Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Soilless Cultivation
2.2. Determination of Flower Production, Stigma Yield and Corm Growth
2.3. Preparation of the Saffron Extract
2.4. Determination of Saffron Quality by ISO 3632
2.5. Determination of Bioactive Compounds by HPLC
2.6. Phytochemical Characterisation
2.7. AMF Evaluation
2.8. Chemicals and Reagents
2.9. Statistical Analysis
3. Results and Discussion
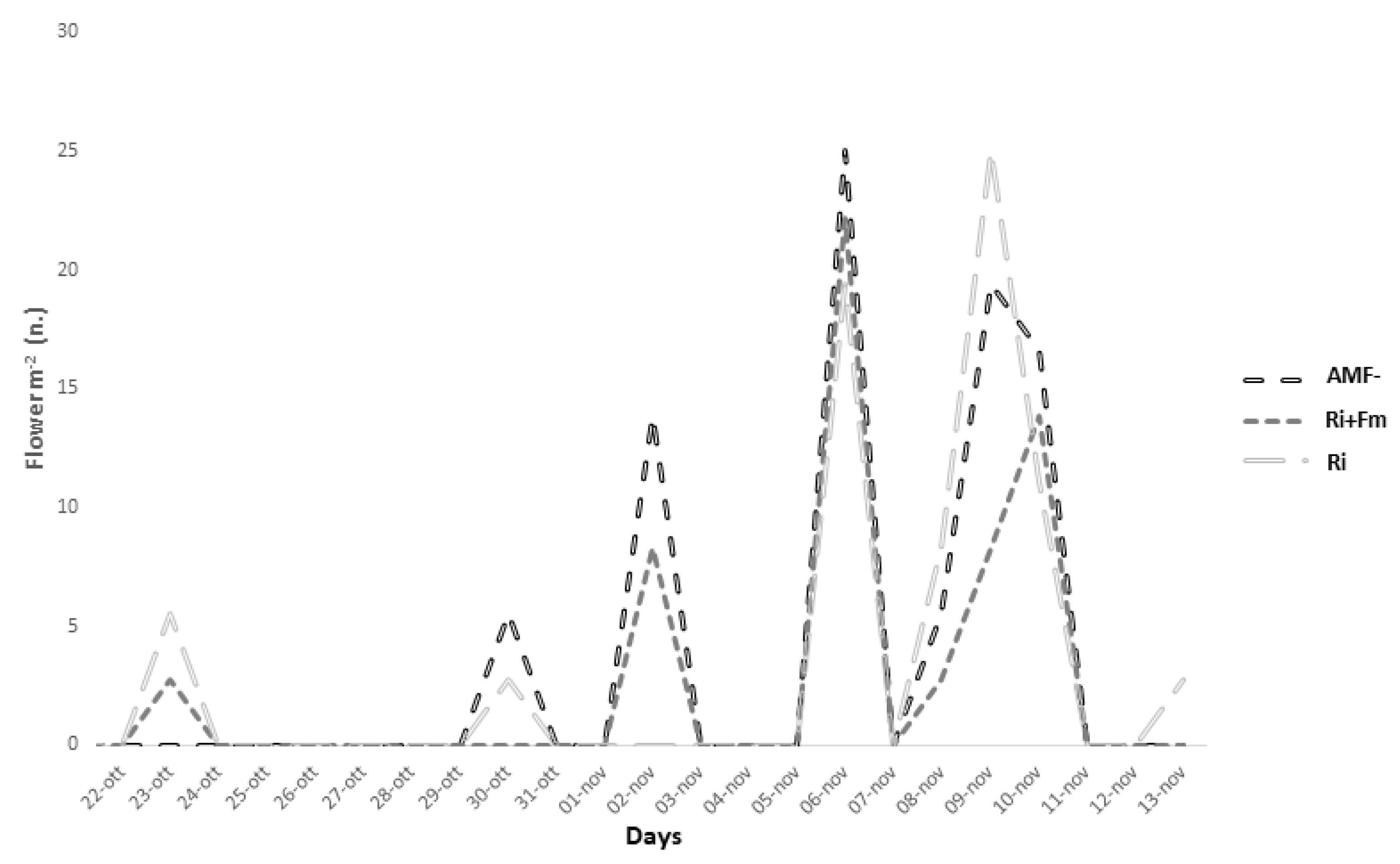
3.1. Crop Performance, Quality and Secondary Metabolite Content of Saffron in Soilless Cultivation
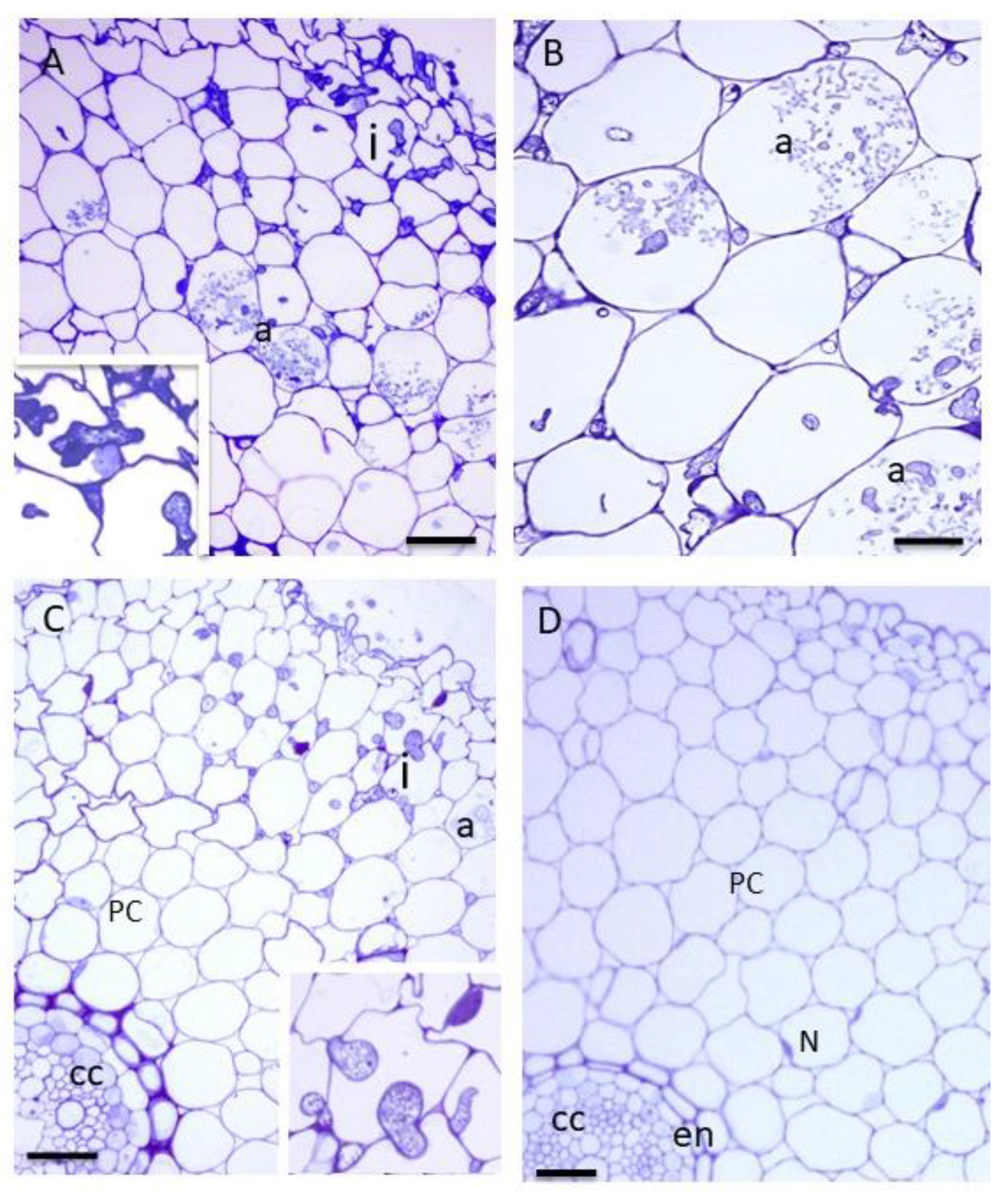
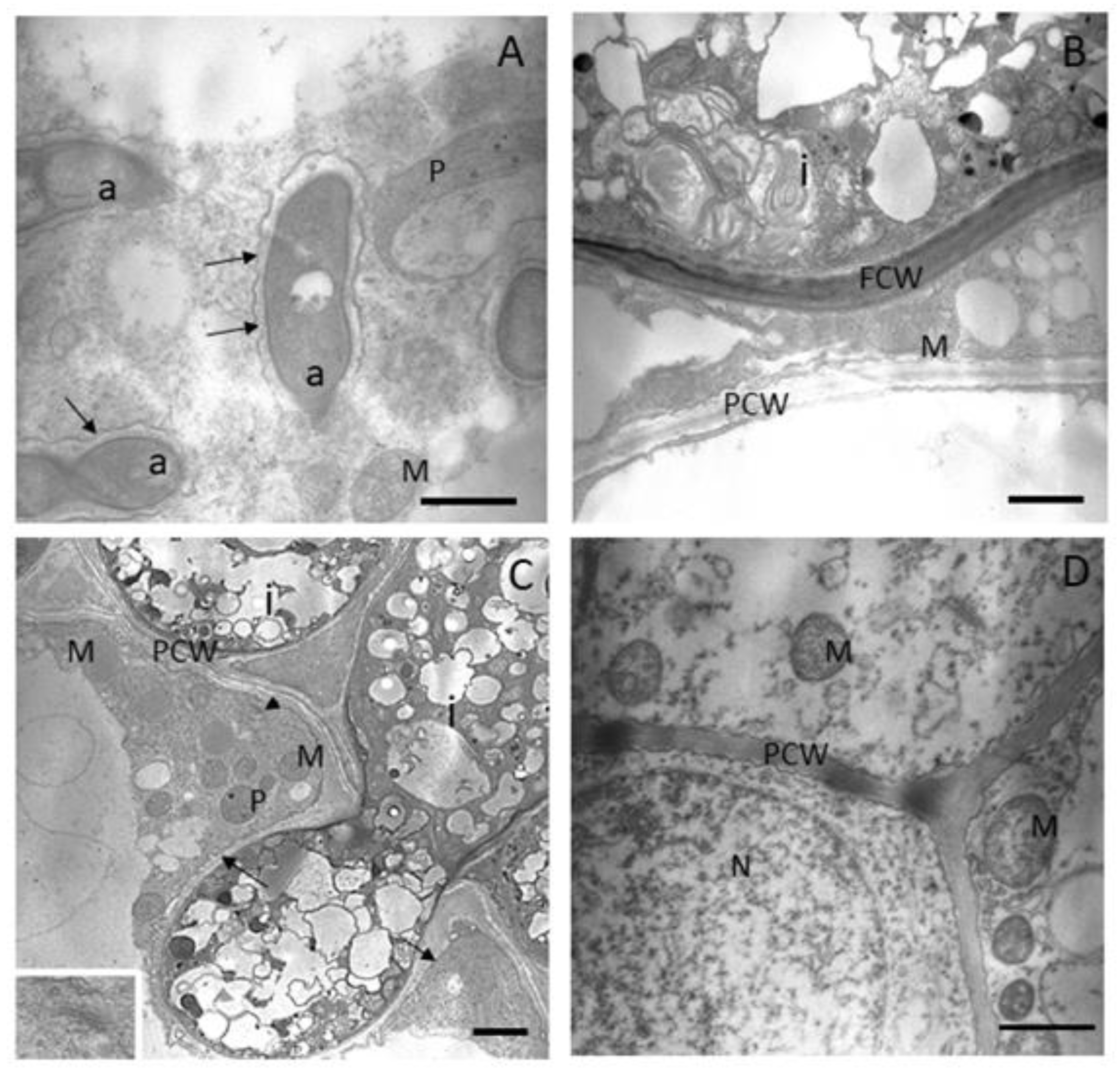
3.2. AMF Colonisation
3.3. Impact of AMF on Saffron in Soilless Cultivation
3.3.1. Crop Performance and Quality Classification
3.3.2. Saffron Metabolic Profiling Comparing to Other Foods
3.3.3. Soilless Cultivation vs. Open Field
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gresta, F.; Santonoceto, C.; Avola, G. Crop rotation as an effective strategy for saffron (Crocus sativus L.) cultivation. Sci. Hortic. 2016, 211, 34–39. [Google Scholar] [CrossRef]
- Maleki, F.; Kazemi, H.; Siahmarguee, A.; Kamkar, B. Development of a land use suitability model for saffron (Crocus sativus L.) cultivation by multi-criteria evaluation and spatial analysis. Eco Eng. 2017, 106, 140–153. [Google Scholar] [CrossRef]
- Giorgi, A.; Scheurer, T. Alpine resources: Assets for a promising future-conclusions from the forum alpinum. Mount. Res. Dev. 2015, 35, 414–415. [Google Scholar] [CrossRef]
- Husaini, A.; Shafiq, A.; Parvaze, S.; Rather, G.; Parray, A.; Shikari, A.; Javid, M. Bioinformatics for saffron (Crocus sativus L.) improvement. Comm. Biomet. Crop. Sci. 2009, 4, 3–8. [Google Scholar]
- Gresta, F.; Lombardo, G.; Siracusa, L.; Ruberto, G. Saffron an alternative crop for sustainable agricultural systems. A review. Agron. Sustain. Dev. 2008, 28, 95–112. [Google Scholar] [CrossRef]
- Gresta, F.; Avola, G. Hypothesis on the relation between meteorological data and flowering peaks in saffron (Crocus sativus L.) crop. Acta Hortic. 2017, 1184, 189–194. [Google Scholar] [CrossRef]
- Elgazar, A.; Rezq, A.; Bukhari, H. Anti-Hyperglycemic Effect of Saffron Extract in Alloxan-Induced Diabetic Rats. Europ. J. Biol. Sci. 2013, 5, 14–22. [Google Scholar]
- Baba, S.; Mohiudin, T.; Basu, S.; Swarnkar, M.; Malik, A.; Abbas, N.; Singh, A.; Ashraf, N. Comprehensive transcriptome analysis of Crocus sativus for discovery and expression of genes involved in apocarotenoid biosynthesis. BMC Genom. 2015, 16, 698. [Google Scholar] [CrossRef] [PubMed]
- Lahmass, I.; Touria, L.; Delporte, C.; Sikdar, S.; Antwerpen, P.; Saalaoui, E.; Mégalizzi, V. The waste of saffron crop, a cheap source of bioactive compounds. J. Funct. Foods 2017, 35, 341–351. [Google Scholar] [CrossRef]
