Screening for Changes on Iris germanica L. Rhizomes Following Inoculation with Arbuscular Mycorrhiza Using Fourier Transform Infrared Spectroscopy
Abstract
1. Introduction
- Test potential of FT-IR to screen for variation in rhizome composition;
- Identify if rhizome quality markers display response associated with treatments;
- Define fingerprint ranges that could be used to confirm presence of main metabolites.
2. Materials and Methods
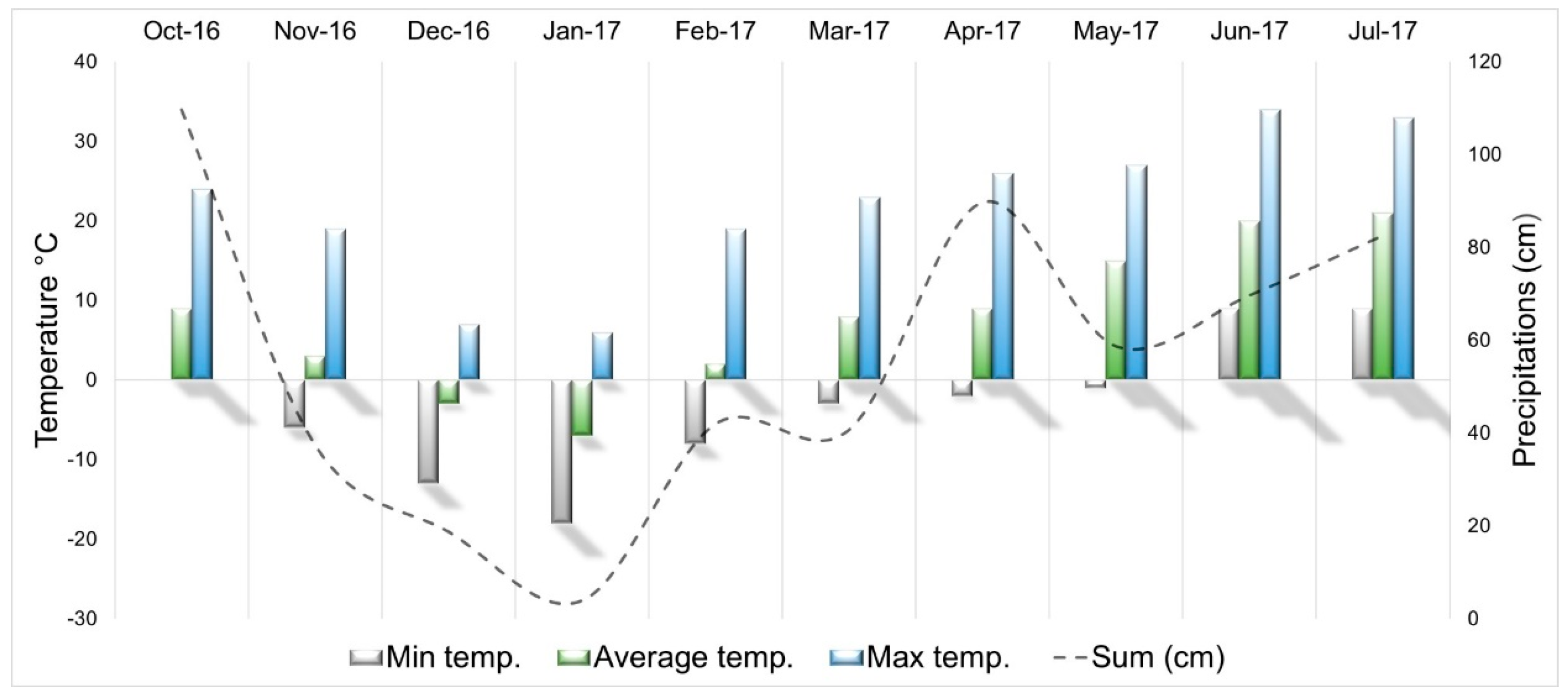
2.1. Location and Conditions
2.2. Treatments
2.3. Assays
2.3.1. AM Analysis
2.3.2. FT-IR Analysis
3. Results and Discussion
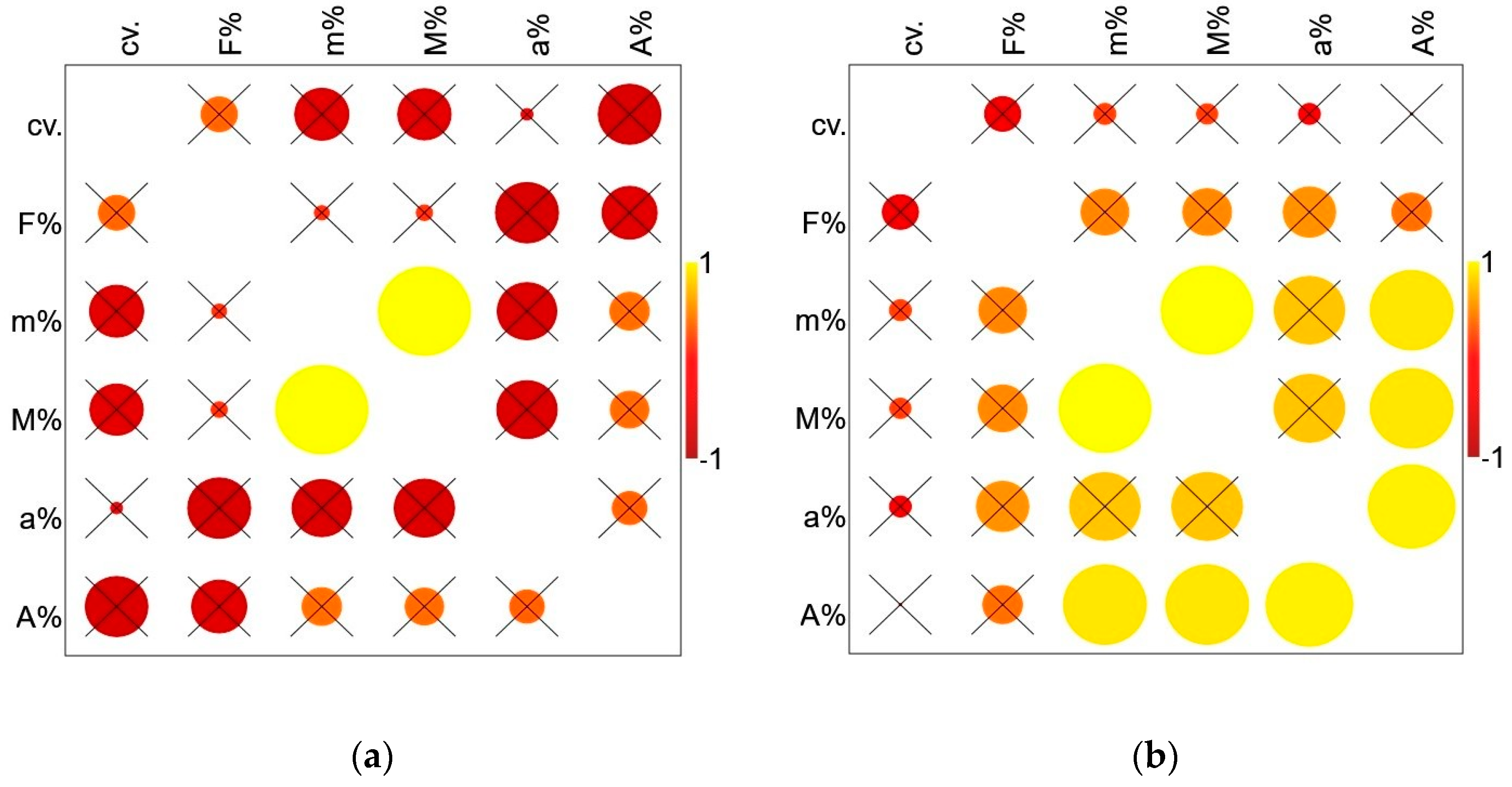
3.1. Root AM Colonization
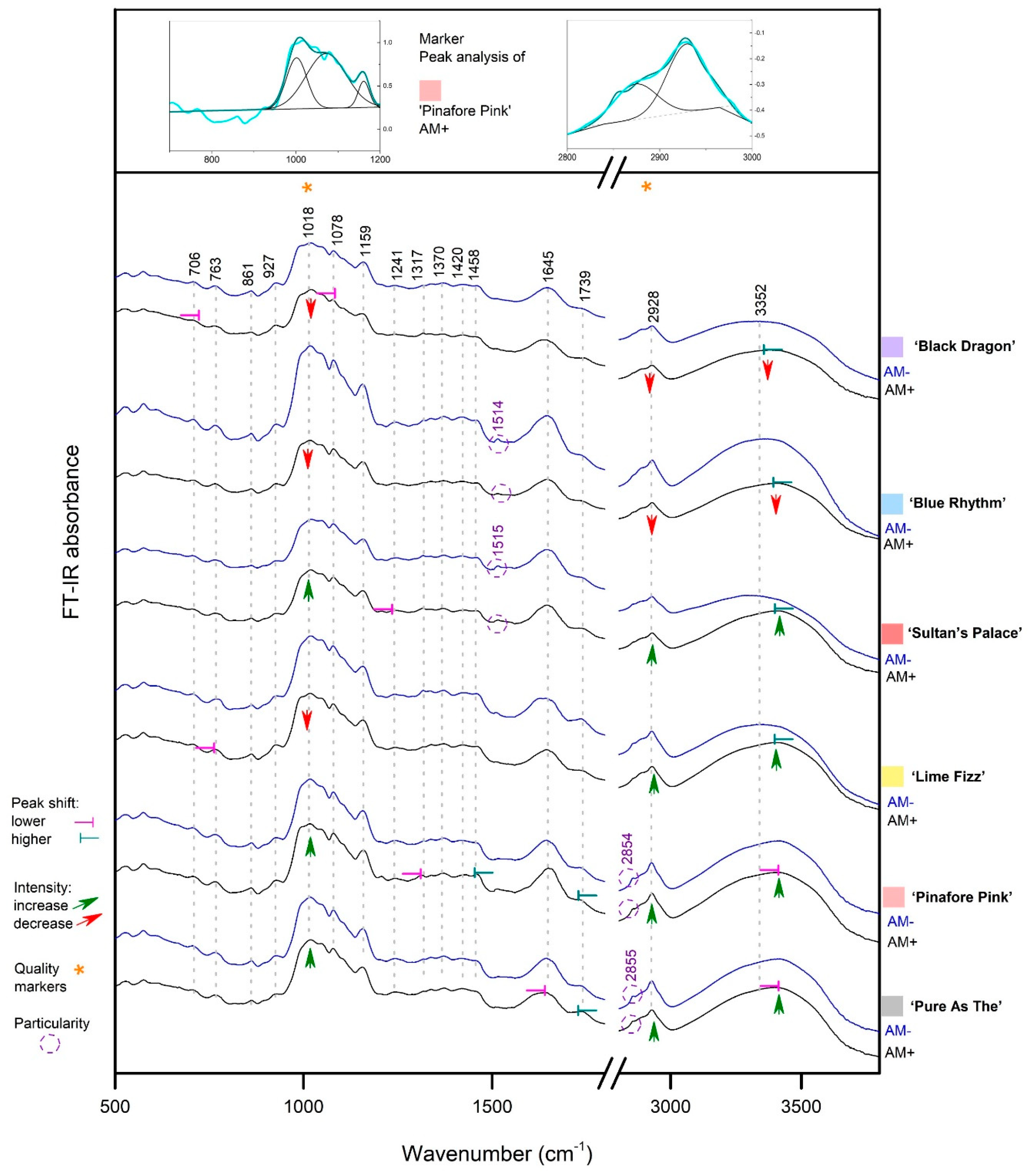
3.2. Rhizome FT-IR Spectra
3.2.1. Starch Presence in Rhizomes
3.2.2. Myristic Acid in Rhizomes
3.2.3. Other Metabolites in Rhizomes
3.2.4. Significance of the Findings
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crișan, I.; Vidican, R.; Stoian, V.; Stoie, A. Wild Iris spp. from Romanian meadows and their importance for ornamental plant breeding. Rom. J. Grassl. Forage Crops 2017, 16, 21–32. [Google Scholar]
- Crișan, I.; Cantor, M. New perspectives on medicinal properties and uses of Iris sp. Hop Med. Plant. 2016, 24, 24–36. [Google Scholar]
- Lim, T.K. Iris x germanica. In Edible Medicinal and Non-medicinal Plants; Modified stems, roots and bulbs; Springer: Cham, Switzerland, 2016; Volume 11, pp. 27–40. [Google Scholar] [CrossRef]
