Archaeological Starch
Abstract
1. Introduction
2. Properties of Starch Relevant to Archaeology
3. Starch as an Archaeological Tool
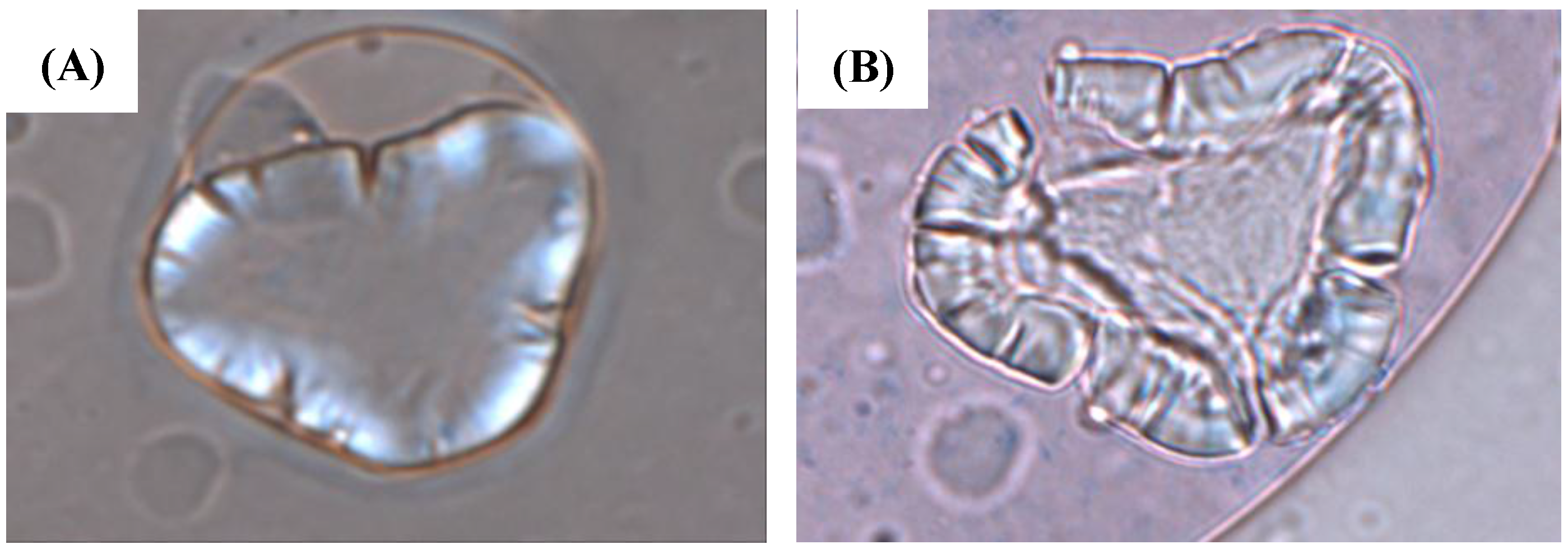
4. Analysis of Archaeological Starch
5. Starch from Archaeological Materials
5.1. Residues from Grinding Stones and Pots
5.2. Dental Calculus
6. Starch and the Evolution of the Modern Human Phenotype
7. Challenges in the Use of Starch in Archaeology
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bruier, F.L. New Clues to stone tool function: Plant and animal residues. Am. Antiq. 1976, 41, 478–484. [Google Scholar] [CrossRef]
- Hardy, K.; Brand Miller, J.; Brown, K.J.; Thomas, M.G.; Copeland, L. The importance of dietary carbohydrate in human evolution. Q. Rev. Biol. 2015, 90, 251–268. [Google Scholar] [CrossRef] [PubMed]
- Copeland, L.; Blazek, J.; Salman, H.; Tang, C.M. Form and functionality of starch. Food Hydrocoll. 2009, 23, 1527–1534. [Google Scholar] [CrossRef]
- Delcour, J.A.; Hoseney, R.C. Principles of Cereal Science and Technology, 3rd ed.; AACC-International: St Paul, MN, USA, 2010; Chapter 2; pp. 23–51. ISBN 978-1-891127-63-2. [Google Scholar]
- Perez, S.; Bertoft, E. The molecular structures of starch components and their contribution to the architecture of starch granules: A comprehensive review. Starch/Stärke 2010, 62, 389–420. [Google Scholar] [CrossRef]
- Eliasson, A.-C.; Bergenståhl, B.; Nilsson, L.; Sjöö, M. From molecules to products: Some aspects of structure–function relationships in cereal starches. Cereal Chem. 2013, 90, 326–334. [Google Scholar] [CrossRef]
- Bertoft, E. Understanding starch structure: Recent progress. Agronomy 2017, 7, 56. [Google Scholar] [CrossRef]
- Tetlow, I.J.; Emes, M.J. Starch Biosynthesis in the Developing Endosperms of Grasses and Cereals. Agronomy 2017, 7, 81. [Google Scholar] [CrossRef]
- Geigenberger, P. Regulation of Starch Biosynthesis in Response to a Fluctuating Environment. Plant Physiol. 2011, 155, 1566–1577. [Google Scholar] [CrossRef] [PubMed]
- Beckles, D.B.; Thitisaksakul, M. How environmental stress affects starch composition and functionality in cereal endosperm. Starch/Stärke 2014, 66, 58–71. [Google Scholar] [CrossRef]
- Hoover, R. The impact of heat-moisture treatment on molecular structures and properties of starches isolated from different botanical sources. Crit. Rev. Food Sci. Nutr. 2010, 50, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Copeland, L. Molecular disassembly of starch granules during gelatinization and its effect on starch digestibility: A review. Food Funct. 2013, 4, 1564–1580. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, C.; Copeland, L.; Niu, Q.; Wang, S. Starch Retrogradation: A Comprehensive Review. Comp. Rev. Food Sci. Food Saf. 2015, 14, 568–585. [Google Scholar] [CrossRef]
- Goesaert, H.; Brijs, K.; Veraverbeke, W.S.; Courtin, C.M.; Gebruers, K.; Delcour, J.A. Wheat flour constituents: How they impact bread quality and how to impact their functionality. Trends Food Sci. Technol. 2005, 16, 12–30. [Google Scholar] [CrossRef]
