Rhizoctonia solani and Bacterial Inoculants Stimulate Root Exudation of Antifungal Compounds in Lettuce in a Soil-Type Specific Manner
Abstract
:1. Introduction
2. Results
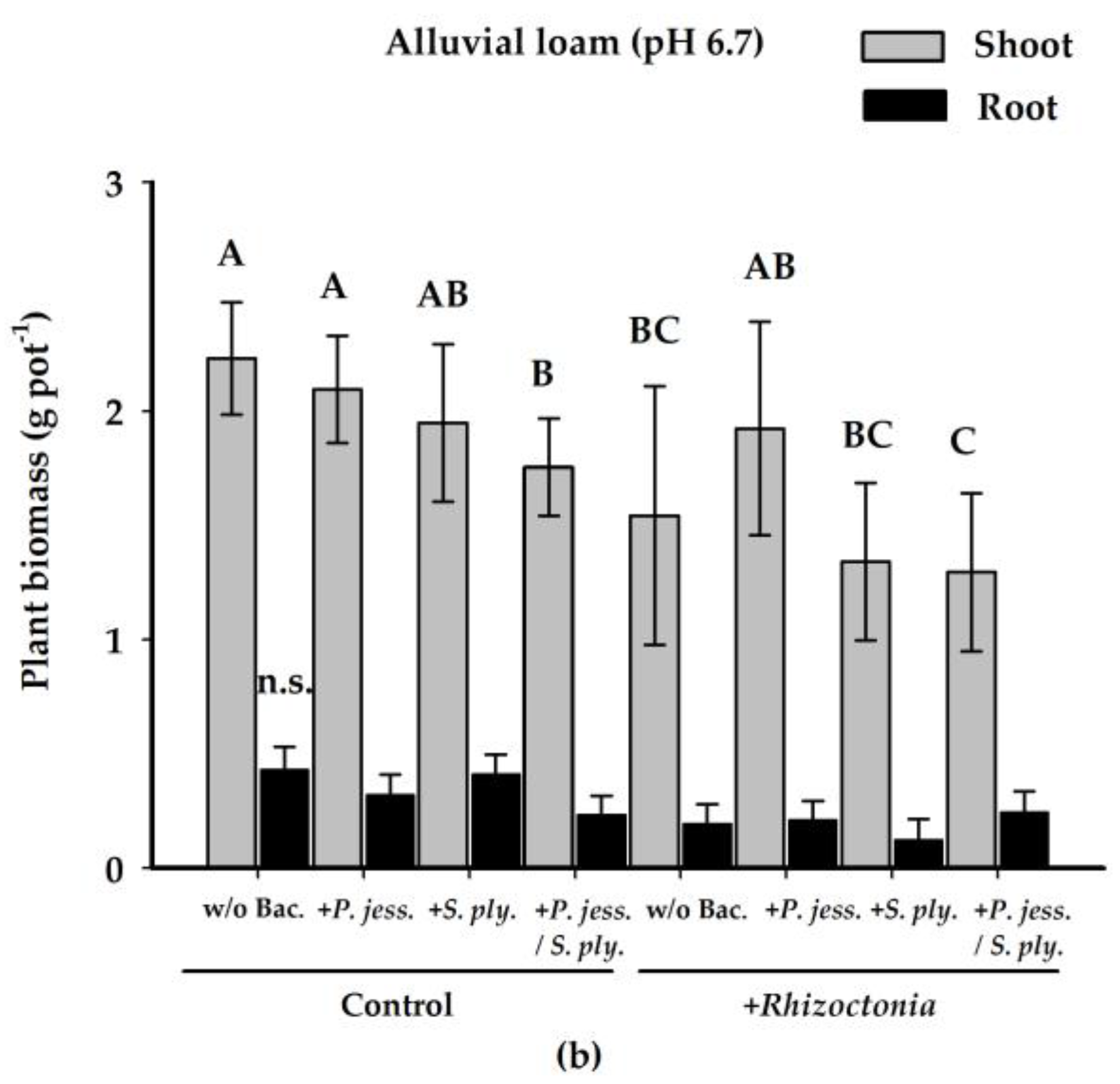
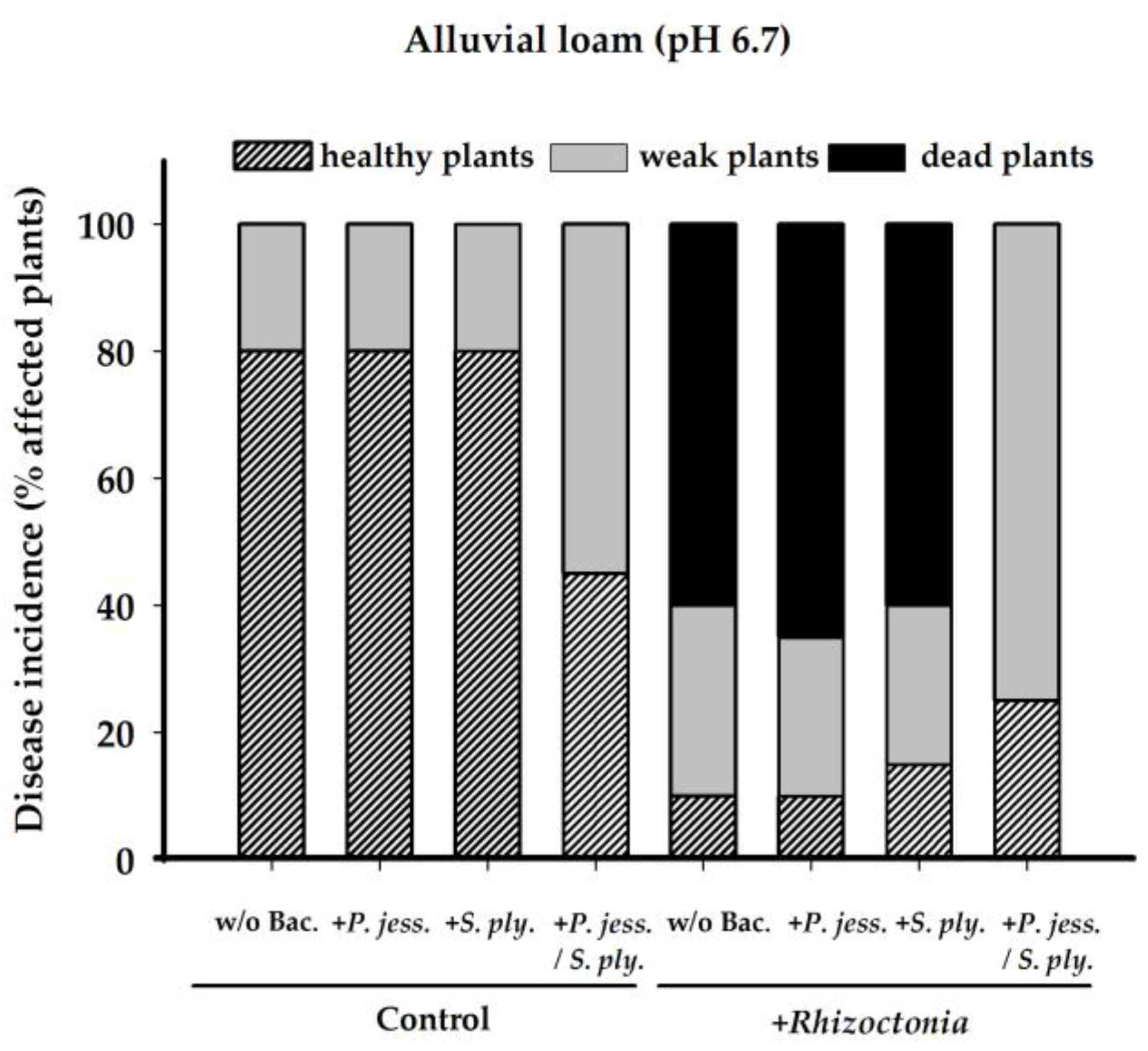
2.1. Plant Biomass Production is Affected by Rhizoctonia solani AG1-IB
2.2. Biocontrol Activity of Pseudomonas jessenii RU47 and Serratia plymuthica 3Re-4-18
2.2.1. Effects on Shoot Growth
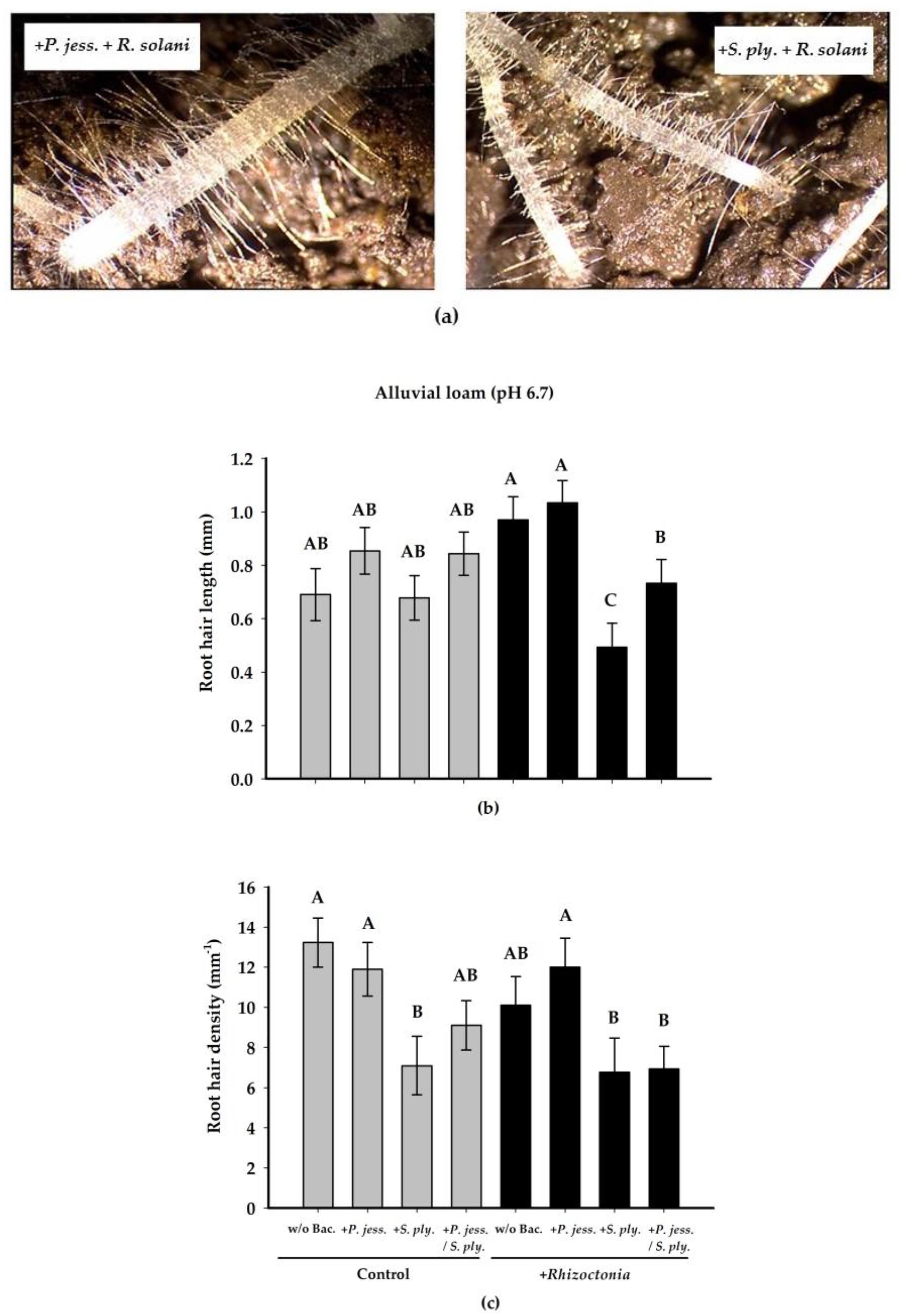
2.2.2. Effects on Root Growth and Morphology
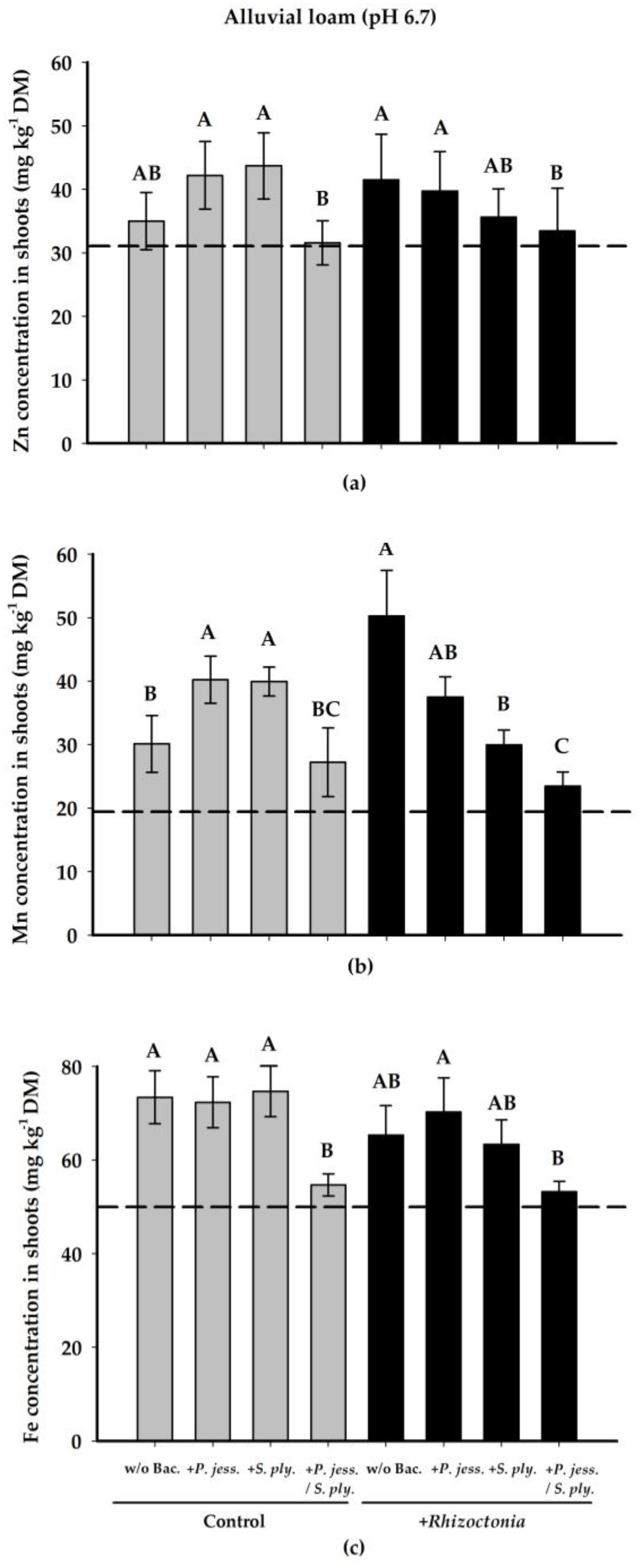
2.2.3. Plant-Nutritional Status
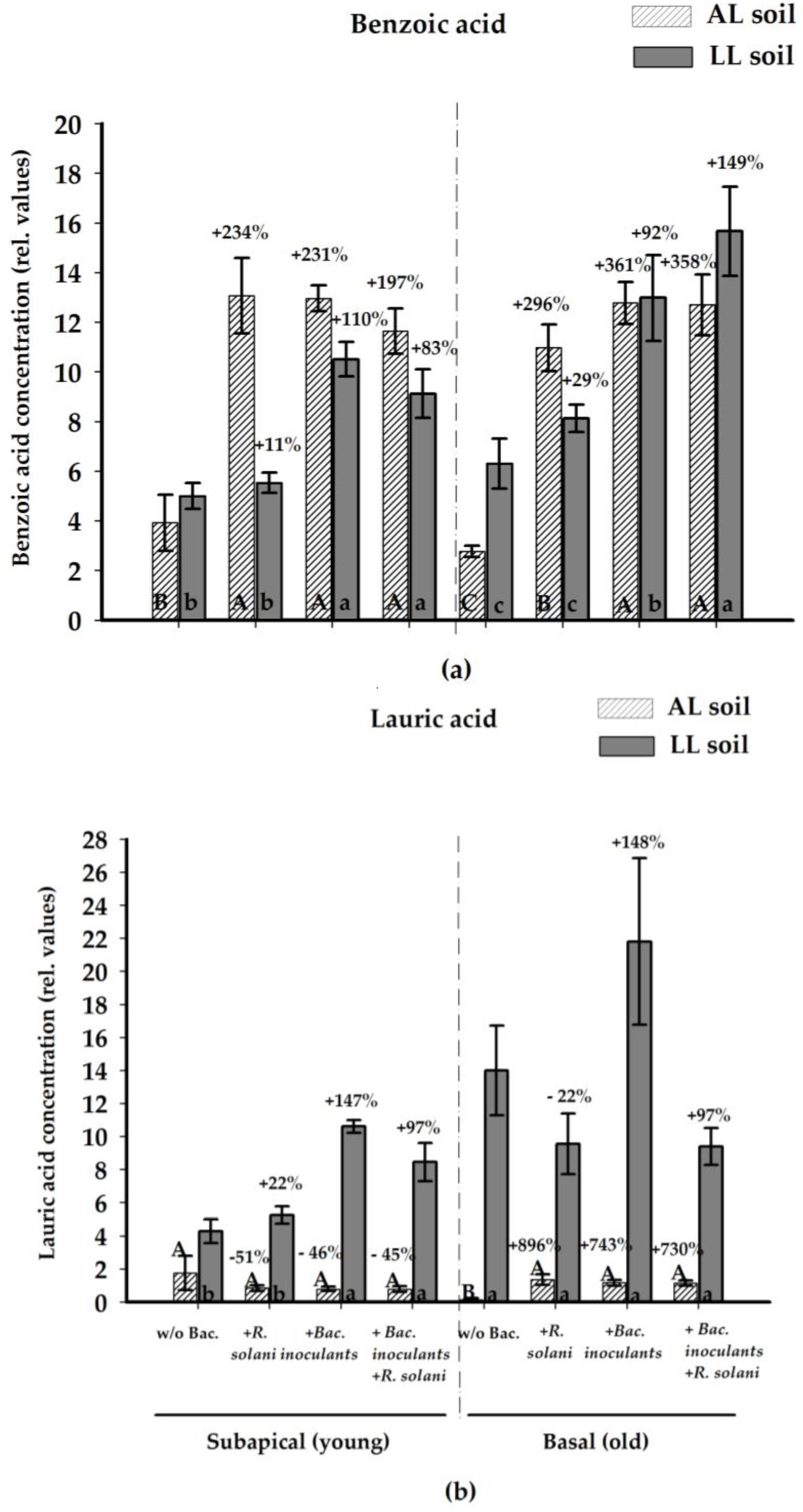
2.2.4. Antifungal Compounds in the Rhizosphere Soil Solution
3. Discussion
3.1. Plant Pathogen Interactions
3.2. Disease Suppression by Bacterial Inoculants
3.3. Negative Side Effects of the Bacterial Inoculants
4. Materials and Methods
4.1. Lettuce Cultivation
4.2. Inoculation with Bacterial Inoculants
4.3. Pathogen Inoculation
4.4. Nutrient Analysis of Plant Biomass
4.5. Analysis of Rhizosphere Soil Solution
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Oerke, E.-C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Grosch, R.; Schneider, J.H.M.; Kofoet, A.; Feller, C. Impact of continuous cropping of lettuce on the disease dynamics of bottom rot and genotypic diversity of Rhizoctonia solani AG 1-IB. J. Phytopathol. 2011, 159, 35–44. [Google Scholar] [CrossRef]
- Schreiter, S.; Sandmann, M.; Smalla, K.; Grosch, R. Soil type dependent rhizosphere competence and biocontrol of two bacterial inoculant strains and their effects on the rhizosphere microbial community of field-grown lettuce. PLoS ONE 2014, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hallmann, J. Einführung in die Thematik- Bedeutung bodenbürtiger Schaderreger; Julius KühnInstitut: Münster, Germany, 2010; Volume 15, pp. 2–9. [Google Scholar]
