Effect of Fertilization on Yield and Quality of Sisymbrium officinale (L.) Scop. Grown as Leafy Vegetable Crop
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Non-Destructive Determination: Chlorophyll a Fluorescence Determination
2.3. Destructive Determination
2.3.1. Total Chlorophylls and Carotenoids Measurements
2.3.2. Phenolic Index and Anthocyanins
2.3.3. Nitrate Determination
2.3.4. Total Sugar Determination
2.3.5. Glucosinolates Determination
2.4. Statistical Analyses
3. Results
3.1. Chlorophyll a Fluorescence Measurement
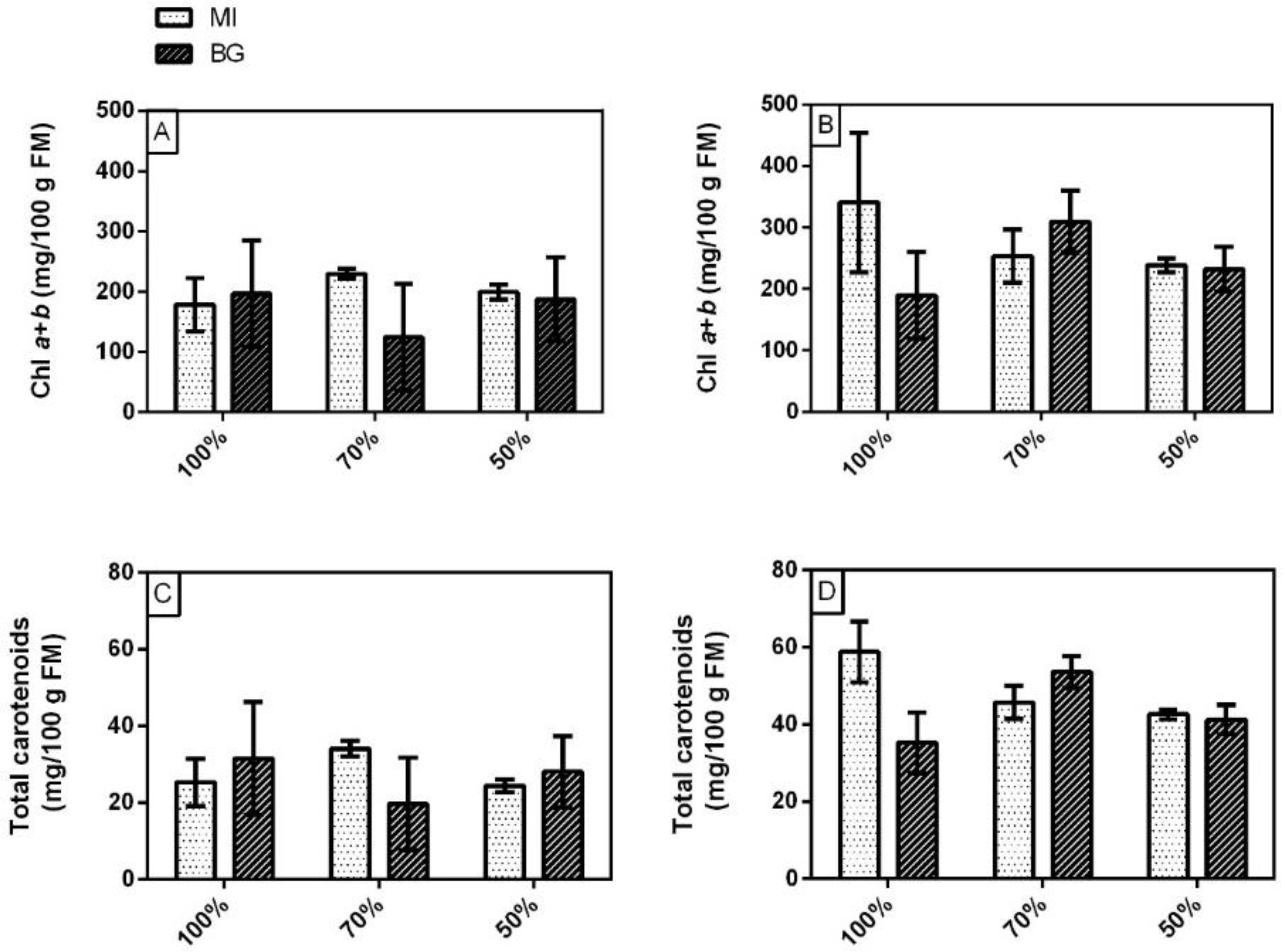
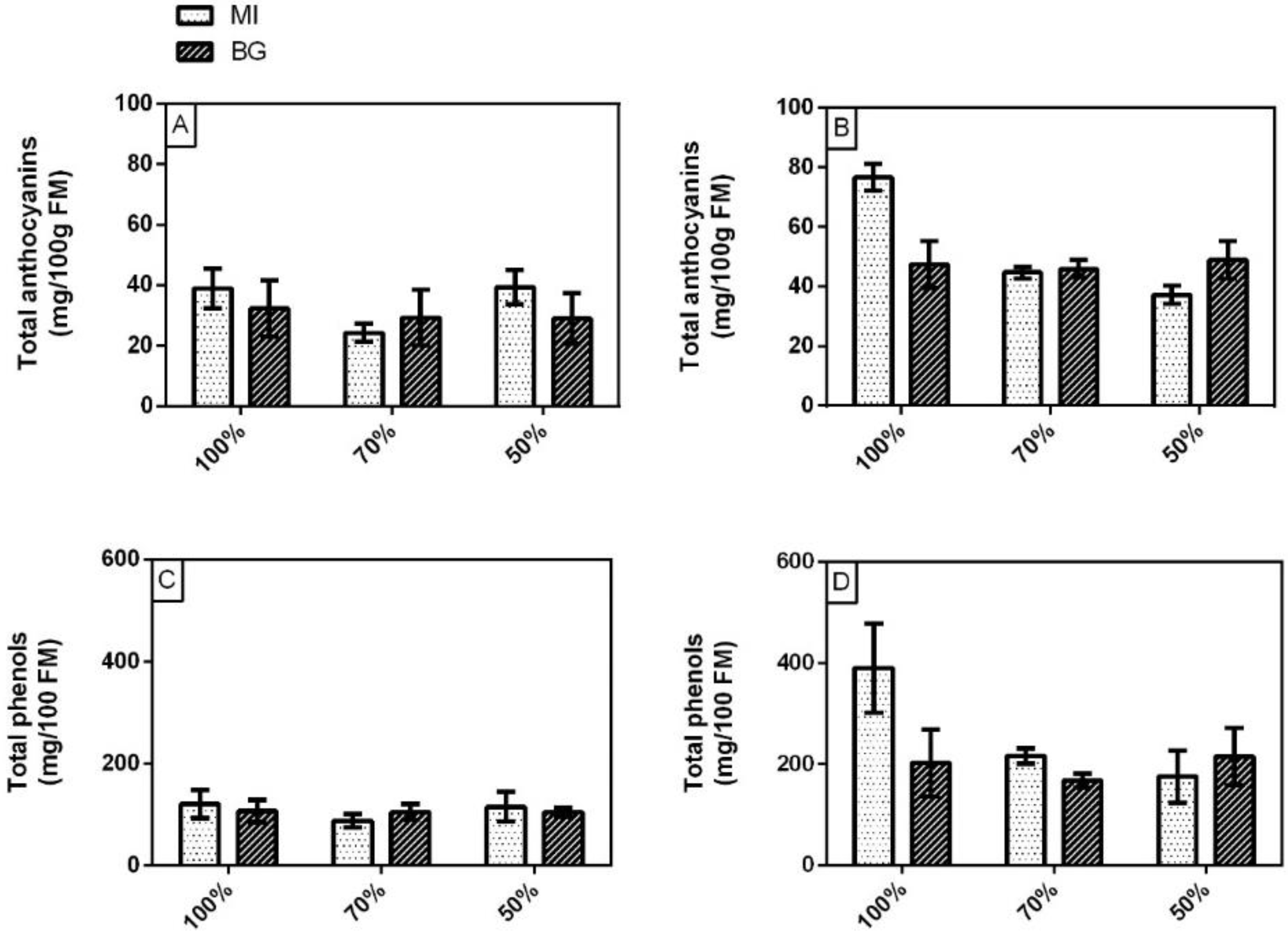
3.2. Yield, Total Chlorophylls, Carotenoids, Phenolic Index and Total Anthocyanins
