1. Introduction
Common bean (
Phaseolus vulgaris L.) is the most important grain legume in Rwanda [
1,
2]. Although there has been an increase in bean production due to new policies aimed at the expansion of beans into marginal agricultural lands, the productivity per unit area of land has continued to decline [
2,
3]. Typical bean yields obtained on the farmers’ fields are only 20 to 30% of the genetic potential of improved genotypes [
4,
5]. These low yields have been attributed to a number of constraints, the most important of which are diseases, insect pests, low soil fertility and periodic water stress [
6,
7]. Ascochyta blight of the common bean, caused by
Phoma exigua var.
diversispora (Bubak) Boerema or
Ascochyta phaseolorum Sacc. as a synonym of this pathogen, is one of the most damaging diseases of the common bean in Africa [
8,
9].
The disease is particularly favoured by cool temperatures and a high relative humidity [
10]. It infects all major bean parts, including the leaves, stems and pods, and is seed-transmitted, sometimes resulting in total crop losses, especially when infected seed is planted [
11,
12]. In Africa, particularly in Rwanda, Uganda, Burundi and the Eastern DRC, where beans constitute the most important source of dietary protein, ascochyta blight is a significant constraint on bean production [
12,
13,
14].
In Rwanda, most market-class bean genotypes, including the recently-released ones and landraces, are susceptible to ascochyta blight. In severe situations, this disease reduces expected harvests significantly, causing food shortages and a loss of income for Rwandan farmers. Ascochyta blight is prevalent in the highland region of Rwanda where beans are extensively grown [
14,
15]. Although bean ascochyta blight is major disease in Rwanda, its prevalence and impact on yield has not been well documented in Rwanda. In addition, no screening has been done for resistance to the disease in Rwanda.
Since the fungus is seed-borne and can also be spread through debris and the air, the carry-over of the disease from season to season has always been possible due to the small-scale farming systems and seed recycling that is practiced by the small-landholder farmers in Rwanda. Small-scale farmers in the highland regions of Rwanda, like the other parts of sub-Saharan Africa, are compelled to rely upon traditional disease management practices [
6,
7], mainly due to financial constraints.
Poor crop management practices, as well as the adverse biophysical environment, leads to a build-up of field inoculum [
16]. A range of ascochyta blight management methods have been suggested for beans, including traditional cultural practices such as crop rotation, planting mixed bean varieties, planting mature seed, weeding, timely planting, intercropping and uprooting and burying infected plants. The use of genetic diversity (local landraces) and chemicals have been used also to control ascochyta blight [
16,
17,
18]. However, many of these methods are not feasible in Rwanda, due to the varying growing conditions and limited resources that characterize the small-scale farming system. The use of resistant genotypes, combined with other disease management practices, is regarded as the most practical approach for disease control at farm level.
Farmers can exploit the diversity available in landraces and genotypes to reduce the risk of ascochyta blight infection [
19]. The identification of useful sources of resistance to the most important diseases is valuable, in that such sources could be used to confer resistance to locally-adapted germplasm [
20]. However, breeding programs need to place more emphasis on the development of genotypes with durable mechanisms of resistance. Resistance genes may be present within the landrace populations, due to long-term co-evolution between crops and disease, natural selection and intentional selection by farmers.
Very little research has been conducted to identify sources of resistance to ascochyta blight in the common bean germplasm. Schmit and Baudoin [
21] evaluated 200 populations of
P. coccineus L. and
P. polyanthus L. for ascochyta resistance, at two highland stations of Ronegro and Popayan in Colombia. However, only low levels of resistance to
Phoma exigua var.
diversispora were detected among them. Similarly, research conducted at CIAT and the National Bean Programme in Guatemala showed some differences in reactions to the ascochyta pathogen. Most of the evaluated genotypes were either susceptible, or had low levels of resistance. A high level of resistance or immunity was present in genotypes of
P. coccineus L., particularly in the sub-species
P.
polyanthus and in interspecific hybrids that were obtained by crossing these two species [
22].
