Inter-Taxa Differences in Iodine Uptake by Plants: Implications for Food Quality and Contamination
Abstract
:1. Introduction
2. Results and Discussion
2.1. Inter-Species Differences in I Concentration
| “Class” | “Subclass” | “Superorder” | Order | Family | Genus + Species | Relative Mean (I) | Studies |
|---|---|---|---|---|---|---|---|
| MONOCOTS | Lilianae | Commelinids | Commelinales | Commelinaceae | Commelina coelestis | 2.646 | 30 |
| Zingiberales | Cannanaceae | Canna indica | 2.333 | 31 | |||
| Zingiberaceae | Zingiber officinale | 0.016 | 31 | ||||
| Poales | Cyperaceae | Carex nigra | 0.842 | 30 | |||
| Juncaceae | Juncus effusus | 0.393 | 31 | ||||
| Poaceae | Agrostis tenuis | 1.871 | 1, 16 | ||||
| Agrostis alba | 2.021 | 16 | |||||
| Arrenatherum elatius | 1.887 | 16 | |||||
| Cynosuarus cristatus | 2.244 | 16 | |||||
| Dactylis glomerata | 1.804 | 1, 16 | |||||
| Festuca arundinacea | 2.138 | 16 | |||||
| Festuca rubra | 1.733 | 16 | |||||
| Festuca pratensis | 2.021 | 16 | |||||
| Holcus lanatus | 0.512 | 1,19 | |||||
| Hordeum vulgare | 2.802 | 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 | |||||
| Lolium perenne | 2.31 | 1, 16, 20, 21, 22, 23 | |||||
| Lolium multiflorum | 2.299 | 16 | |||||
| Lolium hybridum | 1.702 | 1 | |||||
| Phleum pratense | 2.296 | 16, 20, 21, 22, 23 | |||||
| Poa annua | 0.666 | 1 | |||||
| Poa trivialis | 1.59 | 1, 16 | |||||
| Poa pratense | 2.021 | 16 | |||||
| Triticum aesitvum | 2.852 | 24, 25, 26 | |||||
| Zea mays | 0.108 | 15, 29 | |||||
| Non-Commelenids | Asparagales | Iridaceae | Sisyrinchium striatum | 1.484 | 32 | ||
| Amaryllidaceae | Allium cepa | 0.622 | 27, 30, 36, 37 | ||||
| Allium sativum | 2.321 | 27 | |||||
| Allium porum | 2.565 | 27 | |||||
| EUDICOTS | Ranunculanae | ranunculiids | Ranunculales | Papaveraceae | Papaver commutatum | −0.004 | 33 |
