Similarities and Differences in the Acquisition of Fe and P by Dicot Plants
Abstract
1. Fe and P Nutrition in Dicot Plants
2. Hormones and Signaling Substances in the Regulation of Fe and P Deficiency Responses in Dicot Plants
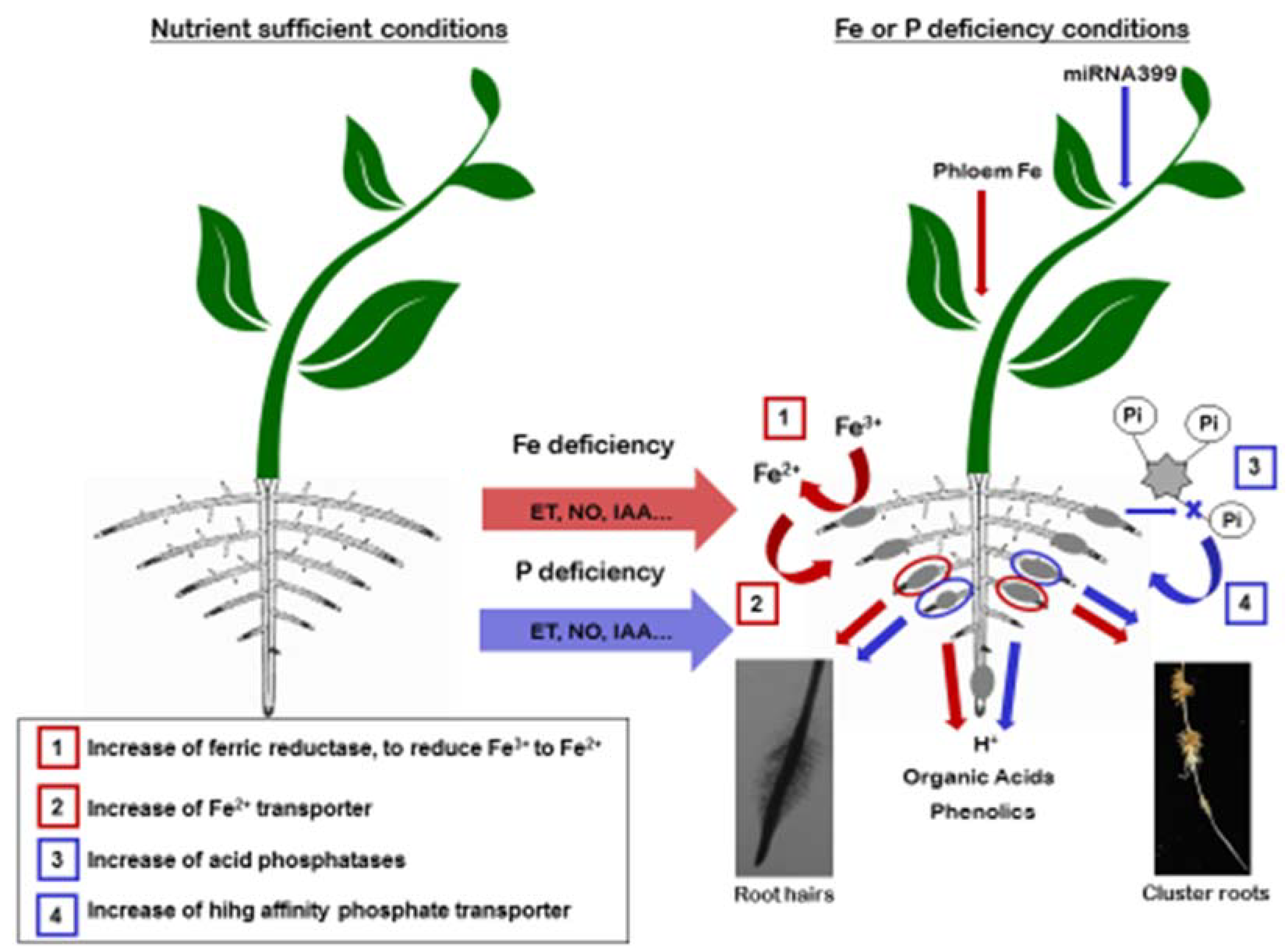
3. Crosstalk among Fe and P Deficiencies
4. Effect of P on Fe Nutrition and Vice Versa
5. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: London, UK, 1995; ISBN 0-12-473541-X. [Google Scholar]
- Zhang, Z.; Hong, L.; William, J.L. Molecular mechanisms underlying phosphate sensing, signaling and adaptation in plants. J. Integr. Plant Biol. 2014, 56, 192–220. [Google Scholar] [CrossRef] [PubMed]
- García, M.J.; Romera, F.J.; Lucena, C.; Alcántara, E.; Pérez-Vicente, R. Ethylene and the regulation of physiological and morphological responses to nutrient deficiencies. Plant Physiol. 2015, 169, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Lucena, C.; Romera, F.J.; García, M.J.; Alcántara, E.; Pérez-Vicente, R. Ethylene Participates in the Regulation of Fe Deficiency Responses in Strategy I Plants and in Rice. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Liu, D. Ethylene and plant responses to phosphate deficiency. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Stetter, M.G.; Benz, M.; Ludewig, U. Increased root hair density by loss of WRKY6 in Arabidopsis thaliana. PeerJ 2017, 5, e2891. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, W.L. Chemical reactions in soils that affect iron availability to plants. A quantitative approach. In Iron Nutrition in Soils and Plants; Abadía, J., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherland, 1995; pp. 7–14. [Google Scholar]
- Korcak, R.F. Iron deficiency chlorosis. Hortic. Rev. 1987, 9, 133–186. [Google Scholar]
- Fuentes, M.; Bacaicoa, E.; Rivero, M.; Zamarreño, A.M.; García-Mina, J.M. Complementary Evaluation of Iron Deficiency Root Responses to Assess the Effectiveness of Different Iron Foliar Applications for Chlorosis Remediation. Front. Plant Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chaney, R.L. Diagnostic practices to identify iron deficiency in higher plants. J. Plant Nutr. 1984, 7, 47–67. [Google Scholar] [CrossRef]
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus uptake by plants: From soil to cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Raghothama, K.G. Phosphorus acquisition. Ann. Rev. Plant Physiol. 1999, 50, 665–693. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.B.; McCall, I.; Jackson, B.; Whipker, B.E. Growth response of herbaceous ornamental to phosphorus fertilization. Hortscience 2017, 52, 1362–1367. [Google Scholar] [CrossRef]
- Hirsch, J.; Marin, E.; Floriani, M.; Chiarenza, S.; Richaud, P.; Nussaume, L.; Thibaud, M.C. Phosphate deficiency promotes modification of iron distribution in Arabidopsis plants. Biochimie 2006, 88, 1767–1771. [Google Scholar] [CrossRef] [PubMed]
- Matar, A.; Torrent, J.; Ryan, J. Soil and fertilizer phosphorus and crop responses in the dryland Mediterranean zone. Adv. Soil Sci. 1992, 18, 81–146. [Google Scholar]
- Salisbury, F.B.; Ross, C.W. Plant physiology, 4th ed.; Wodsworth Publishing: Belmont, CA, USA, 1992; 728p. [Google Scholar]
- Alscher, R.G.; Erturk, N.; Heath, L.S. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J. Exp. Bot. 2002, 53, 1331–1341. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.A.; Guerinot, M.L. Mining iron: Iron uptake and transport in plants. FEBS Lett. 2007, 581, 2273–2280. [Google Scholar] [CrossRef] [PubMed]
- Neumann, G. The Role of Ethylene in Plant Adaptations for Phosphate Acquisition in Soils—A Review. Front. Plant Sci. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Ann. Rev. Plant Biol. 2012, 63, 131–152. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, R.; Brumbarova, T.; Bauer, P. Fitting into the harsh reality: Regulation of iron-deficiency responses in dicotyledonous plants. Mol. Plant 2012, 5, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.L.; Connolly, E.L. Time to pump iron: Iron-deficiency signaling mechanisms of higher plants. Curr. Opin. Plant Biol. 2008, 11, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Römheld, V.; Marschner, H. Mobilization of iron in the rhizosphere of different plant species. Adv. Plant Nutr. 1986, 2, 155–204. [Google Scholar]
