Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability
Abstract
1. Introduction
2. Materials and Methods
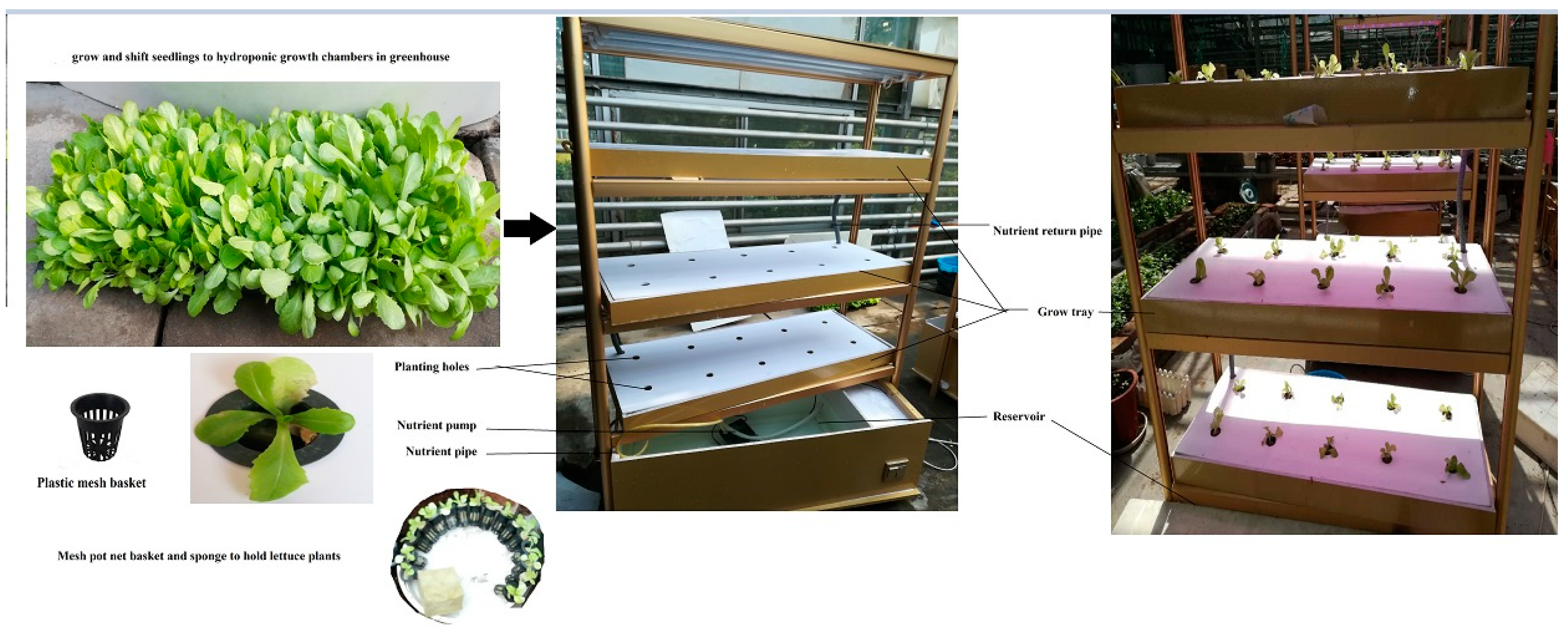
2.1. Plant Material and Growth Conditions
2.2. Application of Three Amino Acids on Lettuce
2.3. Application of L-Methionine Concentrations on Lettuce
2.4. Data Collection and Analysis
2.4.1. Vegetative Growth Parameters
2.4.2. Physiological Measurements
2.5. Statistical Analysis
3. Results
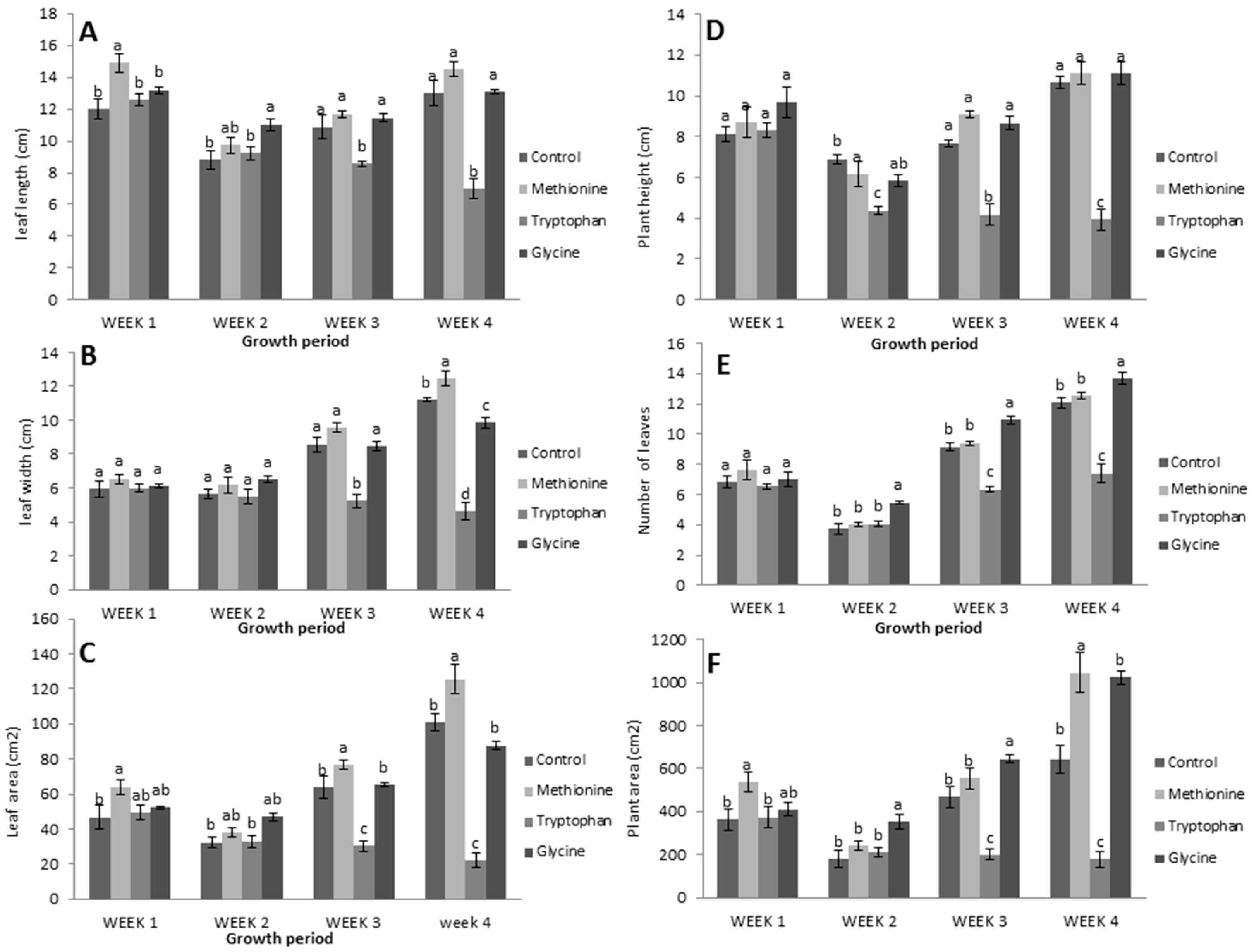
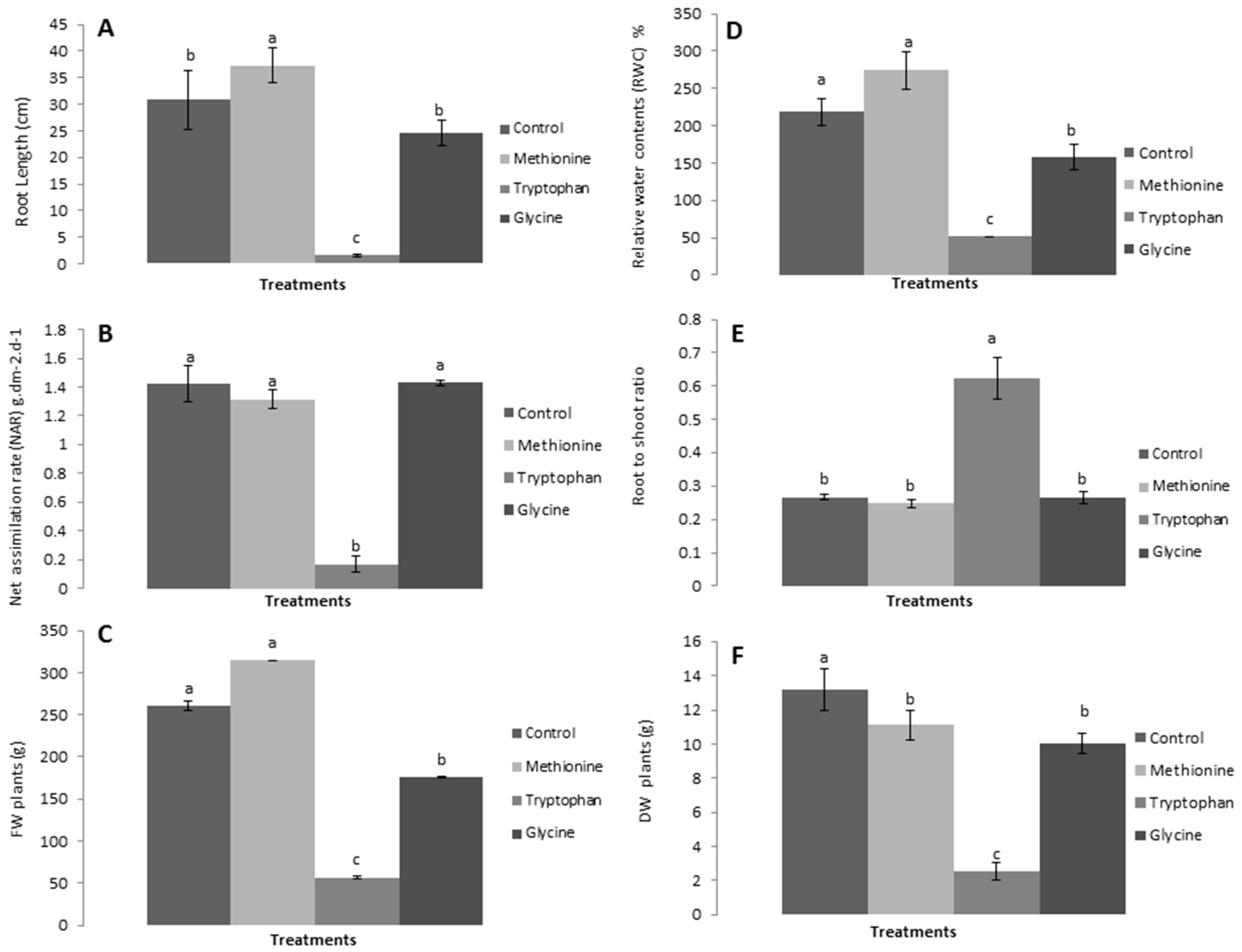
3.1. Application of Three Amino Acids on Lettuce
3.1.1. Photosynthetic Measurements
3.1.2. Nutrient Contents
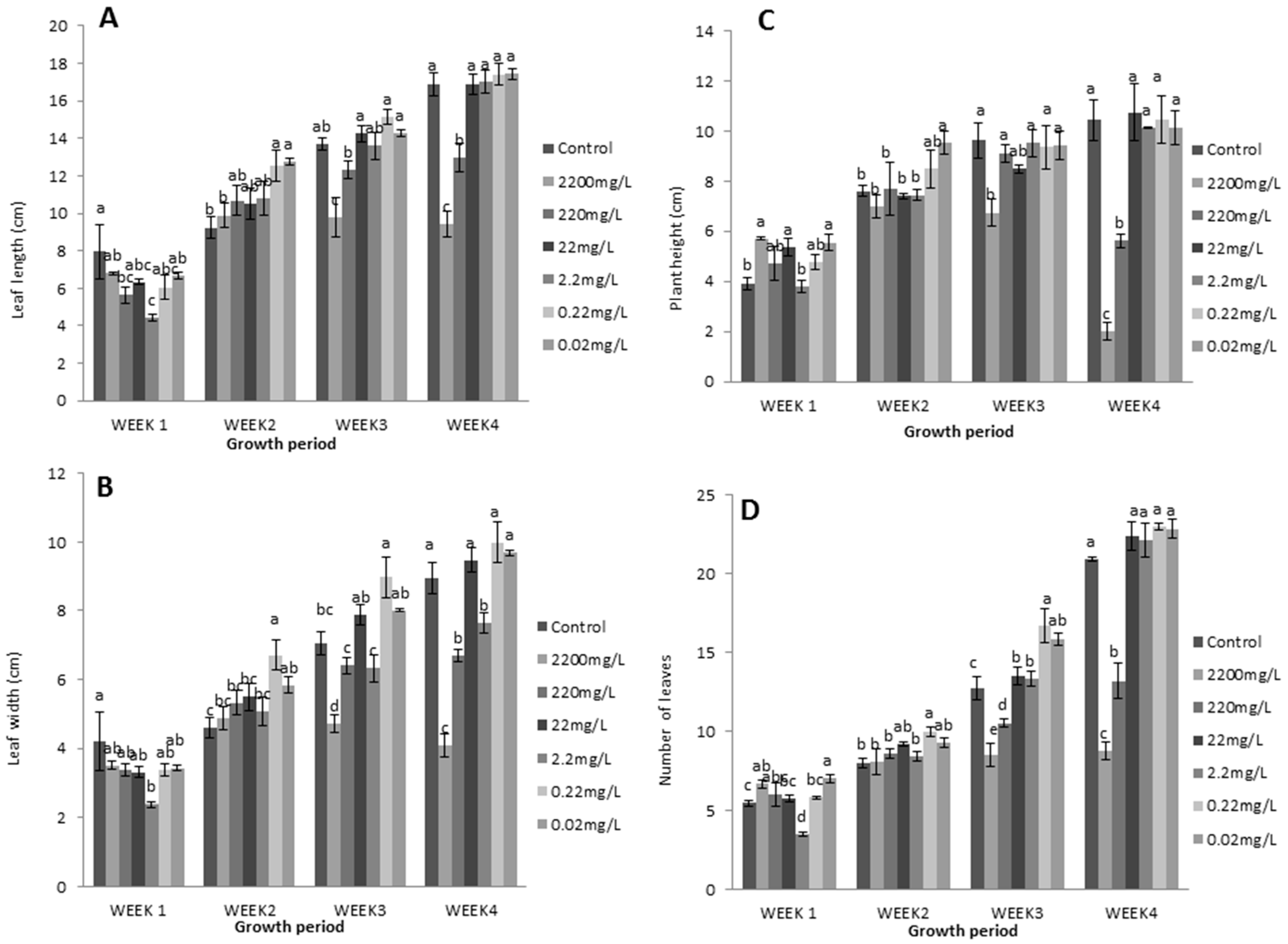
3.2. Application of Different L-Methionine Concentrations on Lettuce
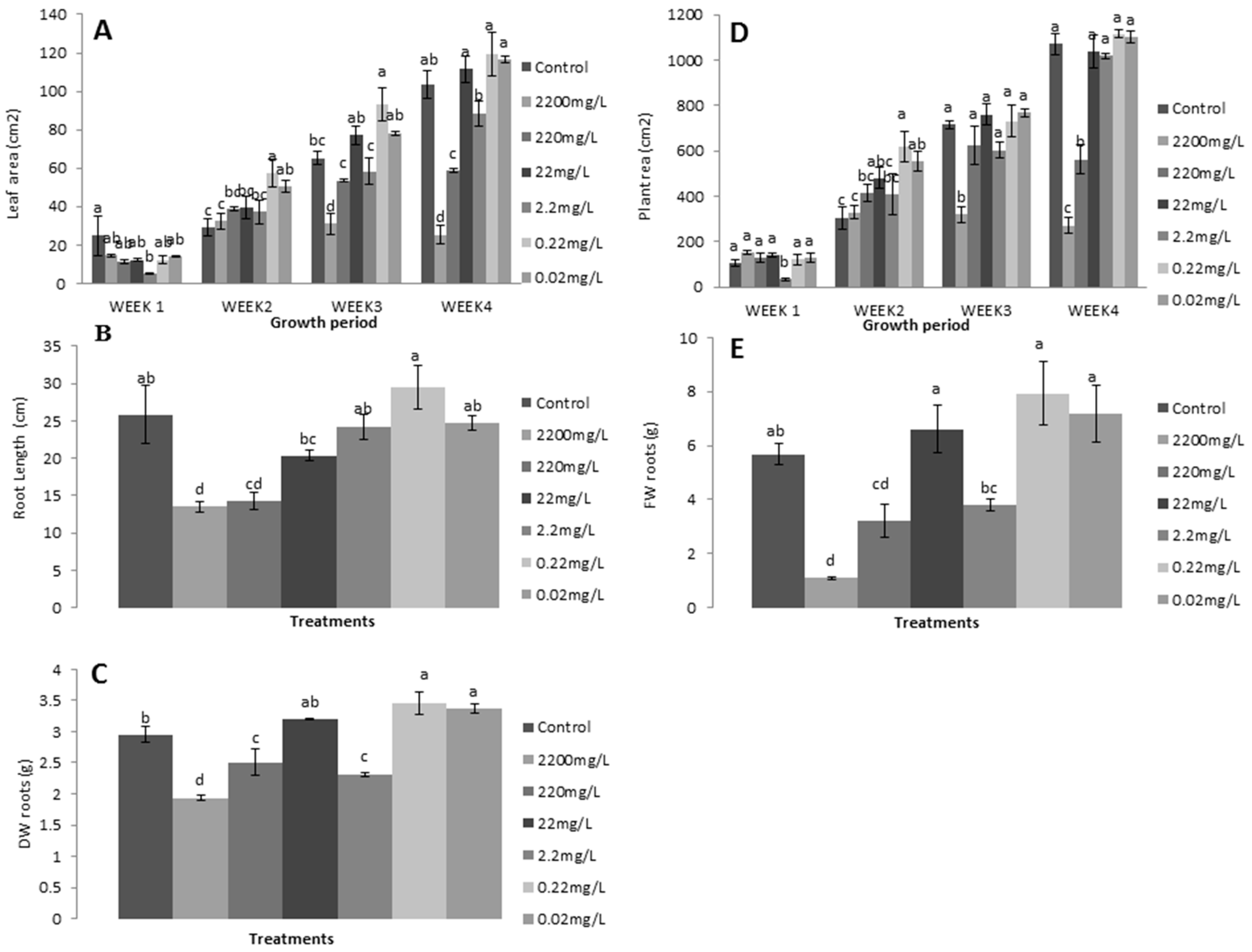
3.2.1. Relative Growth Measurements
3.2.2. Photosynthetic Measurements
3.2.3. Nutrient Contents
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Samuolienė, G.; Urbonavičiūtė, A.; Duchovskis, P.; Bliznikas, Z.; Vitta, P.; Žukauskas, A. Decrease in nitrate concentration in leafy vegetables under a solid-state illuminator. HortScience 2009, 44, 1857–1860. [Google Scholar] [CrossRef]
- Son, K.H.; Oh, M.M. Leaf shape, growth, and antioxidant phenolic compounds of two lettuce cultivars grown under various combinations of blue and red light-emitting diodes. HortScience 2013, 48, 988–995. [Google Scholar]
- Christopoulou, M.; Wo, S.R.C.; Kozik, A.; McHale, L.K.; Truco, M.J.; Wroblewski, T.; Michelmore, R.W. Genome-wide architecture of disease resistance genes in lettuce. G3 Genes Genomes Genet. 2015, 5, 2655–2669. [Google Scholar] [CrossRef] [PubMed]
- Amoozgar, A.; Mohammadi, A.; Sabzalian, M. Impact of light-emitting diode irradiation on photosynthesis, phytochemical composition and mineral element content of lettuce cv. Grizzly. Photosynthetica 2017, 55, 85–95. [Google Scholar] [CrossRef]
- Nawaz, M.A.; Wang, L.; Jiao, Y.; Chen, C.; Zhao, L.; Mei, M.; Yu, Y.; Bie, Z.; Huang, Y. Pumpkin rootstock improves nitrogen use efficiency of watermelon scion by enhancing nutrient uptake, cytokinin content, and expression of nitrate reductase genes. Plant Growth Regul. 2017, 82, 233–246. [Google Scholar] [CrossRef]
- Nawaz, M.A.; Chen, C.; Shireen, F.; Zheng, Z.; Sohail, H.; Afzal, M.; Ali, M.A.; Bie, Z.; Huang, Y. Genome-wide expression profiling of leaves and roots of watermelon in response to low nitrogen. BMC Genom. 2018, 19, 456. [Google Scholar] [CrossRef] [PubMed]
- Scheible, W.-R.; Morcuende, R.; Czechowski, T.; Fritz, C.; Osuna, D.; Palacios-Rojas, N.; Schindelasch, D.; Thimm, O.; Udvardi, M.K.; Stitt, M. Genome-wide reprogramming of primary and secondary metabolism, protein synthesis, cellular growth processes, and the regulatory infrastructure of Arabidopsis in response to nitrogen. Plant Physiol. 2004, 136, 2483–2499. [Google Scholar] [CrossRef]