| Rosanae | fabids | Cucurbitales | Cucurbitaceae | Cucumis sativa | −2.131 | 27 | |
| Cucumis melo | 1.872 | 27 | |||||
| Cucurbita maxima | 0.611 | 27 | |||||
| Potentilla anserina | −0.367 | 18 | |||||
| Fabales | Fabaceae | Cicer arietinum | 2.719 | 29 | |||
| Faba vulgaris | −1.117 | 27 | |||||
| Lupinus angustifolius | 0.412 | 29 | |||||
| Medicago sativa | 2.099 | 31 | |||||
| Medicago lupulina | 1.844 | 17 | |||||
| Phaseolus vulgaris | 1.292 | 15, 27 | |||||
| Pisum sativum | 2.505 | 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 27 | |||||
| Trifolium repens | 1.355 | 1, 16, 17, 19, 20, 21, 22, 23 | |||||
| Trifolium subterraneum | 1.611 | 1 | |||||
| Trifolium pratense | 0.923 | 1,17,18,20,21,22,23 | |||||
| Malpighiales | Euphorbiaceae | Euphorbia cyparasissus | −0.148 | 32 | |||
| Euphorbia myrsinites | −0.232 | 31,32 | |||||
| Linaceae | Linum lewisii | 0.328 | 31,33 | ||||
| Linum usitatissimum | 0.47 | 31 | |||||
| Violaceae | Viola wittrockiana | −4.134 | 33 | ||||
| Rosales | Cannabaceae | Humulus japonica | 2.701 | 35 | |||
| Rosaceae | Fragaria vesca | 2.583 | 32 | ||||
| malvids | Brassicales | Brassicaceae | Aubretia x cultorum | 0.184 | 33 | ||
| Brassica napus | 3.258 | 27 | |||||
| Brassica oleracea | 1.072 | 27, 36 | |||||
| Brassica chinensis | 0.857 | 37 | |||||
| Eruca vesicaria | 1.461 | 29 | |||||
| Lepidium sativum | 0.138 | 15 | |||||
| Raphanus sativus | 2.642 | 15, 27, 36 | |||||
| Geraniales | Geraniaceae | Geranium pratense | 1.155 | 31 | |||
| Saxifrgales | Saxifrgaceae | Bergenia cordifolia | −0.497 | 30 | |||
| Malvales | Cistaceae | Helianthemum nummularium | −0.838 | 33 | |||
| Caryophyllanae | caryophylids | Caryophyllales | Amaranthaceae | Amaranthus paniculatus | 1.912 | 34 | |
| Beta rapa | 3.718 | 27 | |||||