- Mitsuro, H.; Tadao, U. The Role of the overwintered Plant Debris and Sclerotia as Inoculum in the field occurred with Sugarbeet Root Rot. Ann. Phytopathol. Soc. Jpn. 1982, 48, 628–633. [Google Scholar]
- Grosch, R.; Faltin, F.; Lottmann, J.; Kofoet, A.; Berg, G. Effectiveness of 3 antagonistic bacterial isolates to control Rhizoctonia solani Kühn on lettuce and potato. Can. J. Microbiol. 2005, 51, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, N.; Gutiérrez-Barranquero, J.A.; De Vicente, A.; Cazorla, F.M. Enhancing soil quality and plant health through suppressive organic amendments. Diversity 2012, 4, 475–491. [Google Scholar] [CrossRef]
- Weller, D.M.; Raaijmakers, J.M.; Gardener, B.B.M.; Thomashow, L.S. Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu. Rev. Phytopathol. 2002, 40, 309–348. [Google Scholar] [CrossRef] [PubMed]
- Garbeva, P.; Postma, J.; Van Veen, J.A.; Van Elsas, J.D. Effect of above-ground plant species on soil microbial community structure and its impact on suppression of Rhizoctonia solani AG3. Environ. Microbiol. 2006, 8, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Bakker, M.G.; Manter, D.K.; Sheflin, A.M.; Weir, T.L.; Vivanco, J.M. Harnessing the rhizosphere microbiome through plant breeding and agricultural management. Plant Soil 2012, 360, 1–13. [Google Scholar] [CrossRef]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef] [PubMed]
- Raaijmakers, J.M.; Paulitz, T.C.; Steinberg, C.; Alabouvette, C.; Moënne-Loccoz, Y. The rhizosphere: A playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 2009, 321, 341–361. [Google Scholar] [CrossRef]
- Badri, D.V.; Vivanco, J.M. Regulation and function of root exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Chaparro, J.M.; Badri, D.V.; Bakker, M.G.; Sugiyama, A.; Manter, D.K.; Vivanco, J.M. Root Exudation of Phytochemicals in Arabidopsis Follows Specific Patterns That are Developmentally Programmed and Correlate with Soil Microbial Functions. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Marschner, P.; Neumann, G.; Kania, A.; Weiskopf, L.; Lieberei, R. Spatial and temporal dynamics of the microbial community structure in the rhizosphere of cluster roots of white lupin (Lupinus albus L.). Plant Soil 2002, 246, 167–174. [Google Scholar] [CrossRef]
- Neumann, G.; Bott, S.; Ohler, M.A.; Mock, H.-P.; Lippmann, R.; Grosch, R.; Smalla, K. Root exudation and root development of lettuce (Lactuca sativa L. cv. Tizian) as affected by different soils. Front. Microbiol. 2014, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Neumann, G.; Römheld, V. The release of root exudates as affected by the plant physiological status. In The Rhizosphere: Biochemistry and Organic Substances at the Soil-Plant Interface; Pinton, R., Varanini, Z., Nannipieri, P., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 23–72. [Google Scholar]
- Neumann, G. Root exudates and nutrient cycling. In Soil Biology Volume 10; Marschner, P., Rengel, Z., Eds.; Springer: Berlin, Germany, 2007; pp. 123–157. [Google Scholar]
- Aziz, N.H.; El-Fouly, M.Z.; El-Essawy, A.A.; Khalaf, M.A. Influence of bean seedling root exudates on the rhizosphere colonization by Trichoderma lignorum for the control of Rhizoctonia solani. Bot. Bull. Acad. Sin. 1997, 38, 33–38. [Google Scholar]
- Alabouvette, C.; Olivain, C.; Steinberg, C. Biological control of plant diseases: The European situation. Eur. J. Plant Pathol. 2006, 114, 329–341. [Google Scholar] [CrossRef]
- Scherwinski, K.; Grosch, R.; Berg, G. Effect of bacterial antagonists on lettuce: Active biocontrol of Rhizoctonia solani and negligible, short-term effects on nontarget microorganisms. FEMS Microbiol. Ecol. 2008, 64, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Grosch, R.; Dealtry, S.; Schreiter, S.; Berg, G.; Mendonça-Hagler, L.; Smalla, K. Biocontrol of Rhizoctonia solani: Complex interaction of biocontrol strains, pathogen and indigenous microbial community in the rhizosphere of lettuce shown by molecular methods. Plant Soil 2012, 361, 343–357. [Google Scholar] [CrossRef]
