Different Responses of the Quality Parameters of Coriandrum sativum to Organic Substrate Mixtures and Fertilization
Abstract
:1. Introduction
2. Results and Discussion
2.1. Physicochemical Characterization of Substrate Mixtures
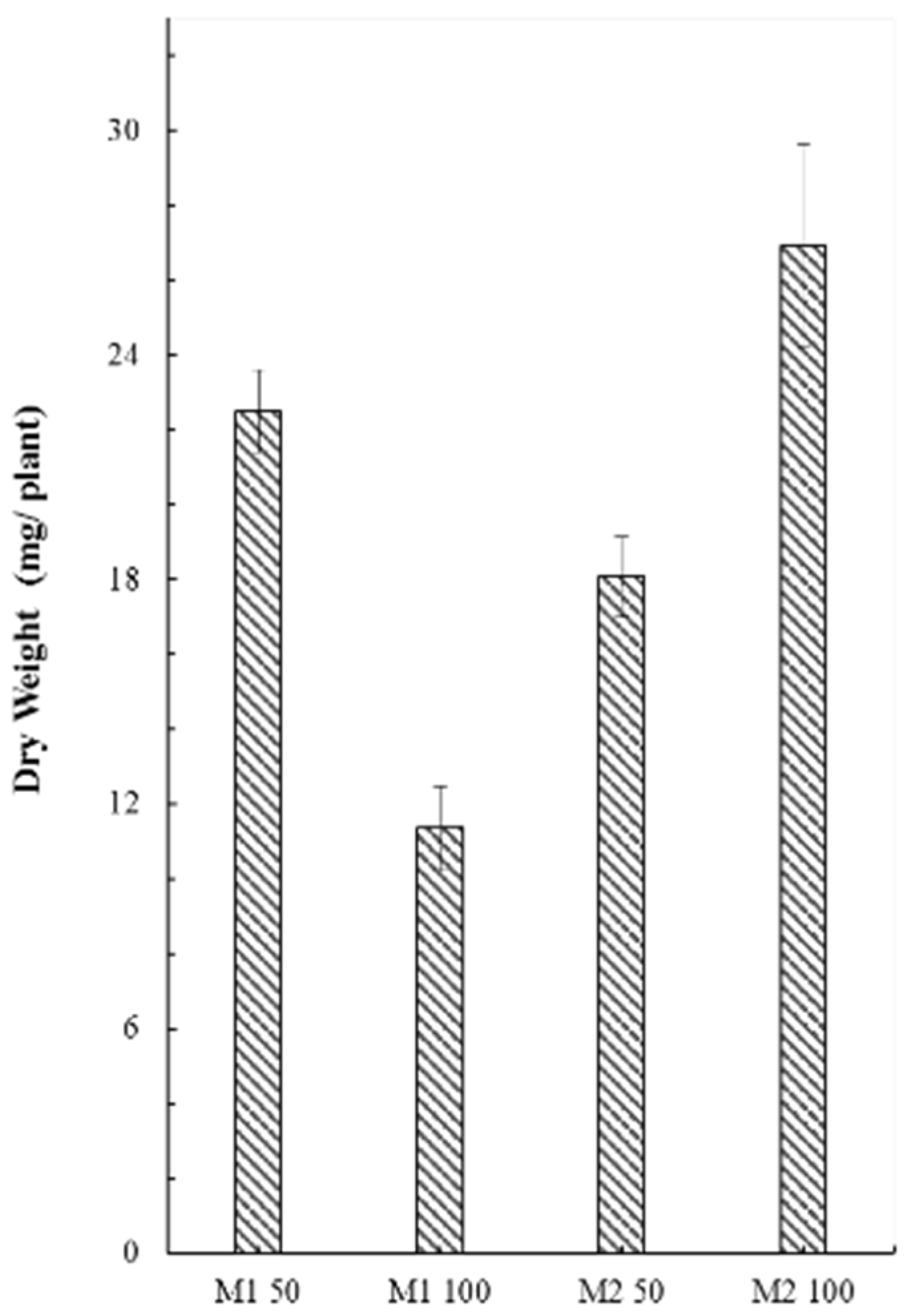
2.2. Evaluation of the Size Parameters of Coriander Cultivated on Two Substrate Mixtures with Two Different Concentrations of Fertilizer
2.3. Evaluation of Antioxidant Capacity
2.4. Determination of the Variation of Volatile Compounds Responsible for Aroma in Coriander Plants Cultivated in Different Substrate Mixtures and Two Fertilizer Concentrations
3. Experimental Section
3.1. Establishment of the Experimental Design and Determination of Sample Size to Ensure Statistical Reliability
3.2. Harvest
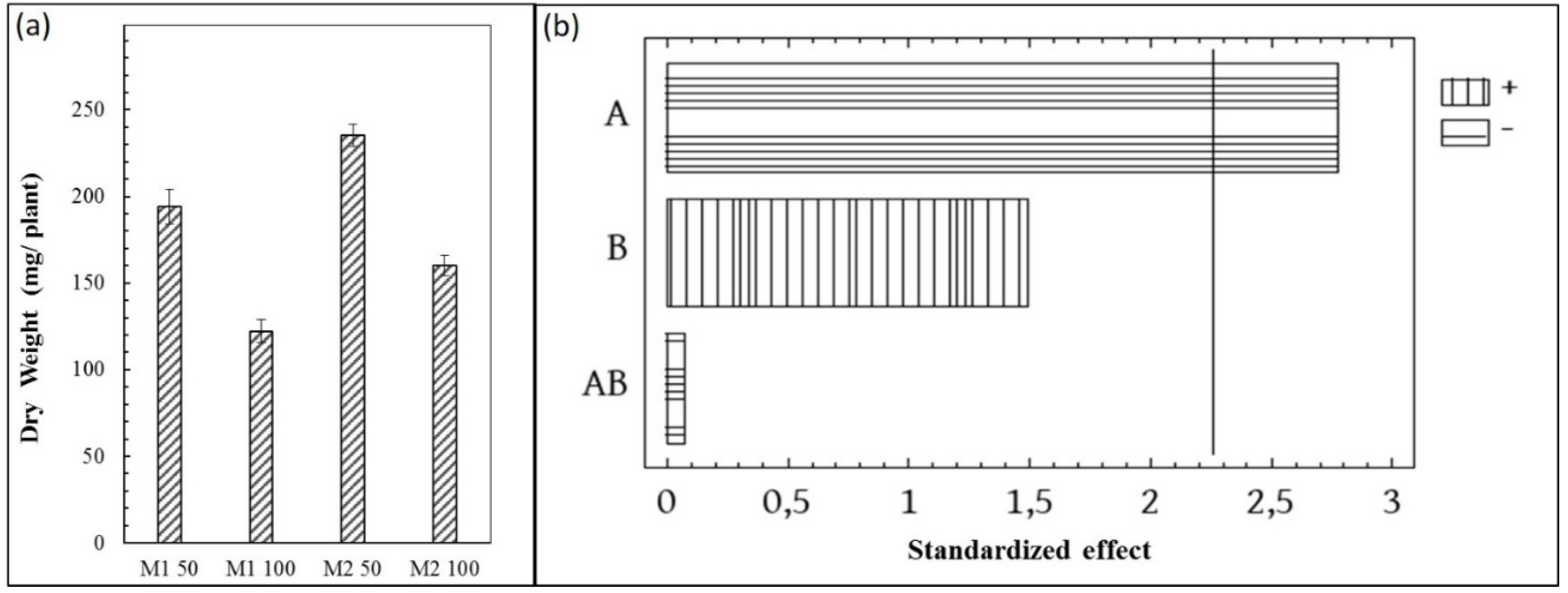
3.3. Evaluation of Fresh Weight (FW) and Dry Weight (DW)
3.4. Evaluation of Antioxidant Capacity
3.5. Solvent Extraction by Soxhlet
3.6. Sample Concentration
3.7. Analysis by Gas Chromatography
3.8. Identification of Volatile Compounds
3.9. Gas Chromatography-Mass Analysis (GC-MS)
3.10. Statistical Analysis of the Results
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gemelli, M. Mercadeo de los Productos Orgánicos en Bolivia: El caso Asopec FAO. Ali&no Editrice: Perugia, Italy, 2003. [Google Scholar]
- Gitli, E.; Arce, R. Consideraciones Sobre El Comercio Internacional de los Productos Orgánicos en Centroamérica. Ideas Sobre Costa Rica; Sistema de Gestión de Información Técnica infoagro.net/Instituto Interamericano de Cooperación para la Agricultura IICA: San Jose, Costa Rica, 2001. [Google Scholar]
- Sagarpa-Se-Bancomext. Official Mark: PC-041-2005 Pliego de condiciones para el uso de la marca oficial méxico calidad suprema en cilantro. Available online: http://s3.esoft.com.mx/esofthands/include/upload_files/4/Archivos/PC_041_2005_Cilantro.pdf (accessed on 20 March 2016).
