I.S. Determination of thiamine by high-performance liquid chromatography. J. Chromatography A 2000, 881, 267–284. [Google Scholar] [CrossRef]
- Quest Diagnostics Vitamin B1 (Thiamine), Blood, LC/MS/MS. Available online: https://testdirectory.questdiagnostics.com/test/test-detail/5042/vitamin-b1-thiamine-blood-lc-ms-ms?cc=MASTER (accessed on 16 July 2021).
- LabCorp Vitamin B1, Whole Blood. Available online: https://www.labcorp.com/tests/121186/vitamin-b-sub-1-sub-whole-blood (accessed on 16 July 2021).
- Burch, H.B.; Bessey, O.A.; Love, R.H.; Lowry, O.H. The determination of thiamine and thiamine phosphates in small quantities of blood and blood cells. J. Biol. Chem. 1952, 198, 477–490. [Google Scholar] [CrossRef]
- Lu, J.; Frank, E.L. Rapid HPLC measurement of thiamine and its phosphate esters in whole blood. Clin. Chem. 2008, 54, 901–906. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.S.; Parkington, D.A.; Cox, L.J.; Koulman, A. Erythrocyte transketolase activity coefficient (ETKAC) assay protocol for the assessment of thiamine status. Ann. N. Y. Acad. Sci. 2020, 1498, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Medscape. What is the Role Erythrocyte Transketolase Levels in the Workup of Wernicke Encephalopathy (WE)? Available online: https://www.medscape.com/answers/794583-156135/what-is-the-role-erythrocyte-transketolase-levels-in-the-workup-of-wernicke-encephalopathy-we (accessed on 15 September 2021).
- Talwar, D.; Davidson, H.; Cooney, J.; St. JO’Reilly, D. Vitamin B1 status assessed by direct measurement of thiamin pyrophosphate in erythrocytes or whole blood by HPLC: Comparison with erythrocyte transketolase activation assay. Clin. Chem. 2000, 46, 704–710. [Google Scholar] [CrossRef] [Green Version]
- Quest Diagnostics. Vitamin B1 (Thiamine), Plasma/Serum, LC/MS/MS. Available online: https://testdirectory.questdiagnostics.com/test/test-detail/90353/vitamin-b1-thiamine-plasma-serum-lc-ms-ms?cc=MASTER (accessed on 16 July 2021).
- Rieck, J.; Halkin, H.; Almog, S.; Seligman, H.; Lubetsky, A.; Olchovsky, D.; Ezra, D. Urinary loss of thiamine is increased by low doses of furosemide in healthy volunteers. J. Lab. Clin. Med. 1999, 134, 238–243. [Google Scholar] [CrossRef]
- Ziporin, Z.Z.; Nunes, W.T.; Powell, R.C.; Waring, P.P.; Sauberlich, H.E. Excretion of thiamine and its metabolites in the urine of young adult males receiving restricted intakes of the vitamin. J. Nutr. 1965, 85, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Tasevska, N.; Runswick, S.A.; McTaggart, A.; Bingham, S.A. Twenty-four-hour urinary thiamine as a biomarker for the assessment of thiamine intake. Eur. J. Clin. Nutr. 2008, 62, 1139–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998; Chapter 4: Thiamin; Available online: https://www.ncbi.nlm.nih.gov/books/NBK114331/ (accessed on 16 July 2021).
- Stiebeling, H.K. Family Food Consumption and Dietary Levels: Five Regions (No. 405); US Government Printing Office: Washington, DC, USA, 1941.
- Committee on Use of Dietary Reference Intakes in Nutrition Labeling. Overview of Food Fortification in the United States and Canada. Dietary Reference Intakes: Guiding Principles for Nutrition Labeling and Fortification; Institute of Medicine: Washington, DC, USA, 2003; pp. 45–56. [Google Scholar]
- Newman, J.C.; Malek, A.M.; Hunt, K.J.; Marriott, B.P. Nutrients in the US diet: Naturally occurring or enriched/fortified food and beverage sources, plus dietary supplements: NHANES 2009–2012. J. Nutr. 2019, 149, 1404–1412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- NIH, Department of Dietary Services. Thiamin Fact Sheet for Health Professionals. 2021. Available online: https://ods.od.nih.gov/factsheets/Thiamin-HealthProfessional/ (accessed on 24 May 2021).
- Carrodeguas, L.; Kaidar-Person, O.; Szomstein, S.; Antozzi, P.; Rosenthal, R. Preoperative thiamine deficiency in obese population undergoing laparoscopic bariatric surgery. Surg. Obes. Relat. Dis. 2005, 1, 517–522. [Google Scholar] [CrossRef]
- Flancbaum, L.; Belsley, S.; Drake, V.; Colarusso, T.; Tayler, E. Preoperative nutritional status of patients undergoing Roux-en-Y gastric bypass for morbid obesity. J. Gastrointest. Surg. 2006, 10, 1033–1037. [Google Scholar] [CrossRef]
- Kerns, J.C.; Arundel, C.; Chawla, L.S. Thiamin deficiency in people with obesity. Adv. Nutr. 2015, 6, 147–153. [Google Scholar] [CrossRef] [Green Version]
- Oudman, E.; Wijnia, J.W.; van Dam, M.; Biter, L.U.; Postma, A. Preventing Wernicke encephalopathy after bariatric surgery. Obes. Surg. 2018, 28, 2060–2068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aasheim, E.T. Wernicke Encephalopathy After Bariatric Surgery. Ann. Surg. 2008, 248, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Adult Obesity Facts. Available online: https://www.cdc.gov/obesity/data/adult.html (accessed on 24 May 2021).
- Prevalence of Obesity. Available online: https://www.worldobesity.org/about/about-obesity/prevalence-of-obesity (accessed on 24 May 2021).