- Cusano, E.; Consonni, R.; Petrakis, E.A.; Astraka, K.; Cagliani, L.R.; Polissiou, M.G. Integrated analytical methodology to investigate bioactive compounds in Crocus sativus L. flowers. Phytochem. Anal. Phytochem. Analysis 2018, 29, 1–11. [Google Scholar] [CrossRef]
- Menghini, L.; Leporini, L.; Vecchiotti, G.; Locatelli, M.; Carradori, S.; Ferrante, C.; Zengin, G.; Recinella, L.; Chiavaroli, A.; Leone, S.; et al. Crocus sativus L. stigmas and byproducts: Qualitative fingerprint, antioxidant potentials and enzyme inhibitory activities. Food Res. Int. 2018, 109, 91–98. [Google Scholar] [CrossRef]
- Caser, M.; Maxaieie Victorino, Í.M.; Demasi, S.; Berruti, A.; Donno, D.; Lumini, E.; Bianciotto, V.; Scariot, V. Saffron cultivation in marginal Alpine environments: How AMF inoculation modulates yield and bioactive compounds. Agronomy 2019, 9, 12. [Google Scholar] [CrossRef]
- Talaei, A.; Moghadam, H.; Tabassi, S.; Mohajeri, S. Crocin, the main active saffron constituent, as an adjunctive treatment in major depressive disorder: A randomized, double-blind, placebo-controlled, pilot clinical trial. J. Affect. Disord. 2015, 174, 51–56. [Google Scholar] [CrossRef]
- Landi, R. Lo zafferano: Tradizione e qualità. Camera di commercio: Industria, Artigianato e Agricoltura di Firenze; CCIAA (Camera di Commercio: Industria, Artigianato e Agricoltura): Firenze, Italy, 2007; Volume 4, p. 65. [Google Scholar]
- Lachaud, C. La Bible du Safranier. Tout savoir sur le Crocus sativus et sur le Safran, 1st ed.; La Paradisière: Correze, France, 2012; ISBN 978-2-7466-4412-0. [Google Scholar]
- De Juan, J.A.; Córcoles, H.L.; Muñoz, R.M.; Picornell, M.R. Yield and yield components of saffron under different cropping systems. Ind. Crops Prod. 2009, 30, 212–219. [Google Scholar] [CrossRef]
- Yarami, N.; Kamgar-Haghighi, A.A.; Sepaskhah, A.R.; Zand-Parsa, S. Determination of the potential evapotranspiration and crop coefficient for saffron using a water-balance lysimeter. Arch. Agr. Soil Sci. 2011, 57, 727–740. [Google Scholar] [CrossRef]
- Molina, R.; Valero, M.; Navarro, Y.; Guardiola, J.; García-Luis, A. Temperature effects on flower formation in saffron (Crocus sativus L.). Sci. Hortic. 2005, 103, 361–379. [Google Scholar] [CrossRef]
- Maggio, A.; Raimondi, G.; Martino, A.; De Pascale, S. Soilless cultivation of saffron in mediterranean environment. Acta Hortic. 2006, 718, 515–522. [Google Scholar] [CrossRef]
- Benschop, M. Crocus; De Hertogh, A., Le Nard, M., Eds.; The Physiology of Flower Bulbs, Elsevier Science Publishers: Amsterdam, The Netherlands, 1993; pp. 257–272. [Google Scholar]
- De Mastro, G.; Ruta, C. Relation between corm size and saffron (Crocus sativus L.) flowering. Acta Hortic. 1993, 334, 512–517. [Google Scholar] [CrossRef]