- Kara, N.; Gürbüzer, G. Effect of harvest times on rhizoma yield, essential oil content and composition in Iris germanica L. species. TURJAF 2019, 7, 707–713. [Google Scholar] [CrossRef]
- DeBaggio, T.; Tucker, A.O. The Encyclopedia of Herbs: A Comprehensive Reference to Herbs of Flavor and Fragrance; Timber Press: Portland, ME, USA, 2009. [Google Scholar]
- Belletti, G.; Fani, E.; Marescotti, A.; Scaramuzzi, S. The role of traditional products in the valorisation of marginal rural areas: The case of Iris pallida. SJAR 2013, 4, 11–23. [Google Scholar] [CrossRef]
- Firmin, L.; Courtois, D.; Pétiard, V.; Ehret, C.; Lerch, K. Evaluation of the natural variability in irone content and selection of Iris sp. for perfume production. HortScience 1998, 33, 1046–1047. [Google Scholar] [CrossRef]
- Roger, B.; Fernandez, X.; Jeannot, V.; Chahboun, J. An alternative method for irones quantification in iris rhizomes using headspace solid-phase microextraction. Phytochem. Anal. 2010, 21, 483–488. [Google Scholar] [CrossRef]
- Price of Orris Butter. Available online: https://hermitageoils.com/product/orris-butter-essential-oil-13-irones/ (accessed on 26 August 2019).
- Meyer, C.J.; Peterson, C.A.; Steudle, E. Permeability of Iris germanica’s multiseriate exodermis to water, NaCl, and ethanol. J. Exp. Bot. 2011, 62, 1911–1926. [Google Scholar] [CrossRef]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and drought stresses in crops and approaches for their mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef]