Apart from the yield reduction caused by diseases such as ascochyta blight, unstable climatic conditions are a persistent problem in Rwanda. Therefore, the adaptation of bean genotypes to such environments requires a genotype with a wide spectrum of tolerance to climatic variations, in addition to disease resistance. The seriousness and distribution of bean ascochyta varies depending on the location and the season.
According to Ceccarelli and Grando [
23], when different genotypes of a given crop are evaluated under a range of environments, genotype (G) × environment (E) (GE) interactions of cross-over types appear to be quite common. Significant GE interactions cannot be disregarded. The options are to manage them by selecting genotypes that are broadly adapted to a whole range of target environments, or to basically carry out selection for an array of genotypes, whereby each is adapted to a specific environment [
24]. Such selection requires separate GE analyses, namely, genotype (G) × year (Y) (GY), which is highly unpredictable, and genotype × location (L) (GL), which identifies a distinct target environment [
24]. Selecting for specific adaptation is important, predominantly for crops grown under unfavorable conditions, as unfavorable environments can be very different from each other [
23]. Therefore, a breeding strategy to identify genotypes suitable for unfavorable environment and variable seasonal conditions will exploit the analysis of GE components [
24]. This is because seasonal variation in bean ascochyta infection, rainfall patterns, and a negative or low correlation between farmer fields and research stations, complicate the breeder’s selection process. This may hamper the positive identification of superior materials for the intended specific target environment or a wide range of environments.
The objectives of this study were therefore (1) to identify sources of resistance to bean ascochyta blight available in landraces and other collections; and (2) to determine the effect of seasonal climatic variation on common bean genotypes in relation to bean ascochyta blight severity in Rwanda.
2. Materials and Methods
The reactions to ascochyta blight caused by Phoma exigua var. diversispora (Bubak) Boerema was evaluated in the field, using 75 bush and climber genotypes. The 75 genotypes were collected from different collections within and outside Rwanda. The genotypes were then screened in the field for ascochyta resistance for two growing seasons; 2014B (from February 2014 to June 2014) and 2015A (from September 2014 to January 2015) at the Nyamagabe, Musanze and Rwerere Research Stations, where conditions are favorable for disease development and establishment.
2.1. Experimental Sites
The field experiments were carried out at three selected locations in Rwanda. The locations are the major research sites of the Rwanda Agriculture Board (RAB) in areas known for their bean production and ascochyta epidemics. The Musanze site is in the highlands of the northern agricultural zone and is located at coordinates 1°14′59.7″ South and 36°44′28.8″ East at an altitude of 1820 m above sea level (masl). The area receives an average rainfall of 1746 mm annually, and has a mean maximum temperature of 23 °C and mean minimum temperature of 12 °C. It represents the highland of volcanic soils. Rwerere is located at an altitude of 2060–2312 masl on a longitude of 29°19′ East and a latitude of 1°36′ South, with an annual rainfall and temperature of 1200 mm and 20 °C, respectively. The soil is volcanic and represents the highlands of Buberuka. Nyamagabe is located at an altitude of 2080 masl on a longitude of 29°33′ East and latitude of 1°33′ South, with the annual rainfall and temperature being 1600 mm and 19 °C, respectively. The soil type is sandy clay. It represents of the highlands of the Congo/Nile Divide [
14].
In warm and moist regions, such as the tropical highlands of Rwanda, inoculum is always present due to the continuous cropping of beans, which combine with the suitable conditions for ascochyta occurrence and spread. In most bean-growing areas in Rwanda, the average annual precipitation ranges from 1200 mm to over 1800 mm. In general, rainfall is bimodal, with a minor peak occurring in October and a major peak in April. High elevations and low latitudes combine to form a moderate temperature regime, with an average annual temperature of about 16 °C [
25].
2.2. Planting Material
A total of 75 bush and climbing genotype lines (
Table 1) were tested for resistance to ascochyta. Screening was done in the field, using ascochyta-infected debris as the inoculum, which had been previously collected from the same region.
