Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Non-Stoichiometric Interpolyelectrolyte Complexes (NPEC) of Chitosan-Polyacrylic Acid (Cs-PAA) and Cs-PAA + Se
2.2. Plant Material and Treatments
2.3. Sampling
2.4. Yield and Biomass Production
2.5. Biochemical Analysis
2.5.1. Total Proteins
2.5.2. Catalase Activity (CAT) (EQ 1.11.1.6)
2.5.3. Glutathione Peroxidase Activity (GPX) (EQ 1.11.1.9)
2.5.4. Glutathione (GSH)
2.5.5 Total Phenols Content
2.6. Selenium Content
2.7. Statistical Analysis
3. Results and Discussion
3.1. Yield and Biomass Production
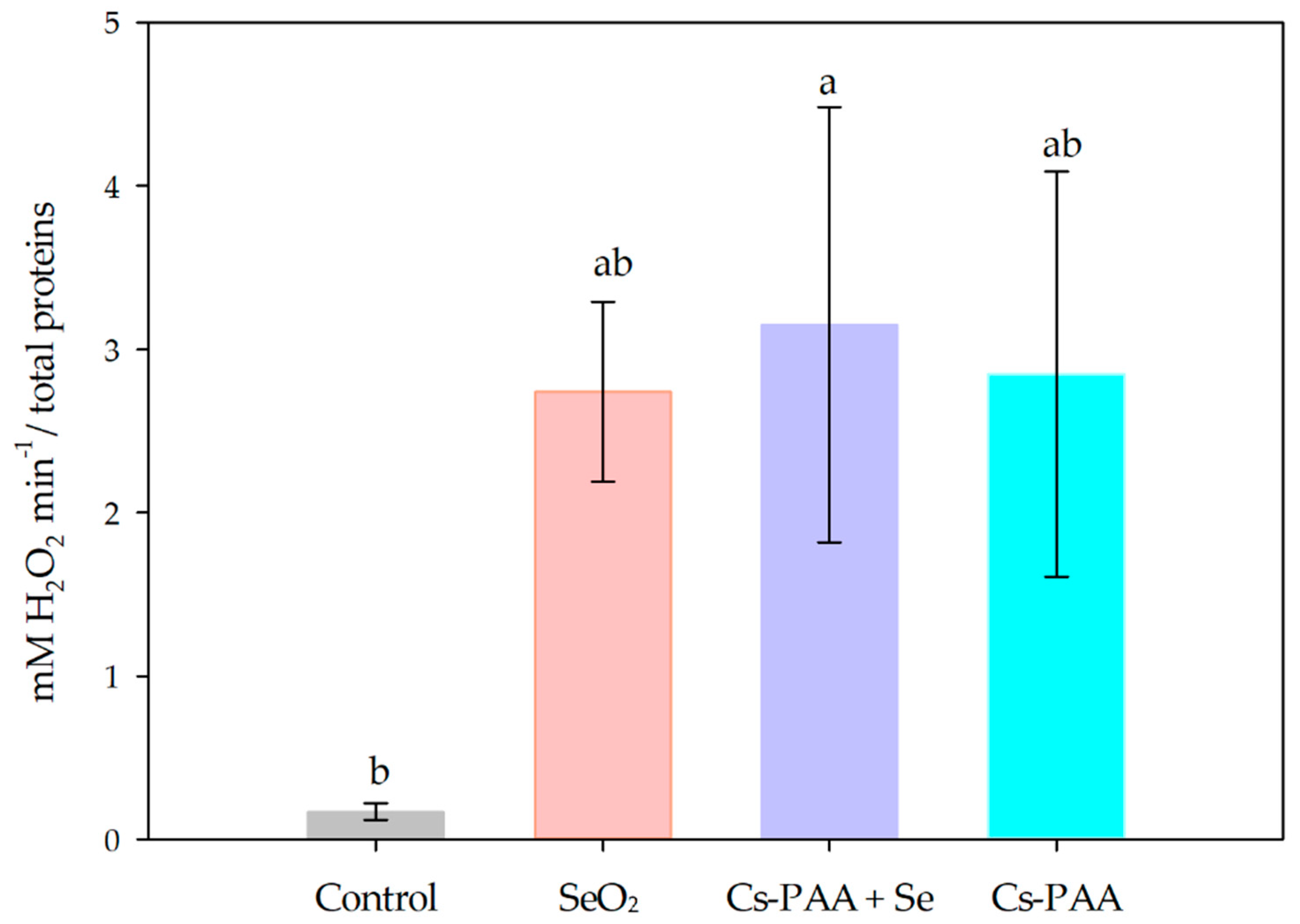
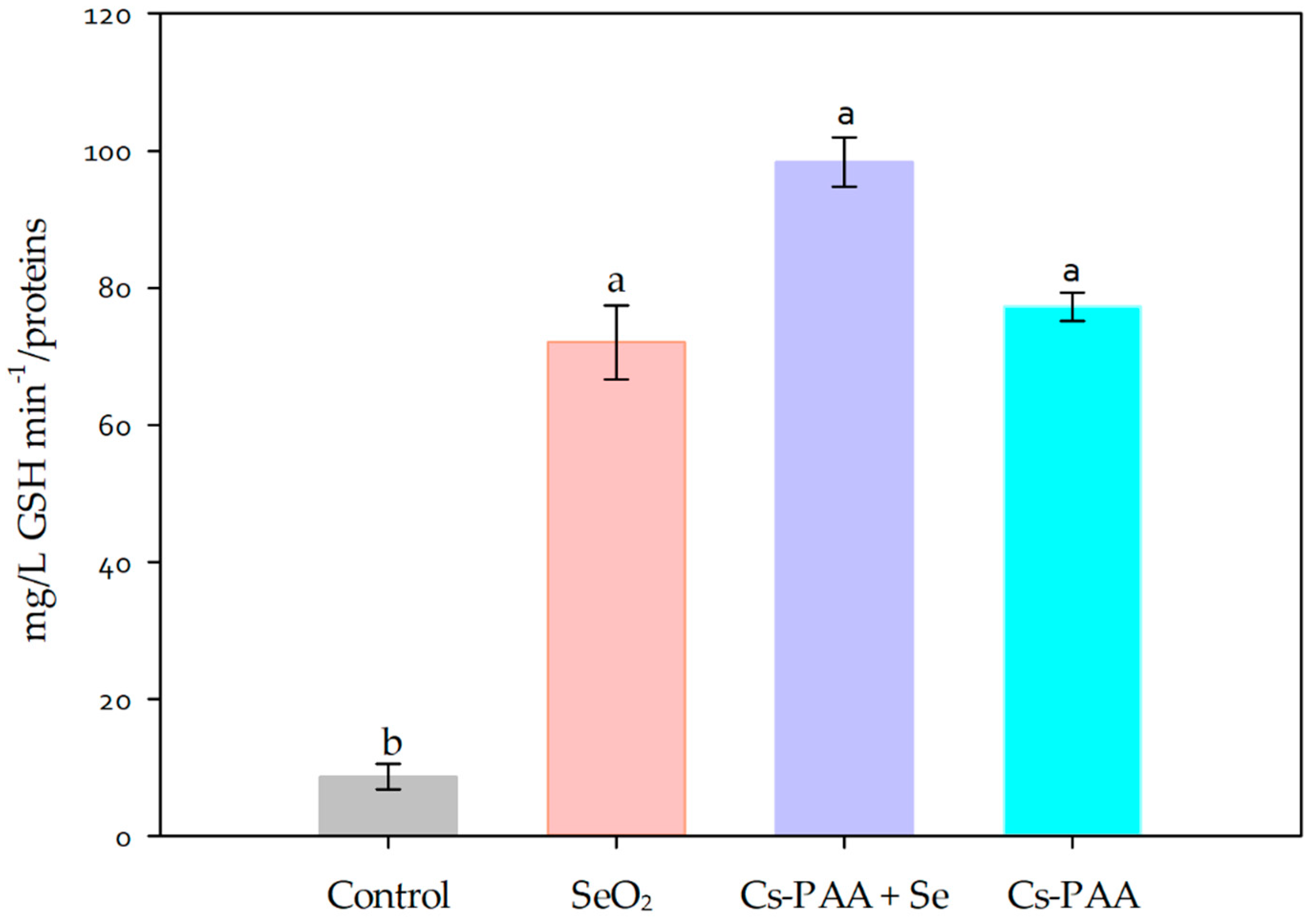
3.2. Biochemical Variables
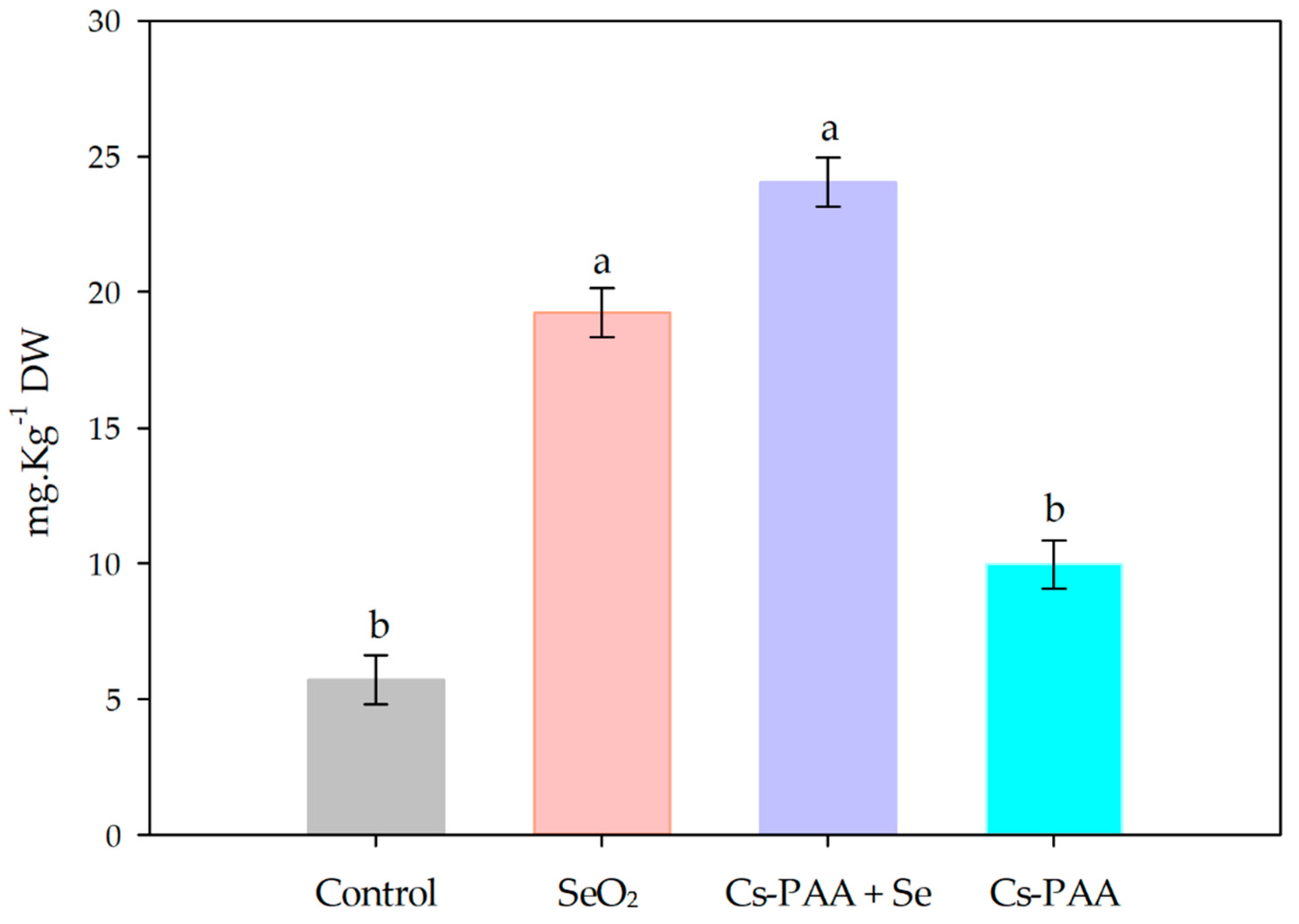
3.3. Selenium Biofortification
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Papp, L.V.; Holmgren, A.; Khanna, K.K. Selenium and selenoproteins in health and disease. Antioxid. Redox Signal. 2010, 12, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Sanmartín, C.; Plano, D.; Sharma, A.K.; Palop, J.A. Selenium compounds, apoptosis and other types of cell death: An overview for cancer therapy. Int. J. Mol. Sci. 2012, 13, 9649–9672. [Google Scholar] [CrossRef] [PubMed]
- El-Ramady, H.; Abdalla, N.; Taha, H.S.; Alshaal, T.; El-Henawy, A.; Faizy, S.E.D.A.; Shams, M.S.; Youssef, S.M.; Shalaby, T.; Bayoumi, Y.; et al. Selenium and nano-selenium in plant nutrition. Environ. Chem. Lett. 2016, 14, 123–147. [Google Scholar] [CrossRef]
- Castillo-Godina, R.G.; Foroughbakhch-Pournavab, R.; Benavides-Mendoza, A. Effect of selenium on elemental concentration and antioxidant enzymatic activity of tomato plants. J. Agric. Sci. Technol. 2016, 18, 233–244. [Google Scholar]
