Cultivar Variety and Added Potassium Influence the Nutraceutical and Antioxidant Content in Hydroponically Grown Basil (Ocimum basilicum L.)
Abstract
1. Introduction
2. Materials and Methods
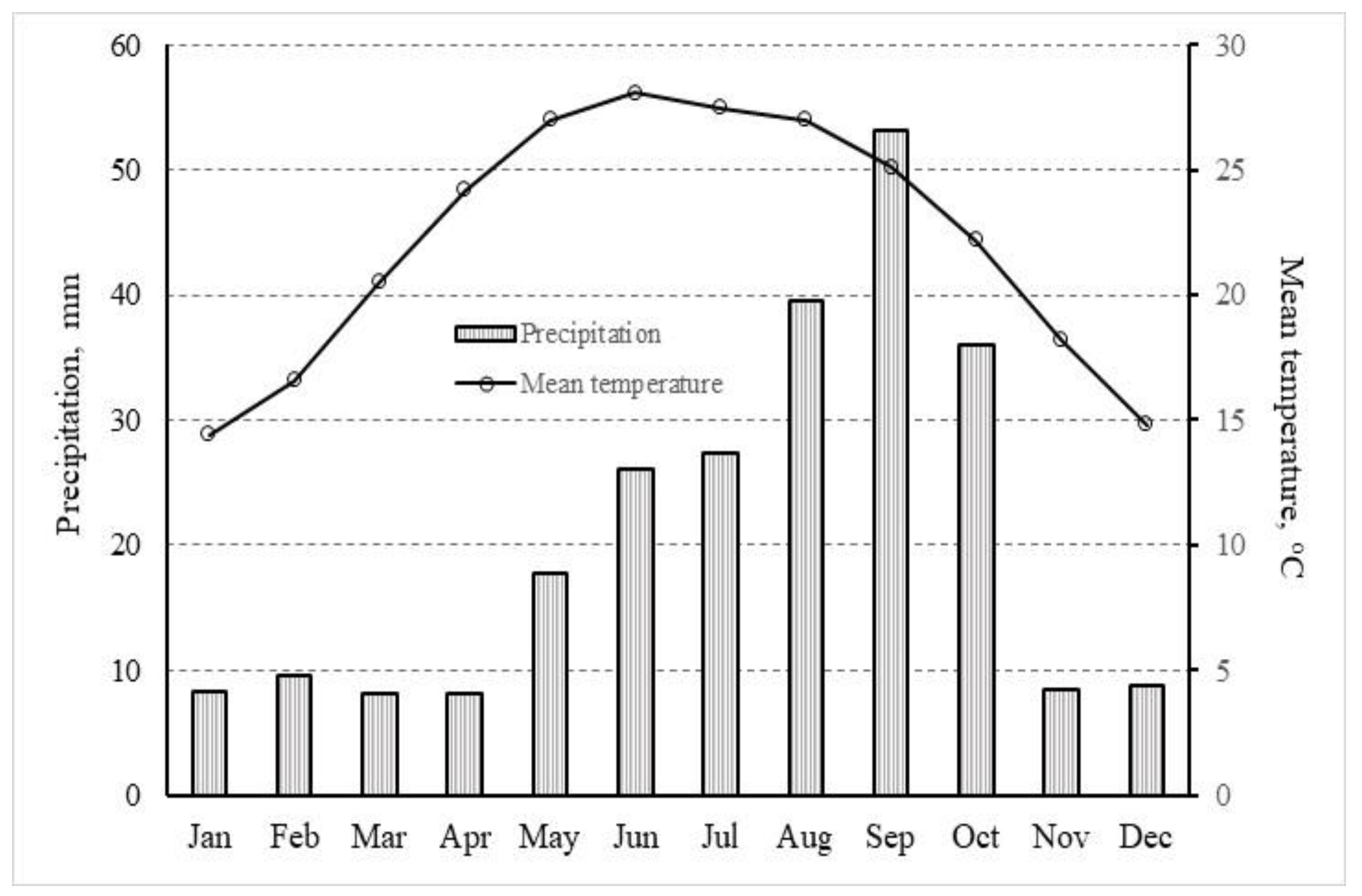
2.1. Study Area
2.2. Experimental Site
2.3. Plant Materials and Treatments
2.4. Sample Preparation for Nutraceutical Compounds and Antioxidant Capacity Analyses
2.5. Chemicals
2.6. Determination of Total Phenolic Compounds
2.7. Total Flavonoids Test
2.8. Anthocyanins Content
2.9. Chlorophyll
2.10. Vitamin C
2.11. Total Antioxidant Capacity
3. Results
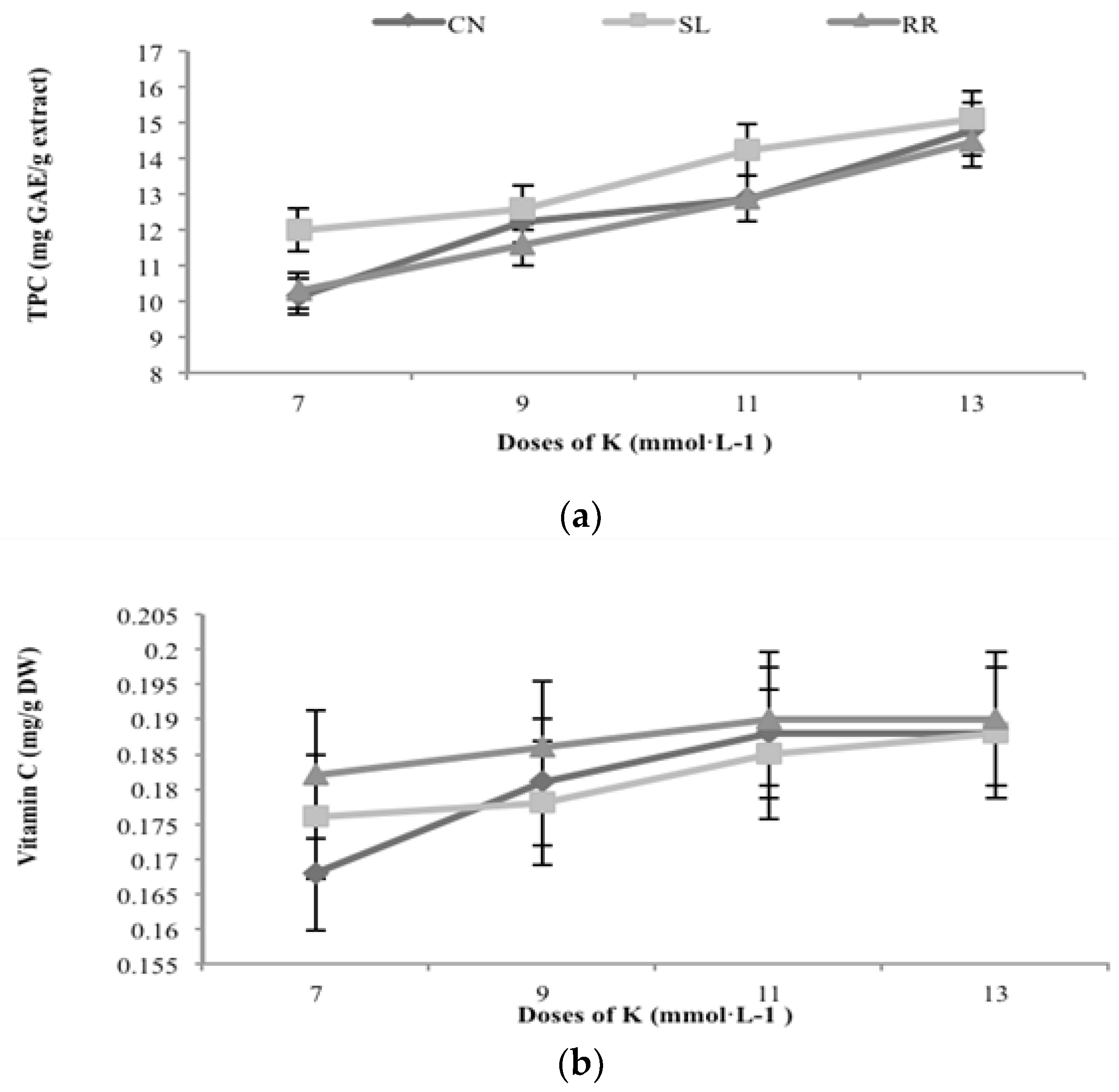
3.1. Total Phenolic Compounds and Flavonoids Content
3.2. Anthocyanins Content
3.3. Chlorophylls
3.4. Vitamin C
3.5. Correlation Analysis
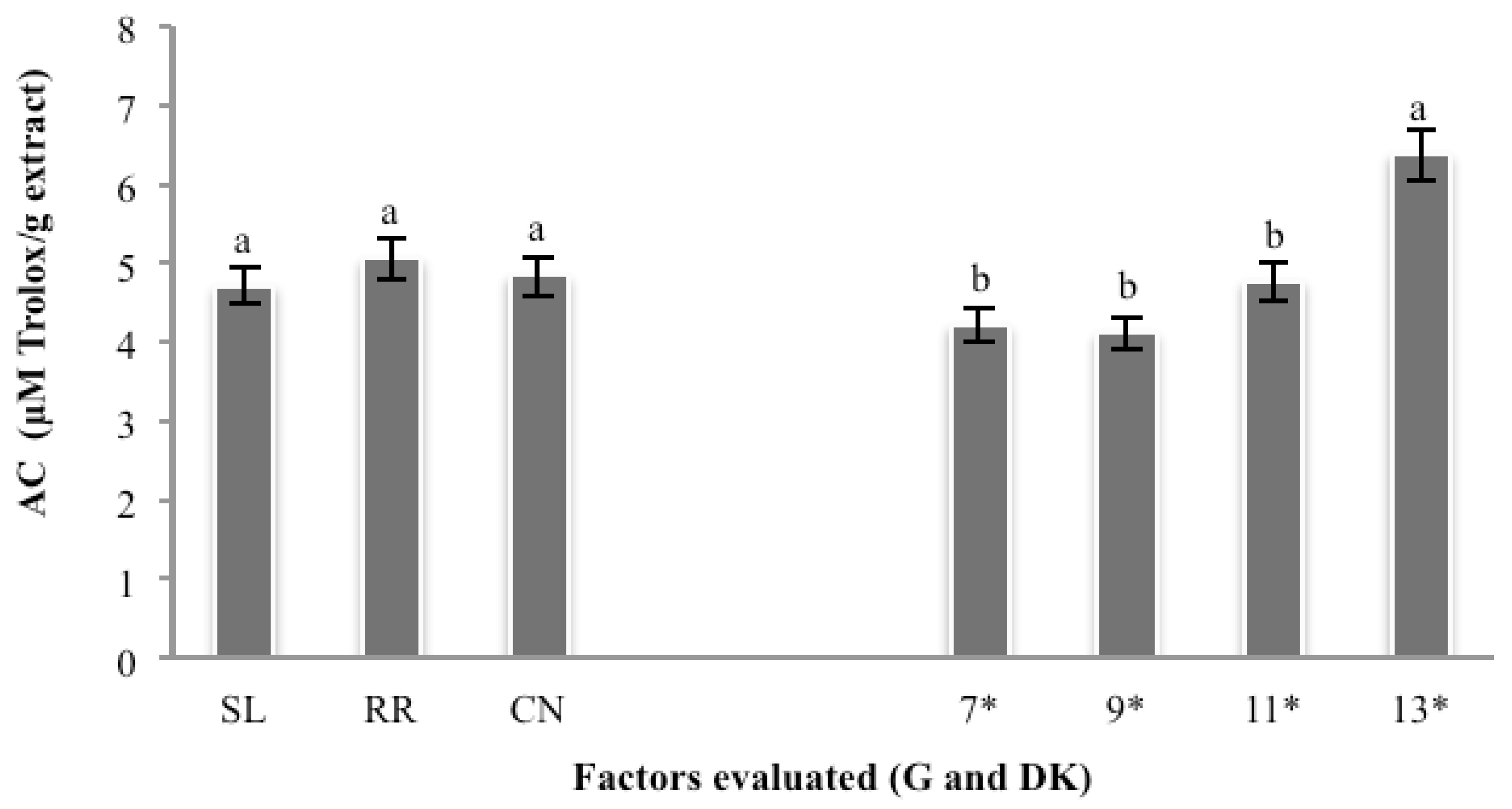
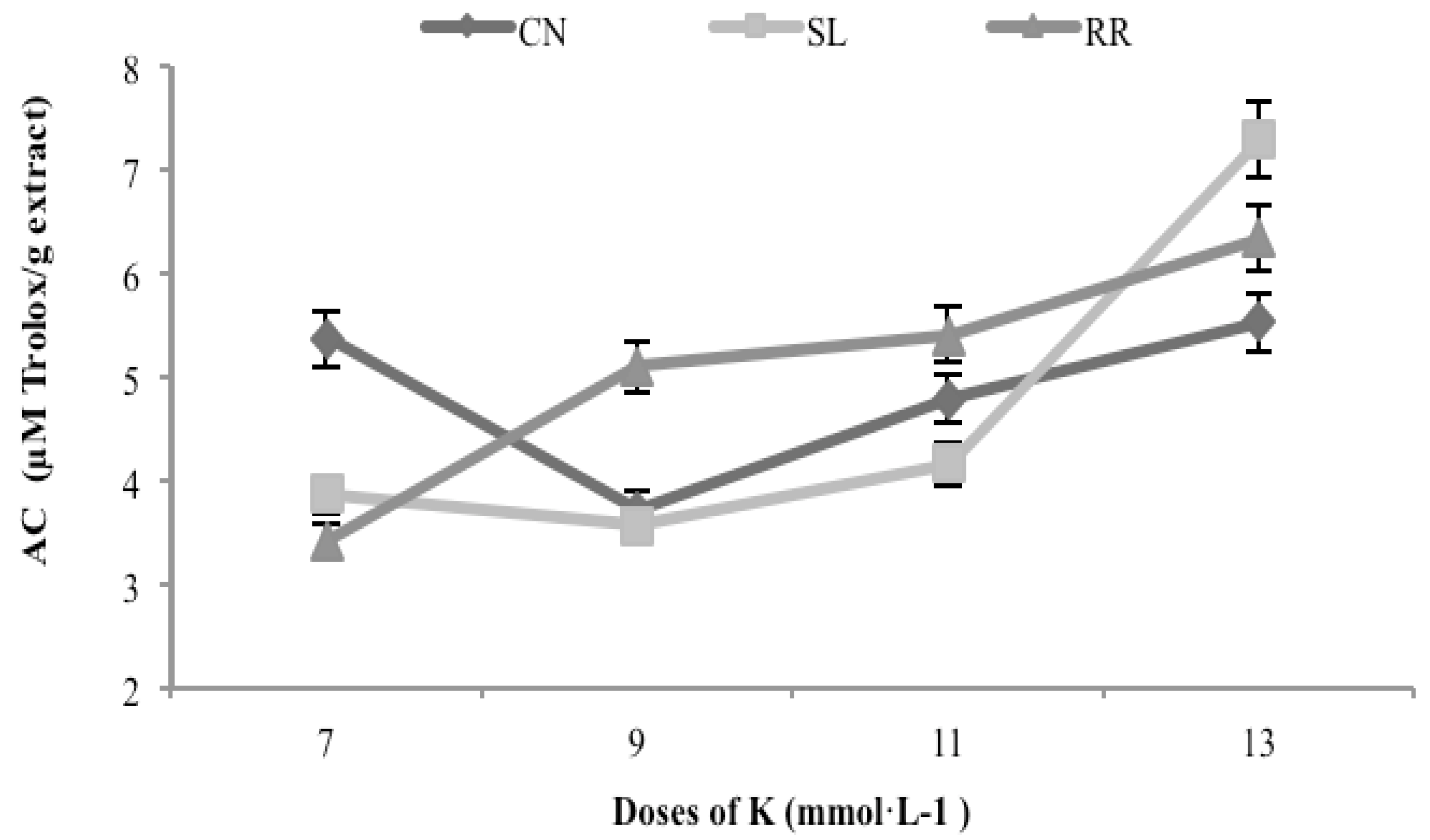
3.6. Antioxidant Capacity
3.7. Interaction Analysis
4. Discussion