The 75 genotypes comprised of 39 bush (Types I, II and III) bean genotypes and 36 climbers (Type IV). The entries include ascochyta-resistant lines acquired from the Centro Internacional de Agricultura Tropicale (CIAT) in Colombia, landraces from the National Gene Bank of Rwanda and improved genotypes, mainly released from the Rwanda Agriculture Board (RAB), as well as checks which were high yielding new released genotypes and mixture of varieties.
In order to identify the adapted genotypes to be used for the experiments, a preliminary screening trial was first conducted during Season 2014A (from September 2013 to January 2014) before the main trials. A complete set of 75 genotypes was then assembled for the main trials.
2.3. Experimental Design
Trials were conducted using an 10 × 4 row by column lattice design for bush and a 9 × 4 row by column lattice design for climbers, with 10 blocks of four plots for bush and nine blocks of four plots for climbers each, with two replications. For climbers, the check has been used twice in the trial in order to complete a 10 × 4 lattice. All genotypes were established in four-row plots of 2 m length and 2 m width with an inter-row spacing of 0.6 m and an intra-row spacing of 0.4 m. Experiments were established under rain-fed conditions. The susceptible genotype Colta was planted as a border of spreader rows around each trial, to serve as a source of inoculum.
Fertilizer was applied in the form of N17-P17-K17 at a rate of 10.5 kg ha−1 in split applications at planting and ridging. Fungicides were not applied, and the trials were maintained with the conventional cultural practices. Weeds were controlled by hand.
The genotypes were inoculated with the bean field debris collected in the previous season from plants showing symptoms that were characteristic of ascochyta. The inoculation was done 14 days after planting by the uniform spreading of 0.25 kg/plot of ascochyta-infected debris in the field trials.
2.4. Data Collection
The data collected included ascochyta disease severity and plot yield weight. Ascochyta symptoms were assessed six times starting 21 days after planting. The two inner rows (ten plants/row) of each plot were visually rated at 14-day intervals for the percentage of the leaf stem and pod area with ascochyta symptoms. The square root transformation has been used to verify whether there is possible deviation and make the data follow a normal distribution. The percentage of the diseased foliage of individual plants was visually estimated. The plant assessments were converted to a single value for each plot and the mean percentage diseased foliar area per plot was calculated [
13,
26,
27]. This was done by comparing the green and non-green leaf portions affected by the disease and by using a percentage scale. The evaluations continued up to physiological maturity.
2.5. Data Analysis
Based on all plots and assessment dates, the area under the disease progress curve (AUDPC) was calculated for each genotype, using the midpoint rule method as per the equation suggested by Madden et al. [
28] in Equation (1) below:
where
Xi = the disease percentage representing the affected foliage at each at
ith evaluation day;
ti+1 −
ti = times (in days) between two diseases scores and
n = the total number of observations. Means were compared by the least significant difference at
p ≤ 0.05.
The estimates of AUDPC were normalized by dividing with the total area of the graph (i.e., the number of days from first appearance of the disease till the end of the observation period), in order to facilitate a better visual comparison among host genotypes, over the seasons and the sites tested [
29]. The normalized AUDPC was referred to as the relative area under the disease progress curve (RAUDPC).
In Equation (2) above, Ti is the ith day when an estimation of percentage foliar blight is made and Di is the estimated percentage of area with diseased foliage at Ti. Ttotal is the number of days at which the final assessment was recorded.
In addition, the correlations between mean RAUDPC values and the mean yields for Seasons A and B were determined. The RAUDPC was used to evaluate and select the parents to be used in a breeding programme. All the collected quantitative data were subjected to residual (or restricted) maximum likelihood (REML) spatial model analysis to fit the variance-components, using the computer software programme GENSTAT Version 17.
From the observed RAUDPC and yield data, the confidence interval have been computed at the confidence level of 95% using the equation as suggested by Neyman [
28]:
where
Z = Critical value;
µ = Sample mean;
n = number of observation and
σ = standard deviation.
The severity score ranges between 0 and 100, with the genotypes having a RAUDPC value >50 considered resistant, 50–100 as having intermediate resistance and those having RAUDPC >100 being susceptible for bush, and genotypes with a RAUDPC value of >40 being considered resistant, 40–80 as having intermediate resistance and RAUDPC >80 being considered susceptible for climbers.