- Vorwieger, A.; Gryczka, C.; Czihal, A.; Douchkov, D.; Tiedemann, J.; Mock, H.P.; Jakoby, M.; Weisshaar, B.; Saalbach, I.; Baumlein, H. Iron assimilation and transcription factor controlled synthesis of riboflavin in plants. Planta 2007, 226, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Celma, J.; Schmidt, W. Reduction-based iron uptake revisited. On the role of secreted iron-binding compounds. Plant Signal. Behav. 2013, 8, e26116. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Günther, C.; Weber, M.; Spörlein, C.; Loscher, S.; Böttcher, C.; Schobert, R.; Clemens, S. Metabolome analysis of Arabidopsis thaliana roots identifies a key metabolic pathway for iron acquisition. PLoS ONE 2014, 9, e102444. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Wu, H.; Wang, N.; Li, J.; Zhao, W.; Du, J.; Wang, D.; Ling, H.Q. FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res. 2008, 18, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Brumbarova, T.; Bauer, P.; Ivanov, R. Molecular mechanisms governing Arabidopsis iron uptake. Trends Plant Sci. 2015, 20, 124–133. [Google Scholar] [CrossRef] [PubMed]
- García, M.J.; Lucena, C.; Romera, F.J.; Alcántara, E.; Pérez-Vicente, R. Ethylene and nitric oxide involvement in the up-regulation of key genes related to iron acquisition and homeostasis in Arabidopsis. J. Exp. Bot. 2010, 61, 3885–3899. [Google Scholar] [CrossRef] [PubMed]
- Palmer, C.M.; Hindt, M.N.; Schmidt, H.; Clemens, S.; Guerinot, M.L. MYB10 and MYB72 are required for growth under iron-limiting conditions. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Shuler, M.; Rellán-Álvarez, R.; Fink-Straube, C.; Abadía, J.; Bauer, P. Nicotianamine functions in the phloem based transport of iron to sink organs, in pollen development and tuve growth in Arabidopsis. Plant Cell 2012, 24, 2380–2400. [Google Scholar] [CrossRef] [PubMed]
- Romera, F.J.; Lucena, C.; Alcántara, E. Plant hormones influencing iron uptake in plants. In Iron Nutrition in Plants and Rhizospheric Microorganisms; Barton, L.L., Abadía, J., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 251–278. [Google Scholar]
- Del Pozo, J.C.; Allona, I.; Rubio, V.; Leyva, A.; de la Peña, A.; Aragoncillo, C.; Paz-Ares, J. A type 5 acid phosphatase gene from Arabidopsis thaliana is induced by phosphate starvation and by som other types of phosphate mobilising/oxidative stress conditions. Plant J. 1999, 19, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Tian, J.; Lim, B.L.; Yan, X.; Liao, H. Overexpressing AtPAP15 enhances phosphorus efficiency in soybean. Plant Physiol. 2009, 151, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Robinson, W.D.; Park, J.; Tran, H.T.; Del Vecchio, H.A.; Ying, S.; Zins, J.L.; Patel, K.; McKnight, T.D.; Plaxton, W.C. The secreted purple acid phosphatase isozymes AtPAP12 and AtPAP26 play a pivotal role in extracellular phosphate-scavenging by Arabidopsis thaliana. J. Exp. Bot. 2012, 63, 6531–6542. [Google Scholar] [CrossRef] [PubMed]
- Valentinuzzi, F.; Pii, Y.; Vigani, G.; Lehmann, M.; Cesco, S.; Mimmo, T. Phosphorus and iron deficiencies incude a metabolic reprograming and affect the exudation traits of the woody plant Fragariaxananassa. J. Exp. Bot. 2015, 66, 6483–6495. [Google Scholar] [CrossRef] [PubMed]
- Steffen, A. Phosphate sensing in root development. Curr. Opin. Plant Biol. 2011, 14, 303–309. [Google Scholar] [CrossRef]
- Ticconi, C.A.; Delatorre, C.A.; Lahner, B.; Salt, D.E.; Abel, S. Arabidopsis pdr reveals a phosphate sensitive checkpoint in root development. Plant J. 2004, 37, 801–814. [Google Scholar] [CrossRef] [PubMed]
- Schachtman, D.P.; Shin, R. Nutrient sensing and signalling: NPKS. Ann. Rev. Plant Biol. 2007, 58, 47–69. [Google Scholar] [CrossRef] [PubMed]
- Rubio, V.; Linhares, F.; Solano, R.; Martín, A.C.; Iglesias, J.; Leyva, A.; Paz-Ares, J. A conversed MYB transcrption factor involved in phosphate starvation signaling both in vascular plants and in unicelular algae. Genes Dev. 2001, 15, 2122–2133. [Google Scholar] [CrossRef] [PubMed]
- Todd, C.D.; Zeng, P.; Rodríguez, A.M.; Hoyos, M.E.; Polacco, J.C. Transcripts of MYB-like genes respond to phosphorus and nitrogen deprivation in Arabidopsis. Planta 2004, 219, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Franco-Zorrilla, J.M.; González, E.; Bustos, R.; Linhares, F.; Leyva, A.; Paz-Ares, J. The transcriptional control of plant responses to phosphate limitation. J. Exp. Bot. 2004, 55, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Bari, R.; Pant, B.D.; Stitt, M.; Scheibl, W.-R. PHO2, microRNA399, and PHR1 define a phosphate-signaling pathway in plants. Plant Physiol. 2006, 141, 988–999. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.; Morant, M.; Jarmer, H.; Nilsson, L.; Nielsen, T.H. Genomewide analysis of the Arabidopsis leaf transcriptome reveals interaction of phosphate and sugar metabolism. Plant Physiol. 2007, 143, 156–171. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Brown, K.M. Topsoil foraging-an architectural adaptation of plants to low phosphorus availability. Plant Soil 2001, 237, 225–237. [Google Scholar] [CrossRef]
