Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Determination of Brix and pH from Juices
2.3. Preparation of Crude Extracts
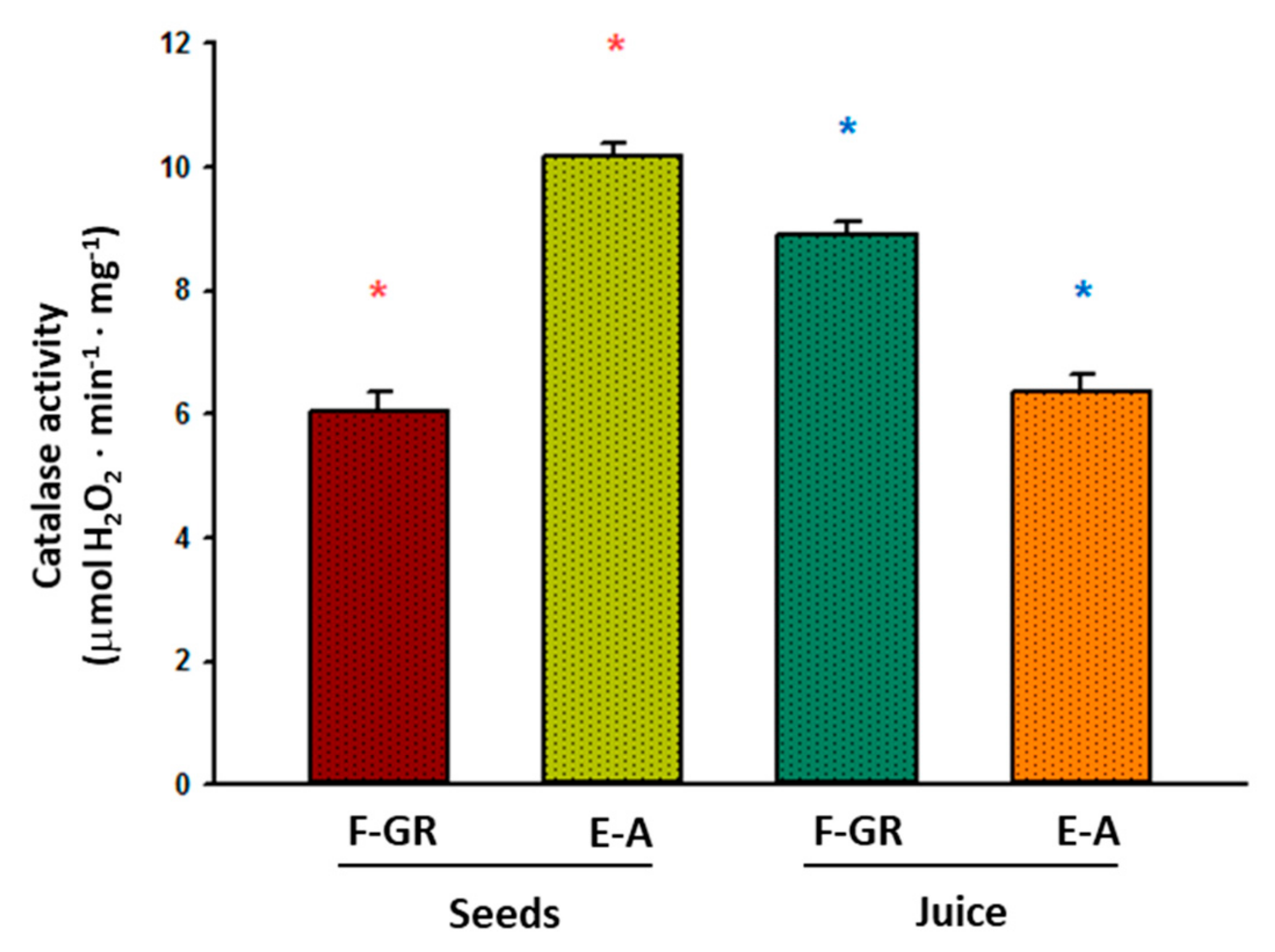
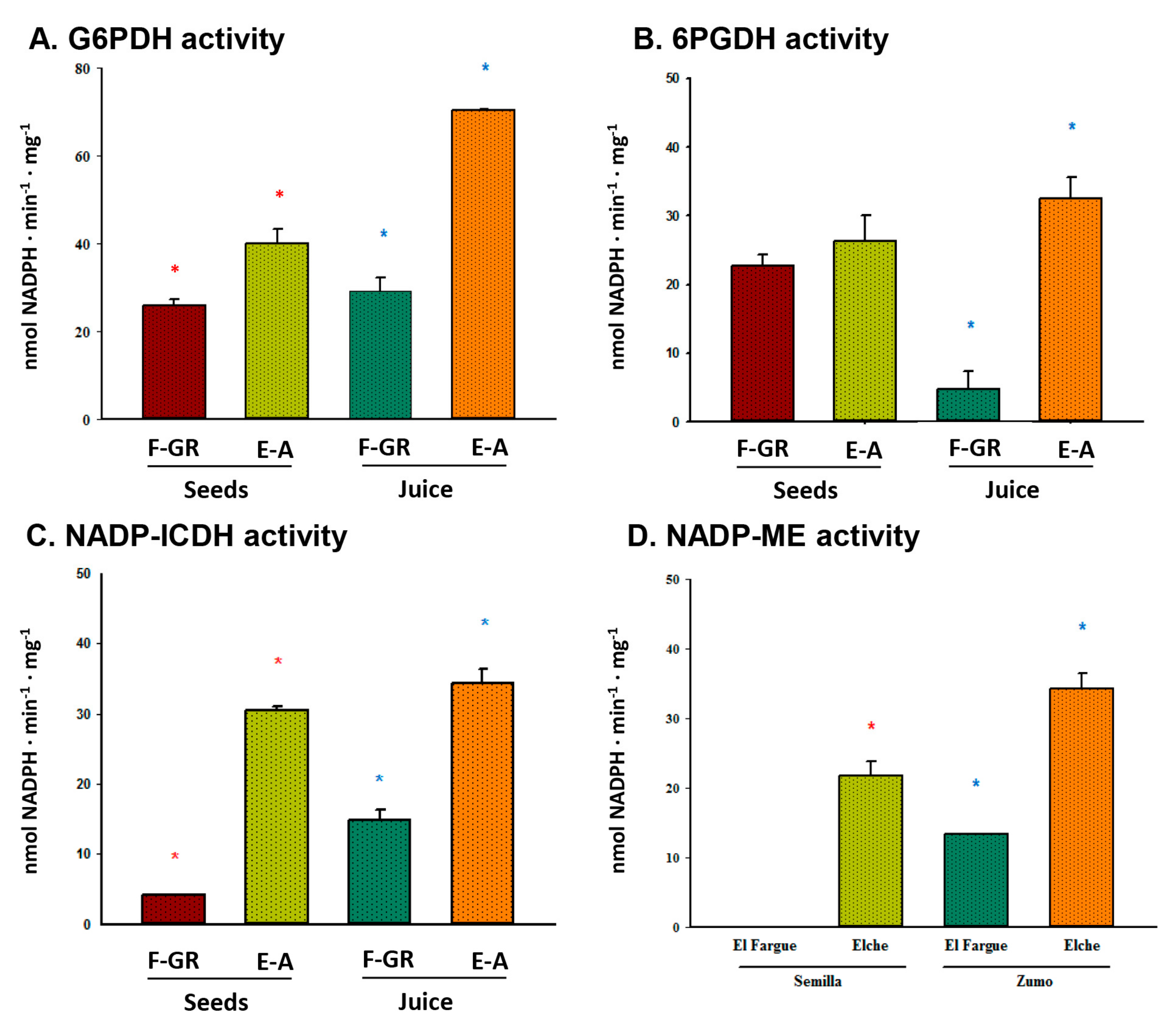
2.4. Enzyme Activity Assays
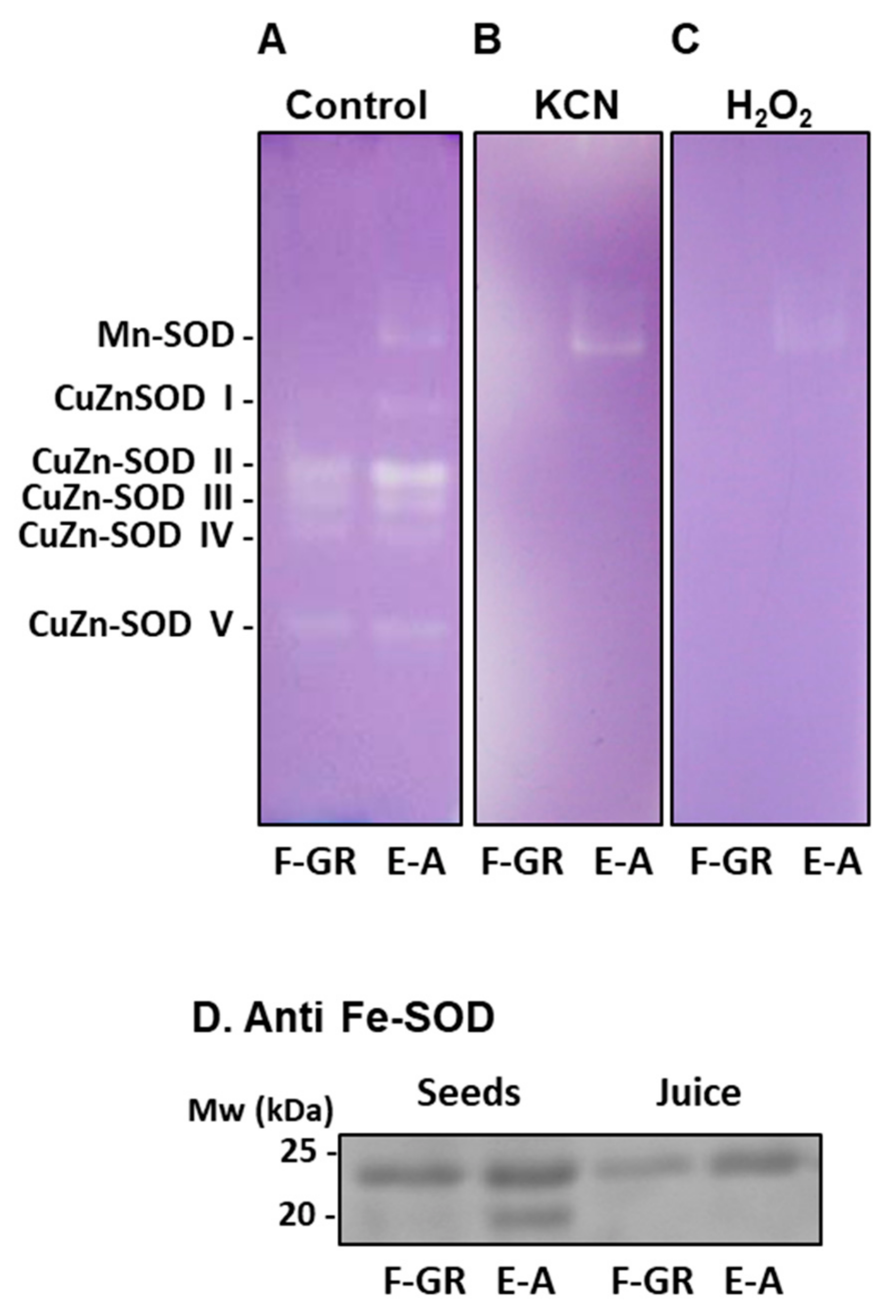
2.5. Non-Denaturing Electrophoresis and SOD Detection in Gels
2.6. SDS-PAGE and Western Blotting
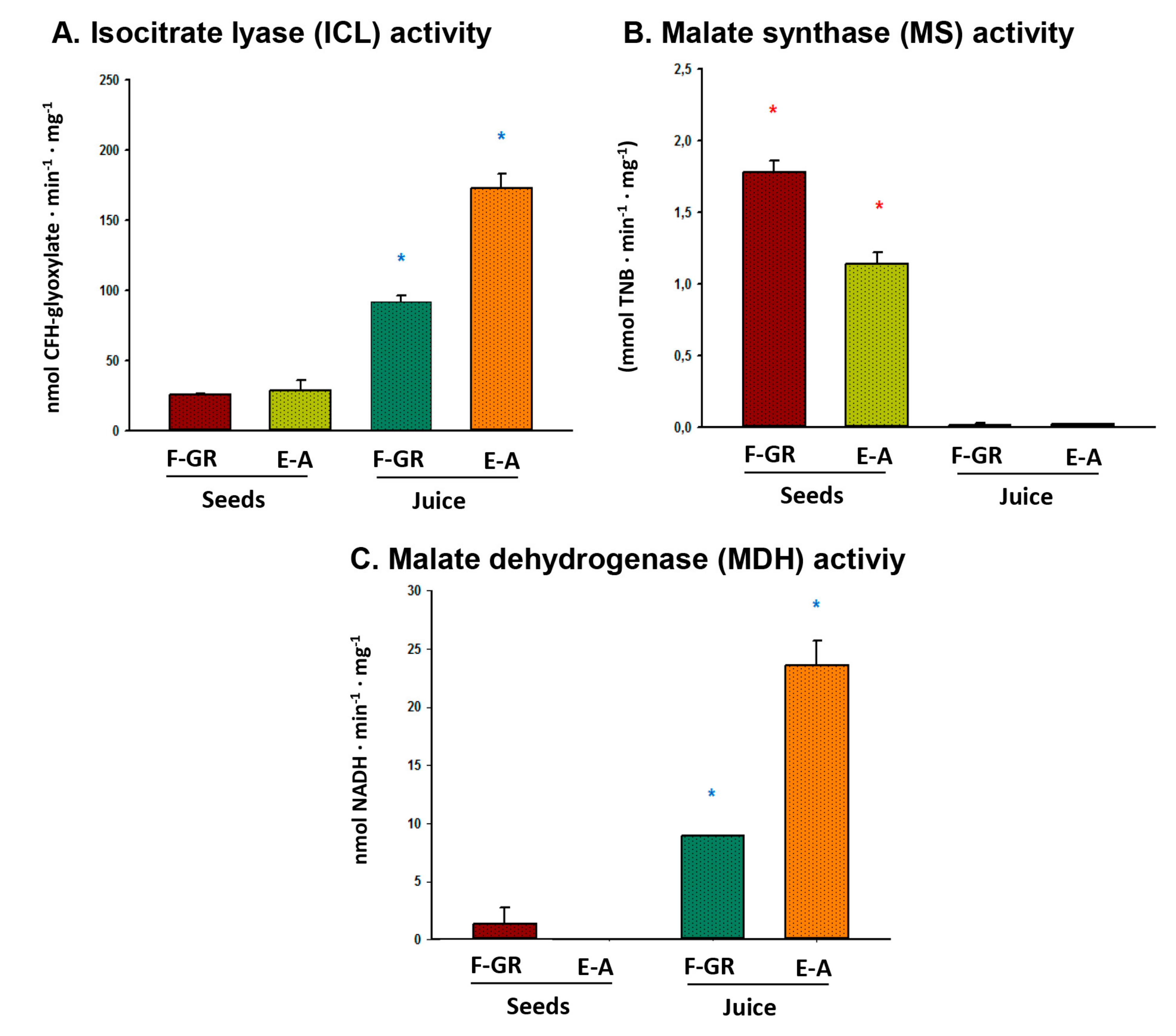
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Karimi, M.; Sadeghi, R.; Kokini, J. Pomegranate as a promising opportunity in medicine and nanotechnology. Trends Food Sci. Technol. 2017, 69, 59–73. [Google Scholar] [CrossRef]
- Wu, S.; Tian, L. Diverse phytochemicals and bioactivities in the ancient fruit and modern functional food pomegranate. Molecules 2017, 22, 1606. [Google Scholar] [CrossRef] [PubMed]
- Pareek, S.; Valero, D.; Serrano, M. Postharvest biology and technologies of pomegranate. J. Sci. Food Agric. 2017, 95, 2360–2379. [Google Scholar] [CrossRef] [PubMed]
- Alcaraz-Mármol, F.; Nuncio-Jáuregui, N.; García-Sánchez, F.; Martínez-Nicolás, J.J.; Hernández, F. Characterization of twenty pomegranate (Punica granatum L.) cultivars grown in Spain: Aptitudes for fresh consumption and processing. Sci. Hortic. 2017, 219, 152–160. [Google Scholar] [CrossRef]
- Holland, D.; Hatib, K.; Bar-Ya’akov, I. Pomegranate: Botany, horticulture, breeding. Hortic. Rev. 2009, 35, 127–191. [Google Scholar]
- Ahmed, M.S.; Ahmed Gardezi, M.S.; Awa, S.I. Germplasm Resources and Biodiversity of Pomegranate: Estimation of Heritable Components and Morphological Diversity of Different Ecotypes of Wild Pomegranate; VDM Verlag: Saarbrucken, Germany, 2010. [Google Scholar]
- Bar-Ya’akov, I.; Tian, L.; Amir, R.; Holland, D. Primary metabolites, anthocyanins, and hydrolyzable tannins in the pomegranate fruit. Front. Plant Sci. 2019, 10, 620. [Google Scholar] [CrossRef] [PubMed]
- Nuncio-Jáuregui, N.; Calín-Sánchez, A.; Carbonell-Barrachina, A.A.; Hernández, F. Changes in quality parameters, proline, antioxidant activity and color of pomegranate (Punica granatum L.) as affected by fruit position within tree, cultivar and ripening stage. Sci. Hortic. 2014, 165, 181–189. [Google Scholar] [CrossRef]