At the end of each growing season, the plants were harvested and the dry seed yields for each genotype (kg ha−1) were recorded. At the end of each growing season, the plants were harvested and the dry seed yields for each genotype (kg ha−1) were recorded. Total plot weight (based on the middle inner row) was measured and expressed in tons per hectare. The bush genotypes with the yield (kg ha−1) value <350 were considered low yielding, 350–500 as medium yielding and those of >500 kg/ha being high yielder.
For climbing genotypes, the mean yield across two seasons >1400 kg/ha being considered high yielder, 1000–1300 as medium yield and Yield/ha >1000 being considered low yield.
Genotypes, location and year were considered fixed terms, while replications, rows and columns were considered random terms, as shown in the model below:
where
Yijklm = observed value;
M = general mean;
Repi = effect of the
ith replication (where
i = 1, 2, 3);
Row(
Rep)
ij = row effect nested within rep (where
j = 1, 2 …
n);
Col(
Rep)
ik = column effect nested within rep (where
k = 1, 2 …
n);
Vi = effect of the
Lth genotype (where
L = 1, 2, …,
n);
Lm = effect of the
mth location (
m = 1, 2);
VLlm = interaction effect of the
Lth genotype and
mth location;
VLjk = interaction genotype × location; and
eijklm = random error. Means were compared by the least significance difference (LSD) test, using a suitable error term.
The variation due to genotypes and G × E for the ascochyta disease was explained using the genotype and genotype × environment (GGE) biplot based on the principal component analysis (PCA) of environment centred data [
30]. The GGE biplot states a visual relation among the genotypes and test environments and was performed using a model based on singular value decomposition (SVD) of the first two principal components [
30] as follows:
where
Yij is the yield mean of
ith genotype in
jth environment,
µ is the grand mean,
Bj is the main effect of environment
j,
µ +
Bj is the mean yield across all genotypes in environment
j,
λ1 and
λ2 are the singular values (SV) for the first and second principal component (PC1 and PC2), respectively,
εi1 and
εi2 are the eigen vectors of genotypes
i for PC1 and PC2, respectively,
nj1 and
n j2 are the eigen vectors of environment
j for PC1 and PC2, respectively and
eij is the residual associated with genotypes
i in environment
j.
3. Results
3.1. Weather Data
Weather conditions were conducive to the development of ascochyta. There was regular rainfall and mean temperatures were around 18 °C (
Table 2) throughout the two growing seasons, which promoted the development of ascochyta.
Cropping seasons (year), location and genotypes, were significantly different for RAUDPC (
Table 3) for all bean types. The two-way interaction between the year and the genotypes was significant (
p ≤ 0.001) for this trait. The interaction between location and genotypes was also significant, while the three-way interaction genotype, year and location were not significant for the RAUDPC. The interaction between season and location has been significant only for climbing genotypes.
The means across all sites for the severity of ascochyta measured as RAUDPC (
Table 4 and
Table 5) were significantly higher in Season 2014B for both bush (106.70) and climber (50.5) types, than in the Season 2015A. The reaction of the genotypes to the pathogen was different and there was a high positive correlation between the RAUDPC of Seasons A and B (
r = 0.86).
The mean RAUDPC value across two seasons (M (A&B)) for bush genotypes ranged from 18.9 to 236.3, whereby genotypes with the RAUDPC value >50 were considered resistant, 50–100 as having intermediate resistance and those having RAUDPC >100 were susceptible.
For climbing genotypes, the mean RAUDPC values across two seasons ranged from 15.8 to 88.3, with a RAUDPC value of >40 being considered resistant, 40–80 as having intermediate resistance and RAUDPC >80 being considered susceptible.
The majority of bush genotypes (46%) showed a susceptible reaction to the pathogen, with severe symptoms on leaves, stems and pods. A total of 36% of the genotypes showed an intermediate reaction, with disease symptoms limited to small lesions, and only seven genotypes (18%) showed resistance to the pathogen. In some resistant plants, a few symptoms were observed, mainly on the primary leaves.
