Bacillus subtilis CBR05 for Tomato (Solanum lycopersicum) Fruits in South Korea as a Novel Plant Probiotic Bacterium (PPB): Implications from Total Phenolics, Flavonoids, and Carotenoids Content for Fruit Quality
Abstract
1. Introduction
2. Material and Methods
2.1. Chemicals and Reagents
2.2. Bacterial Strain and Culture Conditions
2.3. Plant and Growth Conditions
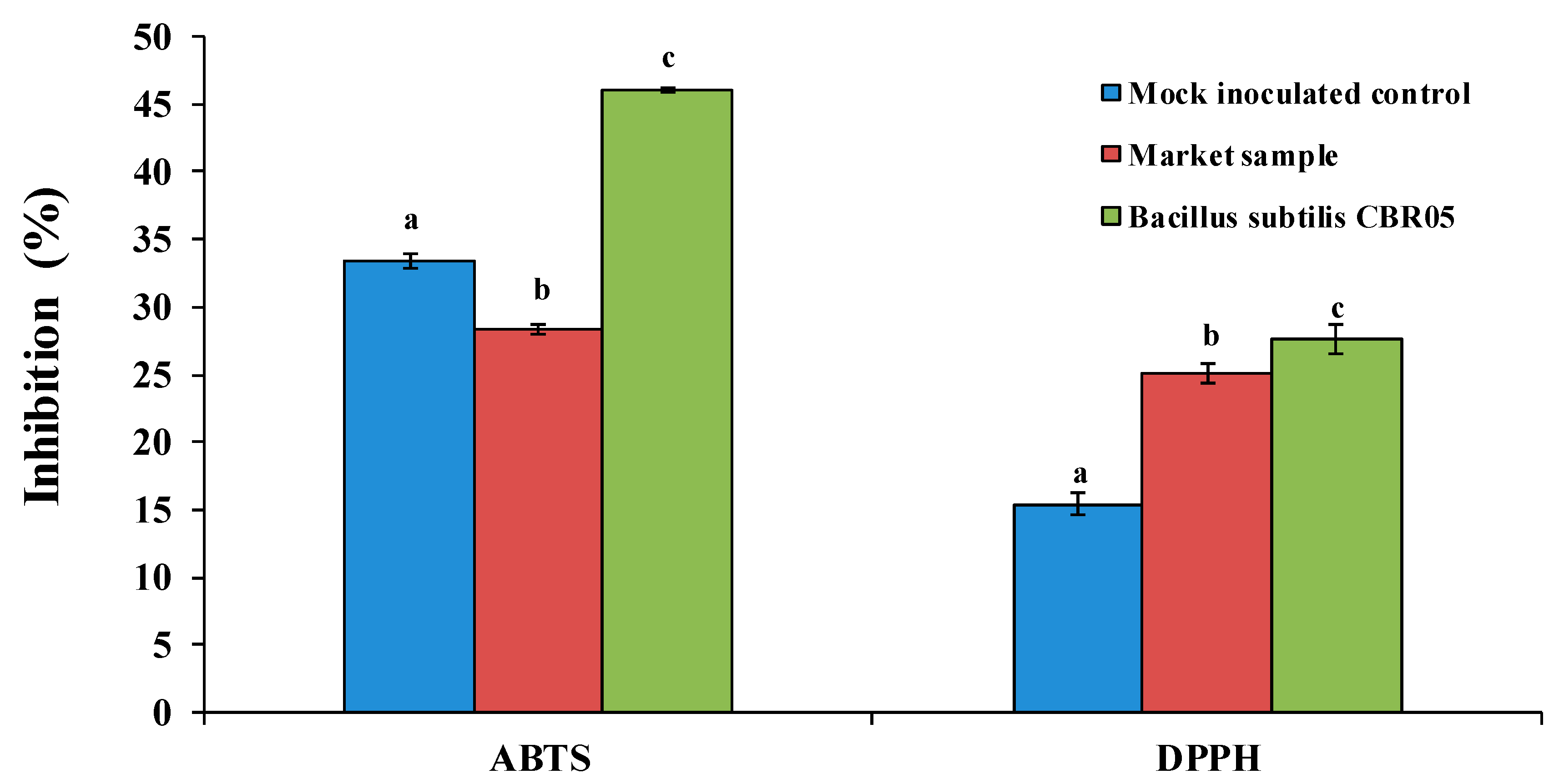
2.4. Antioxidant Assays
2.4.1. DPPH Assay
2.4.2. ABTS (2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) Assay
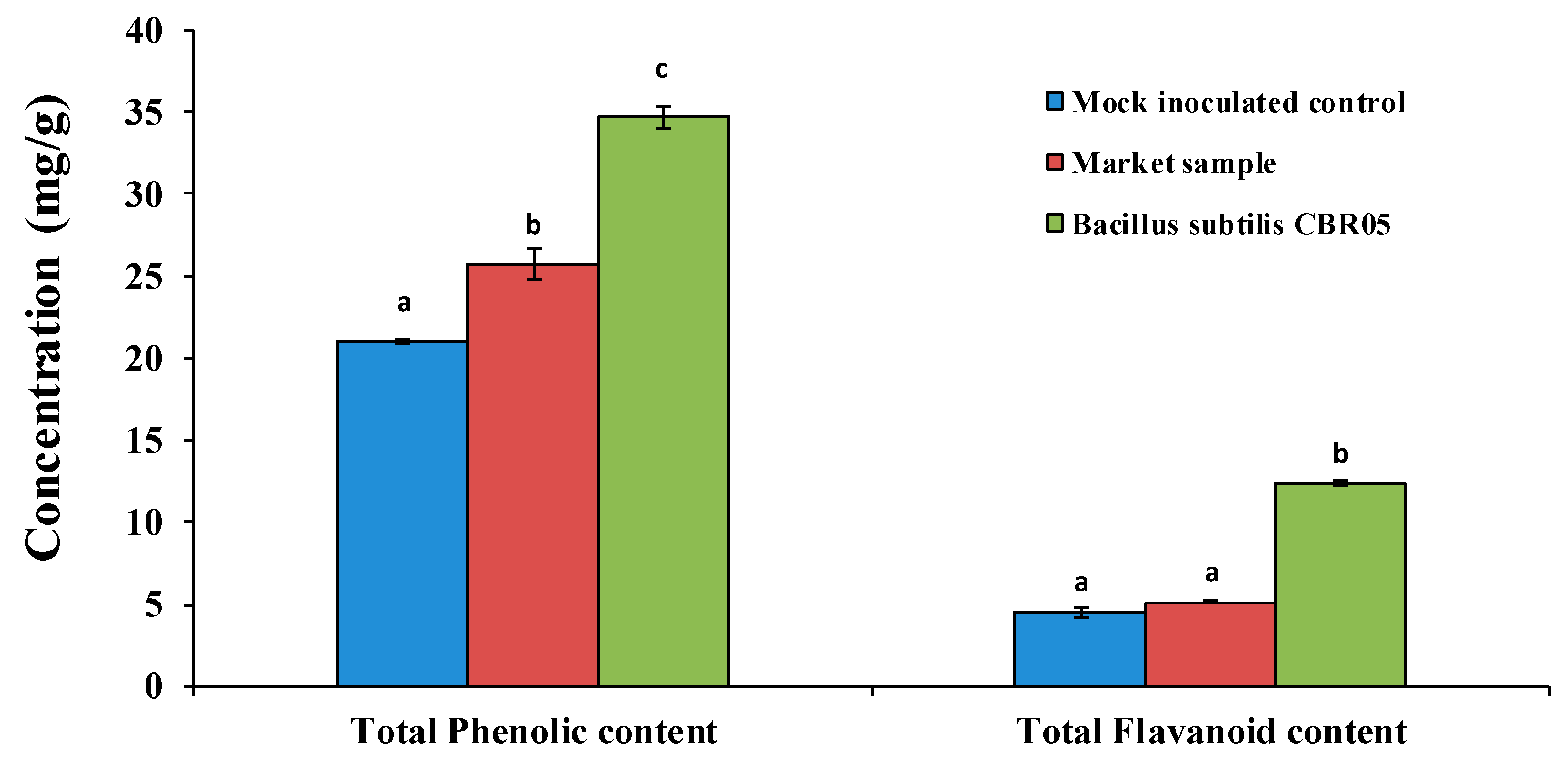
2.5. Determination of Total Phenolic Contents
2.6. Determination of Total Flavonoid Contents
2.7. Extraction and Quantification of Carotenoids
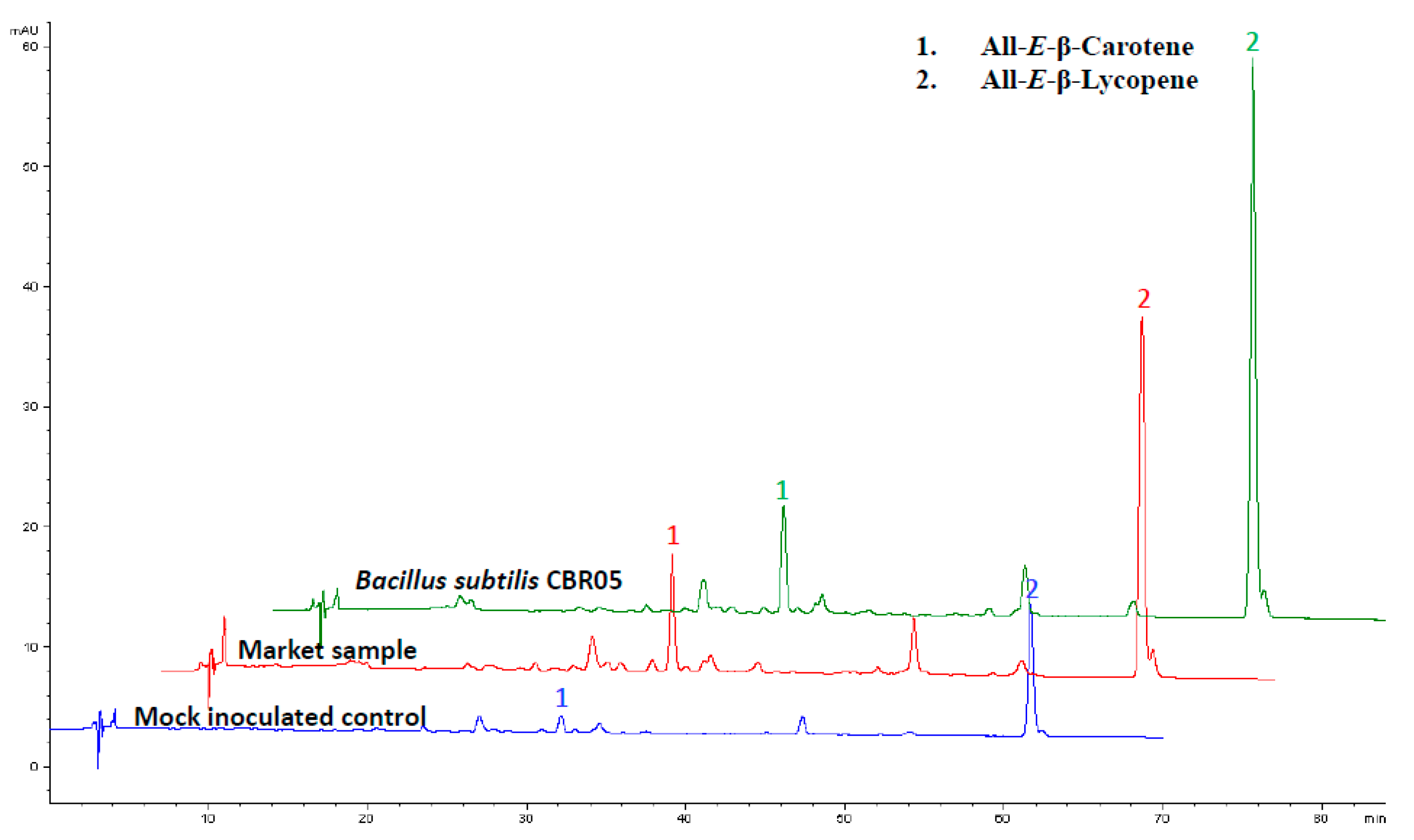
2.8. HPLC Analysis
2.9. Statistics
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Choi, S.H.; Kim, D.S.; Kozukue, N.; Kim, H.J.; Nishitani, Y.; Mizuno, M.; Levin, C.E.; Friedman, M. Protein, free amino acid, phenolic, β-carotene, and lycopene content, and antioxidative and cancer cell inhibitory effects of 12 greenhouse-grown commercial cherry tomato varieties. J. Food Compos. Anal. 2014, 34, 115–127. [Google Scholar] [CrossRef]
- Franceschi, S.; Bidoli, E.; La Vecchia, C.; Talamini, R.; D’Avanzo, B.; Negri, E. Tomatoes and risk of digestive-tract cancers. Int. J. Cancer 1994, 59, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Zamany, A.J.; Keum, Y.S. Ripening improves the content of carotenoid, alpha-tocopherol, and polyunsaturated fatty acids in tomato (Solanum lycopersicum L.) fruits. 3 Biotech 2017, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Klee, H.J.; Giovannoni, J.J. Genetics and control of tomato fruit ripening and quality attributes. Annu. Rev. Genet. 2011, 45, 41–59. [Google Scholar] [CrossRef]
