Biochar for Horticultural Rooting Media Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biochars Produed
2.2. Rooting Medium Testing
2.2.1. Physical Characterization of Biochars
2.2.2. Water Holding Capacity
2.2.3. Chemical Characterization of Biochars
2.2.4. Acid-Neutralizing Capacity
2.2.5. Phytotoxicity Test
2.2.6. Oxygen Uptake Rate
2.3. Climate Chamber Experiment with Gerbera and Powdery Mildew
2.3.1. Treatments
- Standard rooting medium (milled white peat)
- Standard rooting medium with 20% v/v biochar
- Standard rooting medium + fungicide (0.1% triflumizool)
- Standard rooting medium + SAR elicitor (chemically induced disease resistance)
2.3.2. Assessment and Harvest
2.4. Greenhouse Experiment with Added Microorganisms and Chrysanthemum
2.4.1. Treatments
- Peat
- Peat + plant-growth promoting rhizobacteria
- Peat + biochar
- Peat + plant-growth promoting rhizobacteria
2.4.2. Cultivation
2.4.3. Bacterial Analysis
3. Results
3.1. Suitability of Biochars as a Rooting Medium
3.1.1. Physical Properties
3.1.2. Water Holding Capacity
3.1.3. Chemical Composition and Nutrient Values of Biochar
3.1.4. Acid-Neutralizing Capacity
3.1.5. Phytotoxicity
3.1.6. Stability
3.2. Effect of Biochar on Plant Growth and Powdery Mildew Infection of Gerbera
3.2.1. Effect of Biochar Addition on Rooting Medium Quality
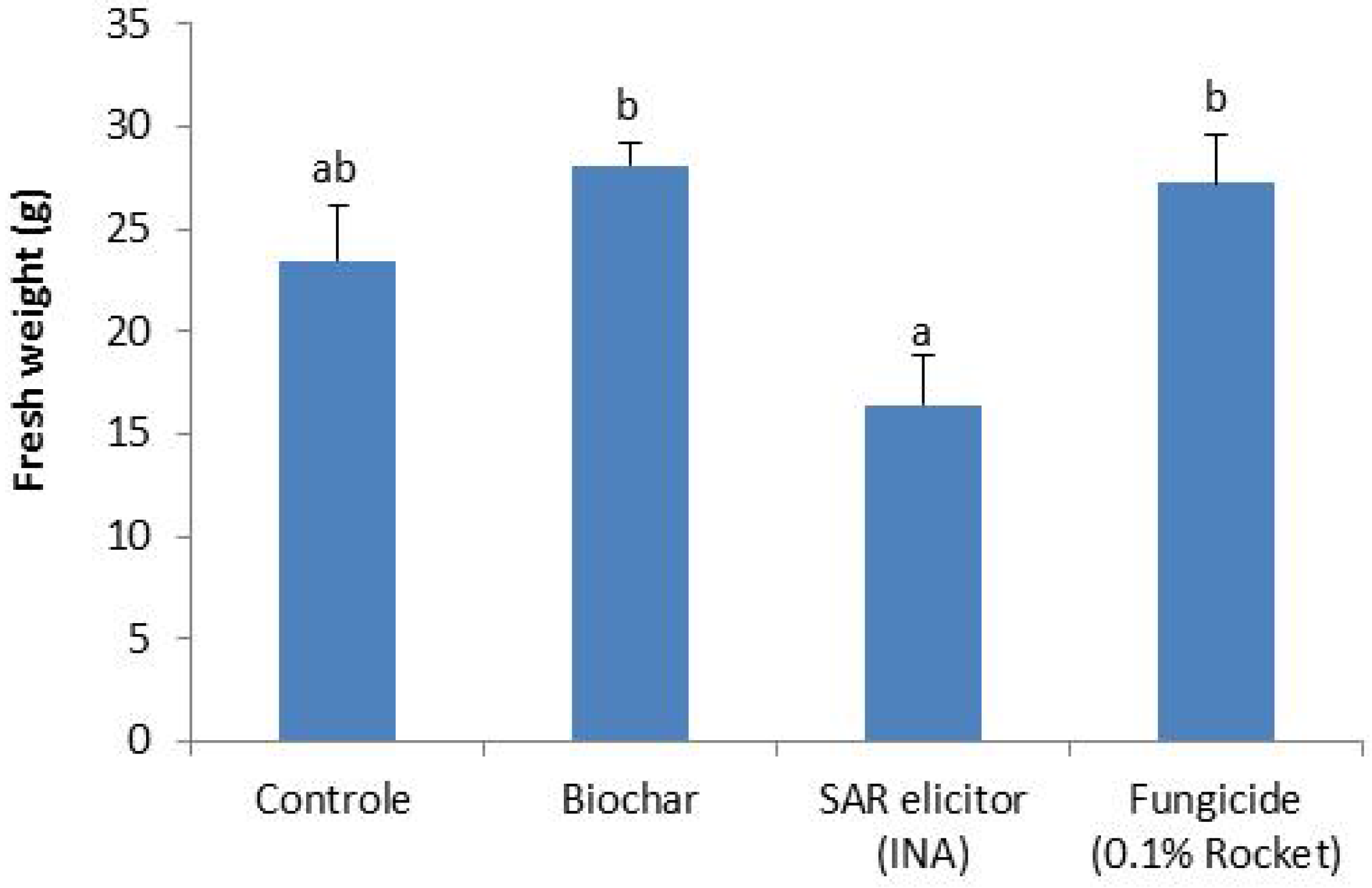
3.2.2. Effect of Biochar Addition on Plant Growth and Disease Suppression
3.3. Effect of Biochar and Added Microorganisms on Plant Growth of Chrysanthemum
3.3.1. Effect of Biochar Addition on Rooting Medium Quality
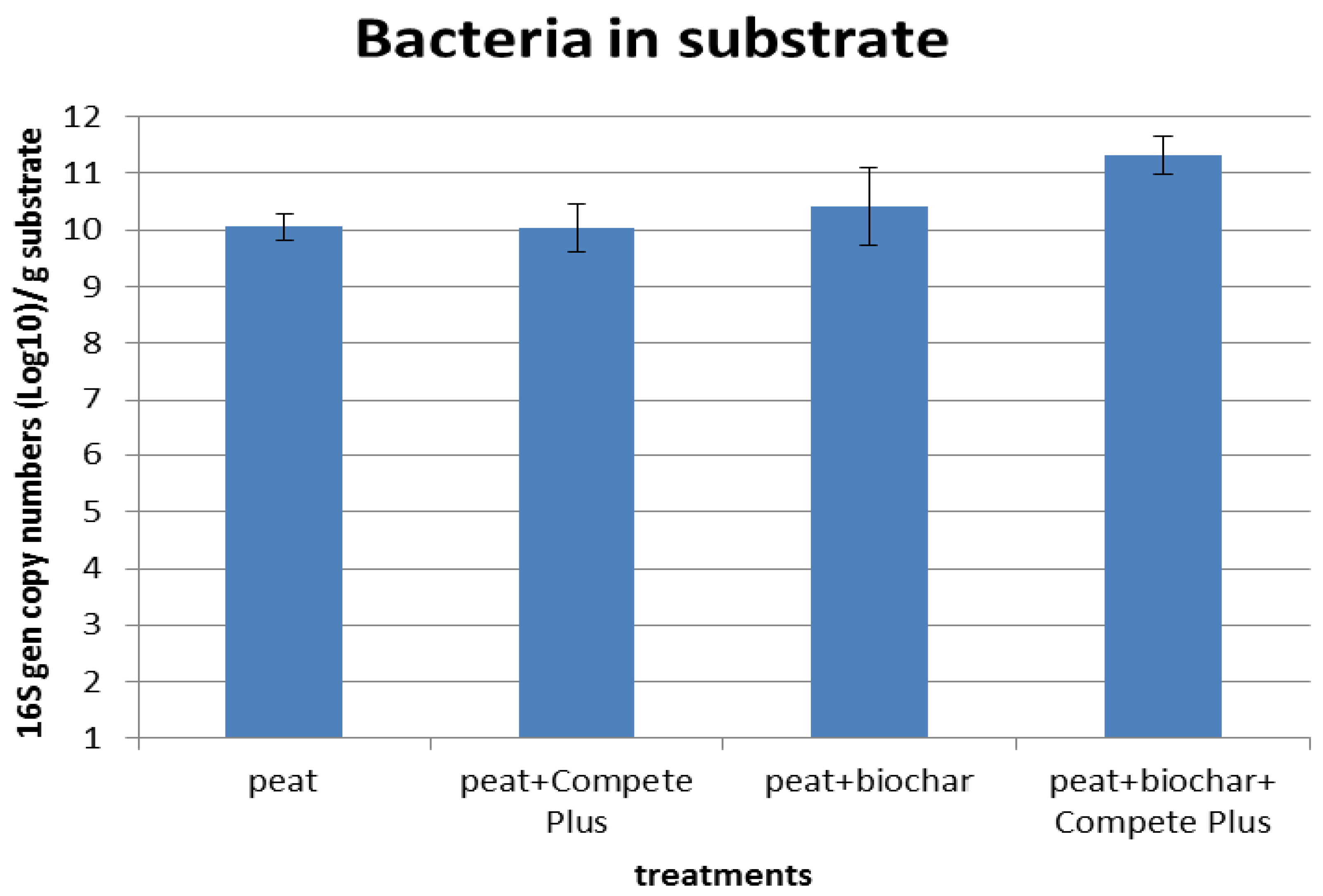
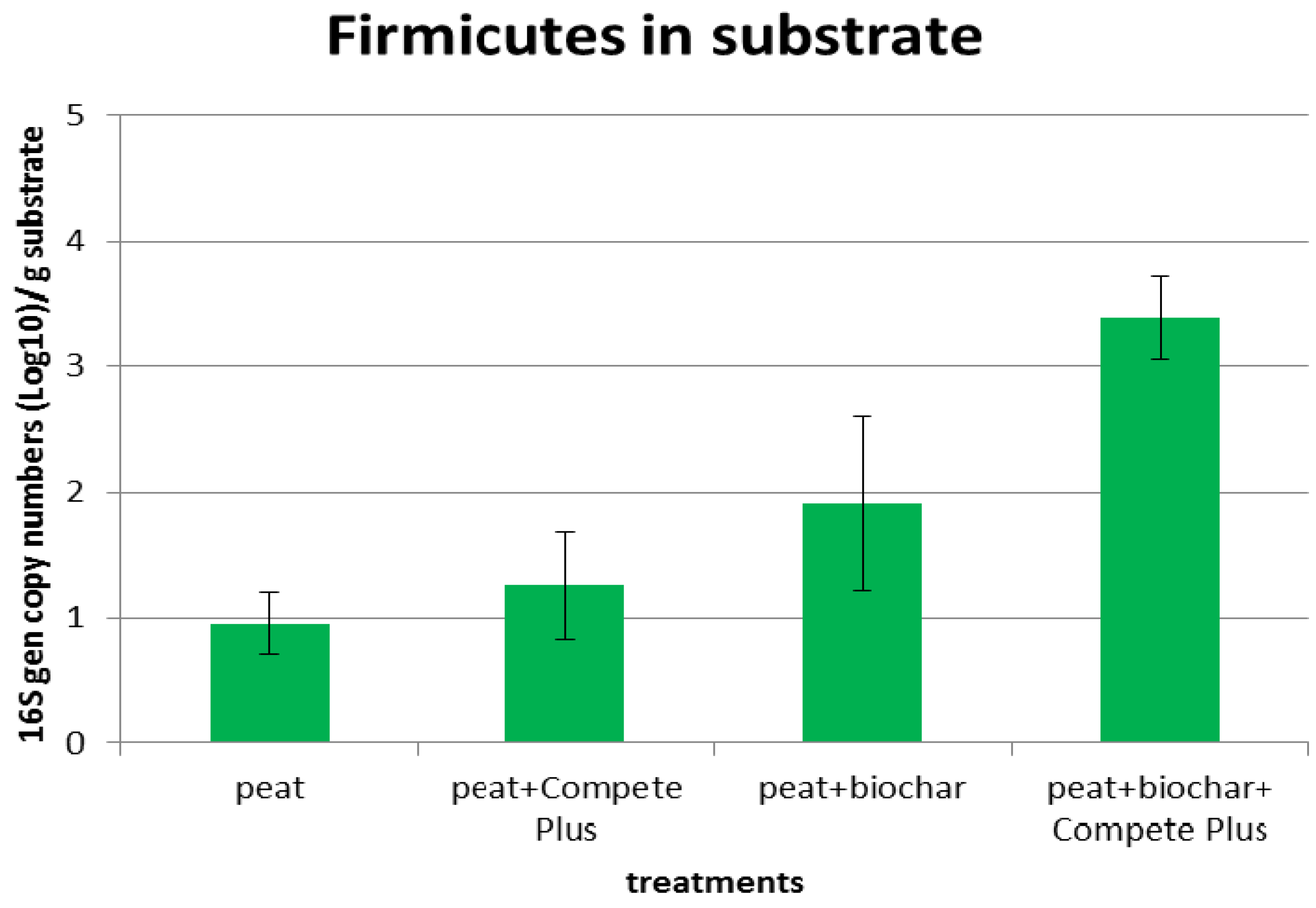
3.3.2. Effect of Biochar Addition on Plant Growth and Added Microorganisms
3.4. Suitability of Feedstocks for Application as Rooting Medium
4. Discussion
4.1. Feedstock Related Effects of Biochar
4.2. Production Setting Related Effects of Biochar
4.3. The Survival Rate of Beneficial Microorganisms as Affected by Biochar
4.4. Horticultural Perspectives of Biochar
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: Science and Technology, 2nd ed.; Earthscan: London, UK, 2015. [Google Scholar]
- Laird, D.A. The charcoal vision: A win-win-win scenario for simultaneously producing bioenergy, permanently sequestering carbon, while improving soil and water quality. Agron. J. 2008, 100, 178–181. [Google Scholar] [CrossRef]
- Atkinson, C.; Fitzgerald, J.; Hipps, N. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil 2010, 337, 1–18. [Google Scholar] [CrossRef]