- Cummings, J.H.; Englyst, H.N. Gastrointestinal effects of food carbohydrate. Am. J. Clin. Nutr. 1995, 61, 938S–945S. [Google Scholar] [PubMed]
- Tester, R.F.; Qi, X.; Karkalas, J. Hydrolysis of native starches with amylases. Anim. Feed Sci. Technol. 2006, 130, 39–54. [Google Scholar] [CrossRef]
- Butterworth, P.J.; Warren, F.J.; Ellis, P.R. Human α-amylase and starch digestion: An interesting marriage. Starch/Stärke 2011, 63, 395–405. [Google Scholar] [CrossRef]
- Wang, S.; Wang, S.; Liu, L.; Wang, S.; Copeland, L. Structural Orders of Wheat Starch Do Not Determine the In Vitro Enzymatic Digestibility. J. Agric. Food Chem. 2017, 65, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Balter, M. Starch Reveals Crop Identities. Science 2007, 316, 1834. [Google Scholar] [CrossRef] [PubMed]
- Denham, T.P.; Haberle, S.G.; Lentfer, C.; Fullagar, R.; Field, J.; Therin, M.; Porch, N.; Winsborough, B. Origins of agriculture at Kuk Swamp in the Highlands of New Guinea. Science 2003, 301, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Dickau, R.; Ranere, A.J.; Cooke, R.G. Starch grain evidence for the preceramic dispersals of maize and root crops into tropical dry and humid forests of Panama. Proc. Natl. Acad. Sci. USA 2007, 104, 3651–3656. [Google Scholar] [CrossRef] [PubMed]
- Fullagar, R.; Field, J.; Denham, T.; Lentfer, C. Early and mid-Holocene tool-use and processing of taro (Colocasia esculenta), yam (Dioscorea sp.) and other plants at Kuk Swamp in the highlands of Papua New Guinea. J. Arch. Sci. 2006, 33, 595–614. [Google Scholar] [CrossRef]
- Horrocks, M.; Irwin, G.; Jones, M.; Sutton, D. Starch grains and xylem cells of sweet potato (Ipomoea batatas) and bracken (Pteridium esculentum) in archaeological deposits from northern New Zealand. J. Arch. Sci. 2004, 31, 251–258. [Google Scholar] [CrossRef]
- Horrocks, M.; Nunn, P.D. Evidence for introduced taro (Colocasia esculenta) and lesser yam (Dioscorea esculenta) in Lapita-era (c. 3050–2500 cal. years BP) deposits from Bourewa, southwest Viti Levu Island, Fiji. J. Arch. Sci. 2007, 34, 739–748. [Google Scholar] [CrossRef]
- Horrocks, M.; Grant-Mackie, J.; Matisoo-Smith, E. Introduced taro (Colocasia esculenta) and yams (Dioscorea spp.) in Podtanean (2700–1800 years BP) deposits from Mé Auré Cave (WMD007), Moindou, New Caledonia. J. Arch. Sci. 2008, 35, 169–180. [Google Scholar] [CrossRef]
- Iriarte, J.; Holst, I.; Marozzi, O.; Listopad, C.; Alonso, E.; Rinderknecht, A.; Montana, J. Evidence for cultivar adoption and emerging complexity during the mid-Holocene in the La Plata basin. Nature 2004, 432, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Field, J.; Fullagar, R.; Bestel, S.; Chen, X.; Ma, X. What did grinding stones grind? New light on Early Neolithic subsistence economy in the Middle Yellow River Valley, China. Antiquity 2010, 84, 816–833. [Google Scholar] [CrossRef]