- Yuan, L.; Yuan, Y.; Du, J.; Sun, J.; Guo, S. Effects of 24-epibrassinolide on nitrogen metabolism in cucumber seedlings under Ca(NO3)2 stress. Plant Physiol. Biochem. 2012, 61, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Amalfitano, C.; Gomez, L.D.; Frendo, P.; De Pascale, S.; Pepe, O.; Simister, R.; Ventorino, V.; Agrelli, D.; Borrelli, C.; McQueen-Mason, S.J.; et al. Plant–Rhizobiumsymbiosis, seed nutraceuticals, and waste quality for energy production of Vicia faba L. as affected by crop management. Chem. Biol. Technol. Agric. 2018, 5, 15. [Google Scholar] [CrossRef]
- Jämtgård, S.; Näsholm, T.; Huss-Danell, K. Nitrogen compounds in soil solutions of agricultural land. Soil Biol. Biochem. 2010, 42, 2325–2330. [Google Scholar] [CrossRef]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Academic Press: New York, NY, USA, 2011. [Google Scholar]
- Caruso, G.; Conti, S.; La Rocca, G. Influence of crop cycle and nitrogen fertilizer form on yield and nitrate content in different species of vegetables. Adv. Hort. Sci. 2011, 25, 81–89. [Google Scholar]
- Silva, L.F.; Hower, J.C.; Izquierdo, M.; Querol, X. Complex nanominerals and ultrafine particles assemblages in phosphogypsum of the fertilizer industry and implications on human exposure. Sci. Total Environ. 2010, 408, 5117–5122. [Google Scholar] [CrossRef]
- Verger, P.J.; Boobis, A.R. Reevaluate pesticides for food security and safety. Science 2013, 341, 717–718. [Google Scholar] [CrossRef]
- du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Paschalidis, K.A.; Roubelakis-Angelakis, K.A. Spatial and temporal distribution of polyamine levels and polyamine anabolism in different organs/tissues of the tobacco plant. Correlations with age, cell division/expansion, and differentiation. Plant Physiol. 2005, 138, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Su, X.; Li, X.; Zhao, X.; Zang, L.; Pan, W. Development of prolonged release microspheres of metformin hydrochloride using ion exchange resins. J. Chin. Pharm. Sci. 2006, 15, 155. [Google Scholar]
- Pang, J.; Ross, J.; Zhou, M.; Mendham, N.; Shabala, S. Amelioration of detrimental effects of waterlogging by foliar nutrient sprays in barley. Funct. Plant Biol. 2007, 34, 221–227. [Google Scholar] [CrossRef]
- Paungfoo-Lonhienne, C.; Thierry, G.; Lonhienne, A.; Doris, R.; Nicole, R.; Michael, C.; Richard, I.W.; Gamage, H.K.; Bernard, J.C.; Peer, M.S.; et al. Plants can use protein as a N source without assistance from other organisms. Proc. Natl. Acad. Sci. USA 2008, 105, 4524–4529. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Colla, G. Synergistic Biostimulatory Action: Designing the Next Generation of Plant Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 1655. [Google Scholar] [CrossRef]
- Rouphael, Y.; Spíchal, L.; Panzarová, K.; Casa, R.; Colla, G. High-Throughput Plant Phenotyping for Developing Novel Biostimulants: From Lab to Field or From Field to Lab? Front. Plant Sci. 2018, 9, 1197. [Google Scholar] [CrossRef]
- Kowalczyk, K.; Zielony, T.; Gajewski, M. Effect of Aminoplant and Asahi on yield and quality of lettuce grown on rockwool. Biostimulators Mod. Agric. Veg. Crops 2008, 35–43. [Google Scholar]