| Beta cycla | 1.025 | 27 | |||||
| Beta vulgaris | 2.749 | 15, 29 | |||||
| Celosia cristata | 1.184 | 30 | |||||
| Spinacia oleracea | 1.072 | 15, 27, 33, 37 | |||||
| Caryophyllaceae | Cerastium holosteiodes | 2.443 | 17, 18 | ||||
| Polygonaceae | Dianthus caryophyllus | 1.4 | 30 | ||||
| Rheum tataricum | 0.986 | 30 | |||||
| Rumex acetosa | 1.801 | 17, 18, 19, 27 | |||||
| Rumex obtusifolius | 2.485 | 17, 18 | |||||
| Asteranae | lamiids | Ericales | Polemoniaceae | Gilia tricolor | 1.755 | 31 | |
| Gentianales | Rubiaceae | Cinchona pubescens | −5.533 | 35 | |||
| Galium vernum | −0.299 | 31 | |||||
| Lamiales | Lamiaceae | Glechoma hederacea | 2.488 | 18 | |||
| Mentha spicata | 1.933 | 30 | |||||
| Plectranthus blumei | 2.22 | 34 | |||||
| Salvia Blaze of Fire' | 2.199 | 33 | |||||
| Oleacae | Fraxinus excelsior | −1.835 | 31 | ||||
| Plantaginaceae | Plantago major | 1.763 | 17, 18, 19 | ||||
| Plantago lanceolata | 1.911 | 17, 18, 19 | |||||
| Antirrhinum major | 1.624 | 31 | |||||
| Digitalis purpurea | 1.58 | 31 | |||||
| Veronica spicata | 2.366 | 32 | |||||
| Veronica chamaedrys | 2.488 | 18 | |||||
| Solanales | Solanceae | Lycopersicon esculentum | 3.615 | 27, 32 | |||
| Solanum tuberosum | −6.828 | 27 | |||||
| Solanum melongena | 0.08 | 27, 36 | |||||
| Solanum macrocarpon | 0.587 | 30 | |||||
| Convolulaceae | Ipomea aquatica | 2.385 | 37 | ||||
| campanulids | Apiales | Apiaceae | Apium graveolens | −0.928 | 30, 37 | ||
| Daucus carota | 2.265 | 27, 29, 37 | |||||
| Heracleum sphondylium | −6.828 | 18 | |||||
| Petroselinum crispum | 2.719 | 27 | |||||
| Scandix cerifolium | 1.813 | 27 | |||||
| Asterales | Asteraceae | Aster x frikartii | 1.153 | 30 | |||
| Lactuca sativa | 0.159 | 15, 24, 25, 26, 27, 29, 36 | |||||
| Helianthus annuus | 2.862 | 29 | |||||
| Taraxicum officinales | −6.828 | 17, 18 | |||||