- Mimmo, T.; Tiziani, R.; Valentinuzzi, F.; Lucini, L.; Nicoletto, C.; Sambo, P.; Scampicchio, M.; Pii, Y.; Cesco, S. Selenium biofortification in Fragaria × Ananassa: Implications on strawberry fruits quality, content of bioactive health beneficial compounds and metabolomic profile. Front. Plant Sci. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, M.; Dall’Acqua, S.; Mietto, A.; Pilon-Smits, E.A.H.; Sambo, P.; Masi, A.; Malagoli, M. Selenium fertilization alters the chemical composition and antioxidant constituents of tomato (Solanum lycopersicon L.). J. Agric. Food Chem. 2013, 61, 10542–10554. [Google Scholar] [CrossRef] [PubMed]
- Narváez-Ortiz, W.; Becvort-Azcurra, A.; Fuentes-Lara, L.; Benavides-Mendoza, A.; Valenzuela-García, J.; González-Fuentes, J.; Narváez-Ortiz, W.A.; Becvort-Azcurra, A.A.; Fuentes-Lara, L.O.; Benavides-Mendoza, A.; et al. Mineral composition and antioxidant status of tomato with application of selenium. Agronomy 2018, 8, 185. [Google Scholar] [CrossRef]
- Tomasi, N.; Pinton, R.; Gottardi, S.; Mimmo, T.; Scampicchio, M.; Cesco, S. Selenium fortification of hydroponically grown corn salad (Valerianella locusta). Crop Pasture Sci. 2015, 66, 1128–1136. [Google Scholar] [CrossRef]
- Vinceković, M.; Jalšenjak, N.; Topolovec-Pintarić, S.; Dermić, E.; Bujan, M.; Jurić, S. Encapsulation of biological and chemical agents for plant nutrition and protection: Chitosan/alginate microcapsules loaded with copper cations and trichoderma viride. J. Agric. Food Chem. 2016, 64, 8073–8083. [Google Scholar] [CrossRef] [PubMed]
- Benavides-Mendoza, A.; Burgos-Limón, D.; Ortega-Ortiz, H.; Ramírez, H. Benzoic acid and poly (acrylic acid)—Chitosan in tomato quality and yield in calcareous soil. Terra Latinoam. 2007, 24, 261–268. [Google Scholar]
- Ortega-Ortíz, H.; Benavides-Mendoza, A.; Flores-Olivas, A.; Ledezma-Pérez, A. Use of the interpolyelectrolyte complexes of poly(acrylic acid)—Chitosan as inductors of tolerance against pathogenic fungi in tomato (Lycopersicon esculentum Mill. var. Floradade). Macromol. Biosci. 2003, 3, 566–570. [Google Scholar] [CrossRef]