- Zhao, Y. Auxin biosynthesis and its role in plant development. Annu. Rev. Plant Biol. 2010, 61, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Dudareva, N. The shikimate pathway and aromatic amino acid biosynthesis in plants. Annu. Rev. Plant Biol. 2012, 63, 73–105. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Nelson, L.; Kloepper, J. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Forde, B.G.; Roberts, M.R. Glutamate receptor-like channels in plants: A role as amino acid sensors in plant defence? F1000Prime Rep. 2014, 6, 37. [Google Scholar] [CrossRef]
- Miller, A.J.; Fan, X.; Shen, Q.; Smith, S.J. Amino acids and nitrate as signals for the regulation of nitrogen acquisition. J. Exp. Bot. 2007, 59, 111–119. [Google Scholar] [CrossRef]
- Walch-Liu, P.; Forde, B.G. L-Glutamate as a novel modifier of root growth and branching: What’s the sensor? Plant Signal. Behav. 2007, 2, 284–286. [Google Scholar] [CrossRef]
- Weiland, M.; Mancuso, S.; Baluska, F. Signalling via glutamate and GLRs in Arabidopsis thaliana. Funct. Plant Biol. 2016, 43, 1–25. [Google Scholar] [CrossRef]
- Halpern, M.; Bar-Tal, A.; Ofek, M.; Minz, D.; Muller, T.; Yermiyahu, U. The Use of Biostimulants for Enhancing Nutrient Uptake. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: New York, NY, USA, 2015; Volume 130, pp. 141–174. [Google Scholar]
- Teixeira, W.F.; Fagan, E.B.; Soares, L.H.; Umburanas, R.C.; Reichardt, K.; Neto, D.D. Foliar and seed application of amino acids affects the antioxidant metabolism of the soybean crop. Front. Plant Sci. 2017, 8, 327. [Google Scholar] [CrossRef]
- Ertani, A.; Pizzeghelio, D.; Altissimo, A.; Nardi, S. Use of meat hydrolyzate derived from tanning residues as plant biostimulant for hydroponically grown maize. J. Plant Nutr. Soil Sci. 2013, 176, 287–296. [Google Scholar] [CrossRef]
- Hildebrandt, T.M.; Nesi, A.N.; Araújo, W.L.; Braun, H.-P. Amino acid catabolism in plants. Mol. Plant 2015, 8, 1563–1579. [Google Scholar] [CrossRef] [PubMed]
- Persson, J.; Högberg, P.; Ekblad, A.; Högberg, M.N.; Nordgren, A.; Näsholm, T. Nitrogen acquisition from inorganic and organic sources by boreal forest plants in the field. Oecologia 2003, 137, 252–257. [Google Scholar] [CrossRef]
- Gioseffi, E.; de Neergaard, A.; Schjørring, J.K. Interactions between uptake of amino acids and inorganic nitrogen in wheat plants. Biogeosciences 2012, 9, 1509–1518. [Google Scholar] [CrossRef]
- Koukounaras, A.; Tsouvaltzis, P.; Siomos, A.S. Effect of root and foliar application of amino acids on the growth and yield of greenhouse tomato in different fertilization levels. J. Food Agric. Environ. 2013, 11, 644–648. [Google Scholar]
- El-Aal, M.A.; Eid, R.S. Effect of foliar spray with lithovit and amino acids on growth, bioconstituents, anatomical and yield features of soybean plant. Plant Biotechnol. 2018, 187–201. [Google Scholar]
- SH SADAK, M.; Abdelhamid, M.T.; Schmidhalter, U. Effect of foliar application of aminoacids on plant yield and some physiological parameters in bean plants irrigated with seawater. Acta Biol. Colomb. 2014, 20, 141–152. [Google Scholar]
