Hydrochar-Amended Substrates for Production of Containerized Pine Tree Seedlings under Different Fertilization Regimes
Abstract
1. Introduction
2. Materials and Methods
2.1. Hydrochar Preparation and Properties: Powder and Pellets
2.2. Experimental Design and Growth Condition
2.3. Seedling Measurements
2.4. Mycorrhizal Colonization Test
2.5. Data Analysis
3. Results
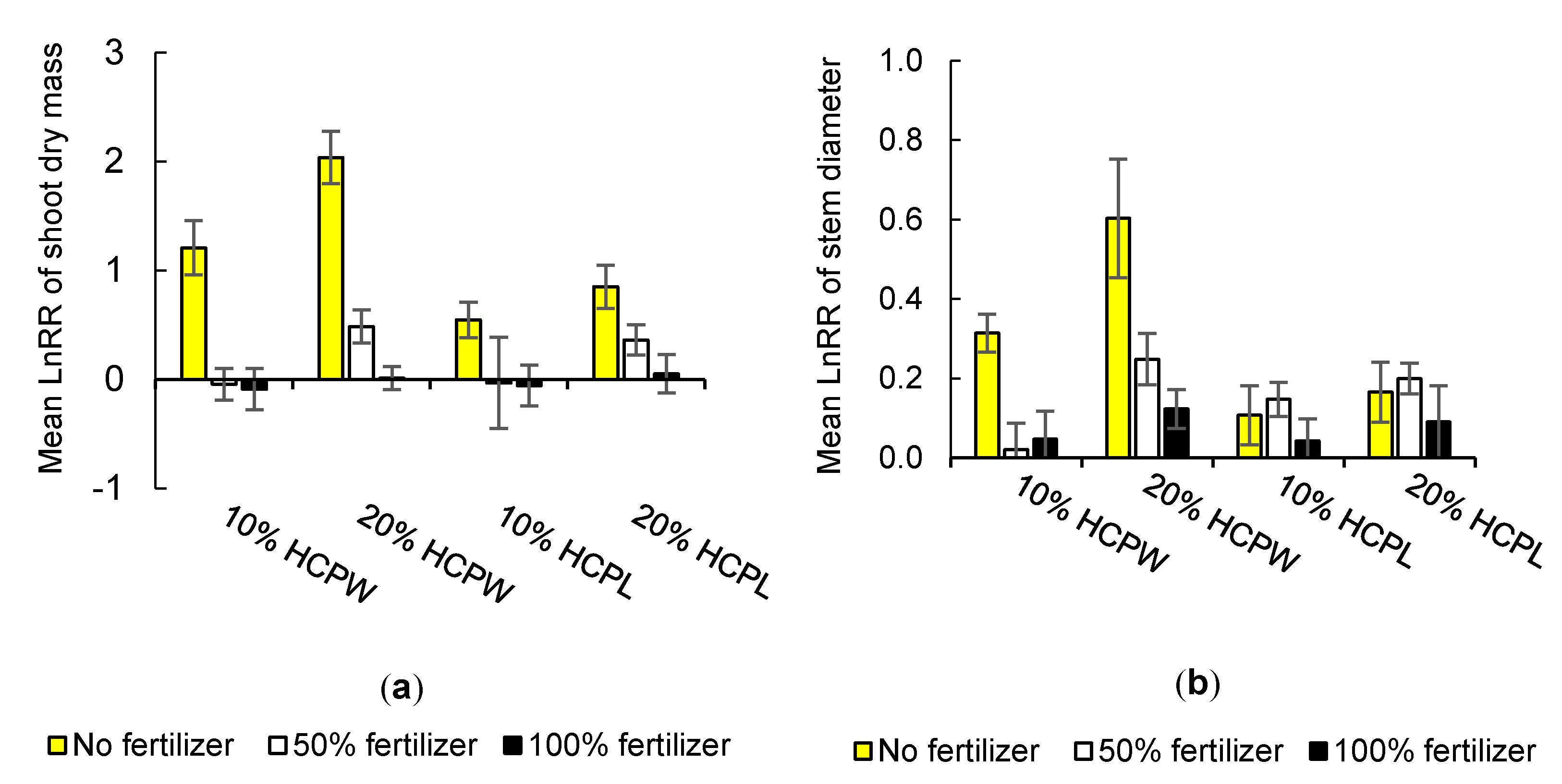
3.1. Effects of Hydrochar on Growth Parameters
3.1.1. Shoot Dry Mass
3.1.2. Stem Diameter
3.1.3. Quality Index of Dickson (QID)
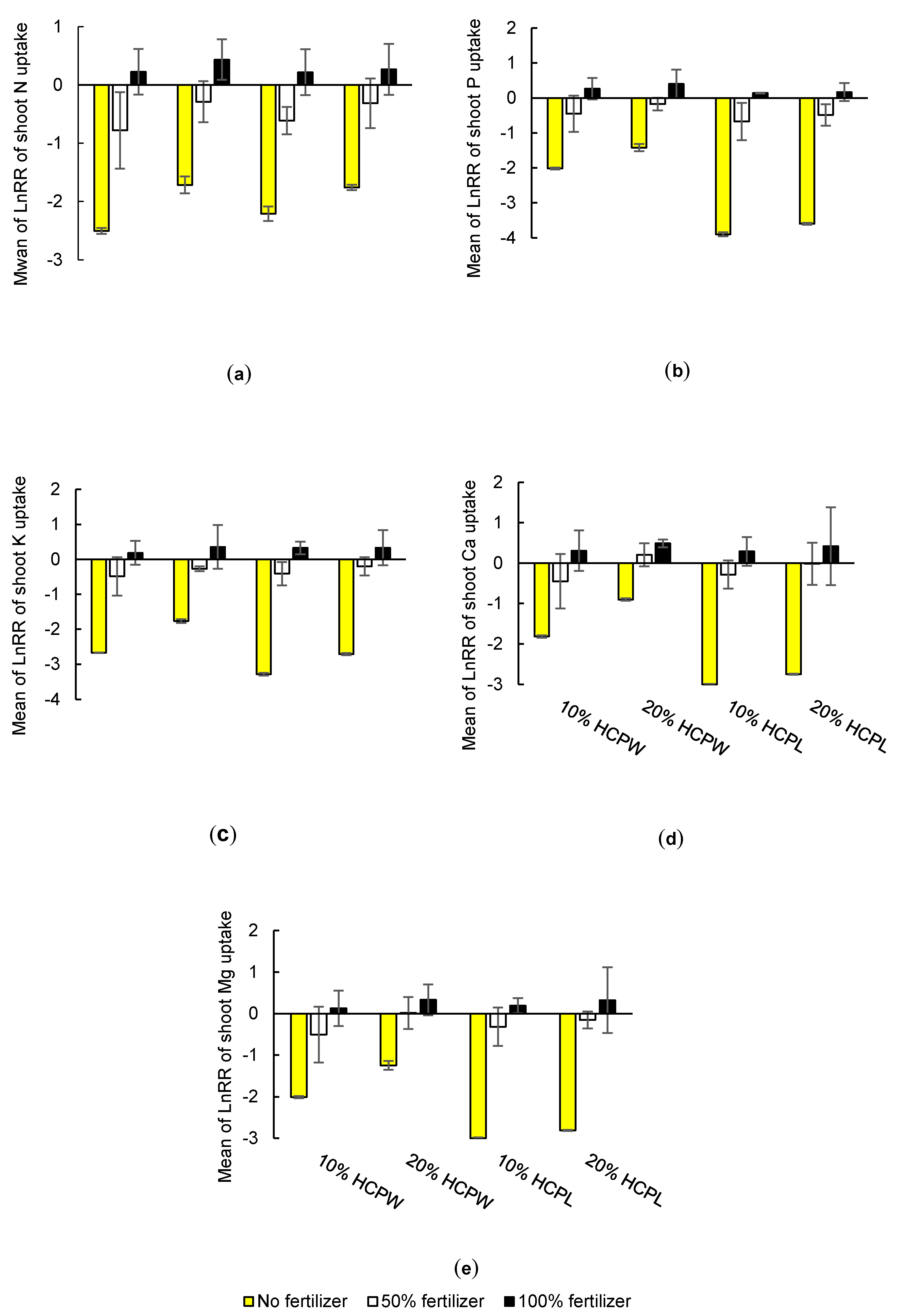