Most of the climbing beans had intermediate resistance (69.5%). Only six genotypes (16.5%) showed a resistant reaction and five genotypes showed a susceptible reaction (14%) (
Table 5). The ascochyta was most severe at the Rwerere and lowest at Nyamagabe sites, compared to the other sites, for both bush and climber.
Using the RAUDPC values obtained in the trials, the climbing beans had a lower number of resistant genotypes, compared to the bush types. The data show that all the genotypes that gave resistant reactions to ascochyta possesses pink and red flowers. Findings further revealed that all resistant genotypes were small-seeded.
On resistant genotypes, the lesions caused by the ascochyta pathogen were limited to the primary leaves. In susceptible bush genotypes, symptoms were scattered throughout the canopy on the leaves, stems and pods. Lesions caused by the pathogen produced brown or black concentric lesions 1–3 cm on leaves and pods as well as collapsed and black nodes, petioles and stems. These symptoms varied in intensity, depending on the resistance of the different genotypes.
3.2. Relationship between Ascochyta Blight and Yield
The analysis of variance shows that significant differences were observed in the yield obtained from the different genotypes, locations, year and two-way interactions of year × location, location × genotype and year × genotype, for both bush and climber type (
Table 6).
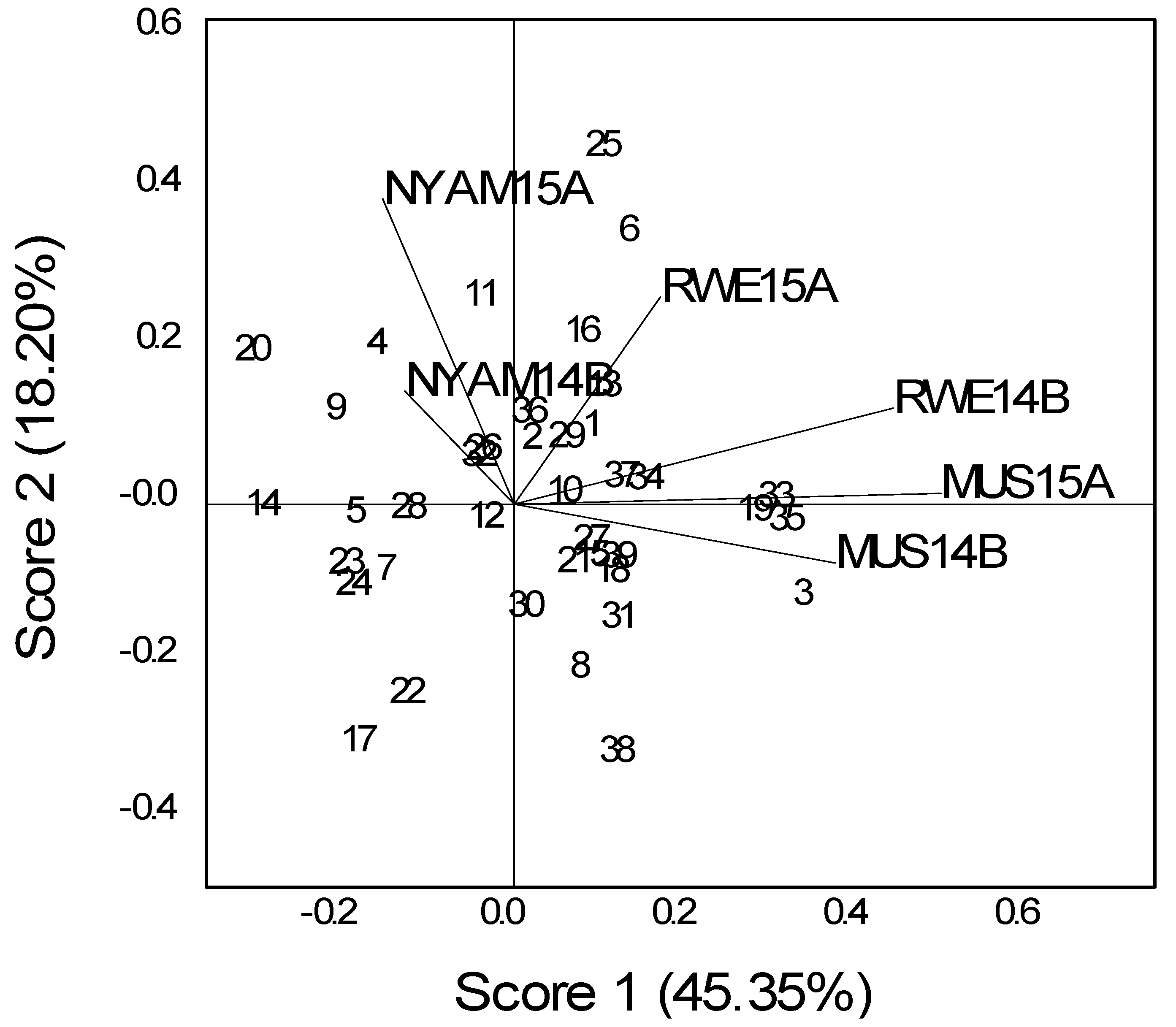
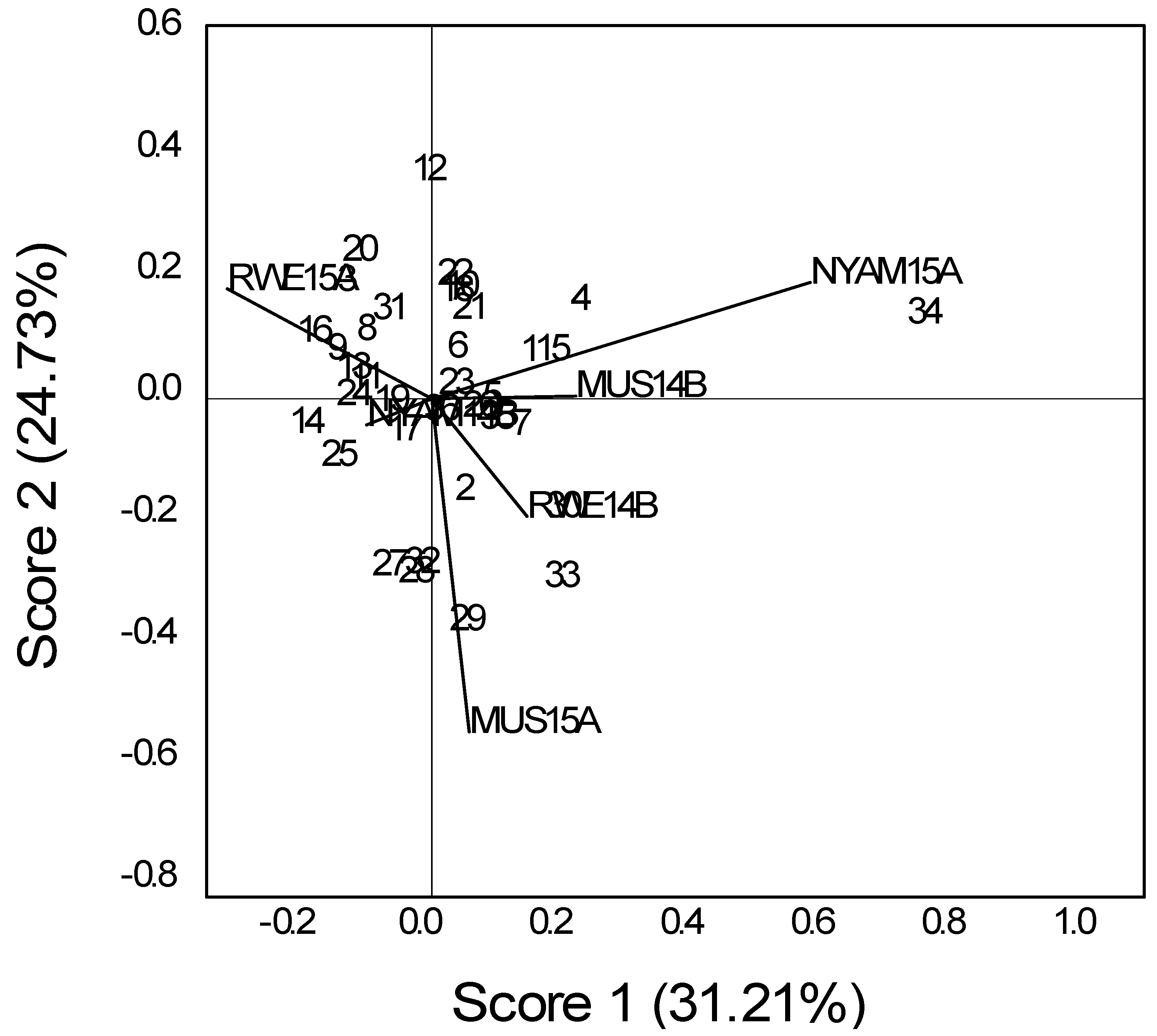
A significant YG interaction for seed yield indicates that seasonal variation affected the relative yield performance of the genotypes under the natural infection of bean ascochyta. The seed yield of bean was affected by seasonal variation, and genotypic variation was observed among the tested genotypes (