- Engel, R.; Szabo, K.; Abranko, L.; Rendes, K.; Füzy, A.; Takács, T. Effect of arbuscular mycorrhizal fungi on the growth and polyphenol profile of marjoram, lemon balm, and marigold. J. Agric. Food Chem. 2016, 64, 3733–3742. [Google Scholar] [CrossRef]
- Scagel, C.F.; Lee, J. Phenolic composition of basil plants is differentially altered by plant nutrient status and inoculation with mycorrhizal fungi. HortScience 2012, 47, 660–671. [Google Scholar] [CrossRef]
- Schweiger, R.; Mueller, C. Leaf metabolome in arbuscular mycorrhizal symbiosis. Curr. Opin. Plant Biol. 2015, 26, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Guo, L.-P.; Chen, B.-D.; Hao, Z.-P.; Wang, J.-Y.; Huang, L.-Q.; Yang, G.; Cui, X.-M.; Yang, L.; Wu, Z.-X. Arbuscular mycorrhizal symbiosis and active ingredients of medicinal plants: Current research status and prospectives. Mycorrhiza 2013, 23, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Pedone-Bonfim, M.V.L.; da Silva, F.S.B.; Maia, L.C. Production of secondary metabolites by mycorrhizal plants with medicinal or nutritional potential. Acta Physiol. Plant. 2015, 37, 27. [Google Scholar] [CrossRef]
- Dutta, S.C.; Neog, B. Accumulation of secondary metabolites in response to antioxidant activity of turmeric rhizomes co-inoculated with native arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria. Sci. Hortic. 2016, 204, 179–184. [Google Scholar] [CrossRef]
- Raghuwanshi, R.; Sinha, S. Linking mycorrhizal technology with medicinal plant secondary metabolites. In Microbial Diversity and Biotechnology in Food Security; Springer: New Dehli, India, 2014; pp. 121–132. [Google Scholar]