- Alcaraz-Mármol, F.; Nuncio-Jáuregui, N.; Calín-Sánchez, A.; Carbonell-Barrachina, A.A.; Martínez-Nicolás, J.J.; Hernández, F. Determination of fatty acid composition in arils of 20 pomegranatescultivars grown in Spain. Sci. Hortic. 2015, 197, 712–718. [Google Scholar] [CrossRef]
- Nuncio-Jáuregui, N.; Munera-Picazo, S.; Calín-Sánchez, A.; Wojdyło, A.; Hernández, F.; Carbonell-Barrachina, A.A. Bioactive compound composition of pomegranate fruits removed during thinning. J. Food Compos. Anal. 2015, 37, 11–19. [Google Scholar] [CrossRef]
- Matthaeus, B.; Ozcan, M.M. Pomegranate plant (Punica granatum L.) composition, antioxidant activity, therapeutic effect, antimicrobial activity–A review. Z. Arznei Gewurzpflanzen 2016, 4, 160–167. [Google Scholar]
- AlMatar, M.; Islam, M.R.; Albarri, O.; Var, I.; Kokal, F. Pomegranate as a possible treatment in reducing risk of developing wound healing, obesity, neurodegenerative disorders, and Diabetes Mellitus. Mini Rev. Med. Chem. 2018, 18, 507–526. [Google Scholar] [CrossRef]
- Khwairakpam, A.D.; Bordoloi, D.; Thakur, K.K.; Monisha, J.; Arfuso, F.; Sethi, G.; Mishra, S.; Kumar, A.P.; Kunnumakkara, A.B. Possible use of Punica granatum (Pomegranate) in cancer therapy. Pharmacol. Res. 2018, 133, 53–64. [Google Scholar] [CrossRef]
- Ceci, C.; Lacal, P.M.; Tentori, L.; De Martino, M.G.; Miano, R.; Graziani, G. Experimental evidence of the antitumor, antimetastatic and antiangiogenic activity of ellagic acid. Nutrients 2018, 10, 1756. [Google Scholar] [CrossRef] [PubMed]
- Khajebishak, Y.; Payahoo, L.; Alivand, M.; Alipour, B. Punicic acid: A potential compound of pomegranate seed oil in Type 2 Diabetes mellitus management. J. Cell. Physiol. 2019, 234, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Proestos, C. Superfoods: Recent data on their role in the prevention of diseases. Curr. Res. Nutr. Food Sci. 2018, 6, 576–593. [Google Scholar] [CrossRef]
- Wang, D.D.; Ozen, C.; Abu-Reidah, I.M.; Chigurupati, S.; Patra, J.K.; Horbanczuk, J.O.; Jozwik, A.; Tzvetkov, N.T.; Uhrin, P.; Atanasov, A.G. Vasculoprotective effects of pomegranate (Punica granatum L.). Front. Pharmacol. 2018, 9, 544. [Google Scholar] [CrossRef] [PubMed]