Figure 1 and
Figure 2).
The differences of mean grain yield across geographic locations indicate that not only the genotypes and locations but also variations in seasons or environmental conditions during different seasons greatly influence the grain yield performance. Similarly, grain yields obtained for different locations in the first season were different from those obtained during second.
From the GGE biplots, the first two principal components (PCs) explain 31.2–45.4% of the total GGE variation for seed yield. This biplot indicated the most yielding genotype for each environment. The biplots show that the bush genotypes LSA 142 (12), ECAB06 (10) and the climber genotypes CAB 2 (2), Nyirabukara (23) and MAC (19) attained values relatively close to zero and hence were more stable and widely adaptable genotypes across all locations (
Figure 1 and
Figure 2).
The average dry seed yield of the different genotypes across seasons and locations ranged from 239.8.0 to 708.7 kg ha
−1 and from 723.1 to 1401.5 kg ha
−1, for bush and climbers, respectively (
Table 7 and
Table 8). Considering the reaction to ascochyta, the mean yield varied widely. For the resistant genotypes, the yield range was between 292.0–709.0 kg ha
−1 and 870.2–1322.9 kg ha
−1 for bush and climber resistant genotypes, respectively.
The genotypes with intermediate resistance showed a yield range of 271.19–645.2 kg ha
−1 and 723.1–1401.5 kg ha
−1 for bush and climber types, respectively, whereas for susceptible genotypes the yield range was from 239.7 to 661.1 kg ha
