The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes
Abstract
:1. Introduction
2. Review of Literature
2.1. An Overview of Redox Processes in Soils with Biochar
- (1)
- Electron transfer from organic matter to Fe(III) (hydr) oxides via C oxidation [14].
- (2)
- Reduction of NO3− to NO2− with the oxidation of Fe2+ to Fe3+.
- (3)
- Mineralisation of organic N to NH4+ [15] and the oxidation of NH4+ to NO2− with the consequent reduction of Fe 3+ to Fe2+.
- (4)
- Oxidation of NH4+ to NO2− with the consequent reduction of Fe3+, formation and oxidation of FeS minerals in the sulphur (S) cycle [7].
- (5)
- Cycling of S from solid to soluble liquid species driven by oxidation or reduction of Fe species [7].
2.2. The Electrochemical Properties of Biochars, Summary of Literature
2.3. Measurement of the Electrochemical Properties of Fresh Biochars and Soil/Biochar Systems
- (i)
- High variability of soil Eh in space and time: Eh is largely influenced by hydric conditions (water activity), temperature, microbial activity and respiration of living organisms [27,28]. As a consequence, it is difficult to obtain stable measurements, especially in soils with low poising ability (that is, soils with low organic matter and clay content).
- (ii)
- Irreversibility of redox reactions at the surface of the electrodes, which makes it difficult to conduct Eh measurements over long time periods [29].
- (iii)
