Effect of the Intensity of Weed Harrowing with Spike-Tooth Harrow in Barley-Pea Mixture on Yield and Mycobiota of Harvested Grains
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Experiment
- Control (without harrowing or herbicide application);
- HT (herbicide application, Chwastox Extra 300 SL 1.5 L per ha at the full tillering stage of barley 23–26; the growth stage of barley according to Biologische Bundesanstalt, Bundessortenamt und Chemische Industrie “BBCH” scale);
- Z-1-0 (one pass of spike-tooth harrow at the beginning of the barley tillering stage; BBCH 21);
- Z-0-1 (one pass of spike-tooth harrow at full tillering stage of barley; BBCH 23–26);
- Z-1-1 (one pass of spike-tooth harrow at the beginning of barley tillering stage and one pass at full tillering stage of barley; BBCH 21 + 23–26);
- Z-2-1 (two passes of spike-tooth harrow at the beginning of the barley tillering stage and one pass at full tillering stage of barley; BBCH 21 + 23–26);
- Z-2-2 (two passes of spike-tooth harrow at the beginning of the barley tillering stage and two passes at full tillering stage of barley; BBCH 21 + 23–26).
2.2. Assessment of the Yield of Pea and Barley
2.3. Measurement of Pea and Spring Barley Grains Moisture
2.4. Mycological Assessment of Pea and Barley Grains
2.5. Data Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Fungi Species | Treatment | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | HT | Z-1-0 | Z-0-1 | Z-1-1 | Z-2-1 | Z-2-2 | |||
| Spring barley | disinfected | Alternaria alternata (Fr.) Keissl. | 60 aC1 | 56 aD | 60 aC | 75 aA | 66 aB | 51 aE | 41 aF |
| Botrytis cinerea Pers. | 0 eA | 1 efA | 0 dA | 0 gA | 1 efA | 0 eA | 1 efA | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 0 eA | 1 efA | 0 dA | 0 gA | 1 efA | 0 eA | 1 efA | ||
| Cladosporium herbarum (Pers.) Link | 2 dB | 1 efBC | 1 dBC | 0 gC | 5 bA | 1 deBC | 5 dA | ||
| Drechslera avenae (Eidam) S. Ito | 0 eC | 1 efB | 0 dC | 0 gC | 1 efB | 0 eC | 6 dA | ||
| Epicoccum nigrum Link | 1 deDE | 1 efDE | 0 dE | 3 deBC | 5 bA | 2 cdCD | 2 eCD | ||
| Fusarium avenaceum (Fr.) Sacc. | 1 deD | 3 cdC | 4 cC | 7 bB | 4 bcC | 3 cC | 11 bA | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 10 bA | 1 efD | 4 cC | 4 cdC | 2 deD | 7 bB | 2 eD | ||
| Fusarium equiseti (Corda) Sacc. | 0 eC | 4 cA | 1 dB | 1 fgB | 0 fC | 1 deB | 1 efB | ||
| Fusarium graminearum Schwabe | 8 cAB | 7 bB | 8 bAB | 5 cC | 3 cdD | 7 bB | 9 cA | ||
| Fusarium oxysporum Schltdl. | 1 deAB | 2 deA | 1 dAB | 0 gB | 2 deA | 0 eB | 0 fB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 0 eC | 1 efB | 3 cA | 0 gC | 0 fC | 0 eC | 0 fC | ||
| Sclerotinia sclerotiorum (Lib.) de Bary | 0 eB | 0 fB | 0 dB | 1 fgA | 0 fB | 1 deA | 0 fB | ||
| Sordaria fimicola (Roberge ex Desm.) Ces. & De Not. | 1 deAB | 0 fB | 0 dB | 2 efA | 1 efAB | 2 cdA | 0 fB | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 71 aA | 48 aD | 65 aB | 49 aD | 55 aC | 49 aD | 45 aE | |
| Botrytis cinerea Pers. | 0 fB | 0 hB | 0 fB | 0 fB | 0 fB | 7 cdA | 0 gB | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 3 dB | 2 fgC | 0 fD | 0 fD | 0 fD | 0 gD | 5 eA | ||
| Cladosporium herbarum (Pers.) Link | 3 dA | 3 efA | 1 efB | 0 fB | 0 fB | 0 gB | 0 gB | ||
| Drechslera avenae (Eidam) S. Ito | 0 fE | 3 efD | 0 fE | 8 cB | 5 dC | 1 gE | 11 bA | ||
| Epicoccum nigrum Link | 5 cD | 18 bA | 13 cB | 9 cC | 17 bA | 10 bC | 9 cC | ||
| Fusarium avenaceum (Fr.) Sacc. | 2 deC | 5 dB | 1 efD | 9 cA | 5 dB | 4 fB | 4 efB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 0 fD | 4 deC | 6 dB | 0 fD | 9 cA | 8 cA | 9 cA | ||
| Fusarium equiseti (Corda) Sacc. | 1 efCD | 0 hD | 2 eBC | 2 eBC | 3 eB | 6 deA | 3 fB | ||
| Fusarium graminearum Schwabe | 12 bC | 8 cD | 15 bB | 18 bA | 1 fE | 4 fH | 7 dD | ||
| Fusarium oxysporum Schltdl. | 1 efB | 1 ghB | 0 fB | 0 fB | 4 deA | 5 efA | 1 gB | ||
