1. Introduction
Loquat (
Eriobotrya japonica Lindl.) is a subtropical evergreen fruit tree that presents an annual cycle reverse to that of the well-known temperate fruit crops. Loquat rests during summer, blooms in autumn, develops its fruit through winter and ripens them in early spring. Regulated deficit irrigation (RDI) is a water-saving strategy that has the potential to modify this unusual phenology, making loquat more profitable thanks to an earliness improvement of harvest dates [
1]. This is possible because RDI advances loquat bloom due to earlier summer rest abandonment [
2].
However, earliness achieved by RDI at bloom results in being noticeably trimmed at harvest in terms of calendar days. In a previous study, we found that the displacement of loquat bloom to an earlier date results in a lower daily accumulation of the heat needed to ripen fruits because fruit maturation takes place then on cooler dates [
3]. Such a phenology effect accounts for most of the partial loss of the precocity previously achieved. However, some differences in heat requirements were still observed between fully-irrigated trees and trees experiencing prolonged postharvest water stress. Because of this unexpected finding, we have decided to compare mild and severe regulated deficit irrigation strategies and deficit irrigations carried out in different months always prior to bloom, to evaluate their effects on chronological and thermal time requirements from flowering to harvest.
As it is well known, poikilothermic organisms require of a certain amount of external heat to develop from one stage to another in their life cycle. Zalom et al. (1983) [
4] proposed five methods of different complexity to estimate the daily accumulation of heat in poikilothermic organisms, considering the minimum and maximum daily air temperature. The heat requirement is frequently quantified by determining the growing degree days (GDD) needed between two developmental stages. For a plant genotype, the GDD are assumed to be constant if cultivation is not otherwise limited [
5]. A valid GDD estimate for a given genotype depends on a precise determination of its base temperature (Tb), the lowest temperature at which development will proceed [
6]. Tb can be determined biologically by following plant development under a controlled temperature in a growth chamber [
7,
8]. This method is not suitable for fruit trees. More commonly, Tb is inferred statistically. The accuracy of Tb estimation can be measured by the least coefficient of variation or by the least standard deviation of GDD resulting from data of different years or locations [
9,
10]. Using this approach, we have determined in a previous work that Tb in ‘Algerie’ loquat is 3 °C [
1].
As stated before, temperature is the main meteorological variable in determining plant development [
11]. However, if the range of temperature is adequate, then other factors such as soil moisture may be important and the reason for the variations in accumulated degree days between sites and years. The different RDI strategies we have applied to the same plant material in the same location may allow therefore determining if the amount of heat needed to mature the fruit is related to the severity and duration of the water stress imposed. Although the previous experiments were not specifically designed with this objective in mind, the evaluation of the effects of several RDI treatments on bloom and harvest date and the estimation of fruit development rate and sink-source parameters should provide some insight into the reasons behind the higher heat demand for fruit ripening in water-stressed loquats.
The aim of this study is to compare calendar days and heat units needed between bloom and ripening in fully- and pre-bloom deficit-irrigated trees and to speculate on the reasons behind the differences found.
2. Materials and Methods
The study was conducted during seven consecutive seasons, from 1999/2000 to 2005/2006, in an experimental orchard of loquat located at the Experimental Station of Cajamar Foundation in El Ejido (Almería, SE Spain; 36°48’ N, 2°43’ W). According to the agroclimatic classification of Papadakis [
12], the area presents a semi-arid subtropical climate with an average rainfall of 231 mm and evaporation from an “A” pan (Epan) of 1922 mm per year. The orchard soil is a well-aerated sandy-loam (72.4% sand, 14.6% loam and 13.0% clay) of pH 7.8. The soil gravimetric moisture content is 13.4% at field capacity and 5.1% at the wilting point.