- Loy, T.H.; Spriggs, M.; Wickler, S. Direct evidence for human use of plants 28,000 years ago: Starch residues on stone artefacts from the northern Solomon Islands. Antiquity 1992, 66, 898–912. [Google Scholar] [CrossRef]
- Lu, H.; Yang, X.; Ye, M.; Liu, K.-B.; Xia, Z.; Ren, X.; Cai, L.; Wu, N.; Liu, T.-S. Millet noodles in Late Neolithic China. Nature 2005, 437, 967–968. [Google Scholar] [CrossRef] [PubMed]
- Neumann, K. New Guinea: A cradle of agriculture. Science 2003, 301, 180–181. [Google Scholar] [CrossRef] [PubMed]
- Pearsall, D.M.; Chandler-Ezell, K.; Zeidler, J.A. Maize in ancient Ecuador: Results of residue analysis of stone tools from the Real Alto site. J. Arch. Sci. 2004, 31, 423–442. [Google Scholar] [CrossRef]
- Perry, L.; Dickau, R.; Zarrillo, S.; Holst, I.; Pearsall, D.M.; Piperno, D.R.; Berman, M.J.; Cooke, R.G.; Rademaker, K.; Ranere, A.J.; et al. Starch fossils and the domestication and dispersal of chilli peppers (Capsicum spp. L.) in the Americas. Science 2007, 315, 986–988. [Google Scholar] [CrossRef] [PubMed]
- Piperno, D.R.; Holst, I. The presence of starch grains on prehistoric stone tools from the humid neotropics: Indications of early tuber use and agriculture in Panama. J. Arch. Sci. 1998, 25, 765–776. [Google Scholar] [CrossRef]
- Piperno, D.R.; Ranere, A.J.; Holst, I.; Iriarte, J.; Dickau, R. Starch grain and phytolith evidence for early ninth millennium B.P. maize from the Central Balsas River Valley, Mexico. Proc. Natl. Acad. Sci. USA 2009, 106, 5019–5024. [Google Scholar] [CrossRef] [PubMed]
- Ranere, A.J.; Piperno, D.R.; Holst, I.; Dickau, R.; Iriarte, J. The cultural and chronological context of early Holocene maize and squash domestication in the Central Balsas River Valley, Mexico. Proc. Natl. Acad. Sci. USA 2009, 106, 5014–5018. [Google Scholar] [CrossRef] [PubMed]
- Zarrillo, S.; Pearsall, D.M.; Raymond, J.S.; Tisdale, M.A.; Quon, D.J. Directly dated starch residues document early formative maize (Zea mays L.) in tropical Ecuador. Proc. Natl. Acad. Sci. USA 2008, 105, 5006–5011. [Google Scholar] [CrossRef] [PubMed]
- Duncan, N.; Pearsall, D.M.; Benfer, R.A., Jr. Gourd and squash artifacts yield starch grains of feasting foods from preceramic Peru. Proc. Natl. Acad. Sci. USA 2009, 106, 13202–13206. [Google Scholar] [CrossRef] [PubMed]
- Piperno, D.R.; Dillehay, T.D. Starch grains on human teeth reveal early broad crop diet in northern Peru. Proc. Natl. Acad. Sci. USA 2008, 105, 19622–19627. [Google Scholar] [CrossRef] [PubMed]
- Barker, G.; Barton, H.; Bird, M.; Daly, P.; Datan, I.; Dykes, A.; Farr, L.; Gilbertson, D.; Harrisson, B.; Hunt, C.; et al. The ‘human revolution’ in lowland tropical Southeast Asia: The antiquity and behavior of anatomically modern humans at Niah Cave (Sarawak, Borneo). J. Hum. Evol. 2007, 52, 243–261. [Google Scholar] [CrossRef] [PubMed]