- Thornalley, P.J.; Babaei-Jadidi, R.; Al Ali, H.; Rabbani, N.; Antonysunil, A.; Larkin, J.; Ahmed, A.; Rayman, G.; Bodmer, C.W. High prevalence of low plasma thiamine concentration in diabetes linked to a marker of vascular disease. Diabetologia 2007, 50, 2164–2170. [Google Scholar] [CrossRef] [Green Version]
- Nix, W.A.; Zirwes, R.; Bangert, V.; Kaiser, R.P.; Schilling, M.; Hostalek, U.; Obeid, R. Vitamin B status in patients with type 2 diabetes mellitus with and without incipient nephropathy. Diabetes Res. Clin. Pract. 2015, 107, 57–165. [Google Scholar] [CrossRef] [Green Version]
- Larkin, J.R.; Zhang, F.; Godfrey, L.; Molostvov, G.; Zehnder, D.; Rabbani, N.; Thornalley, P.J. Glucose-induced down regulation of thiamine transporters in the kidney proximal tubular epithelium produces thiamine insufficiency in diabetes. PLoS ONE 2012, 7, e53175. [Google Scholar] [CrossRef] [PubMed]
- Baker, H.; DeAngelis, B.; Holland, B.; Gittens-Williams, L.; Barrett, T., Jr. Vitamin profile of 563 gravidas during trimesters of pregnancy. J. Am. Coll. Nutr. 2002, 21, 33–37. [Google Scholar] [CrossRef]
- Sánchez, D.J.; Murphy, M.M.; Bosch-Sabater, J.; Fernández-Ballart, J. Enzymic evaluation of thiamin, riboflavin and pyridoxine status of parturient mothers and their newborn infants in a Mediterranean area of Spain. Eur. J. Clin. Nutr. 1999, 53, 27–38. [Google Scholar] [CrossRef] [Green Version]
- Oudman, E.; Wijnia, J.W.; Oey, M.; van Dam, M.; Painter, R.C.; Postma, A. Wernicke’s encephalopathy in hyperemesis gravidarum: A systematic review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 236, 84–93. [Google Scholar] [CrossRef]
- Dhir, S.; Tarasenko, M.; Napoli, E.; Giulivi, C. Neurological, psychiatric, and biochemical aspects of thiamine deficiency in children and adults. Front. Psychiatry 2019, 10, 207. [Google Scholar] [CrossRef] [Green Version]
- Carney, M.W.; Ravindran, A.; Rinsler, M.G.; Williams, D.G. Thiamine, riboflavin and pyridoxine deficiency in psychiatric in-patients. Br. J. Psychiatry 1982, 141, 271–272. [Google Scholar] [CrossRef]
- Zhang, G.; Ding, H.; Chen, H.; Ye, X.; Li, H.; Lin, X.; Ke, Z. Thiamine nutritional status and depressive symptoms are inversely associated among older Chinese adults. J. Nutr. 2013, 143, 53–58. [Google Scholar] [CrossRef] [Green Version]
- Ghaleiha, A.; Davari, H.; Jahangard, L.; Haghighi, M.; Ahmadpanah, M.; Seifrabie, M.A.; Bajoghli, H.; Holsboer-Trachsler, E.; Brand, S. Adjuvant thiamine improved standard treatment in patients with major depressive disorder: Results from a randomized, double-blind, and placebo-controlled clinical trial. Eur. Arch. Psychiatry Clin. Neurosci. 2016, 266, 695–702. [Google Scholar] [CrossRef]
- Quoc Lu’o’ng, K.V.; Nguyen, L.T.H. The impact of thiamine treatment on generalized anxiety disorder. Int. J. Clin. Med. 2011, 2, 439. [Google Scholar]
- McCormick, L.M.; Buchanan, J.R.; Onwuameze, O.E.; Pierson, R.K.; Paradiso, S. Beyond alcoholism: Wernicke-Korsakoff syndrome in patients with psychiatric disorders. Cogn. Behav. Neurol. Off. J. Soc. Behav. Cogn. Neurol. 2011, 24, 209. [Google Scholar] [CrossRef]
- Nichols, H.K.; Basu, T.K. Thiamin status of the elderly: Dietary intake and thiamin pyrophosphate response. J. Am. Coll. Nutr. 1994, 13, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Pepersack, T.; Garbusinski, J.; Robberecht, J.; Beyer, I.; Willems, D.; Fuss, M. Clinical relevance of thiamine status amongst hospitalized elderly patients. Gerontology 1999, 45, 96–101. [Google Scholar] [CrossRef]
- Wilkinson, T.J.; Hanger, H.C.; George, P.M.; Sainsbury, R. Is thiamine deficiency in elderly people related to age or co-morbidity? Age Ageing 2000, 29, 111–116. [Google Scholar] [CrossRef] [Green Version]
- Sang, S.; Pan, X.; Chen, Z.; Zeng, F.; Pan, S.; Liu, H.; Jin, L.; Fei, G.; Wang, C.; Ren, S.; et al. Thiamine diphosphate reduction strongly correlates with brain glucose hypometabolism in Alzheimer’s disease, whereas amyloid deposition does not. Alzheimer’s Res. Ther. 2018, 10, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Håglin, L.; Domellöf, M.; Bäckman, L.; Forsgren, L. Low plasma thiamine and phosphate in male patients with Parkinson’s disease is associated with mild cognitive impairment. Clin. Nutr. ESPEN 2020, 37, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Montojo, M.T.; Aganzo, M.; González, N. Huntington’s disease and diabetes: Chronological sequence of its association. J. Huntingt. Dis. 2017, 6, 179–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibson, G.E.; Hirsch, J.A.; Fonzetti, P.; Jordon, B.D.; Cirio, R.T.; Elder, J. Vitamin B1 (thiamine) and dementia. Ann. N. Y. Acad. Sci. 2016, 1367, 21. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Ke, Z.; Luo, J. Thiamine deficiency and neurodegeneration: The interplay among oxidative stress, endoplasmic reticulum stress, and autophagy. Mol. Neurobiol. 2017, 54, 5440–5448. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, C.P.; Obeid, O.A.; Powell-Tuck, J. The thiamin, riboflavin and pyridoxine status of patients on emergency admission to hospital. Clin. Nutr. 1999, 18, 87–91. [Google Scholar] [CrossRef]