4.1. Total Phenolic Compounds, Flavonoids and Anthocyanins Content
4.2. Chlorophyll
4.3. Vitamin C
4.4. Antioxidant Capacity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tilman, D.; Clark, M. Global diets link environmental sustainability and human health. Nature 2014, 515, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Cardeñosa, V.; Barreira, J.; Barros, L.; Arenas-Arenas, F.; Moreno-Rojas, J.; Ferreira, I. Variety and Harvesting Season Effects on Antioxidant Activity and Vitamins Content of Citrus sinensis Macfad. Molecules 2015, 20, 8287–8302. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Fu, Y.; Liu, H. Nutritional status and ion uptake response of Gynura bicolor DC between Porous-tube and traditional hydroponic growth systems. Acta Astronaut. 2015, 113, 13–21. [Google Scholar] [CrossRef]
- Katsoulas, N.; Savvas, D.; Kitta, E.; Bartzanas, T.; Kittas, C. Extension and evaluation of a model for automatic drainage solution management in tomato crops grown in semi-closed hydroponic systems. Comput. Electron. Agric. 2015, 113, 61–71. [Google Scholar] [CrossRef]
- Sgherri, C.; Cecconami, S.; Pinzino, C.; Navari-Izzo, F.; Izzo, R. Levels of antioxidants and nutraceuticals in basil grown in hydroponics and soil. Food Chem. 2010, 123, 416–422. [Google Scholar] [CrossRef]
- Constán-Aguilar, C.; Leyva, R.; Blasco, B.; Sánchez-Rodríguez, E.; Soriano, T.; Ruiz, J.M. Biofortification with potassium: Antioxidant responses during postharvest of cherry tomato fruits in cold storage. Acta Physiol. Plant. 2014, 36, 283–293. [Google Scholar] [CrossRef]
- Demiral, M.A.; Küseoglu, A.T. Effect of Potassium on Yield, Fruit Quality, and Chemical Composition of Greenhouse-Grown Galia Melon. J. Plant Nutr. 2005, 28, 93–100. [Google Scholar] [CrossRef]
- Lester, G.E.; Jifon, J.L.; Makus, D.J. Impact of potassium nutrition on postharvest fruit quality: Melon (Cucumis melo L.) case study. Plant Soil 2010, 335, 117–131. [Google Scholar] [CrossRef]