| Mucor mucedo Fresen. | 1 efB | 0 hC | 0 fC | 0 fC | 0 fC | 0 gC | 3 fA | ||
| Penicillium chrysogenum Thom | 0 fB | 4 deA | 0 fB | 0 fB | 1 fB | 0 gB | 0 gB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 3 dC | 4 deBC | 0 fD | 6 dA | 0 fD | 5 efAB | 0 gF | ||
| Sclerotinia sclerotiorum (Lib.) de Bary | 0 fB | 0 hB | 1 efA | 0 fB | 0 fB | 0 gB | 0 gB | ||
| Sordaria fimicola (Roberge ex Desm.) Ces. & De Not. | 0 fC | 0 hC | 2 eB | 0 fC | 0 fC | 0 gC | 5 eA | ||
| Pea | disinfected | Alternaria alternata (Fr.) Keissl. | 24 aAB | 20 aBC | 29 aA | 17 aBC | 18 aBC | 28 aA | 15 aC |
| Aspergillus niger Tiegh. | 1 cA | 1 cA | 0 dB | 0 dB | 0 dB | 0 dB | 1 bcA | ||
| Botrytis cinerea Pers. | 0 cB | 0 cB | 0 dB | 3 cdA | 2 dAB | 0 dB | 2 bcAB | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 3 cAB | 0 cB | 2 cdAB | 3 cdAB | 3 cdAB | 7 cA | 1 bcB | ||
| Cladosporium herbarum (Pers.) Link | 2 cAB | 4 bcAB | 4 cAB | 0 dB | 7 bA | 0 dB | 5 bcAB | ||
| Epicoccum nigrum Link | 0 cC | 2 efAB | 1 cdC | 0 dC | 0 dC | 4 cdA | 0 cC | ||
| Fusarium avenaceum (Fr.) Sacc. | 0 cC | 0 cC | 0 dC | 3 cdBC | 0 dC | 13 bA | 6 bB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 3 cB | 8 bAB | 10 bA | 11 bA | 8 bAB | 2 dB | 2 bcB | ||
| Fusarium equiseti (Corda) Sacc. | 1 cB | 2 cAB | 0 dC | 4 cAB | 6 bcA | 4 cdAB | 2 bcAB | ||
| Fusarium graminearum Schwabe | 8 bAB | 5 bcABC | 10 bA | 3 cdBC | 1 dC | 1 dC | 1 bcC | ||
| Fusarium oxysporum Schltdl. | 0 cB | 0 cB | 0 dB | 0 dB | 0 dB | 0 dB | 1 bcA | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 3 cAB | 0 cB | 0 dB | 0 dB | 0 dB | 0 dB | 5 bcA | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 53 aD | 47 aE | 71 aB | 44 aF | 67 aC | 83 aA | 55 aD | |
| Aspergillus niger Tiegh. | 0 dB | 2 defA | 2 cdA | 1 cdAB | 1 cAB | 0 eB | 1 cdAB | ||
| Botrytis cinerea Pers. | 3 cdA | 0 fB | 0 dB | 0 dB | 0 cB | 2 deA | 0 dB | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 7 cBC | 2 defCD | 0 dD | 8 cBC | 0 cD | 12 cAB | 15 bA | ||
| Cladosporium herbarum (Pers.) Link | 38 bBC | 22 bC | 62 bA | 33 bBC | 59 bA | 47 bAB | 51 aAB | ||
| Epicoccum nigrum Link | 4 cdAB | 7 cdA | 6 cAB | 3 cdB | 5 cAB | 7 cdA | 3 cdB | ||
| Fusarium avenaceum (Fr.) Sacc. | 0 dB | 1 efA | 1 cdA | 0 dB | 0 cB | 1 eA | 0 dB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 1 cdC | 9 cA | 0 dC | 3 cdBC | 1 cC | 4 deABC | 7 cAB | ||
| Fusarium equiseti (Corda) Sacc. | 7 cA | 3 defAB | 0 dB | 6 cdAB | 1 cAB | 0 eB | 0 dB | ||
| Fusarium graminearum Schwabe | 3 cdA | 1 efB | 4 cdA | 0 dB | 0 cB | 3 deA | 0 dB | ||
| Fusarium oxysporum Schltdl. | 1 cdA | 0 fB | 0 dB | 0 dB | 0 cB | 0 eB | 1 cdA | ||
| Penicillium chrysogenum Thom | 0 dB | 0 fB | 0 dB | 0 dB | 0 cB | 1 eA | 0 dB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 1 cdAB | 6 cdeA | 0 dB | 4 dAB | 0 cB | 5 deAB | 1 cdAB | ||
| Sclerotinia sclerotiorum (Lib.) de Bary | 0 dB | 1 efAB | 4 cdA | 0 dB | 2 cAB | 0 eB | 2 cdAB | ||
| Fungi Species | Treatment | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | HT | Z-1-0 | Z-0-1 | Z-1-1 | Z-2-1 | Z-2-2 | |||
| Spring barley | disinfected | Alternaria alternata (Fr.) Keissl. | 62 aB1 | 60 aC | 52 aE | 78 aA | 56 aD | 48 aF | 42 aG |
| Botrytis cinerea Pers. | 0 fB | 0 eB | 0 eB | 0 eB | 0 fB | 0 eB | 8 cA | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 0 fC | 0 eC | 2 dB | 0 eC | 4 dA | 2 dB | 2 eB | ||
| Epicoccum nigrum Link | 0 fC | 0 eC | 0 eC | 1 deBC | 2 eB | 0 eC | 12 bA | ||
| Fusarium avenaceum (Fr.) Sacc. | 2 eD | 4 cC | 4 cC | 4 cC | 6 cB | 8 bA | 2 eD | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 8 bA | 0 eE | 0 eE | 2 dD | 4 dC | 0 eE | 6 dB | ||
| Fusarium equiseti (Corda) Sacc. | 0 fC | 4 cB | 9 bA | 0 eC | 0 fC | 0 eC | 0 fC | ||
| Fusarium graminearum Schwabe | 6 cC | 12 bA | 2 dD | 2 dD | 8 bB | 2 dD | 2 eD | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 0 fB | 2 dA | 0 eB | 0 eB | 0 fB | 0 eB | 0 fB | ||
