Sensitivity of Three Phosphate Extraction Methods to the Application of Phosphate Species Differing in Immediate Plant Availability
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Soils
2.2. Experimental Treatments
2.3. CAL Analysis
2.4. Olsen Analyses
2.5. DGT in Standard Solution
2.6. DGT Analysis in Soil
2.7. Statistical Analysis
3. Results and Discussion
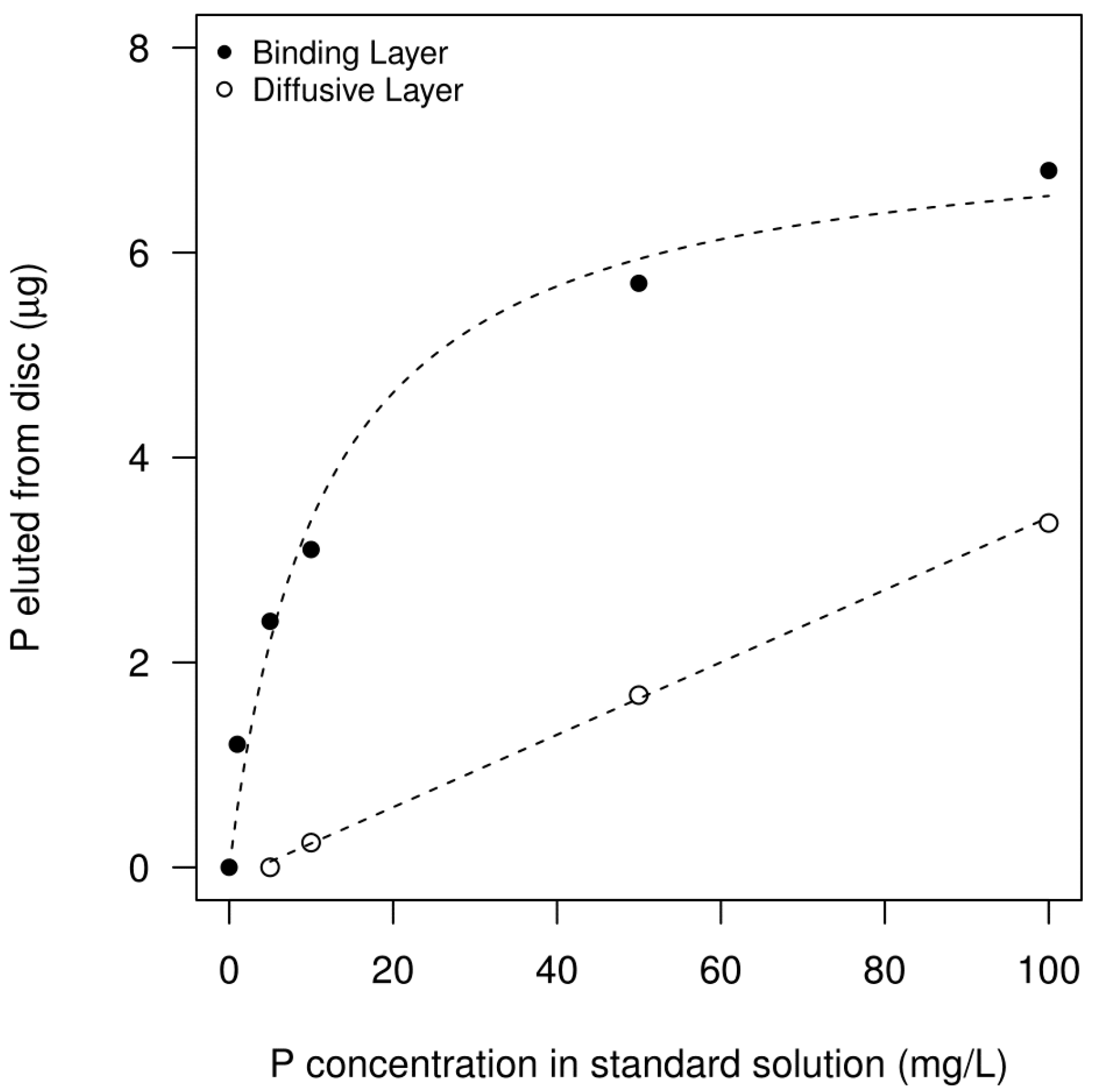
3.1. DGT-Standard Curve
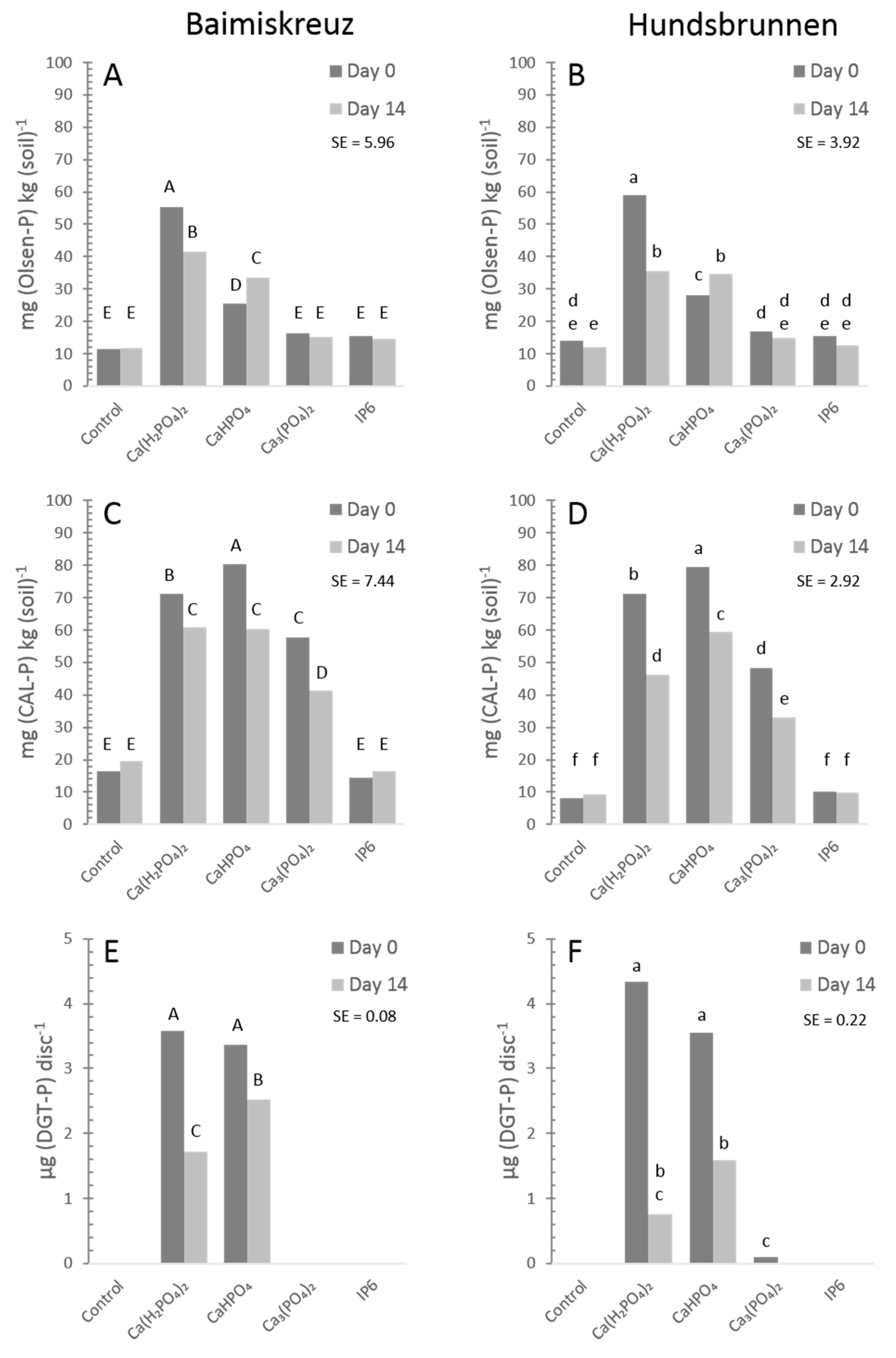
3.2. Comparison of Extraction Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cordell, D.; White, S. Sustainable Phosphorus Measures: Strategies and Technologies for Achieving Phosphorus Security. Agronomy 2013, 3, 86–116. [Google Scholar] [CrossRef]
- Kruse, J.; Abraham, M.; Amelung, W.; Baum, C.; Bol, R.; Kühn, O.; Lewandowski, H.; Niederberger, J.; Oelmann, Y.; Rüger, C.; et al. Innovative methods in soil phosphorus research: A review. J. Plant Nutr. Soil Sci. 2015, 178, 43–88. [Google Scholar] [CrossRef] [PubMed]
- Wünscher, R.; Unterfrauner, H.; Peticzka, R.; Zehetner, F. A comparison of 14 soil phosphorus extraction methods applied to 50 agricultural soils from Central Europe. Plant Soil Environ. 2015, 61, 86–96. [Google Scholar] [CrossRef]
- Schüller, H. Die CAL-Methode, eine neue Methode zur Bestimmung des pflanzenverfügbaren Phosphates in Böden. Z. für Pflanzenernähr. Bodenkd. 1969, 123, 48–63. [Google Scholar] [CrossRef]
- VdLUFA Das VdLUFA Methodenbuch. In Das VdLUFA Methodenbuch; VdLUFA Verlag: Darmstadt, Germany, 2012.
- Hedley, M.J.; Stewart, J.W.B.; Chauhan, B.S. Changes in Inorganic and Organic Soil Phosphorus Fractions Induced by Cultivation Practices and by Laboratory Incubations. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Bowman, R.A.; Cole, C.V. An exploratory method for fractionation of organic phosphorus from grassland soils. Soil Sci. 1978, 125, 95–101. [Google Scholar] [CrossRef]