- Lynch, J.P.; Brown, K.M. Ethylene and plant responses to nutritional stress. Physiol. Plant 1997, 100, 613–619. [Google Scholar] [CrossRef]
- Gahoonia, T.S.; Nielsen, N.E.; Lyshede, O.B. Phosphorus acquisition of cereal cultivars in the field at three levels of P fertilization. Plant Soil 1999, 211, 269–281. [Google Scholar] [CrossRef]
- Gilroy, S.; Jones, D.L. From form to function: Development and nutrient uptake in root hairs. Trends Plant Sci. 2000, 5, 56–60. [Google Scholar] [CrossRef]
- Louis, I.; Racette, S.; Torrey, J.G. Occurrence of cluster roots on Myrica cerífera L. (Myricaceae) in water culture in relation to phosphorus nutrition. New Phytol. 1990, 115, 311–317. [Google Scholar] [CrossRef]
- Dinkelaker, B.; Hengeler, C.; Marschner, H. Distribution and function of proteoid roots and other root clusters. Bot. Acta 1995, 108, 183–200. [Google Scholar] [CrossRef]
- Skene, K.R. Cluster roots: Some ecological considerations. J. Ecol. 1998, 86, 1060–1064. [Google Scholar] [CrossRef]
- Adams, M.A.; Bells, T.L.; Pate, J.S. Phosphorus sources and availability modify growth and distribution of root cluster and nodules of native Australian legumes. Plant Cell Environ. 2002, 25, 837–850. [Google Scholar] [CrossRef]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: Critical adaptations by plants for securing a nonrenewable resource. New Phytol. 2003, 157, 423–447. [Google Scholar] [CrossRef]
- Perea-Garcia, A.; Garcia-Molina, A.; Andres-Colas, N.; Vera-Sirera, F.; Perez-Amador, M.A.; Puig, S.; Penarrubia, L. Arabidopsis Copper Transport Protein COPT2 Participates in the Cross Talk between Iron Deficiency Responses and Low-Phosphate Signaling. Plant Physiol. 2013, 162, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Lucena, C.; Porras, R.; Romera, F.J.; Alcántara, E.; García, M.J.; Pérez-Vicente, R.; Bacaicoa, E.; García-Mina, J.M. Ethylene involvement in the regulation of physiological responses to Fe and P deficiency: Similarities and differences. In Proceedings of the XVII International Symposium on Iron Nutrition and Interactions in Plants, Quedlinburg, Germany, 6–10 July 2014. [Google Scholar]
- Wang, Z.; Straub, D.; Yang, H.; Kania, A.; Shen, J.; Ludewig, U.; Neumann, G. The regulatory network of cluster-root function and development in phosphatedeficient white lupin (Lupinus albus) identified by transcriptome sequencing. Physiol. Plant 2014, 151, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, Q.; Wang, Y.; Wu, T.; Yang, Y.; Zhang, X.; Han, Z.; Xu, X. Ethylene response factor AtERF72 negatively regulates Arabidopsis thaliana response to iron deficiency. Biochem. Biophys. Commun. 2017, 491, 862–868. [Google Scholar] [CrossRef] [PubMed]
- De Kock, P.C. Hall, Iron nutrition of plants at high pH. Soil Sci. 1955, 79, 167–175. [Google Scholar] [CrossRef]
- Wang, Y.H.; Garvin, D.F.; Kochian, L.V. Rapid introduction of regulatory and transporter genes in response to phosphorus, potassium, and iron deficiencies in tomato roots. Evidence for cross talk and root/rhizosphere-mediated signals. Plant Physiol. 2002, 130, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Misson, J.; Raghothama, K.G.; Jain, A.; Jouhet, J.; Block, M.A.; Bligny, R.; Ortet, P.; Creff, A.; Somerville, S.; Rolland, N.; et al. A genome-wide transcriptional analysis using Arabidopsis thaliana Affymetrix gene chips determined plant responses to phosphate deprivation. Proc. Natl. Acad. Sci. USA 2005, 102, 11934–11939. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.T.; Lahner, B.; Yakubova, E.; Salt, D.E.; Raghothama, K.G. The effect of iron on the primary root elongation of Arabidopsis during phosphate deficiency. Plant Physiol. 2008, 147, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Huang, F.; Narsai, R.; Wu, J.; Giraud, E.; He, F.; Cheng, L.; Wang, F.; Wu, P.; Whelan, J.; et al. Physiological and transcriptome analysis of iron and phosphorus interaction in rice seedlings. Plant Physiol. 2009, 151, 262–274. [Google Scholar] [CrossRef] [PubMed]
- Romera, F.J.; Alcantara, E.; de la Guardia, M.D. Effects of bicarbonate, phosphate and high pH on the reducing capacity of Fe-deficient sunflower and cucumber plants. J. Plant Nutr. 1992, 15, 1519–1530. [Google Scholar] [CrossRef]
- Sánchez-Rodriguez, A.R.; del Campillo, M.C.; Torrent, J. Phosphate aggravates iron chlorosis in carbonate−iron oxide systems. Plant Soil 2013, 373, 31–42. [Google Scholar] [CrossRef]