| Sordaria fimicola (Roberge ex Desm.) Ces. & De Not. | 4 dC | 0 eD | 0 eD | 12 bA | 0 fD | 8 cB | 0 fD | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 82 aA | 56 aD | 64 aC | 64 aC | 76 aB | 55 aD | 46 aE | |
| Botrytis cinerea Pers. | 0 gB | 0 gB | 0 hB | 0 fB | 0 gB | 0 eB | 3 hA | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 4 eA | 4 eA | 0 hB | 0 fB | 0 gB | 0 eB | 0 iB | ||
| Cladosporium herbarum (Pers.) Link | 2 fB | 2 fB | 0 hC | 0 fC | 0 gC | 0 eC | 5 gA | ||
| Epicoccum nigrum Link | 0 gF | 1 fgE | 10 cA | 4 dD | 4 eD | 6 dC | 9 cdB | ||
| Fusarium avenaceum (Fr.) Sacc. | 8 cB | 12 cA | 4 fD | 4 dD | 12 bA | 6 dC | 7 efB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 0 gE | 6 dC | 6 eC | 2 eD | 8 cB | 12 bA | 6 fgC | ||
| Fusarium equiseti (Corda) Sacc. | 0 gE | 0 gE | 2 gD | 6 cC | 8 cB | 6 dC | 15 bA | ||
| Fusarium graminearum Schwabe | 16 bC | 18 bB | 18 bB | 34 bA | 6 dE | 10 cD | 0 iF | ||
| Fusarium oxysporum Schltdl. | 0 gB | 0 gB | 0 hB | 0 fB | 0 gB | 0 eB | 10 cA | ||
| Mucor mucedo Fresen. | 0 gB | 2 fA | 2 gA | 2 eA | 0 gB | 0 eB | 0 iB | ||
| Penicillium chrysogenum Thom | 0 gC | 6 dA | 0 hC | 2 eB | 0 gC | 0 eC | 0 iC | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 6 dB | 6 dB | 0 hC | 6 cB | 0 gC | 6 dB | 8 deA | ||
| Sordaria fimicola (Roberge ex Desm.) Ces. & De Not. | 0 gC | 0 gC | 8 dA | 0 fC | 2 fB | 0 eC | 0 iC | ||
| Pea | disinfected | Alternaria alternata (Fr.) Keissl. | 22 aAB | 13 aB | 32 aAB | 20 aB | 42 aA | 20 aB | 18 aB |
| Aspergillus niger Tiegh. | 0 cB | 0 cB | 4 bA | 1 cAB | 0 cB | 0 cB | 4 bcA | ||
| Botrytis cinerea Pers. | 2 bcAB | 0 cB | 0 bB | 0 cB | 0 cB | 0 cB | 4 bcA | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 0 cB | 2 cAB | 2 bAB | 2 cAB | 4 bcA | 2 cAB | 0 cB | ||
| Cladosporium herbarum (Pers.) Link | 6 bcAB | 1 cB | 8 bAB | 4 bcAB | 10 bAB | 12 bA | 4 bcAB | ||
| Epicoccum nigrum Link | 2 bcA | 0 cB | 2 bA | 0 cB | 0 cB | 0 cB | 0 cB | ||
| Fusarium avenaceum (Fr.) Sacc. | 4 bcB | 0 cB | 2 bB | 0 cB | 0 cB | 0 cB | 10 abA | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 0 cB | 2 cA | 0 bB | 0 cB | 0 cB | 0 cB | 0 cB | ||
| Fusarium equiseti (Corda) Sacc. | 2 bcAB | 5 bcA | 0 bB | 0 cB | 0 cB | 0 cB | 0 cB | ||
| Fusarium graminearum Schwabe | 4 bcBC | 12 abA | 8 bABC | 10 bAB | 4 bcBC | 2 cC | 2 bcC | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 8 bAB | 2 cBC | 0 bC | 3 cABC | 0 cC | 6 bcABC | 10 abA | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 88 aA | 52 aAB | 78 aAB | 50 aB | 72 bAB | 89 aA | 72 aAB | |
| Aspergillus niger Tiegh. | 0 cB | 2 cdA | 2 bA | 0 cB | 0 cB | 0 dB | 0 cB | ||
| Botrytis cinerea Pers. | 0 cB | 1 cdA | 0 bB | 0 cB | 0 cB | 0 dB | 0 cB | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 10 B | 0 dC | 0 bC | 0 cC | 0 cC | 15 cA | 12 bAB | ||
| Cladosporium herbarum (Pers.) Link | 69 bAB | 37 bC | 87 aA | 55 aB | 88 aA | 76 bA | 73 aAB | ||
| Epicoccum nigrum Link | 3 cA | 4 cdA | 5 bA | 4 cA | 8 cA | 6 cdA | 3 bcA | ||
| Fusarium avenaceum (Fr.) Sacc. | 4 cA | 0 dB | 4 bA | 0 cB | 0 cB | 0 dB | 0 cB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 0 cB | 0 dB | 0 bB | 0 cB | 0 cB | 0 dB | 2 bcA | ||
| Fusarium equiseti (Corda) Sacc. | 4 cA | 0 dC | 0 bC | 2 cB | 0 cC | 0 dC | 0 cC | ||
| Fusarium graminearum Schwabe | 0 cC | 0 dC | 2 bB | 0 cC | 6 cA | 0 dC | 0 cC | ||
| Penicillium chrysogenum Thom | 2 cAB | 0 dC | 0 bC | 0 cC | 1 cBC | 3 cdA | 0 cC | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 0 cA | 12 cB | 4 bD | 16 bA | 4 cD | 0 dA | 8 bcC | ||
| Fungi Species | Treatment | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | HT | Z-1-0 | Z-0-1 | Z-1-1 | Z-2-1 | Z-2-2 | |||
| Spring barley | disinfected | Alternaria alternata (Fr.) Keissl. | 61 aC1 | 60 aC | 76 aA | 66 aB | 58 aD | 48 aF | 52 aE |