- Ivanoff, D.B.; Reddy, K.R.; Robinson, S. Chemical fractionation of organic phosphorus in selected histosols. Soil Sci. 1998, 163, 36–45. [Google Scholar] [CrossRef]
- Nawara, S.; Van Dael, T.; Merckx, R.; Amery, F.; Elsen, A.; Odeurs, W.; Vandendriessche, H.; Mcgrath, S.; Roisin, C.; Jouany, C.; et al. A comparison of soil tests for available phosphorus in long-term field experiments in Europe. Eur. J. Soil Sci. 2017, 68, 873–885. [Google Scholar] [CrossRef]
- Bissani, C.A.; Tedesco, M.J.; Camargo, F.D.O.; Miola, G.L.; Gianello, C. Anion-exchange resins and iron oxide-impregnated filter paper as plant available phosphorus indicators in soils. Commun. Soil Sci. Plant Anal. 2002, 33, 1119–1130. [Google Scholar] [CrossRef]
- Degryse, F.; Smolders, E.; Zhang, H.; Davison, W. Predicting availability of mineral elements to plants with the DGT technique: A review of experimental data and interpretation by modelling. Environ. Chem. 2009, 6, 198–218. [Google Scholar] [CrossRef]
- Davison, W.; Zhang, H. Progress in understanding the use of diffusive gradients in thin films (DGT)—Back to basics. Environ. Chem. 2012, 9, 1–13. [Google Scholar] [CrossRef]
- Zhang, C.; Ding, S.; Xu, D.; Tang, Y.; Wong, M.H. Bioavailability assessment of phosphorus and metals in soils and sediments: A review of diffusive gradients in thin films (DGT). Environ. Monit. Assess. 2014, 186, 7367–7378. [Google Scholar] [CrossRef]