- Alexander, L.; Grierson, D. Ethylene biosynthesis and action in tomato: A model for climacteric fruit ripening. J. Exp. Bot. 2002, 53, 2039–2055. [Google Scholar] [CrossRef]
- Konozy, E.H.E.; Rogniaux, H.; Causse, M.; Faurobert, M. Proteomic analysis of tomato (Solanum lycopersicum) secretome. J. Plant Res. 2012, 126, 251–266. [Google Scholar] [CrossRef]
- Barrett, D.M.; Weakley, C.; Diaz, J.V.; Watnik, M. Qualitative and nutritional differences in processing tomatoes grown under commercial organic and conventional production system. J. Food Sci. 2007, 72, 441–451. [Google Scholar] [CrossRef]
- Erba, D.; Casiraghi, M.C.; Ribas-Agustí, A.; Cáceres, R.; Marfà, O.; Castellari, M. Nutritional value of tomatoes (Solanum lycopersicum L.) grown in greenhouse by different agronomic techniques. J. Food Compos. Anal. 2013, 31, 245–251. [Google Scholar] [CrossRef]
- Dumas, Y.; Dadomo, M.; Di Lucca, G.; Grolier, P. Effects of environmental factors and agricultural techniques on antioxidant content of tomatoes. J. Sci. Food Agric. 2003, 83, 369–382. [Google Scholar] [CrossRef]
- Wang, X.; Xing, Y. Evaluation of the effects of irrigation and fertilization on tomato fruit yield and quality: A principal component analysis. Sci. Rep. 2017, 7, 350. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.; Whitfield, S.; Challinor, A. Crop modelling: Towards locally relevantand climate-informed adaptation. Clim. Chang. 2018, 147, 475–489. [Google Scholar] [CrossRef]
- Challinor, A.J.; Müller, C.; Asseng, S.; Deva, C.; Nicklin, K.J.; Wallach, D.; Vanuytrecht, E.; Whitfield, S.; Ramirez-Villegas, J.; Koehler, A.K. Improving the use of crop models for risk assessment and climate change adaptation. Agric. Syst. 2018, 159, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, M.M.; Gatta, G.; Cappelli, G.; Gagliardi, A.; Donatelli, M.; Fanchini, D.; De Nart, D.; Mongiano, G.; Bregaglio, S. Identifying the most promising agronomic adaptation strategies for the tomato growing systems in Southern Italy via simulation modeling. Eur. J. Agron. 2019, 111, 125937. [Google Scholar] [CrossRef]