- Zhang, H.; Sun, J.; Bao, Y. Advances in studies on plant secondary metabolites influenced by arbuscular mycorrhizal fungi. J. Agric. biotechnol. 2015, 23, 1093–1103. [Google Scholar]
- Kilam, D.; Sharma, P.; Agnihotri, A.; Kharkwal, A.; Varma, A. Microbial symbiosis and bioactive ingredients of medicinal plants. In Mycorrhiza-eco-physiology, Secondary Metabolites, Nanomaterials; Springer: Cham, Switzerland, 2017; pp. 283–302. [Google Scholar]
- Crișan, I.; Vidican, R.; Stoian, V. Induced modifications on secondary metabolism of aromatic and medicinal plants—an endomycorrhizal approach. Hop Med. Plant. 2018, 26, 15–29. [Google Scholar]
- Kosa, G.; Shapaval, V.; Kohler, A.; Zimmermann, B. FTIR spectroscopy as a unified method for simultaneous analysis of intra-and extracellular metabolites in high-throughput screening of microbial bioprocesses. Microb. Cell Fact. 2017, 16, 195. [Google Scholar] [CrossRef]
- Ţucureanu, V.; Matei, A.; Avram, A.M. FTIR Spectroscopy for Carbon Family Study. Crit. Rev. Anal. Chem. 2016, 46, 502–520. [Google Scholar] [CrossRef]
- Ciobanu, I.; Cantor, M.; Stefan, R.; Erzsebet, B.; Magyari, K.; Monica, B. The influence of storage conditions on the biochemical composition and morphology of dahlia tubers. Not. Bot. Horti Agrobo. Cluj-Napoc. 2016, 44, 459–465. [Google Scholar] [CrossRef][Green Version]
- Robu, T. A Monograph of Genus Iris; Ion Ionescu de la Brad: Iași, Romania, 2005; ISBN 973-7921-60-7. [Google Scholar]
- Davidescu, D.; Calancea, L.; Davidescu, V.; Lixandru, G.; Țârdea, C. Agrochimie; Editura Didactică și Pedagogică: Bucharest, Romania, 1981. [Google Scholar]
