Trace Element Management in Rice
Abstract
:1. Introduction
| TEs | Health Disorder |
|---|---|
| Se | Keshena (China), bone, arthritis, cardio vascular and Cancer |
| Zn | Dwarfness (Iran & Egypt) infertility, impaired taste and smell |
| Cu | Anemia, Skeletal defects |
| Mn | Tremors, Stiff muscles |
| I | Goiter |
| Fe | Anemia |
| Mg | Depression, Nervous system disorders |
| Mo | Mouth/esophageal cancer, Neurologic damage |
| Co | Anemia |
| Na | Coma |
| Toxic TEs | Toxicity |
| Hg | Mina-Mata, Neurological disorders |
| Cd | Itai-Itai, Bone crippling, Cancer, Heart problems |
| F | Yellowing of teeth, Skeletal deformities, Dental caries |
| Pb | Lung cancer, Neurological disorders |
| As | Skin, Lung cancer |
| Cr | Lung cancer, Kidney dysfunction, Skin ulcer |
2. Nutrient Cycle and Plant Uptake of TEs

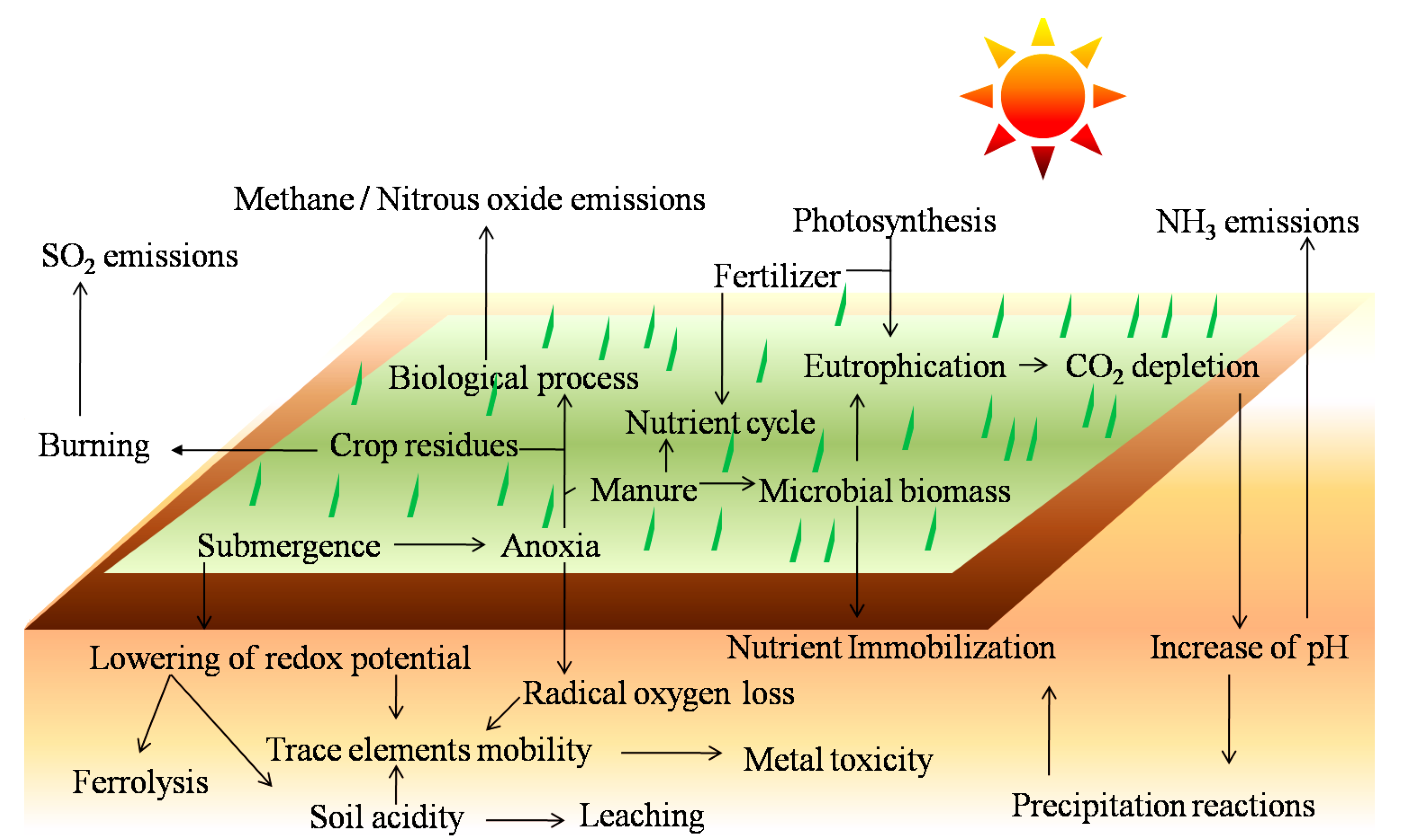
2.1. Regulatory Role of Macronutrients
2.2. Regulatory Role of Micronutrients
| TEs | Occurrence |
|---|---|
| Mn | Mn2+ |
| Fe | Fe2+, Fe(OH)2+, Fe(OH)2+, Fe3+ |
| Zn | Zn2+, Zn(OH)+ |
| Mo | MoO42−, HMoO4− |
| B | H3BO3− |
| Cu | Cu2+,Cu(OH)+ |
| Co | Co2+ |
| Toxic TEs | Occurence |
| Pb | Pb2+ |
| Hg | Hg2+ |
| Cd | Cd2+,Cd1+ |
| As | As(III), As(V) |
| Cr | Cr3+,Cr6+ |
3. Soil Dynamics Act on Mobilization of TEs
3.1. Redox Process
3.2. Soil pH
3.3. Alkalinity
3.4. Cation Exchange Capacity
3.5. Biological Processes
3.6. Fertilization
4. Nutrient Acquisition Directs TE Accumulation
4.1. Root Phenology
| Gene | Function | Refs. |
|---|---|---|
| OsGNOM1 | PIN protein expression and auxin response | [101] |
| OsWOX3A | Inhibition of PIN protein expression | [102] |
| OsHO1 | Latteral root initiation | [103] |
| PSTOL1 | Crown root initiation | [104] |
| ARL1 | Adventitous root initiation | [105] |
| OsABF2/OsTIR1/OsCYP2 | Inhibition of lateral roots emergence | [106,107,108] |
| OsIAA11/OsIAA13 | Lateral root initiation | [109,110] |
| OsLBD3-2 | Crown and latteral root initiation | [106,111] |
| OsIAA23 | Quicent center inhibition | [112] |
| OsSCR1 | Quicent center activation | [113] |
| OsSHR1 | Endoderm diffewrentiation | [114] |
| OsCAND1/OsPIN1 | Crown root emergence | [115] |
| OsRPK1 | Inhibition of root growth | [116] |
| DRO1 | Root growth | [117] |
| OSGLU3/OSDGL1/OsEXPA8 | Cell elogation related root growth | [118,119,120] |
| OsORC3 | Lateral root growth | [121] |
| DES | Root thickness | [122] |
| OsRR1 | Crown root initiation | [123] |
| RAL1,RAL2,RAL3 | Radicle initiation | [124,125] |
4.2. Molecular Physiological Process
4.2.1. Photosynthesis
4.2.2. Transition Metal Transporters
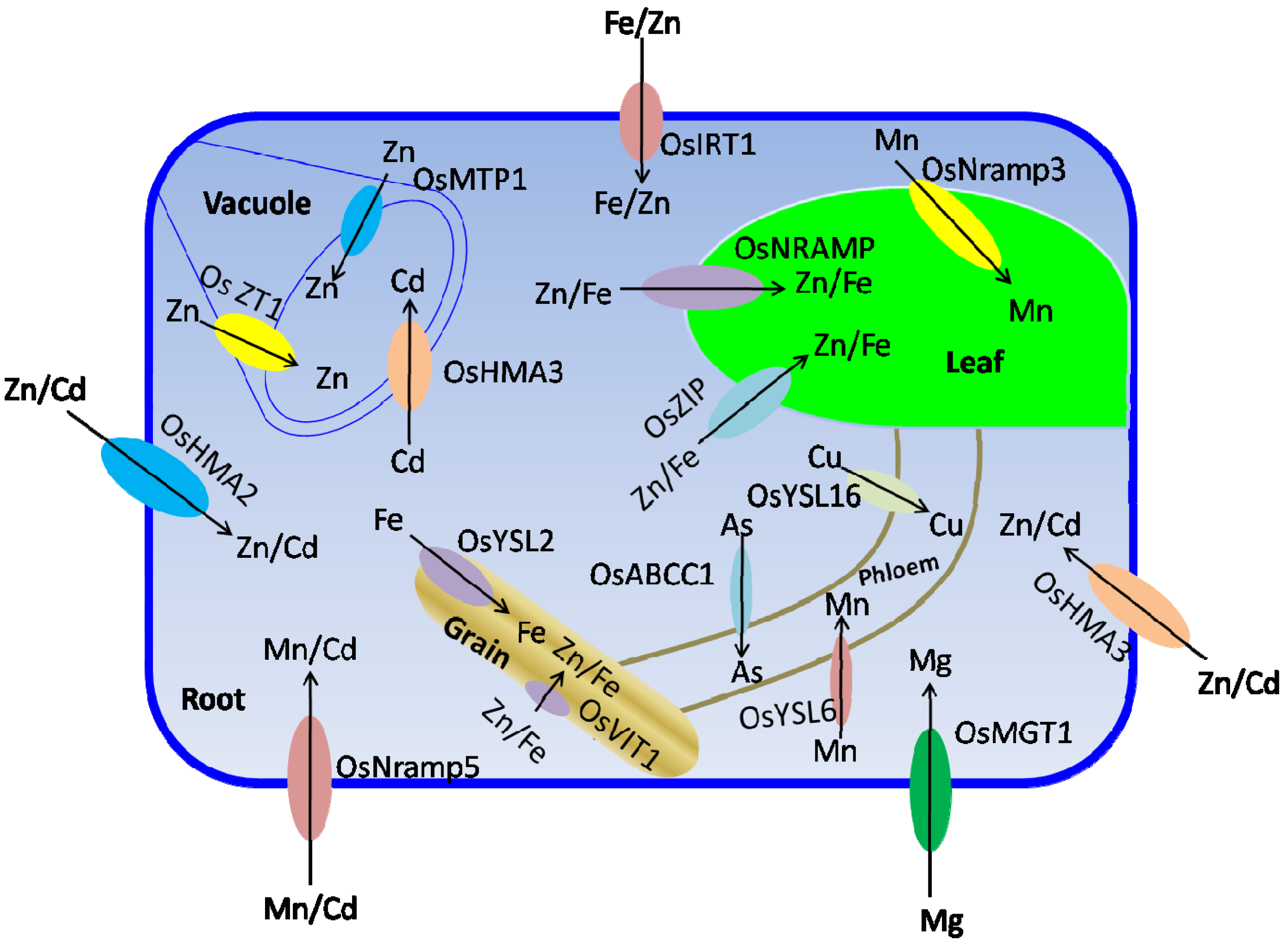
5. Trace Element Fortification

5.1. TEs Supplements
| Nutrient | Mode of Application |
|---|---|
| Zn | Addition of compost, Zincated urea and Gypsum (20 Kg Zn/ha), Periodic draining, Foliar spraying or seed treatment of 0.2 to 0.5% ZnSO4 |
| Fe | Addition of farm yard manure, FeSO4, Fe-EDTA, and Fe-EDDHA (25 Kg Fe/ha). Foliar spraying or seed treatment of 1%–2% FeNH4SO4 (pH5.2). |
| B | Application of Borax andH3BO3 (1Kg/ha), Foliar spray of 0.5% H3BO3 |
| Mn | Application of MnSO4 (2–5 Kg/ha)or Farm yard manure (10 ton/hectare), Foliar Spray of 1% MnSO4 at tillering |
| Mo | Liming of acid soils to a pH 6.2–6.5, Dustingof (NH4)2MoO4 at 100–500 g/ha, Foliar spray of 0.1% (NH4)2MoO4 |
| Cu | Addition of CuSO4 (5 Kg/ha at an interval of 5 years), Root dipping in 1% CuSO4 solution |
| Si | Irrigation with Si rich water, Application of Ca or K silicate (60 Kg/ha), Foliar spray of 0.1% Na2O3Si |
5.2. Plant Breeding
5.3. Genetic Manipulations
6. Outlook
Acknowledgements
Conflicts of Interest
References
- Bashir, K.; Takahash, R.; Nakanishi, H.; Nishizawa, N.K. The road to micronutrient biofortification of rice: Progress and prospects. Front. Plant Sci. 2013, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Lucca, P.; Hurrell, R.; Potrykus, I. Genetic engineering approaches to improve the bioavailability and the level of iron in rice grains. Theor. Appl. Genet. 2001, 102, 392–397. [Google Scholar] [CrossRef]
- Witt, C.; Dobermann, A.; Abdulrachman, S.; Gines, H.C.; Wang, G.; Nagarajan, R.; Satawatananont, S.; Tran, T.S.; Pham, S.T.; Le, V.T.; et al. Internal nutrient efficiencies of irrigated lowland rice in tropical and subtropical Asia. Field Crops Res. 1999, 63, 113–138. [Google Scholar] [CrossRef]
- Andriesse, J.P.; Schelhaas, R.M. A monitoring study of nutrient cycles in soils used for shifting cultivation under various climatic conditions in tropical Asia. II. Nutrient stores in biomass and soil—Results of baseline studies. Agric. Ecosyst. Environ. 1987, 19, 285–310. [Google Scholar] [CrossRef]
- Chase, P.; Singh, O.P. Soil nutrients and fertility in three traditional land use systems of Khonoma, Nagaland, India. Resour. Environ. 2014, 4, 181–189. [Google Scholar]
- Kögel-Knabner, I.; Amelung, W.; Cao, Z.; Fiedler, S.; Frenzel, P.; Jahn, R.; Kalbitz, K.; Kölbl, A.; Schloter, M. Biogeochemistry of paddy soils. Geoderma 2010, 157, 1–14. [Google Scholar] [CrossRef]
- Takahashi, Y.; Minamikawa, R.; Hattori, K.H.; Kurishima, K.; Kihou, N.; Yuita, K. Arsenic behavior in paddy fields during the cycle of flooded and non-flooded periods. Environ. Sci. Technol. 2004, 38, 1038–1044. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Gonzalez, R.; Simünek, J.; Sauvé, S.; Adriano, D. Mechanisms and pathways of trace element mobility in soils. Adv. Agron. 2006, 91, 113–180. [Google Scholar]