- Douglas, M.H.; Smallfield, B.M.; Wallace, A.R.; McGimpsey, J.A. Saffron (Crocus sativus L.): The effect of mother corm size on progeny multiplication, flower and stigma production. Sci. Hortic. 2014, 166, 50–58. [Google Scholar] [CrossRef]
- Kumar, R. Calibration and validation of regression model for non-destructive leaf area estimation of saffron (Crocus sativus L.). Sci. Hortic. 2009, 122, 142–145. [Google Scholar] [CrossRef]
- Mollafilabi, A.; Koocheki, A.; Moeinerad, H.; Kooshki, L. Effect of plant density and weight of corm on yield and yield components of saffron (Crocus sativus L.) under soil, hydroponic and plastic tunnel cultivation. Acta Hortic. 2013, 997, 51–58. [Google Scholar] [CrossRef]
- Putra, P.A.; Yuliando, H. Soilless culture system to support water use efficiency and product quality: A review. Agric. Agricult. Sci. Proc. 2015, 3, 283–288. [Google Scholar] [CrossRef]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Helal Beigi, Y.; Khoshgoftar, M.; Shamsi, A.; Zamani, N. Possibility of saffron corm growth in different bed of cultivation in soilless system. In Proceedings of 1st Congress of Hydroponic in Greenhouse Production; Isfahan University of Technology: Isfahan, Iran, 2009. [Google Scholar]
- Sourell, F.; Weathers, P. The growth of saffron (Crocus sativus L.) in aeroponics and hydroponics. J. Herb. Spices Med. Plants 2000, 7, 113–127. [Google Scholar]
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi acta s biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Berruti, A.; Lumini, E.; Balestrini, R.; Bianciotto, V. Arbuscular mycorrhizal fungi as natural biofertilizers: Let’s benefit from past successes. Front. Microbiol. 2016, 6, 1559. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis; Academic Press: London, UK, 1997; Volume 40, p. 154. [Google Scholar]
- Todeschini, V.; Lahmidi, N.A.; Mazzucco, E.; Marsano, F.; Gosetti, F.; Robotti, E.; Bona, E.; Massa, N.; Bonneau, L.; Marengo, E.; et al. Impact of beneficial microorganisms on strawberry growth, fruit production, nutritional quality, and volatilome. Front. Plant. Plant Sci. 2018, 9, 1611. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, E.; Bedini, S. Enhancing ecosystem services in sustainable agriculture: Biofertilization and biofortification of chickpea (Cicer arietinum L.) by arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2014, 68, 429–439. [Google Scholar] [CrossRef]
- Zeng, Y.; Guo, L.; Chen, B.; Hao, Z.; Wang, J.; Huang, L.; Yang, G.; Cui, X.; Yang, L.; Wu, Z.; et al. Arbuscular mycorrhizal symbiosis and active ingredients of medicinal plants: Current research status and prospectives. Mycorrhiza 2013, 23, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Giovannetti, M.; Avio, L.; Sbrana, C. Improvement of nutraceutical value of food by plant symbionts; Ramawat, K.G., Merillon, J.M., Eds.; Natural