- Christel, W.; Lemming, C.; Mundus, S.; Bruun, S.; Magid, J.; Jensen, L.S. Measuring Phosphorus Availability in Recently Fertilized Soils with the Diffusive Gradient in Thin Films (DGT) Method—Challenges and Opportunities. Commun. Soil Sci. Plant Anal. 2016, 47, 563–570. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Ritz, C.; Streibig, J. Bioassay Analysis Using R. J. Stat. Softw. 2005, 12, 1–22. [Google Scholar] [CrossRef]
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Findenegg, G.R.; Nelemans, J.A. The effect of phytase on the availability of P from myo-inositol hexaphosphate (phytate) for maize roots. Plant Soil 1993, 154, 189–196. [Google Scholar] [CrossRef]
- Gerke, J. Phytate (Inositol Hexakisphosphate) in Soil and Phosphate Acquisition from Inositol Phosphates by Higher Plants. A Review. Agronomy 2015, 4, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.S.; Pratt, P.F. Effects of Rate of Application and Time on Phosphorus Sorption by Soils1. Soil Sci. Soc. Am. J. 1982, 46, 926–928. [Google Scholar] [CrossRef]
- Nziguheba, G.; Palm, C.A.; Buresh, R.J.; Smithson, P.C. Soil phosphorus fractions and adsorption as affected by organic and inorganic sources. Plant Soil 1998, 198, 159–168. [Google Scholar] [CrossRef]
- Burkitt, L.L.; Mason, S.D.; Dougherty, W.J.; Sale, P.W.G. The ability of the DGT soil phosphorus test to predict pasture response in Australian pasture soils—A preliminary assessment. Soil Use Manag. 2015, 32, 27–35. [Google Scholar] [CrossRef]
- Six, L.; Pypers, P.; Degryse, F.; Smolders, E.; Merckx, R. The performance of DGT versus conventional soil phosphorus tests in tropical soils—An isotope dilution study. Plant Soil 2012, 359, 267–279. [Google Scholar] [CrossRef]
- Six, L.; Smolders, E.; Merckx, R. The performance of DGT versus conventional soil phosphorus tests in tropical soils—Maize and rice responses to P application. Plant Soil 2013, 366, 49–66. [Google Scholar] [CrossRef]