| Drechslera avenae (Eidam) S. Ito | 10 dC | 12 bB | 4 dE | 8 cD | 10 cC | 12 bB | 18 bA | ||
| Epicoccum nigrum Link | 2 eC | 8 cA | 0 fD | 2 fC | 8 dA | 6 dB | 6 dB | ||
| Fusarium avenaceum (Fr.) Sacc. | 0 fC | 6 dB | 0 fC | 6 dB | 12 bA | 0 eC | 12 cA | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 12 cA | 0 fE | 6 cB | 4 eC | 2 fD | 12 bA | 4 eC | ||
| Fusarium graminearum Schwabe | 14 bB | 8 cD | 12 bC | 16 bA | 8 dD | 8 cD | 2 fE | ||
| Fusarium oxysporum Schltdl. | 0 fC | 2 eB | 0 fC | 0 gC | 4 eA | 0 eC | 0 gC | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 0 fB | 0 fB | 2 eA | 0 gB | 0 gB | 0 eB | 0 gB | ||
| Sclerotinia sclerotiorum (Lib.) de Bary | 0 fB | 2 eA | 0 fB | 0 gB | 0 gB | 0 eB | 0 gB | ||
| Sordaria fimicola (Roberge ex Desm.) Ces. & De Not. | 0 fB | 2 fA | 0 fB | 0 gB | 0 gB | 0 eB | 0 gB | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 54 aB | 28 bF | 62 aA | 44 aD | 34 aE | 47 aC | 44 aD | |
| Drechslera avenae (Eidam) S. Ito | 8 dE | 22 cC | 14 cD | 24 bB | 24 cB | 14 cD | 38 bA | ||
| Epicoccum nigrum Link | 12 cF | 32 aA | 14 cE | 20 cC | 26 bB | 26 bB | 16 cD | ||
| Fusarium avenaceum (Fr.) Sacc. | 0 gC | 0 hC | 0 eC | 6 dA | 4 eB | 6 dA | 0 fC | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 2 fE | 6 fC | 8 dB | 4 eD | 2 fE | 0 fF | 14 dA | ||
| Fusarium equiseti (Corda) Sacc. | 4 eA | 4 gA | 0 eB | 0 gB | 0 gB | 4 eA | 0 fB | ||
| Fusarium graminearum Schwabe | 18 bA | 12 dB | 18 bA | 2 fE | 2 fE | 6 dD | 10 eC | ||
| Fusarium oxysporum Schltdl. | 0 gC | 0 hC | 0 eC | 2 fB | 4 eA | 0 fC | 0 fC | ||
| Penicillium chrysogenum Thom | 0 gB | 2 hA | 0 eB | 0 gB | 0 gB | 0 fB | 0 fB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 3 efC | 8 eB | 0 eD | 4 eC | 14 dA | 0 fD | 0 fD | ||
| Pea | disinfected | Alternaria alternata (Fr.) Keissl. | 20 aBC | 20 bBC | 22 bB | 16 bCD | 8 cE | 30 bA | 14 aD |
| Botrytis cinerea Pers. | 0 fD | 2 cdC | 0 fD | 4 deB | 8 cA | 0 dD | 4 dB | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 0 fB | 2 cdA | 0 fB | 2 efA | 0 fB | 0 dB | 0 eB | ||
| Cladosporium herbarum (Pers.) Link | 0 fB | 0 eB | 2 eB | 0 fB | 0 fB | 0 dB | 4 dA | ||
| Fusarium avenaceum (Fr.) Sacc. | 0 fC | 0 eC | 0 fC | 2 efA | 0 fC | 0 dC | 1 eB | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 12 cC | 24 aB | 32 aA | 32 aA | 25 aB | 34 aA | 12 abC | ||
| Fusarium equiseti (Corda) Sacc. | 4 eDE | 3 cE | 6 cCD | 12 cA | 10 bAB | 8 cBC | 10 bcAB | ||
| Fusarium graminearum Schwabe | 16 bA | 3 cC | 6 cB | 6 dB | 4 dBC | 0 dD | 4 dBC | ||
| Fusarium oxysporum Schltdl. | 0 fB | 1 deAB | 0 fB | 0 fB | 2 eA | 0 dB | 1 eAB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 10 dA | 2 cdE | 4 dD | 6 dC | 4 dD | 0 dF | 8 cB | ||
| non-disinfected | Alternaria alternata (Fr.) Keissl. | 39 aF | 54 aD | 80 aB | 44 aE | 74 aC | 86 aA | 52 aD | |
| Aspergillus niger Tiegh. | 0 gB | 0 fB | 0 dB | 2 deA | 0 dB | 0 dB | 0 eB | ||
| Botrytis cinerea Pers. | 7 deA | 0 fC | 0 dC | 1 deBC | 0 dC | 2 dB | 0 eC | ||
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 2 efgC | 0 fD | 0 dD | 8 cA | 4 dB | 0 dD | 8 cA | ||
| Cladosporium herbarum (Pers.) Link | 26 bDE | 9 cE | 58 bA | 30 bCD | 46 bABC | 34 bBCD | 52 aAB | ||
| Epicoccum nigrum Link | 3 defgB | 3 deB | 7 cA | 0 eC | 0 dC | 3 dB | 0 eC | ||
| Fusarium avenaceum (Fr.) Sacc. | 1 fgC | 0 fD | 0 dD | 0 eD | 2 dB | 4 dA | 0 eD | ||
| Fusarium culmorum (W.G. Sm.) Sacc. | 15 cB | 22 bA | 4 cdE | 10 cD | 10 cD | 12 cCD | 14 bBC | ||
| Fusarium equiseti (Corda) Sacc. | 8 dA | 0 fE | 4 cdC | 6 cdB | 2 dD | 4 dC | 4 cdeC | ||
| Fusarium graminearum Schwabe | 6 defA | 0 fC | 0 dC | 0 eC | 0 dC | 2 dB | 0 eC | ||
| Fusarium oxysporum Schltdl. | 0 gC | 4 dA | 0 dC | 2 deB | 0 dC | 0 dC | 2 deB | ||
| Rhizopus stolonifer (Ehrenb.) Vuill. | 1 fgC | 8 cA | 0 dD | 0 eD | 0 dD | 4 dB | 0 eD | ||