| Cichorum intybus | −6.828 | 27 | |||||
| Cichorum angustifolium | 1.684 | 27 | |||||
| Dipsacales | Dipsacaceae | Dipsacus fullonium | 1.096 | 34 | |||
| Lamium album | 3.076 | 18 |
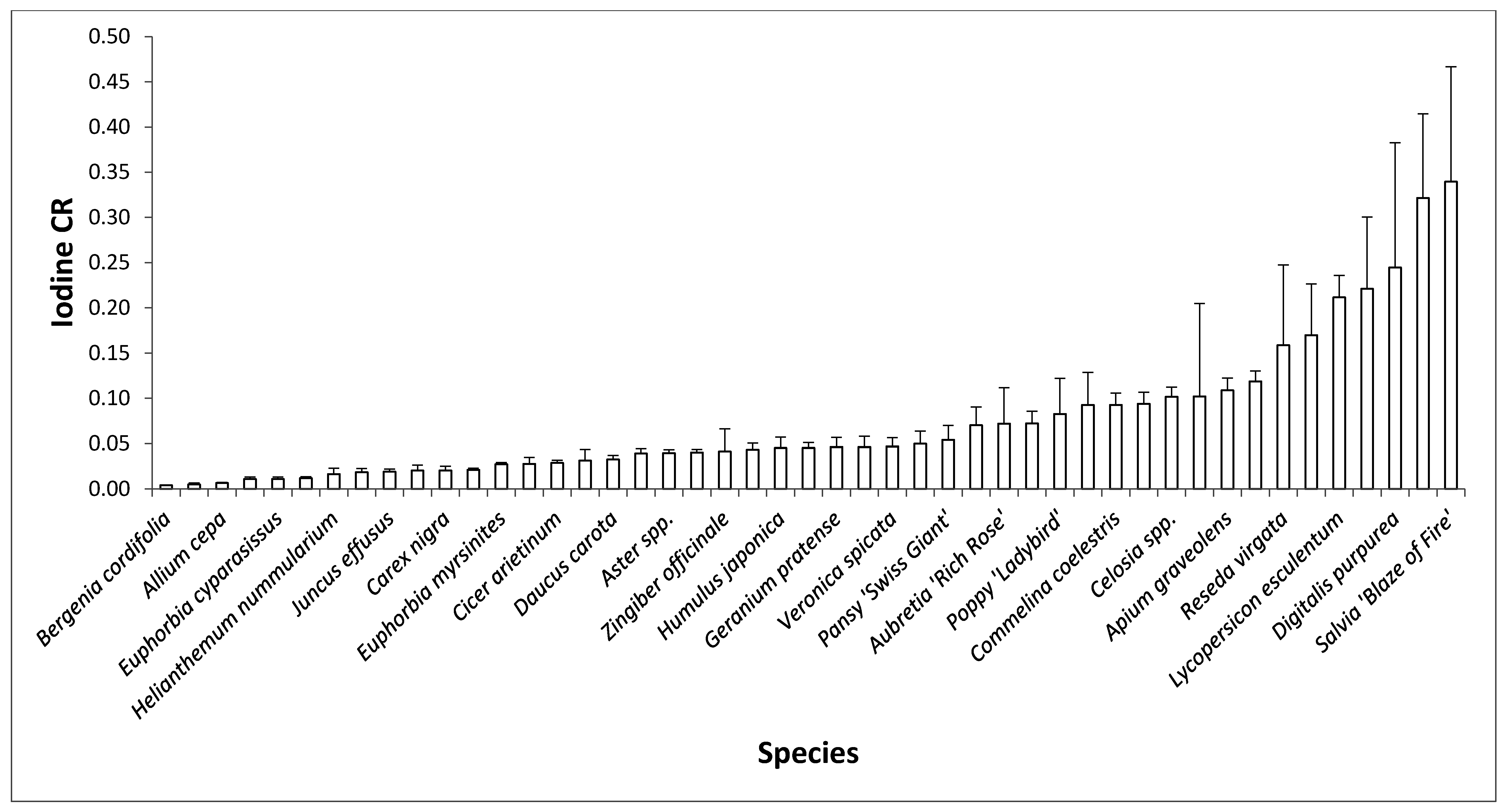
2.2. Taxonomic Influence on Relative Mean I Concentration
| Factor | df | SS | %SS | Cumulative SS | MS | F | p-value |
|---|---|---|---|---|---|---|---|
| “Class” | 2 | 38.28 | 4.80 | 4.02 | 19.14 | 6.26 | 0.053 |
| “Subclass” | 3 | 7.37 | 0.92 | 5.72 | 2.45 | 2.53 | 0.136 |
| “Superorder” | 3 | 1.96 | 0.24 | 5.97 | 0.65 | 0.15 | 0.928 |
| Order | 12 | 48.21 | 6.05 | 12.02 | 4.01 | 1.03 | 0.494 |
| Family | 12 | 50.37 | 6.32 | 18.34 | 4.19 | 0.82 | 0.623 |
| Genus | 45 | 239.06 | 30.0 | 48.34 | 5.31 | 2.78 | 0.004 |
| Species | 24 | 45.79 | 5.74 | 54.09 | 1.90 | 0.35 | 0.895 |
| Residual | 98 | 365.78 | 45.90 | 100 |
| Factor | df | SS | %SS | Cumulative SS | MS | F | p-value |
|---|---|---|---|---|---|---|---|
| “Class” | 2 | 67.16 | 3.32 | 3.32 | 33.58 | 2.85 | 0.211 |
| “Subclass” | 3 | 35.57 | 1.76 | 4.08 | 11.86 | 1.39 | 0.358 |
| “Superorder” | 3 | 24.71 | 1.22 | 6.30 | 8.23 | 0.64 | 0.603 |
| Order | 12 | 157.34 | 7.78 | 14.08 | 13.11 | 1.19 | 0.394 |
| Family | 12 | 129.00 | 6.38 | 20.45 | 10.75 | 1.28 | 0.254 |
| Genus | 45 | 355.43 | 17.57 | 38.02 | 7.89 | 0.78 | 0.774 |
| Species | 24 | 244.65 | 12.09 | 50.12 | 10.19 | 14.62 | 0.204 |
| Residual | 98 | 1009.18 | 49.88 | 100 |
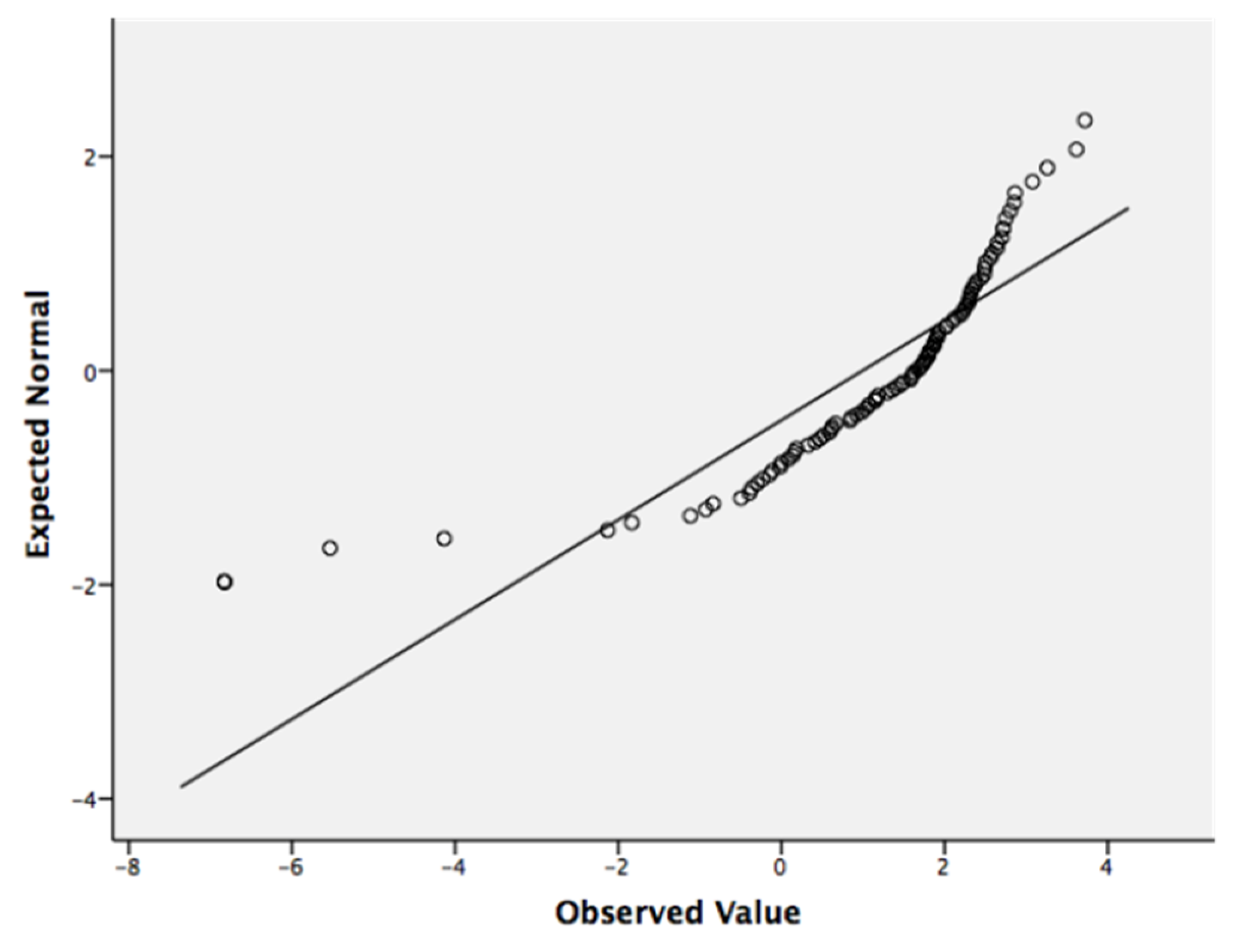
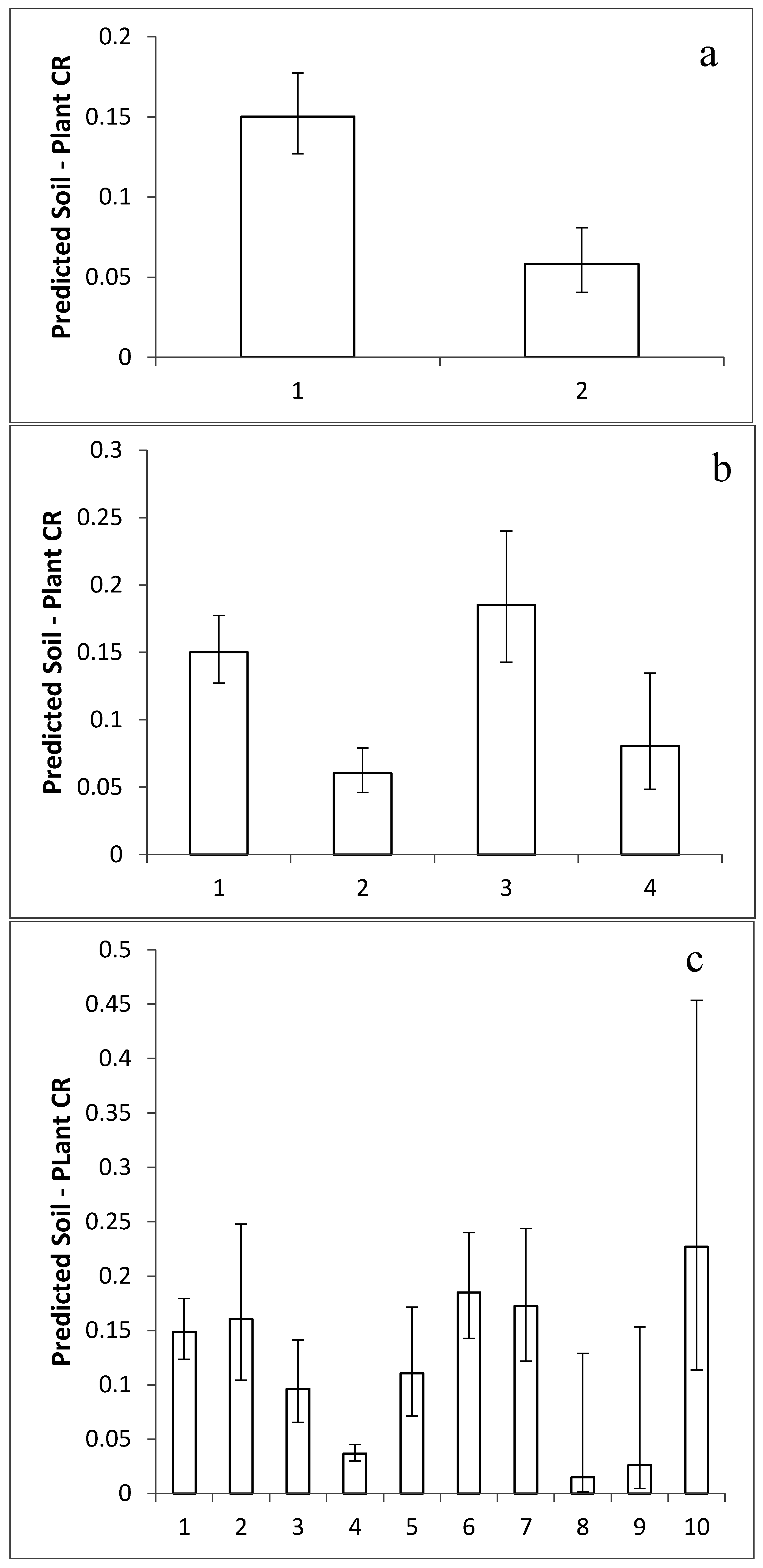
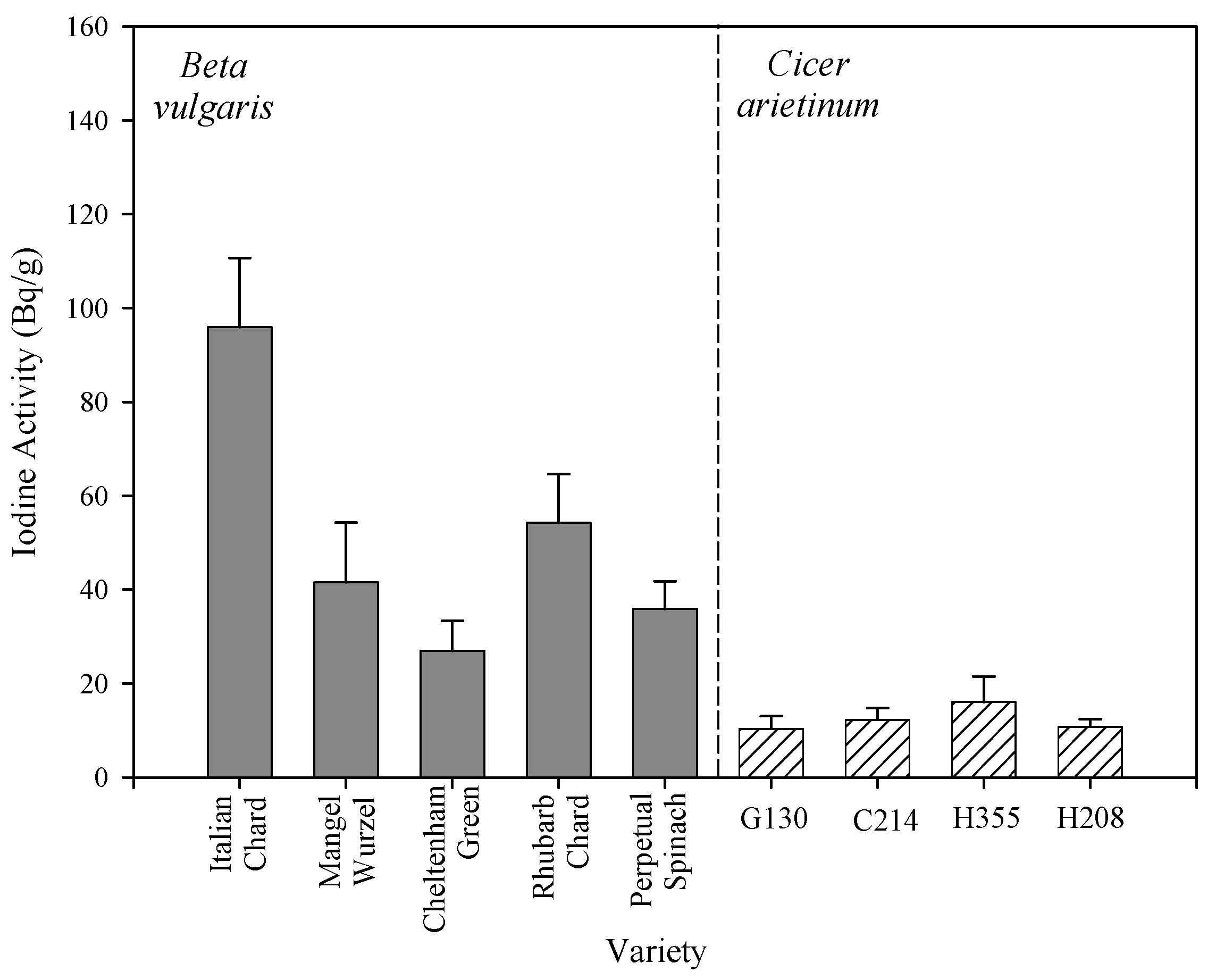
2.3. Comparison of Taxonomic Effects
3. Discussion
4. Experimental Section
4.1. Plant Growth
4.2. Radiolabeling with 125I
4.3. Residual Maximum Likelihood Analysis (REML)
4.4. Analysis of Taxonomic Effects