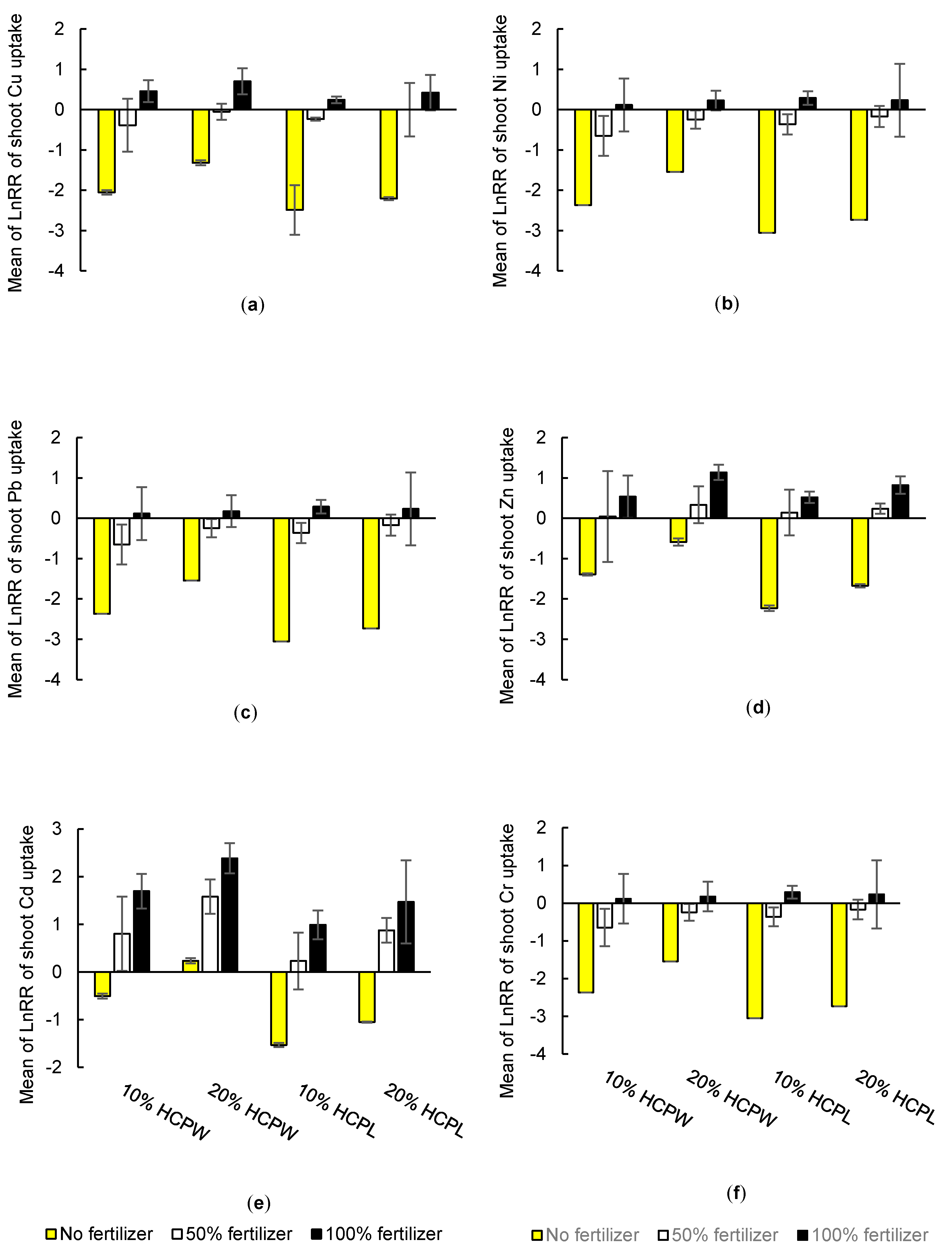
3.2. Effects of Hydrochar on Above-Ground Nutrient and Heavy Metal Uptake
3.3. Mycorrhizal Colonization Response to Hydrochar
4. Discussion
4.1. Shoot Dry Mass and Stem Diameter
4.2. Quality of The Pine Tree Seedlings
4.3. Nutrient and Heavy Metal Contents
4.4. Colonization with Ectomycorrhizal Fungi
4.5. Environmental and Economic Implications of Using Hydrochar
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Percentage of Peat Replaced (v/v) | |||||||
|---|---|---|---|---|---|---|---|
| 0 | 10 | 20 | |||||
| Fertilizer rate | 100% | 0% | 50% | 100% | 0% | 50% | 100% |
| Peat | 100 | ||||||
| Hydrochar Powder | 66 | 94 | 96 | 72 | 97 | 100 | |
| Hydrochar Pellet | 61 | 92 | 92 | 61 | 90 | 93 | |
References
- The Swedish Forest Industries’ Statistics. Economic Importance. Available online: http://www.forestindustries.se/forest-industry/statistics/economic-importance/ (accessed on 4 April 2019).
- Nilsson, P. Forest Land. Available online: https://www.slu.se/en/Collaborative-Centres-and-Projects/the-swedish-national-forest-inventory/forest-statistics/forest-statistics/skogsmark (accessed on 21 August 2018).