- Devi, B.S.R.; Kim, Y.J.; Selvi, S.K.; Gayathri, S.; Altanzul, K.; Parvin, S.; Yang, D.U.; Lee, O.R.; Lee, S.; Yang, D.C. Influence of potassium nitrate on antioxidant level and secondary metabolite genes under cold stress in Panax ginseng. Russ. J. Plant Phys. 2012, 59, 318–325. [Google Scholar] [CrossRef]
- Ibrahim, M.; Jaafar, H.; Karimi, E.; Ghasemzadeh, A. Primary, secondary metabolites, photosynthetic capacity and antioxidant activity of the Malaysian herb Kacip Fatimah (Labisia pumila Benth) exposed to potassium fertilization under greenhouse conditions. Int. J. Mol. Sci. 2012, 13, 15321–15342. [Google Scholar] [CrossRef] [PubMed]
- Al-Kateb, H.; Mottram, D.S. The relationship between growth stages and aroma composition of lemon basil Ocimum citriodorum Vis. Food Chem. 2014, 152, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Flanigan, P.M.; Niemeyer, E.D. Effect of cultivar on phenolic levels, anthocyanin composition, and antioxidant properties in purple basil (Ocimum basilicum L.). Food Chem. 2014, 164, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Javanmardi, J.; Stushnoff, C.; Locke, E.; Vivanco, J.M. Antioxidant activity and total phenolic content of Iranian Ocimum accessions. Food Chem. 2003, 83, 547–550. [Google Scholar] [CrossRef]
- Inzunza-López, J.O.; López-Ariza, B.; Valdez-Cepeda, R.D.; Mendoza, B.; Sánchez-Cohen, I.; García-Herrera, G. La variación de las temperaturas extremas en la Comarca Lagunera y cercanías. Rev. Chapingo Ser. Cienc. For. Ambient. 2011, 17, 45–61. [Google Scholar] [CrossRef]
- Atmospheric Science Data Center (ASDC). NASA Surface Meteorology and Solar Energy—Location. NASA Langley Research Center. USA. Available online: https://eosweb.larc.nasa.gov/cgi-bin/sse/grid.cgi?email=skip@larc.nasa.gov (accessed on 27 October 2017).