- Chemical disequilibrium in soils [30].
- (iv)
- Polarisation of and/or leakage from electrodes.
- (v)
- The influence of electromagnetic fields on water and living organisms, which can greatly perturb Eh measurements in soil samples through an induced current in the electrode [31].
- (vi)
2.4. Electrochemical Properties of Biochar as Measured Using Solid State Cyclic Voltammetry (SSCV), SEM and TEM
- (1)
- A wood biochar (Jarrah) produced at 600 °C in a vertical retort [34]. This biochar had a high surface area, high fixed C, high concentration of stable aromatic C and low concentration of functional groups and low ash.
- (2)
- Acacia saligna biochar produced at 380 °C in a rotating drum kiln. This biochar has high labile C content and a relative low surface area compared with the Jarrah biochar [35].
- (3)
- Chicken litter high mineral ash biochar produced at 400 °C in a rotating drum kiln [4].
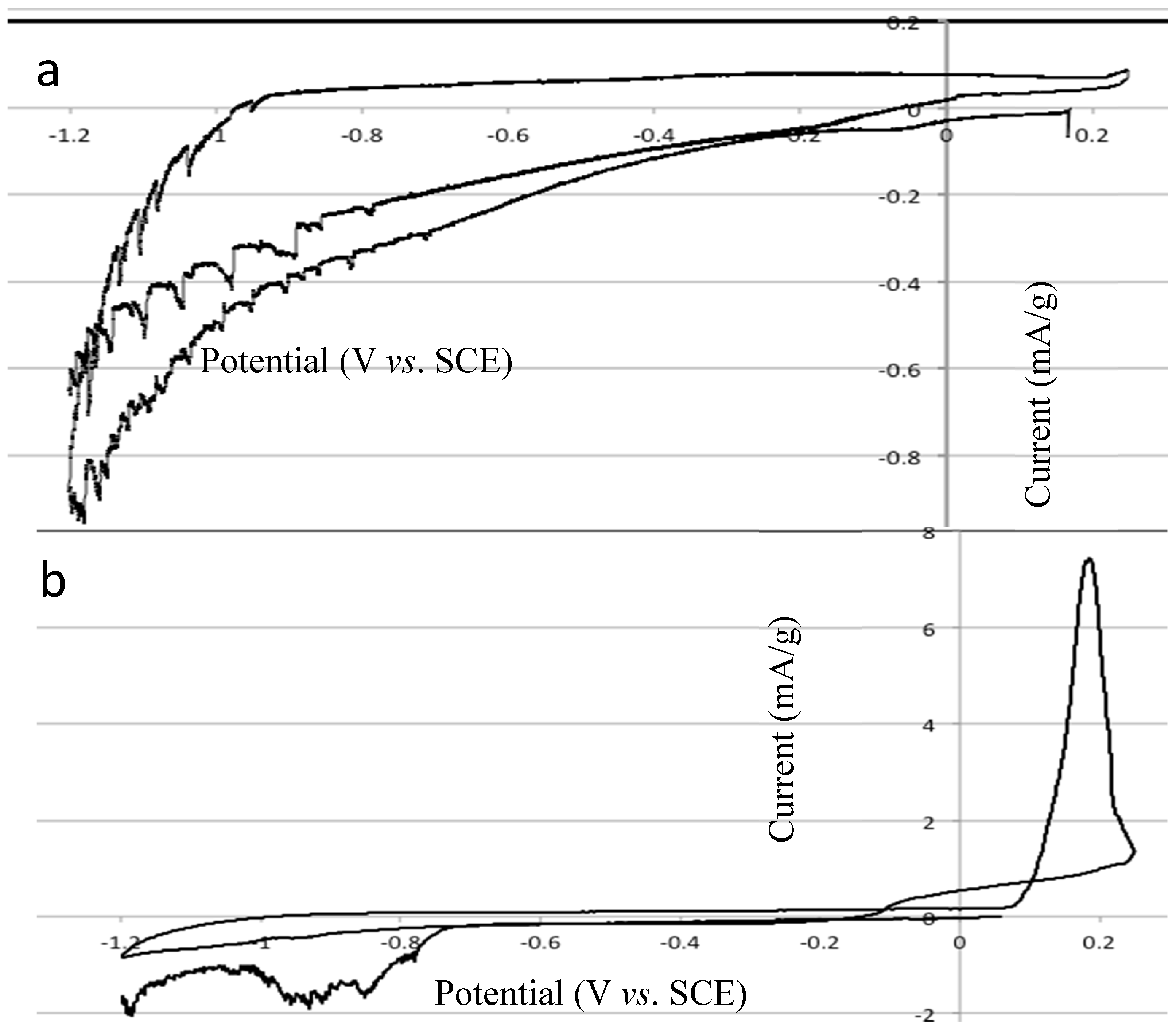

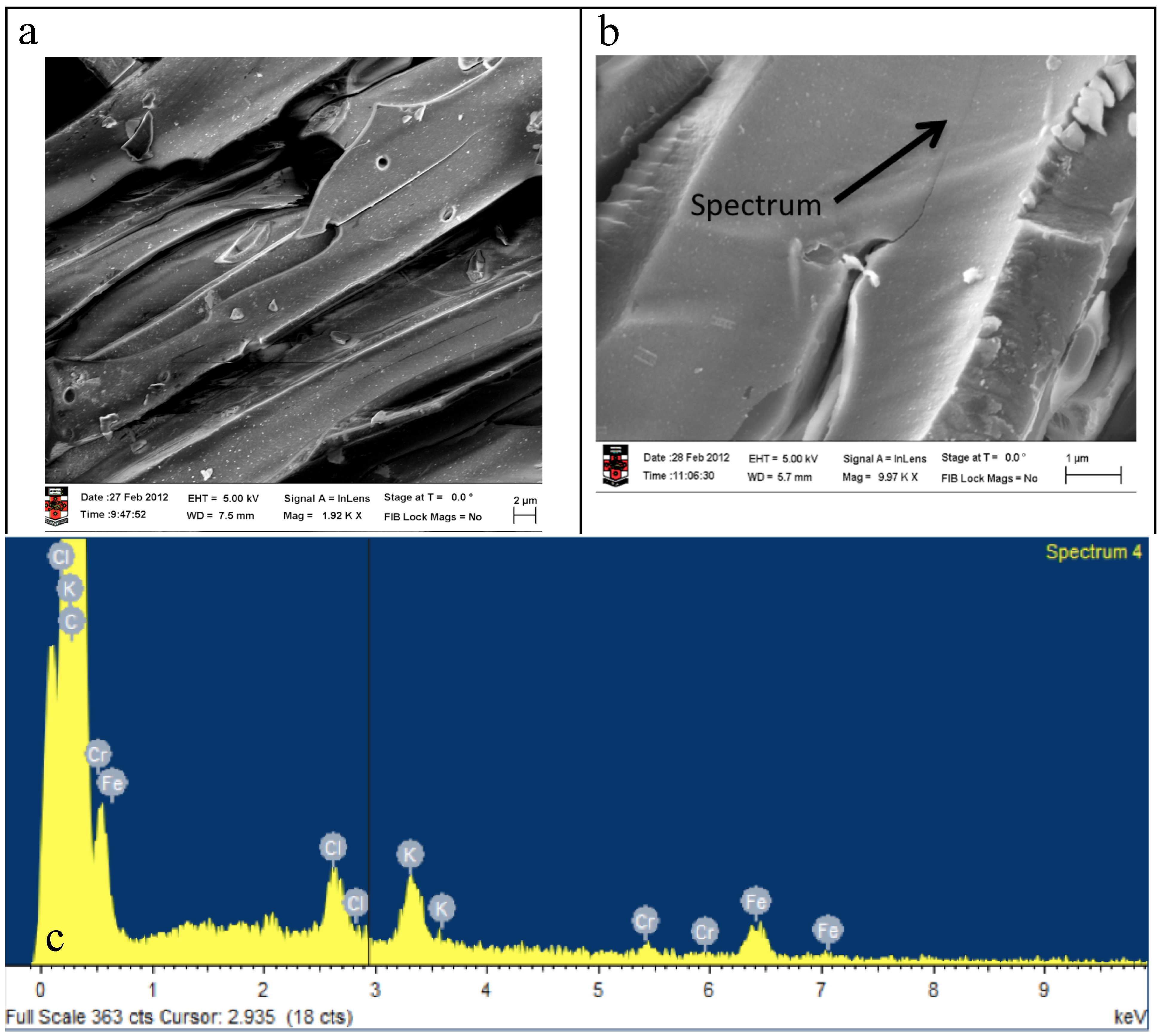
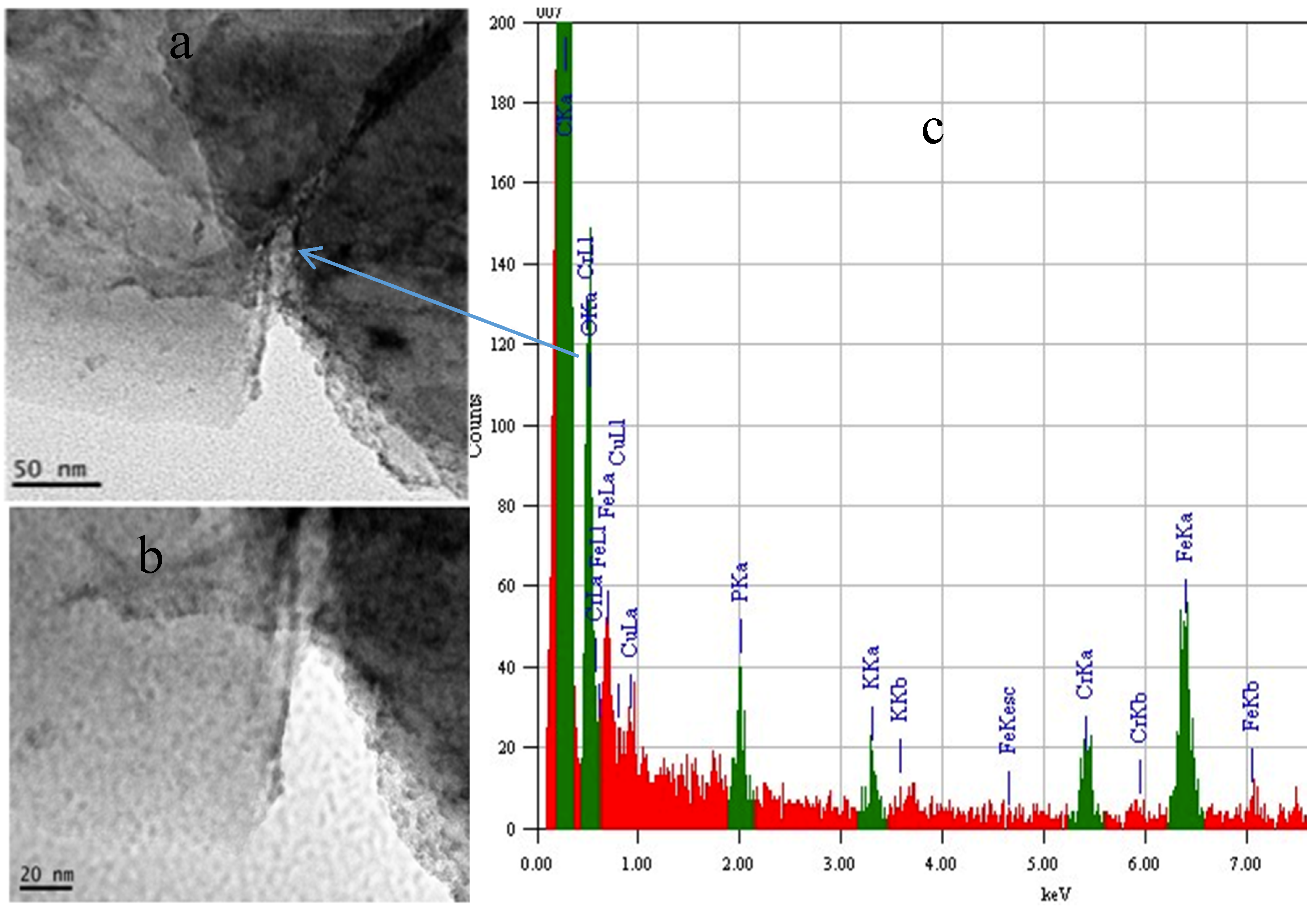

2.5. Changes in Eh and pH When Biochars are Added to Soil Using the In-Situ Measurement Technique
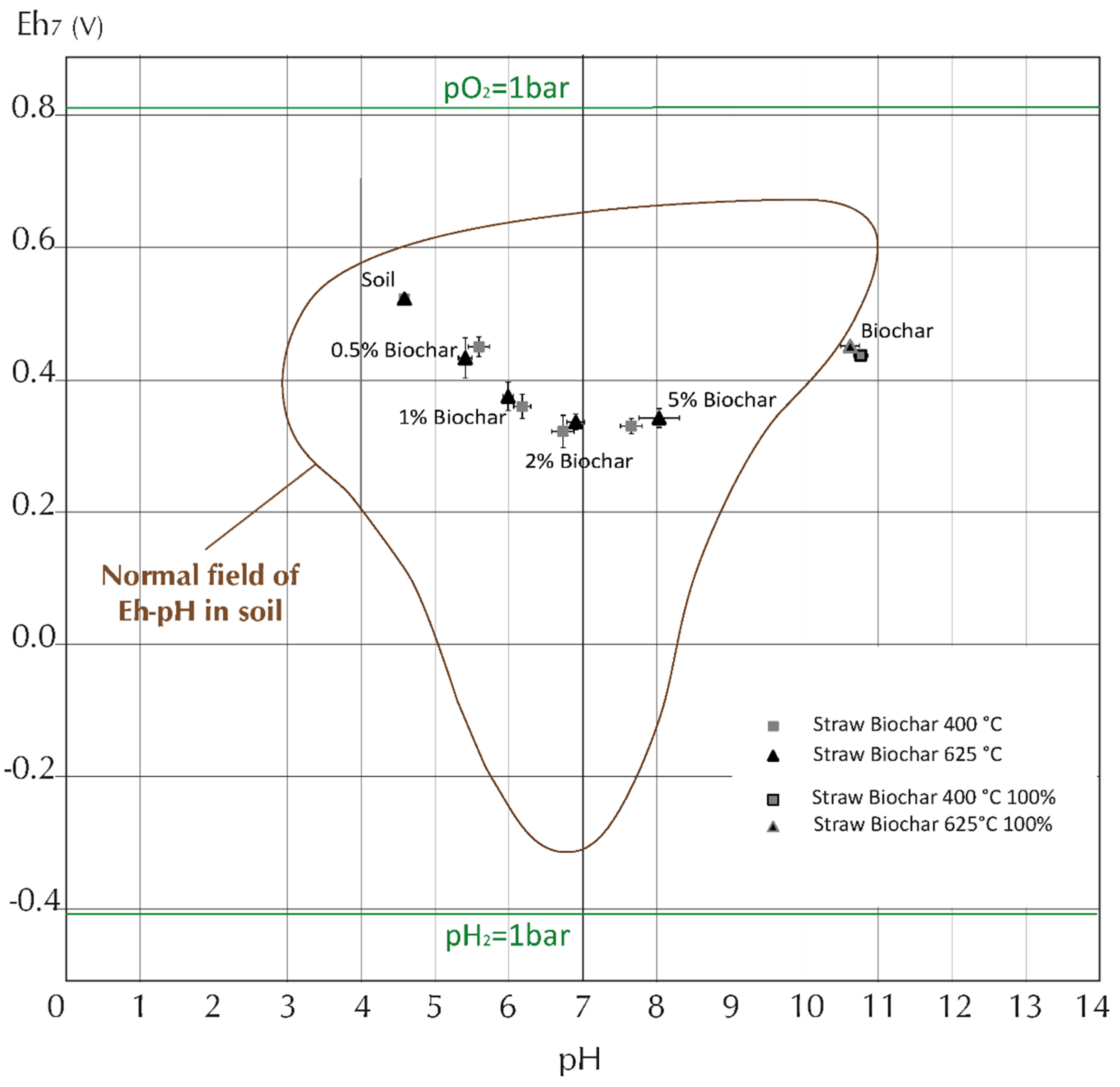
3. Conclusions and Future Directions
- Are the redox properties of biochars responsible for some or any of the different effects that biochars have been reported to have in the integrated soil/plant/rhizosphere microbiome system?