- Wissuwa, M.; Ismail, A.M.; Yanagihara, S. Effects of zinc deficiency on rice growth and genetic factors contributing to tolerance. Plant Physiol. 2006, 142, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Fieldler, S.; Vepraskas, M.J.; Richardson, J.L. Soil redox potential importance, field measurements and observations. Adv. Agron. 2007, 94, 41–57. [Google Scholar]
- Sebastian, A.; Prasad, M.N.V. Cadmium minimization in rice: A review. Agron. Sustain. Dev. 2014, 34, 155–173. [Google Scholar] [CrossRef]
- Williams, C.H.; David, D.J. The accumulation in soil of cadmium residues from phosphate fertilizers and their effect on the cadmium content of plants. Soil Sci. 1976, 121, 86–93. [Google Scholar] [CrossRef]
- Bolan, N.S.; Duraisamy, V.P. Role of inorganic and organic soil amendments on immobilization and phytoavailability of heavy metals: A review involving specific case studies. Aust. J. Soil Res. 2003, 41, 533–535. [Google Scholar] [CrossRef]
- Czarnecki, S.; Düring, R.A. Influence of long-term mineral fertilization on metal contents and properties of soil samples taken from different locations in Hesse, Germany. Soil 2015, 1, 23–33. [Google Scholar] [CrossRef]
- Pan, J.; Plant, J.A.; Voulvoulis, N.; Oates, C.J.; Ihlenfeld, C. Cadmium levels in Europe: Implications for human health. Environ. Geochem. Health 2010, 32, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P. Nutrient cycling and nutrient use efficiency. Am. Nat. 1987, 119, 553–572. [Google Scholar] [CrossRef]
- Salt, D.E.; Blaylock, M.; Kumar, N.P.B.A.; Dushenkov, V.; Ensley, D.; Chet, I.; Raskin, I. Phytoremediation: A novel strategy for the removal of toxic metals from the environment using plants. Biotechnology 1995, 13, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Sessitsch, A.; Kuffner, M.; Kidd, P.; Vangronsveld, J.; Wenzel, W.W.; Fallmann, K.; Puschenreiter, M. The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol. Biochem. 2013, 60, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Tsheboeng, G.; Bonyongo, M.; Murray-Hudson, M. Flood variation and soil nutrient content in flood plain vegetation communities in the Okavango Delta. S. Afr. J. Sci. 2014, 110, 1–5. [Google Scholar] [CrossRef]
- Chen, Y.; Stevenson, F.J. Soil organic matter interactions with trace elements. Dev. Plant Soil Sci. 1986, 25, 73–116. [Google Scholar]
- Chaoui, H.I.; Zibilske, L.M.; Ohno, T. Effects of earthworm casts and compost on soil microbial activity and plant nutrient availability. Soil Biol. Biochem. 2003, 35, 295–302. [Google Scholar] [CrossRef]
- Kabarta-Pendias, A. Behavioural properties of trace metals in soils. Appl. Geochem. 1993, 2, 3–9. [Google Scholar] [CrossRef]
- Lu, Y.H.; Watanabe, A.; Kimura, M. Contribution of plant-derived carbon to soil microbial biomass dynamics in a paddy rice microcosm. Biol. Fertil. Soils 2002, 36, 136–142. [Google Scholar] [CrossRef]
- Gao, Y.; Kan, A.T.; Tomson, M.B. Critical evaluation of desorpion phenomena of heavy metals from natural sediments. Environ. Sci. Technol. 2003, 37, 5566–5573. [Google Scholar] [CrossRef] [PubMed]
- Meunier, N.; Drogui, P.; Montané, C.; Hausler, R.; Mercier, G.; Blais, J.F. Comparison between electrocoagulation and chemical precipitation for metals removal from acidic soil leachate. J. Hazard. Mater. 2006, 137, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Rajmohan, N.; Elango, L. Mobility of major ions and nutrients in the unsaturated zone during paddycultivation: A field study and solute transport modelling approach. Hydrol. Process. 2007, 21, 2698–2712. [Google Scholar] [CrossRef]
- Blair, N.; Faulkner, R.D.; Till, A.R.; Poulton, P.R. Long-term management impacts on soil C, N and physical fertility. Part I. Broadbalk experiment. Soil Tillage Res. 2006, 91, 30–38. [Google Scholar] [CrossRef]
- Kothawala, D.N.; Moore, T.R. Adsorption of dissolved nitrogen by forest mineral soils. Can. J. For. Res. 2009, 39, 2381–2390. [Google Scholar] [CrossRef]
- Kanda, K.; Tsuruta, H.; Minami, K. Emission of dimethyl sulfide, carbonyl sulfide and carbon disulfide from paddy fields. Soil Sci. Plant Nutr. 1992, 38, 709–716. [Google Scholar] [CrossRef]
- Whiticar, M.J. Carbon and hydrogen isotope systematics of bacterial formation and oxidation of methane. Chem. Geol. 1999, 161, 291–314. [Google Scholar] [CrossRef]
- Kuo, S. Application of modified Langmuir isotherm to phosphate sorption by some acid soils. Soil Sci. Soc. Am. J. 1988, 52, 97–102. [Google Scholar] [CrossRef]
- Brennan, R.F.; Bolland, M.D.A.; Bowden, J.W. Potassium deficiency, and molybdenum deficiency and aluminium toxicity due to soil acidification have become problems for cropping sandy soils in south-western Australia. Aust. J. Exp. Agric. 2004, 44, 1031–1039. [Google Scholar] [CrossRef]
- Inal, A.; Gunes, A.; Sahin, O.; Taskin, M.B.; Kaya, E.C. Impacts of biochar and processed poultry manure, applied to a calcareous soil, on the growth of bean and maize. Soil Use Manag. 2015, 31, 106–113. [Google Scholar] [CrossRef]
- Green, C.H.; Heil, D.M.; Cardon, G.E.; Butters, G.L.; Kelly, E.F. Solubilization of manganese and trace metals in soils affected by acid mine runoff. J. Environ. Qual. 2003, 32, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Greipsson, S. Effect of iron plaque on roots of rice on growth of plants in excess zinc and accumulation of phosphorus in plants in excess copper or nickel. J. Plant Nutr. 1995, 18, 1659–1665. [Google Scholar] [CrossRef]
- Adriano, D.C.; Wenzel, W.W.; Vangronsveld, J.; Bolan, N.S. Role of assisted natural remediation in environmental cleanup. Geoderma 2004, 122, 121–142. [Google Scholar] [CrossRef]
- Coleman, D.C.; Reid, C.P.P.; Cole, C.V. Biological strategies of nutrient cycling in soil systems. Adv. Ecol. Res. 1983, 13, 1–55. [Google Scholar]