- Singh, B.; Singh, B.P.; Cowie, A.L. Characterisation and evaluation of biochars for their application as a soil amendment. Soil Res. 2010, 48, 516–525. [Google Scholar] [CrossRef]
- Glaser, B.; Wiedner, K.; Seelig, S.; Schmidt, H.-P.; Gerber, H. Biochar organic fertilizers from natural resources as substitute for mineral fertilizers. Agron. Sustain. Dev. 2014, 35, 667–678. [Google Scholar] [CrossRef]
- Olmo, M.; Villar, R.; Salazar, P.; Alburquerque, J. Changes in soil nutrient availability explain biochar’s impact on wheat root development. Plant Soil 2016, 399, 333–343. [Google Scholar] [CrossRef]
- Akhter, A.; Hage-Ahmed, K.; Soja, G.; Steinkellner, S. Compost and biochar alter mycorrhization, tomato root exudation, and development of Fusarium oxysporum f. Sp. lycopersici. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Minchin, T.; Kimber, S.; van Zwieten, L.; Gilbert, J.; Munroe, P.; Joseph, S.; Thomas, T. Comparative analysis of the microbial communities in agricultural soil amended with enhanced biochars or traditional fertilisers. Agric. Ecosyst. Environ. 2014, 191, 73–82. [Google Scholar] [CrossRef]
- Elad, Y.; David, D.R.; Harel, Y.M.; Borenshtein, M.; Kalifa, H.B.; Silber, A.; Graber, E.R. Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 2010, 100, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Nemati, M.R.; Simard, F.; Fortin, J.-P.; Beaudoin, J. Potential use of biochar in growing media. Vadose Zone J. 2015, 14. [Google Scholar] [CrossRef]
- Nieto, A.; Gascó, G.; Paz-Ferreiro, J.; Fernández, J.M.; Plaza, C.; Méndez, A. The effect of pruning waste and biochar addition on brown peat based growing media properties. Sci. Hortic. 2016, 199, 142–148. [Google Scholar] [CrossRef]
- Steiner, C.; Harttung, T. Biochar as growing media additive and peat substitute. Solid Earth Discuss. 2014, 6, 1023–1035. [Google Scholar] [CrossRef]
- Verhagen, J.; van den Akker, J.; Blok, C.; Diemont, H.; Joosten, H.; Schouten, M.; Schrijver, R.; Verweij, P.; Wösten, H. Climate Change. Scientific Assessment and Policy Analysis: Peatlands and Carbon Flows: Outlook and Importance for the Netherlands; Wab 500102 027; Netherlands Environmental Assessment Agency PBL: Bilthoven, The Netherlands, 2009. [Google Scholar]
- Graber, E.R.; Harel, Y.M.; Kolton, M.; Cytryn, E.; Silber, A.; David, D.R.; Tsechansky, L.; Borenshtein, M.; Elad, Y. Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant Soil 2010, 337, 481–496. [Google Scholar] [CrossRef]
- Jaiswal, A.; Frenkel, O.; Elad, Y.; Lew, B.; Graber, E. Non-monotonic influence of biochar dose on bean seedling growth and susceptibility to rhizoctonia solani: The “shifted rmax-effect”. Plant Soil 2015, 395, 125–140. [Google Scholar] [CrossRef]