- Shaygannia, E.; Bahmani, M.; Zamanzad, B.; Rafieian-Kopaei, M. A Review Study on Punica granatum L. J. Evid. Based Complement. Altern. Med. 2016, 21, 221–227. [Google Scholar] [CrossRef]
- Bekir, J.; Bouajila, J.; Mars, M. The effect of cultivar and ripening on antioxidant system and PAL activity of pomegranate (Punica granatum L.) grown in Tunisia. J. Food Biochem. 2015, 39, 575–584. [Google Scholar] [CrossRef]
- Jannatizadeh, A. Exogenous melatonin applying confers chilling tolerance in pomegranate fruit during cold storage. Sci. Hortic. 2019, 246, 544–549. [Google Scholar] [CrossRef]
- Babalar, M.; Pirzad, F.; Sarcheshmeh, M.A.A.; Talaei, A.; Lessani, H. Arginine treatment attenuates chilling injury of pomegranate fruit during cold storage by enhancing antioxidant system activity. Postharvest Biol. Technol. 2018, 137, 31–37. [Google Scholar] [CrossRef]
- Calzone, A.; Podda, A.; Lorenzini, G.; Maserti, B.E.; Carrari, E.; Deleanu, E.; Hoshika, Y.; Haworth, M.; Nali, C.; Badea, O.; et al. Cross-talk between physiological and biochemical adjustments by Punica granatum cv. Dente di cavallo mitigates the effects of salinity and ozone stress. Sci. Total Environ. 2019, 656, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Ebtedaie, M.; Shekafandeh, A. Antioxidant and carbohydrate changes of two pomegranate cultivars under deficit irrigation stress. Span. J. Agric. Res. 2016, 14, e0809. [Google Scholar] [CrossRef]
- Dokhanieh, A.Y.; Aghdam, M.S.; Sarcheshmeh, M.A.A. Impact of postharvest hot salicylic acid treatment on aril browning and nutritional quality in fresh-cut pomegranate. Hortic. Environ. Biotechnol. 2016, 57, 378–384. [Google Scholar] [CrossRef]
- Daniel, J.; Kliebenstein, R.-A.M.; Robert, L. Superoxide dismutase in Arabidopsis: An eclectic enzyme family with disparate regulation and protein localization. Plant Physiol. 1998, 118, 637–650. [Google Scholar]
- Shigeoka, S.; Ishikawa, T.; Tamoi, M.; Miyagawa, Y.; Takeda, T.; Yabuta, Y.; Yoshimura, K. Regulation and function of ascorbate peroxidase isoenzymes. J. Exp. Bot. 2012, 53, 1305–1319. [Google Scholar] [CrossRef]