- Hanninen, S.A.; Darling, P.B.; Sole, M.J.; Barr, A.; Keith, M.E. The prevalence of thiamin deficiency in hospitalized patients with congestive heart failure. J. Am. Coll. Cardiol. 2006, 47, 354–361. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, M.; Azizi-Namini, P.; Yan, A.T.; Keith, M. Thiamin deficiency and heart failure: The current knowledge and gaps in literature. Heart Fail. Rev. 2015, 20, 1–11. [Google Scholar] [CrossRef]
- Mates, E.; Alluri, D.; Artis, T.; Riddle, M.S. A Retrospective Case Series of Thiamine Deficiency in Non-Alcoholic Hospitalized Veterans: An Important Cause of Delirium and Falling? J. Clin. Med. 2021, 10, 1449. [Google Scholar] [CrossRef]
- Costa, N.A.; Gut, A.L.; de Souza Dorna, M.; Pimentel, J.A.C.; Cozzolino, S.M.F.; Azevedo, P.S.; Fernandes, A.A.H.; Zornoff, L.A.M.; de Paiva, S.A.R.; Minicucci, M.F. Serum thiamine concentration and oxidative stress as predictors of mortality in patients with septic shock. J. Crit. Care 2014, 29, 249–252. [Google Scholar] [CrossRef] [Green Version]
- Sweet, R.L.; Zastre, J.A. HIF1-α-mediated gene expression induced by vitamin B. Int. J. Vitam. Nutr. Res. 2013, 83, 188–197. [Google Scholar] [CrossRef]
- Lee, J.W.; Ko, J.; Ju, C.; Eltzschig, H.K. Hypoxia signaling in human diseases and therapeutic targets. Exp. Mol. Med. 2019, 51, 1–13. [Google Scholar] [CrossRef]
- Rich, P.R. The molecular machinery of Keilin’s respiratory chain. Biochem. Soc. Trans. 2003, 31, 1095–1105. [Google Scholar] [CrossRef]
- McGettrick, A.F.; O’Neill, L.A. The role of HIF in immunity and inflammation. Cell Metab. 2020, 32, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D.; Marrs, C. Thiamine Deficiency Disease, Dysautonomia, and High Calorie Malnutrition; Academic Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Xiao, W.; Wang, R.S.; Handy, D.E.; Loscalzo, J. NAD (H) and NADP (H) redox couples and cellular energy metabolism. Antioxid. Redox Signal. 2018, 28, 251–272. [Google Scholar] [CrossRef]
- Patra, K.C.; Hay, N. The pentose phosphate pathway and cancer. Trends Biochem. Sci. 2014, 39, 347–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, G.L.J.; Laight, D.; Cummings, M.H. TD in diabetes mellitus and the impact of thiamine replacement on glucose metabolism and vascular disease. Int. J. Clin. Pract. 2011, 65, 684–690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alam, S.S.; Riaz, S.; Akhtar, M.W. Effect of high dose thiamine therapy on risk factors in type 2 diabetics. J. Diabetes Metab. 2012, 3, 1000233. [Google Scholar]
- Gralak, M.A.; Dębski, B.; Drywień, M. TD affects glucose transport and β-oxidation in rats. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1629–1635. [Google Scholar] [CrossRef] [PubMed]
- Strumiło, S. Short-term regulation of the mammalian pyruvate dehydrogenase complex. Acta Biochim. Pol. 2005, 52, 759–764. [Google Scholar] [CrossRef]
- Seheult, J.; Fitzpatrick, G.; Boran, G. Lactic acidosis: An update. Clin. Chem. Lab. Med. 2017, 55, 322–333. [Google Scholar] [CrossRef]
- Eys, J.V. Alpha-glycerophosphate and lactic dehydrogenase activities in tissues of thiamine-deficient rats. J. Nutr. 1961, 73, 403–408. [Google Scholar] [CrossRef]
- Moore, R.O.; Yontz, F.D. Effect of TD in rats on adipose tissue lactate dehydrogenase isozyme distribution. J. Nutr. 1969, 98, 325–329. [Google Scholar] [CrossRef] [Green Version]
- Zera, K.; Zastre, J. Stabilization of the hypoxia-inducible transcription factor-1 alpha (HIF-1α) in TD is mediated by pyruvate accumulation. Toxicol. Appl. Pharmacol. 2018, 355, 180–188. [Google Scholar] [CrossRef]
- Casteels, M.; Sniekers, M.; Fraccascia, P.; Mannaerts, G.P.; Van Veldhoven, P.P. The role of 2-hydroxyacyl-CoA lyase, a thiamin pyrophosphate-dependent enzyme, in the peroxisomal metabolism of 3-methyl-branched fatty acids and 2-hydroxy straight-chain fatty acids. Biochem. Soc. Trans. 2007, 35, 876–880. [Google Scholar] [CrossRef] [Green Version]
- Adult Refsum’s Disease, Phytanic Acid Calculator. Available online: http://www.refsumdisease.org/clinicians/phytanicacidcalculator.shtml (accessed on 16 July 2021).
- Vesper, H.; Schmelz, E.M.; Nikolova-Karakashian, M.N.; Dillehay, D.L.; Lynch, D.V.; Merrill, A.H., Jr. Sphingolipids in food and the emerging importance of sphingolipids to nutrition. J. Nutr. 1999, 129, 1239–1250. [Google Scholar] [CrossRef] [Green Version]
- Refsum’s Disease Information Page. Available online: https://www.ninds.nih.gov/Disorders/All-Disorders/Refsum-Disease-Information-Page (accessed on 12 August 2021).