| Sclerotinia sclerotiorum (Lib.) de Bary | 1 fgC | 0 fC | 4 cdB | 1 deC | 0 dC | 0 dC | 6 cdA | ||
| Trichoderma harzianum Rifai | 0 gB | 2 eA | 0 dB | 0 eB | 0 dB | 0 dB | 0 eB | ||
References
- Ecobichon, D.J. Our changing perspectives on benefit and risks of pesticides: A historical overview. Neurotoxicology 2000, 21, 211–218. [Google Scholar] [PubMed]
- Lejman, A.; Ogórek, R.; Sobkowicz, P. The effect of mechanical weed control in barley–pea mixture on colonization of barley grain by fungi. Part 1. Pol. J. Environ. Stud. 2015, 24, 141–149. [Google Scholar] [CrossRef]
- Lejman, A.; Ogórek, R.; Sobkowicz, P. The effect of mechanical weed control in barley–pea mixture on colonization of pea seeds by fungi. Part 2. Pol. J. Environ. Stud. 2015, 26, 2485–2492. [Google Scholar] [CrossRef]
- Aktar, W.M.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdiscip. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef] [PubMed]
- WHO. Geneva: World Health Organization, Public Health Impact of Pesticides Used in Agriculture; WHO/UNEP Working Group: Geneva, Switzerland, 1990; p. 88. [Google Scholar]
- Environews Forum. Killer environment. EHP 1999, 107, A62. [Google Scholar] [CrossRef]
- Bradbery, S.M.; Watt, B.E.; Proudfoot, A.T.; Vale, J.A. Mechanisms of toxicity, clinical features, and management of acute chlorophenoxy herbicide poisoning: a review. Clin. Toxicol. 2000, 38, 111–122. [Google Scholar] [CrossRef]
- Wally, O.; Punaj, Z.K. Genetic engineering for increasing fungal and bacterial disease resistance in crop plants. GM crops 2010, 1, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Bakhsh, A.; Khabbazi, S.D.; Baloch, F.S.; Demirel, U.; Çalişkan, M.E.; Hatipoğlu, R.; Özcan, S.; Özkan, H. Insect-resistant transgenic crops: Eetrospect and challenges. Turk. J. Aric. For. 2015, 39, 531–548. [Google Scholar] [CrossRef]
- Denner, F.D.N.; Millard, C.P.; Wehner, F.C. Effect of soil solarisation and mouldboard ploughing black dot of potato, caused by Colletotrichum coccodes. Potato Res. 2000, 43, 195–201. [Google Scholar] [CrossRef]
- Malézieux, E.; Crozat, Y.; Dupraz, C.; Laurans, M.; Makowski, D.; Ozier-Lafontaine, H.; Rapidel1, B.; de Tourdonnet, S.; Valantin-Morison, M. Mixing plant species in cropping systems: Concepts, tools and models. A review. Agron. Sustain. 2009, 29, 43–62. [Google Scholar] [CrossRef]
- Paulsen, H.M.; Schochow, M.; Ulber, B.; Kühne, S.; Rahmann, G. Mixed cropping systems for biological control of weeds and pests in organic oilseed crops. Asp. Appl. Biol. 2006, 79, 215–220. [Google Scholar]
- Sobkowicz, P. Competition between triticale and field beans in additive intercrops. Plant Soil Environ. 2006, 52, 47–56. [Google Scholar] [CrossRef]
- Kang, Z.; Buchenauer, H.; Huang, L.; Han, Q.; Zhang, C.H. Cytological and immunocytochemical studies on responses of wheat spikes of the resistant Chinese cv. Sumai 3 and the susceptible cv. Xiaoyan 22 to infection by Fusarium graminearum. Eur. J. Plant Pathol. 2008, 120, 383–396. [Google Scholar] [CrossRef]
- Pasanen, A.L.; Rautiala, S.; Kasanen, J.P.; Raunio, P.; Rantamäki, J.; Kalliokoski, P. The relationship between measured moisture conditions and fungal concentrations in water-damaged building materials. Indoor Air 2000, 10, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.; Svenningsen, T. Selective weed harrowing in cereals. Biol. Agric. Hortic. 1995, 12, 29–46. [Google Scholar] [CrossRef]
- Malschi, D.; Ivaş, A.D.; Valean, A.M.; Cheţan, F.; Tritean, N.; Cheţan, C.; Pataki, I. Current Importance of wheat pests in the cultural technologies of soil no-tillage conservative system and of antierosional agroforestry curtains system, in Transylvania. ProEnviron. Promediu 2015, 8, 159–169. [Google Scholar]
- Rasmussen, J.; Ascard, J. Weed Control in Organic Farming Systems; Department for Environment, Food and Rural Affairs: Lundon, UK, 1995; pp. 49–67.