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhu, Y.-G.; Huang, Y.-Z.; Hu, Y.; Liu, Y.-X. Iodine uptake by spinach (Spinacia oleracea L.) plants grown in solution culture: Effects of iodine species and solution concentrations. Environ. Int. 2003, 29, 33–37. [Google Scholar] [CrossRef]
- Sheppard, S.C. Interpolation of solid/liquid partition coefficients, Kd, for iodine in soils. J. Environ. Radioact. 2003, 70, 21–27. [Google Scholar] [CrossRef]
- Welch, R.M.; Graham, R.D. A new paradigm for world agriculture: Meeting human needs—Productive, sustainable, nutritious. Field Crops Res. 1999, 60, 1–10. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Andersson, M. Prevalence of iodine deficiency in Europe in 2010. Ann. Endocrinol. 2011, 72, 164–166. [Google Scholar] [CrossRef] [PubMed]
- Fréchou, C.; Calmet, D. 129I in the environment of the La Hague nuclear fuel reprocessing plant—From sea to land. J. Environ. Radioact. 2003, 70, 43–59. [Google Scholar] [CrossRef]
- Ashworth, D.J.; Shaw, G.; Butler, A.P.; Cociani, L. Soil transport and plant uptake of radio-iodine from near-surface groundwater. J. Environ. Radiact. 2003, 70, 99–114. [Google Scholar] [CrossRef]
- Sheppard, M.; Thibault, D.H. Default soil solid/liquid partition coefficients, Kds, for four major soil types: A compendium. Health Phys. 1990, 59, 471–482. [Google Scholar] [PubMed]
- Sheppard, S.C.; Motycka, M. Is the Akgare phenomenon important to iodine uptake by wild rice (Zizania aquatica)? J. Environ. Radioact. 1997, 37, 339–353. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Chloride in soils and its uptake and movement within the plant. Ann. Bot. 2001, 88, 967–988. [Google Scholar] [CrossRef]