Products, Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Springer: Berlin/Heidelberg, Germany, 2013; pp. 2641–2662. [Google Scholar]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Response of PGPR and AM fungi toward growth and secondary metabolite production in medicinal and aromatic plants; Hakeem, K., Akhtar, M., Eds.; Plant, Soil and Microbes; Springer: Heidelberg, Germany, 2016. [Google Scholar]
- Avio, L.; Turrini, A.; Giovannetti, M.; Sbrana, C. Designing the ideotype mycorrhizal symbionts for the production of healthy food. Front. Plant Sci. 2018, 9, 1089. [Google Scholar] [CrossRef] [PubMed]
- De Sousa Oliveira, M.; da Silva Campos, M.; de Albuquerque, U.P.; da Silva, F.S.B. Arbuscular mycorrhizal fungi (AMF) affects biomolecules content in Myracrodruon urundeuva seedlings. Ind. Crops Prod. 2013, 50, 244–247. [Google Scholar] [CrossRef]
- Aimo, S.; Gosetti, F.; D´Agostino, G.; Gamalero, E.; Gianotti, V.; Bottaro, M.; Gennaro, M.; Berta, G. Use of arbuscular mycorrhizal fungi and beneficial soil bacteria to improve yield and quality of saffron (Crocus sativus L.). Acta Hortic. 2010, 850, 159–162. [Google Scholar] [CrossRef]
- Lone, R.; Shuab, R.; Koul, K. AMF association and their effect on metabolite mobilization, mineral nutrition and nitrogen assimilating enzymes in saffron (Crocus sativus) plant. J. Plant Nutr. 2016, 39, 1852–1862. [Google Scholar] [CrossRef]
- Shajari, M.A.; Moghaddam, P.R.; Ghorbani, R.; Koocheki, A. Increasing saffron (Crocus sativus L.) corm size through the mycorrhizal inoculation, humic acid application and irrigation managements. J. Plant Nutr. 2018, 41, 1047–1064. [Google Scholar] [CrossRef]
- Mohebi-Anabat, M.; Riahi, H.; Zanganeh, S.; Sadeghnezhad, E. Effects of arbuscular mycorrhizal inoculation on the growth, photosynthetic pigments and soluble sugar of Crocus sativus L. (saffron) in autoclaved soil. Int. J. Agron. Agric. Res. 2015, 6, 296–304. [Google Scholar]
- ISO 3632-1:2011. Spices—Saffron (Crocus sativus L.); Food products SC 7, Spices, culinary herbs and condiments; International Organization for Standardization: Geneva, Switzerland, September 2011. [Google Scholar]
- Giorgi, A.; Pentimalli, D.; Giupponi, L.; Panseri, S. Quality traits of saffron (Crocus sativus L.) produced in the Italian Alps. Open Agriculture 2017, 2, 52–57. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.G.; Prgomet, Z.; Beccaro, G.L. Advances in Ribes x nidigrolaria Rud. Bauer & A. Bauer fruits as potential source of natural molecules: A preliminary study on physico-chemical traits of an underutilized berry. Sci. Hortic. 2018, 237, 20–27. [Google Scholar]