- Kurstjens, D.A.G.; Perdock, U.D.; Goense, D. Selective uprooting by weed harrowing on sandy soils. Weed Res. 2000, 40, 431–447. [Google Scholar] [CrossRef]
- Rasmussen, I.A. The effect of sowing date, stale seedbed row width and mechanical weed control on weeds and yields of organic winter wheat. Weed Res. 2004, 44, 12–20. [Google Scholar] [CrossRef]
- Rasmussen, J.; Kurtzmann, J.I.; Jensen, A. Tolerance of competitive spring barley cultivars to weed harrowing. Weed Res. 2004, 44, 446–452. [Google Scholar] [CrossRef]
- Lejman, A. Weed Control in Mixture of Spring Barley and Field Pea. Ph.D. Thesis, Wrocław University of Environmental and Life Sciences, Wrocław, Poland, 2015; 108p. [Google Scholar]
- Wnuk, A.; Górny, A.G.; Bocianowski, J.; Kozak, M. Visualizing harvest index in crops. CBCS 2013, 8, 48–59. [Google Scholar]
- Dugan, F.M. The Identification of Fungi: An Illustrated Introduction with Keys, Glossary, and Guide to Literature; Amer Phytopathological Society: St. Paul, MN, USA, 2006. [Google Scholar]
- Simmons, E.G. Alternaria: An identification manual; CBS Fungal Biodiversity Centre: Utrecht, The Netherlands, 2008. [Google Scholar]
- Watanabe, T. Pictorial Atlas of Soil and Seed Fungi: Morphologies of Cultured Fungi and Key to Species, 3rd ed.; CRC Press: London, UK, 2010. [Google Scholar]
- Bliss, C.T. The method of probits. Science 1934, 79, 38–39. [Google Scholar] [CrossRef] [PubMed]
- Cerkauskas, R.F.; Dhingra, D.; Sinclair, J.B.; Asmus, G. Amaranthus spinosus, Leonotis nepetaefolia, and Leonurus sibiricus: New hosts of Phomopsis spp. in Brazil. Plant Dis. 1983, 67, 821–824. [Google Scholar] [CrossRef]
- Helbig, J.B.; Carroll, R.B. Dicotyledonous weeds as a source of Fusarium oxysporum pathogenic on soybean. Plant Dis. 1984, 68, 694–696. [Google Scholar] [CrossRef]
- Baturo, A. Effect of biopreparations on the health of grain of spring barley (Hordeum vulgare) in organic system. Phytopatholog 2009, 53, 19–30. [Google Scholar]
- Gleń, K.; Boligłowa, E.; Gospodarek, J. Fungi colonizing broad bean seeds depending on protection method. Pol. J. Agron. 2013, 12, 9–16. [Google Scholar]
- Woudenberg, J.H.; Groenewald, J.Z.; Binder, M.; Crous, P.W. Alternaria redefined. Stud. Mycol. 2013, 75, 171–212. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Spieksma, F.T.M. Aerobiologic and clinical aspects of mould allergy in Europe. Allergy 1995, 50, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Zare, L. The casual agent of barley black point disease in certified seed loads in Iran. IJACS 2013, 5, 332–335. [Google Scholar]
- Rademacher, B. Mechanische Unkrautbekämpfung im Getreide. Landtechnik 1962, 17, 194–198. [Google Scholar]