- Rauthan, B.S.; Schnitzer, M. Effects of a soil fulvic acid on the growth and nutrient content of cucumber (cucumis sativus) plants. Plant Soil 1981, 63, 491–495. [Google Scholar] [CrossRef]
- Chiang, K.; Wang, Y.; Wang, M.; Chiang, P. Low-molecular-weight organic acids and metal speciation in rhizosphere and bulk soils of a temperate rain forest in Chitou, Taiwan. Taiwan J. For. Sci. 2006, 21, 327–337. [Google Scholar]
- Vinolas, L.C.; Healey, J.R.; Jones, D.L. Kinetics of soil microbial uptake of free amino acids. Biol. Fertil. Soils 2001, 33, 67–74. [Google Scholar] [CrossRef]
- Sivapullaiah, P.V.; Prakash, B.S.N.; Suma, S. Electrokinetic removal of heavy metals from soil. J. Electrochem. Sci. Eng. 2015, 5, 47–65. [Google Scholar] [CrossRef]
- Chapin, F.S.; Bloom, A.J.; Field, C.B.; Waring, R.H. Plant responses to multiple environmental factors. J. Biosci. 1987, 37, 49–57. [Google Scholar] [CrossRef]
- Ehrenfeld, J.G. Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 2003, 6, 503–523. [Google Scholar] [CrossRef]
- Hattenschwiler, S.; Vitousek, P. The role of polyphenols in terrestrial ecosystem nutrient cycling. Trends Ecol. Evol. 2000, 15, 238–243. [Google Scholar] [CrossRef]
- Katoh, M.; Murase, J.; Hayashi, M.; Matsuya, K.; Kimura, M. Nutrient leaching from the plow layer by water percolation and accumulation in the subsoil in an irrigated paddy field. Soil Sci. Plant Nutr. 2004, 50, 721–729. [Google Scholar] [CrossRef]
- Zhou, W.; Lv, T.; Chen, Y.; Westby, A.P.; Ren, W. Soil physicochemical and biological properties of paddy-upland rotation: A review. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.M.; Tsadilas, C.D.; Rinklebe, J. Immobilization of soil copper using organic and inorganic amendments. Plant Nutr. Soil Sci. 2015, 178, 112–117. [Google Scholar] [CrossRef]
- Sharma, A.; Weindorf, D.C.; Wang, D.; Chakraborty, S. Characterizing soils via portable X-ray fluorescence spectrometer: 4. Cation exchange capacity (CEC). Geoderma 2015, 239–240, 130–134. [Google Scholar] [CrossRef]
- Charlet, L.; Markelova, E.; Parsons, C.; Couture, R.; Madé, B. Redox oscillation impact on natural and engineered biogeochemical systems: Chemical resilience and implications for contaminant mobility. Proced. Earth Planet. Sci. 2013, 7, 135–138. [Google Scholar] [CrossRef]
- Hoffman, M.R.; Yost, E.C.; Eisenrich, S.J.; Maier, W.J. Characterization of soluble and colloidal phase metal complexes in river water by ultra-filtration: A mass balance approach. Environ. Sci. Technol. 1981, 15, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.; Wu, H.B.; Xi, Y.; Lo, X.W. Mixed transition-metal oxides: Design, synthesis, and energy-related applications. Angew. Chem. 2014, 53, 1488–1504. [Google Scholar] [CrossRef] [PubMed]
- Scharpenseel, H.W.; Pfeiffer, E.M.; Becker-Heidmann, P. Organic carbon storage in tropical hydromorphic soils. In Structure Andorganic Matter Storage in Agricultural Soils; Carter, M.R., Stewart, B.A., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1996; pp. 361–392. [Google Scholar]
- Diaz, R.J. Overview of hypoxia around world. J. Environ. Qual. 2001, 30, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, S.; Richards, B.K.; Steenhuis, T.S.; McBride, M.B.; Baveye, P.; Dousset, S. Microbial acidification and pH effects on trace element release from sewage sludge. Environ. Pollut. 2004, 132, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Olaniran, A.O.; Balgobind, A.; Pillay, B. Bioavailability of heavy metals in soil: Impact on microbial biodegradation of organic compounds and possible improvement strategies. Int. J. Mol. Sci. 2013, 14, 10197–10228. [Google Scholar] [CrossRef] [PubMed]
- Brinkman, R. Ferrolysis: Chemical and Mineralogical Aspects of Soil Formation in Seasonally Wet Acid Soils, and Some Practical Implications; International Rice Research Institute: Los Banos, Philippines, 1978; pp. 295–303. [Google Scholar]
- Van Ranst, E.; Dumon, M.; Tolossa, A.R.; Cornelis, J.T.; Stoops, G.; Vandenberghe, R.E.; Deckers, J. Revisiting ferrolysis processes in the formation of Planosols for rationalizing the soils with stagnic properties in WRB. Geoderma 2011, 163, 265–274. [Google Scholar] [CrossRef]
- Fageria, N.K.; Carvalho, G.D.; Santos, A.B.; Ferreira, E.P.B.; Knupp, A.M. Chemistry of lowland rice soils and nutrient availability. Commun. Soil Sci. Plant Anal. 2011, 42, 1913–1933. [Google Scholar] [CrossRef]
- Sahrawat, K.L.; Sika, M. Comparative tolerance of Oryza sativaand Oryza glaberrimarice cultivars for iron toxicity in West Africa. Int. Rice Res. Notes 2002, 27, 30–31. [Google Scholar]
- Barak, P.; Jobe, B.O.; Krueger, A.R.; Peterson, L.A.; Laird, D.A. Effects of long term soil acidification due to nitrogen inputs in Wisconsin. Plant Soil 1997, 197, 61–69. [Google Scholar] [CrossRef]
- Behera, S.K.; Shukla, A.K. Spatial distribution of surface soil acidity, electrical conductivity, soil organic carbon content and exchangeable potassium, calcium and magnesium in some cropped acid soils of India. Land Degrad. Dev. 2015, 26, 71–79. [Google Scholar] [CrossRef]
- Magdoff, F.R.; Bartlett, R.J. Soil pH buffering revisited. Soil Sci. Soc. Am. J. 1985, 49, 145–148. [Google Scholar] [CrossRef]
- Pan, Y.; Koopmans, G.F.; Bonten, L.T.; Song, J.; Luo, Y.; Temminghoff, E.J.; Comans, R.N. Influence of pH on the redox chemistry of metal (hydr) oxides and organic matter in paddy soils. J. Soils Sediments 2014, 14, 1713–1726. [Google Scholar] [CrossRef]
- Evangelou, V.P.; Philipps, R.E. Cation exchange in soils. In Chemical Processes in Soil; Tabatabai, M.A., Sparks, D.L., Eds.; Soil Science Society of America: Madison, WI, USA, 2005; pp. 343–410. [Google Scholar]