- Schreiter, S.; Ding, G.C.; Heuer, H.; Neumann, G.; Sandmann, M.; Grosch, R.; Kropf, S.; Smalla, K. Effect of the soil type on the microbiome in the rhizosphere of field-grown lettuce. Front. Microbiol. 2014, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.; Seo, K.H.; Lee, S.H.; Choi, G.J.; Jang, K.S.; Choi, Y.H.; Cha, B.; Kim, J.-C. Antifungal Activity of Benzoic Acid from Bacillus subtilis GDYA-1 against Fungal Phytopathogens. Res. Plant Dis. 2012, 18, 109–116. [Google Scholar] [CrossRef]
- Walters, D.R.; Walker, R.L.; Walker, K.C. Lauric Acid Exhibits Antifungal Activity against Plant Pathogenic Fungi. J. Phytopathol. 2003, 151, 228–230. [Google Scholar] [CrossRef]
- Lee, J.G.; Lee, B.Y.; Lee, H.J. Accumulation of phytotoxic organic acids in reused nutrient solution during hydroponic cultivation of lettuce (Lactuca sativa L.). Sci. Hortic. 2006, 110, 119–128. [Google Scholar] [CrossRef]
- Bergmann, W. Ernährungsstörungen bei Kulturpflanzen; Gustav Fischer Verlag: Jena, Germany, 1988. [Google Scholar]
- Grosch, R.; Schneider, J.H.M.; Kofoet, A. Characterisation of Rhizoctonia solani anastomosis groups causing bottom rot in field-grown lettuce in Germany. Eur. J. Plant Pathol. 2004, 110, 53–62. [Google Scholar] [CrossRef]
- Lehtonen, M.J.; Somervuo, P.; Valkonen, J.P.T. Infection with Rhizoctonia solani induces defense genes and systemic resistance in potato sprouts grown without light. Phytopathology 2008, 98, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Li, X.G.; Zhang, T.L.; Wang, X.X.; Hua, K.; Zhao, L.; Han, Z.M. The composition of root exudates from two different resistant peanut cultivars and their effects on the growth of soil-borne pathogen. Int. J. Biol. Sci. 2013, 9, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, J.; Bhuvaneswari, R.; Rabindran, R.; Muthukrishnan, S.; Velazhahan, R. Oxalic acid-induced resistance to Rhizoctonia solani in rice is associated with induction of phenolics, peroxidase and pathogenesis-related proteins. J. Plant Interact. 