- El-Awadi, M.; El-Bassiony, A.; Fawzy, Z.; El-Nemr, M. Response of snap bean (Phaseolus vulgaris L) plants to nitrogen fertilizer and foliar application with methionine and tryptophan. Nat. Sci. 2011, 9, 87–94. [Google Scholar]
- Yong, J.W.H.; Letham, S.D.; Wong, C.S.; Graham, D.F. Rhizobium-induced elevation in xylem cytokinin delivery in pigeon pea induces changes in shoot development and leaf physiology. Funct. Plant Biol. 2014, 41, 1323–1335. [Google Scholar] [CrossRef]
- Paciorek, T.; Zazímalová, E.; Ruthardt, N.; Petrásek, J.; Stierhof, Y.D.; Kleine-Vehn, J.; Morris, D.A.; Emans, N.; Jürgens, G.; Geldner, N.; et al. Auxin inhibits endocytosis and promotes its own efflux from cells. Nature 2005, 435, 1251–1256. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Aleksandr, A.L.; Ildus, A.Y.; Patrick, H.B. Biostimulants in Plant Science: A Global Perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef]
- Shekari, G.; Javanmardi, J. Effects of Foliar Application Pure Amino Acid and Amino Acid Containing Fertilizer on Broccoli (Brassica oleracea L. var. italica) Transplant. Adv. Crop Sci. Tech. 2017, 5, 280. [Google Scholar] [CrossRef]
- Jiang, W.J. New Techniques for Soilless Cultivation of Vegetables, rev. ed.; Beijing Jindun Publishing House: Beijing, China, 2007; pp. 197–200. [Google Scholar]
- Hunt, R. Plant growth curves. In The Functional Approach to Plant Growth Analysis; Edward Arnold Ltd.: London, UK, 1982. [Google Scholar]
- Yoshida, S.; Kitano, M.; Eguchi, H. growth of lettuce plants (Lactuca sativa L.) under control of dissolved O2 concentration in hydroponics. Biotronics 1997, 26, 39–45. [Google Scholar]
- Woltz, S.; Jackson, C. Production of yellow strapleaf of chrysanthemum & similar disorders by amino acid treatment. Plant Physiol. 1961, 36, 197. [Google Scholar] [PubMed]
- León, A.P.; Martín, J.P.; Chiesa, A. Vermicompost application and growth patterns of lettuce (Lactuca sativa L.). Agric. Tropica Subtrop. 2012, 45, 134–139. [Google Scholar] [CrossRef][Green Version]
- Medek, D.E.; Ball, M.C.; Schortemeyer, M. Relative contributions of leaf area ratio and net assimilation rate to change in growth rate depend on growth temperature: Comparative analysis of subantarctic and alpine grasses. New Phytol. 2007, 175, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yu, H.; Liu, P.; Ma, C.; Li, Q.; Jiang, W. Ending composting during the thermophilic phase improves cultivation substrate properties and increasing winter cucumber yield. Waste Manag. 2018, 79, 260–272. [Google Scholar] [CrossRef]
- Ahn, Y.S. Plant analysis for evaluating plant nutrition. In International Training Workshop on Soil Test and Plant Analysis; RDA& FFTC/ASPAC, 1987. [Google Scholar]
- Snedecor, G.; Cochran, W. Statistical Method, 7th ed.; The Iowa State University Press: Ames, IA, USA, 1980; pp. 39–63. [Google Scholar]