Seven-year-old (at the beginning of the experimentation) ‘Algerie’ loquat trees grafted on ‘Provence’ quince were used for the experiment. The trees were vase-trained and spaced 5 m × 2.5 m during the first four seasons. After the fourth season, every second tree in the row was removed to minimize canopy shading. Light pruning consisting of a moderate shoot thinning was performed in June, soon after harvest. Loquat is heavily pollinated by bees and bumble bees and may set more than 12 fruits per panicle, so, in addition to pruning, flower and fruit thinning to a level of four to five fruits per panicle were carried out to achieve commercial fruit size.
Nine irrigation treatments were applied. The first treatment was a control with a crop evapotranspiration (ET
c) of about 725 mm, according to an ET
0 of 1283 mm, and the following crop coefficients (Kc): 0.50 from shoot growth to bloom; 0.70 from bloom to fruit set; 0.75 from the fruit growth phase; and 0.70 in fruit ripening [
13]. The second treatment was a prolonged deficit irrigation strategy (RDI-long) from mid-May-June to August (45% water cuts for the period; 20% water deficit on a year basis) proven successful in previous experiences [
13]. This treatment was applied seven consecutive years from season 1999/2000 to season 2005/2006. From 1999/2000 to 2001/2002, a continuous deficit irrigation (CDI) was also applied to the same orchard. In this strategy, the trees suffered the same 20% water cut, but uniformly reduced along the year [
1]. Others sets of data come from trees suffering deficit irrigation either in June (RDIJn), July (RDIJl) or August (RDIAu) (a monthly water cut of 66–75% was established depending on the season). These treatments were applied during seasons 2002/2003 and 2003/2004 [
14]. Finally, three more strategies have been explored for their effects on loquat bloom and harvest dates. In this case, during two more seasons, (2004/2005 and 2005/2006), the trees experienced a deficit irrigation schedule limited to a period of six weeks (from mid-June to the end of July), with different severity (50, 75 and 100% water cut; RDI50, RDI75 and RDI100, respectively) [
15]. A concise description of the treatments is presented in
Table 1. Plant water status was monitored in all treatments by measuring predawn leaf water potential (ψpd) with a pressure chamber (model 3000, Soil Moisture Equipment Corp., Santa Barbara, CA, USA). Measurements were performed in fully-expanded leaves (one per tree) randomly selected but located near the tree trunk. Minimum values of predawn leaf water potential reached by each treatment are reported to illustrate the maximum level of water stress reached. Changes in plant water status along time can be consulted in previous experimentation [
13,
14,
15].
Chronological and thermal time between bloom and harvest were compared for these trees. Chronological time was compared counting the days from full bloom to mean harvest date for each treatment and season. Thermal time was calculated using the double sine method to estimate daily heat summation [
6], in the period between bloom and harvest for each treatment and season incorporating a lower threshold temperature of 3 °C previously determined for loquat [
1]. Maximum and minimum daily temperature used for calculations were retrieved from a nearby weather station. Growing degree days from full bloom to harvest were calculated for each treatment as the average of the values obtained from the seasons available for that particular treatment.
Flowering phenology of six trees per treatment (two per replication) was followed twice per week [
16] using phenological stages described by Cuevas et al. (1997) [
17]. Full bloom and mean harvest dates were established for each treatment based on these observations. Full bloom was dated as the day when more than 50% of the panicles show most of the flowers open. Mean harvest was dated as the day in which 50% of the yield was harvested. Harvest was based on skin color by sight using color charts. Total soluble solids content confirms the appropriateness of the harvest operations. Several operations were needed to pick all fruits, since maturation was not uniform for all fruit in the canopy. Sink and source strength were assessed by flower size and seed number, as well as by leaf number, size and activity (gas exchange), respectively. Flower size was measured by their dry weight, collecting at the balloon stage a subsample of 60 king flowers per treatment (10 flowers per tree) each season. Leaf number was counted in 8–10 tagged shoots per tree and 6 trees per treatment (two per replication). Final leaf size was measured in mature leaves using an AM300 leaf area meter (Dynamax Co., Houston, TX, USA). As in other pome fruit crops, fruit development and final size depend at this regard on the number and size of the leaves of the fruitful shoot and on the photosynthetic activity taking place. Stomatal conductance (gs) using a porometer Model AP4 (Delta-T Devices, Cambridge, UK) was evaluated in season 2004/2005. Tree nutritional status was also determined through foliar analysis carried out on November of every year.