- Premuzic, Z.; Bargiela, M.; Garcia, A.; Rendina, A.; Iorio, A. Calcium, iron, potassium, phosphorus, and vitamin C content of organic and hydroponic tomatoes. HortScience 1998, 33, 255–257. [Google Scholar]
- Montero, S.M.; Sing, B.K.; Taylor, R. Evaluaci¢n de seis estructuras de producciónn hidroponica diversificada en el trópico húmedo de Costa Rica. Tierra Trop. 2006, 2, 27–37. [Google Scholar]
- Álvarez, R.C.; Chávez, P.C.; Concepción, N.C. Tecnología para la fertilización orgánica en organopónicos. Agricultura Orgánica 2012, 18, 29. [Google Scholar]
- Bracho, J.; Pierre, F.; Quiroz, A. Caracterización de componentes de sustratos locales para la producciónn de plántulas de hortalizas en el estado lara, venezuela. Bioagro 2009, 21, 117–124. [Google Scholar]
- Villanueva-Couoh, E.; Alcántar-González, G.; Sánchez-García, P.; Soria-Fregoso, M.; Larque-Saavedra, A. Nutrición mineral con nitrógeno, fósforo y potasio para la producción de Chrysanthemum morifolium ramat. Con sustratos regionales en Yucatán, México. Terra Latinoam. 2010, 28, 43–52. [Google Scholar]
- Kroetsc, D.W.C. Particle size distribution. In Soil Sampling and Methods of Analysis; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 161–171. [Google Scholar]
- Carrubba, A. Organic and chemical n fertilization on coriander (Coriandrum sativum L.) in a mediterranean environment. Ind. Crops Prod. 2014, 57, 174–187. [Google Scholar] [CrossRef]
- Franco, R.; Schoneveld, O.; Georgakilas, A.G.; Panayiotidis, M.I. Oxidative stress, DNA methylation and carcinogenesis. Cancer Lett. 2008, 266, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Karanjawala, Z.E.; Murphy, N.; Hinton, D.R.; Hsieh, C.-L.; Lieber, M.R. Oxygen metabolism causes chromosome breaks and is associated with the neuronal apoptosis observed in DNA double-strand break repair mutants. Curr. Biol. 2002, 12, 397–402. [Google Scholar] [CrossRef]
- Campanella, L.; Bonanni, A.; Favero, G.; Tomassetti, M. Determination of antioxidant properties of aromatic herbs, olives and fresh fruit using an enzymatic sensor. Anal. Bioanal. Chem. 2003, 375, 1011–1016. [Google Scholar] [PubMed]
- Dapkevicius, A.; Venskutonis, R.; Beek, V.; Linssen, J. Antioxidant activity of extracts obtained by different isolation procedures from some aromatic herbs grown in lithuania. J. Sci. Food Agric. 1998, 77, 140–146. [Google Scholar] [CrossRef]
- Poiana, M.-A.; Alexa, E.; Mateescu, C. Tracking antioxidant properties and color changes in low-sugar bilberry jam as effect of processing, storage and pectin concentration. Chem. Cent. J. 2012, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.R.; Dissanayake, A.A.; Kevseroglu, K.; Nair, M.G. Evaluation of coriander spice as a functional food by using in vitro bioassays. Food Chem. 2015, 167, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Dua, A.; Agrawal, S.; Kaur, A.; Ritu, M. Antioxidant profile of coriandrum sativum merhanolic extract. Int. Res. J. Pharm. 2014, 5, 220–224. [Google Scholar] [CrossRef]
- Yang, H.; Mu, J.; Chen, L.; Feng, J.; Hu, J.; Li, L.; Zhou, J.-M.; Zuo, J. S-nitrosylation positively regulates ascorbate peroxidase activity during plant stress responses. Plant Physiol. 2015, 167, 1604–1615. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.; Palencia, E.; Hinton, D. Abiotic and biotic plant stress-tolerant and beneficial secondary metabolites produced by endophytic bacillus species. In Plant Microbes Symbiosis: Applied Facets; Arora, N.K., Ed.; Springer India: New Delhi, India, 2015; pp. 163–177. [Google Scholar]
- Salvador, G.-P.; Luis Humberto, R.-C.; Tábata, R.-R. Análisis de compuestos volátiles en cilantro (Coriandrum sativum L.). Acta Univ. 2010, 20, 19–24. [Google Scholar]