- Colla, G.; Nardi, S.; Cardarelli, M.; Ertani, A.; Lucini, L.; Canaguier, R.; Rouphael, Y. Protein hydrolysates as biostimulants in horticulture. Sci. Hortic. 2015, 196, 28–38. [Google Scholar] [CrossRef]
- Liu, X.Q.; Chen, H.Y.; Ni, Q.X.; Kyu, S.L. Evaluation of the role of mixed amino acids in nitrate uptake and assimilation in leafy radish by using 15N-labeled nitrate. Agric. Sci. China 2008, 7, 1196–1202. [Google Scholar] [CrossRef]
- Tsouvaltzis, P.; Koukounaras, A.; Siomos, A.S. Application of amino acids improves lettuce crop uniformity and inhibits nitrate accumulation induced by the supplemental inorganic nitrogen fertilization. Int. J. Agric. Biol. 2014, 16, 951–955. [Google Scholar]
- Wang, H.J.; Wu, L.H.; Wang, M.Y.; Zhu, Y.H.; Tao, Q.N.; Zhang, F.S. Effects of amino acids replacing nitrate on growth, nitrate accumulation, and macroelement concentrations in pakchoi (Brassica chinensis L.). Pedosphere 2007, 17, 595–600. [Google Scholar] [CrossRef]
- Mobini, M.; Khoshgoftarmanesh, A.H.; Ghasemi, S. The effect of partial replacement of nitrate with arginine, histidine, and a mixture of amino acids extracted from blood powder on yield and nitrate accumulation in onion bulb. Sci. Hortic. 2014, 176, 232–237. [Google Scholar] [CrossRef]
- Vincill, E.D.; Bieck, A.M.; Spalding, E.P. Ca2+ conduction by an amino acid-gated ion channel related to glutamate receptors. Plant Physiol. 2012, 159, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Santi, C.; Zamboni, A.; Varanini, Z.; Pandolfini, T. Growth stimulatory effects and genome-wide transcriptional changes produced by protein hydrolysates in maize seedlings. Front. Plant Sci. 2017, 8, 433. [Google Scholar] [CrossRef] [PubMed]
- Price, C.A. Leaf Gui: Analyzing the geometry of veins and areoles using image segmentation algorithms. In High-Throughput Phenotyping in Plants; Springer: New York, NY, USA, 2012; pp. 41–49. [Google Scholar]
- Singh, R.; Parihar, P.; Prasad, S.M. Sulfur and calcium simultaneously regulate photosynthetic performance and nitrogen metabolism status in As-challenged Brassica juncea L. seedlings. Front. Plant Sci. 2018, 9, 772. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, A.; Prasad, M. Vertisol prevent cadmium accumulation in rice: Analysis by ecophysiological toxicity markers. Chemosphere 2014, 108, 85–92. [Google Scholar] [CrossRef]
- El-Shiaty, O.H.; El-Sharabasy, S.F.; Abd El-Kareim, A.H. Effect of some amino acids and biotin on callus and proliferation of date palm (Phoenix dactylifera L.) Sewy cultivar. Arab. J. Biotechnol. 2004, 7, 265–272. [Google Scholar]
- Kakkar, R.; Nagar, P.; Ahuja, P.; Rai, V. Polyamines and plant morphogenesis. Biol. Plant. 2000, 43, 1–11. [Google Scholar] [CrossRef]
- Padgett, P.E.; Leonard, R.T. Free amino acid levels and the regulation of nitrate uptake in maize cell suspension cultures. J. Exp. Bot. 1996, 47, 871–883. [Google Scholar] [CrossRef][Green Version]
- Shafeek, M.; Helmy, Y.; Magda, A.; Shalaby, F.; Nadia, M.O. Response of onion plants to foliar application of sources and levels of some amino acid under sandy soil conditions. J. Appl. Sci. Res. 2012, 8, 5521–5527. [Google Scholar]