A randomized complete-block design was used for the experiments with three replications per treatment. Each replication consisted of one row of trees. The central trees of each row were chosen for measurements. Analyses of variance were carried to compare control and DI treatments applied each season. A comparison among DI treatments carried out in different seasons is facilitated by presenting the average values and the standard errors of the different parameters. The association between these parameters and heat requirements was explored by means of correlation and regression analyses using the Statistix software package, version 9.0 (Analytical Software, Tallahassee, FL, USA).
3. Results and Discussion
Control trees require an average number of 159 days between bloom and harvest. Water-stressed trees after harvest need more calendar days to ripen their fruits. The average number of days for this period was 168 for RDI-long trees. The most severe deficit irrigation treatments, as RDI100 and RDI75, required also more time than their counterpart treatment (RDI50) and than control trees (
Table 2). The limitation of water stress to a period of six weeks did not limit the increase in time for ripening. Indeed, RDI50, RDI75 and RDI100 trees needed more days to mature their fruits than RDI-long trees despite the prolongation of water deficit period in the latter (
Table 2). The greater the advancement in full bloom achieved by deficit irrigation, the longer the period between bloom and harvest resulted (
Table 2). Such a pattern indicates that the main effect in terms of calendar days is due to a displacement of the reproductive phenology to earlier dates. This pattern is explained because deficit irrigation brings late fruit development and ripening from the warmer days of late April to milder early April days, resulting in the need for more days to reach ripening.