- Deumlich, D.; Thiere, J.; Altermann, M. Characterization of cation exchange capacity (CEC) for agricultural land-use areas Arch. Agron. Soil Sci. 2015, 6, 767–784. [Google Scholar] [CrossRef]
- Anda, M.; Suryani, E.; Husnain; Subardja, D. Strategy to reduce fertilizer application in volcanic paddy soils: Nutrient reserves approach from parent materials. Soil Till. Res. 2015, 150, 10–20. [Google Scholar] [CrossRef]
- Sebastian, A.; Prasad, M.N.V. Vertisol prevents cadmium accumulation in rice: Analysis by ecophysiological toxicity markers. Chemosphere 2014, 108, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Sun, G.; Zhu, Y.G. Effects of microbial processes on the fate of arsenic in paddy soil. Chin. Sci. Bull. 2013, 58, 186–193. [Google Scholar] [CrossRef]
- Nayak, A.K.; Raja, R.; Rao, K.S.; Shukla, A.K.; Mohanty, S.; Shahid, M.; Tripathi, R.; Panda, B.B.; Bhattacharyya, P.; Kumar, A.; et al. Effect of fly ash application on soil microbial response and heavy metal accumulation in soil and rice plant. Ecotoxicol. Environ. Saf. 2014. [Google Scholar] [CrossRef] [PubMed]
- Lomax, C.; Liu, W.J.; Wu, L.; Xue, K.; Xiong, J.; Zhou, J.; McGrath, S.P.; Meharg, A.A.; Miller, A.J.; Zhao, F.J. Methylated arsenic species in plants originate from soil microorganisms. New Phytol. 2012, 193, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, M.; Ae, N.; Prasad, M.N.V.; Freitas, H. Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnol. 2010, 28, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Loaces, I.; Ferrando, L.; Scavino, A.F. Dynamics, diversity and function of endophytic siderophore-producing bacteria in rice. Microb. Ecol. 2010, 61, 606–618. [Google Scholar] [CrossRef] [PubMed]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Noori, M.S.S.; Saud, H.M. Potential plant growth-promoting activity of Pseudomonassp. isolated from paddy soil in Malaysia as biocontrol agent. J. Plant Pathol. Microb. 2012, 3, 120. [Google Scholar] [CrossRef]
- Deshwal, V.K.; Kumar, P. Plant growth promoting activity of Pseudomonads in Rice crop. Int. J. Curr. Microbiol. App. Sci. 2013, 2, 152–157. [Google Scholar]
- Doi, R.; Pitiwut, S. From maximization to optimization: A paradigm shift in rice production in thailand to improve overall quality of life of stakeholder. Sci. World J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Bodelier, P.L.E.; Sorrell, B.; Drake, H.L.; Küsel, K.; Hurek, T.; Reinhold-Hurek, B.; Lovell, C.; Megonigal, P.; Frenzel, P. Ecological aspects of microbes and microbial communities inhabiting the rhizosphere of wetland plants. In Wetlands as a Natural Resource; Bobbink, R., Beltman, B., Verhoeven, J.T.A., Whigham, D.F., Eds.; Springer-Verlag: New York, NY, USA, 2006; pp. 205–238. [Google Scholar]
- Redeker, K.; Wang, N.; Low, J.; McMillan, A.; Tyler, S.; Cicerone, R. Emissions of methyl halides and methane from rice paddies. Science 2000, 290, 966–969. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 2012, 63, 131–152. [Google Scholar] [CrossRef] [PubMed]
- Kochian, L.V.; Hoekenga, A.O.; Pineros, A.M. How do crop plants tolerate acid soils? Mechanism of Aluminium tolerance and Phosphorous efficiency. Annu. Rev. Plant Biol. 2004, 55, 459–493. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.-J.; Su, J.-Q.; Xu, H.-J.; Jia, Z.-J.; Zhu, Y.-G. Long-term nitrogen fertilization of paddy soil shifts iron-reducing microbial community revealed by RNA-13C-acetate probing coupled with pyrosequencing. ISME J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.F.; Bian, R.J.; Pan, G.X.; Cui, L.Q.; Hussain, Q.; Li, L.Q.; Zheng, J.W.; Zheng, J.F.; Zhang, X.H.; Han, X.J.; et al. Effect of biochar amendment on soil quality, crop yield and greenhouse gas emission in a Chinese rice paddy: A field study of 2 consecutive rice growing cycles. Field Crop Res. 2012, 127, 153–160. [Google Scholar] [CrossRef]
- Yang, C.; Yang, L.; Yang, Y.; Ouyang, Z. Rice root growth and nutrient uptake as influenced by organic manure in continuously and alternately flooded paddy soils. Agric. Water Manag. 2004, 70, 67–81. [Google Scholar] [CrossRef]
- Dobermann, A.; Witt, C.; Dawe, D. Performance of site-specific nutrient management in intensive rice cropping systems of Asia. Better Crops Int. 2002, 16, 25–30. [Google Scholar]
- Eriksson, J.E. Effects of nitrogen-containing fertilizers on solubility and plant uptake of cadmium. Water Air Soil Pollut. 1990, 49, 355–368. [Google Scholar] [CrossRef]
- Das, S.K. Role of micronutrient in rice cultivation and management strategy in organic agriculture—A reappraisal. Agric. Sci. 2014, 5, 765–769. [Google Scholar] [CrossRef]
- Wong, M.C.; Ma, K.K.; Fang, K.M.; Cheung, C. Utilization of a manure compost for organic farming in Hong Kong. Biores. Technol. 1999, 67, 43–46. [Google Scholar] [CrossRef]
- Tang, J.C.; Inoue, Y.; Yasuta, T.; Yoshida, S.; Katayama, A. Chemical and microbial properties of various compost products. Soil Sci. Plant Nutr. 2003, 49, 273–280. [Google Scholar] [CrossRef]
- Sebastian, A.; Prasad, M.N.V. Cadmium accumulation retard activity of functional components of photo assimilation and growth of rice cultivars amended with vermicompost. Int. J. Phytoremediation 2013, 15, 965–978. [Google Scholar] [CrossRef] [PubMed]
- Shibaharaa, F.; Inubushiab, K. Effects of organic matter application on microbial biomass and available nutrients in various types of paddy soils. Soil Sci. Plant Nutr. 1997, 43, 191–203. [Google Scholar] [CrossRef]