- Wang, F.; Kang, S.; Du, T.; Li, F.; Qiu, R. Determination of comprehensive quality index for tomato and its response to different irrigation treatments. Agric. Water Manag. 2011, 98, 1228–1238. [Google Scholar] [CrossRef]
- Shi, S.; Chen, D.; Fu, J.; Liu, L.; Wei, Y.; Shu, B.; Li, W.; Liang, Q.; Liu, C. Comprehensive evaluation of fruit quality traits in longan ‘Fengliduo’ × ‘Dawuyuan’ sexual progenies. Sci. Hortic. 2015, 192, 54–59. [Google Scholar] [CrossRef]
- Wang, C.; Gu, F.; Chen, J.; Yang, H.; Jiang, J.; Du, T.; Zhang, J. Assessing the response of yield and comprehensive fruit quality of tomato grown in greenhouse to deficit irrigation and nitrogen application strategies. Agric. Water Manag. 2015, 161, 9–19. [Google Scholar] [CrossRef]
- Luo, H.; Li, F. Tomato yield, quality and water use efficiency under different drip fertigation strategies. Sci. Hortic. 2018, 235, 181–188. [Google Scholar] [CrossRef]
- Mader, P.; Fliessbach, A.; Dubois, D.; Gunst, L.; Fried, P.; Niggli, U. Soil fertility and biodiversity in organic farming. Science 2002, 296, 1694–1697. [Google Scholar] [CrossRef] [PubMed]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Mena-Violante, H.G.; Olalde-Portugal, V. Alteration of tomato fruit quality by root inoculation with plant growth-promoting rhizobacteria (PGPR): Bacillus subtilis BEB-13bs. Sci. Hortic. 2007, 113, 103–106. [Google Scholar] [CrossRef]