- Crișan, I.; Vidican, R.; Stoian, V.; Cantor, M. The effects of supplementary mycorrhization regarding some vegetative characteristics at iris germanica L. Lucrări Științifice Seria Agronomie USAMV Iași 2017, 60, 209–214. [Google Scholar]
- Crișan, I.; Vidican, R.; Stoian, V.; Vâtcă, S.; Stoie, A. Leaf stomatal parameters of Iris germanica L. influenced by cultivar and arbuscular mycorrhizae inoculation in field conditions, Romania. Sci. Papers Series B Hortic. 2018, 62, 619–628. [Google Scholar]
- Weather Cluj-Napoca. Available online: https://www.wunderground.com/history/daily/ro/cluj-napoca/LRCL (accessed on 21 May 2019).
- Root Grow AMF. Available online: http://rootgrow.co.uk/ (accessed on 22 November 2019).
- Vierheilig, H.; Coughlan, A.P.; Wyss, U.R.S.; Piché, Y. Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl. Environ. Microbiol. 1998, 64, 5004–5007. [Google Scholar] [PubMed]
- Lab Procedures, Trouvelot Method. Available online: https://www2.dijon.inra.fr/mychintec/Protocole/Workshop_Procedures.html#1.5 (accessed on 21 May 2019).
- Mycocalc Software; Institut National de la Recherche Agronomique. Available online: https://www2.dijon.inra.fr/mychintec/Mycocalc-prg/download.html (accessed on 21 May 2019).
- Hammer, Ø. Past 3.x Software; Natural History Museum, University of Oslo. Available online: https://folk.uio.no/ohammer/past/ (accessed on 28 November 2019).
- Origin: Data Analysis and Graphing Software; Origin Lab. Available online: https://www.originlab.com/ (accessed on 28 November 2019).