- Fullagar, R.; Field, J. Pleistocene seed-grinding implements from the Australian arid zone. Antiquity 1997, 71, 301–310. [Google Scholar] [CrossRef]
- Henry, A.; Brooks, A.; Piperno, D.R. Microfossils in calculus demonstrate consumption of plants and cooked foods in Neanderthal diets (Shanidar III, Iraq; Spy I and II, Belgium). Proc. Natl. Acad. Sci. USA 2011, 108, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Hardy, K.; Blakeney, T.; Copeland, L.; Kirkham, J.; Wrangham, R.; Collins, M. Starch granules, dental calculus and new perspectives on ancient diet. J. Arch. Sci. 2009, 36, 248–255. [Google Scholar] [CrossRef]
- Hardy, K.; Buckley, S.; Collins, M.J.; Estalrrich, A.; Brothwell, D.; Copeland, L.; García-Tabernero, A.; García-Vargas, S.; de la Rasilla, M.; Lalueza-Fox, C.; et al. Neanderthal medics? Evidence for food, cooking and medicinal plants entrapped in dental calculus. Naturwissenschaften 2012, 99, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Buckley, S.; Usai, D.; Jakob, T.; Radini, A.; Hardy, K. Dental calculus reveals unique insights into food items, cooking and plant processing in prehistoric central Sudan. PLoS ONE 2014, 9, e100808. [Google Scholar] [CrossRef] [PubMed]
- Hardy, K.; Radini, A.; Buckley, S.; Sarig, R.; Copeland, L.; Gopher, A.; Barkai, R. Dental calculus reveals potential respiratory irritants and ingestion of essential plant-based nutrients at Lower Palaeolithic Qesem Cave Israel. Quat. Int. 2016, 398, 129–135. [Google Scholar] [CrossRef]
- Hardy, K.; Radini, A.; Buckley, S.; Blasco, R.; Copeland, L.; Burjachs, F.; Girbal, J.; Yll, R.; Carbonell, E.; Bermúdez de Castro, J.M. Diet and environment 1.2 million years ago revealed through analysis of dental calculus from Europe’s oldest hominin at Sima del Elefante, Spain. Sci. Nat. 2017, 104, 2. [Google Scholar] [CrossRef] [PubMed]
- Weyrich, L.S.; Duchene, S.; Soubrier, J.; Arriola, L.; Llamas, B.; Breen, J.; Morris, A.G.; Alt, K.W.; Caramelli, D.; Dresely, V.; et al. Neanderthal behaviour, diet and disease inferred from ancient DNA in dental calculus. Nature 2017, 544, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Lombard, M. Evidence of hunting and hafting during the Middle Stone Age at Sibidu Cave, KwaZulu-Natal, South Africa: A multianalytical approach. J. Hum. Evol. 2005, 48, 279–300. [Google Scholar] [CrossRef] [PubMed]
- Mercader, J. Mozambican grass seed consumption during the Middle Stone Age. Science 2009, 326, 1680–1683. [Google Scholar] [CrossRef] [PubMed]
- Piperno, D.R.; Weiss, E.; Holst, I.; Nadel, D. Processing of wild cereal grains in the Upper Palaeolithic revealed by starch grain analysis. Nature 2004, 430, 670–673. [Google Scholar] [CrossRef] [PubMed]
- Revedin, A.; Aranguren, B.; Becattini, R.; Longo, L.; Marconi, E.; Mariotti Lippi, M.; Skakun, N.; Sinitsyn, A.; Spiridonova, E.; Svoboda, J. Thirty thousand-year-old evidence of plant food processing. Proc. Natl. Acad. Sci. USA 2010, 107, 18815–18819. [Google Scholar] [CrossRef] [PubMed]