- Kumpiene, J.; Lagerkvist, A.; Maurice, C. Stabilization of As, Cr, Cu, Pb and Zn in soil using amendments—A review. Waste Manag. 2008, 28, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Horikawa, Y. Stability behavior of soil colloidal suspensions in relation to sequential reduction of soils. II Turbidity changes by submergence of paddy soils at different temperatures. Soil Sci. Plant Nutr. 1997, 43, 911–919. [Google Scholar] [CrossRef]
- Baligar, V.C.; Fageria, N.K.; He, Z. Nutrient use efficiency in plants. Commun. Soil Sci. Plant Anal. 2001, 31, 921–950. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C. Improving nutrient use efficiency of annual crops in Brazilian acid soils for sustainable crop production. Commun. Soil Sci. Plant Anal. 2001, 32, 1303–1319. [Google Scholar] [CrossRef]
- Raun, W.R.; Johnson, G.V. Improving nitrogen use efficiency for cereal production. Agron. J. 1999, 91, 357–363. [Google Scholar] [CrossRef]
- Yoo, Y.; Choi, H.; Jung, K. Genome-wide identification and analysis of genes associated with lysigenous aerenchyma formation in rice roots. J. Plant Biol. 2015, 58, 117–127. [Google Scholar] [CrossRef]
- Mai, C.D.; Phung, N.T.P.; To, H.T.M.; Gonin, M.; Hoang, G.T.; Nguyen, K.L.; Do, V.N.; Courtois, B.; Gantet, P. Genes controlling root development in rice. Rice 2014, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Cheng, S. Root genetic research, an opportunity and challenge to rice improvement. Field Crops Res. 2014, 165, 111–124. [Google Scholar] [CrossRef]
- Gowdaa, V.R.P.; Henrya, A.; Yamauchi, A.; Shashidhar, H.E.; Serraj, R. Root biology and genetic improvement for drought avoidance in rice. Field Crops Res. 2011, 122, 1–13. [Google Scholar] [CrossRef]
- Thanh, N.D.; Zheng, H.G.; Dong, N.V.; Trinh, L.N.; Ali, M.L.; Nguyen, H.T. Genetic variation in root morphology and microsatellite DNA loci in upland rice (Oryza sativaL.) from Vietnam. Euphytica 1999, 105, 43–55. [Google Scholar] [CrossRef]
- Liu, S.; Wang, J.; Wang, L.; Wang, X.; Xue, Y.; Wu, P.; Shou, H. Adventitious root formation in rice requires OsGNOM1 and is mediated by the OsPINs family. Cell Res. 2009, 19, 1110–1119. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.C.; Cho, S.H.; Paek, N.C. Rice WUSCHEL-related homeobox 3A (OsWOX3A) modulates auxin-transport gene expression in lateral root and root hair development. Plant Signal Behav. 2013, 8, e25929. [Google Scholar] [CrossRef]
- Hsu, Y.Y.; Chao, Y.-Y.; Kao, C.H. Biliverdin-promoted lateral root formation is mediated through heme oxygenase in rice. Plant Signal Behav. 2012, 7, 885–887. [Google Scholar] [CrossRef] [PubMed]
- Gamuyao, R.; Chin, J.H.; Pariasca-Tanaka, J.; Pesaresi, P.; Catausan, S.; Dalid, C.; Slamet-Loedin, I.; Tecson-Mendoza, E.M.; Wissuwa, M.; Heuer, S. The protein kinase Pstol1 from traditional rice confers tolerance of phosphorus deficiency. Nature 2012, 488, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, S.; Yu, X.; Yu, J.; He, X.; Zhang, S.; Shou, H.; Wu, P. ARL1, a LOB-domain protein required for adventitious root formation in rice. Plant J. 2005, 43, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Cho, J.I.; Han, M.; Ahn, C.H.; Jeon, J.S.; An, G.; Park, P.B. The ABRE-binding transcription factor OsABF2 is a positive regulator of abiotic stress and ABA signaling in rice. J Plant Physiol. 2010, 167, 1512–1520. [Google Scholar] [CrossRef] [PubMed]
- Xia, K.; Wang, R.; Ou, X.; Fang, Z.; Tian, C.; Duan, J.; Wang, Y.; Zhang, M. OsTIR1and OsAFB2downregulation via OsmiR393 overexpression leads to more tillers, early flowering and less tolerance to salt and drought in rice. PLoS ONE 2012, 7, e30039. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.; Zhang, Z.; Wang, L.; Zheng, L.; Mao, W.; Li, M.; Wu, Y.; Wu, P.; Mo, X. OsCYP2, a chaperone involved in degradation of auxin-responsive proteins, plays crucial roles in rice lateral root initiation. Plant J. 2013, 74, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.X.; Liu, Y.; Liu, S.J.; Mao, C.Z.; Wu, Y.R.; Wu, P. A gain-of-function mutation in OsIAA11 affects lateral root development in rice. Mol. Plant 2012, 5, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Kitomi, Y.; Inahashi, H.; Takehisa, H.; Sato, Y.; Inukai, Y. OsIAA13-mediated auxin signaling is involved in lateral root initiation in rice. Plant Sci. 2012, 190, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, S.; Guan, X.; Chen, L.; He, Y.; Wang, J.; Lu, G. Genome-wide identification and transcriptional profiling analysis of auxin response-related gene families in cucumber. BMC Res. Notes 2014, 7, 218. [Google Scholar] [CrossRef] [PubMed]
- Jun, N.; Gaohang, W.; Zhenxing, Z.; Huanhuan, Z.; Yunrong, W.; Ping, W. OsIAA23-mediated auxin signaling defines postembryonic maintenance of QC in rice. Plant J. 2011, 68, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Shen, Y.; Zhang, Y.; Wu, P. Definition and stabilization of the quiescent center in rice roots. Plant Biol. 2014, 16, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Levesque, M.P.; Vernoux, T.; Jung, J.W.; Paquette, A.J.; Gallagher, K.L.; Wang, J.Y.; Blilou, I.; Scheres, B.; Benfey, P.N. An evolutionarily conserved mechanism delimiting SHR movement defines a single layer of endodermis in plants. Science 2007, 316, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.F.; He, F.F.; Ma, X.X.; Mao, C.Z.; Hodgman, C.; Lu, C.G.; Wu, P. OsCAND1 is required for crown root emergence in rice. Mol. Plant 2010, 4, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Zhu, L.; Shou, H.; Wu, P. A PIN1 family gene, OsPIN1, involved in auxin-dependent adventitious root emergence and tillering in rice. Plant Cell Physiol. 2005, 46, 1674–1681. [Google Scholar] [CrossRef] [PubMed]