- Elad, Y.; Cytryn, E.; Meller Harel, Y.; Lew, B.; Graber, E.R. The biochar effect: Plant resistance to biotic stresses. Phytopathol. Mediterr. 2012, 50, 335–349. [Google Scholar]
- De Tender, C.A.; Debode, J.; Vandecasteele, B.; D’Hose, T.; Cremelie, P.; Haegeman, A.; Ruttink, T.; Dawyndt, P.; Maes, M. Biological, physicochemical and plant health responses in lettuce and strawberry in soil or peat amended with biochar. Appl. Soil Ecol. 2016, 107, 1–12. [Google Scholar] [CrossRef]
- Altland, J.E.; Locke, J.C. Biochar affects macronutrient leaching from a soilless substrate. HortScience 2012, 47, 1136–1140. [Google Scholar]
- Kloss, S.; Zehetner, F.; Wimmer, B.; Buecker, J.; Rempt, F.; Soja, G. Biochar application to temperate soils: Effects on soil fertility and crop growth under greenhouse conditions. J. Plant Nutr. Soil Sci. 2014, 177, 3–15. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Heiskanen, J.; Englund, K.; Tervahauta, A. Pelleted biochar: Chemical and physical properties show potential use as a substrate in container nurseries. Biomass Bioenergy 2011, 35, 2018–2027. [Google Scholar] [CrossRef]
- Bedussi, F.; Zaccheo, P.; Crippa, L. Pattern of pore water nutrients in planted and non-planted soilless substrates as affected by the addition of biochars from wood gasification. Biol. Fertil. Soils 2015, 51, 625–635. [Google Scholar] [CrossRef]
- Dunlop, S.J.; Arbestain, M.C.; Bishop, P.A.; Wargent, J.J. Closing the loop: Use of biochar produced from tomato crop green waste as a substrate for soilless, hydroponic tomato production. Hortscience 2015, 50, 1572–1581. [Google Scholar]
- Vaughn, S.F.; Eller, F.J.; Evangelista, R.L.; Moser, B.R.; Lee, E.; Wagner, R.E.; Peterson, S.C. Evaluation of biochar-anaerobic potato digestate mixtures as renewable components of horticultural potting media. Ind. Crops Prod. 2015, 65, 467–471. [Google Scholar] [CrossRef]
- Altland, J.E.; Locke, J.C. Gasified rice hull biochar is a source of phosphorus and potassium for container-grown plants. J. Environ. Hortic. 2013, 31, 138–144. [Google Scholar]
- Locke, J.C.; Altland, J.E.; Ford, C.W. Gasified rice hull biochar affects nutrition and growth of horticultural crops in container substrates. J. Environ. Hortic. 2013, 31, 195–202. [Google Scholar]
- Trifonova, R.; Postma, J.; Ketelaars, J.J.M.H.; van Elsas, J.D. Torrefied grass fibres as a substitute for peat in potting soil. In Presented at ORBIT Conference, Wageningen, The Netherlands, 13–15 October 2008.