- Caverzan, A.; Passaia, G.; Barcellos Rosa, S.; Werner Ribeiro, C.; Lazzarotto, F.; Margis-Pinheiro, M. Plant responses to stresses: Role of ascorbate peroxidase in the antioxidant protection. Genet. Mol. Biol. 2012, 35, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Mhamdi, A.; Queval, G.; Chaouch, S.; Vanderauwera, S.; Van Breusegem, F. Catalase function in plants: A focus on Arabidopsis mutants as stress-mimic models. J. Exp. Bot. 2010, 61, 4198–4220. [Google Scholar] [CrossRef]
- Mhamdi, A.; Noctor, G.; Baker, A. Plant catalases: Peroxisomal redox guardians. Arch. Biochem. Biophys. 2012, 525, 181–194. [Google Scholar] [CrossRef]
- Yang, H.X.; Li, M.L.; Qi, X.; Lv, C.Y.; Deng, J.J.; Zhao, G.H. Identification of seven water-soluble non-storage proteins from pomegranate (Punica granatum L.) seeds. Food Sci. Technol. Int. 2012, 18, 329–338. [Google Scholar] [CrossRef]
- Cao, S.Y.; Niu, J.; Cao, D.; Li, H.X.; Xue, H.; Chen, L.N.; Zhang, F.H.; Zhao, D.G. Comparative proteomics analysis of pomegranate seeds on fruit maturation period (Punica granatum L.). J. Integr. Agric. 2015, 14, 2558–2564. [Google Scholar] [CrossRef]
- Niu, J.; Cao, D.; Li, H.X.; Xue, H.; Chen, L.; Liu, B.; Cao, S.Y. Quantitative proteomics of pomegranate varieties with contrasting seed hardness during seed development stages. Tree Genet. Genom. 2018, 14, 14. [Google Scholar] [CrossRef]
- Luo, X.; Cao, D.; Li, H.X.; Zhao, D.G.; Xue, H.; Niu, J.; Chen, L.; Zhang, F.H.; Cao, S.Y. Complementary iTRAQ-based proteomic and RNA sequencing-based transcriptomic analyses reveal a complex network regulating pomegranate (Punica granatum L.) fruit peel color. Sci. Rep. 2018, 8, 12362. [Google Scholar] [CrossRef] [PubMed]
- Rouholamin, S.; Zahedi, B.; Nazarian-Firouzabadi, F.; Saei, A. Expression analysis of anthocyanin biosynthesis key regulatory genes involved in pomegranate (Punica granatum L.). Sci. Hortic. 2015, 186, 84–88. [Google Scholar] [CrossRef]
- Harel-Beja, R.; Tian, L.; Freilich, S.; Habashi, R.; Borochov-Neori, H.; Lahav, T.; Trainin, T.; Doron-Faigenboim, A.; Ophir, R.; Bar-Ya’akov, I.; et al. Gene expression and metabolite profiling analyses of developing pomegranate fruit peel reveal interactions between anthocyanin and punicalagin production. Tree Genet. Genom. 2019, 15, 22. [Google Scholar] [CrossRef]