- Mattsson, A. Reforestation challenges in Scandinavia. Reforesta 2016, 67–85. [Google Scholar] [CrossRef]
- Bohlin, C.; Holmberg, P. Peat: Dominating growing medium in Swedish horticulture. In Proceedings of the International Symposium on Growing Media and Hydroponics, Alnarp, Sweden, 8–14 September 2001; pp. 177–181. [Google Scholar]
- Cleary, J.; Roulet, N.T.; Moore, T.R. Greenhouse Gas Emissions from Canadian Peat Extraction, 1990–2000: A Life-cycle Analysis. Ambio J. Hum. Environ. 2005, 34, 456–461. [Google Scholar] [CrossRef]
- Alexander, P.; Bragg, N.; Meade, R.; Padelopoulos, G.; Watts, O. Peat in horticulture and conservation: The UK response to a changing world. Mires Peat 2008, 3, 8. [Google Scholar]
- Dombrowsky, M.; Dixon, M. Sustainable growing substrates for potted greenhouse gerbera production. In Proceedings of the International Symposium on Responsible Peatland Management and Growing Media Production, Quebec City, Canada, 13–17 June 2011; pp. 61–68. [Google Scholar]
- Tian, Y.; Sun, X.; Li, S.; Wang, H.; Wang, L.; Cao, J.; Zhang, L. Biochar made from green waste as peat substitute in growth media for Calathea rotundifola cv. Fasciata. Sci. Hortic. 2012, 143, 15–18. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Eller, F.J.; Evangelista, R.L.; Moser, B.R.; Lee, E.; Wagner, R.E.; Peterson, S.C. Evaluation of biochar-anaerobic potato digestate mixtures as renewable components of horticultural potting media. Ind. Crop. Prod. 2015, 65, 467–471. [Google Scholar] [CrossRef]
- Dumroese, K.R.; Pinto, J.R.; Heiskanen, J.; Tervahauta, A.; McBurney, K.G.; Page-Dumroese, D.S.; Englund, K. Biochar Can Be a Suitable Replacement for Sphagnum Peat in Nursery Production of Pinus ponderosa Seedlings. Forests 2018, 9, 232. [Google Scholar] [CrossRef]
- Matt, C.P.; Keyes, C.R.; Dumroese, R.K. Biochar effects on the nursery propagation of 4 northern Rocky Mountain native plant species. Nativ. Plants J. 2018, 19, 14–26. [Google Scholar] [CrossRef]
- Huber, G.W.; Iborra, S.; Corma, A. Synthesis of transportation fuels from biomass: Chemistry, catalysts, and engineering. Chem. Rev. 2006, 106, 4044–4098. [Google Scholar] [CrossRef]
- Glaser, B.; Haumaier, L.; Guggenberger, G.; Zech, W. The ‘Terra Preta’ phenomenon: A model for sustainable agriculture in the humid tropics. Naturwissenschaften 2001, 88, 37–41. [Google Scholar] [CrossRef]
- Marris, E. Black is the new green. Nature 2006, 442, 624–626. [Google Scholar] [CrossRef]
- Mohammadi, A.; Cowie, A.; Anh Mai, T.L.; de la Rosa, R.A.; Kristiansen, P.; Brandão, M.; Joseph, S. Biochar use for climate-change mitigation in rice cropping systems. J. Clean. Prod. 2016, 116, 61–70. [Google Scholar] [CrossRef]
- Mohammadi, A.; Cowie, A.L.; Anh Mai, T.L.; Brandão, M.; Anaya de la Rosa, R.; Kristiansen, P.; Joseph, S. Climate-change and health effects of using rice husk for biochar-compost: Comparing three pyrolysis systems. J. Clean. Prod. 2017, 162, 260–272. [Google Scholar] [CrossRef]
- Mohammadi, A.; Cowie, A.L.; Cacho, O.; Kristiansen, P.; Anh Mai, T.L.; Joseph, S. Biochar addition in rice farming systems: Economic and energy benefits. Energy 2017, 140, 415–425. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.; Yu, S.-H.; Antonietti, M.; Titirici, M.-M. Engineering Carbon Materials from the Hydrothermal Carbonization Process of Biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Laird, D.; Fleming, P.; Wang, B.; Horton, R.; Karlen, D. Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma 2010, 158, 436–442. [Google Scholar] [CrossRef]
- Di Lonardo, S.; Baronti, S.; Vaccari, F.P.; Albanese, L.; Battista, P.; Miglietta, F.; Bacci, L. Biochar-based nursery substrates: The effect of peat substitution on reduced salinity. Urban For. Urban Green. 2017, 23, 27–34. [Google Scholar] [CrossRef]
- Sarauer, J.L.; Coleman, M.D. Biochar as a growing media component for containerized production of Douglas-fir. Can. J. For. Res. 2018, 48, 581–588. [Google Scholar] [CrossRef]
- Paneque, M.; De la Rosa, J.M.; Franco-Navarro, J.D.; Colmenero-Flores, J.M.; Knicker, H. Effect of biochar amendment on morphology, productivity and water relations of sunflower plants under non-irrigation conditions. Catena 2016, 147, 280–287. [Google Scholar] [CrossRef]