- Blok, C.; Rijpsma, E.; Ketelaars, J.J.M.H. New growing media and value added organic waste processing. ISHS Acta Hortic. 2016, 1112, 269–280. [Google Scholar] [CrossRef]
- Quintero, M.F.; Ortega, D.; Valenzuela, J.L.; Guzman, M. Variation of hydro-physical properties of burnt rice husk used for carnation crops: Improvement of fertigation criteria. Sci. Hortic. 2013, 154, 82–87. [Google Scholar] [CrossRef]
- Singh, B.; Macdonald, L.M.; Kookana, R.S.; van Zwieten, L.; Butler, G.; Joseph, S.; Weatherley, A.; Kaudal, B.B.; Regan, A.; Cattle, J.; et al. Opportunities and constraints for biochar technology in australian agriculture: Looking beyond carbon sequestration. Soil Res. 2014, 52, 739–750. [Google Scholar] [CrossRef]
- Graber, E.; Tsechansky, L.; Gerstl, Z.; Lew, B. High surface area biochar negatively impacts herbicide efficacy. Plant Soil 2012, 353, 95–106. [Google Scholar] [CrossRef]
- Abiven, S.; Schmidt, M.W.I.; Lehmann, J. Biochar by design. Nat. Geosci. 2014, 7, 326–327. [Google Scholar] [CrossRef]
- Schimmelpfennig, S.; Glaser, B. One step forward toward characterization: Some important material properties to distinguish biochars. J. Environ. Qual. 2012, 41, 1001–1013. [Google Scholar] [CrossRef] [PubMed]
- International Biochar Initiative. Standardized Product Definition and Product Testing Guidelines for Biochar That Is Used in Soil; International Biochar Initiative: Westerville, OH, USA, 2012. [Google Scholar]
- Mukherjee, A.; Zimmerman, A.R.; Harris, W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 2011, 163, 247–255. [Google Scholar] [CrossRef]
- Blok, C.; Kreij, C.D.; Baas, R.; Wever, G. Analytical Methods Used in Soilless Cultivation. In Soilless Culture : Theory and Practice; Elsevier: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Kloss, S.; Zehetner, F.; Dellantonio, A.; Hamid, R.; Ottner, F.; Liedtke, V.; Schwanninger, M.; Gerzabek, M.H.; Soja, G. Characterization of slow pyrolysis biochars: Effects of feedstocks and pyrolysis temperature on biochar properties. J. Environ. Qual. 2012, 41, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- Morales, V.L.; Pérez-Reche, F.J.; Hapca, S.M.; Hanley, K.L.; Lehmann, J.; Zhang, W. Reverse engineering of biochar. Bioresour. Technol. 2015, 183, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Mimmo, T.; Panzacchi, P.; Baratieri, M.; Davies, C.A.; Tonon, G. Effect of pyrolysis temperature on miscanthus (Miscanthus × giganteus) biochar physical, chemical and functional properties. Biomass Bioenergy 2014, 62, 149–157. [Google Scholar] [CrossRef]