- Rohaeti, E.; Rafi, M.; Syafitri, U.D.; Heryanto, R. Fourier transform infrared spectroscopy combined with chemometrics for discrimination of Curcuma longa, Curcuma xanthorrhiza and Zingiber cassumunar. Spectrochim. Acta A 2015, 137, 1244–1249. [Google Scholar] [CrossRef] [PubMed]
- Taber, R.A.; Trappe, J.M. Vesicular-arbuscular mycorrhiza in rhizomes, scale-like leaves, roots, and xylem of ginger. Mycologia 1982, 74, 156–161. [Google Scholar] [CrossRef]
- Abdullah, A.H.D.; Chalimah, S.; Primadona, I.; Hanantyo, M.H.G. Physical and chemical properties of corn, cassava, and potato starch. IOP Conf. Ser. Earth Environ. Sci. 2018, 160, 012003. [Google Scholar] [CrossRef]
- Sharma, N.; Khajuria, Y.; Sharma, J.; Gondal, M.A.; Kumar, V.; Dwivedi, Y.; Singh, V.K. Spectroscopic analysis of rhizomes of black turmeric (Curcuma caesia). In Proceedings of the AIP Conference Proceedings, Jammu, India, 17–18 April 2018; AIP Publishing: New York, NY, USA, 2018; pp. 030036-1–030036-6. [Google Scholar] [CrossRef]
- Lee, B.-J.; Kim, H.-Y.; Lim, S.R.; Huang, L.; Choi, H.-K. Discrimination and prediction of cultivation age and parts of Panax ginseng by Fourier-transform infrared spectroscopy combined with multivariate statistical analysis. PLoS ONE 2017, 12, e0186664. [Google Scholar] [CrossRef]
- Yang, Y.; Ou, X.; Yang, G.; Xia, Y.; Chen, M.; Guo, L.; Liu, D. Arbuscular Mycorrhizal Fungi Regulate the Growth and Phyto-Active Compound of Salvia miltiorrhiza Seedlings. Appl. Sci. 2017, 7, 68. [Google Scholar] [CrossRef]
- da Silva, M.F.; Pescador, R.; Rebelo, R.A.; Stürmer, S.L. The effect of arbuscular mycorrhizal fungal isolates on the development and oleoresin production of micropropagated Zingiber officinale. Braz. J. Plant Physiol. 2008, 20, 119–130. [Google Scholar] [CrossRef]
- Gupta, M.L.; Prasad, A.; Ram, M.; Kumar, S. Effect of the vesicular–arbuscular mycorrhizal (VAM) fungus Glomus fasciculatum on the essential oil yield related characters and nutrient acquisition in the crops of different cultivars of menthol mint (Mentha arvensis) under field conditions. Bioresour. Technol. 2002, 81, 77–79. [Google Scholar] [CrossRef]
- Myristic Acid. Available online: https://www.makingcosmetics.com/Myristic-Acid_p_1045.html?locale=en (accessed on 22 November 2019).
- Arctander, S. Perfume and Flavor Materials of Natural Origin; Lulu Press Inc: Morrisville, NC, USA, 2017; p. 358. [Google Scholar]
- Trivedi, M.K.; Tallapragada, R.M.; Branton, A.; Trivedi, D.; Nayak, G.; Mishra, R.K.; Snehasis, J. Physical, spectroscopic and thermal characterization of biofield treated myristic acid. J. Fundam. Renew. Energy Appl. 2015, 5, 3–6. [Google Scholar] [CrossRef]
- Nille, G.C.; Singh, V.K.; Reddy, K. Investigation of Talapotaka Churna using FTIR spectroscopy. JAHM 2016, 4, 3–11. [Google Scholar]
- Zeier, J.; Schreiber, L. Fourier transform infrared-spectroscopic characterisation of isolated endodermal cell walls from plant roots: Chemical nature in relation to anatomical development. Planta 1999, 209, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, N.H.; Selvaraj, G.; Wei, Y.; King, J. Role of lignification in plant defense. Plant Signal. Behav. 2009, 4, 158–159. [Google Scholar] [CrossRef] [PubMed]