- Zehnder, G.W.; Murphy, I.F.; Sikora, E.J.; Kloepper, J.W. Application to rhizobacteria for induced resistance. Eur. J. Plant Pathol. 2001, 107, 39–50. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Chun, S.C. Induction of defence-related enzymes in tomato (Solanum lycopersicum) plants treated with Bacillus subtilis CBR05 against Xanthomonas campestris pv. vesicatoria. Biocontrol Sci. Technol. 2016, 26, 1366–1378. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Chun, S.C. Expression of PR-protein genes and induction of defense-related enzymes by Bacillus subtilis CBR05 in tomato (Solanum lycopersicum) plants challenged with Erwinia carotovora subsp. carotovora. Biosci. Biotechnol. Biochem. 2016, 80, 2277–2283. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R.; Penrose, D.M.; Li, J. A model for the lowering of plant ethylene concentrations by plant growth promoting bacteria. J. Theor. Biol. 1998, 190, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Verheul, M.; Maessen, H.; Grimstad, O.S. Optimizing a year-round cultivation system of tomato under artificial light. Acta Hortic. 2012, 956, 389–394. [Google Scholar] [CrossRef]
- Moco, S.; Capanoglu, E.; Tikunov, Y.; Bino, R.J.; Boyacioglu, D.; Hall, R.D.; Vervoort, J.; De Vos, R.C. Tissue specialization at the metabolite level is perceived during the development of tomato fruit. J. Exp. Bot. 2007, 58, 4131–4146. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Ramos, A.; Rivero, R.; Victoria, M.C.; Visozo, A.; Piloto, J.; Garcia, A. Assessment of mutagenicity in Parthenium hysterophorus L. J. Ethnopharmacol. 2001, 77, 25–30. [Google Scholar] [CrossRef]
- Chen, L.; Xin, X.; Yuan, Q.; Su, D.; Liu, W. Phytochemical properties and antioxidant capacities of various colored berries. J. Sci. Food Agric. 2014, 94, 180–188. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. A Guide to Carotenoid Analysis in Foods; ILsi Press: Washington, DC, USA, 2001. [Google Scholar]