- Summerhayes, G.; Leavesley, M.; Fairbairn, A.; Mandui, H.; Field, J.; Ford, A.; Fullagar, R. Human Adaptation and Plant Use in Highland New Guinea 49,000 to 44,000 Years Ago. Science 2010, 330, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Wesolowski, V.; Ferraz Mendonça de Souza, S.M.; Reinhard, K.J.; Ceccantini, G. Evaluating microfossil content of dental calculus from Brazilian sambaquis. J. Arch. Sci. 2010, 37, 1326–1338. [Google Scholar] [CrossRef]
- Mercader, J.; Barton, H.; Gillespie, J.; Harris, J.; Kuhn, S.; Tyler, R.; Boesch, C. 4300-Year-old chimpanzee sites and the origins of percussive stone technology. Proc. Natl. Acad. Sci. USA 2007, 104, 3043–3048. [Google Scholar] [CrossRef] [PubMed]
- Lentfer, C.; Therin, M.; Torrence, R. Starch grains and environmental reconstruction: a modern test case from West New Britain, Papua New Guinea. J. Arch. Sci. 2002, 29, 687–698. [Google Scholar] [CrossRef]
- Power, R.C.; Salazar-García, D.C.; Wittig, R.M.; Freiberg, M.; Henry, A.G. Dental calculus evidence of Taï Forest Chimpanzee plant consumption and life history transitions. Sci. Rep. 2015, 5, 15161. [Google Scholar] [CrossRef] [PubMed]
- Haslam, M. Potential misidentification of in situ archaeological tool-residues: Starch and conidia. J. Arch. Sci. 2006, 33, 114–121. [Google Scholar] [CrossRef]
- Copeland, L.; Blakeney, A.B.; Hardy, K. Exposure of starch granules from dental calculus to alpha-amylase. The University of Sydney: Sydney, Australia, Unpublished data. 2017. [Google Scholar]
- Reichert, E.T. The Differentiation and Specificity of Starches in Relation to Genera, Species, etc.: Stereochemistry Applied to Protoplasmic Processes and Products and As a Strictly Scientific Basis for the Classification of Plants and Animals; Carnegie Institution of Washington: Washington, DC, USA, 1913. [Google Scholar]
- Jane, J.L.; Kasemasuwan, T.; Leas, S.; Zobel, H.; Robyt, J.F. Anthology of starch granule morphology by scanning electron microscopy. Starch/Stärke 1994, 46, 121–129. [Google Scholar] [CrossRef]
- Torrence, R.; Wright, R.; Conway, R. Identification of starch granules using image analysis and multivariate techniques. J. Arch. Sci. 2004, 31, 519–532. [Google Scholar] [CrossRef]
- Arráiz, H.; Barbarin, N.; Pasturel, M.; Beaufort, L.; Domínguez-Rodrigo, M.; Barboni, D. Starch granules identification and automatic classification based on an extended set of morphometric and optical measurements. J. Arch. Sci. Rep. 2016, 7, 169–179. [Google Scholar] [CrossRef]
- Coster, A.C.; Field, J.H. What starch grain is that? A geometric morphometric approach to determining plant species origin. J. Arch. Sci. 2015, 58, 9–25. [Google Scholar] [CrossRef]
- Wilson, J.; Hardy, K.; Allen, R.; Copeland, L.; Wrangham, R.; Collins, M. Automated classification of starch granules using supervised pattern recognition of morphological properties. J. Arch. Sci. 2010, 37, 594–604. [Google Scholar] [CrossRef]