- Sahu, V.; Dhongade, H.J.; Sawarkar, H.A.; Sahu, P.; Sahu, R.; Patel, D.; Kashyap, P. Isolation and characterization of valerenic acid from Valeriana wallichii (Valerianaceae). IJBPAS 2016, 5, 1230–1243. [Google Scholar]
- Pakkirisamy, M.; Kalakandan, S.K.; Ravichandran, K. Phytochemical screening, GC-MS, FT-IR analysis of methanolic extract of Curcuma caesia Roxb (Black Turmeric). Pharmacogn. J. 2017, 9. [Google Scholar] [CrossRef]
- Ardelean, F.; Moaca, E.A.; Pacurariu, C.; Antal, D.S.; Dehelean, C.; Toma, C.-C.; Dragan, S. Invasive Polygonum cuspidatum: Physico-chemical analysis of a plant extract with pharmaceutical potential. Studia Universitatis” Vasile Goldis” Arad. Seria Stiintele Vietii (Life Sciences Series) 2016, 26, 415. [Google Scholar]
- Pachurekar, P.; Dixit, A.K. Phytochemical screening and spectroscopic characterization of phytoconstituents from rhizome extract of Hedychium coronarium J. Koenig. Int. J. Res. BioSci. 2018, 7, 34–40. [Google Scholar] [CrossRef]
- Orujei, Y.; Shabani, L.; Sharifi-Tehrani, M. Induction of glycyrrhizin and total phenolic compound production in licorice by using arbuscular mycorrhizal fungi. Russ. J. Plant Physiol. 2013, 60, 855–860. [Google Scholar] [CrossRef]
- Irises: Plant Care and Collection of Varieties. Available online: https://garden.org/plants/group/irises/ (accessed on 10 September 2019).
- Ryan, M.H.; Graham, J.H. Little evidence that farmers should consider abundance or diversity of arbuscular mycorrhizal fungi when managing crops. New Phytol. 2018, 220, 1092–1107. [Google Scholar] [CrossRef]

| Parameter | Method | Results |
|---|---|---|
| pH | Potentiometric | 6.72 |
| Humus | Walkley–Black | 1.35% |
| Nitrogen | Kjeldahl | 0.461% |
| Phosphorus | Colorimetric | 68 ppm |
| Potassium | Flame photometry | 312 ppm |
| Basic cation saturation | Kappen | 20.96 me/100 g |
| Hydrolytic acidity | Extraction | 1.92 me/100 g |
| Base saturation (V) | Calculation | 92 |
| Granulometric | Kacinscki | Coarse sand 14.42, fine sand 25.08, silt I 7.95, silt II 13.65, clay 38.90 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crișan, I.; Vidican, R.; Olar, L.; Stoian, V.; Morea, A.; Ștefan, R. Screening for Changes on Iris germanica L. Rhizomes Following Inoculation with Arbuscular Mycorrhiza Using Fourier Transform Infrared Spectroscopy. Agronomy 2019, 9, 815. https://doi.org/10.3390/agronomy9120815
Crișan I, Vidican R, Olar L, Stoian V, Morea A, Ștefan R. Screening for Changes on Iris germanica L. Rhizomes Following Inoculation with Arbuscular Mycorrhiza Using Fourier Transform Infrared Spectroscopy. Agronomy. 2019; 9(12):815. https://doi.org/10.3390/agronomy9120815
Chicago/Turabian StyleCrișan, Ioana, Roxana Vidican, Loredana Olar, Vlad Stoian, Adriana Morea, and Răzvan Ștefan. 2019. "Screening for Changes on Iris germanica L. Rhizomes Following Inoculation with Arbuscular Mycorrhiza Using Fourier Transform Infrared Spectroscopy" Agronomy 9, no. 12: 815. https://doi.org/10.3390/agronomy9120815
APA StyleCrișan, I., Vidican, R., Olar, L., Stoian, V., Morea, A., & Ștefan, R. (2019). Screening for Changes on Iris germanica L. Rhizomes Following Inoculation with Arbuscular Mycorrhiza Using Fourier Transform Infrared Spectroscopy. Agronomy, 9(12), 815. https://doi.org/10.3390/agronomy9120815