- Davies, P.J. Plant Hormones: Biosynthesis, Signal Transduction, Action! Springer: New York, NY, USA, 2004. [Google Scholar]
- Colla, G.; Cardarelli, M.; Bonini, P.; Rouphael, Y. Foliar Applications of Protein Hydrolysate, Plant and Seaweed Extracts Increase Yield but Differentially Modulate Fruit Quality of Greenhouse Tomato. HortScience 2017, 52, 1214–1220. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; Giordano, M.; El-Nakhel, C.; Kyriacou, M.C.; De Pascale, S. Foliar applications of a legume-derived protein hydrolysate elicit dosedependent increases of growth, leaf mineral composition, yield and fruit quality in two greenhouse tomato cultivars. Sci. Hortic. 2017, 226, 353–360. [Google Scholar] [CrossRef]
- Bahari, A.; Pirdashti, H.; Yaghubi, M. The effects of amino acid fertilizers spraying on photosynthetic pigments and antioxidant enzymes of wheat (Triticum aestivum L.) under salinity stress. Int. J. Agron. Plant Prod. 2013, 4, 787–793. [Google Scholar]
- Anne, C.; Thomas, S. Regulation of chloroplast development and function by cytokinin. J. Exp. Bot. 2015, 66, 4999–5013. [Google Scholar]
- Fawzy, Z.; El-Shal, Z.; Yunsheng, L.; Zhu, O.; Sawan, O.M. Response of garlic (Allium Sativum L.) plants to foliar spraying of some bio-stimulants under sandy soil condition. J. Appl. Sci. Res. 2012, 8, 770–776. [Google Scholar]
- Romero, I.; Téllez, J.; Yamanaka, L.E.; Steindel, M.; Romanha, A.J.; Grisard, E.C. Transsulfuration is an active pathway for cysteine biosynthesis in Trypanosoma rangeli. Parasites Vectors 2014, 7, 197. [Google Scholar] [CrossRef] [PubMed]
- Abbas, S.H.; Sohail, M.; Saleem, M.; Mahmood, T.; Aziz, I.; Qamar, M.; Majeed, A.; Arif, M. Effect of L-tryptophan on plant weight and pod weight in chickpea under rainfed conditions. SciTech Dev. 2013, 32, 277–280. [Google Scholar]
- Herve, J.J. Biostimulants, a new concept for the future; prospects offered by the chemistry of synthesis and biotechnology. C. R. Acad. Agric. Fr. 1994, 80, 91–102. [Google Scholar]
- Yunna, Z.; Gang, L.; Houcheng, L.; Guangwen, S.; Riyuan, C.; Shiwei, S. Effects of partial replacement of nitrate with different nitrogen forms on the yield, quality and nitrate content of Chinese kale. Commun. Soil Sci. Plant. Anal. 2018, 1384–1393. [Google Scholar]
- Chen, G.L.; Gao, X.R.; Zhang, X.B. Effect of partial replacement of nitrate by amino acid and urea on nitrate content of non-heading Chinese cabbage and lettuce in hydroponic condition. Agric. Sci. China 2002, 444–449. [Google Scholar]
- Valpuesta, V.; Botella, M. Biosynthesis of L-ascorbic acid in plants: New pathways for an old antioxidant. Trends Plant Sci. 2004, 9, 573–577. [Google Scholar] [CrossRef]
- Podsedek, A. Natural antioxidants and antioxidant capacity of Brassica vegetables: A review. LWT-Food Sci. Technol. 2007, 40, 1–11. [Google Scholar] [CrossRef]
- John, S.J.; Guha-Mukherjee, S. Plant Molecular Biology and Biotechnology, 1st ed.; Narosa Pub. House: New Delhi, India, 1997; pp. 17–28. [Google Scholar]