2010, 5, 147–157. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Dekkers, L. What makes Pseudomonas bacteria rhizosphere competent. Environ. Microbiol. 1999, 1, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Berg, G. Plant-microbe interactions promoting plant growth and health: Perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Biotechnol. 2009, 84, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Zachow, C.; Fathehi, J.; Cardinale, M.; Tilcher, R.; Berg, G. Strain-specific colonization pattern of Rhizoctonia antagonists in the root system of sugar beet. FEMS Microb. Ecol 2010, 74, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.C.; Bakker, P.A.H.M. PGPR: Biocontrol and Biofertilization. In Springer, Dordrecht, The Netherlands; Siddiqui, Z.A., Ed.; Springer: Dordrecht, The Netherlands, 2005; pp. 39–66. [Google Scholar]
- Adesina, M.F.; Grosch, R.; Lembke, A.; Vatchev, T.D.; Smalla, K. In vitro antagonists of Rhizoctonia solani tested on lettuce: Rhizosphere competence, biocontrol efficiency and rhizosphere microbial community response: Research article. FEMS Microbiol. Ecol. 2009, 69, 62–74. [Google Scholar] [CrossRef] [PubMed]
- De Vleesschauwer, D.; Chernin, L.; Höfte, M.M. Differential effectiveness of Serratia plymuthica IC1270-induced systemic resistance against hemibiotrophic and necrotrophic leaf pathogens in rice. BMC Plant Biol. 2009, 16, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Adesina, M.F.; Lembke, A.; Costa, R.; Speksnijder, A.; Smalla, K. Screening of bacterial isolates from various European soils for in vitro antagonistic activity towards Rhizoctonia solani and Fusarium oxysporum: Site-dependent composition and diversity revealed. Soil Biol. Biochem. 2007, 39, 2818–2828. [Google Scholar] [CrossRef]