- Romera, F.J.; Alcántara, E. Iron-deficiency stress responses in cucumber (Cucumis sativus L.) roots. A possible role for ethylene? Plant Physiol. 1994, 105, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Borch, K.; Bouma, T.J.; Lynch, J.P.; Brown, K.M. Ethylene: A regulator of root architectural responses to soil phosphorus availability. Plant Cell Environ. 1999, 22, 425–431. [Google Scholar] [CrossRef]
- Lucena, C.; Waters, B.M.; Romera, F.J.; García, M.J.; Morales, M.; Alcántara, E.; Pérez-Vicente, R. Ethylene could influence ferric reductase, iron transporter and H−-ATPase gene expression by affecting FER (or FER-like) gene activity. J. Exp. Bot. 2006, 57, 4145–4154. [Google Scholar] [CrossRef] [PubMed]
- García, M.J.; Suarez, V.; Romera, F.J.; Alcántara, E.; Pérez-Vicente, R. A new model involving ethylene, nitric oxide and Fe to explain the regulation of Fe-acquisition genes in Strategy I plants. Plant Physiol. Biochem. 2011, 49, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Zhu, C.; Liu, Y.; Karthikeyan, A.S.; Bressan, R.A.; Raghothama, K.G.; Liu, D. Ethylene signaling is involved in regulation of phosphate starvation-induced gene expression and production of acid phosphatases and anthocyanin in Arabidopsis. New Phytol. 2011, 189, 1084–1095. [Google Scholar] [CrossRef] [PubMed]
- Roldán, M.; Dinh, P.; Leung, S.; McManus, M.T. Ethylene and the responses of plants to phosphate deficiency. AoB Plants 2013, 5. [Google Scholar] [CrossRef]
- Wang, N.; Cui, Y.; Liu, Y.; Fan, H.; Du, J.; Huang, Z.; Yuan, Y.; Wu, H.; Ling, H. Requirement and functional redundancy of Ib sibgroup bHLH proteins for iron deficiency responses and uptake in Arabidopsis thaliana. Mol. Plant 2013, 6, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Lingam, S.; Mohrbacher, J.; Brumbarova, T.; Potuschak, T.; Fink-Straube, C.; Blondet, E.; Genschik, P.; Bauer, P. Interaction between the bHLH transcription factor FIT and the ETHILENE INSENSITIVE3/ETHYLENE INSENSITIRE3-LIKE1 reveals molecular linkage between the regulation of iron acquisition and ethylene signaling in Arabidopsis. Plant Cell 2011, 23, 1815–1829. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ou, B.; Zhang, J.; Si, W.; Gu, H.; Qin, G.; Qu, L. The Arabidopsis mediator subunit MED16 regulates iron homeostasis by associating with EIN3/EIL1 through subunit MED25. Plant J. 2014, 77, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P. Roots phenes for enhanced soil exploration and phosphorus acquisition: Tools for future crops. Plant Physiol. 2011, 156, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Graziano, M.; Lamattina, L. Nitric oxide accumulation is required for molecular and physiological responses to iron deficiency in tomato roots. Plant J. 2007, 52, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.W.; Yang, J.L.; Qin, C.; Jin, C.W.; Mo, J.H.; Ye, T.; Zheng, S.J. Nitric oxide acts ownstream of auxin to trigger root ferric-chelate reductase activity in response to iron deficiency in Arabidopsis thaliana. Plant Physiol. 2010, 154, 810–819. [Google Scholar] [CrossRef] [PubMed]
- Bacaicoa, E.; Mora, V.; Zamarreño, A.M.; Fuentes, M.; Casanova, E.; García-Mina, J.M. Auxin: A major player in the shoot-to-root regulation of root Fe-stress physiological responses to Fe deficiency in cucumber plants. Plant Physiol. Biochem. 2011, 49, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Meiser, J.; Lingam, S.; Bauer, P. Post-transcriptional regulation of the Fe deficiency bHLH transcription factor FIT is affected by iron and nitric oxide. Plant Physiol. 2011, 157, 2154–2166. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, L.; Simontacchi, M.; Murgia, I.; Zabaleta, E.; Lamattina, L. Nitric oxide, nitrosyl iron complexes ferritin and frataxin: A well-equipped team to preserve plant iron homeostasis. Plant Sci. 2011, 181, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.B.; Chen, L.Q.; Suo, D.; Li, G.X.; Tang, C.X.; Zheng, S.J. Nitric oxide is the shared signalling molecule in phosphorus- and iron deficiency- induced formation of cluster roots in white lupin (Lupinus albus). Ann. Bot. 2012, 109, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zhang, H.T.; Wang, Y.; Jia, W.S.; Xu, X.F.; Zhang, X.Z.; Han, Z.H. Induction of root Fe (III) reductase activity and proton extrusion by iron deficiency is mediated by auxin-based systemic signallingin Malusxiaojinensis. J. Exp. Bot. 2012, 63, 859–870. [Google Scholar] [CrossRef] [PubMed]