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Gray, M.; Johnson, M.; Dragila, M.I.; Kleber, M. Water uptake in biochars: The roles of porosity and hydrophobicity. Biomass Bioenergy 2014, 61, 196–205. [Google Scholar] [CrossRef]
- Fryda, L.; Visser, R. Biochar for soil improvement: Evaluation of biochar from gasification and slow pyrolysis. Agriculture 2015, 5, 1076–1115. [Google Scholar] [CrossRef]
- Urrestarazu Gavilán, M. Tratado de Cultivo Sin Suelo; Mundi-Prensa: Madrid, Spain, 2004. [Google Scholar]
- CEN, European Committee for Standardization. Soil Improvers and Growing Media—Determination of Physical Properties—Dry Bulk Density, Air Volume, Water Volume, Shrinkage Value and Total Pore Space; prEN 13041; Technical Committee 223: Berlin, Germany, 2006. [Google Scholar]
- CEN, European Committee for Standardization. Soil Improvers and Growing Media—Determination of Organic Content and Ash; prEN 13039; Technical Committee 223: Berlin, Germany, 2007. [Google Scholar]
- CEN, European Committee for Standardization. Soil Improvers and Growing Media—Determination of Electrical Conductivity; prEN 13038; Technical Committee 223: Berlin, Germany, 2008. [Google Scholar]
- Verhagen, J.B.G.M. Cec and the saturation of the adsorption complex of coir dust. Acta Hortic. 1999, 481, 151–155. [Google Scholar] [CrossRef]
- ISO/DIS 11260. Soil Quality—Determination of Effective Cation Exchange Capacity and Base Saturation Level Using Barium Chloride Solution; Technical Committee 190: Berlin, Germany, 2011. [Google Scholar]
- Blok, C.; Kaarsemaker, R. Ph in rockwool propagation blocks: A method to measure the ph buffer capacity of rockwool and other mineral wool media. Acta Hortic. 2013, 1013, 65–72. [Google Scholar] [CrossRef]
- Blok, C.; Wever, G.; Persoone, G. A practical and low cost microbiotest to assess the phytotoxic potential of growing media and soil. Acta Hortic. 2008, 779, 367–374. [Google Scholar] [CrossRef]
- CEN, European Committee for Standardization. Soil Improvers and Growing Media—Determination of Plant Response—Part 2: Petri Dish Test; EN 16086; Technical Committee 223: Berlin, Germany, 2008. [Google Scholar]