- What are the mutual redox interactions of different biochars in soils of different types?
- Do biochars increase the poising capacity of soils and why?
- Are some biochars more effective than others in altering Eh fluctuations, especially in systems (e.g., rice) where flooding and drying cycles occur?
- Does the penetration of root hairs into the pores of biochar and/or the attachment of roots to the biochar surface change the potential across the plant cell wall and change the take up of specific nutrients? If so, why does this occur?
- Do Fe and Mn/Oxide particles with diameters less than 20 nm redox active particles on the surfaces of biochar assist in the breakdown of organic matter, increase P availability and reduce the production of greenhouse gases?
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: Science, Technology and Implementation; Routledge: Abingdon, UK, 2015. [Google Scholar]
- Graber, E.; Tschansky, L.; Cohen, E. Reducing capacity of water extracts of biochars and their solubilization of soil Mn and Fe. Eur. J. Soil Sci. 2014, 65, 162–172. [Google Scholar] [CrossRef]
- Klüpfel, L.; Keiluweit, M.; Kleber, M.; Sander, M. Redox properties of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2014, 48, 5601–5611. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.D.; Camps-Arbestain, M.; Lin, Y.; Munroe, P.; Chia, C.H.; Hook, J.; van Zweiten, L.; Kimber, S.; Cowie, A.; Singh, B.P.; et al. An investigation into the reactions of biochars in soil. Aust. J. Soil Res. 2010, 48, 501–515. [Google Scholar] [CrossRef]
- Joseph, S.; Graber, E.R.; Chia, C.; Munroe, P.; Donne, S.; Thomas, T.; Nielsen, S.; Marjo, C.; Rutlidge, H.; Pan, G.X.; et al. Shifting paradigms on biochar: Micro/nano-structures and soluble components are responsible for its plant-growth promoting ability. Carbon Manag. 2013, 4, 323–343. [Google Scholar] [CrossRef]
- Bartlett, R.J.; James, B.R. Redox chemistry of soils. Adv. Agron. 1993, 50, 151–208. [Google Scholar]
- Li, Y.R.; Yu, S.R.; Strong, J.; Wang, H. Are the biogeochemical cycles of carbon, nitrogen, sulfur, and phosphorus driven by the “FeIII–FeII redox wheel” in dynamic redox environments? J. Soils Sediments 2012, 12, 683–693. [Google Scholar] [CrossRef]
- Chesworth, W. Redox, soils, and carbon sequestration. Edafología 2004, 11, 37–43. [Google Scholar]
- Chen, S.; Rotaru, A.; Shrestha, P.; Malvankar, S.; Liu, F.; Fan, W.; Nevin, K.P.; Lovley, D.R. Promoting interspecies electron transfer with biochar. Sci. Rep. 2014, 4, 5019. [Google Scholar] [CrossRef] [PubMed]
- Husson, O. Redox potential (Eh) and pH as drivers of soil/plant/microorganism systems: A transdisciplinary overview pointing to integrative opportunities for agronomy. Plant Soil 2013, 362, 389–417. [Google Scholar] [CrossRef]
- Pidello, A. Environmental redox potential and redox capacity concept using a simple polarographic experiment. J. Chem. Educ. 2003, 80, 68–70. [Google Scholar] [CrossRef]
- Glinski, J.; Stepniewski, W. Soil Aeration and its Role for Plants; CRC Press: Boca Raton, FL, USA, 1985. [Google Scholar]
- Van Zwieten, L.; Kammann, C.; Cayuela, M.-L.; Singh, B.-P.; Joseph, S.; Kimber, S.; Clough, T.; Spokas, K. Biochar Effects on Nitrous Oxide and Methane Emissions from Soil. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: Abingdon, UK, 2015. [Google Scholar]
- Davidson, E.A.; Dail, D.B.; Chorover, J. Iron interference in the quantification of nitrate in soil extracts and its effect on hypothesized abiotic immobilization of nitrate. Biogeochemistry 2008, 90, 65–73. [Google Scholar] [CrossRef]