−1 and 837.6–1307.6 kg ha
−1 for bush and climbing genotypes, respectively (
Table 4,
Table 5,
Table 7 and
Table 8).
There were genotypes with intermediate resistant reactions that yielded significantly higher than genotypes with a resistant reaction. This observation was also true for the susceptible, versus the intermediate resistant, genotypes (
Table 4,
Table 5,
Table 7 and
Table 8). Although there was a strong correlation (r = 0.62;
p ≤ 0.05) between the yields of the two seasons, mean yields obtained in the second season (Season 2014B) were significantly higher than those obtained in the first season for bush and climbers. The results also indicate a significant negative correlation between the RAUDPC and the yield (r = −0.51).
Bush genotypes RWR 281, ASC 87 and ASC 92, and climbing genotypes Vuninkingi, Nyiramagorori and RWV 2872, are adapted to the short rainy season (Season A). Bush genotypes, RWR 2154, ASC 92 and NUA 377, and climbing Garukurare, G 2331 and RWV 2269, are best adapted to the long rainy season (Season B)
Considering the genotypic performance based on the geometric mean (M (A&B)), which is associated with yield performance of different sites, a number of bush genotypes, both landraces and bean ascochyta resistant genotypes (introductions from CIAT), consistently outperformed the local checks, indicating their broad adaptation under a varied environment (
Table 7 and
Table 8). For climbers, the best check from Rwandan germplasm (RWV 2269) was outstanding for its yield. A range of seed sizes, from small to large, existed among the genotypes. The top two performing lines had relatively small seed sizes.