- Mencarelli, C.; Martinez–Martinez, P. Ceramide function in the brain: When a slight tilt is enough. Cell. Mol. Life Sci. 2013, 70, 181–203. [Google Scholar] [CrossRef] [Green Version]
- Koves, T.R.; Ussher, J.R.; Noland, R.C.; Slentz, D.; Mosedale, M.; Ilkayeva, O.; Bain, J.; Stevens, R.; Dyck, J.R.; Newgard, C.B.; et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008, 7, 45–56. [Google Scholar] [CrossRef] [Green Version]
- Gar, C.; Rottenkolber, M.; Prehn, C.; Adamski, J.; Seissler, J.; Lechner, A. Serum and plasma amino acids as markers of prediabetes, insulin resistance, and incident diabetes. Crit. Rev. Clin. Lab. Sci. 2018, 55, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Rutkowsky, J.M.; Knotts, T.A.; Ono-Moore, K.D.; McCoin, C.S.; Huang, S.; Schneider, D.; Singh, S.; Adams, S.H.; Hwang, D.H. Acylcarnitines activate proinflammatory signaling pathways. Am. J. Physiol.-Endocrinol. Metab. 2014, 306, E1378–E1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Entry to the citric acid cycle and metabolism through it are controlled. In Biochemistry, 5th ed.; Freeman W.H.: New York, NY, USA, 2002. [Google Scholar]
- Tretter, L.; Adam-Vizi, V. Alpha-ketoglutarate dehydrogenase: A target and generator of oxidative stress. Philos. Trans. R. Soc. B Biol. Sci. 2005, 360, 2335–2345. [Google Scholar] [CrossRef] [Green Version]
- Panov, A.; Scarpa, A. Independent modulation of the activity of α-ketoglutarate dehydrogenase complex by Ca2+ and Mg2+. Biochemistry 1996, 35, 427–432. [Google Scholar] [CrossRef] [PubMed]
- McLain, A.L.; Szweda, P.A.; Szweda, L.I. α-Ketoglutarate dehydrogenase: A mitochondrial redox sensor. Free Radic. Res. 2011, 45, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauhan, A.; Srivastva, N.; Bubber, P. TDinduced dietary disparity promotes oxidative stress and neurodegeneration. Indian J. Clin. Biochem. 2018, 33, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Mallat, J.; Lemyze, M.; Thevenin, D. Do not forget to give thiamine to your septic shock patient! J. Thorac. Dis. 2016, 8, 1062. [Google Scholar] [CrossRef] [PubMed]
- Bubber, P.; Ke, Z.J.; Gibson, G.E. Tricarboxylic acid cycle enzymes following thiamine deficiency. Neurochem. Int. 2004, 45, 1021–1028. [Google Scholar] [CrossRef]
- Organic Market Trends. Available online: https://www.ers.usda.gov/topics/natural-resources-environment/organic-agriculture/organic-market-summary-and-trends/ (accessed on 22 August 2021).
- Poti, J.M.; Mendez, M.A.; Ng, S.W.; Popkin, B.M. Is the degree of food processing and convenience linked with the nutritional quality of foods purchased by US households? Am. J. Clin. Nutr. 2015, 101, 1251–1262. [Google Scholar] [CrossRef] [Green Version]
- Shepherd, S.J.; Gibson, P.R. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. 2013, 26, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Vudhivai, N.; Ali, A.; Pongpaew, P.; Changbumrung, S.; Vorasanta, S.; Kwanbujan, K.; Charoenlarp, P.; Migasena, P.; Schelp, F.P. Vitamin B1, B2 and B6 status of vegetarians. J. Med Assoc. Thail. = Chotmaihet Thangphaet 1991, 74, 465–470. [Google Scholar]
- Zhao, R.; Goldman, I.D. Folate and thiamine transporters mediated by facilitative carriers (SLC19A1-3 and SLC46A1) and folate receptors. Mol. Asp. Med. 2013, 34, 373–385. [Google Scholar] [CrossRef] [Green Version]
- Enogieru, O.J.; Koleske, M.L.; Vora, B.; Ngo, H.; Yee, S.W.; Chatad, D.; Sirota, M.; Giacomini, K.M. The Effects of Genetic Mutations and Drugs on the Activity of the Thiamine Transporter, SLC19A2. AAPS J. 2021, 23, 35. [Google Scholar] [CrossRef]
- Liang, X.; Yee, S.W.; Chien, H.C.; Chen, E.C.; Luo, Q.; Zou, L.; Piao, M.; Mifune, A.; Chen, L.; Calvert, M.E.; et al. Organic cation transporter 1 (OCT1) modulates multiple cardiometabolic traits through effects on hepatic thiamine content. PLoS Biol. 2018, 16, e2002907. [Google Scholar] [CrossRef] [Green Version]
- Uniprot Q9HC21 (TPC_HUMAN). Available online: https://www.uniprot.org/uniprot/Q9HC21 (accessed on 28 August 2021).
- Zhang, K.; Huentelman, M.J.; Rao, F.; Sun, E.I.; Corneveaux, J.J.; Schork, A.J.; Wei, Z.; Waalen, J.; Miramontes-Gonzalez, J.P.; Hightower, C.M.; et al. Genetic implication of a novel thiamine transporter in human hypertension. J. Am. Coll. Cardiol. 2014, 63, 1542–1555. [Google Scholar] [CrossRef]
- Zang, X.L.; Han, W.Q.; Yang, F.P.; Ji, K.D.; Wang, J.G.; Gao, P.J.; He, G.; Wu, S.N. Association of a SNP in SLC35F3 gene with the risk of hypertension in a Chinese han population. Front. Genet. 2016, 7, 108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nabokina, S.M.; Subramanian, V.S.; Said, H.M. The human colonic thiamine pyrophosphate transporter (hTPPT) is a glycoprotein and N-linked glycosylation is important for its function. Biochim. Biophys. Acta 2016, 1858, 866–871. [Google Scholar] [CrossRef]
- Marcé-Grau, A.; Martí-Sánchez, L.; Baide-Mairena, H.; Ortigoza-Escobar, J.D.; Pérez-Dueñas, B. Genetic defects of thiamine transport and metabolism: A review of clinical phenotypes, genetics, and functional studies. J. Inherit. Metab. Dis. 2019, 42, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Ikehata, M.; Ueda, K.; Iwakawa, S. Different involvement of DNA methylation and histone deacetylation in the expression of solute-carrier transporters in 4 colon cancer cell lines. Biol. Pharm. Bull. 2012, 35, 301–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bay, A.; Keskin, M.; Hizli, S.; Uygun, H.; Dai, A.; Gumruk, F. Thiamine-responsive megaloblastic anemia syndrome. Int. J. Hematol. 2010, 92, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Tabarki, B.; Al-Hashem, A.; Alfadhel, M. Biotin-Thiamine-Responsive Basal Ganglia Disease. In GeneReviews®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.A., Eds.; University of Washington: Seattle, WA, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK169615/ (accessed on 28 August 2021).