- Velykis, A.; Maiksteniene, S.; Arlauskiene, A.; Kristaponyte, I.; Satkus, A. Mechanical weed control in organically grown spring oat and field pea crops. Agron. Res. 2009, 7, 542–547. [Google Scholar]
- Padro, G.; Cirujeda, A.; Aibar, J.; Cavero, J.; Zaragoza, C. Weed harrowing in winter cereal under semi-arid conditions. Span. J. Agric. Res. 2008, 6, 661–670. [Google Scholar]
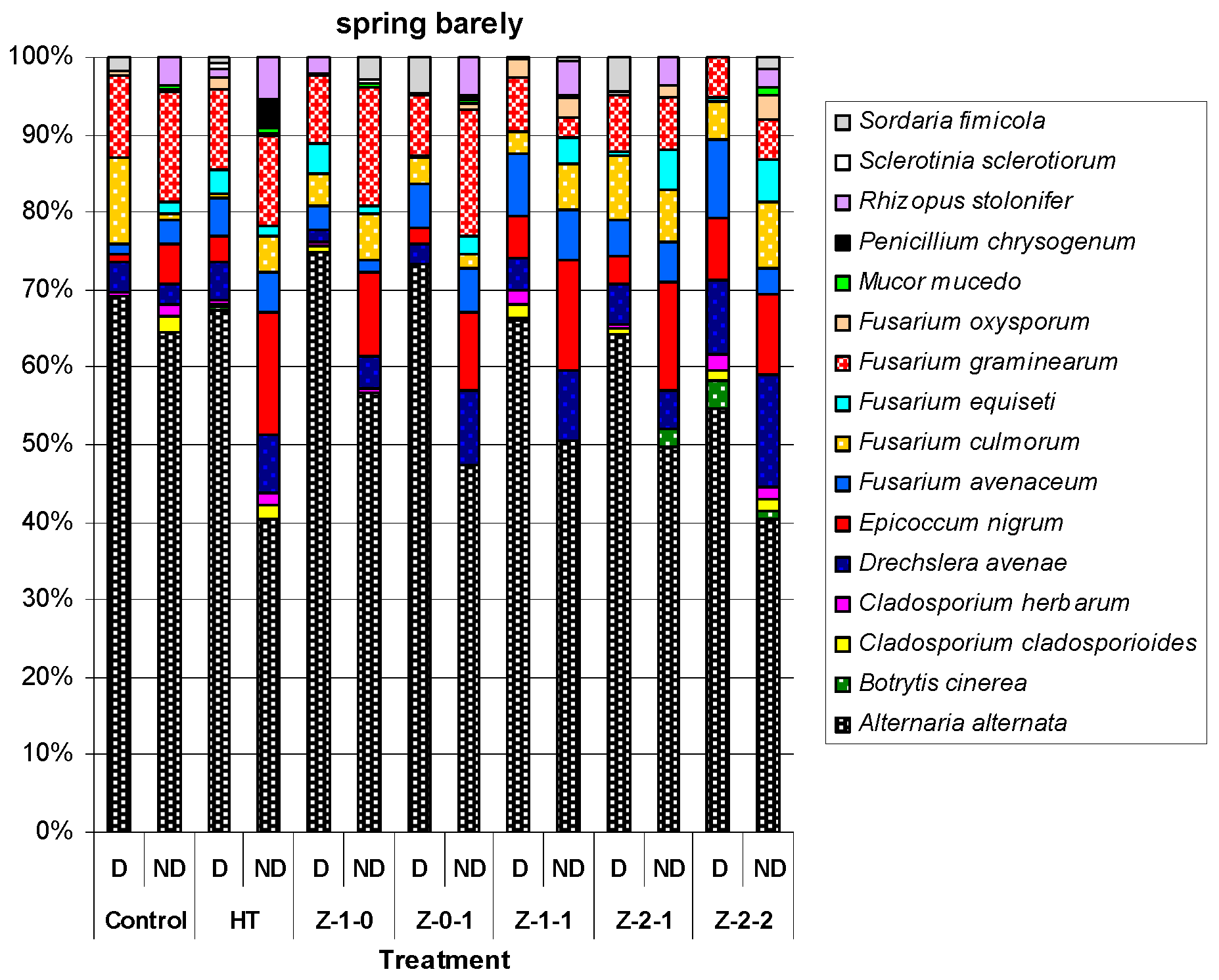
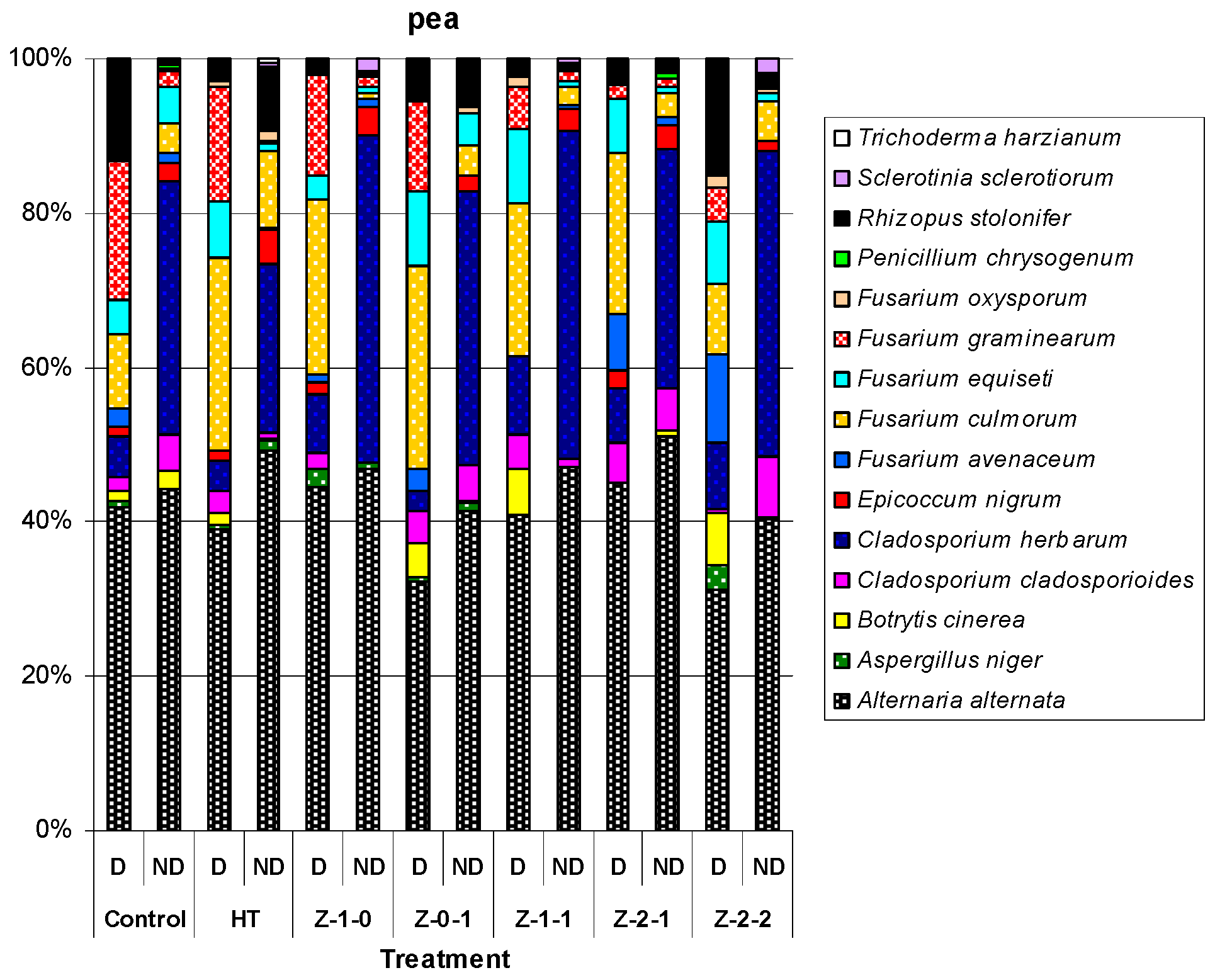
| Treatment | 2010 | 2011 | 2012 | Mean for 2010–2012 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Yield | Harvest Index | ||||||||||
| Spring barley | Control | 141 | aAB1 | 256 | abA | 132 | aB | 176.33 | a | 0.49 | a |
| HT | 154 | aB | 322 | aA | 241 | aAB | 239.00 | a | 0.56 | a | |
| Z-1-0 | 147 | aB | 270 | abA | 174 | aAB | 197.00 | a | 0.52 | a | |
| Z-0-1 | 179 | aA | 226 | abA | 218 | aA | 207.67 | a | 0.52 | a | |
| Z-1-1 | 151 | aA | 268 | abA | 213 | aA | 210.67 | a | 0.52 | a | |
| Z-2-1 | 160 | aA | 184 | bA | 212 | aA | 185.33 | a | 0.51 | a | |
| Z-2-2 | 167 | aA | 213 | abA | 213 | aA | 197.67 | a | 0.52 | a | |
| Pea | Control | 111 | aA | 102 | aA | 89 | bA | 100.67 | a | 0.47 | a |
| HT | 89 | aB | 112 | aB | 216 | aA | 139.00 | a | 0.47 | a | |
| Z-1-0 | 101 | aA | 86 | aA | 93 | bA | 93.33 | a | 0.47 | a | |
| Z-0-1 | 72 | aA | 104 | aA | 92 | bA | 89.33 | a | 0.47 | a | |
| Z-1-1 | 70 | aA | 78 | aA | 105 | bA | 84.33 | a | 0.45 | a | |
| Z-2-1 | 83 | aA | 111 | aA | 71 | bA | 88.33 | a | 0.47 | a | |
| Z-2-2 | 93 | aA | 147 | aA | 101 | bA | 113.67 | a | 0.51 | a | |
| Treatment | 2010 | 2011 | 2012 | Mean for 2010–2012 | |||||
|---|---|---|---|---|---|---|---|---|---|
| Spring barley | Control | 4.43 | aAB1 | 5.93 | aA | 2.34 | aB | 4.23 | a |
| HT | 4.00 | aA | 5.09 | aA | 2.39 | aA | 3.83 | a | |
| Z-1-0 | 5.20 | aAB | 6.89 | aA | 2.75 | aB | 4.97 | a | |
| Z-0-1 | 5.30 | aB | 6.47 | aA | 3.45 | aC | 5.07 | a | |
| Z-1-1 | 4.86 | aA | 4.80 | aA | 4.12 | aA | 4.59 | a | |
| Z-2-1 | 4.45 | aA | 4.64 | aA | 3.25 | aA | 4.11 | a | |
| Z-2-2 | 4.89 | aA | 5.06 | aA | 3.84 | aA | 4.60 | a | |
| Mean | 4.93 | A | 5.89 | A | 3.24 | A | — | ||
| Pea | Control | 6.32 | aAB | 4.10 | aB | 8.04 | aA | 6.15 | a |
| HT | 7.98 | aA | 5.27 | aB | 10.11 | aA | 7.79 | a | |
| Z-1-0 | 7.80 | aA | 7.10 | aA | 8.22 | aA | 7.71 | a | |
| Z-0-1 | 7.89 | aA | 7.37 | aA | 7.67 | aA | 7.64 | a | |
| Z-1-1 | 6.56 | aA | 5.09 | aA | 6.89 | aA | 6.18 | a | |
| Z-2-1 | 6.63 | aA | 5.28 | aB | 7.21 | aA | 6.37 | a | |
| Z-2-2 | 6.45 | aA | 4.99 | aA | 7.09 | aA | 6.18 | a | |
| Mean | 7.09 | A | 5.60 | A | 7.89 | A | — | ||
| Treatment | 2010 | 2011 | 2012 | Mean for 2010–2012 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Spring barley | disinfected | Control | 84 | bB1 | 82 | bB | 99 | aA | 88.3 | a |