- García, M.J.; Romera, F.J.; Stacey, M.G.; Stacey, G.; Villar, E.; Alcántara, E.; Pérez-Vicente, R. Shoot to root communication is necessary to control the expression of iron-acquisition genes in Strategy I plants. Planta 2013, 237, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Romera, F.J.; García, M.J.; Alcántara, E.; Pérez-Vicente, R. Latest findings about the interplay or auxin, ethylene and nitric oxide in the regulation of Fe deficiency responses by Stratagy I plants. Plant Signal Behav. 2011, 6, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Durret, T.P.; Gassmann, W.; Rogers, E.E. The FRD3-mediated afflux of citrate into the root vasculature is necessary for efficient iron translocation. Plant Physiol. 2007, 144, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Roschzttardtz, H.; Séguéla-Arnaud, M.; Briat, J.F.; Vert, G.; Curie, C. The FRD3 citrate effluxer promotes iron nutrition between simplistically disconnected tissues throughout Arabidopsis development. Plant Cell 2011, 23, 2725–2737. [Google Scholar] [CrossRef] [PubMed]
- Rogers, E.E.; Guerinot, M.L. FRD3, a member of the multidrug and toxin efflux family, controls iron deficiency responses in Arabidopsis. Plant Cell 2002, 14, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Stacey, M.G.; Patel, A.; McClain, W.E.; Mathieu, M.; Remley, M.; Rogers, E.E.; Gassmann, W.; Blevins, D.G.; Satacey, G. The Arabidopsis AtOPT3 protein functions in metal homeostasis and movement of iron to developing seeds. Plant Physiol. 2018, 146, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Stacey, M.G.; Osawa, H.; Patel, A.; Gassmann, W.; Satacey, G. Expression analyses of Aradidopsis oligopeptide transporters during seed germination, vegetative growth and reproduction. Planta 2006, 223, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.; Gayomba, S.R.; Jung, H.; Vimalakumari, N.K.; Piñeros, M.; Craft, E.; Rutzke, M.A.; Danku, J.; Lahner, B.; Punshon, T.; et al. OPT3 is a Phloem-specific iron transporter that is essential for systemic iron signaling and redistriburtion of iron and cadmium in Arabidopsis. Plant Cell 2014. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Cózatl, D.G.; Xie, Q.; Akmakjian, G.Z.; Jobe, T.O.; Patel, A.; Stacey, M.G.; Song, L.; Demoin, D.W.; Jurisson, S.S.; Stacey, G.; et al. OPT3 Is a component of the iron-signaling network between leaves and roots and misregulation of opt3 leads to an over-accumulation of cadmium in seeds. Mol. Plant 2014. [CrossRef] [PubMed]
- He, C.J.; Morgan, P.W.; Drew, M.C. Enhanced sensitivity to ethylene in nitrogen- or phosphate-starved roots of Zea mays L. during aerenchyma formation. Plant Physiol. 1992, 98, 137–142. [Google Scholar] [CrossRef] [PubMed]
- López-Bucio, J.; Hernández-Abreu, E.; Sánchez-Calderón, L.; Nieto-Jacobo, M.F.; Simpson, J.; Herrera-Estrella, L. Phosphate availability alters architecture and causes changes in hormone sensitivity in the Arabidopsis root system. Plant Physiol. 2002, 129, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Baskin, T.I.; Brown, K.M.; Lynch, J.P. Regulation of root elongation under phosphorus stress involves changes in ethylene responsiveness. Plant Physiol. 2003, 131, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Zaid, H.; El Morabet, R.; Diem, H.G.; Arahou, M. Does Ethylene mediate cluster root formation under iron deficiency? Ann. Bot. 2003, 92, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Lynch, J.P.; Brown, K.I.M. Ethylene and phosphorus availability have interacting yet distinct effects on root hair development. J. Exp. Bot. 2003, 54, 2351–2361. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W. From faith to fate: Ethylene signalling in morphogenic responses to P and Fe deficiency. J. Plant Nutr. Soil Sci. 2001, 164, 147–154. [Google Scholar] [CrossRef]
- Nagarajan, V.K.; Smith, A.P. Ethylene’s role in phosphate starvation signaling: More than just a root growth regulator. Plant Cell Physiol. 2012, 53, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dong, J.; Gao, Z.; Liu, D. The Arabidopsis gene Hypersensitive to phosphate starvation 3 encodes ethylene overproduction 1. Plant Cell Physiol. 2012, 53, 1093–1105. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.S.; Gao, Y.; Tian, Q.Y.; Shi, F.L.; Li, L.H.; Zhang, L.Z. Stimulation of root acid phosphatase by phosphorus deficiency is regulated by ethylene in Medicago falcata. Environ. Exp. Bot. 2011, 71, 114–120. [Google Scholar] [CrossRef]