4. Discussion
Sources of good resistance are an important tool to pursue as the principal element in a breeding programme. The best possible method for identifying resistant sources is to expose the potential sources of resistance to all dominant pathogens over different production areas, in order to eliminate the highly susceptible genotypes [
13,
31,
32]. In this study, a germplasm collection of 75 dry bean genotypes was screened to establish whether there was any genotype which could be used as effective sources of ascochyta resistance. The use of the relative area under the disease progress curve (RAUDPC), as a tool for the plant resistance evaluation, helps to reflect the progress of the disease throughout the growing season [
27]. In this study, the highest RAUDPC values represented genotypes with the highest disease infection. There were differences in the RAUDPC values between genotypes within the seasons and between the seasons. The differences that were observed suggest differences in the resistance of the individual genotypes or are explained by variability in virulence pattern of the pathogen. On the other hand, the differences observed between seasons could be explained by the differences in the climatic conditions. These differences in climatic conditions between test sites in the two growing seasons differentially affected the yield of the genotypes. Related studies conducted by Hanson et al. [
33] on bean ascochyta blight show that climatic conditions have a strong influence on the yield.
The season with the highest disease infection was also observed to have a better yield performance. The inconsistency in results could be explained by the Gaunt [
34] theory, which states that the green leaf area and the green area duration is directly correlated to yield, in both the healthy and diseased crop species. The observations from the study show that Season B, which had higher disease infection, also had better climatic conditions, resulting in longer green leaf area duration, which culminated in higher yields.
Furthermore, a strong negative correlation between disease and yield would be expected but, as reported in this study, the correlation between these two factors, although negative, was only weak and could not fully explain all the yield variations. A partial explanation could be offered with regard to the tolerance observed in some genotypes, which resulted in high yields despite high disease infection (RUADPC). According to Gaunt and Bryson [
35], the absence of a strong negative relationship between yield and RAUDPC is more common when data from different seasons are used, as was the case in this study. The analysis of data for the individual seasons showed a moderate correlation (r = −0.42 and r = 0.51 for Seasons A and B, respectively). This weak correlation between yield and RAUDPC could partially be explained by the variation in defoliation, the variation in growth habits and the differences in yield potential that was exhibited by the different genotypes. In addition, measuring disease by visual rating lacked precision and accuracy. As revealed by O’Brien and Van Bruggen [
36], the inaccuracies made while measuring disease in the field are a major constraint, when relating disease to yield, and in some cases, there may be no relationship between these two variables. Similar studies by Waggoner and Berger [
37], Gaunt [
34] and Filho et al. [
38] have indicated that the measurement of disease severity, based on lesion number or leaf area, may be less related to yield.
This study indicates the relationship between crop resistance and some phenotypic traits. Results showed that a number of resistant genotypes had either pink or red flowers and were small-seeded. A large percent of bush germplasm that was collected for use in this study was susceptible. Many of the Phaseolus coccineus genotypes were categorised as resistant. It was also observed that most of resistant genotypes, yielded far better than those showing an intermediate or susceptible disease reactions. These genotypes could be described as being tolerant to Phoma exigua. With the exception of the thirteen genotypes obtained from outside Rwanda, the remaining two resistant genotypes originated from Rwanda (SMC 18 and G 2331). It is possible that resistant genotypes may have been selected by farmers as a result of the high disease pressure, but this is a hypothesis which needs further investigation.
Finally, the use of RAUDPC as the measure of resistance was very useful in this study, as it was able to show that, out of the 75 germplasm genotypes, 13 consistently had resistant reactions to the ascochyta pathogen in Rwanda, 39 had intermediate resistance reactions and 23 were susceptible. It is therefore suggested that the identified resistant lines in the development of an ascochyta breeding programme are used for the Rwandan common bean genotypes. There is also a need for further studies to determine the quality of resistance exhibited by the resistant genotypes.