- Yin, X.; Lv, X.; Jiang, M.; Zou, Y. Research progress of the coupling process of Fe and N in wetland soils. Chin. J. Environ. Sci. 2010, 31, 2254–2259. (In Chinese) [Google Scholar]
- Melton, E.; Swanner, E.D.; Behrens, S.; Schmidt, C.; Kappler, A. The interplay of microbially mediated and abiotic reactions in the biogeochemical Fe cycle. Nat. Rev. Microbiol. 2014, 12, 797–808. [Google Scholar] [CrossRef] [PubMed]
- Kappler, A.; Wuestner, M.L.; Ruecker, A.; Harter, J.; Halama, M.; Behrens, S. Biochar as an Electron Shuttle between Bacteria and Fe(III) Minerals. Environ. Sci. Technol. 2014, 1, 339–344. [Google Scholar] [CrossRef]
- Ishihara, S. Recent trend of advanced carbon materials from wood charcoals. Mokuzai Gakkai Shi 1996, 42, 717–723. [Google Scholar]
- Bourke, J.; Manley-Harris, M.; Fushimi, C.; Dowaki, K.; Nunoura, T.; Antal, M.J. Do all carbonized charcoals have the same chemical structure? 2. A model of the chemical structure of carbonized charcoal. Ind Eng Chem Res. 2007, 46, 5954–5967. [Google Scholar] [CrossRef]
- Arends, J.; Verstraede, W. 100 years of microbial electricity production:three concepts for the future. Microbial Biotech. 2012, 5, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, A.; Petter, B.; Madari, E. Biochar: Agronomic and environmental potential in Brazilian savannah soils. Rev. Bras. Eng. Agríc. Ambient. 2012, 16, 761–768. [Google Scholar]
- Suda, F.; Matsuo, T.; Ushioda, D. Transient changes in the power output from the concentration difference cell (dialytic battery) between seawater and river water. Energy 2007, 32, 165–173. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joseph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 2010, 327, 235–246. [Google Scholar] [CrossRef]
- Tulloch, J.; Allen, J.; Wibberley, L.; Donne, S. Influence of selected coal contaminants on graphitic carbon electro-oxidation for application to the direct carbon fuel cell. J. Power Sources 2014, 260, 140–149. [Google Scholar] [CrossRef]
- Husson, O.; Huson, B.; Brunet, A.; Babre, D.; Alary, K.; Sarthou, J.; Charpentier, H.; Durand, M.; Benada, J.; Henry, M. Practical improvements in soil redox potential (Eh) measurement for characterisation of soil properties. Application for comparison of conventional and conservation agriculture cropping systems. Anal. Chim. Acta 2015. under review. [Google Scholar]
- Fiedler, S.; Vepraskas, M.J.; Richardson, J.L. Soil redox potential: Importance, field measurements, and observations. Adv. Agron. 2007, 94, 1–54. [Google Scholar]
- Thomas, C.R.; Miao, S.L.; Sindhoj, E. Environmental factors affecting temporal and spatial patterns of soil redox potential in Florida Everglades wetlands. Wetlands 2009, 29, 1133–1145. [Google Scholar] [CrossRef]
- Benada, J. A non invasive method for redox potential measurement. Obilnarske Listy 2009, 14, 15–18. [Google Scholar]
- Whitfield, A.E.; Ullman, D.E.; German, T.L. Tomato spotted wilt virus glycoprotein G(C) is cleaved at acidic pH. Virus Res. 2005, 110, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Grundl, T. A review of current understanding of redox capacity in natural, disequilibrium systems. Chemosphere 1994, 28, 613–626. [Google Scholar] [CrossRef]
- Colic, M.; Morse, D. The elusive mechanism of the magnetic “memory” of water. Colloids Surf. A: Physicochem. Eng. Asp. 1999, 154, 167–174. [Google Scholar] [CrossRef]
- Del Giudice, E.; Spinetti, P.R.; Tedeschi, A. Water dynamics at the root of metamorphosis in living organisms. Water 2010, 2, 566–586. [Google Scholar] [CrossRef]