- Adam, E.; Müller, H.; Erlacher, A.; Berg, G. Complete genome sequences of the Serratia plymuthica strains 3Rp8 and 3Re4-18, two rhizosphere bacteria with antagonistic activity towards fungal phytopathogens and plant growth promoting abilities. Stand. Genom. Sci. 2016, 11, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Brandel, J.; Humbert, N.; Elhabiri, M.; Schalk, I.J.; Mislin, G.L.A.; Abrecht-Gary, A.-M. Pyochelin, a siderophore of Pseudomonas aeruginosa: Physicochemical characterization of the iron (III), copper (II) and zinc (II) complexes. Dalton Trans. 2012, 41, 2820–2834. [Google Scholar] [CrossRef] [PubMed]
- Audenaert, K.; Patterey, T.; Cornelis, P.; Höfte, M. Induction of Systemic Resistance to Botrytis cinerea in Tomato by Pseudomonas aeruginosa 7NSK2: Role of Salicylic Acid, Pyochelin, and Pyocyanin. Mol. Plant-Microbe Interact. 2002, 15, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- Neumann, G.; Römheld, V. Root-induced changes in the availability of nutrients in the rhizosphere. In Plant Roots, The Hidden Half; Waisel, Y., Eshel, A., Kafkafi, U., Eds.; CRC Press: Boca Raton, FL, USA, 2002; pp. 617–649. [Google Scholar]
- Glick, B.R.; Cheng, Z.; Czarny, J.; Duan, J. Promotion of plant growth by ACC deaminase-producing soil bacteria. In New Perspect. Approaches Plant Growth-Promoting Rhizobacteria Resarch; Bakker, P.A.H.M., Raaijmakers, J.M., Bloemberg, G., Höfte, M., Lemanceau, P., Cooke, B.M., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 329–339. [Google Scholar]