- Ortigoza-Escobar, J.D.; Molero-Luis, M.; Arias, A.; Oyarzabal, A.; Darín, N.; Serrano, M.; Garcia-Cazorla, A.; Tondo, M.; Hernández, M.; Garcia-Villoria, J.; et al. Free-thiamine is a potential biomarker of thiamine transporter-2 deficiency: A treatable cause of Leigh syndrome. Brain 2016, 139, 31–38. [Google Scholar] [CrossRef]
- Bottega, R.; Perrone, M.D.; Vecchiato, K.; Taddio, A.; Sabui, S.; Pecile, V.; Said, H.M.; Faletra, F. Functional analysis of the third identified SLC25A19 mutation causative for the thiamine metabolism dysfunction syndrome 4. J. Hum. Genet. 2019, 64, 1075–1081. [Google Scholar] [CrossRef]
- Moshfegh, A.; Goldman, J.; Ahuja, J.; Rhodes, D.; LaComb, R. What We Eat in America, NHANES 2005–2006: Usual Nutrient Intakes from Food and Water Compared to 1997 Dietary Reference Intakes for Vitamin D, Calcium, Phosphorus, and Magnesium; US Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2009.
- Lonsdale, D. Thiamine and magnesium deficiencies: Keys to disease. Med. Hypotheses 2015, 84, 129–134. [Google Scholar] [CrossRef]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional Roles of B-Vitamins in the Gut and Gut Microbiome. Mol. Nutr. Food Res. 2020, 64, 2000426. [Google Scholar] [CrossRef]
- Said, H.M. Intestinal absorption of water-soluble vitamins in health and disease. Biochem. J. 2011, 437, 357–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of dietary and microbial vitamin B family in the regulation of host immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraft, C.E.; Angert, E.R. Competition for vitamin B1 (thiamin) structures numerous ecological interactions. Q. Rev. Biol. 2017, 92, 151–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, J.; Robinson, C.; Danhof, H.; Knetsch, C.W.; van Leeuwen, H.C.; Lawley, T.D.; Auchtung, J.M.; Britton, R.A. Dietary trehalose enhances virulence of epidemic Clostridium difficile. Nature 2018, 553, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Costliow, Z.A.; Degnan, P.H. Thiamine acquisition strategies impact metabolism and competition in the gut microbe Bacteroides thetaiotaomicron. MSystems 2017, 2, e00116-17. [Google Scholar] [CrossRef] [Green Version]
- Lakhani, S.V.; Shah, H.N.; Alexander, K.; Finelli, F.C.; Kirkpatrick, J.R.; Koch, T.R. Small intestinal bacterial overgrowth and thiamine deficiency after Roux-en-Y gastric bypass surgery in obese patients. Nutr. Res. 2008, 28, 293–298. [Google Scholar] [CrossRef]
- Jenkins, A.H.; Schyns, G.; Potot, S.; Sun, G.; Begley, T.P. A new thiamin salvage pathway. Nat. Chem. Biol. 2007, 3, 492–497. [Google Scholar] [CrossRef]
- Gangolf, M.; Czerniecki, J.; Radermecker, M.; Detry, O.; Nisolle, M.; Jouan, C.; Martin, D.; Chantraine, F.; Lakaye, B.; Wins, P.; et al. Thiamine status in humans and content of phosphorylated thiamine derivatives in biopsies and cultured cells. PLoS ONE 2010, 5, e13616. [Google Scholar] [CrossRef] [Green Version]
- Tallaksen, C.M.E.; Sande, A.; Bøhmer, T.; Bell, H.; Karlsen, J. Kinetics of thiamin and thiamin phosphate esters in human blood, plasma and urine after 50 mg intravenously or orally. Eur. J. Clin. Pharmacol. 1993, 44, 73–78. [Google Scholar] [CrossRef]
- Laird, E.; Molloy, A.M. Water-Soluble Vitamins and Essential Nutrients. Ref. Modul. Biomed. Sci. 2014. [Google Scholar] [CrossRef]
- Donnino, M.W.; Carney, E.; Cocchi, M.N.; Barbash, I.; Chase, M.; Joyce, N.; Chou, P.P.; Ngo, L. Thiamine deficiency in critically ill patients with sepsis. J. Crit. Care 2010, 25, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Elmadfa, I.; Majchrzak, D.; Rust, P.; Genser, D. The thiamine status of adult humans depends on carbohydrate intake. Int. J. Vitam. Nutr. Res. 2001, 71, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Gruber, M. Nature of the Vitamin B 1-sparing Action of Fat. Nature 1950, 166, 78. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S. The influence of pH, water activity, and reducing sugars on kinetics of thermal thiamin breakdown in model systems and ground pork. In Retrospective Theses and Dissertations; Iowa State University: Ames, IA, USA, 1988. [Google Scholar]
- Watson, A.J.S.; Walker, J.F.; Tomkin, G.H.; Finn, M.M.R.; Keogh, J.A.B. Acute Wernickes encephalopathy precipitated by glucose loading. Ir. J. Med Sci. 1981, 150, 301. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, S.; Nelson-Piercy, C. Management of nausea and vomiting in pregnancy. BMJ 2011, 342, d3606. [Google Scholar] [CrossRef] [Green Version]
- Schabelman, E.; Kuo, D. Glucose before thiamine for Wernicke encephalopathy: A literature review. J. Emerg. Med. 2012, 42, 488–494. [Google Scholar] [CrossRef]
- Lustig, R.H. Ultraprocessed food: Addictive, toxic, and ready for regulation. Nutrients 2020, 12, 3401. [Google Scholar] [CrossRef]
- Ferder, L.; Ferder, M.D.; Inserra, F. The role of high-fructose corn syrup in metabolic syndrome and hypertension. Curr. Hypertens. Rep. 2010, 12, 105–112. [Google Scholar] [CrossRef]
- Åkesson, A.; Larsson, S.C.; Discacciati, A.; Wolk, A. Low-risk diet and lifestyle habits in the primary prevention of myocardial infarction in men: A population-based prospective cohort study. J. Am. Coll. Cardiol. 2014, 64, 1299–1306. [Google Scholar] [CrossRef] [Green Version]