- Saini, R.K.; Nile, S.H.; Park, S.W. Carotenoids from fruits and vegetables: Chemistry, analysis, occurrence, bioavailability and biological activities. Food Res. Int. 2015, 76, 735–750. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; Valadez-Blanco, R.; Salas-Coronado, R.; Sustaita-Rivera, F.; Hernández-Carlos, B.; García-Ortega, S.; Santos-Sánchez, N.F. Effect of nitrogen fertilization and Bacillus licheniformis biofertilizer addition on the antioxidants compounds and antioxidant activity of greenhouse cultivated tomato fruits (Solanum lycopersicum L. var. Sheva). Sci. Hortic. 2016, 201, 338–345. [Google Scholar] [CrossRef]
- Baset Mia, M.A.; Shamsuddin, Z.H. Nitrogen fixation and transportation by rhizobacteria: A scenario of rice and banana. Int. J. Bot. 2010, 6, 235–242. [Google Scholar]
- Bar-Ness, E.; Chen, Y.; Hadar, Y.; Marchner, H.; Romheld, V. Siderophores of Pseudomonas putida as an iron source for dicot and monocot plants. Plant Soil 1991, 130, 231–241. [Google Scholar] [CrossRef]
- Lucas, G.J.A.; Probanza, A.; Ramos, B.; Palomino, M.R.; Gutierrez Mañero, F.J. Effect of inoculation of Bacillus licheniformis on tomato and pepper. Agronomie 2004, 24, 169–176. [Google Scholar]
- Martí, R.; Roselló, S.; Cebolla-Cornejo, J. Tomato as a source of carotenoids and polyphenols targeted to cancer prevention. Cancer 2016, 8, 58. [Google Scholar] [CrossRef]
- Ronen, G.; Cohen, M.; Zamir, D.; Hirschberg, J. Regulation of carotenoid biosynthesis during tomato fruit development: Expression of the gene for lycopene epsilon-cyclase is downregulated during ripening and is elevated in the mutant Delta. Plant J. Cell Mol. Biol. 1999, 17, 341–351. [Google Scholar] [CrossRef]