- Comisión Nacional del Agua (CONAGUA). Servicio Meteorológico Nacional—Normales Climatológicas. México. Available online: http://smn.cna.gob.mx/es/component/content/article?id=42 (accessed on 27 October 2017).
- Lee, J.; Scagel, C.F. Chicoric acid found in basil (Ocimum basilicum L.) leaves. Food Chem. 2009, 115, 650–656. [Google Scholar] [CrossRef]
- Steiner, A.A. A universal method for preparing nutrient solutions of a certain desired composition. Plant Soil 1961, 15, 134–154. [Google Scholar] [CrossRef]
- Rivera, J.R.E.; Stone, M.B.; Stushnoff, C.; Pilon-Smits, E.; Kendall, P.A. Effects of ascorbic acid applied by two hydrocooling methods on physical and chemical properties of green leaf lettuce stored at 5 C. J. Food Sci. 2006, 71, 270–276. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Abdel-Aal, E.S.M.; Hucl, P. A rapid method for quantifying total anthocyanins in blue aleurone and purple pericarp wheats. Cereal Chem. 1999, 76, 350–354. [Google Scholar] [CrossRef]
- Asai, H.; Samson, B.K.; Stephan, H.M.; Songyikhangsuthor, K.; Homma, K.; Kiyono, Y.; Inove, Y.; Shiraiwa, T.; Horie, T. Biochar amendment techniques for upland rice production in Northern Laos: 1. Soil physical properties, leaf SPAD and grain yield. Field Crop Res. 2009, 111, 81–84. [Google Scholar] [CrossRef]
- Davis, S.H.R.; Masten, S.J. Spectrophotometric method for ascorbic acid using dichlorophenolindophenol: Elimination of the interference due to iron. Anal. Chim. Acta 1991, 248, 225–227. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Statistical Analysis System (SAS). SAS/STAT® 9.2. In User’s Guide; SAS Institute Inc.: Cary, NC, USA, 2008; 74p. [Google Scholar]
- Tanaka, Y.; Sasaki, N.; Ohmiya, A. Biosynthesis of plant pigments: Anthocyanins, betalains and carotenoids. Plant J. 2008, 54, 733–749. [Google Scholar] [CrossRef] [PubMed]
- Kwee, E.M.; Niemeyer, E.D. Variations in phenolic composition and antioxidant properties among 15 basil (Ocimum basilicum L.) cultivars. Food Chem. 2011, 128, 1044–1050. [Google Scholar] [CrossRef]
- Nguyen, P.M.; Kwee, E.M.; Niemeyer, E.D. Potassium rate alters the antioxidant capacity and phenolic concentration of basil (Ocimum basilicum L.) leaves. Food Chem. 2010, 123, 1235–1241. [Google Scholar] [CrossRef]
- Phippen, W.B.; Simon, J.E. Anthocyanins in basil (Ocimum basilicum L.). J. Agric. Food Chem. 1998, 46, 1734–1738. [Google Scholar] [CrossRef]
- Kong, J.M.; Chia, L.S.; Goh, N.K.; Chia, T.F.; Brouillard, R. Analysis and biological activities of anthocyanins. Phytochemistry 2003, 64, 923–933. [Google Scholar] [CrossRef]
- Finazzi, G.; Petroutsos, D.; Tomizioli, M.; Flori, S.; Sautron, E.; Villanova, V.; Rolland, N.; Seigneurin-Berny, D. Ions channels/transporters and chloroplast regulation. Cell Calcium 2015, 58, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Martinoia, E.; Szabo, I. Organellar channels and transporters. Cell Calcium 2015, 58, 1–10. [Google Scholar] [CrossRef] [PubMed]