- Muñoz-Vargas, M.A.; González-Gordo, S.; Cañas, A.; López-Jaramillo, J.; Palma, J.M.; Corpas, F.J. Endogenous hydrogen sulfide (H2S) is up-regulated during sweet pepper (Capsicum annuum L.) fruit ripening. In vitro analysis shows that NADP-dependent isocitrate dehydrogenase (ICDH) activity is inhibited by H2S and NO. Nitric Oxide 2018, 81, 36–45. [Google Scholar]
- Corpas, F.J.; Barroso, J.B. NADPH-generating dehydrogenases: Their role in the mechanism of protection against nitro-oxidative stress induced by adverse environmental conditions. Front. Environ. Sci. 2014, 2, 55. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Hossain, M.A.; Asada, K. Inactivation of ascorbate peroxidase in spinach chloroplasts on dark addition of hydrogen peroxide: Its protection by ascorbate. Plant Cell Physiol. 1984, 25, 1285–1295. [Google Scholar]
- Quessada, M.P.; Macheix, J.J. Caractérisation d’une peroxydase impliquée spécifiquement dans la lignification, en relation avec l’incompatibilité au greffage chez l’Abricotier. Physiol. Veg. 1984, 22, 533–540. [Google Scholar]
- Barroso, J.B.; Peragón, J.; Contreras-Jurado, C.; García-Salguero, L.; Corpas, F.J.; Esteban, F.J.; Peinado, M.A.; de la Higuera, M.; Lupiáñez, J.A. Impact of starvation-refeeding on kinetics and protein expression of trout liver NADPH-production systems. Am. J. Physiol. 1998, 274, R1578–R1587. [Google Scholar] [CrossRef]
- Leterrier, M.; del Río, L.A.; Corpas, F.J. Cytosolic NADP-isocitrate dehydrogenase of pea plants: Genomic clone characterization and functional analysis under abiotic stress conditions. Free Radic. Res. 2007, 41, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Mateos, R.M.; Bonilla-Valverde, D.; del Río, L.A.; Palma, J.M.; Corpas, F.J. NADP-dehydrogenases from pepper fruits: Effect of maturation. Physiol. Plant. 2009, 135, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Archer, E.K.; Ting, B.L. A virescent plastid mutation in tobacco decreases peroxisome enzyme activities in seedlings. J. Plant Physiol. 1996, 149, 520–526. [Google Scholar] [CrossRef]