- De la Monte, S.M.; Wands, J.R. Alzheimer’s disease is type 3 diabetes—Evidence reviewed. J. Diabetes Sci. Technol. 2008, 2, 1101–1113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araújo, J.; Cai, J.; Stevens, J. Prevalence of optimal metabolic health in American adults: National Health and Nutrition Examination Survey 2009–2016. Metab. Syndr. Relat. Disord. 2019, 17, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, N.; Thornalley, P.J. Emerging role of thiamine therapy for prevention and treatment of early-stage diabetic nephropathy. Diabetes, Obes. Metabol. 2011, 13, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Bloomgarden, Z. Diabetes and branched-chain amino acids: What is the link? J. Diabetes 2018, 10, 350–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karwi, Q.G.; Uddin, G.M.; Ho, K.L.; Lopaschuk, G.D. Loss of metabolic flexibility in the failing heart. Front. Cardiovasc. Med. 2018, 5, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lonsdale, D. A review of the biochemistry, metabolism and clinical benefits of thiamin (e) and its derivatives. Evid.-Based Complementary Altern. Med. 2006, 3, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Alshakhshir, N.; Zhao, L. Glycolytic Metabolism, Brain Resilience, and Alzheimer’s Disease. Front. Neurosci. 2021, 15, 476. [Google Scholar]
- Kohda, Y.; Tanaka, T.; Matsumura, H. Effect of thiamine repletion on cardiac fibrosis and protein O-glycosylation in diabetic cardiomyopathy. J. Diabetes Metab. 2013, 4. [Google Scholar]
- Babaei-Jadidi, R.; Karachalias, N.; Ahmed, N.; Battah, S.; Thornalley, P.J. Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes 2003, 52, 2110–2120. [Google Scholar] [CrossRef] [Green Version]
- Katare, R.G.; Caporali, A.; Oikawa, A.; Meloni, M.; Emanueli, C.; Madeddu, P. Vitamin B1 analog benfotiamine prevents diabetes-induced diastolic dysfunction and heart failure through Akt/Pim-1–mediated survival pathway. Circ. Heart Fail. 2010, 3, 294–305. [Google Scholar] [CrossRef] [Green Version]
- Pan, X.; Gong, N.; Zhao, J.; Yu, Z.; Gu, F.; Chen, J.; Sun, X.; Zhao, L.; Yu, M.; Xu, Z.; et al. Powerful beneficial effects of benfotiamine on cognitive impairment and β-amyloid deposition in amyloid precursor protein/presenilin-1 transgenic mice. Brain 2010, 133, 1342–1351. [Google Scholar] [CrossRef]
- González-Ortiz, M.; Martínez-Abundis, E.; Robles-Cervantes, J.A.; Ramírez-Ramírez, V.; Ramos-Zavala, M.G. Effect of thiamine administration on metabolic profile, cytokines and inflammatory markers in drug-naïve patients with type 2 diabetes. Eur. J. Nutr. 2011, 50, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Nikolić, A.; Kačar, A.; Lavrnić, D.; Basta, I.; Apostolski, S. The effect of benfothiamine in the therapy of diabetic polyneuropathy. Srpski Arhiv Celokupno Lekarstvo 2009, 137, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Al-Attas, O.; Al-Daghri, N.; Alokail, M.; Abd-Alrahman, S.; Vinodson, B.; Sabico, S. Metabolic benefits of six-month thiamine supplementation in patients with and without diabetes mellitus type 2. Clin. Med. Insights: Endocrinol. Diabetes 2014, 7, CMED-S13573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eshak, E.S.; Arafa, A.E. TD and cardiovascular disorders. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 965–972. [Google Scholar] [CrossRef]
- Seligmann, H.; Halkin, H.; Rauchfleisch, S.; Kaufmann, N.; Tal, R.; Motro, M.; Vered, Z.V.I.; Ezra, D. TD in patients with congestive heart failure receiving long-term furosemide therapy: A pilot study. Am. J. Med. 1991, 91, 151–155. [Google Scholar] [CrossRef]
- Schoenenberger, A.W.; Schoenenberger-Berzins, R.; Der Maur, C.A.; Suter, P.M.; Vergopoulos, A.; Erne, P. Thiamine supplementation in symptomatic chronic heart failure: A randomized, double-blind, placebo-controlled, cross-over pilot study. Clin. Res. Cardiol. 2012, 101, 159–164. [Google Scholar] [CrossRef]
- Martínez Steele, E.; Khandpur, N.; da Costa Louzada, M.L.; Monteiro, C.A. Association between dietary contribution of ultra-processed foods and urinary concentrations of phthalates and bisphenol in a nationally representative sample of the US population aged 6 years and older. PLoS ONE 2020, 15, e0236738. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Jiang, M.; Huang, D.; Zhang, H.; Feng, L.; Chen, Y.; Zhu, X.; Wang, S.; Han, J. Synergistic effects of the enhancements to mitochondrial ROS, p53 activation and apoptosis generated by aspartame and potassium sorbate in HepG2 cells. Molecules 2019, 24, 457. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.N.; Leung, M.C.; Rooney, J.P.; Sendoel, A.; Hengartner, M.O.; Kisby, G.E.; Bess, A.S. Mitochondria as a target of environmental toxicants. Toxicol. Sci. 2013, 134, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.; Ahn, S.Y.; Song, I.C.; Chung, M.H.; Jang, H.C.; Park, K.S.; Lee, K.U.; Pak, Y.K.; Lee, H.K. Chronic exposure to the herbicide, atrazine, causes mitochondrial dysfunction and insulin resistance. PLoS ONE 2009, 4, e5186. [Google Scholar] [CrossRef]