- Ngang, J.J.; Nyegue, M.A.; Ndoye, F.C.; Tchuenchieu Kamgain, A.D.; Sado Kamdem, S.L.; Lanciotti, R.; Gardini, F.; Etoa, F.X. Characterization of Mexican coriander (Eryngium foetidum) essential oil and its inactivation of listeria monocytogenes in vitro and during mild thermal pasteurization of pineapple juice. J. Food Prot. 2014, 77, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Kohara, K.; Sakamoto, Y.; Hasegawa, H.; Kozuka, H.; Sakamoto, K.; Hayata, Y. Fluctuations in volatile compounds in leaves, stems, and fruits of growing coriander Coriandrum sativum L. Plants. J. Jpn. Soc. Hortic. Sci. 2006, 75, 267–269. [Google Scholar] [CrossRef]
- Rathore, S.S.; Sharma, L.K.; Agarwal, D.; Sing, B.; Saxena, S.N. Assessment of variability in leaf essential oil of three coriander (Coriandrum sativum L.) genotypes. Int. J. Seed Spices 2015, 5, 86–88. [Google Scholar]
- Gladys, B.C. Aproximación al muestreo estadistico en investigaciones cientificas. Rev. Actual. Clín. Investig. 2011, 10, 476–480. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Vasko, L.; Vaskova, J.; Fejercakova, A.; Mojzisova, G.; Poracova, J. Comparison of some antioxidant properties of plant extracts from origanum vulgare, salvia officinalis, eleutherococcus senticosus and stevia rebaudiana. In Vitro Cell. Dev. Biol. Anim. 2014, 50, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.C.; Sharma, N.; Rao, C.V. Comparison of the antioxidant activity and total phenolic, flavonoid content of aerial part of Cleome viscosa L. Int. J. Phytomed. 2011, 3, 386–391. [Google Scholar]
| PT (%) | Dr (g/cm3) | C (dS·m−1) | pH | Moisture (%) | Water Absorbance Capacity (Times Their Weight) | Distribution of Particle Size | |||
|---|---|---|---|---|---|---|---|---|---|
| >2 mm | 2–1 mm | <1 mm | |||||||
| M1 | 50.15 | 0.61 | 0.73 | 6.3 | 7.26 | 1.0 | 28.58 | 17.66 | 53.76 |
| M2 | 71.21 | 0.46 | 0.99 | 5.1 | 11.00 | 1.0 | 40.80 | 15.02 | 45.26 |
| Treatment No. | Substrate Mixture | Fertilizer Concentration (%) | Length of Plants (cm) | Number of Leaves | Stem Diameter (mm) |
|---|---|---|---|---|---|
| 1 | 1 | 50 | 15.07 | 3.30 | 1.62 |
| 2 | 2 | 50 | 14.68 | 3.57 | 1.56 |
| 3 | 1 | 100 | 16.07 | 3.13 | 1.95* |
| 4 | 2 | 100 | 14.00 | 3.33 | 1.43 |
| Treatment | % of DPPH Degradation (Compared to Control) |
|---|---|
| M1 50 | 43.71 |
| M2 50 | 42.31 |
| M1 100 | 48.95 |
| M2 100 | 51.87 |
| Treatments | 2-Octenal (E) | (E)–2 Dodecenal | Decanal |
|---|---|---|---|
| M1 50 | 42.26 c | 11.035 c | 5.085 d |
| M1 100 | 41.125 b | 10.985 b | 4.885 c |
| M2 50 | 38.03 a | 9.6 a | 3.37 a |
| M2 100 | 43.03 d | 11.075 c | 4.395 b |
| No. of Treatment | Coded Factor | Decoded Factor | ||
|---|---|---|---|---|
| Factor A | Factor B | Substrate Mixture | Fertilizer Concentration (%) | |
| 1 | − | − | 1 | 50 |
| 2 | + | − | 2 | 50 |
| 3 | − | + | 1 | 100 |
| 4 | + | + | 2 | 100 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacheco L., N.A.; Cano-Sosa, J.; Poblano C., F.; Rodríguez-Buenfil, I.M.; Ramos-Díaz, A. Different Responses of the Quality Parameters of Coriandrum sativum to Organic Substrate Mixtures and Fertilization. Agronomy 2016, 6, 21. https://doi.org/10.3390/agronomy6020021
Pacheco L. NA, Cano-Sosa J, Poblano C. F, Rodríguez-Buenfil IM, Ramos-Díaz A. Different Responses of the Quality Parameters of Coriandrum sativum to Organic Substrate Mixtures and Fertilization. Agronomy. 2016; 6(2):21. https://doi.org/10.3390/agronomy6020021
Chicago/Turabian StylePacheco L., Neith A., Julia Cano-Sosa, Fernando Poblano C., Ingrid M. Rodríguez-Buenfil, and Ana Ramos-Díaz. 2016. "Different Responses of the Quality Parameters of Coriandrum sativum to Organic Substrate Mixtures and Fertilization" Agronomy 6, no. 2: 21. https://doi.org/10.3390/agronomy6020021
APA StylePacheco L., N. A., Cano-Sosa, J., Poblano C., F., Rodríguez-Buenfil, I. M., & Ramos-Díaz, A. (2016). Different Responses of the Quality Parameters of Coriandrum sativum to Organic Substrate Mixtures and Fertilization. Agronomy, 6(2), 21. https://doi.org/10.3390/agronomy6020021