- Mateos, R.M.; León, A.M.; Sandalio, L.M.; Gómez, M.; del Río, L.A.; Palma, J.M. Peroxisomes from pepper fruits (Capsicum annuum L.): Purification, characterisation and antioxidant activity. J. Plant Physiol. 2003, 160, 1507–1516. [Google Scholar] [CrossRef] [PubMed]
- Hock, B.; Beevers, H. Development and decline of glyoxylate-cycle enzymes in watermelon seedlings (Citrullus vulgaris Schrad)-effects of dactinomycin and cycloheximide. Z. Pflanzenphysiol. 1966, 55, 405–414. [Google Scholar]
- Gerhardt, B. Localization of beta-oxidation enzymes in peroxisomes isolated from nonfatty plant tissues. Planta 1983, 159, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Heukeshoven, J.; Dernick, R. Simplified method for silver staining of proteins in polyacrylamide gels and the mechanism of silver staining. Electrophoresis 1985, 6, 103–112. [Google Scholar] [CrossRef]
- Corpas, F.J.; Barroso, J.B.; Sandalio, L.M.; Distefano, S.; Palma, J.M.; Lupiáñez, J.A.; del Río, L.A. A dehydrogenase-mediated recycling system of NADPH in plant peroxisomes. Biochem. J. 1998, 330, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Corpas, F.J.; Fernández-Ocaña, A.; Carreras, A.; Valderrama, R.; Luque, F.; Esteban, F.J.; Rodríguez-Serrano, M.; Chaki, M.; Pedrajas, J.R.; Sandalio, L.M.; et al. The expression of different superoxide dismutase forms is cell-type dependent in olive (Olea europaea L.) leaves. Plant Cell Physiol. 2006, 47, 984–994. [Google Scholar] [CrossRef] [PubMed]
- Mateos, R.M.; Jiménez, A.; Román, P.; Romojaro, F.M.; Bacarizo, S.; Leterrier, M.; Gómez, M.; Sevilla, F.; del Río, L.A.; Corpas, F.J.; et al. Antioxidant systems from pepper (Capsicum annuum L.): Involvement in the response to temperature changes in ripe fruits. Int. J. Mol. Sci. 2013, 14, 9556–9580. [Google Scholar] [CrossRef] [PubMed]
- Houmani, H.; Rodríguez-Ruiz, M.; Palma, J.M.; Abdelly, C.; Corpas, F.J. Modulation of superoxide dismutase (SOD) isozymes by organ development and high long-term salinity in the halophyte Cakile maritima. Protoplasma 2016, 253, 885–894. [Google Scholar] [CrossRef] [PubMed]