- Steiner, A.A. A universal method for preparing nutrient solutions of a certain desired composition. Plant Soil 1961, 15, 134–154. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Cansev, A.; Gulen, H.; Eris, A. The activities of catalase and ascorbate peroxidase in olive (Olea europaea L. cv. Gemlik) under low temperature stress. Hortic. Environ. Biotechnol. 2011, 52, 113–120. [Google Scholar] [CrossRef]
- Xue, T.; Hartikainen, H.; Piironen, V. Antioxidative and growth-promoting effect of selenium on senescing lettuce. Plant Soil 2001, 237, 55–61. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1998, 299, 152–178. [Google Scholar] [CrossRef]
- Kalra, P.Y. Handbook of Reference Methods for Plant Analysis; CRC Press: Boca Raton, FL, USA, 1998; Volume 38, ISBN 9781574441246. [Google Scholar]
- Schiavon, M.; Berto, C.; Malagoli, M.; Trentin, A.; Sambo, P.; Dall’Acqua, S.; Pilon-Smits, E.A.H. Selenium biofortification in radish enhances nutritional quality via accumulation of methyl-selenocysteine and promotion of transcripts and metabolites related to glucosinolates, phenolics, and amino acids. Front. Plant Sci. 2016, 7, 1371. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.F.; Zheng, W.J.; Wong, Y.S.; Yang, F. Selenium-induced changes in activities of antioxidant enzymes and content of photosynthetic pigments in spirulina platensis. J. Integr. Plant Biol. 2008, 50, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.M. Selenium pretreatment regulates the antioxidant defense system and reduces oxidative stress on drought-stressed wheat (Triticum aestivum L.) plants. Asian J. Plant Sci. 2014, 13, 120–128. [Google Scholar] [CrossRef]