- CEN, European Committee for Standardization. Pren_00223089 2009 Determination of Aerobic Biological actIvity—Part 1: Oxygen Uptake Rate (Our); EN 16087; Technical Committee 223: Berlin, Germany, 2009. [Google Scholar]
- Spencer, D.M. Standardized methods for the evaluation of fungicides to control cucumber powdery mildew. In Crop Protection Agents—Their Biological Evaluation; McFarlane, N.R., Ed.; Academic Press: London, UK, 1977; pp. 455–464. [Google Scholar]
- Verhagen, J.B.G.M. Oxygen diffusion in relation to physical characteristics of growing media. Acta Hortic. 2013, 1013, 313–318. [Google Scholar] [CrossRef]
- Londra, P.A. Simultaneous determination of water retention curve and unsaturated hydraulic conductivity of substrates using a steady-state laboratory method. HortScience 2010, 45, 1106–1112. [Google Scholar]
- Trifonova, R.; Postma, J.; Verstappen, F.W.A.; Bouwmeester, H.J.; Ketelaars, J.J.; Van Elsas, J.-D. Removal of phytotoxic compounds from torrefied grass fibres by plant-beneficial microorganisms. FEMS Microbiol. Ecol. 2008, 66, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, F.; Consoli, S. Reusing perlite substrates in soilless cultivation: Analysis of particle size, hydraulic properties, and solarization effects. J. Irrig. Drain. Eng. 2016, 142. [Google Scholar] [CrossRef]
- Dede, O.H.; Dede, G.; Ozdemir, S.; Abad, M. Physicochemical characterization of hazelnut husk residues with different decomposition degrees for soilless growing media preparation. J. Plant Nutr. 2011, 34, 1973–1984. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
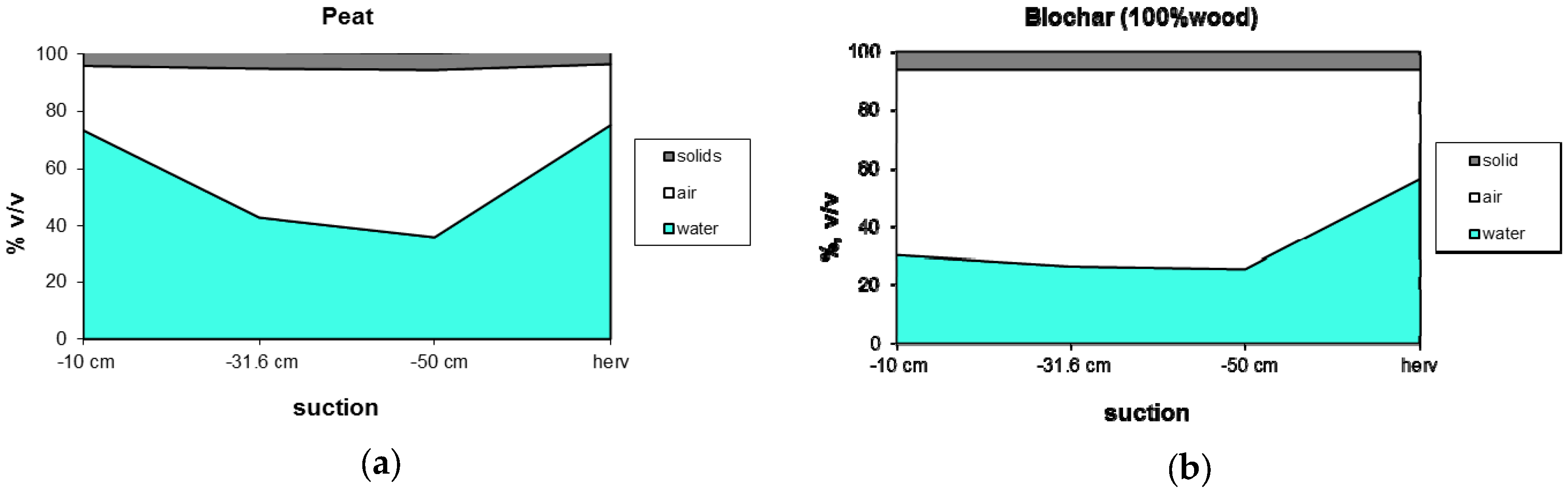
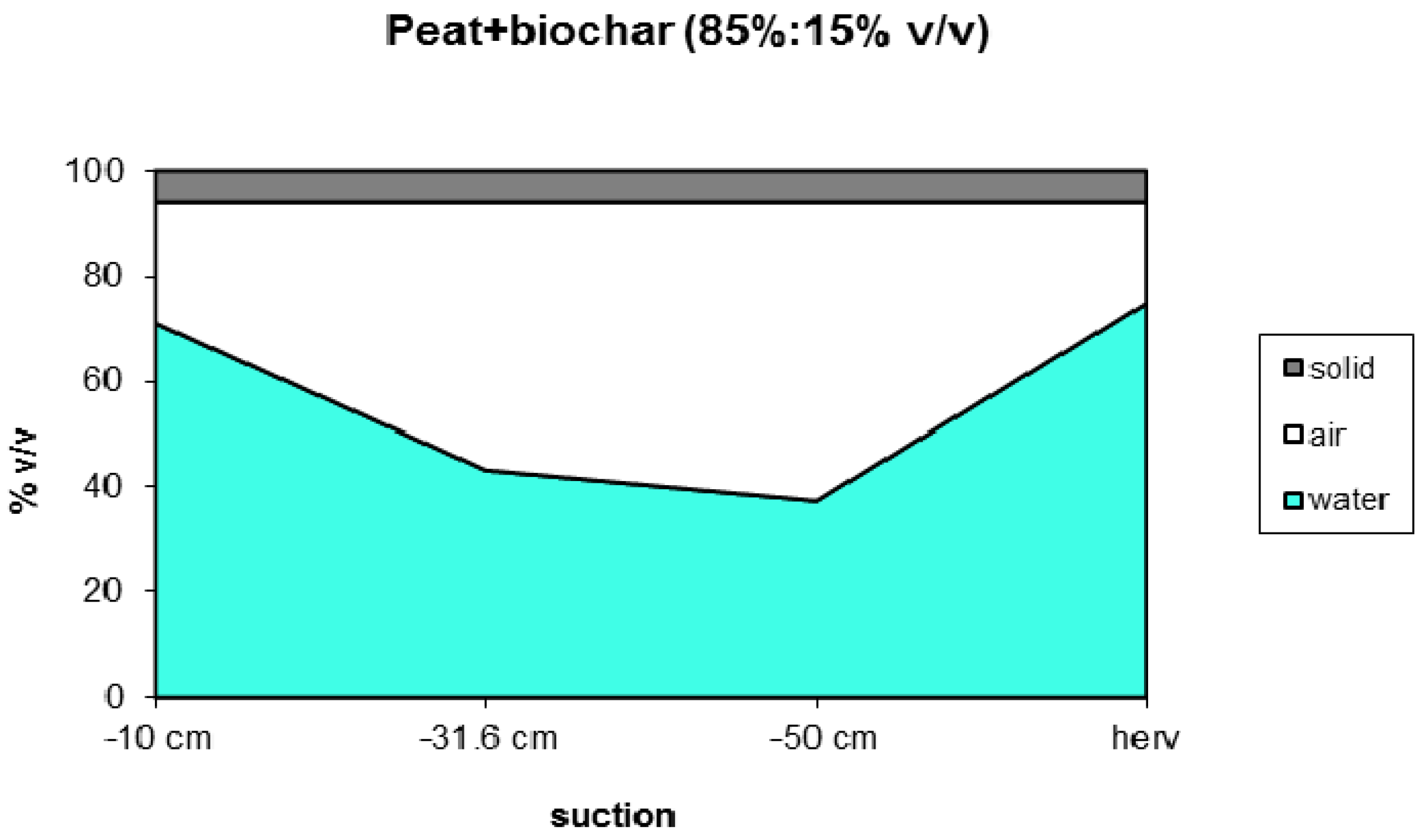
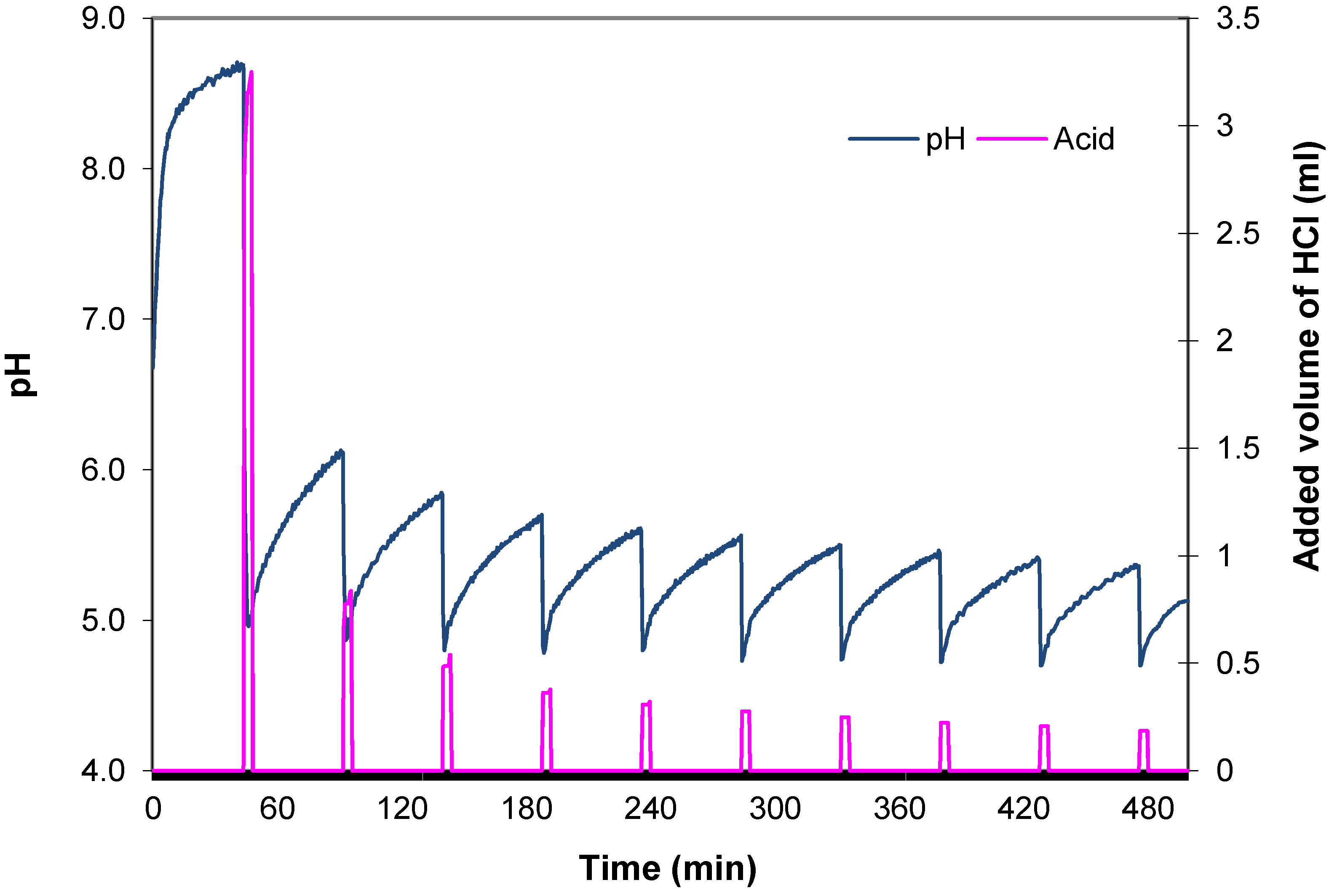
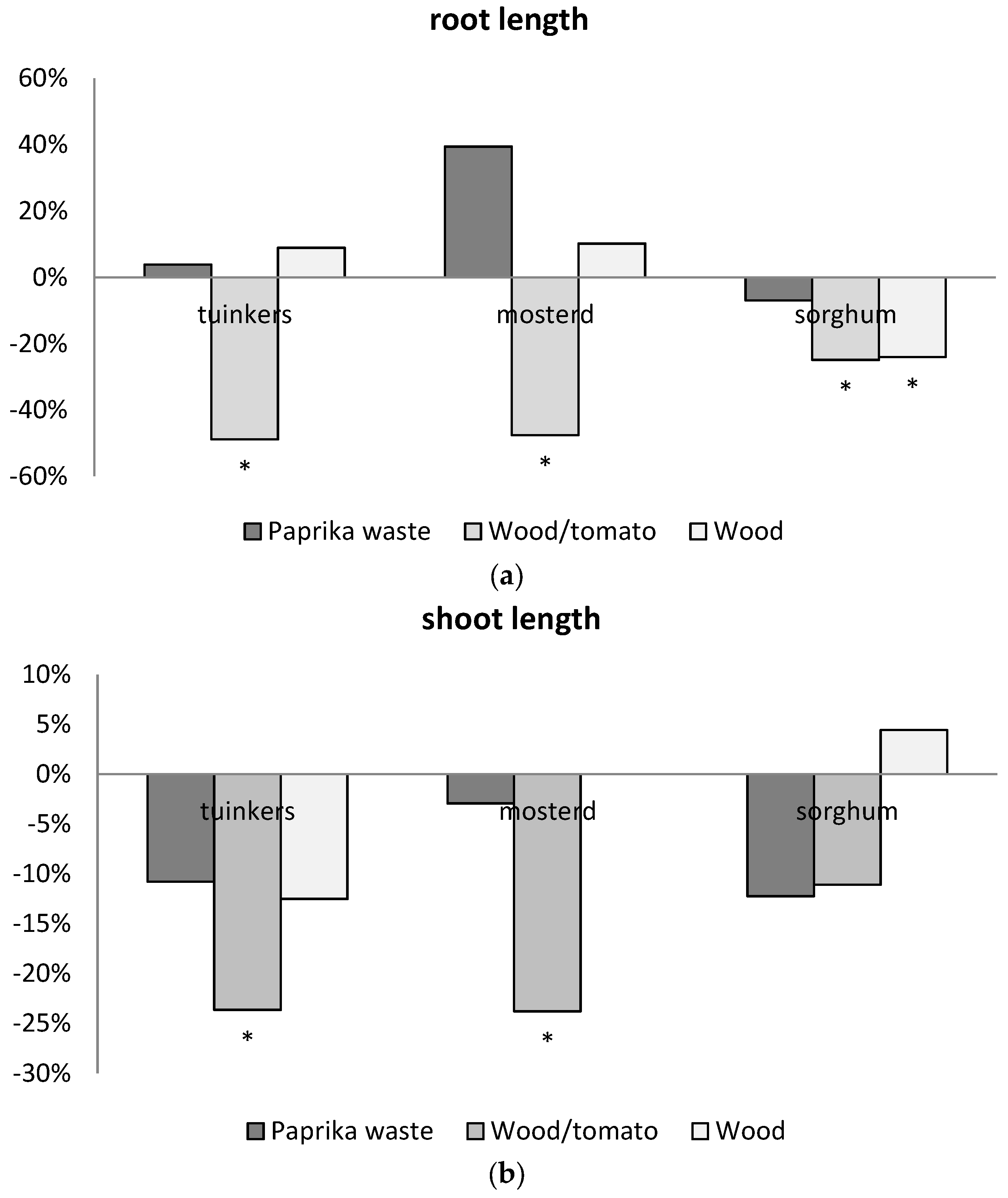

| Code | Biomass | T | Moisture | Ash | Volatiles | C | HHV |
|---|---|---|---|---|---|---|---|
| °C | %, a.r. | %, d.m. | %, d.m. | %, d.m. | MJ/kg | ||
| 1. Beech/Tomato | 80% beech wood + 20% tomato leaves | 670 | 5.4 | 17.5 | 12.3 | 70.2 | 27.5 |
| 2. Wood/Tomato | 80% wood chips-1 * + 20% tomato leaves | 670 | 3.3 | 22.5 | 13.3 | 64.2 | 24.4 |
| 3. Wood chips-1 | Batch spring 2015 * | 670 | - | - | - | - | |
| 4. Sweet pepper waste | Vegetable residues (Spain) | 670 | 4.5 | 33.6 | 14.7 | 51.7 | 21.3 |
| 5. Sweet pepper waste | Vegetable residues (Spain) | 750 | 4.5 | 26.5 | 15.1 | 58.4 | 21.2 |