- Poirier, Y.; Thoma, S.; Somerville, C.; Schiefelbein, J. A mutant of Arabidopsis deficient in xylem loading of phosphate. Plant Physiol. 1991, 97, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Delhaize, E.; Randall, P.J. Characterization of a phosphate accumulator mutant of Arabidopsis thaliana. Plant Physiol. 1995, 107, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Burleigh, S.H.; Harrison, M.J. The down-regulation of Mt4.like genes by phosphate fertilization occurs systemically and involves phosphate translocation to the shoots. Plant Physiol. 1999, 119, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.Y.; Lin, W.Y.; Huang, T.K.; Chiou, T.J. MicroRNA-mediated surveillance of phosphate transporters on the move. Trends Plant Sci. 2014, 19, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Wege, S.; Khan, G.A.; Jung, J.Y.; Vogiatzaki, E.; Pradervand, S.; Aller, I.; Meyer, A.J.; Poirier, Y. The EXS domain of PHO1 participates in the response of shoots to phosphate deficiency via a root-to-shoot signal. Plant Physiol. 2016, 170, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.K.; Han, C.L.; Lin, S.I.; Chen, Y.J.; Tsai, Y.C.; Chen, Y.R.; Chen, J.W.; Lin, W.Y.; Chen, P.M.; Liu, T.Y.; et al. Identification of downstream components of ubiquitin-conjugating enzyme PHOSPHATE2 by quantitative membrane proteomic in Arabidopsis roots. Plant Cell 2013, 25, 4044–4060. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Verma, S.; Trivedi, P.K. Involvement of Small RNAs in phosphorus and sulfur sensing, signaling and etress: Current update. Front. Plant Sci. 2017, 8, 285. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Rengel, Z.; Delhaize, E. Uptake and translocation of phosphate by pho2 mutant and wild-type seedlings of Arabidopsis thaliana. Planta 1998, 205, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Liu, W.; Meng, Q.; Chen, A.; Sun, S.B. Identification of microRNAs in six solanaceous plants and their potential link with phosphate and mycorrhizal signalings. J. Integr. Plant Biol. 2014, 56, 1164–1178. [Google Scholar] [CrossRef] [PubMed]
- Shane, M.W.; De Vos, M.; De Roock, S.; Lambers, H. Shoot P status regulates cluster-root growth and citrate exudation in Lupinus albus grown with a divided root system. Plant Cell Environ. 2003, 26, 265–273. [Google Scholar] [CrossRef]
- Chiou, T.J. The role of microRNAs in sensing nutrient stress. Plant Cell Environ. 2007, 30, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Grennan, A.K. Arabidopsis microRNAs. Plant Physiol. 2008, 146, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Sunkar, R.; Zhu, J.K. Novel and stress.regulated microRNAs and other small RNAs from Arabidopsis. Plant Cell 2004, 16, 2001–2019. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Chiou, T.J.; Lin, S.I.; Aung, K.; Zhu, J.K. A miRNA involved in phosphate-starvation response in Arabidopsis. Curr. Biol. 2005, 15, 2038–2043. [Google Scholar] [CrossRef] [PubMed]
- Chiou, T.J.; Aung, K.; Lin, S.I.; Wu, C.C.; Chiang, S.F.; Su, C.L. Regulation of phosphate homeostasis by microRNA in Arabidopsis. Plant Cell 2006, 18, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Navarro, L.; Dunoyer, P.; Jay, F.; Arnold, B.; Dharmasiri, N.; Estelle, M.; Voinnet, O.; Jones, J.D.G. A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 2006, 312, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Sunkar, R.; Kapoor, A.; Zhu, J.K. Post-transcriptional induction of two Cu/Zn superoxide dismutase genes in Arabidopsis in mediated by downregulation of mir398 and important for oxidative stress tolerance. Plant Cell 2006, 18, 2051–2065. [Google Scholar] [CrossRef] [PubMed]
- Bartel, B.; Bartel, D.P. MicroRNAs: At the root of plant development? Plant Physiol. 2003, 132, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Buhtz, A.; Pieritz, J.; Springer, F.; Kehr, J. Phloem small RNAs, nutrient stress responses, and systemic mobility. BMC Plant Biol. 2010, 10, 64. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, X.; Zhang, S.; Korpelainen, H.; Li, C. Physiological and transcriptional responses of two contrasting Populus clones to nitrogen stress. Tree Physiol. 2016, 36, 628–642. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Tiwari, M.; Pandey, A.; Bhatia, C.; Sharma, A.; Trivedi, P.K. MicroRNA858 is a potential regulator of phenyl propanoid pathway and plant development in Arabidopsis. Plant Physiol. 2016, 171, 944–959. [Google Scholar] [CrossRef] [PubMed]
- Dugas, D.; Bartel, B. Sucrose induction of Arabidopsis miR398 repress two Cu/Zn superoxide dismutase. Plant Mol. Biol. 2008, 67, 403–417. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Yoo, B.C.; Kragler, F.; Varkonyi-Gasic, E.; Haywood, V.; Archer-Evans, S.; Lee, Y.M.; Lough, T.J.; Lucas, W.J. A systemic small RNA signaling system in plants. Plant Cell 2004, 16, 1979–2000. [Google Scholar] [CrossRef] [PubMed]
- Kidner, C.A.; Martienssen, R.A. The developmental role of microRNA in plants. Curr. Opin. Plant Biol. 2005, 8, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Rus, A.; Sharkhuu, A.; Yokoi, S.; Karthikeyan, A.S.; Raghothama, K.G.; Baek, D.; Koo, Y.D.; Jin, J.B.; Bressan, R.A.; et al. The Arabidopsis SUMO E3 ligase SIZ controls phosphate deficiency responses. Proc. Natl. Acad. Sci. USA 2005, 102, 7760–7765. [Google Scholar] [CrossRef] [PubMed]