- Uga, Y.; Sugimoto, K.; Ogawa, S.; Rane, J.; Ishitani, M.; Hara, N.; Kitomi, Y.; Inukai, Y.; Ono, K.; Uga, Y.; et al. Dro1, a major QTL involved in deep rooting of rice under upland field conditions. J. Exp. Bot. 2011, 62, 2485–2494. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.W.; Xu, L.; Wu, Y.R.; Chen, X.A.; Liu, Y.; Zhu, S.H.; Ding, W.N.; Wu, P.; Yi, K.K. OsGLU3, a putative membrane-bound endo-1,4-beta-glucanase, is required for root cell elongation and division in rice (Oryza sativa L.). Mol. Plant 2012, 5, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Li, Y.; Gan, J.; Wang, W.; Zhang, H.; Liu, Y.; Wu, P. OsDGL1, a homolog of an oligosaccharyltransferase complex subunit, is involved in N-glycosylation and root development in rice. Plant Cell Physiol. 2013, 54, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.; Wang, Y.; Qiu, S.; Kang, Z.; Che, S.; Wang, G.; Huang, J. Overexpression of OsEXPA8, a root-specific gene, improves rice growth and root system architecture by facilitating cell extension. PLoS ONE 2013, 8, e75997. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shi, J.; Hao, X.; Liu, H.; Wu, Y.; Wu, Z.; Chen, M.; Wu, P.; Mao, C. OsORC3 is required for lateral root development in rice. Plant J. 2013, 74, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Nakazaki, T.; Ikehashi, H. Analyses of genetic loci for diameter of seminal root with marker genes in rice. Breed. Sci. 1996, 46, 75–77. [Google Scholar]
- Kitomi, Y.; Ito, H.; Hobo, T.; Aya, K.; Kitano, H.; Inukai, Y. The auxin responsive AP2/ERF transcription factorCROWN ROOTLESS5is involved in crown root initiation in rice through the induction ofOsRR1, a type-A response regulator of cytokinin signaling. Plant J. 2011, 67, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Scarpella, E.; Rueb, E.; Meijer, A.H. TheRADICLELESS1gene is required for vascular pattern formation in rice. Development 2003, 130, 645–665. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.K.; Aoki, T.; Kitano, H.; Satoh, H.; Nagato, Y. Phenotypic diversity of 188 rice embryo mutants. Dev. Genet. 1995, 16, 298–310. [Google Scholar] [CrossRef]
- Siebers, N.; Godlinski, F.; Leinweber, P. Bone char as phosphorus fertilizer involved in cadmium immobilization in lettuce, wheat, and potato cropping. J. Plant Nutr. Soil Sci. 2014, 177, 75–83. [Google Scholar] [CrossRef]
- Clark, R.B. Physiology of cereals for mineral nutrient uptake, use and efficiency. In Crops Enhancers of Nutrient Use; Baligar, V.C., Duncan, R.R., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 131–209. [Google Scholar]
- Gahoonia, T.S.; Nielsen, N.E. Root traits as tools for creating phosphorus efficient crop varieties. Plant Cell Environ. 2004, 260, 47–57. [Google Scholar]
- Gourley, C.J.P.; Allan, D.L.; Russlle, M.P. Plant nutrient effi-ciency: A comparison of definitions and suggested improvement. Plant Soil 1994, 158, 29–37. [Google Scholar] [CrossRef]
- Tanner, W.; Beevers, H. Transpiration, a prerequisite for long-distance transport of minerals in plants? Proc. Natl. Acad. Sci. USA 2001, 98, 9443–9447. [Google Scholar] [CrossRef] [PubMed]
- Palmgren, M.G. Plant plasma membrane H+-ATPases: Powerhouses for nutrient uptake. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 817–845. [Google Scholar] [CrossRef] [PubMed]
- Sondergaard, T.E.; Schulz, A.; Palmgren, M.G. Energization of transport processes in plants. Roles of the plasma membrane H+-ATPase. Plant Physiol. 2004, 136, 2475–2482. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Siguad-Kutner, T.; Leitão, M.; Okamoto, O.K.; Morse, D.; Colepicolo, P. Heavy metal-induced oxidative stress in algae. J. Phycol. 2003, 39, 1008–1018. [Google Scholar] [CrossRef]
- Rauser, W.E. Structure and function of metal chelators produced by plants: The case for organic acids, amino acids, phytin and metallothioneins. Cell Biochem. Biophys. 1999, 31, 19–48. [Google Scholar] [CrossRef] [PubMed]
- Azura, A.E.; Shamshuddin, J.; Fauziah, C.I. Root elongation, root surface area and organic acid by rice seedling under Al3+ and/or H+ Stress. Am. J. Agric. Biol. Sci. 2011, 6, 324–331. [Google Scholar] [CrossRef]
- Wertin, T.M.; Teskey, R.O. Close coupling of whole-plant respiration to net photosynthesis and carbohydrates. Tree Physiol. 2008, 28, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Liu, P.; Zheng, G.; Zhang, J.; Xu, G. Effects of potassium on organic acid metabolism of Fe-sensitive and Fe-resistant rices (Oryza sativa L.). Aust. J. Crop Sci. 2013, 7, 843–848. [Google Scholar]
- Dong, J.; Mao, W.H.; Zhang, G.P.; Wu, F.B.; Cai, Y. Root excretion and plant tolerance to cadmium toxicity—A review. Plant Soil Environ. 2007, 53, 193–200. [Google Scholar]
- Zhu, X.F.; Zheng, C.; Hu, Y.T.; Jiang, T.; Liu, Y.; Dong, N.Y.; Yang, J.L.; Zheng, S.J. Cadmium-induced oxalate secretion from root apex is associated with cadmium exclusion and resistance in Lycopersicon esulentum. Plant Cell Environ. 2011, 34, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Negishi, T.; Nakanishi, H.; Yazaki, J.; Kishimoto, N.; Fujii, F.; Shimbo, K.; Yamamoto, K.; Sakata, K.; Sasaki, T.; Kikuchi, S.; et al. cDNA microarray analysis of gene expression during Fe-deficiency stress in barley suggests that polar transport of vesicles is implicated in phytosiderophore secretion in Fe-deficient barley roots. Plant J. 2002, 30, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Raskin, I.; Kumar, P.B.A.N.; Dushenkov, S.; Salt, D.E. Bioconcentration of heavy metals by plants. Curr. Opin. Biotechnol. 1994, 5, 285–290. [Google Scholar] [CrossRef]