- De la Rosa, J.M.; Paneque, M.; Miller, A.Z.; Knicker, H. Relating physical and chemical properties of four different biochars and their application rate to biomass production of Lolium perenne on a Calcic Cambisol during a pot experiment of 79days. Sci. Total Environ. 2014, 499, 175–184. [Google Scholar] [CrossRef]
- Mohammadi, A.; Sandberg, M.; Venkatesh, G.; Eskandari, S.; Dalgaard, T.; Joseph, S.; Granström, K. Environmental analysis of producing biochar and energy recovery from pulp and paper mill biosludge. J. Ind. Ecol. 2019. [Google Scholar] [CrossRef]
- Mohammadi, A.; Sandberg, M.; Venkatesh, G.; Eskandari, S.; Dalgaard, T.; Joseph, S.; Granström, K. Environmental performance of end-of-life handling alternatives for paper-and-pulp-mill sludge: Using digestate as a source of energy or for biochar production. Energy 2019, 182, 594–605. [Google Scholar] [CrossRef]
- Kambo, H.S.; Dutta, A. A comparative review of biochar and hydrochar in terms of production, physico-chemical properties and applications. Renew. Sustain. Energy Rev. 2015, 45, 359–378. [Google Scholar] [CrossRef]
- George, E.; Ventura, M.; Panzacchi, P.; Scandellari, F.; Tonon, G. Can hydrochar and pyrochar affect nitrogen uptake and biomass allocation in poplars? J. Plant Nutr. Soil Sci. 2017, 180, 178–186. [Google Scholar] [CrossRef]
- Toufiq, M.R.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal carbonization of biomass for energy and crop production. Appl. Bioenergy 2014, 1, 11–29. [Google Scholar] [CrossRef]
- Fornes, F.; Belda, R.M. Biochar versus hydrochar as growth media constituents for ornamental plant cultivation. Sci. Agric. 2018, 75, 304–312. [Google Scholar] [CrossRef]
- Belda, R.M.; Lidón, A.; Fornes, F. Biochars and hydrochars as substrate constituents for soilless growth of myrtle and mastic. Ind. Crop. Prod. 2016, 94, 132–142. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Heiskanen, J.; Englund, K.; Tervahauta, A. Pelleted biochar: Chemical and physical properties show potential use as a substrate in container nurseries. Biomass Bioenergy 2011, 35, 2018–2027. [Google Scholar] [CrossRef]
- Robertson, S.J.; Rutherford, P.M.; Lopez-Gutierrez, J.C.; Massicotte, H.B. Biochar enhances seedling growth and alters root symbioses and properties of sub-boreal forest soils. Can. J. Soil Sci. 2012, 92, 329–340. [Google Scholar] [CrossRef]
- Rillig, M.C.; Wagner, M.; Salem, M.; Antunes, P.M.; George, C.; Ramke, H.-G.; Titirici, M.-M.; Antonietti, M. Material derived from hydrothermal carbonization: Effects on plant growth and arbuscular mycorrhiza. Appl. Soil. Ecol. 2010, 45, 238–242. [Google Scholar] [CrossRef]
- Busch, D.; Kammann, C.; Grunhage, L.; Muller, C. Simple biotoxicity tests for evaluation of carbonaceous soil additives: Establishment and reproducibility of four test procedures. J. Environ. Qual. 2012, 41, 1023–1032. [Google Scholar] [CrossRef]
- Baronti, S.; Alberti, G.; Camin, F.; Criscuoli, I.; Genesio, L.; Mass, R.; Vaccari, F.P.; Ziller, L.; Miglietta, F. Hydrochar enhances growth of poplar for bioenergy while marginally contributing to direct soil carbon sequestration. GCB Bioenergy 2017, 9, 1618–1626. [Google Scholar] [CrossRef]
- Nara, K. Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol. 2006, 169, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Ahlroth, M.; Bialik, M.; Jensen, A. Hydrothermal Carbonization of Pulp-and Paper Mill Effluent Sludge. Bioresource Technol. 2016, 200, 15–489. [Google Scholar]
- Chen, H.; Zhou, Y.; Zhao, H.; Li, Q. A comparative study on behavior of heavy metals in pyrochar and hydrochar from sewage sludge. Energy Sources Part A Recovery Util. Environ. Eff. 2018, 40, 565–571. [Google Scholar] [CrossRef]
- Jackson, D.P.; Dumroese, K.R.; Barnett, J.P. Nursery response of container Pinus palustris seedlings to nitrogen supply and subsequent effects on outplanting performance. For. Ecol. Manag. 2012, 265, 1–12. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Pinto, J.R.; Montville, M.E. Using container weights to determine irrigation needs: A simple method. Nativ. Plants J. 2015, 16, 67–71. [Google Scholar] [CrossRef]
- Dickson, A.; Leaf, A.L.; Hosner, J.F. Quality appraisal of white spruce and white pine seedling stock in nurseries. For. Chron. 1960, 36, 10–13. [Google Scholar] [CrossRef]
- Brundrett, M.; Bougher, N.; Dell, B.; Grove, T.; Malajczuk, N. Working with Mycorrhizas in Forestry and Agriculture; Australian Centre for International Agricultural Research Canberra: Canberra, Australia, 3 January 1996.