- Ban-Nai, T.; Muramatsu, Y. Transfer factors of radioiodine from volcanic-ash soil (Andosol) to crops. J. Radiat. Res. 2003, 44, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Broadley, M.R.; Willey, N.J.; Mead, A. A method to assess taxonomic variation in shoot caesium concentration among flowering plants. Environ. Pollut. 1999, 106, 341–349. [Google Scholar] [CrossRef]
- APG (Angiosperm Phylogeny Group) II. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG II. Bot. J. Linnean Soc. 2003, 141, 399–436. [Google Scholar]
- Soltis, P.S.; Soltis, D.E.; Chase, M.W. Angiosperm phylogeny inferred from multiple genes as a research tool for comparative biology. Nature 1999, 402, 402–404. [Google Scholar] [CrossRef] [PubMed]
- Broadley, M.R.; Willey, N.J.; Wilkins, J.; Baker, A.J.M.; Mead, A.; White, P.J. Phylogenetic variation in heavy metal accumulation in angiosperms. New Phytol. 2001, 152, 9–27. [Google Scholar] [CrossRef]
- Broadley, M.R.; Bowen, H.C.; Cotterill, H.L.; Hammond, J.P.; Meacham, M.C.; Mead, A.; White, P.J. Variation in the shoot calcium content of angiosperms. J. Exp. Bot. 2003, 54, 1–16. [Google Scholar] [CrossRef]
- Broadley, M.R.; Bowen, H.C.; Cotterill, H.L.; Hammond, J.P.; Meacham, M.C.; Mead, A.; White, P.J. Phylogenetic variation in the shoot mineral concentration of angiosperms. J. Exp. Bot. 2004, 55, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Willey, N.J.; Tang, S.; Watt, N. Predicting inter-taxa differences in plant uptake of 134/137Cs. J. Environ. Qual. 2005, 34, 1478–1489. [Google Scholar] [CrossRef] [PubMed]
- Willey, N.J.; Fawcett, K. Species selection for phytoremediation of 36Cl/35Cl using angiosperm phylogeny and inter-taxa differences in uptake. Int. J. Phytoremediation 2005, 7, 295–306. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency. Handbook of Parameter Values for the Prediction of Radionuclide Transfer in Terrestrial and Freshwater Environments; Technical Report Series; IAEA: Vienna, Austria, 2010; p. 472. [Google Scholar]