- San Miguel-Chávez, R.; Hernández-Sequera, V.; Rosas-Calleja, D.; Trinidad-Santos, A.; Larqué-Saavedra, A. Efecto del potasio sobre la conductancia estomatica y contenido de clorofila en Amaranto (Amaranthus hypochondriacus L.). Rev. Chapingo Ser. Hortic. 1999, 5, 19–22. [Google Scholar] [CrossRef]
- Limantara, L.; Dettling, M.; Indrawati, R.; Brotosudarmo, T.H.P. Analysis on the Chlorophyll Content of Commercial Green Leafy Vegetables. Proc. Chem. Soc. 2015, 14, 225–231. [Google Scholar] [CrossRef]
- Loewus, F.A. Biosynthesis and metabolism of ascorbic acid in plants and of analogs of ascorbic acid in fungi. Phytochemistry 1999, 52, 193–210. [Google Scholar] [CrossRef]
- Pignocchi, C.; Foyer, C.H. Apoplastic ascorbate metabolism and its role in the regulation of cell signalling. Curr. Opin. Plant Biol. 2003, 6, 379–389. [Google Scholar] [CrossRef]
- Cakmak, I. Potassium for better crop production and quality. Plant Soil 2010, 335, 1–2. [Google Scholar] [CrossRef]
- Yurtseven, E.; Kesmez, G.D.; Ünlükara, A. The effects of water salinity and potassium levels on yield, fruit quality and water consumption of a native central anatolian tomato species (Lycopersicon esculantum). Agric. Water Manag. 2005, 78, 128–135. [Google Scholar] [CrossRef]
- Ali, L.; Alsanius, B.W.; Rosberg, A.K.; Svensson, B.; Nielsen, T.; Olsson, M.E. Effects of nutrition strategy on the levels of nutrients and bioactive compounds in blackberries. Eur. Food Res. Technol. 2012, 234, 33–44. [Google Scholar] [CrossRef]
- Koca, N.; Karaman, S. The effects of plant growth regulators and l-phenylalanine on phenolic compounds of sweet basil. Food Chem. 2015, 166, 515–521. [Google Scholar] [CrossRef] [PubMed]
| K+ Dose | Genotypes Replicates Distribution | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G1,2,3 | ||||||||||||||||||
| D1 | G1 | G3 | G2 | G3 | G1 | G2 | G2 | G3 | G1 | G2 | G3 | G1 | G3 | G1 | G2 | G1 | G2 | G3 |
| D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | D1 | |
| D3 | G1 | G3 | G2 | G3 | G1 | G2 | G2 | G3 | G1 | G2 | G3 | G1 | G3 | G1 | G2 | G1 | G2 | G3 |
| D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | D3 | |
| D2 | G1 | G3 | G2 | G3 | G1 | G2 | G2 | G3 | G1 | G2 | G3 | G1 | G3 | G1 | G2 | G1 | G2 | G3 |
| D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | D2 | |
| D4 | G1 | G3 | G2 | G3 | G1 | G2 | G2 | G3 | G1 | G2 | G3 | G1 | G3 | G1 | G2 | G1 | G2 | G3 |
| D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | D4 | |
| Solution | N | P | K+ | Ca++ | Mg++ | Mn | Cu | Mo | Fe | B | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| mg L−1 | |||||||||||