- Brose, P.H.; Dey, D.C.; Phillips, R.J.; Waldrop, T.A. A Meta-Analysis of the Fire-Oak Hypothesis: Does Prescribed Burning Promote Oak Reproduction in Eastern North America? For. Sci. 2012, 59, 322–334. [Google Scholar] [CrossRef]
- Loydi, A.; Donath, T.W.; Eckstein, R.L.; Otte, A. Non-native species litter reduces germination and growth of resident forbs and grasses: Allelopathic, osmotic or mechanical effects? Biol. Invasions 2015, 17, 581–595. [Google Scholar] [CrossRef]
- Goldberg, D.E.; Scheiner, S.M. ANOVA and ANCOVA: Field competition experiments. In Design and Analysis of Ecological Experiments, 2nd ed.; Scheiner, S.M., Gurevitch, J., Eds.; Oxford University Press Inc: Oxford, UK, 2001; pp. 77–98. [Google Scholar]
- Selander, J.; Immonen, A.; Raukko, P. Resistance of naturally regenerated and nursery-raised Scots pine seedlings to the large pine weevil. Folia For. 1990, 766, 19. [Google Scholar]
- Thorsen, Å.A.; Mattsson, S.; Weslien, J. Influence of Stem Diameter on the Survival and Growth of Containerized Norway Spruce Seedlings Attacked by Pine Weevils (Hylobius spp.). Scand. J. For. Res. 2001, 16, 54–66. [Google Scholar] [CrossRef]
- Köster, E.; Pumpanen, J.; Köster, K. Biochar as a possible new tool for afforestation practices. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 8–13 April 2018; p. 6082. [Google Scholar]
- Aung, A.; Han, S.H.; Youn, W.B.; Meng, L.; Cho, M.S.; Park, B.B. Biochar effects on the seedling quality of Quercus serrata and Prunus sargentii in a containerized production system. For. Sci. Technol. 2018, 14, 112–118. [Google Scholar] [CrossRef]
- Steinbeiss, S.; Gleixner, G.; Antonietti, M. Effect of biochar amendment on soil carbon balance and soil microbial activity. Soil Biol. Biochem. 2009, 41, 1301–1310. [Google Scholar] [CrossRef]
- Rezende, F.A.; Santos, V.A.H.F.D.; Maia, C.M.B.D.F.; Morales, M.M. Biochar in substrate composition for production of teak seedlings. Pesqui. Agropec. Bras. 2016, 51, 1449–1456. [Google Scholar] [CrossRef]