- Krämer, U. Metal hyperaccumulation in plants. Ann. Rev. Plant Biol. 2010, 61, 517–534. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, A.; Prasad, M.N.V. Red and blue lights induced oxidative stress tolerance promote cadmium rhizocomplexation in Oryza sativa. J. Photochem. Photobiol. B 2014, 137, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Stephan, U.W.; Schmidke, I.; Stephan, V.W.; Scholz, G. The nicotianamine molecule is made-to-measure for complexation of metal micronutrients in plants. Biometals 1996, 9, 84–90. [Google Scholar] [CrossRef]
- Von Wirén, N.; Klair, S.; Bansal, S.; Briat, J.F.; Khodr, H.; Shioiri, T.; Leigh, R.A.; Hider, R.C. Nicotianamine chelates both FeIIIand FeII. Implications for metal transport in plants. Plant Physiol. 1999, 119, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Errécalde, O.; Seidl, M.; Campbell, P.G.C. Influence of a low molecular weight metabolite (citrate) on the toxicity of cadmium and zinc to the unicellular green alga Selenastrum capricornutum: An exception to the free ion activity model. Water Res. 1998, 32, 419–429. [Google Scholar] [CrossRef]
- Kramer, U.; Talke, I.N.; Hanikenne, M. Transition metal transport. FEBS Lett. 2007, 581, 2263–2272. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Yamaji, N.; Yokosho, K.; Ma, J.F. Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell 2012, 24, 2155–2167. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, N.; Sasaki, A.; Xia, J.X.; Yokosho, K.; Ma, J.F. Anode-based switch for preferential distribution of manganese in rice. Nat. Commun. 2013, 4, 2442. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.C.; Yamaji, N.; Motoyama, R.; Nagamura, Y.; Ma, J.F. Up-regulation of a magnesium transporter gene OsMGT1 is required for conferring aluminum tolerance in rice. Plant Physiol. 2012, 159, 1624–1633. [Google Scholar] [CrossRef] [PubMed]
- Ranade-malvi, U. Interaction of micronutrients with major nutrients with special reference to potassium. KarnatakaJ. Agric. Sci. 2011, 24, 106–109. [Google Scholar]
- Yamaji, N.; Xia, J.X.; Mitani-Ueno, N.; Yokosho, K.; Ma, J.F. Preferential delivery of Zn to developing tissues in rice is mediated by a P-type ATPases, OsHMA2. Plant Physiol. 2013, 162, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; An, G. Over-expression of OsIRT1 leads to increased iron and zinc accumulations in rice. Plant Cell Environ. 2009, 32, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Yamaji, N.; Ma, J.F. Overexpression of OsHMA3 enhances Cd tolerance and expression of Zn transporter genes in rice. J. Exp. Bot. 2014, 65, 6013–6021. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Yamaji, N.; Yokosho, K.; Ma, J.F. YSL16 is a phloem-localized transporter of the copper-nicotianamine complex that is responsible for copper distribution in rice. Plant Cell 2012, 37, 3767–3782. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Yamaki, T.; Yamaji, N.; Ko, D.; Jung, K.; Fujii-Kashino, M.; An, G.; Martinoia, E.; Lee, Y.; Ma, J.F. A rice ABC transporter, OsABCC1, reduces arsenic accumulation in the grain. Proc. Natl. Acad. Sci. USA 2014, 111, 15699–15704. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, Y.; Masuda, H.; Bashir, K.; Inoue, H.; Tsukamoto, T.; Takahashi, M.; Nakanishi, H.; Aoki, N.; Hirose, T.; Ohsugi, R.; et al. Rice metal-nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J. 2010, 62, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Yamaji, N.; Xia, J.; Ma, J.F. OsYSL6 is involved in the detoxification of excess manganese in rice. Plant Physiol. 2011, 157, 1832–1840. [Google Scholar] [CrossRef] [PubMed]
- Beyer, P. Golden Rice and “Golden” crops for human nutrition. New Biotechnol. 2010, 27, 478–481. [Google Scholar] [CrossRef] [PubMed]
- Ravindrababu, V. Importance and advantages of rice biofortification with iron and zinc. J. SAT Agric. Res. 2013, 11, 1–6. [Google Scholar]
- Piccoli, N.B.; Grede, N.; de Pee, S.; Singhkumarwong, A.; Roks, E.; Moench-Pfanner, R.; Bloem, M.W. Rice fortification: Its potential for improving micronutrient intake and steps required for implementation at scale. Food Nutr.Bull. 2012, 33, S360–S372. [Google Scholar] [PubMed]
- Clemens, S. Molecular mechanisms of plant metal tolerance and homeostasis. Planta 2001, 212, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.L.; Williams, L.E. Transition metal transporters in plants. J. Exp. Bot. 2003, 54, 2601–2613. [Google Scholar] [CrossRef] [PubMed]
- Sahrawat, K.L. Soil fertility in flooded and non-flooded irrigated rice systems. Arch. Agron. Soil Sci. 2012, 58, 423–436. [Google Scholar] [CrossRef]
- Shahid, M.; Nayak, A.K.; Shukla, A.K.; Tripathi, R.; Kumar, A.; Raja, R.; Panda, B.B.; Meher, J.; Bhattacharyya, P.; Dash, D. Mitigation of iron toxicity and iron, zinc, and manganese nutrition of wetland rice cultivars (Oryza sativa L.) grown in iron-toxic soil. Soil Air Water 2014, 42, 1604–1609. [Google Scholar] [CrossRef]
- Chen, W.; Yao, X.; Cai, K.; Chen, J. Silicon alleviates drought stress of rice plants by improving plant water status, photosynthesis and mineral nutrient absorption. Biol. Trace Elem. Res. 2011, 142, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Gregorio, G.B. Progress in breeding for traceminerals in staple crops. J. Nutr. 2002, 132, 500–502. [Google Scholar]
- Patne, N.; Ravindrababu, V.; Usharani, G.; Reddy, T.D. Grain iron and zinc association studies in rice (Oryza sativa L.) F1 progenies. Arch. Appl. Sci. Res. 2012, 4, 696–702. [Google Scholar]