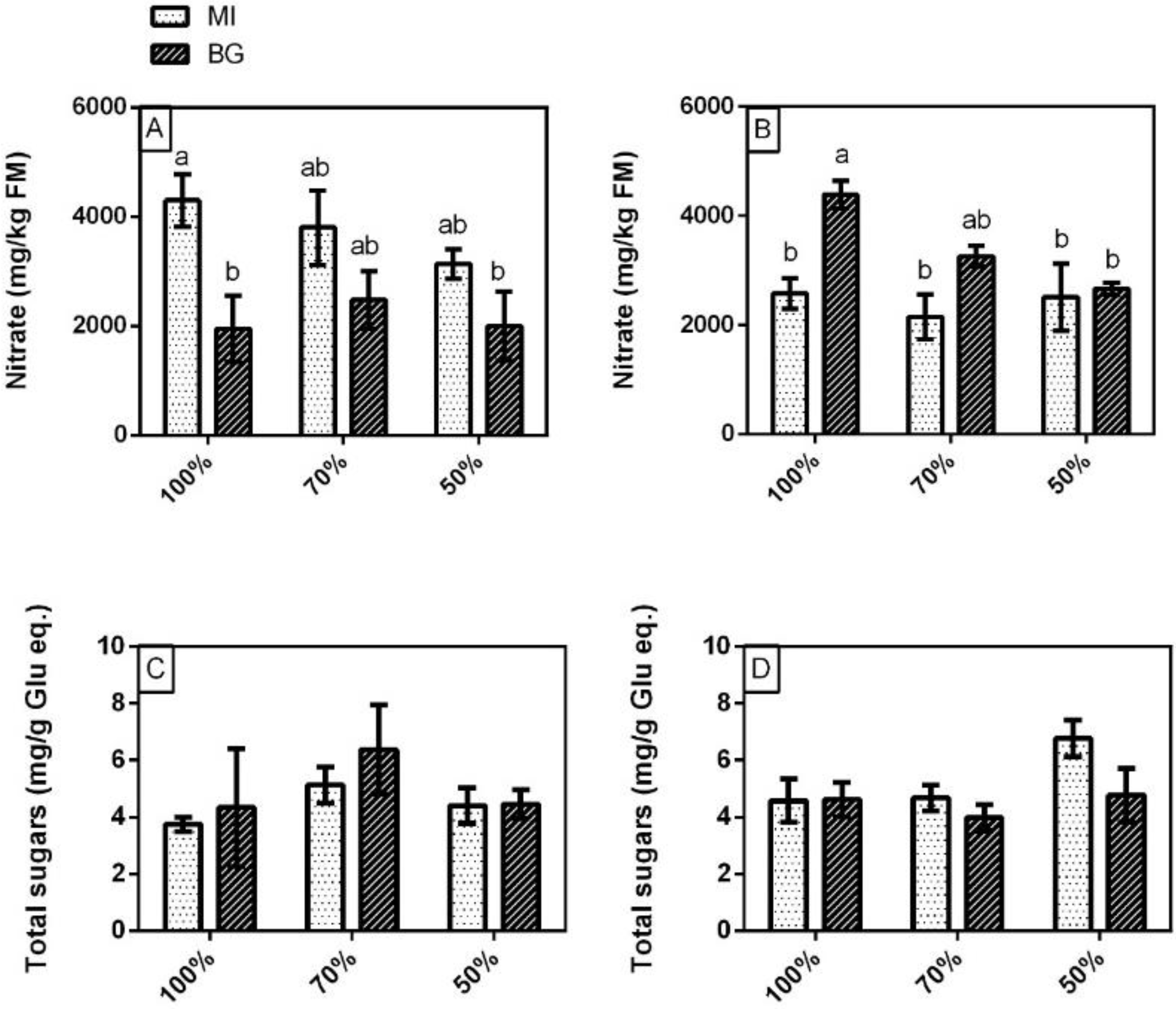
3.3. Nitrate and Total Sugars
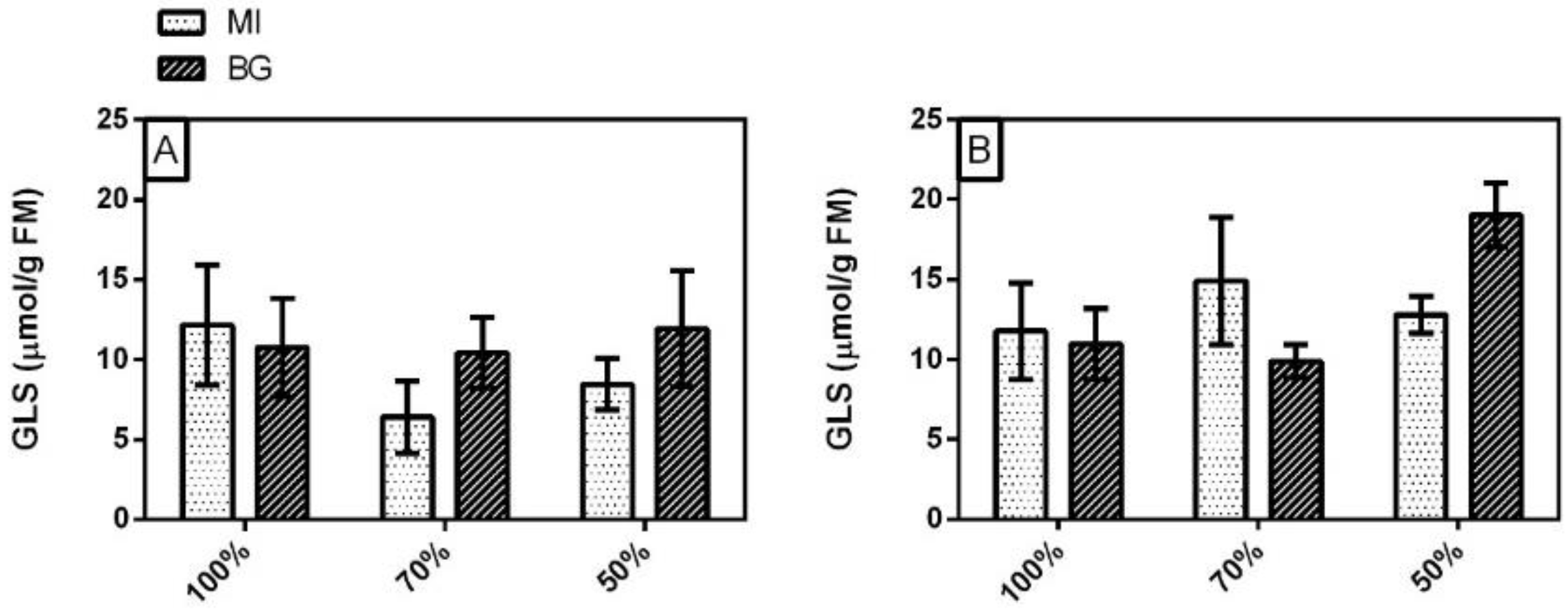
3.4. Glucosinolates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Turner, N.J.; Luczaj, L.; Migliorini, P.; Pieroni, A.; Dreon, A.L.; Sacchetti, L.E.; Paoletti, M.G. Edible and Tende Wild Plants, Traditional Ecological Knowledge and Agroecology. Crit. Rev. Plant Sci. 2011, 30, 198–225. [Google Scholar] [CrossRef]
- Bacchetta, L.; Visioli, F.; Cappelli, G.; Caruso, E.; Martin, G.; Nemeth, E.; Bacchetta, G.; Bedini, G.; Wezel, A.; Van Asseldonk, T.; et al. A manifesto for the valorisation of wild edible plants. J. Ethnopharmacol. 2016, 191, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.F.R.; Morales Gómez, P.; Morales, P.; Barros, L. Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications; John Wiley Sons Ltd.: Hoboken, NJ, USA, 2017; pp. 159–179. [Google Scholar]
- Guarrera, P.M.; Savo, V. Perceived health properties of wild and cultivated food plants in local and popular traditions of Italy: A Review. J. Ethnopharmacol. 2013, 146, 659–680. [Google Scholar] [CrossRef] [PubMed]
- European Commision. Conservation of natural habitats and wild fauna and flora. Council Directive (ECC) N. 92/43. Off. J. Eur. Communities 1992, L206, 7–50. [Google Scholar]
- Pinela, J.; Carvalho, A.M.; Ferreira, I.C.F.R. Wild edible plants: Nutritional and toxicological characteristics, retrieval strategies and importance for today’s society. Food Chem. Toxicol. 2017, 11, 165–188. [Google Scholar] [CrossRef] [PubMed]
- Luczaj, L.; Pieroni, A.; Tardío, J.; Pardo-de-Santayana, M.; Sõukand, R.; Svanberg, I.; Kalle, R. Wild food plant use in 21st century Europe, the disappearance of old traditions and the search for new cuisines involving wild edibles. Acta Soc. Bot. Pol. 2012, 81, 359–370. [Google Scholar] [CrossRef]
- Mazzucotelli, C.A.; González-Aguilar, G.A.; Villegas-Ochoa, M.A.; Domínguez-Avila, A.J.; Ansorena, M.R.; Di Scala, K.C. Chemical characterization and functional properties of selected leafy vegetables for innovative mixed salads. J. Food Biochem. 2018, 42, e12461. [Google Scholar] [CrossRef]
- Sánchez-Mata, M.C.; Cabrera Loera, R.D.; Morales, P.; Fernández-Ruiz, V.; Cámara, M.; Diez Marqués, C.; Pardo-de-Santayana, M.; Tardío, J. Wild vegetables of the Mediterranean area as valuable sources of bioactive compounds. Genet. Resour. Crop Evol. 2012, 59, 431–443. [Google Scholar] [CrossRef]
- Pignatti, S. Flora d’Italia; Edizioni Edagricole: Bologna, Italy, 1982; Volume 2, p. 337. [Google Scholar]
- Acta Plantarum. Available online: https://www.actaplantarum.org/flora/flora_info.php?id=501324 (accessed on 14 May 2019).
- Portal to the Flora of Italy. Available online: http://dryades.units.it/floritaly/index.php?procedure=taxon_page&tipo=all&id=1324 (accessed on 14 May 2019).