- Sunkar, R.; Li, Y.F.; Jagadeeswaran, G. Functions of microRNAs in plant stress responses. Trends Plant Sci. 2012, 17, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, G.A.; Knight, J.D.; Vances, C.P.; Allan, D.L. Does auxin play a role in the adaptations of white lupin roots to phosphate deficiency? Plant Physiol. 1997, 114, S-31. [Google Scholar]
- Gilbert, G.A.; Knight, J.D.; Vances, C.P.; Allan, D.L. Proteoid root development of phosphorus deficient lupin in mimicked by auxin and phosphonate. Ann. Bot. 2000, 85, 921–928. [Google Scholar] [CrossRef]
- Lamont, B.B. Structure, ecology and physiology of root clusters-a review. Plant Soil 2003, 248, 1–19. [Google Scholar] [CrossRef]
- Neumann, G.; Massonneau, A.; Langlade, N.; Dinkelaker, B.; Hengeler, C.; Römheld, V. Physiological aspects of cluster root function and development in phosphorus-deficient white lupin (Lupinus albus L.). Ann. Bot. 2000, 85, 909–919. [Google Scholar] [CrossRef]
- Skene, K.R.; James, W.M. A comparison of the effects of auxin on cluster root initiation and development in Grevillea robusta Cunn. ex R. Br. (Proteaceae) and in the genus Lupinus (Leguminosae). Plant Soil 2000, 219, 221–229. [Google Scholar] [CrossRef]
- Salama, A.; Wareing, P.F. Effects of mineral nutrition on endogenous cytokinins in plants of sunflower (Helianthus annus L.). J. Exp. Bot. 1979, 30, 971–981. [Google Scholar]
- Horgan, J.M.; Wareing, P.F. Cytokinins and the growth responses of seedlings of Betula pendula Roth. and Acer pseudoplatanus L. to nitrogen and phosphorus deficiency. J. Exp. Bot. 1980, 31, 525–532. [Google Scholar] [CrossRef]
- Martin, A.C.; Del Pozo, J.C.; Iglesias, J.; Rubio, V.; Solano, R.; De la Peña, A.; Leyva, A.; Paz-Ares, J. Influence of cytokinins on the expression of phosphate starvation responsive genes in Arabidopsis. Plant J. 2000, 24, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Ségelá, M.; Briat, J.F.; Vert, G.; Curie, C. Cytokinins negatively regulate the root iron uptake machinery in Arabidopsis through a growth-dependent pathway. Plant J. 2008, 55, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Romera, F.J.; Alcántara, E. Ethylene involvement in the regulation of Fe deficiency stress responses by Strategy I plants. Funct. Plant Biol. 2004, 31, 315–328. [Google Scholar] [CrossRef]
- Bernal, M.; Casero, D.; Singh, V.; Wilson, G.T.; Grande, A.; Yang, H.; Dodani, S.C.; Pellegrini, M.; Peter Huijser, P.; Connolly, E.L.; et al. Transcriptome sequencing identifies SPL7-regulated copper acquisition genes FRO4/FRO5 and the copper dependence of iron homeostasis in Arabidopsis. Plant Cell 2012, 24, 738–761. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Shin, R.; Schachtman, D.P. Ethylene mediates response and tolerance to potassium deprivation in Arabidopsis. Plant Cell 2009, 21, 607–621. [Google Scholar] [CrossRef] [PubMed]
- Maruyama-Nakashita, A.; Nakamura, Y.; Tohge, T.; Saito, K.; Takahashi, H. Arabidopsis SLIM1 is a central transcriptional regulator of plant sulfur response and metabolism. Plant Cell 2006, 18, 3235–3251. [Google Scholar] [CrossRef] [PubMed]
- Moniuszko, G.; Skoneczny, M.; Zientara-Rytter, K.; Wawrzyñska, A.; Glów, D.; Cristescu, S.M.; Harren, F.J.M.; Sirko, A. Tobacco LSU-like protein couples sulphur-deficiency response with ethylene signalling pathway. J. Exp. Bot. 2013, 64, 5173–5182. [Google Scholar] [CrossRef] [PubMed]
- Rai, V.; Sanagala, R.; Sinilal, B.; Yadav, S.; Sarkar, A.K.; Dantu, P.K.; Jain, A. Iron availability affects phosphate deficiency-mediated responses, and evidence of crosstalk with auxin and zinc in Arabidopsis. Plant Cell Physiol. 2015, 56, 1107–1123. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.; Ibrikci, H.; Delgado, A.; Torrent, J.; Sommer, R.; Rashid, A. Significance of phosphorus for agriculture and the environment in the West Asia and North Africa region. Adv. Agron. 2012, 114, 91–153. [Google Scholar]
- Correll, D.L. Phosphorus: A rate limiting nutrient in surface waters. Poultry Sci. 1999, 78, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Foy, R.H. The return of the phosphorus paradigm: Agricultural phosphorus and eutrophication. In Phosphorus: Agriculture and the Environment; Sims, J.T., Sharpley, A.N., Eds.; ASA, CSSA and SSSA: Madison, WI, USA, 2005; pp. 911–939. [Google Scholar]
- Delgado, A.; Scalenghe, R. Aspects of phosphorus transfer from soils in Europe. J. Plant Nutr. Soil Sci. 2008, 171, 552–575. [Google Scholar] [CrossRef]