- Ishikawa, S.; Abe, T.; Kuramata, M.; Yamaguchi, M.O.T.; Yamamoto, T.; Yano, M. Major quantitative trait locus for increasing cadmium specific concentration in rice grain is located on the short arm of chromosome 7. J. Exp. Bot. 2010, 61, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Ueno, D.; Koyama, E.; Kono, I.O.T.; Yano, M.; Ma, J.F. Identification of a novel major quantitative trait locus controlling distribution of Cd between roots and shoots in rice. Plant CellPhysiol. 2009, 50, 2223–2233. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.; Chen, M.; Zhang, G. Mapping of QTLs associated with cadmium tolerance and accumulation during seedling stage in rice (Oryza sativaL.). Euphytica 2009, 165, 587–596. [Google Scholar] [CrossRef]
- Goto, F.; Yoshihara, T.; Shigemoto, N.; Toki, S.; Takaiwa, F. Iron fortification of rice seed by the soybean ferritin gene. Nat. Biotechnol. 1999, 17, 282–286. [Google Scholar] [PubMed]
- Qu, L.Q.; Yoshihara, T.; Ooyama, A.; Goto, F.; Takaiwa, F. Iron accumulation does not parallel the high expression level of ferritin in transgenic rice seeds. Planta 2005, 222, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Ali, N.; Gayen, D.; Datta, S.K.; Datta, K. Molecular breeding of Osfer2 gene to increase iron nutrition in rice grain. GM Crops Food 2012, 3, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Masuda, H.; Kobayashi, T.; Ishimaru, Y.; Takahashi, M.; Aung, M.S.; Nakanishi, H.; Mori, S.; Nishizawa, N.K. Iron-biofortification in rice by the introduction of three barley genes participated in mugineic acid biosynthesis with soybean ferritin gene. Front. Plant Sci. 2013. [Google Scholar] [CrossRef] [PubMed]
- Wirth, J.; Poletti, S.; Aeschlimann, B.; Yakandawala, N.; Drosse, B.; Osorio, S.; Tohge, T.; Fernie, A.R.; Günther, D.; Gruissem, W.; et al. Rice endosperm iron biofortification by targeted and synergistic action of nicotianamine synthase and ferritin. Plant Biotechnol. J. 2009, 7, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Higuchi, K.; Takahashi, M.; Nakanishi, H.; Mori, S.; Nishizawa, N.K. Three rice nicotianamine synthase genes, OsNAS1, OsNAS2and OsNAS3 are expressed in cells involved in long-distance transport of iron and differentially regulated by iron. Plant J. 2003, 36, 366–381. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jeon, U.S.; Lee, S.J.; Kim, Y.K.; Persson, D.P.; Husted, S.; Schjørring, J.K.; Kakei, Y.; Masuda, H.; Nishizawa, N.K.; et al. Iron fortification of rice seeds through activation of the nicotianamine synthase gene. Proc. Natl. Acad. Sci. USA 2009, 106, 22014–22019. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.T.; Kyriacou, B.; Callahan, D.L.; Carruthers, L.; Stangoulis, J.; Lombi, E.; Tester, M. Constitutive overexpression of the OsNAS gene family reveals single-gene strategies for effective iron- and zinc-biofortification of rice endosperm. PLoS ONE 2011, 6, e24476. [Google Scholar] [CrossRef] [PubMed]
- Ogo, Y.; Itai, R.N.; Kobayashi, T.; Aung, M.S.; Nakanishi, H.; Nishizawa, N.K. OsIRO2 is responsible for iron utilization in rice and improves growth and yield in calcareous soil. Plant Mol. Biol. 2011, 75, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Venkata, T.V.G.N.; Kotla, A.; Mangrauthia, S.K.; Neelamraju, S. Expression patterns of QTL based and other candidate genes in Madhukar × Swarna RILs with contrasting levels of iron and zinc in unpolished rice grains. Gene 2014, 546, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Chandel, G.P.; Samuel, M.; Dubey, R.; Meena, R.K. In silico expression analysis of QTL specific candidate genes for grain micronutrient (Fe/Zn) content using ESTs and MPSS signature analysis in rice (Oryza sativa L.). Plant Genet. Transgenics 2011, 2, 11–22. [Google Scholar]
- Lee, S.; Chiecko, J.C.; Kim, S.A.; Walker, E.L.; Lee, Y.; Guerinot, M.L.; An, G. Disruption of OsYSL15 leads to iron inefficiency in rice plants. Plant Physiol. 2009, 150, 786–800. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, Y.; Masuda, H.; Suzuki, M.; Bashir, K.; Takahashi, M.; Nakanishi, H.; Mori, S.; Nishizawa, N.K. Overexpression of the OsZIP4 zinc transporter confers disarrangement of zinc distribution in rice plants. J. Exp. Bot. 2007, 58, 2909–2915. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jeong, H.; Kim, S.; Lee, J.; Guerinot, M.; An, G. OsZIP5 is a plasma membrane zinc transporter in rice. Plant Mol. Biol. 2010, 73, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Huang, J.; Ye, Q.; Wu, D.; Chen, Z. Modified accumulation of selected heavy metals in Bt transgenic rice. J. Environ. Sci. 2009, 21, 1607–1612. [Google Scholar] [CrossRef]
- Zhan, J.; Wei, S.; Niu, R.; Li, Y.; Wang, S.; Zhu, J. Identification of rice cultivar with exclusive characteristic to Cd using a field-polluted soil and its foreground application. Environ. Sci. Pollut. Res. Int. 2013, 20, 2645–2650. [Google Scholar] [CrossRef] [PubMed]
- Antosiewicz, D.M.; Henning, J. Overexpression of LTC1in tobacco enhances the protective action of calcium against cadmium toxicity. Environ. Pollut. 2004, 129, 237–245. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sebastian, A.; Prasad, M.N.V. Trace Element Management in Rice. Agronomy 2015, 5, 374-404. https://doi.org/10.3390/agronomy5030374
Sebastian A, Prasad MNV. Trace Element Management in Rice. Agronomy. 2015; 5(3):374-404. https://doi.org/10.3390/agronomy5030374
Chicago/Turabian StyleSebastian, Abin, and Majeti Narasimha Vara Prasad. 2015. "Trace Element Management in Rice" Agronomy 5, no. 3: 374-404. https://doi.org/10.3390/agronomy5030374
APA StyleSebastian, A., & Prasad, M. N. V. (2015). Trace Element Management in Rice. Agronomy, 5(3), 374-404. https://doi.org/10.3390/agronomy5030374