- Elfalleh, W.; Hannachi, H.; Guetat, A.; Tlili, N.; Guasmi, F.; Ferchichi, A.; Ying, M. Storage protein and amino acid contents of Tunisian and Chinese pomegranate (Punica granatum L.) cultivars. Genet. Res. Crop Evol. 2012, 59, 999–1014. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, T.; Masuda, T.; Lv, C.; Sun, L.; Qu, G.; Zhao, G. Chitinase III in pomegranate seeds (Punica granatum Linn.): A high-capacity calcium-binding protein in amyloplasts. Plant J. 2011, 68, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Capriotti, A.L.; Caruso, G.; Cavaliere, C.; Foglia, P.; Piovesana, S.; Samperi, R.; Laganà, A. Proteome investigation of the non-model plant pomegranate (Punica granatum L.). Anal. Bioanal. Chem. 2013, 405, 9301–9309. [Google Scholar] [CrossRef]
- Mertens-Talcott, S.U.; Jilma-Stohlawetz, P.; Rios, J.; Hingorani, L.; Derendorf, H. Absorption, Metabolism, and antioxidant effects of pomegranate (Punica granatum L.) polyphenols after ingestion of a standardized extract in healthy human volunteers. J. Agric. Food Chem. 2006, 54, 8956–8961. [Google Scholar] [CrossRef]
- Opara, L.U.; Al-Ani, M.R.; Al-Shuaibi, Y.S. Physico-chemical properties, vitamin C content, and antimicrobial properties of pomegranate fruit (Punica granatum L.). Food Bioprocess Technol. 2009, 2, 315–321. [Google Scholar] [CrossRef]
- Venkata, C.; Prakash, S.; Prakash, I. Bioactive chemical constituents from pomegranate (Punica granatum) juice, seed and peel–A review. Int. J. Res. Chem. Environ. 2011, 1, 1–18. [Google Scholar]
- Meighani, H.; Ghasemnezhad, M.; Bakhshi, D. An evaluation of the phytochemical properties of some pomegranate cultivars during fruit development and ripening. Int. J. Hortic. Sci. Technol. 2017, 4, 193–204. [Google Scholar]
- Palma, J.M.; Gupta, D.K.; Corpas, F.J. Metalloenzymes involved in the metabolism of reactive oxygen species and heavy metal stress. In Heavy Metal Stress in Plants; Prasad, M.N.V., Ed.; Springer: Berlin, Germany, 2013; pp. 1–17. [Google Scholar]
- Palma, J.M.; Sevilla, F.; Jiménez, A.; del Río, L.A.; Corpas, F.J.; Álvarez de Morales, P.; Camejo, D.M. Physiology of pepper fruits and the metabolism of antioxidants: Chloroplasts, mitochondria and peroxisomes. Ann. Bot. 2015, 116, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Corpas, F.J.; Barroso, J.B.; Palma, J.M.; Rodríguez-Ruiz, M. Plant peroxisomes: A nitro-oxidative cocktail. Redox Biol. 2017, 11, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Del Río, L.A.; Pastori, G.M.; Palma, J.M.; Sandalio, L.M.; Sevilla, F.; Corpas, F.J.; Jiménez, A.; López-Huertas, E.; Hernández, J.A. The activated oxygen role of peroxisomes in senescence. Plant Physiol. 