- Dandurishvili, N.; Toklikishvili, N.; Ovadis, M.; Eliashvili, P.; Giorgobiani, N.; Keshelava, R.; Tediashvili, M.; Vainstein, A.; Khmel, I.; Szegedi, E.; et al. Broad-range antagonistic rhizobacteria Pseudomonas fluorescens and Serratia plymuthica suppress Agrobacterium crown gall tumours on tomato plants. J. Appl. Microbiol. 2010, 110, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Gericke, S.; Kurmis, B. Die kolorimetrische Phosphorsäurebestimmung mit Ammonium-Vanadat-Molybdat und ihre Anwendung in der Pflanzenanalyse. Zeitschrift der Pflanzenernährung/Bodenkd 1958, 59, 235–247. [Google Scholar]
- Haase, S.; Neumann, G.; Kania, A.; Kuzyakov, Y.; Römheld, V.; Kandeler, E. Elevation of atmospheric CO2 and N-nutritional status modify nodulation, nodule-carbon supply, and root exudation of Phaseolus vulgaris L. Soil Biol. Biochem. 2007, 39, 2208–2221. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: London, UK, 1995. [Google Scholar]
- Weisskopf, L.; Abou-Mansour, E.; Fromin, N.; Tomasi, N.; Santelia, D.; Edelkott, I.; Neumann, G.; Aragno, M.; Tabacchi, R.; Martinoia, E. White lupin has developed a complex strategy to limit microbial degradation of secreted citrate required for phosphate acquisition. Plant Cell Environ. 2006, 29, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Lippmann, R.; Kaspar, S.; Rutten, T.; Melzer, M.; Kumlehn, J.; Matros, A.; Mock, H.P. Protein and metabolite analysis reveals permanent induction of stress defense and cell regeneration processes in a tobacco cell suspension culture. Int. J. Mol. Sci. 2009, 10, 3012–3032. [Google Scholar] [CrossRef] [PubMed]

| Treatment | Diluvial Sand | Alluvial Loam | Loess Loam |
|---|---|---|---|
| Control | 0.22 ± 0.2 | 1.13 ± 0.1 a | 1.33 ± 0.5 a |
| +Rhizoctonia | ++ | 0.70 ± 0.3 b | 1.45 ± 0.2 a |
| Treatment | Benzoic Acid | Lauric Acid | ||||||
|---|---|---|---|---|---|---|---|---|
| Subapical (Young) | Basal (Old) | Subapical (Young) | Basal (Old) | |||||
| AL | LL | AL | LL | AL | LL | AL | LL | |
| Control | 3.92 a | 5.00 a | 2.77 a | 6.30 a | 1.76 a | 4.29 a | 0.14 a | 14.00 a |
| +R. solani | 13.07 b | 5.53 a | 10.97 b | 8.12 a | 0.85 a | 5.25 a | 1.36 ab | 9.56 a |