- El-sharabasy, S.; Fatma, I.; Gehan, H.; El-Dawayaty, M. Effect of different amino acids at different concentrations on multiplication and rooting stage of in vitro propagation of strawberries (Fragaria X Ananassa Duch cv. Chandler). Egypt. J. Genet. Cytol. 2015, 44, 31–34. [Google Scholar]
- Rosati, A.; Day, K.; DeJong, T. Distribution of leaf mass per unit area and leaf nitrogen concentration determine partitioning of leaf nitrogen within tree canopies. Tree Physiol. 2000, 20, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Thimann, K.V. Auxins and the inhibition of plant growth. Biol. Rev. 1939, 14, 314–337. [Google Scholar] [CrossRef]
| Treatments | Net Photosynthesis Rate (Pn) (µmol CO2 m−2 s−1) | Stomatal Conductance (mol m−2 s−1) | Ci (µmol/mol) | Transpiration Rate (Tr) (mol H2O m−2 s−1) | Total Chlorophyll Content (SPAD Values) |
|---|---|---|---|---|---|
| Control | 7.31 a | 0.07 a | 335.78 b | 1.86 a | 16.5 n.s. |
| L-methionine (Meth) | 4.33 b | 0.03 b | 369.55 ab | 0.97 bc | 16.7 n.s. |
| L-tryptophan (Try) | 3.08 c | 0.03 b | 426.67 a | 0.83 c | 15.6 n.s. |
| L-glycine (Gly) | 4.63 b | 0.04 b | 390.34 ab | 1.53 b | 17.4 n.s. |
| Treatments | LAI (cm2 cm−2) | LDMC (g g−1) | RMR (g g−1) | SLA (cm2 g−1) | LAR (cm2 g−1) | RGR (g g−1 d−1) |
|---|---|---|---|---|---|---|
| Control | 2.73 b | 0.05 b | 0.01 b | 0.07 n.s. | 22.67 bc | 4.44 b |
| L-methionine | 2.06 b | 0.03 b | 0.01 b | 0.04 n.s. | 6.38 c | 1.18 c |
| L-tryptophan | 26.18 a | 0.58 a | 0.15 a | 0.10 n.s. | 63.65 a | 1.34 c |
| L-glycine | 3.85 b | 0.05 b | 0.01 b | 0.07 n.s. | 35.78 b | 7.31 a |
| Treatments | Vitamin C (mg 100 g−1) | Dry Matter (%) |
|---|---|---|
| Control | 0.25 n.s. | 13.4 bc |
| L-methionine | 0.3 n.s. | 10.1 c |
| L-tryptophan | 0.2 n.s. | 94.5 a |
| L-glycine | 0.3 n.s. | 24.7 b |
| Treatments | N | P | K | S | Ca | Mg | Fe | Cu | Na | Zn | Al |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (% DW) | (mg/g) | ||||||||||
| Control | 1.4 b | 12.3 d | 208.1 b | 9.8 b | 93.7 a | 22.9 a | 6.8 a | 0.1 c | 18.3 a | 0.2 b | 13.1 a |
| L-methionine | 4.3 a | 32.2 b | 420.7 a | 10.1 b | 66.3 c | 12.6 b | 2.1 b | 0.06 d | 4.48 b | 0.4 a | 7.1 b |
| L-tryptophan | 3.7 a | 36.8 a | 421.4 a | 13.1 a | 81.5 b | 13.4 b | 2.8 b | 0.14 b | 3.95 b | 0.4 a | 9.0 ab |
| L-glycine | 4.3 a | 21.7 c | 230.8 b | 13.3 a | 44.6 d | 12.6 b | 6.9 a | 0.23 a | 3.40 b | 0.3 a | 9.6 ab |
| Treatment (mg/L) | LAI (cm2 cm−2) | RMR (g g−1) | SLA (cm2 g−1) | LAR (cm2 g−1) | RGR (g g−1 d−1) | RWC (%) |
|---|---|---|---|---|---|---|
| Control | 0.24 a | 0.04 b | 1679.50 a | 51.5 ab | 0.33 c | 91.2 a |
| 2200 | 0.06 c | 0.34 a | 1259.19 ab | 76.6 a | 0.34 c | 11.11 c |
| 220 | 0.17 b | 0.06 b | 572.02 b | 42.2 ab | 0.23 c | 72.22 b |
| 22 | 0.25 a | 0.02 b | 1118.38 ab | 36.9 b | 0.27 c | 93.72 a |
| 2.2 | 0.24 a | 0.02 b | 1937.15 a | 46.1 ab | 0.52 c | 92.96 a |
| 0.22 | 0.26 a | 0.01 b | 1496.32 a | 34.5 b | 1.6 b | 93.28 a |