- Kalkhajeh, Y.K.; Sørensen, H.; Huang, B.; Guan, D.-X.; Luo, J.; Hu, W.; Holm, P.E.; Hansen, H.C.B. DGT technique to assess P mobilization from greenhouse vegetable soils in China: A novel approach. Sci. Total Environ. 2018, 630, 331–339. [Google Scholar] [CrossRef] [PubMed]
| Field | pH | Sand (%) | Silt (%) | Clay (%) | Texture | PCAL (mg kg−1) | POlsen (mg kg−1) | PTotal (mg kg−1) |
|---|---|---|---|---|---|---|---|---|
| Baimiskreuz | 6.4 | 7 | 61 | 32 | silty clay loam | 16.5 | 11.6 | 510 |
| Hundsbrunnen | 7.4 | 5 | 66 | 29 | silty clay loam | 8.2 | 13.9 | 500 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartmann, T.E.; Wollmann, I.; You, Y.; Müller, T. Sensitivity of Three Phosphate Extraction Methods to the Application of Phosphate Species Differing in Immediate Plant Availability. Agronomy 2019, 9, 29. https://doi.org/10.3390/agronomy9010029
Hartmann TE, Wollmann I, You Y, Müller T. Sensitivity of Three Phosphate Extraction Methods to the Application of Phosphate Species Differing in Immediate Plant Availability. Agronomy. 2019; 9(1):29. https://doi.org/10.3390/agronomy9010029
Chicago/Turabian StyleHartmann, Tobias Edward, Iris Wollmann, Yawen You, and Torsten Müller. 2019. "Sensitivity of Three Phosphate Extraction Methods to the Application of Phosphate Species Differing in Immediate Plant Availability" Agronomy 9, no. 1: 29. https://doi.org/10.3390/agronomy9010029
APA StyleHartmann, T. E., Wollmann, I., You, Y., & Müller, T. (2019). Sensitivity of Three Phosphate Extraction Methods to the Application of Phosphate Species Differing in Immediate Plant Availability. Agronomy, 9(1), 29. https://doi.org/10.3390/agronomy9010029