| Water | 7.67 | nd | 0.22 | 23 | 0.95 | na | na | na | na | na | na |
| ENS | 190.6 | 34 | w, x, y, z | 125 | 27 | 1.6 | 0.11 | 0.5 | 8 | 0.865 | 0.023 |
| Factor | Cultivar | TPC | TF | A | Ch | VC |
|---|---|---|---|---|---|---|
| mg GAE/g Extract | mg QE/g Extract | mg AE/g Extract | Units SPAD | mg AA/g DW | ||
| G | SL | 13.53 a | 5.85 b | 0.69 b | 38.16 a | 0.18 a |
| RR | 12.30 b | 6.09 a | 0.72 a | 32.50 b | 0.18 a | |
| CN | 12.51 b | 6.15 a | 0.74 a | 40.00 a | 0.18 a | |
| Dose, mmol L−1 | ||||||
| DK | 7 | 10.81 d | 5.38 d | 0.64 d | 35.88 a | 0.17 b |
| (mmol·L−1) | 9 | 12.14 c | 5.83 c | 0.69 c | 34.18 a | 0.18 a |
| 11 | 13.38 b | 6.25 b | 0.74 b | 36.02 a | 0.18 a | |
| 13 | 14.80 a | 6.64 a | 0.79 a | 35.82 a | 0.18 a | |
| G × DK | ** | ns | ns | ns | ** |
| Nutraceutical Contents | Cultivars | ||
|---|---|---|---|
| CN | SL | RR | |
| DK-TPC | 0.970 ** | 0.950 ** | 0.998 ** |
| DK-TF | 0.993 ** | 0.993 ** | 0.998 ** |
| DK-A | 0.982 ** | 0.973 ** | 0.983 ** |
| DK-Ch | 0.109 ns | 0.198 ns | 0.107 ns |
| DK-VC | 0.723 * | 0.847 ** | 0.847 ** |
| DK-AC | 0.988 ** | 0.867 ** | 0.923 ** |
| Parameters | TPC | TF | A | Ch | VC | AC |
|---|---|---|---|---|---|---|
| TPC | 1 | |||||
| TF | 0.822 * | 1 | ||||
| A | 0.757 * | 0.979 * | 1 | |||
| Ch | 0.344 | 0.288 | 0.144 | 1 | ||
| VC | 0.523 | 0.708 * | 0.682 * | 0.291 | 1 | |
| AC | 0.549 | 0.621 * | 0.655 * | −0.206 | 0.304 | 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salas-Pérez, L.; Fornari-Reale, T.; Preciado-Rangel, P.; García-Hernández, J.L.; Sánchez-Chávez, E.; Troyo-Diéguez, E. Cultivar Variety and Added Potassium Influence the Nutraceutical and Antioxidant Content in Hydroponically Grown Basil (Ocimum basilicum L.). Agronomy 2018, 8, 13. https://doi.org/10.3390/agronomy8020013
Salas-Pérez L, Fornari-Reale T, Preciado-Rangel P, García-Hernández JL, Sánchez-Chávez E, Troyo-Diéguez E. Cultivar Variety and Added Potassium Influence the Nutraceutical and Antioxidant Content in Hydroponically Grown Basil (Ocimum basilicum L.). Agronomy. 2018; 8(2):13. https://doi.org/10.3390/agronomy8020013
Chicago/Turabian StyleSalas-Pérez, Lilia, Tiziana Fornari-Reale, Pablo Preciado-Rangel, José L. García-Hernández, Esteban Sánchez-Chávez, and Enrique Troyo-Diéguez. 2018. "Cultivar Variety and Added Potassium Influence the Nutraceutical and Antioxidant Content in Hydroponically Grown Basil (Ocimum basilicum L.)" Agronomy 8, no. 2: 13. https://doi.org/10.3390/agronomy8020013
APA StyleSalas-Pérez, L., Fornari-Reale, T., Preciado-Rangel, P., García-Hernández, J. L., Sánchez-Chávez, E., & Troyo-Diéguez, E. (2018). Cultivar Variety and Added Potassium Influence the Nutraceutical and Antioxidant Content in Hydroponically Grown Basil (Ocimum basilicum L.). Agronomy, 8(2), 13. https://doi.org/10.3390/agronomy8020013