- Hartikainen, H.; Xue, T.; Piironen, V. Selenium as an anti-oxidant and pro-oxidant in ryegrass. Plant Soil 2000, 225, 193–200. [Google Scholar] [CrossRef]
- Filek, M.; Keskinen, R.; Hartikainen, H.; Szarejko, I.; Janiak, A.; Miszalski, Z.; Golda, A. The protective role of selenium in rape seedlings subjected to cadmium stress. J. Plant Physiol. 2008, 165, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Liu, S.; Zhao, J.; Wang, F.; Du, Y.; Zou, S.; Li, H.; Wen, D.; Huang, Y. Comparative responses to silicon and selenium in relation to antioxidant enzyme system and the glutathione-ascorbate cycle in flowering Chinese cabbage (Brassica campestris L. ssp. chinensis var. utilis) under cadmium stress. Environ. Exp. Bot. 2017, 133, 1–11. [Google Scholar] [CrossRef]
- Khan, M.I.R.; Nazir, F.; Asgher, M.; Per, T.S.; Khan, N.A. Selenium and sulfur influence ethylene formation and alleviate cadmium-induced oxidative stress by improving proline and glutathione production in wheat. J. Plant Physiol. 2015, 173, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Põldma, P.; Moor, U.; Tõnutare, T.; Herodes, K.; Rebane, R. Selenium treatment under field conditions effects mineral nutrition, yield and antioxidant properties of bulb onion (Allium cepa L.). Acta. Sci. Pol. Hortorum Cultus 2013, 12, 167–181. [Google Scholar]
- Ramos, S.J.; Faquin, V.; Guilherme, L.R.G.; Castro, E.M.; Ávila, F.W.; Carvalho, G.S.; Bastos, C.E.A.; Oliveira, C. Selenium biofortification and antioxidant activity in lettuce plants fed with selenate and selenite. Plant. Soil Environ. 2010, 56, 584–588. [Google Scholar] [CrossRef]
- Smoleń, S.; Kowalska, I.; Sady, W. Assessment of biofortification with iodine and selenium of lettuce cultivated in the NFT hydroponic system. Sci. Hortic. 2014, 166, 9–16. [Google Scholar] [CrossRef]