- Butler, G.W.; Johnson, J.M. Factors influencing the iodine content of pasture herbage. Nature 1957, 179, 216–217. [Google Scholar] [CrossRef]
- Moiseyev, I.T.; Tikhomirov, F.A.; Perevezentsev, V.M.; Rerikh, L.A. Role of soil properties, inter-specific plant differences, and other factors affecting the accumulation of radioactive iodine in crops. Sov. Soil Sci. 1984, 16, 60–66. [Google Scholar]
- Cline, J.F.; Klepper, B. Iodine-125 accumulation in plant parts: Influence of water use rate and stable iodine content of soil. Health Phys. 1975, 28, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Hartmans, J. Factors affecting the herbage iodine content. Neth. J. Agric. Sci. 1974, 22, 195–206. [Google Scholar]
- Sheppard, S.C.; Evenden, W.G.; Amiro, B.D. Investigation of the soil-to-plant pathway for I, Br, Cl and F. J. Environ. Radioact. 1993, 21, 9–32. [Google Scholar] [CrossRef]
- Whitehead, D.C. Uptake and distribution of iodine in grass and clover plants grown in solution culture. J. Sci. Food Agric. 1973, 24, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Kashparov, V.; Colle, C.; Zvarich, S.; Yoshenko, V.; Levchuk, S.; Lundin, S. Soil-to-plant halogen transfer studies. J. Environ. Radioact. 2005, 79, 187–204. [Google Scholar] [CrossRef] [PubMed]
- Bourchet, M.P. Sur l’absorption de l’iode par les végétaux. Acad. Sci. Paris 1899, 129, 768–770. [Google Scholar]
- Dai, J.L.; Zhu, Y.G.; Zhang, M.; Huang, Y.Z. Selecting Iodine-Enriched Vegetables and the residual effect of iodate application to soil. Biol. Trace Elem. Res. 2004, 101, 265–276. [Google Scholar] [CrossRef]
- White, P.J.; Bowen, H.C.; Willey, N.J.; Broadley, M.R. Selecting plants to minimise radiocaesium contamination of food chains. Plant Soil 2003, 249, 177–186. [Google Scholar] [CrossRef]