| 0.02 | 0.27 a | 0.01 b | 1641.48 a | 45.5 ab | 3.7 a | 90.77 a |
| Treatment (mg/L) | Net Photosynthesis Rate (Pn) (µmol CO2 m−2 s−1) | Stomatal Conductance (mol m−2 s−1) | Ci (µmol/mol) | Transpiration Rate (Tr) (mol H2O m−2 s−1) | Total Chlorophyll Content (SPAD) |
|---|---|---|---|---|---|
| Control | 5.36 b | 0.10 abc | 363.22 bc | 1.94 b | 24.7 cd |
| 2200 | 1.67 e | 0.03 c | 379.51 ab | 0.64 d | 23.7 d |
| 220 | 2.37 de | 0.05 bc | 398.91 a | 1.11 c | 29.9 a |
| 22 | 3.07 d | 0.15 a | 395.1 a | 1.35 c | 26.2 bc |
| 2.2 | 4.47 c | 0.06 bc | 347.86 c | 1.27 c | 27.3 b |
| 0.22 | 7.16 a | 0.12 ab | 369.23 bc | 2.41 a | 28.1 ab |
| 0.02 | 6.61 a | 0.09 abc | 349.91 c | 1.92 b | 27.3b |
| Treatment (mg/L) | N | P | K | S | Ca | Mg | Fe | Cu | Na | Zn | Al |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (% DW) | (mg/g) | ||||||||||
| Control | 5.3 c | 17.5 d | 128.3 c | 9.7 d | 67.1 c | 11.3 d | 2.5 c | 0.03 c | 4.3 d | 0.5 c | 7.6 b |
| 2200 | 2.1 e | 27 bc | 164.1 c | 23.8 a | 106.1 ab | 19.2 abc | 5.5 a | 0.08 a | 15.4 a | 0.8 b | 9.9 ab |
| 220 | 2.3 e | 27.1 bc | 233.1 b | 19.6 b | 127.9 a | 24.5 a | 4.4 ab | 0.04 bc | 11 b | 0.4 c | 10.4 a |
| 22 | 4.2 d | 25.2 c | 254.3 b | 12.4cd | 83.2 bc | 16.27 cd | 3.2 bc | 0.04 bc | 6 c | 0.4 c | 8.7 ab |
| 2.2 | 5.7 bc | 39.0 a | 351.8 a | 14.1 c | 108.8 bc | 22.7 ab | 3.0 c | 0.05 b | 11 b | 1.1 a | 9.3 ab |
| 0.22 | 6.7 a | 34.9 ab | 336.4 a | 13 cd | 107.1 ab | 19.8 abc | 3.2 bc | 0.04 bc | 9.4 b | 0.4 c | 9 ab |
| 0.02 | 6.1 b | 29.5 bc | 246.5 b | 12.5 cd | 99.2 abc | 17.3 bc | 3.4 bc | 0.05 b | 6.6 b | 0.4 c | 9.1 ab |
| Treatment (mg/L) | Vitamin C (mg 100 g−1) | LDMC (g g−1) | DM (%) | Mean Fresh Weight (g) | Mean Dry Weight (g) |
|---|---|---|---|---|---|
| Control | 0.21 a | 0.13 b | 4.7 b | 37.97 bc | 4.8 c |
| 2200 | 0.11 b | 0.49 a | 48.8 a | 0.86 d | 0.8 d |
| 220 | 0.19 a | 0.12 b | 11.9 ab | 10.89 cd | 2.3 d |
| 22 | 0.19 a | 0.06 b | 5.69 b | 58.89 ab | 5.4 bc |
| 2.2 | 0.21 a | 0.06 b | 5.62 b | 44.29 ab | 7.97 a |
| 0.22 | 0.23a | 0.05 b | 5.29 b | 70.94 a | 8.7 a |
| 0.02 | 0.21 a | 0.04 b | 4.23 b | 60.89 ab | 7.3 ab |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, S.; Yu, H.; Li, Q.; Gao, Y.; Sallam, B.N.; Wang, H.; Liu, P.; Jiang, W. Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability. Agronomy 2019, 9, 266. https://doi.org/10.3390/agronomy9050266
Khan S, Yu H, Li Q, Gao Y, Sallam BN, Wang H, Liu P, Jiang W. Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability. Agronomy. 2019; 9(5):266. https://doi.org/10.3390/agronomy9050266
Chicago/Turabian StyleKhan, Shumaila, Hongjun Yu, Qiang Li, Yinan Gao, Basheer Noman Sallam, Heng Wang, Peng Liu, and Weijie Jiang. 2019. "Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability" Agronomy 9, no. 5: 266. https://doi.org/10.3390/agronomy9050266
APA StyleKhan, S., Yu, H., Li, Q., Gao, Y., Sallam, B. N., Wang, H., Liu, P., & Jiang, W. (2019). Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability. Agronomy, 9(5), 266. https://doi.org/10.3390/agronomy9050266