- Peixoto, F. Comparative effects of the Roundup and glyphosate on mitochondrial oxidative phosphorylation. Chemosphere 2005, 61, 1115–1122. [Google Scholar] [CrossRef]
- Peixoto, F.; Vicente, J.A.; Madeira, V.M. The herbicide dicamba (2-methoxy-3, 6-dichlorobenzoic acid) interacts with mitochondrial bioenergetic functions. Arch. Toxicol. 2003, 77, 403–409. [Google Scholar] [CrossRef]
- Peillex, C.; Pelletier, M. The impact and toxicity of glyphosate and glyphosate-based herbicides on health and immunity. J. Immunotoxicol. 2020, 17, 163–174. [Google Scholar] [CrossRef]
- Kohlmeier, M. Nutrient Metabolism. Handbook of Nutrients, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2015; Nutrient Metabolism Chapter 10—Water-Soluble Vitamins and Non Nutrients; pp. 567–671. [Google Scholar]
- Rindi, G.; Imarisio, L.; Patrini, C. Effects of acute and chronic ethanol administration on regional thiamin pyrophosphokinase activity of the rat brain. Biochem. Pharmacol. 1986, 35, 3903–3908. [Google Scholar] [CrossRef]
- Laforenza, U.; Patrini, C.; Gastaldi, G.; Rindi, G. Effects of acute and chronic ethanol administration on thiamine metabolizing enzymes in some brain areas and in other organs of the rat. Alcohol Alcohol. 1990, 25, 591–603. [Google Scholar] [CrossRef]
- Van der Werff, A.S.A.; Klooster, A. Relationship of alcohol intake and TD in heart failure. OA Alcohol 2013, 1, 4. [Google Scholar]
- Engen, P.A.; Green, S.J.; Voigt, R.M.; Forsyth, C.B.; Keshavarzian, A. The gastrointestinal microbiome: Alcohol effects on the composition of intestinal microbiota. Alcohol Res. Curr. Rev. 2015, 37, 223. [Google Scholar]
- Srinivasan, P.; Thrower, E.C.; Loganathan, G.; Balamurugan, A.N.; Subramanian, V.S.; Gorelick, F.S.; Said, H.M. Chronic Nicotine Exposure In Vivo and In Vitro Inhibits Vitamin B1 (Thiamin) Uptake by Pancreatic Acinar Cells. PLoS ONE 2015, 10, e0143575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rathanaswami, P.; Pourany, A.; Sundaresan, R. Effects of TDon the secretion of insulin and the metabolism of glucose in isolated rat pancreatic islets. Biochem. Int. 1991, 25, 577–583. [Google Scholar] [PubMed]
- Alexandre, M.; Pandol, S.J.; Gorelick, F.S.; Thrower, E.C. The emerging role of smoking in the development of pancreatitis. Pancreatology 2011, 11, 469–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NCA Releases Atlas of American Coffee. Available online: https://www.ncausa.org/Newsroom/NCA-releases-Atlas-of-American-Coffee (accessed on 12 August 2021).
- Will, Y.; Dykens, J. Mitochondrial toxicity assessment in industry—A decade of technology development and insight. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1061–1067. [Google Scholar] [CrossRef]
- Shobha. Antibiotics and Nutritional Implications- The Drugs-Nutrients Interactions. Acta Sci. Nutr. Health 2019, 3, 51–54. [Google Scholar]
- Vora, B.; Green, E.A.E.; Khuri, N.; Ballgren, F.; Sirota, M.; Giacomini, K.M. Drug–nutrient interactions: Discovering prescription drug inhibitors of the thiamine transporter ThTR-2 (SLC19A3). Am. J. Clin. Nutr. 2019, 111, 110–121. [Google Scholar] [CrossRef]
- Liang, X.; Chien, H.C.; Yee, S.W.; Giacomini, M.M.; Chen, E.C.; Piao, M.; Hao, J.; Twelves, J.; Lepist, E.I.; Ray, A.S.; et al. Metformin Is a Substrate and Inhibitor of the Human Thiamine Transporter, THTR-2 (SLC19A3). Mol. Pharm. 2015, 12, 4301–4310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neustadt, J.; Pieczenik, S.R. Medication-induced mitochondrial damage and disease. Mol. Nutr. Food Res. 2008, 52, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Alston, T.A.; Abeles, R.H. Enzymatic conversion of the antibiotic metronidazole to an analog of thiamine. Arch. Biochem. Biophys. 1987, 257, 357–362. [Google Scholar] [CrossRef]
- Giacomini, M.M.; Hao, J.; Liang, X.; Chandrasekhar, J.; Twelves, J.; Whitney, J.A.; Lepist, E.I.; Ray, A.S. Interaction of 2, 4-diaminopyrimidine–containing drugs including fedratinib and trimethoprim with thiamine transporters. Drug Metab. Dispos. 2017, 45, 76–85. [Google Scholar] [CrossRef]
- Karadima, V.; Kraniotou, C.; Bellos, G.; Tsangaris, G.T. Drug-micronutrient interactions: Food for thought and thought for action. EPMA J. 2016, 7, 10. [Google Scholar] [CrossRef] [Green Version]
- Wallace, K.B. Drug-induced mitochondrial neuropathy in children: A conceptual framework for critical windows of development. J. Child Neurol. 2014, 29, 1241–1248. [Google Scholar] [CrossRef]
- Nixon, P.; Diefenbach, R.; Duggleby, R. Inhibition of transketolase and pyruvate decarboxylase by omeprazole. Biochem. Pharm. 1992, 44, 177–179. [Google Scholar] [CrossRef]
- Suter, P.M.; Haller, J.; Hany, A.; Vetter, W. Diuretic use: A risk for subclinical TD in elderly patients. J. Nutr. Health Aging 2000, 4, 69–71. [Google Scholar] [PubMed]
- Onishi, H.; Sato, I.; Uchida, N.; Takahashi, T.; Furuya, D.; Ebihara, Y.; Yoshioka, A.; Ishida, M. High proportion of TD in referred cancer patients with delirium: A retrospective descriptive study. Eur. J. Clin. Nutr. 2021. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Halli-Tierney, A.D.; Scarbrough, C.; Carroll, D. Polypharmacy: Evaluating risks and deprescribing. Am. Fam. Physician 2019, 100, 32–38. [Google Scholar]