- Su, L.; Diretto, G.; Purgatto, E.; Danoun, S.; Zouine, M.; Li, Z.; Roustan, J.P.; Bouzayen, M.; Giuliano, G.; Chervin, C. Carotenoid accumulation during tomato fruit ripening is modulated by the auxin–ethylene balance. BMC Plant Biol. 2015, 15, 114. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Lv, Q. DFT study on the quenching mechanism of singlet oxygen by lycopene. RSC Adv. 2016, 6, 98498–98505. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Belachew, S.T.; Yoon, E.; Chun, S.C. Expression of β-1,3-glucanase (GLU) and phenylalanine ammonia-lyase (PAL) genes and their enzymes in tomato plants induced after treatment with Bacillus subtilis CBR05 against Xanthomonas campestris pv. vesicatoria. J. Gen. Plant Pathol. 2016, 83, 7–13. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Manivannan, P.; Chun, S.C. Bacillus subtilis CBR05 induces Vitamin B6 biosynthesis in tomato through the de novo pathway in contributing disease resistance against Xanthomonas campestris pv. vesicatoria. Sci. Rep. 2019, 9, 6495. [Google Scholar] [CrossRef]
- Jiménez-Gómez, A.; Flores-Félix, J.D.; García-Fraile, P.; Mateos, P.F.; Menéndez, E.; Velázquez, E.; Rivas, R. Probiotic activities of rhizobium laguerreae on growth and quality of spinach. Sci. Rep. 2018, 8, 295. [Google Scholar] [CrossRef]
- Martínez-Hidalgo, P.; García, J.M.; Pozo, M.J. Induced systemic resistance against Botrytis cinerea by Micromonospora strainsisolated from root nodules. Front. Microbiol. 2015, 6, 922. [Google Scholar] [CrossRef] [PubMed]
- Flores-Felix, J.D.; Silva, L.R.; Rivera, L.P.; Marcos-Garcia, M.; Garcia-Fraile, P.; Martinez-Molina, E.; Mateos, P.F.; Velazquez, E.; Andrade, P.; Rivas, R. Plants probiotics as a tool to produce highly functional fruits: The case of Phyllobacterium and vitamin C in strawberries. PLoS ONE 2015, 10, e0122281. [Google Scholar] [CrossRef]
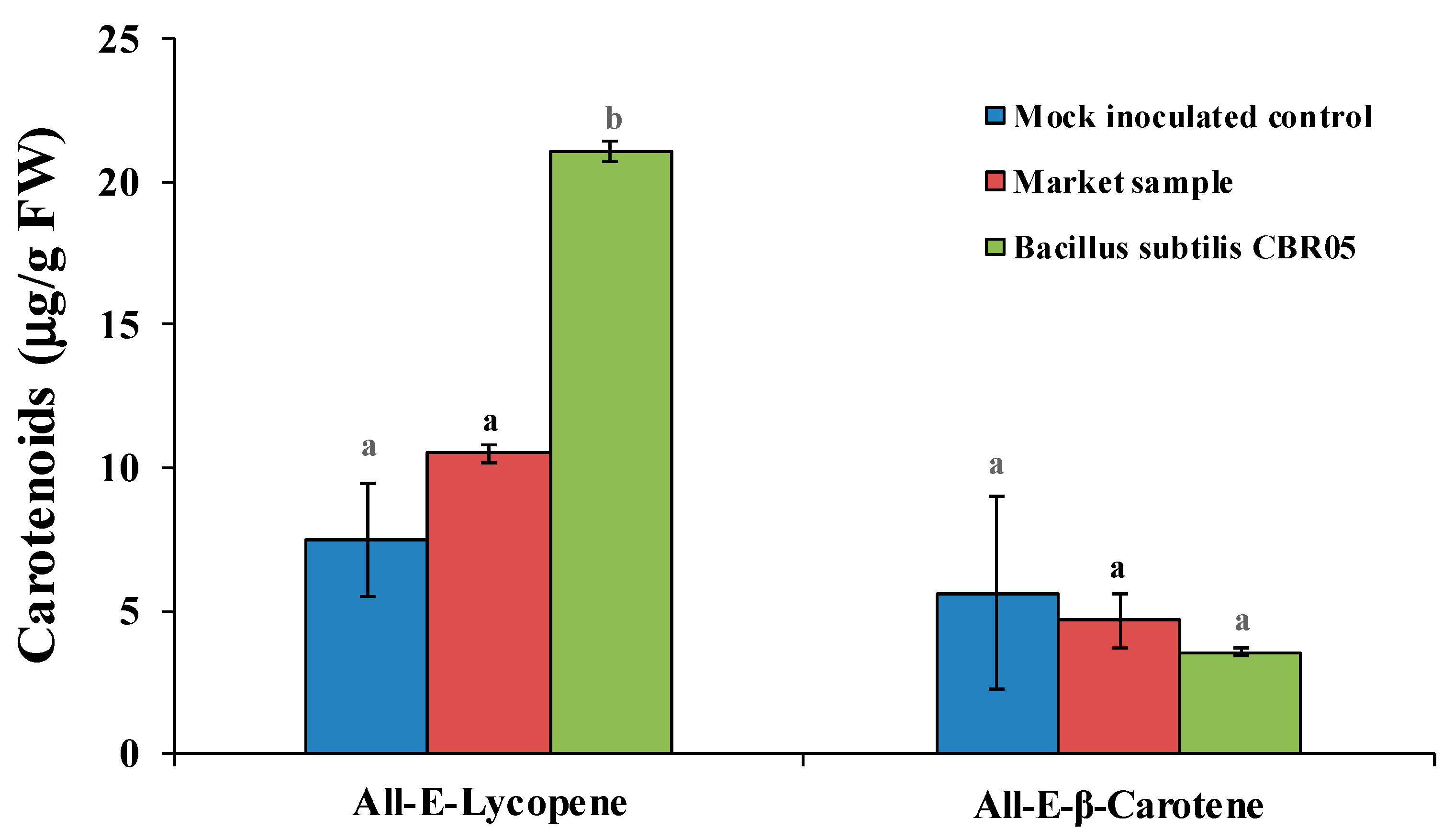
| Treatment | Carotenoids | Content (μg/g FW) |
|---|---|---|
| Mock inoculated control | All-E-β-Carotene | 5.63 ± 0.28 a |