- Donno, D.; Mellano, M.G.; Prgomet, Z.; Cerutti, A.K.; Beccaro, G.L. Phytochemical characterization and antioxidant activity evaluation of Mediterranean medlar fruit (Crataegus azarolus L.): Preliminary study of underutilized genetic resources as a potential source of health-promoting compound for food supplements. J. Food Nutrit. Res. 2017, 56, 18–31. [Google Scholar]
- Caser, M.; D’Angiolillo, F.; Chitarra, W.; Lovisolo, C.; Ruffoni, B.; Pistelli, L.; Pistelli, L.; Scariot, V. Ecophysiological and phytochemical responses of Salvia sinaloensis Fern. to drought stress. Plant Growth Regul. 2018, 84, 383–394. [Google Scholar] [CrossRef]
- Caser, M.; Chitarra, W.; D’Angiolillo, F.; Perrone, I.; Demasi, S.; Lovisolo, C.; Pistelli, L.; Pistelli, L.; Scariot, V. Drought stress adaptation modulates plant secondary metabolite production in Salvia dolomitica Codd. Ind. Crop Prod. 2019, 129, 85–96. [Google Scholar] [CrossRef]
- Hoch, H.C. Freeze-substitution of fungi; Aldrich, H.C., Todd, W.J., Eds.; Ultrastructure techniques of microorganisms; Plenum Press: New York, NY, USA, 1986; pp. 183–211. [Google Scholar]
- Reynolds, E.S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell. Biol. 1963, 17, 208–212. [Google Scholar] [CrossRef]
- Gresta, F.; Avola, G.; Lombardo, G.M.; Siracusa, L.; Ruberto, G. Analysis of flowering, stigmas yield and qualitative traits of saffron (Crocus sativus L.) as affected by environmental conditions. Sci. Hortic. 2009, 119, 320–324. [Google Scholar]
- Cavusoglu, A.; Erkel, E. The effect of different planting areas and corm size on yield and harvest period of saffron (Crocus sativus L.) in Kocaeli province. J. Akdeniz Univ. Faculty Agric. 2005, 18, 179–184. [Google Scholar]
- Koocheki, A.; Seyyedi, S. How irrigation rounds and mother corm size control saffron yield, quality, daughter corms behavior and phosphorus uptake. Sci. Hortic. 2016, 213, 132–143. [Google Scholar] [CrossRef]
- Turhan, H.; Kahriman, F.; Egesel, C.; Kemal Gul, M. The effects of different growing media on flowering and corm formation of saffron (Crocus sativus L.). Afr. J. Biotech. 2007, 6, 2328–2332. [Google Scholar]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H. Evaluation of Crocus sativus L. stigma phenolic and flavonoid compounds and its antioxidant activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef]
- Asdaq, S.; Inamdar, M. Potential of garlic and its active constituent, S-allyl cysteine, as antihypertensive and cardioprotective in presence of captopril. Phytomedicine 2010, 17, 1016–1026. [Google Scholar] [CrossRef] [PubMed]
- Makhlouf, H.; Saksouk, M.; Habib, J.; Chahine, R. Determination of antioxidant activity of saffron taken from the flower of Crocus sativus grown in Lebanon. Afr. J. Biotech. 2011, 10, 8093–8100. [Google Scholar]
- Ordoudi, S.; Befani, C.; Nenadis, N.; Koliakos, G.; Tsimidou, M. Further Examination of Antiradical Properties of Crocus sativus Stigmas Extract Rich in Crocins. J. Agric. Food Chem. 2009, 57, 3080–3086. [Google Scholar] [CrossRef]