- Broadley, M.R.; White, P.J.; Bryson, R.J.; Meacham, M.C.; Bowen, H.C.; Johnson, S.E.; Hawkesford, M.J.; McGrath, S.P.; Zhao, F.-J.; Breward, N.; et al. Biofortification of UK food crops with selenium. Proc. Nutr. Soc. 2006, 65, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Biomass | PROT | GSH | PHEN |
|---|---|---|---|---|
| (g DW) | (mg/g) | (mg/g) | (µg/g) | |
| Control | 51.35 ± 3.19 a¥ | 2.24 ± 0.56 a | 0.81 ± 0.02 a | 63.51 ±7.49 ab |
| SeO2 | 51.67 ± 2.43 a | 2.87 ± 0.51 a | 0.83 ± 0.01 a | 88.4 ± 14.05 a |
| Cs-PAA + SeO2 | 47.27 ± 4.12 a | 2.05 ± 0.35 a | 0.95 ± 0.38 a | 61.1 ± 11.11 ab |
| Cs-PAA | 50.17 ± 2.41 a | 3.57 ± 0.82 a | 0.97 ± 0.58 a | 48.76 ± 6.81 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leija-Martínez, P.; Benavides-Mendoza, A.; Cabrera-De La Fuente, M.; Robledo-Olivo, A.; Ortega-Ortíz, H.; Sandoval-Rangel, A.; González-Morales, S. Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes. Agronomy 2018, 8, 275. https://doi.org/10.3390/agronomy8120275
Leija-Martínez P, Benavides-Mendoza A, Cabrera-De La Fuente M, Robledo-Olivo A, Ortega-Ortíz H, Sandoval-Rangel A, González-Morales S. Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes. Agronomy. 2018; 8(12):275. https://doi.org/10.3390/agronomy8120275
Chicago/Turabian StyleLeija-Martínez, Paola, Adalberto Benavides-Mendoza, Marcelino Cabrera-De La Fuente, Armando Robledo-Olivo, Hortensia Ortega-Ortíz, Alberto Sandoval-Rangel, and Susana González-Morales. 2018. "Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes" Agronomy 8, no. 12: 275. https://doi.org/10.3390/agronomy8120275
APA StyleLeija-Martínez, P., Benavides-Mendoza, A., Cabrera-De La Fuente, M., Robledo-Olivo, A., Ortega-Ortíz, H., Sandoval-Rangel, A., & González-Morales, S. (2018). Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes. Agronomy, 8(12), 275. https://doi.org/10.3390/agronomy8120275