- Benigni, R.; Capra, C.; Cattorini, P.E. Piante Medicinali, Chimica, Farmacologia e Terapia; Inverni e Della Beffa: Milano, Italy, 1964; pp. 558–559. [Google Scholar]
- Politi, M.; Braca, A.; Altinier, G.; Sosa, S.; Ndjoko, K.; Wolfender, J.L.; Hostettmann, K.; Jiménez-Barbero, J. Different approaches to study the traditional remedy of “hierba del canto” Sisymbrium officinale (L.) Scop. Boletin Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas 2008, 7, 30–37. [Google Scholar]
- Calcinoni, O. Sisymbrium Singers Plant Efficacy in Reducing Perceived Vocal Tract Disability. J. Otolaryngol.-ENT Res. 2017, 8, 00243. [Google Scholar] [CrossRef]
- Mithen, R. Sulphur-Cointaining Compounds. In Plant Secondary Metabolites: Occurence, Structure and Role in Human Diet; Blackwell Publishing: Hoboken, NJ, USA, 2006; Chapter 2. [Google Scholar]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Pardossi, A.; Prosdocimi Gianquinto, G.; Santamaria, P.; Incrocci, L. Orticoltura. Principi e Pratica; Edizioni Edagricole: Bologna, Italy, 2018; pp. 136–141. [Google Scholar]
- Incrocci, L.; Dimauro, B.; Santamaria, P.; Pardossi, A. La Concimazione Azotata Degli Ortaggi. Progetto Interregionale “AZORT”; Barone e Bella C.: Ragusa, Italy, 2013; pp. 7–14. [Google Scholar]
- Santamaria, P. Nitrate in vegetables: Toxicity, content, intake and EC regulation. J. Sci. Food Agric. 2006, 86, 10–17. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Nitrate in vegetables. Scientific Opinion of the Panel on Contaminants in Food chain. EFSA J. 2008, 689, 1–79. [Google Scholar]
- European Commission. Maximum Levels for Nitrates in Foodstuffs. Regulation (EU) N. 1258/2011 Amending. Off. J. Eur. Union 2011, L320, 15–17. [Google Scholar]
- Wang, Z.H.; Li, S.X.; Malhi, S. Effects of fertilization and other agronomic measures on nutritional quality of crops. J. Sci. Food Agric. 2008, 88, 7–23. [Google Scholar] [CrossRef]
- Aires, A.; Fernandes, C.; Carvalho, R.; Bennett, R.N.; Saavedra, M.J.; Rosa, E.A. Seasonal effects on bioactive compounds and antioxidant capacity of six economically important Brassica vegetables. Molecules 2011, 16, 6816–6832. [Google Scholar] [CrossRef] [PubMed]
- Podsędek, A. Natural antioxidants and antioxidant capacity of Brassica vegetables: A review. LWT-Food Sci. Technol. 2007, 40, 1–11. [Google Scholar] [CrossRef]
- Di Sotto, A.; Di Giacomo, S.; Toniolo, C.; Nicoletti, M.; Mazzanti, G. Sisymbrium Officinale (L.) Scop. and its Polyphenolic Fractions Inhibit the Mutagenicity of Tert-Butylhydroperoxide in Escherichia coli WP2uvrAR Strain. Phytother. Res. 2016, 30, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Ko, E.Y.; Keum, Y.S. Minimally processed ready-to-eat baby-leaf vegetables: Production, processing, storage, microbial safety, and nutritional potential. Food Rev. Int. 2017, 33, 644–663. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic membranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Giusti, M.M.; Rodríguez-Saona, L.E.; Wrolstad, R.E. Molar absorptivity and color characteristics of acylated and non-acylated pelargonidin-based anthocyanins. J. Agric. Food Chem. 1999, 47, 4631–4637. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, C.A.; Maroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by titration of salicylic acid. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Carnat, A.; Fraisse, D.; Carnat, A.P.; Groubert, A.; Lamaison, J.L. Normalisation de l’erysimum, Sisymbriumofficinale, L. Ann. Pharm. Fr. 1998, 56, 36–39. [Google Scholar] [PubMed]
- Mawlong, I.; Sujith Kumar, M.S.; Gurung, B.; Singh, K.H.; Singh, D. A simple spectrophotometric method for estimating total glucosinolates in mustard de-oiled cake. Int. J. Food Prop. 2017, 20, 3274–3281. [Google Scholar] [CrossRef]
- Borgonovo, G.; Guarise, M.; Zimabaldi, N.; De Nisi, P.; De Petrocellis, L.; Moriello, A.S.; Bassoli, A. Isothiocyanates and glucosinolates from Sisymbrium officinale (the “singers’ plant”): Isolation and in vitro assays on the somatosensory and pain receptors TRPA1 channel. Molecules 2019, 24, 949. [Google Scholar] [CrossRef] [PubMed]
- Tesi, R. Colture Protette. Ortoflorovivaismo in Ambiente Mediterraneo, 6th ed.; Edizioni Edagicole: Bologna, Italy, 2008; p. 223. [Google Scholar]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Krumbein, A.; Schonhof, I.; Schreiner, M. Composition and contents of phytochemicals (glucosinolates, carotenoids and chlorophylls) and ascorbic acid in selected Brassica species (B. juncea, B. rapa subsp. nipposinica var. chinoleifera, B. rapa subsp. chinensis and B. rapa subsp. rapa). J. Appl. Bot. Food Qual. 2005, 79, 168–174. [Google Scholar]
- Duma, M.; Alsina, I.; Zeipina, S.; Lepse, L.; Dubova, L. Leaf vegetables as source of phytochemicals. In Proceedings of the 9th Baltic Conference on Food Science and Technology “Food for Consumer Well-Being” FOODBALT. LLU. P, Latvia, Jelgava, 8–9 May 2014; pp. 262–265. [Google Scholar]
- Kaur, C.; Kapoor, H.C. Anti-oxidant activity and total phenolic content of some Asian vegetables. Int. J. Food Sci. Technol. 2002, 37, 153–161. [Google Scholar] [CrossRef]
- Anjana, S.U.; Iqbal, M. Nitrate accumulation in plants, factors affecting the process, and human health implications. A review. Agron. Sustain. Dev. 2007, 27, 45–57. [Google Scholar] [CrossRef]
- Colla, G.; Kim, H.J.; Kyriacou, M.C.; Rouphael, Y. Nitrate in fruits and vegetables. Sci. Hortic. 2018, 237, 221–238. [Google Scholar] [CrossRef]
- Santamaria, P.; Elia, A.; Serio, F.; Todaro, E. A survey of nitrate and oxalate content in fresh vegetables. J. Sci. Food Agric. 1999, 79, 1882–1888. [Google Scholar] [CrossRef]
- Guadagnin, S.G.; Rath, S.; Reyes, F.G.R. Evaluation of the nitrate content in leaf vegetables produced through different agricultural systems. Food Addit. Contam. 2005, 22, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Guarise, M.; Borgonovo, G.; Bassoli, A.; Ferrante, A. Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable. Horticulturae 2019, 5, 13. [Google Scholar] [CrossRef]