Thermal time, nowadays, provides a common reference of crop heat requirements independent of the calendar days needed for such accumulation. Then, if phenological changes completely explain the longer time for maturing fruits under RDI, the differences among treatments in thermal time should be minimal and not related either to the bloom date or with to duration of the bloom-harvest period. However, what we have found is that trees from most (but not all) deficit irrigation treatments still need more heat to properly ripen their fruits (
Table 2). In this regard, again, a trend for higher heat requirements is observed for more efficient treatments in terms of earlier blooming. For instance, more precocious treatments as RDI100 and RDI75 required more heat than RDI50 for maturing the fruits. Similarly, RDIJn and RDIJl, which advanced the bloom date to a higher extent than RDIAu, needed more heat than trees from RDIAu, the treatment that advanced bloom only one day with respect to control fully-irrigated trees (
Table 2). Minimum (more negative) leaf water potential values of each treatment and season are shown in
Table 3.
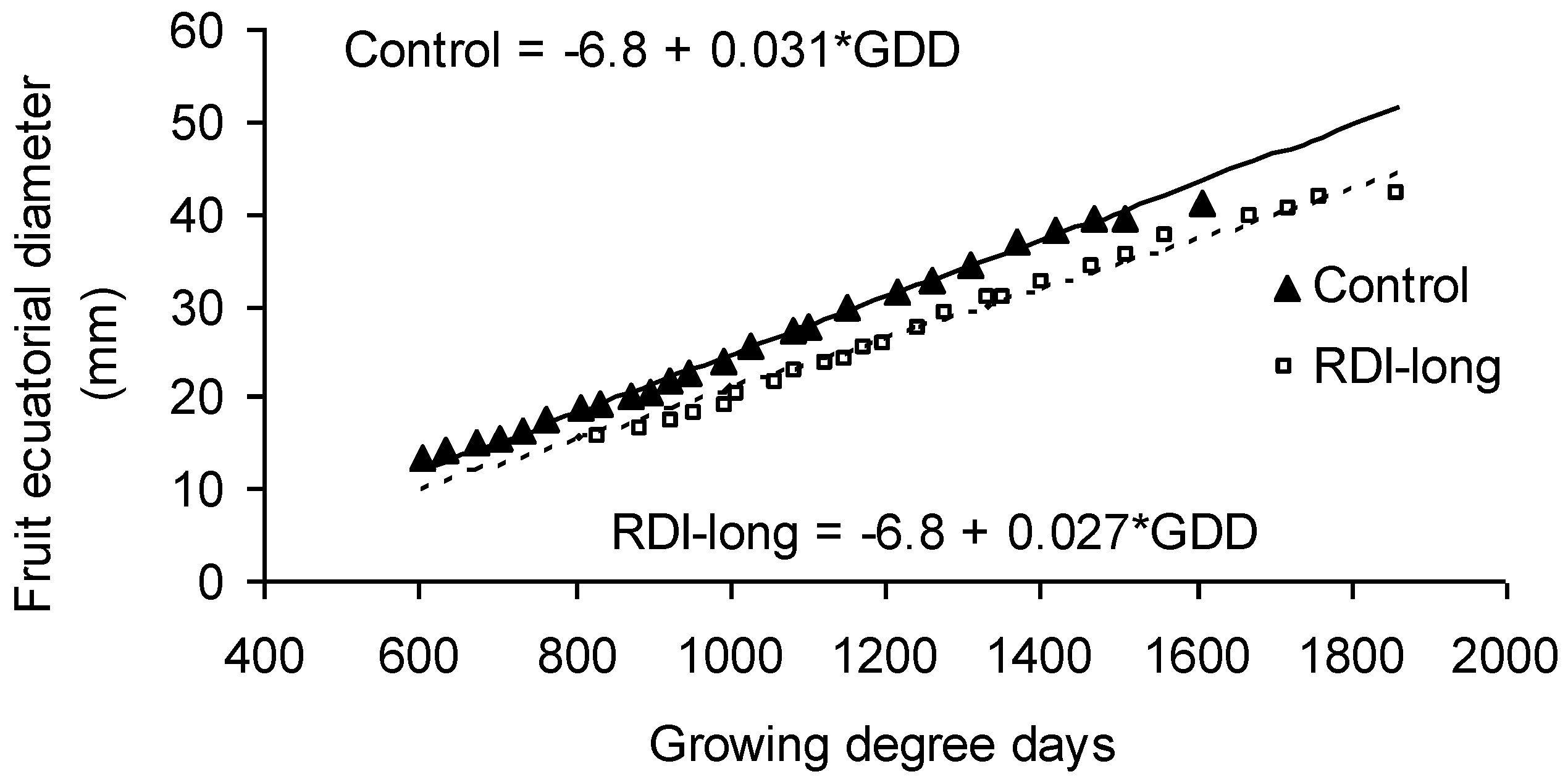
Given that most RDI trees needed more time and heat than control trees to mature their fruits, a slower fruit growth is inferred for the former. The comparison of fruit growth curves in fully-irrigated trees versus RDI-long trees certainly illustrates a reduced development rate in the latter (
p < 0.001;
Figure 1). The differences between these treatments were established in the phase of rapid growth, while the slopes of the curves are not significantly different in the last phase when fruits approach ripening. The lower slope of the curve describing the fruit growth pattern in RDI-long trees cannot be explained by a smaller fruit size in RDI-long. In fact, final fruit size was slightly higher in RDI-long trees compared to controls (48.5 vs. 43.3 g, for RDI-long vs. control, respectively;
p < 0.05) [
1].