- Payne, K.C.; Bowen, H.C.; Hammond, J.P.; Hampton, C.R.; Lynn, J.R.; Mead, A.; Swarup, K.; Bennett, M.J.; White, P.J.; Broadley, M.R. Natural egentic variation in caesium (Cs) accumulation by Arabidopsis thaliana. New Phytol. 2004, 162, 535–548. [Google Scholar] [CrossRef]
- Johnson, C.C.; Fordyce, F.M.; Stewart, A.G. Environmental Controls in Iodine Deficiency Disorders; Project Summary Report, CR/03/058N; British Geological Survey: Keyworth, UK, 2003. [Google Scholar]
- Crush, J.R.; Caradus, J.R. Cyanogenesis potential and iodine concentration in white clover (Trifolium repens L.) cultivars. N. Z. J. Agric. Res. 1995, 38, 309–316. [Google Scholar] [CrossRef]
- Thompson, R.; Welham, S.J. REML analysis of mixed models. In The Guide to Genstat, Part 2—Statistics; Payne, R.W., Ed.; VSN International: Oxford, UK, 2001; pp. 413–503. [Google Scholar]
- Chase, M.W.; Reveal, J.L. A phylogenetic classification of the land plants to accompany APG III. Bot. J. Linnean Soc. 2009, 161, 122–127. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siasou, E.; Willey, N. Inter-Taxa Differences in Iodine Uptake by Plants: Implications for Food Quality and Contamination. Agronomy 2015, 5, 537-554. https://doi.org/10.3390/agronomy5040537
Siasou E, Willey N. Inter-Taxa Differences in Iodine Uptake by Plants: Implications for Food Quality and Contamination. Agronomy. 2015; 5(4):537-554. https://doi.org/10.3390/agronomy5040537
Chicago/Turabian StyleSiasou, Eleni, and Neil Willey. 2015. "Inter-Taxa Differences in Iodine Uptake by Plants: Implications for Food Quality and Contamination" Agronomy 5, no. 4: 537-554. https://doi.org/10.3390/agronomy5040537
APA StyleSiasou, E., & Willey, N. (2015). Inter-Taxa Differences in Iodine Uptake by Plants: Implications for Food Quality and Contamination. Agronomy, 5(4), 537-554. https://doi.org/10.3390/agronomy5040537