1998, 116, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Toriyama, K.; Kondo, M.; Kato, A.; Mano, S.; De Bellis, L.; Hayashi-Ishimaru, Y.; Yamaguchi, K.; Nishimura, M. Functional transformation of plant peroxisomes. Cell Biochem. Biophys. 2000, 32, 295–304. [Google Scholar] [CrossRef]
- Baker, A.; Graham, I. Plant Peroxisomes. Biochemistry, Cell Biology and Biotechnological Applications; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Palma, J.M.; Ruiz, C.; Corpas, F.J. A simple and useful method to apply exogenous no gas to plant systems: Bell pepper fruits as a model. Methods Mol. Biol. Nitric Oxide Methods Prot. 2018, 1747, 3–11. [Google Scholar]
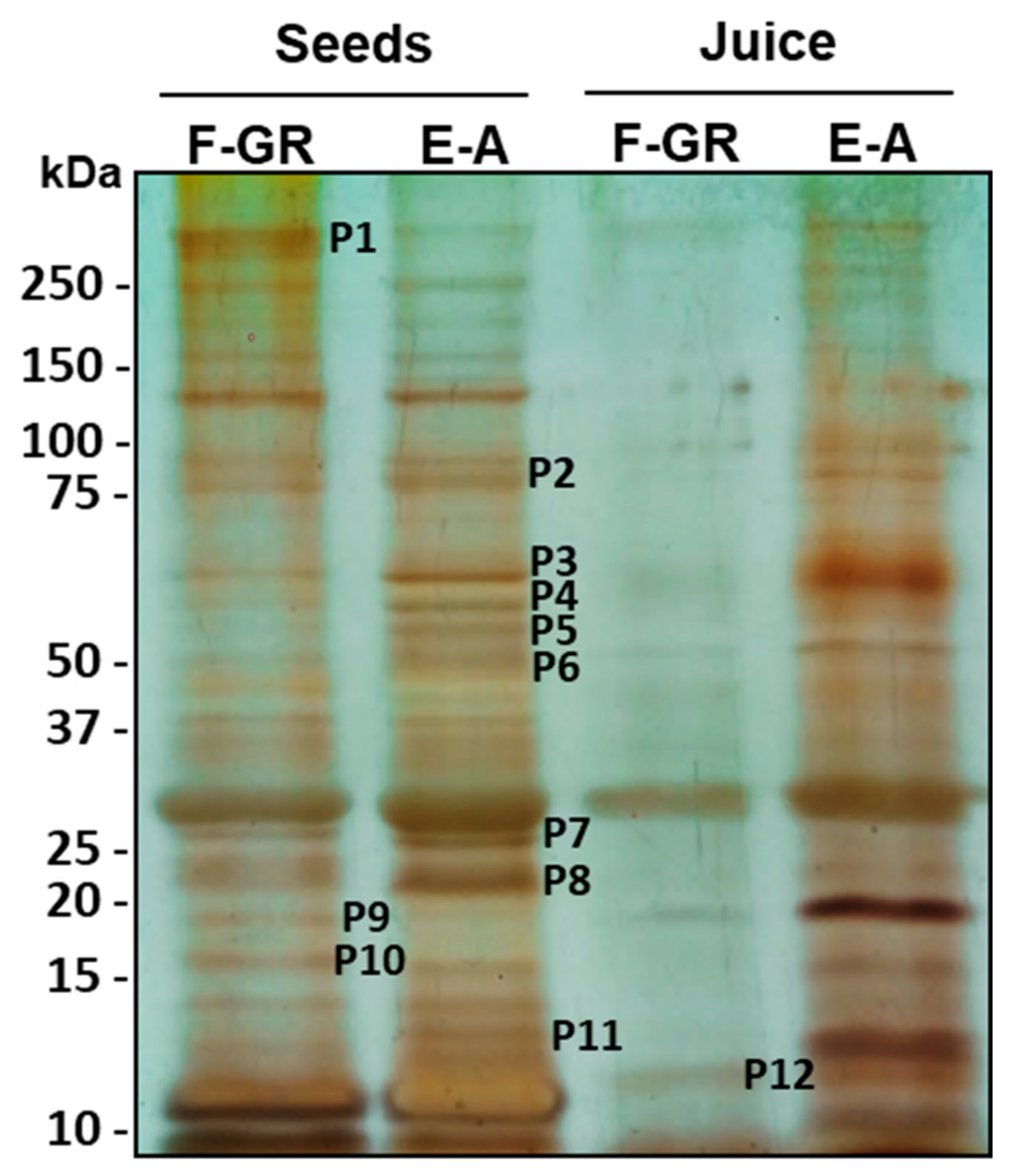
| Polypeptide (Molecular Weight) | F-GR | E-A |
|---|---|---|
| P1 (> 250 kDa) | √ | |
| P2 (87) | √ | |
| P3 (67) | √ | |
| P4 (64) | √ | |
| P5 (60) | √ | |
| P6 (50) | √ | |
| P7 (25) | √ | |
| P8 (22) | √ | |
| P9 (18) | √ | |
| P10 (16) | √ | |
| P11 (13) | √ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinilla, M.; Iglesias-Moya, J.; Jesús Campos, M.; Corpas, F.J.; Palma, J.M. Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System. Agronomy 2019, 9, 338. https://doi.org/10.3390/agronomy9060338
Pinilla M, Iglesias-Moya J, Jesús Campos M, Corpas FJ, Palma JM. Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System. Agronomy. 2019; 9(6):338. https://doi.org/10.3390/agronomy9060338
Chicago/Turabian StylePinilla, Melisa, Jessica Iglesias-Moya, María Jesús Campos, Francisco J. Corpas, and José M. Palma. 2019. "Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System" Agronomy 9, no. 6: 338. https://doi.org/10.3390/agronomy9060338
APA StylePinilla, M., Iglesias-Moya, J., Jesús Campos, M., Corpas, F. J., & Palma, J. M. (2019). Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System. Agronomy, 9(6), 338. https://doi.org/10.3390/agronomy9060338