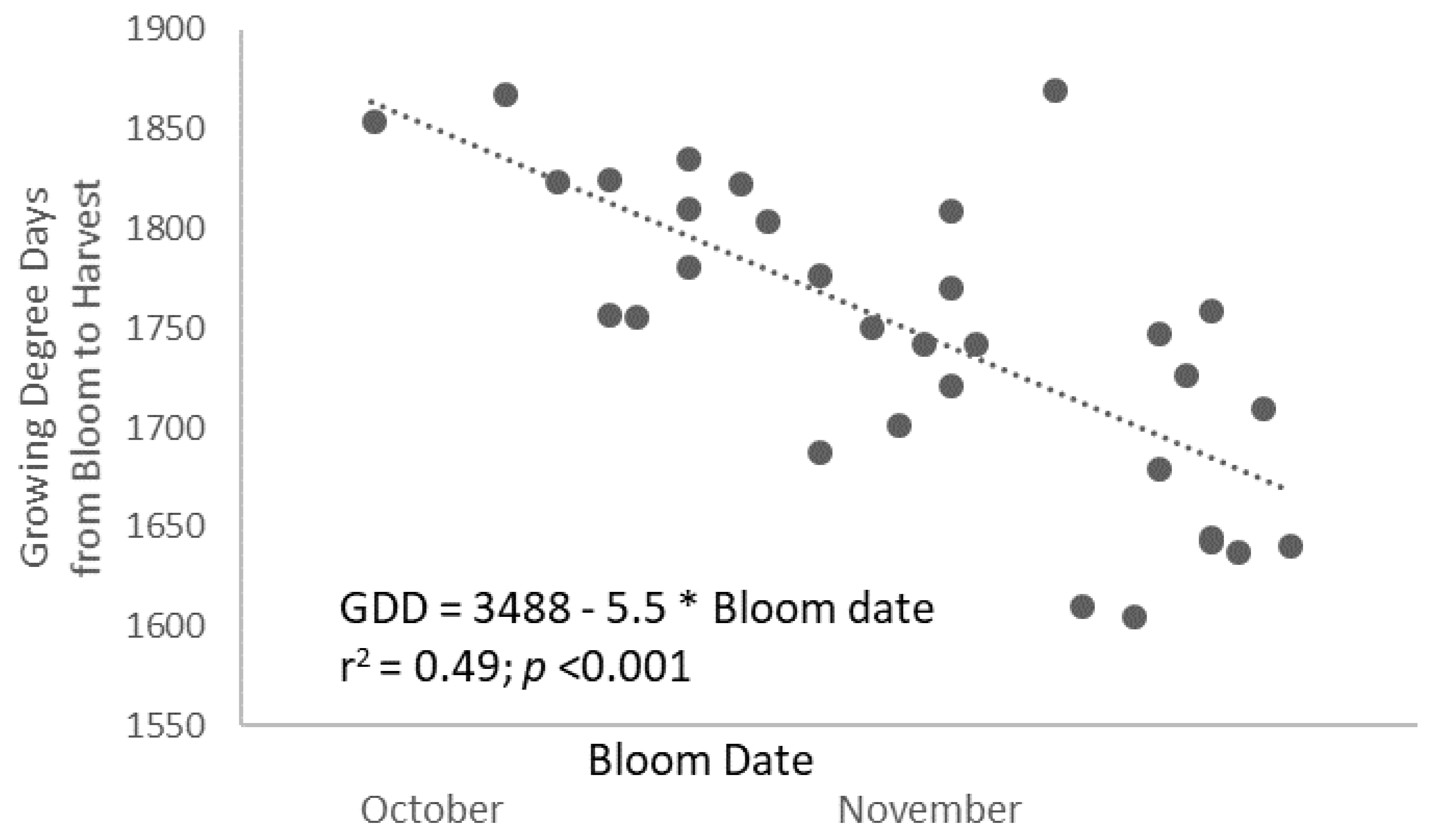
Water stress during the period of fruit development cannot explain either the slower development rate in RDI-long trees because the plants were relieved from deficit irrigation well before bloom, and predawn leaf water potential (ψpd) measurement confirms that the trees recovered from stress shortly after cessation of water deprivation well before bloom (ψpd between −0.2 and −0.3 MPa) [
13]. On the contrary, the common link found between bloom earliness (day of bloom) and heat requirements (as GDD needed to mature fruits) (
r2 = 0.48;
p < 0.001 linear;
Figure 2) and the early establishment of the differences among treatments invoke processes prior to bloom as the determinants of the slower fruit growth and of the higher thermal needs in RDI trees. In this regard, the earlier the bloom was, the higher the heat units needed. On average, every week of bloom advancement implied 39 GDD more needed for ripening the fruits. Using a similar deficit irrigation strategy, Iwasaki and Yamaguchi (2004) [
18] advanced kumquat flowering by causing postharvest water stress of different durations. However, contrary to our observations, an enhancement in fruit maturation due to postharvest deficit irrigation is deduced by the authors since fruits in water-stressed kumquats showed an increase in the color of the rind (although no differences in total soluble solids were found). However, this is not to say that fruit development progressed faster in water-stressed kumquat trees since vegetative growth restraint under deficit irrigation might result also in better fruit color. Water stress during fruit development is largely known to negatively affect fruit size [
19,
20,
21] and advance [
22,
23], rarely delay [
24], fruit maturation. However, no study could be found in the literature dealing with the effects of pre-bloom water stress on fruit development rate.