- Locke, J.C.; Altland, J.E.; Ford, C.W. Gasified Rice Hull Biochar Affects Nutrition and Growth of Horticultural Crops in Container Substrates. J. Environ. Hortic. 2013, 31, 195–202. [Google Scholar] [CrossRef]
- Álvarez, M.L.; Gascó, G.; Plaza, C.; Paz-Ferreiro, J.; Méndez, A. Hydrochars from Biosolids and Urban Wastes as Substitute Materials for Peat. Land Degrad. Dev. 2017, 28, 2268–2276. [Google Scholar] [CrossRef]
- Hytönen, J. Wood ash fertilisation increases biomass production and improves nutrient concentrations in birches and willows on two cutaway peats. Balt 2016, 22, 98–106. [Google Scholar]
- Devi, P.; Saroha, A.K. Risk analysis of pyrolyzed biochar made from paper mill effluent treatment plant sludge for bioavailability and eco-toxicity of heavy metals. Bioresour. Technol. 2014, 162, 308–315. [Google Scholar] [CrossRef]
- Liu, T.; Liu, Z.; Zheng, Q.; Lang, Q.; Xia, Y.; Peng, N.; Gai, C. Effect of hydrothermal carbonization on migration and environmental risk of heavy metals in sewage sludge during pyrolysis. Bioresour. Technol. 2018, 247, 282–290. [Google Scholar] [CrossRef]
- Warnock, D.D.; Lehmann, J.; Kuyper, T.W.; Rillig, M.C. Mycorrhizal responses to biochar in soil—Concepts and mechanisms. Plant Soil 2007, 300, 9–20. [Google Scholar] [CrossRef]
- George, C.; Wagner, M.; Kücke, M.; Rillig, M.C. Divergent consequences of hydrochar in the plant—Soil system: Arbuscular mycorrhiza, nodulation, plant growth and soil aggregation effects. Appl. Soil Ecol. 2012, 59, 68–72. [Google Scholar] [CrossRef]
- Mohammadi, A.; Cowie, A.; Mai, T.L.A.; de la Rosa, R.A.; Brandão, M.; Kristiansen, P.; Joseph, S. Quantifying the Greenhouse Gas Reduction Benefits of Utilising Straw Biochar and Enriched Biochar. Energy Procedia 2016, 97, 254–261. [Google Scholar] [CrossRef]
- Cowie, A.; Woolf, D.; Gaunt, J.; Brandão, M.; Anaya de la Rosa, R.; Cowie, A. Biochar, carbon accounting and climate change. In Biochar for Environmental Management: Science, Technology, and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Milton Park, UK, 2015; pp. 763–794. [Google Scholar]