- Nadel, D.; Piperno, D.R.; Holst, I.; Snir, A.; Weiss, E. New evidence for the processing of wild cereal grains at Ohalo II, a 23,000-year-old campsite on the shore of the Sea of Galilee, Israel. Antiquity 2012, 86, 990–1003. [Google Scholar] [CrossRef]
- Fullagar, R.; Hayes, E.; Stephenson, B.; Field, J.; Matheson, C.; Stern, N.; Fitzsimmons, K. Evidence for Pleistocene seed grinding at Lake Mungo, south-eastern Australia. Archaeol. Ocean. 2015, 50, 3–19. [Google Scholar] [CrossRef]
- Lucarini, G.; Radini, A.; Barton, H.; Barker, G. The exploitation of wild plants in Neolithic North Africa. Use-wear and residue analysis on non-knapped stone tools from the Haua Fteah cave, Cyrenaica, Libya. Quat. Int. 2016, 410, 77–92. [Google Scholar] [CrossRef]
- Perry, L. Starch analyses reveal the relationship between tool type and function: An example from the Orinoco valley of Venezuela. J. Arch. Sci. 2004, 31, 1069–1081. [Google Scholar] [CrossRef]
- Yang, X.; Perry, L. Identification of ancient starch grains from the tribe Triticeae in the North China Plain. J. Arch. Sci. 2013, 40, 3170–3177. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, J.; Perry, L.; Ma, Z.; Wan, Z.; Li, M.; Diao, X.; Lu, H. From the modern to the archaeological: Starch grains from millets and their wild relatives in China. J. Arch. Sci. 2012, 39, 247–254. [Google Scholar] [CrossRef]
- Yang, X.; Wan, Z.; Perry, L.; Lu, H.; Wang, Q.; Zhao, C.; Li, J.; Xie, F.; Yu, J.; Cui, T.; et al. Early millet use in northern China. Proc. Natl. Acad. Sci. USA 2012, 109, 3726–3730. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, J.; Lü, H.; Cui, T.; Guo, J.; Ge, Q. Starch grain analysis reveals function of grinding stone tools at Shangzhai site, Beijing. Sci. China Ser. D Earth Sci. 2009, 52, 1164–1171. [Google Scholar] [CrossRef]
- Liu, L.; Field, J.; Fullagar, R.; Zhao, C.; Chen, X.; Yu, J. A functional analysis of grinding stones from an early Holocene site at Donghulin, North China. J. Arch. Sci. 2010, 37, 2630–2639. [Google Scholar] [CrossRef]
- Tao, D.; Wu, Y.; Guo, Z.; Hill, D.V.; Wang, C. Starch grain analysis for groundstone tools from Neolithic Baiyinchanghan site: Implications for their function in Northeast China. J. Arch. Sci. 2011, 38, 3577–3583. [Google Scholar] [CrossRef]
- Walsh, R.; Lee, G.A.; Liu, L.; Chen, X. Millet grain morphometry as a tool for social inference: A case study from the Yiluo basin, China. Holocene 2016, 26, 1778–1787. [Google Scholar] [CrossRef]
- Crowther, A. Starch residues on undecorated Lapita pottery from Anir, New Ireland. Archaeol. Ocean. 2005, 40, 62–66. [Google Scholar] [CrossRef]
- Yang, X.; Jiang, L. Starch grain analysis reveals ancient diet at Kuahuqiao site, Zhejiang Province. Chin. Sci. Bull. 2010, 55, 1150–1156. [Google Scholar] [CrossRef]
- Crowther, A.; Haslam, M.; Oakden, N.; Walde, D.; Mercader, J. Documenting contamination in ancient starch laboratories. J. Arch. Sci. 2014, 49, 90–104. [Google Scholar] [CrossRef]