Negative effects of DI on sink strength or/and source size were then explored to explain the slower rate of fruit development in RDI trees during the initial phase of rapid fruit growth. On the side of the sink, a negative effect of DI on fruit development rate could not be linked to a smaller size of the flower (
Table 4). During our previous investigations, we learned that deficit irrigation during August (RDIAu) was detrimental to our objectives since it did not advance blooming and diminished the size of the flower [
25] with respect to control well-irrigated trees (
p = 0.72 and
p = 0.046; seasons 2002/2003 and 2003/2004, respectively) (
Table 4). For these reasons, this treatment was removed afterward for future experiments. Nonetheless, despite DI being thereafter time-limited up to July as much, treatments such RDIJn, RDIJl, RDI100 and RDI75 still need more heat than the control to ripen their fruits. Therefore, contrary to our expectations, damages in developing flowers hindering future fruit growth via cell number is not a valid explanation for the lower developmental rate observed in some RDI treatments. Seed number per fruit, a clear determinant of fruit sink strength, was not reduced by RDI, confirming that its diminution is also not the real reason of the slower fruit growth rate in RDI trees.
If not the sink, then we turn onto the source. In this regard, mineral and carbohydrate nutrition are known to affect flower and fruit development [
25,
26,
27], and both processes can be affected by irrigation. In our case, a comparison of the nitrogen levels on leaf in control versus RDI-long during seven consecutive years shows no significant effects of a long period of water deficit (
p = 0.55;
Table 4). The low N content found in leaves of treatments RDIJn, RDIJl and RDIAu is related to changes in the fertilization program of seasons 2002/2003 and 2003/2004 and not the restriction of watering (control trees averaged 1.09% in these two seasons).
Although leaf size diminution is a common response to water stress [
28], RDI did not significantly limit the size of single leaves any year (
Table 4), while leaf number was only occasionally reduced (for instance, in season 2000/2001, when control trees formed six more leaves per shoot than RDI-long trees, but one leaf less per shoot than CDI trees;
p = 0.03). The lack of effects of CDI on source size and heat requirements for ripening (in contrast to RDI-long treatment) agrees with the minimal effects caused by CDI on blooming and harvest dates [
1]. In this regard, CDI trees were largely unaffected by DI because the 20% water cut was distributed uniformly along the year, and therefore, neither sink nor source experienced serious stress at any moment in any season (
Table 3). Finally, stored reserves of carbohydrates and the current season’s photosynthates might also greatly contribute to fruit development rate. The amount of stored carbohydrate has not been properly evaluated in our research; however, we found a significant reduction in stomatal conductance (gs) (40% less on average) in response to water stress during RDI period in season 2004/2005 (0.91 vs. 0.53, for controls and RDI-long, respectively). These figures agree with the response of young seedlings of ‘Jiefangzhong’ loquat to moderate water deficits (40% relative soil water content of controls) [
29]. A low stomatal conductance and a reduced photosynthesis have been found to diminish the build-up of starch reserves in the leaves for future fruit development in peach [
30].