| All-E-Lycopene | 7.48 ± 1.96 a | |
| Market tomato | All-E-β-Carotene | 4.65 ± 0.96 a |
| All-E-Lycopene | 10.51 ± 3.34 a | |
| Bacillus subtilis CBR05 | All-E-β-Carotene | 3.53 ± 0.12 a |
| All-E-Lycopene | 21.08 ± 0.32 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chandrasekaran, M.; Chun, S.C.; Oh, J.W.; Paramasivan, M.; Saini, R.K.; Sahayarayan, J.J. Bacillus subtilis CBR05 for Tomato (Solanum lycopersicum) Fruits in South Korea as a Novel Plant Probiotic Bacterium (PPB): Implications from Total Phenolics, Flavonoids, and Carotenoids Content for Fruit Quality. Agronomy 2019, 9, 838. https://doi.org/10.3390/agronomy9120838
Chandrasekaran M, Chun SC, Oh JW, Paramasivan M, Saini RK, Sahayarayan JJ. Bacillus subtilis CBR05 for Tomato (Solanum lycopersicum) Fruits in South Korea as a Novel Plant Probiotic Bacterium (PPB): Implications from Total Phenolics, Flavonoids, and Carotenoids Content for Fruit Quality. Agronomy. 2019; 9(12):838. https://doi.org/10.3390/agronomy9120838
Chicago/Turabian StyleChandrasekaran, Murugesan, Se Chul Chun, Jae Wook Oh, Manivannan Paramasivan, Ramesh Kumar Saini, and Jesudoss Joseph Sahayarayan. 2019. "Bacillus subtilis CBR05 for Tomato (Solanum lycopersicum) Fruits in South Korea as a Novel Plant Probiotic Bacterium (PPB): Implications from Total Phenolics, Flavonoids, and Carotenoids Content for Fruit Quality" Agronomy 9, no. 12: 838. https://doi.org/10.3390/agronomy9120838
APA StyleChandrasekaran, M., Chun, S. C., Oh, J. W., Paramasivan, M., Saini, R. K., & Sahayarayan, J. J. (2019). Bacillus subtilis CBR05 for Tomato (Solanum lycopersicum) Fruits in South Korea as a Novel Plant Probiotic Bacterium (PPB): Implications from Total Phenolics, Flavonoids, and Carotenoids Content for Fruit Quality. Agronomy, 9(12), 838. https://doi.org/10.3390/agronomy9120838