| 6. Wood chips-2 ** | Batch July 2015 * | 670 | 3.2 | 10.7 | 10.2 | 79.1 | 29.7 |
| 7. Wood chips-3 | Batch August 2015 * | 670 | - | - | - | - | |
| 8. Wood chips *** | Beech wood chips | 670 | 2.7 | 23.8 | 6.0 | 70.2 | 27.1 |
| 1 | 2 * | 3 | 4 | 5 | 6 * | 7 | 8 * | ||
|---|---|---|---|---|---|---|---|---|---|
| Tomato | Wood | Pepper | Pepper | Residual wood | |||||
| Parameter | Unit | Beech | Wood | Beech | 650 °C | 750 °C | Batch 1 ** | Batch 2 | Batch 3 *** |
| pH | - | 11 | 12 | 11 | 12 | 12 | 9.4 | 9.9 | 10 |
| EC | dS·m−1 | 6.8 | 13 | 0.68 | 9.6 | 11 | 0.53 | 0.61 | 0.71 |
| NH4 | mmol·L−1 | <0.1 | 0.1 | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 |
| K | mmol·L−1 | 49.1 | 94.3 | 3.2 | 61 | 84.3 | 3.6 | 3.6 | 4.5 |
| Na | mmol·L−1 | 0.5 | 1 | 0.1 | 5.1 | 7.4 | 0.2 | 0.2 | 0.3 |
| Ca | mmol·L−1 | 0.3 | 3 | 0.3 | 0.3 | 0.5 | 0.2 | 0.3 | 0.2 |
| Mg | mmol·L−1 | 0.1 | <0.1 | <0.1 | 0.2 | 0.1 | 0.2 | 0.2 | 0.1 |
| Si | mmol·L−1 | 0.3 | 0.3 | <0.1 | 0.1 | 0.2 | <0.1 | <0.1 | <0.1 |
| NO3 | mmol·L−1 | <0.1 | 0.2 | <0.1 | 0.1 | 0.1 | <0.1 | 0.1 | <0.1 |
| Cl | mmol·L−1 | 23.6 | 48.5 | 0.1 | 47.8 | 65.2 | 0.2 | 0.4 | 0.6 |
| SO4 | mmol·L−1 | 10.8 | 21.4 | 0.3 | 4.1 | 4.3 | 0.4 | 0.2 | 0.2 |
| HCO3 | mmol·L−1 | 3.3 | 7.2 | 3.6 | 18.9 | 19.5 | 3.2 | 3.7 | 4.3 |
| P | mmol·L−1 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | 0.1 | <0.05 | <0.05 |
| Fe | μmol·L−1 | 0.4 | 0.9 | <0.4 | <0.4 | <0.4 | 0.5 | 0.6 | 0.5 |
| Mn | μmol·L−1 | 0.2 | <0.1 | <0.1 | <0.1 | <0.1 | 2.2 | 0.7 | 0.2 |
| Zn | μmol·L−1 | 0.1 | 0.1 | <0.1 | <0.1 | <0.1 | 0.2 | 0.1 | 0.1 |
| B | μmol·L−1 | 13 | 9 | 10 | 28 | 35 | 8 | 7 | 6 |
| Cu | μmol·L−1 | 0.1 | 5 | <0.1 | 0.2 | 0.1 | <0.1 | <0.1 | <0.1 |
| Mo | μmol·L−1 | 0.5 | 0.9 | <0.1 | <0.1 | <0.1 | <0.1 | 0.10 | <0.1 |
| Dry weight | % | 96.7 | 97.1 | 95.5 | 95.5 | 97.8 | |||
| Bulk density | kg·m−3 | 131 | 113 | 104 | 129 | 102 | |||
| SSA | m2·g−1 d.m. | 59 | 81 | 39 | 29 | 119 | |||
| Ash | % d.m. | 19 | 28 | 35 | 34 | 13 | |||
| total C | % d.m. | 77 | 68 | 59 | 59 | 82 | |||
| total H | % d.m. | 1.3 | 1.4 | 1.2 | 1.3 | 1.5 | |||
| Total N | % d.m. | 0.7 | 1.0 | 0.8 | 0.9 | 0.8 | |||
| C/N ratio | mol·mol−1 | 128 | 79 | 86 | 76 | 119 | |||
| Medium | Acid Buffering Capacity | Base Buffering Capacity | ||
|---|---|---|---|---|
| mmol·kg−1 d.m. | mol·m−3 | mmol·kg−1 d.m. | mol·m−3 | |