| HT | 79 | bcB | 82 | bB | 100 | aA | 87.0 | a | ||
| Z-1-0 | 82 | bB | 69 | dC | 100 | aA | 83.7 | a | ||
| Z-0-1 | 98 | aA | 99 | aA | 102 | aA | 99.7 | a | ||
| Z-1-1 | 91 | abB | 80 | bC | 102 | aA | 91.0 | a | ||
| Z-2-1 | 75 | cB | 68 | dB | 86 | cA | 76.3 | a | ||
| Z-2-2 | 79 | bcB | 74 | cC | 94 | bA | 82.3 | a | ||
| non-disinfected | Control | 102 | abB | 118 | bA | 101 | eB | 107.0 | a | |
| HT | 100 | bB | 113 | cA | 114 | abcA | 109.0 | a | ||
| Z-1-0 | 106 | aB | 114 | cA | 116 | abA | 112.0 | a | ||
| Z-0-1 | 101 | bB | 124 | aA | 106 | cdeB | 110.3 | a | ||
| Z-1-1 | 100 | bB | 116 | bcA | 110 | bcdAB | 108.7 | a | ||
| Z-2-1 | 99 | bA | 101 | eA | 103 | eA | 101.0 | a | ||
| Z-2-2 | 102 | abC | 109 | dB | 122 | aA | 111.0 | a | ||
| Pea | disinfected | Control | 45 | cC | 50 | bB | 62 | cA | 52.3 | a |
| HT | 42 | deB | 37 | dC | 57 | dA | 45.3 | a | ||
| Z-1-0 | 56 | bB | 58 | aB | 72 | bA | 62.0 | a | ||
| Z-0-1 | 44 | cdB | 40 | cdC | 80 | aA | 54.7 | a | ||
| Z-1-1 | 45 | cB | 60 | aA | 61 | cdA | 55.3 | a | ||
| Z-2-1 | 59 | aB | 42 | cC | 72 | bA | 57.7 | a | ||
| Z-2-2 | 41 | eC | 52 | bB | 58 | dA | 50.3 | a | ||
| non-disinfected | Control | 118 | bcB | 180 | aA | 109 | bcB | 135.7 | ab | |
| HT | 101 | cA | 108 | bA | 102 | bcA | 103.7 | b | ||
| Z-1-0 | 150 | abA | 182 | aA | 157 | aA | 163.0 | ab | ||
| Z-0-1 | 102 | cA | 127 | bA | 104 | cA | 111.0 | ab | ||
| Z-1-1 | 136 | abcB | 179 | aA | 138 | abcAB | 151.0 | ab | ||
| Z-2-1 | 165 | aA | 189 | aA | 151 | abA | 168.3 | a | ||
| Z-2-2 | 136 | abcA | 170 | aA | 138 | abcA | 148.0 | ab | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogórek, R.; Lejman, A.; Sobkowicz, P. Effect of the Intensity of Weed Harrowing with Spike-Tooth Harrow in Barley-Pea Mixture on Yield and Mycobiota of Harvested Grains. Agronomy 2019, 9, 103. https://doi.org/10.3390/agronomy9020103
Ogórek R, Lejman A, Sobkowicz P. Effect of the Intensity of Weed Harrowing with Spike-Tooth Harrow in Barley-Pea Mixture on Yield and Mycobiota of Harvested Grains. Agronomy. 2019; 9(2):103. https://doi.org/10.3390/agronomy9020103
Chicago/Turabian StyleOgórek, Rafał, Agnieszka Lejman, and Piotr Sobkowicz. 2019. "Effect of the Intensity of Weed Harrowing with Spike-Tooth Harrow in Barley-Pea Mixture on Yield and Mycobiota of Harvested Grains" Agronomy 9, no. 2: 103. https://doi.org/10.3390/agronomy9020103
APA StyleOgórek, R., Lejman, A., & Sobkowicz, P. (2019). Effect of the Intensity of Weed Harrowing with Spike-Tooth Harrow in Barley-Pea Mixture on Yield and Mycobiota of Harvested Grains. Agronomy, 9(2), 103. https://doi.org/10.3390/agronomy9020103