| +P. jess. | 12.84 b | 10.54 b | 12.98 b | 13.58 ab | 1.00 a | 10.55 b | 1.52 b | 21.80 a |
| +S. ply. | 13.08 b | 10.49 b | 12.75 b | 9.58 ab | 0.51 a | 10.66 b | 1.01 ab | 9.40 a |
| +R. solani +P. jess. | 10.75 b | 9.62 b | 13.65 b | 15.53 b | 0.92 a | 9.29 b | 1.34 ab | 25.03 a |
| +R. solani +S. ply. | 11.71 b | 8.63 b | 12.68 b | 15.79 b | 1.00 a | 7.82 ab | 0.94 ab | 29.87 a |
| +P. jess./S. ply. | 12.84 b | N.d. | 12.63 b | N.d. | 0.85 a | N.d. | 0.93 ab | N.d. |
| +R. solani +P. jess./S. ply. | 12.14 b | N.d. | 11.31 b | N.d. | 0.43 a | N.d. | 1.09 ab | N.d. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Windisch, S.; Bott, S.; Ohler, M.-A.; Mock, H.-P.; Lippmann, R.; Grosch, R.; Smalla, K.; Ludewig, U.; Neumann, G. Rhizoctonia solani and Bacterial Inoculants Stimulate Root Exudation of Antifungal Compounds in Lettuce in a Soil-Type Specific Manner. Agronomy 2017, 7, 44. https://doi.org/10.3390/agronomy7020044
Windisch S, Bott S, Ohler M-A, Mock H-P, Lippmann R, Grosch R, Smalla K, Ludewig U, Neumann G. Rhizoctonia solani and Bacterial Inoculants Stimulate Root Exudation of Antifungal Compounds in Lettuce in a Soil-Type Specific Manner. Agronomy. 2017; 7(2):44. https://doi.org/10.3390/agronomy7020044
Chicago/Turabian StyleWindisch, Saskia, Sebastian Bott, Marc-Andreas Ohler, Hans-Peter Mock, Rico Lippmann, Rita Grosch, Kornelia Smalla, Uwe Ludewig, and Günter Neumann. 2017. "Rhizoctonia solani and Bacterial Inoculants Stimulate Root Exudation of Antifungal Compounds in Lettuce in a Soil-Type Specific Manner" Agronomy 7, no. 2: 44. https://doi.org/10.3390/agronomy7020044
APA StyleWindisch, S., Bott, S., Ohler, M.-A., Mock, H.-P., Lippmann, R., Grosch, R., Smalla, K., Ludewig, U., & Neumann, G. (2017). Rhizoctonia solani and Bacterial Inoculants Stimulate Root Exudation of Antifungal Compounds in Lettuce in a Soil-Type Specific Manner. Agronomy, 7(2), 44. https://doi.org/10.3390/agronomy7020044