- Siracusa, L.; Gresta, F.; Avola, G.; Lombardo, G.M.; Ruberto, G. Influence of corm provenance and environmental condition on yield and apocarotenoid profiles in saffron (Crocus sativus L.). J. Food Comp. Anal. 2010, 23, 394–400. [Google Scholar] [CrossRef]
- Manzo, A.; Panseri, S.; Bertoni, D.; Giorgi, A. Economic and qualitative traits of Italian Alps saffron. J. Mt. Sci. 2015, 12, 1542–1550. [Google Scholar] [CrossRef]
- Sánchez-Vioque, R.; Rodríguez-Conde, M.F.; Reina-Ureña, J.V.; Escolano-Tercero, M.A.; Herraiz-Peñalver, D.; Santana-Méridas, O. In vitro antioxidant and metal chelating properties of corm, tepal and leaf from saffron (Crocus sativus L.). Ind. Crop Prod. 2012, 39, 149–153. [Google Scholar] [CrossRef]
- Rahaiee, S.; Moini, S.; Hashemi, M.; Shojaosadati, S. Evaluation of antioxidant activities of bioactive compounds and various extracts obtained from saffron (Crocus sativus L.): A review. J. Food Sci. Tech. 2015, 52, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Shahi, T.; Assadpour, E.; Jafari, S. Main chemical compounds and pharmacological activities of stigmas and tepals of ‘red gold’; saffron. Trends Food Sci. Technol. 2016, 58, 69–78. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.G.; Raimondo, E.; Cerutti, A.K.; Prgomet, Z.; Beccaro, G.L. Influence of applied drying methods on phytochemical composition in fresh and fried goji fruits by HPLC fingerprint. Eur. Food Res. Technol. 2016, 242, 1961–1974. [Google Scholar] [CrossRef]
- Heidari, M.; Karami, V. Effects of different mycorrhiza species on grain yield, nutrient uptake and oil content of sunflower under water stress. J. Saudi Soc. Agric. Sci. 2014, 13, 9–13. [Google Scholar] [CrossRef]
- Araim, G.; Saleem, A.; Arnason, J.T.; Charest, C. Root colonization by arbuscular mycorrhizal (AM) fungus increases growth and secondary metabolism of purple coneflower, Echinacea purpurea (L.) Moench. J. Agric. Food Chem. 2009, 57, 2255–2258. [Google Scholar] [CrossRef]
- Ceccarelli, N.; Curadi, M.; Martelloni, L.; Sbrana, C.; Picciarelli, P.; Giovannetti, M. Mycorrhizal colonization impacts on phenolic content and antioxidant properties of artichoke leaves and flower heads two years after field transplant. Plant Soil 2010, 335, 311–323. [Google Scholar] [CrossRef]
- Avio, L.; Sbrana, C.; Giovannetti, M.; Frassinetti, S. Arbuscular mycorrhizal fungi affect total phenolics content and antioxidant activity in leaves of oak leaf lettuce varieties. Sci. Hortic. 2017, 224, 265–271. [Google Scholar] [CrossRef]
- Toussaint, J.P.; Smith, F.A.; Smith, S.E. Arbuscular mycorrhizal fungi can induce the production of phytochemicals in sweet basil irrespective of phosphorus nutrition. Mycorrhiza 2007, 17, 291–297. [Google Scholar] [CrossRef]