- Torrent, J.; Barberis, E.; Gil-Sotres, F. Agriculture as a source of phosphorus for eutrophication in southern Europe. Soil Use Manag. 2007, 23, 25–35. [Google Scholar] [CrossRef]
- MacDonald, G.K.; Bennett, E.M.; Potter, P.A.; Ramankutty, N. Agronomic phosphorus imbalances across the world’s croplands. Proc. Natl. Acad. Sci. USA 2011, 108, 3086–3091. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P. Amide biosynthesis in root nodules of temperature legumes. In Prokaryotic Nitrogen Fixation: A Model System for Analysis of a Biological Process; Triplett, E.W., Ed.; Horizon Scientific Press: Wymondham, UK, 2001; pp. 589–607. [Google Scholar]
- Cordell, D.; Drangert, J.O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Rahmatullah, H.; Torrent, J. Phosphorus dynamics and uptake by wheat in a model calcite-ferrihydrite system. Soil Sci. 2000, 165, 803–812. [Google Scholar] [CrossRef]
- Strauss, R.; Brummer, G.; Barrow, N.J. Effects of cristallinity of goethite: II. Rates of sorption and desorption of phosphate. Eur. J. Soil Sci. 1997, 48, 101–114. [Google Scholar] [CrossRef]
- Torrent, J.; Schwertmann, U.; Barron, V. Phosphate sorption by natural hematites. Eur. J. Soil Sci. 1994, 45, 45–51. [Google Scholar] [CrossRef]
- Borggard, O.K. Effect of surface area and mineralogy of iron oxides on their surface charge and anionadsorption properties. Clay Clay Min. 1983, 31, 230–232. [Google Scholar] [CrossRef]
- Borggard, O.K. The influence of iron oxides on phosphate adsorption by soil. J. Soil Sci. 1983, 32, 285–299. [Google Scholar] [CrossRef]
- Brown, J.C.; Lunt, O.R.; Holmes, R.S.; Tiffin, L.O. The bicarbonate ion as an indirect cause of iron chlorosis. Soil Sci. 1959, 88, 260–266. [Google Scholar] [CrossRef]
- Sánchez-Rodriguez, A.R.; del Campillo, M.C.; Torrent, J. The severity of iron chlorosis in sensitive plants is related to soil phosphorus levels. J. Sci. Food Agric. 2014, 94, 2766–2773. [Google Scholar] [CrossRef] [PubMed]
- Mengel, K.; Breiniger, M.T.; Bulb, W. Bicarbonate, the most important factor inducing iron chlorosis in vine grapes on calcareous soils. Plant Soil 1984, 81, 333–344. [Google Scholar] [CrossRef]
- Kovanci, I.; Hakerlerler, H.; Hofner, W. Ursachen der chlorosen a mandarinen (Citrus recitulata ‘blanco’) der aigaaiischen region. Plant Soil 1978, 50, 193–205. [Google Scholar] [CrossRef]
- Mullner, L. Ergebnisse eines chloroseforschungsprojektes. Mitt Klosterneubg. 1979, 29, 141–150. [Google Scholar]
- Mengel, K.; Scherer, H.W.; Malissiovas, N. Die chlorose aus der sicht der bodenchemie und rebenernahrung. Mitt Klosterneubg. 1979, 29, 151–156. [Google Scholar]
- Romera, F.J.; Alcantara, E.; de la Guardia, M.D. Characterization of tolerance to iron chlorosis in different peach rootstocks grown in nutrient solution. Effect of bicarbonate and phosphate. Plant Soil 1991, 130, 121–125. [Google Scholar] [CrossRef]
- Samar, S.M.; Shahabian, M.; Fallahi, E.; Davoodi, M.H.; Bagheri, Y.R.; Noorgholipoor, F. Iron deficiency of apple tree as affected by increasing soil available phosphorus. J. Plant Nutr. 2007, 30, 1–7. [Google Scholar] [CrossRef]
- Balal, R.; Shahid, M.A.; Lidon, V.; Garcia, A.L.; Abbas, T.; Garcia-Sanchez, F.; Ghazanfer, U. Effects of phosphorus fertilization on growth, leaf mineral concentration and xylem-phloem nutrient mobility in two rootstocks of prunus (Prunus persica x Prunus amygdalus and Prunus insititia) in the Mediterranean. Aust. J. Crop Sci. 2011, 5, 1542–1549. [Google Scholar]
- Heuer, S.; Gaxiola, R.; Schilling, R.; Herrera-Estrella, L.; López-Arredondo, D.; Wissuwa, M.; Delhaize, E.; Rouached, H. Improving phosphorus use efficiency: A complex trait with emerging opportunities. Plant J. 2017, 90, 868–885. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucena, C.; Porras, R.; Romera, F.J.; Alcántara, E.; García, M.J.; Pérez-Vicente, R. Similarities and Differences in the Acquisition of Fe and P by Dicot Plants. Agronomy 2018, 8, 148. https://doi.org/10.3390/agronomy8080148
Lucena C, Porras R, Romera FJ, Alcántara E, García MJ, Pérez-Vicente R. Similarities and Differences in the Acquisition of Fe and P by Dicot Plants. Agronomy. 2018; 8(8):148. https://doi.org/10.3390/agronomy8080148
Chicago/Turabian StyleLucena, Carlos, Rafael Porras, Francisco Javier Romera, Esteban Alcántara, María José García, and Rafael Pérez-Vicente. 2018. "Similarities and Differences in the Acquisition of Fe and P by Dicot Plants" Agronomy 8, no. 8: 148. https://doi.org/10.3390/agronomy8080148
APA StyleLucena, C., Porras, R., Romera, F. J., Alcántara, E., García, M. J., & Pérez-Vicente, R. (2018). Similarities and Differences in the Acquisition of Fe and P by Dicot Plants. Agronomy, 8(8), 148. https://doi.org/10.3390/agronomy8080148