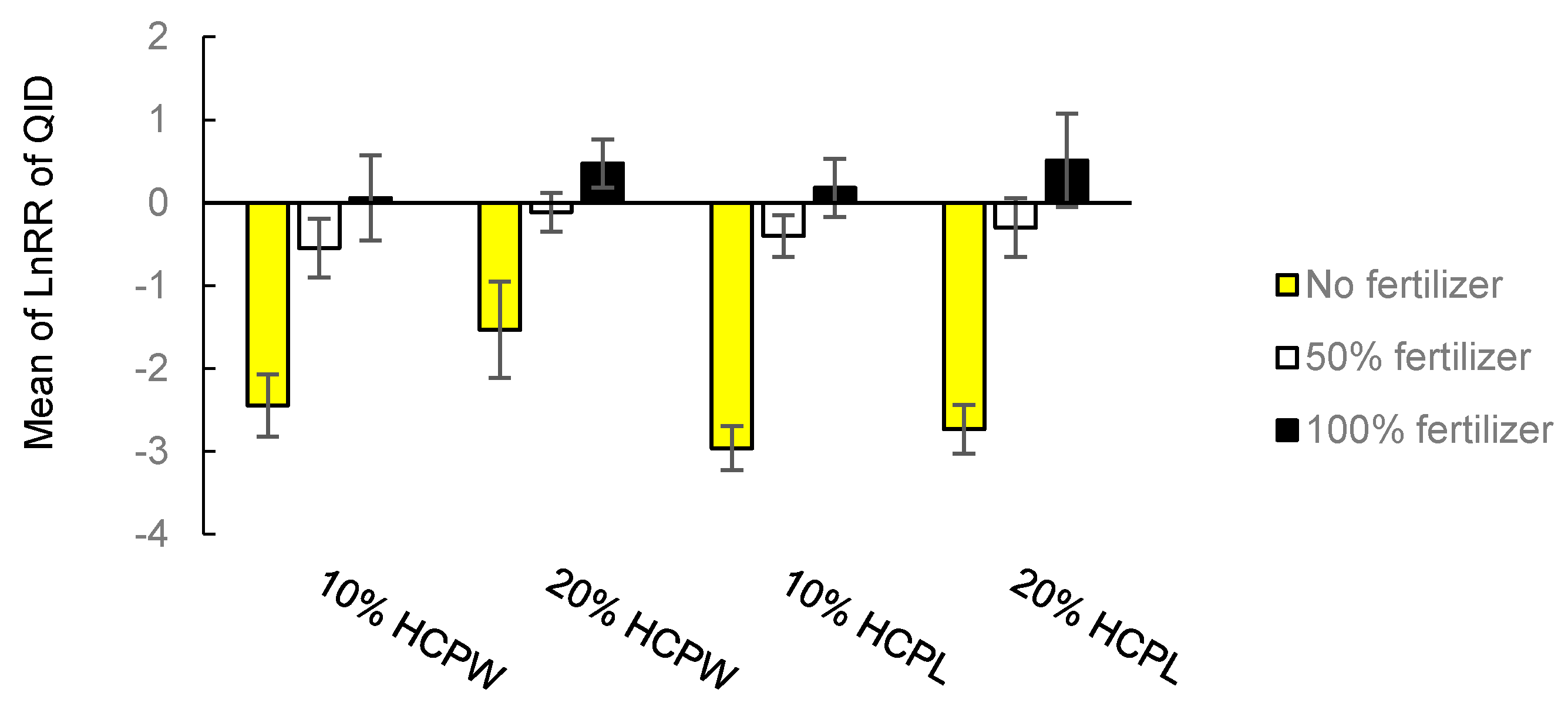
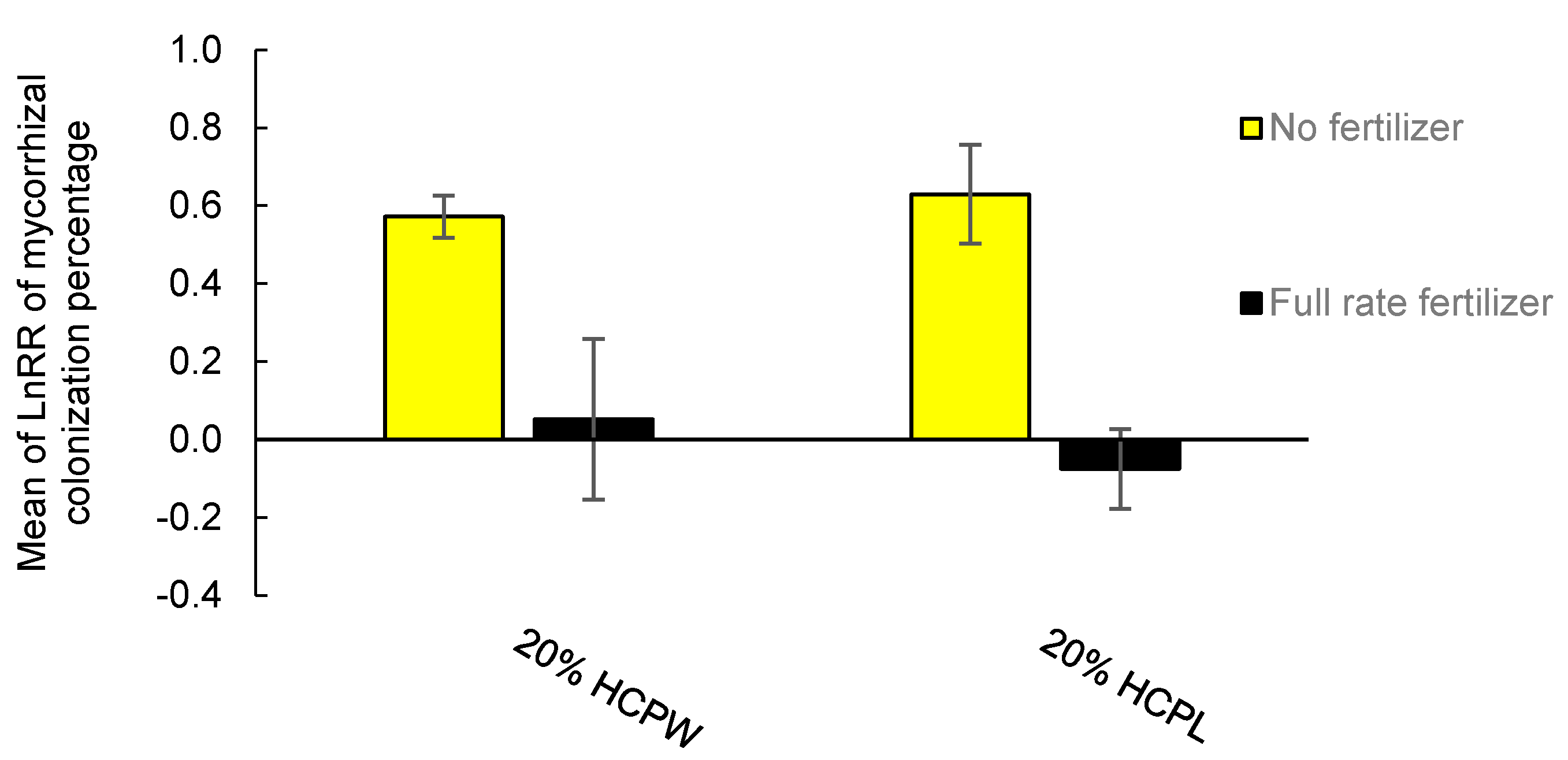
| Parameters | Peat | Hydrochar | Wood Ash |
|---|---|---|---|
| pH | 5.17 ± 0.17 | 5.59 ± 0.12 | 11.95 ± 0.00 |
| Total Carbon (%) | 48.82 ± 0.04 | 49.20 ± 1.51 | 3.16 ± 0.88 |
| Total N (%) | 1.04 ± 0.01 | 2.26 ± 0.02 | 0.05 ± 0.01 |
| P (g/kg) | 0.28 ± 0.00 | 2.77 ± 0.66 | 8.68 ± 0.08 |
| Ca (g/kg) | 7.92 ± 0.08 | 2.72 ± 0.04 | 216.66 ± 4.26 |
| K (g/kg) | 0.39 ± 0.00 | 1.34 ± 0.02 | 19.33 ± 0.12 |
| Mg (g/kg) | 2.39 ± 0.01 | 1.34 ± 0.01 | 9.55 ± 0.13 |
| Na (g/kg) | 0.23 ± 0.00 | 0.81 ± 0.01 | 6.93 ± 0.09 |
| Zn (g/kg) | 0.01 ± 0.00 | 0.43 ± 0.00 | 1.19 ± 0.00 |
| Cu (g/kg) | 0.07 ± 0.00 | 0.06 ± 0.00 | |
| Ni (g/kg) | 0.02 ± 0.00 | 0.01 ± 0.00 | |
| Pb (g/kg) | 0.02 ± 0.00 | 0.02 ± 0.00 | |
| Cd (g/kg) | 0.01 ± 0.00 | ||
| Cr (g/kg) | 0.04 ± 0.00 | 0.06 ± 0.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eskandari, S.; Mohammadi, A.; Sandberg, M.; Eckstein, R.L.; Hedberg, K.; Granström, K. Hydrochar-Amended Substrates for Production of Containerized Pine Tree Seedlings under Different Fertilization Regimes. Agronomy 2019, 9, 350. https://doi.org/10.3390/agronomy9070350
Eskandari S, Mohammadi A, Sandberg M, Eckstein RL, Hedberg K, Granström K. Hydrochar-Amended Substrates for Production of Containerized Pine Tree Seedlings under Different Fertilization Regimes. Agronomy. 2019; 9(7):350. https://doi.org/10.3390/agronomy9070350
Chicago/Turabian StyleEskandari, Samieh, Ali Mohammadi, Maria Sandberg, Rolf Lutz Eckstein, Kjell Hedberg, and Karin Granström. 2019. "Hydrochar-Amended Substrates for Production of Containerized Pine Tree Seedlings under Different Fertilization Regimes" Agronomy 9, no. 7: 350. https://doi.org/10.3390/agronomy9070350
APA StyleEskandari, S., Mohammadi, A., Sandberg, M., Eckstein, R. L., Hedberg, K., & Granström, K. (2019). Hydrochar-Amended Substrates for Production of Containerized Pine Tree Seedlings under Different Fertilization Regimes. Agronomy, 9(7), 350. https://doi.org/10.3390/agronomy9070350