- Nell, M.; Vötsch, M.; Vierheilig, H.; Steinkellner, S.; Zitterl-Eglseer, K.; Franz, C.; Novak, J. Effect of phosphorus uptake on growth and secondary metabolites of garden (Salvia officinalis L.). J. Sci. Food Agric. 2009, 89, 1090–1096. [Google Scholar] [CrossRef]
- Geneva, M.P.; Stancheva, I.V.; Boychinova, M.M.; Mincheva, N.H.; Yonova, P.A. Effects of foliar fertilization and arbuscular mycorrhizal colonization on Salvia officinalis L. growth, antioxidant capacity, and essential oil composition. J. Sci. Food Agric. 2010, 90, 696–702. [Google Scholar]
- Donno, D.; Beccaro, G.; Mellano, M.; Cerutti, A.; Bounous, G. Goji berry fruit (Lycium spp.): Antioxidant compound fingerprint and bioactivity evaluation. J. Funct. Foods 2015, 18, 1070–1085. [Google Scholar] [CrossRef]
- Vasco, C.; Ruales, J.; Kamal-Eldin, A. Total phenolic compounds and antioxidant capacities of majpr fruits from Ecuador. Food Chem. 2008, 111, 816–823. [Google Scholar] [CrossRef]
- Donno, D.; Cerutti, A.K.; Prgomet, I.; Mellano, M.G.; Beccaro, G.L. Foodomics for mulberry fruit (Morus spp.): Analytical fingerprint as antioxidants’ and health properties’ determination tool. Food Res. Int. 2015, 69, 179–188. [Google Scholar] [CrossRef]
- Ma, H.; Johnson, S.L.; Liu, W.; DaSilva, N.A.; Meschwitz, S.; Dain, J.A.; Seeram, N.P. Evaluation of polyphenol anthocyanin-enriched extracts of blackberry, black raspberry, blueberry, cranberry, res raspberry, and strawberry for free radical scavenging, reactive carbonyl species trapping, anti-glycation, anti-β-amyloid aggregation, and microglial neuroprotective effects. Int. J. Mol. Sci. 2018, 19, 461. [Google Scholar]
| HPLC Method | Class | Standard | Stationary Phase | Mobile Phase | Flow (mL min−1) | Time (min) |
|---|---|---|---|---|---|---|
| α | Cinnamic acids | Caffeic acid Chlorogenic acid Coumaric acid Cerulic acid | KINETEX–C18 column (4.6 × 150 mm, 5 µm) | A: 10mM KH2PO4/H3PO4, pH = 2.8 B: CH3CN | 1.5 | 20 + 2 (CT) |
| Flavanols | Hyperoside Isoquercitrin Quercetin Wuercitrin Rutin | |||||
| β | Benzoic acids | Ellagic acid Gallic acid | A: H2O/CH3OH/HCOOH (5:95:0.1 v/v/v), pH = 2.5 B: CH3OH/HCOOH (100:0.1 v/v) | 0.6 | 23 + 2 (CT) | |
| Catechins | Catechin Epicatechin | |||||
| γ | Carotenoids | Crocin I Crocin II Safranal | A: H2O B: CH3CN | 0.6 | 35 + 10 (CT) | |
| δ | Vitamin C | Ascorbic acid Dehydroascorbic acid | A: 5 mM C16H33N(CH3)3Br/50 mM KH2PO4, pH=2.5 B: CH3OH | 0.9 | 10 + 5 (CT) |
| Yield | Replacement corm | Quality category (ISO3632 [44]) | ||||||
|---|---|---|---|---|---|---|---|---|
| Treatment | Flower corm−1 (n) | Saffron flower−1 (mg) | Size (%) | Corm−1 (n) | Weight (%) | Picrocrocin (A1%1cm (λ257) | Safranal (A1%1cm (λ330) | Crocin (A1%1cm (λ440) |
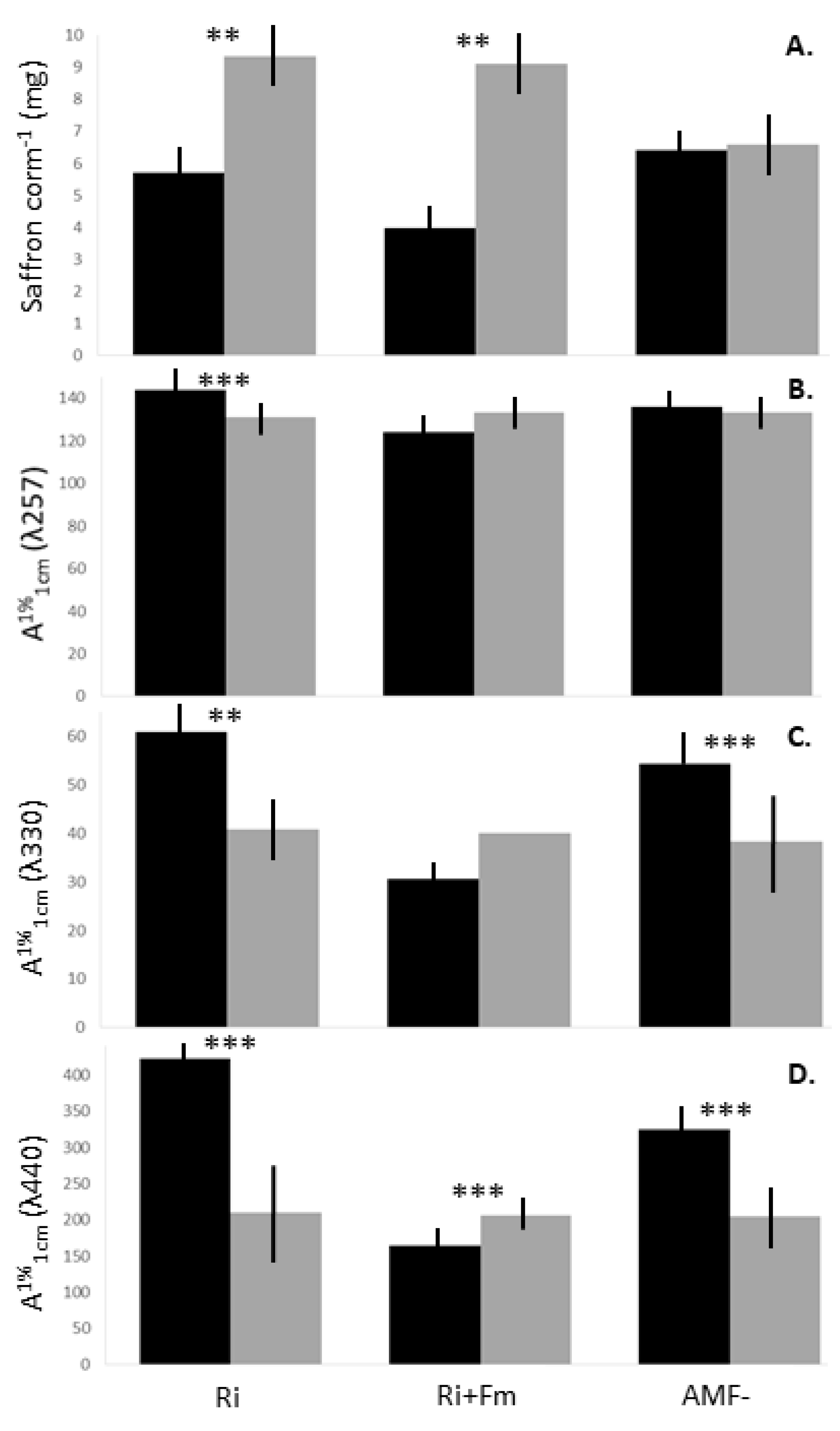