- Schonhof, I.; Blankenburg, D.; Müller, S.; Krumbein, A. Sulfur and nitrogen supply influence growtn, product appearance and glucosinolate concentration of broccoli. J. Plant Nutr. Soil Sci. 2007, 170, 65–72. [Google Scholar] [CrossRef]
- Hu, Y.; Liang, H.; Yuan, Q.; Hong, Y. Determination of glucosinolates in 19 Chinese medicinal plants with spectrophotometry and high-pressure liquid chromatography. Nat. Prod. Res. 2010, 24, 1195–1205. [Google Scholar] [PubMed]
| Cycle | Fertilization Level | Wild Population | Fv/Fm | PI | DIo/RC | RC/CSm |
|---|---|---|---|---|---|---|
| I | 100% | MI | 0.84 ± 0.002 | 2.51 ± 0.045 | 0.43 ± 0.003 | 2162.44 ± 434.5 |
| BG | 0.84 ± 0.006 | 2.33 ± 0.26 | 0.43 ± 0.02 | 2109.75 ± 113.73 | ||
| 70% | MI | 0.84 ± 0.003 | 2.44 ± 0.2 | 0.44 ± 0.025 | 2068.96 ± 129.3 | |
| BG | 0.84 ± 0.008 | 2.49 ± 0.37 | 0.46 ± 0.05 | 2210.5 ± 142.45 | ||
| 50% | MI | 0.84 ± 0.003 | 2.33 ± 0.2 | 0.45 ± 0.02 | 1968.51 ± 110.03 | |
| BG | 0.84 ± 0.004 | 2.28 ± 0.3 | 0.45 ± 0.03 | 2079.49 ± 195.4 | ||
| II | 100% | MI | 0.85 ± 0.004 | 4.56 ± 0.82 a | 0.31 ± 0.03 b | 3159.74 ± 369.6 a |
| BG | 0.83 ± 0.011 | 2.38 ± 0.5 ab | 0.46 ± 0.05 ab | 2085.37 ± 191.55 ab | ||
| 70% | MI | 0.85 ± 0.002 | 4.66 ± 0.47 a | 0.32 ± 0.02 b | 3062.5 ± 222.07 ab | |
| BG | 0.84 ± 0.003 | 2.88 ± 0.35 ab | 0.39 ± 0.03 ab | 2454.64 ± 213.75 ab | ||
| 50% | MI | 0.84 ± 0.006 | 2.67 ± 0.46 ab | 0.42 ± 0.03 ab | 2388.09 ± 194.22 ab | |
| BG | 0.83 ± 0.005 | 1.96 ± 0.38 b | 0.49 ± 0.04 a | 2008.36 ± 238.22 b |
| Cycle | Fertilization Level | Wild Population | Fresh Matter (g/plant) | Kg/m2 | Dry Matter (%) |
|---|---|---|---|---|---|
| I | 100% | MI | 35.5 | 0.25 | 7.6 |
| BG | 67.1 | 0.47 | 8.1 | ||
| 70% | MI | 31.8 | 0.22 | 7.2 | |
| BG | 40.7 | 0.28 | 7.8 | ||
| 50% | MI | 30 | 0.21 | 7.7 | |
| BG | 55.1 | 0.38 | 9.1 | ||
| II | 100% | MI | 33.7 | 0.23 | 13.8 |
| BG | 32.8 | 0.23 | 13.4 | ||
| 70% | MI | 41.3 | 0.29 | 11.4 | |
| BG | 28 | 0.20 | 16.2 | ||
| 50% | MI | 32.1 | 0.22 | 12.6 | |
| BG | 25.9 | 0.18 | 14.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarise, M.; Borgonovo, G.; Bassoli, A.; Ferrante, A. Effect of Fertilization on Yield and Quality of Sisymbrium officinale (L.) Scop. Grown as Leafy Vegetable Crop. Agronomy 2019, 9, 401. https://doi.org/10.3390/agronomy9070401
Guarise M, Borgonovo G, Bassoli A, Ferrante A. Effect of Fertilization on Yield and Quality of Sisymbrium officinale (L.) Scop. Grown as Leafy Vegetable Crop. Agronomy. 2019; 9(7):401. https://doi.org/10.3390/agronomy9070401
Chicago/Turabian StyleGuarise, Marta, Gigliola Borgonovo, Angela Bassoli, and Antonio Ferrante. 2019. "Effect of Fertilization on Yield and Quality of Sisymbrium officinale (L.) Scop. Grown as Leafy Vegetable Crop" Agronomy 9, no. 7: 401. https://doi.org/10.3390/agronomy9070401
APA StyleGuarise, M., Borgonovo, G., Bassoli, A., & Ferrante, A. (2019). Effect of Fertilization on Yield and Quality of Sisymbrium officinale (L.) Scop. Grown as Leafy Vegetable Crop. Agronomy, 9(7), 401. https://doi.org/10.3390/agronomy9070401