- Haslam, M. The decomposition of starch grains in soils: Implications for archaeological residue analyses. J. Arch. Sci. 2004, 31, 1715–1734. [Google Scholar] [CrossRef]
- Laurence, A.R.; Thoms, A.V.; Bryant, V.M.; McDonough, C. Airborne starch granules as a potential contamination source at archaeological sites. J. Ethnobiol. 2011, 31, 213–232. [Google Scholar] [CrossRef]
- Cristiani, E.; Radini, A.; Edinborough, M.; Borić, D. Dental calculus reveals Mesolithic foragers in the Balkans consumed domesticated plant foods. Proc. Natl. Acad. Sci. USA 2016, 113, 10298–10303. [Google Scholar] [CrossRef] [PubMed]
- Mickleburgh, H.L.; Pagán-Jiménez, J.R. New insights into the consumption of maize and other food plants in the pre-Columbian Caribbean from starch grains trapped in human dental calculus. J. Arch. Sci. 2012, 39, 2468–2478. [Google Scholar] [CrossRef]
- Hardy, K.; Kubiak-Martens, L. (Eds.) Wild Harvest: Plants in the Hominin and Pre-Agrarian Human Worlds. In Studying Scientific Archaeology Series; Oxbow Books: Oxford, UK, 2016; pp. 1–17. ISBN 978-178570-123-8. [Google Scholar]
- Radini, A.; Buckley, S.; Nikita, E.; Copeland, L.; Hardy, K. Beyond Food: The rich and varied pathways for inclusion of microscopic remains into ancient dental calculus. Am. J. Phys. Anthropol. 2017, 162, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Leonard, C.; Vashro, L.; O’Connell, J.F.; Henry, A.G. Plant microremains in dental calculus as a record of plant consumption: A test with Twe forager-horticulturalists. J. Arch. Sci. Rep. 2015, 2, 449–457. [Google Scholar] [CrossRef]
- Collins, M.J.; Copeland, L. Ancient starch: Cooked or just old? Proc. Natl. Acad. Sci. USA 2011, 108, E145. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Azevedo, K.; Herculano-Houzel, S. Metabolic constraint imposes trade off between body size and number of brain neurons in human evolution. Proc. Natl. Acad. Sci. USA 2012, 109, 18571–18576. [Google Scholar] [CrossRef] [PubMed]
- Mulquiney, P.J.; Bubb, W.A.; Kuchel, P.W. Model of 2,3-bisphosphoglycerate metabolism in the human erythrocyte based on detailed enzyme kinetic equations: In vivo kinetic characterization of 2,3-bisphosphoglycerate synthase/phosphatase using 13C and 31P NMR. Biochem. J. 1999, 342, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.W.; de Souza, R.; Kendall, C.W.C.; Emam, A.; Jenkins, D.J.A. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroentrol. 2006, 40, 235–243. [Google Scholar] [CrossRef]
- Herrera, E. Metabolic adaptations in pregnancy and their implications for the availability of substrates to the fetus. Eur. J. Clin. Nutr. 2000, 54, S47–S51. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.U.; Deborde, S.; Illsley, N.P. Placental glucose transfer and fetal growth. Endocrine 2002, 19, 13–22. [Google Scholar] [CrossRef]