| Ri | 0.84 ± 0.62 | 6.8 ± 1.3 | 45.8 ± 4.6a | 2.71 ± 1.53 | 7.8 ± 5.6 | 143.8 ± 4.6(I)βa | 61.0 ± 5.3(I)a | 422.6 ± 4.1(I)a |
| Ri+Fm | 0.66 ± 0.60 | 6.0 ± 1.4 | 54.6 ± 6.2a | 2.25 ± 0.95 | 8.6 ± 3.8 | 124.3 ± 3.9(I)c | 30.7 ± 3.4(I)c | 164.2 ± 3.8(III)c |
| AMF- | 0.97 ± 0.53 | 6.6 ± 0.4 | 33.1 ± 6.8b | 2.63 ± 1.06 | 12.6 ± 5.1 | 135.9 ± 3.4(I)b | 54.3 ± 6.7(I)b | 324.7 ± 5.9(I)b |
| p | ns | ns | *** | ns | ns | *** | *** | *** |
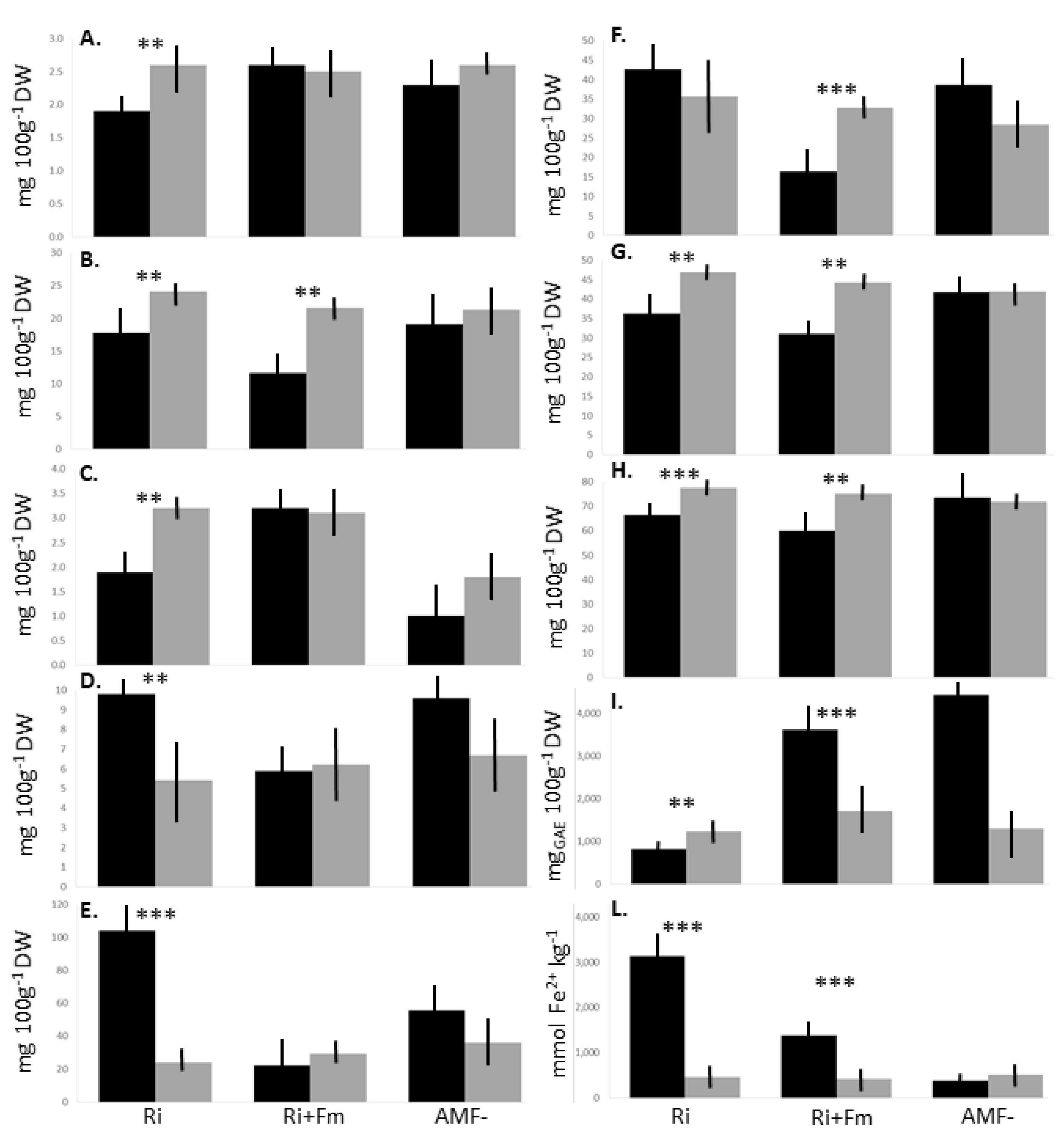
| Class | Compound (mg 100g−1 DW) | Ri | Ri+Fm | AMF- | p |
|---|---|---|---|---|---|
| Cinnamic acids | Coumaric acid | 23.4 ± 3.5 | 23.7 ± 2.6 | 23.7 ± 3.1 | ns |
| Flavonols | Isoquercitrin | 1.9 ± 0.3b | 2.6 ± 0.2a | 2.3 ± 0.3ab | ** |
| Quercitrin | 17.8 ± 4.6 | 11.6 ± 4.1 | 19.1 ± 3.6 | ns | |
| Benzoic acids | Gallic acid | 4.5 ± 1.5 | 5.1 ± 1.3 | 4.9 ± 1.4 | ns |
| Ellagic acid | 1.9 ± 0.5b | 3.2 ± 0.3a | 1.0 ± 0.4b | ** | |
| Catechins | Catechin | 1.9 ± 0.4 | 1.6 ± 0.3 | 1.8 ± 0.3 | ns |
| Epicatechin | 9.8 ± 2.9 | 5.9 ± 2.1 | 9.6 ± 2.5 | ns | |
| Carotenoids | Safranal | 4.0 ± 0.9 | 4.0 ± 1.2 | 4.0 ± 0.7 | ns |
| Crocin I | 104.2 ± 8.6a | 22.1 ± 6.5c | 55.5 ± 8.4b | *** | |
| Crocin II | 42.7 ± 9.6a | 16.4 ± 3.8b | 38.7 ± 12.9ab | ** | |
| Vitamin C | Dehydroascorbic acid | 28.8 ± 6.5 | 30.2 ± 4.1 | 31.8 ± 6.9 | ns |
| Ascorbic acid | 31.1 ± 9.5 | 36.3 ± 6.7 | 41.7 ± 4.8 | ns | |
| Total vitamin C | 59.9 ± 10.2 | 66.5 ± 5.9 | 73.6 ± 8.4 | ns | |
| TAC | Anthocyanin (mgC3G 100g−1 DW) | 640.7 ± 84.6b | 146.4 ± 29.8c | 1654.5 ± 68.4a | * |
| Methods | |||||
| TPC | Folin–Ciocalteu (mgGAE 100g−1 DW) | 816.5±152.7b | 3619.0±400.2a | 4445.4±450.2a | *** |
| AOA | FRAP (mmol Fe2+ kg−1 DW) | 3133.9±1524.3a | 1383.0±589.7ab | 379. 7±128.4b | ** |
| ABTS (μmolTE g−1 DW) | 5.4±0.8a | 3.6±0.4c | 4.5±0.7ab | ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caser, M.; Demasi, S.; Victorino, Í.M.M.; Donno, D.; Faccio, A.; Lumini, E.; Bianciotto, V.; Scariot, V. Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation. Agronomy 2019, 9, 232. https://doi.org/10.3390/agronomy9050232
Caser M, Demasi S, Victorino ÍMM, Donno D, Faccio A, Lumini E, Bianciotto V, Scariot V. Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation. Agronomy. 2019; 9(5):232. https://doi.org/10.3390/agronomy9050232
Chicago/Turabian StyleCaser, Matteo, Sonia Demasi, Íris Marisa Maxaieie Victorino, Dario Donno, Antonella Faccio, Erica Lumini, Valeria Bianciotto, and Valentina Scariot. 2019. "Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation" Agronomy 9, no. 5: 232. https://doi.org/10.3390/agronomy9050232
APA StyleCaser, M., Demasi, S., Victorino, Í. M. M., Donno, D., Faccio, A., Lumini, E., Bianciotto, V., & Scariot, V. (2019). Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation. Agronomy, 9(5), 232. https://doi.org/10.3390/agronomy9050232