| Biochar (wood chips) | 258 | 26 | - | - |
| Peat 2 | - | - | 90 | 9 |
| Substrate | Bulk Density | Solid Fraction | Total Pore Space |
|---|---|---|---|
| kg·m−3 | % v/v | % v/v | |
| Biochar (wood chips) | 100 ± 2.6 | 6.2 ± 0.17 | 93.8 ± 0.17 |
| Peat 2 | 91 ± 0.87 | 5.8 ± 0.06 | 94.2 ± 0.06 |
| Peat/biochar mixture (85%:15%) | 91 ± 1.7 | 5.8 ± 0.11 | 94.2 ± 0.11 |
| Material | OUR Value |
|---|---|
| Biochar Woodchips | 2.5 |
| Peat | 1.9 |
| Compost (for reference) | 5–10 |
| Parameter | Unit | Medium | Medium |
|---|---|---|---|
| Peat | Peat + Biochar | ||
| pH | - | 5.3 | 6.4 |
| EC | dS·m−1 | 0.65 | 0.54 |
| NH4 | mmol·L−1 | <0.1 | <0.1 |
| K | mmol·L−1 | 2.3 | 2.4 |
| Na | mmol·L−1 | 0.3 | 0.4 |
| Ca | mmol·L−1 | 0.7 | 0.4 |
| Mg | mmol·L−1 | 0.5 | 0.3 |
| Si | mmol·L−1 | <0.1 | <0.1 |
| NO3 | mmol·L−1 | 2.7 | 0.6 |
| Cl | mmol·L−1 | 0.1 | 0.1 |
| SO4 | mmol·L−1 | 0.8 | 1.1 |
| HCO3 | mmol·L−1 | <0.1 | <0.1 |
| P | mmol·L−1 | 0.65 | 0.65 |
| Fe | μmol·L−1 | 4.7 | 2.8 |
| Mn | μmol·L−1 | 0.4 | 0.2 |
| Zn | μmol·L−1 | 0.8 | 0.8 |
| B | μmol·L−1 | 6 | 8 |
| Cu | μmol·L−1 | 0.2 | 0.4 |
| Mo | μmol·L−1 | 0.1 | 0.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blok, C.; Van der Salm, C.; Hofland-Zijlstra, J.; Streminska, M.; Eveleens, B.; Regelink, I.; Fryda, L.; Visser, R. Biochar for Horticultural Rooting Media Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis. Agronomy 2017, 7, 6. https://doi.org/10.3390/agronomy7010006
Blok C, Van der Salm C, Hofland-Zijlstra J, Streminska M, Eveleens B, Regelink I, Fryda L, Visser R. Biochar for Horticultural Rooting Media Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis. Agronomy. 2017; 7(1):6. https://doi.org/10.3390/agronomy7010006
Chicago/Turabian StyleBlok, Chris, Caroline Van der Salm, Jantineke Hofland-Zijlstra, Marta Streminska, Barbara Eveleens, Inge Regelink, Lydia Fryda, and Rianne Visser. 2017. "Biochar for Horticultural Rooting Media Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis" Agronomy 7, no. 1: 6. https://doi.org/10.3390/agronomy7010006
APA StyleBlok, C., Van der Salm, C., Hofland-Zijlstra, J., Streminska, M., Eveleens, B., Regelink, I., Fryda, L., & Visser, R. (2017). Biochar for Horticultural Rooting Media Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis. Agronomy, 7(1), 6. https://doi.org/10.3390/agronomy7010006