- Zolkipli-Cunningham, Z.; Falk, M.J. Clinical effects of chemical exposures on mitochondrial function. Toxicology 2017, 391, 90–99. [Google Scholar] [CrossRef]
- Meyer, J.N.; Hartman, J.H.; Mello, D.F. Mitochondrial toxicity. Toxicol. Sci. 2018, 162, 15–23. [Google Scholar] [CrossRef]
- Swanson, N.L.; Hoy, J.; Seneff, S. Evidence that glyphosate is a causative agent in chronic sub-clinical metabolic acidosis and mitochondrial dysfunction. Int. J. Hum. Nutr. Funct. Med. 2016, 4. [Google Scholar]
- Singh, A.P.; Goel, R.K.; Kaur, T. Mechanisms pertaining to arsenic toxicity. Toxicol. Int. 2011, 18, 87. [Google Scholar]
- Williams, R.D.; Mason, H.L.; Power, M.H.; Wilder, R.M. Induced thiamine (vitamin B1) deficiency in man: Relation of depletion of thiamine to development of biochemical defect and of polyneuropathy. Arch. Intern. Med. 1943, 71, 38–53. [Google Scholar] [CrossRef]
| In Vivo | |
|---|---|
| Study | Outcome |
| Effect of thiamine repletion on cardiac fibrosis and protein O-glycosylation in diabetic cardiomyopathy [138]. | In STZ induced diabetic rats, thiamine reduced or reversed hyperglycemia related activation of secondary glucose pathways (polyol/sorbitol, hexosamine, diacylglycerol/PKC, AGE) via upregulation of the PDC enzyme; improved cardiac contractility, reduced cardiac fibrosis and expression of mRNA associated proteins (thrombospondin, fibroconnection, plasminogen activator inhibitor 1, and connective tissue growth factor); and prevented obesity in the overfed arm of the experiment. |
| Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine [139]. | High-dose thiamine and benfotiamine therapy increased TKT and PDC activity in STZ induced diabetic rats, increasing ribose-5-phostphate and reduced microalbuminuria and proteinuria by 70–80%. PKC, AGE and oxidative stress were reduced significantly. |
| Vitamin B1 analog benfotiamine prevents diabetes-induced diastolic dysfunction and heart failure through Akt/Pim-1–mediated survival pathway [140]. | Benfotiamine prevented hyperglycemia induced diastolic dysfunction and heart failure by several mechanisms in STZ induced diabetic rats |
| Powerful beneficial effects of benfotiamine on cognitive impairment and beta-amyloid deposition in amyloid precursor protein/presenilin-1 transgenic mice [141]. | Benfotiamine improved spatial memory, amyloid precursor protein/presenilin-1, reduced amyloid plaques and tau levels dose dependently after 8 weeks of treatment in mouse model. |
| In Vivo | |
| Effect of thiamine administration on metabolic profile, cytokines and inflammatory markers in drug-naïve patients with type 2 diabetes [142]. | A total of 150 mg thiamine daily significantly reduced blood glucose within a month, in randomized, placebo control trial of 24 drug naïve T2D diabetics |
| Effect of high dose thiamine therapy on risk factors in type 2 diabetics [68]. | A 3 month, randomized, placebo controlled trial of 50 T2D patients, given 100 mg 3x thiamine per day. Thiamine significantly improved micro albuminuria, glycated hemoglobin, while decreasing PKC levels. |
| The effect of benfotiamine in the therapy of diabetic polyneuropathy [143]. | After 45 days of benfotiamine and vitamin B6 supplementation, 19/22 patients saw statically significant reductions in pain, symptom scores, neurophysiological and biological markers of diabetic neuropathy |
| Metabolic benefits of six-month thiamine supplementation in patients with and without diabetes mellitus type 2 [144]. | Six month randomized trial, 60 T2D with medication controlled blood sugar and 26 age- and BMI-matched controls. A total of 100 mg thiamine daily, significantly corrected lipid profiles and creatinine levels. |
| Thiamine deficiency and cardiovascular disorders [145]. | One time administration of 100 mg IV thiamine, improved endothelium-dependent vasodilatation in 10 patients with TD2 during an acute glucose tolerance test. |
| Thiamine deficiency in patients with congestive heart failure receiving long-term furosemide therapy: a pilot study [146]. | A total of 200 mg/day of thiamine for 1 week in 6 patients with HF receiving diuretics improved left ventricular ejection fraction (LVEF) in four of the patients from 24% to 37%. |
| Thiamine supplementation in symptomatic chronic heart failure: a randomized, double-blind, placebo-controlled, cross-over pilot study [147]. | A total of 300 mg/day oral thiamine improved LVEF significantly in HF patients on diuretics. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marrs, C.; Lonsdale, D. Hiding in Plain Sight: Modern Thiamine Deficiency. Cells 2021, 10, 2595. https://doi.org/10.3390/cells10102595
Marrs C, Lonsdale D. Hiding in Plain Sight: Modern Thiamine Deficiency. Cells. 2021; 10(10):2595. https://doi.org/10.3390/cells10102595
Chicago/Turabian StyleMarrs, Chandler, and Derrick Lonsdale. 2021. "Hiding in Plain Sight: Modern Thiamine Deficiency" Cells 10, no. 10: 2595. https://doi.org/10.3390/cells10102595
APA StyleMarrs, C., & Lonsdale, D. (2021). Hiding in Plain Sight: Modern Thiamine Deficiency. Cells, 10(10), 2595. https://doi.org/10.3390/cells10102595