- Ungar, P.S.; Sponheimer, M. The diets of early hominins. Science 2011, 334, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Aiello, L.C.; Wheeler, P. The expensive-tissue hypothesis: The brain and the digestive system in human and primate evolution. Curr. Anthropol. 1995, 36, 199–221. [Google Scholar] [CrossRef]
- Lee, S.-H.; Wolpoff, M.H. The pattern of evolution in Pleistocene human brain size. Paleobiology 2009, 29, 186–196. [Google Scholar] [CrossRef]
- Bramble, D.M.; Lieberman, D.E. Endurance running and the evolution of Homo. Nature 2004, 432, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Shahack-Gross, R.; Berna, F.; Karkanas, P.; Lemorini, C.; Gopher, A.; Barkai, R. Evidence for the repeated use of a central hearth at Middle Pleistocene (300 ky ago) Qesem Cave, Israel. J. Arch. Sci. 2014, 44, 12–21. [Google Scholar] [CrossRef]
- Roebroeks, W.; Villa, P. On the earliest evidence for habitual use of fire in Europe. Proc. Natl. Acad. Sci. USA 2011, 108, 5209–5214. [Google Scholar] [CrossRef] [PubMed]
- Gowlett, J.A. The discovery of fire by humans: A long and convoluted process. Philos. Trans. R. Soc. B 2016, 371, 20150164. [Google Scholar] [CrossRef] [PubMed]
- Wrangham, R.W.; Jones, J.H.; Laden, G.; Pilbeam, D.; Conklin-Brittain, N.L. The raw and the stolen: Cooking and the ecology of human origins. Curr. Anthropol. 1999, 40, 567–594. [Google Scholar] [CrossRef] [PubMed]
- Wrangham, R.W.; Carmody, R. Human adaptation to the control of fire. Evol. Anthropol. 2010, 19, 187–199. [Google Scholar] [CrossRef]
- Barton, H.; Matthews, P.J. Taphonomy. In Ancient Starch Research; Torrence, R., Barton, H., Eds.; Left Coast Press Inc.: Walnut Creek, CA, USA, 2006; pp. 5–94. [Google Scholar]
- BeMiller, J.N. Carbohydrate Chemistry for Food Scientists, 2nd ed.; AACC International: St Paul, MN, USA, 2007; Chapter 6; pp. 173–225. ISBN 978-1-891127-3-3. [Google Scholar]

| Geological Epochs | Years before Present | |
| Pleistocene | ||
| Early | 2.6 million–781,000 | |
| Middle | 781,000–126,000 | |
| Late | 126,000–11,500 | |
| Holocene | 11,500–present | |
| Cultural Periods | Years before Present | Examples |
| Palaeolithic | ||
| Lower | 3.3 million-~300,000 | Qesem Cave [45], Sima del Elefante [46] |
| Middle | ~300,000–~45,000 | El Sidrón [43], Shanidar [41], Spy [41] |
| Upper | ~45,000–~11,500 | Various sites, e.g., [50,51] |
| Mesolithic | ~11,500–variable (~8000–~2000) | Various sites e.g., [42,56] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Copeland, L.; Hardy, K. Archaeological Starch. Agronomy 2018, 8, 4. https://doi.org/10.3390/agronomy8010004
Copeland L, Hardy K. Archaeological Starch. Agronomy. 2018; 8(1):4. https://doi.org/10.3390/agronomy8010004
Chicago/Turabian StyleCopeland, Les, and Karen Hardy. 2018. "Archaeological Starch" Agronomy 8, no. 1: 4. https://doi.org/10.3390/agronomy8010004
APA StyleCopeland, L., & Hardy, K. (2018). Archaeological Starch. Agronomy, 8(1), 4. https://doi.org/10.3390/agronomy8010004