- Pollack, G.; Clegg, J. Unexpected Linkage between Unstirred Layers, Exclusion Zones, and Water. In Phase Transitions in Cell Biology; Pollack, G., Chin, W.C., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2008; pp. 143–152. [Google Scholar]
- Nielsen, S.; Minchin, T.; Kimber, S.; van Zwieten, L.; Caporaso, G.; Gilbert, J.; Munroe, P.; Joseph, S.; Thomas, T. Enhanced biochar causes complex shifts in soil microbial communities. Agric. Ecosyst. Environ. 2014, 191, 73–82. [Google Scholar]
- Chia, C.H.; Singh, B.P.; Joseph, S.; Graber, E.; Munroe, P. Characterization of an enriched biochar. J. Anal. Appl. Pyrolysis 2014, 108, 26–34. [Google Scholar] [CrossRef]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Thies, J.; Luizão, F.J.; Petersen, J.; et al. Black carbon increases cation exchange capacity in soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef]
- Nakamura, M.; Nakanishi, M.; Yamamoto, K. Influence of physical properties of activated carbons on characteristics of electric double-layer capacitors. J. Power Sources 1996, 60, 225–231. [Google Scholar] [CrossRef]
- Gallagher, K.G.; Fuller, T.F. Kinetic model of the electrochemical oxidation of graphitic carbon in acidic environments. Phys. Chem. Phys. 2009, 11, 11557–11567. [Google Scholar] [CrossRef] [PubMed]
- Siroma, Z.; Ishii, Z.; Yasuda, K.; Miyazaki, Y.; Inaba, M.; Tasaka, A. Imaging of highly oriented pyrolytic graphite corrosion accelerated by Pt particles. Electrochem. Commun. 2005, 7, 1153–1156. [Google Scholar] [CrossRef]
- Willsau, J.; Heitbaum, J. The influence of Pt-activation on the corrosion of carbon in gas diffusion electrodes—A dems study. Electroanal. Chem. 1984, 161, 93. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Zhang, J.L.; Yu, P.T.; Zhang, J.X.; Makharia, R.; More, K.L.; Stach, E.A. Transmission Electron Microscopy Observation of Corrosion Behaviors of Platinized Carbon Blacks under Thermal and Electrochemical Conditions. J. Electrochem. Soc. 2010, 157, B906–B913. [Google Scholar] [CrossRef]
- Crombie, K.; Mašek, O.; Sohi, S.; Brownsort, P.; Cross, A. The effect of pyrolysis conditions on biochar stability as determined by three methods. GCB Bioenergy 2013, 5, 122–131. [Google Scholar] [CrossRef]
- Youngmi, Y. Study on the degradation of carbon materials for electrocatalytic applications. Ph.D. Thesis, Technical University of Berline, Berlin, Germany, 2014. [Google Scholar]
- Maestrini, B.; Herrmann, A.M.; Nannipieri, P.; Schmidt, M.W.I.; Abiven, S. Ryegrass-derived pyrogenic organic matter changes organic carbon and nitrogen mineralization in a temperate forest soil. Soil Biol. Biochem. 2014, 69, 291–301. [Google Scholar] [CrossRef]
- Zimmerman, A.R.; Gao, M.; Ahn, M.Y. Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol. Biogeochem. 2011, 43, 1169–1179. [Google Scholar] [CrossRef]
- Baas Becking, L.G.M.; Kaplan, I.R.; Moore, D. Limits of the natural environment in terms of pH and oxidation-reduction potentials. J. Geol. 1960, 68, 243–284. [Google Scholar] [CrossRef]
- Kirk, G. The Biogeochemistry of Submerged Soils; John Wiley & Sons: Chichester, UK, 2004; pp. 92–134. [Google Scholar]
- Singla, A.; Iwasa, H.; Inubushi, K. Effect of biogas digested slurry based-biochar and digested liquid on N2O, CO2 flux and crop yield for three continuous cropping cycles of komatsuna (Brassica rapa var. perviridis). Biol. Fertil. Soils 2014, 50, 1201–1209. [Google Scholar] [CrossRef]
- Singla, A.; Dubey, S.K.; Singh, A.; Inubushi, K. Effect of biogas digested slurry-based biochar on methane flux and methanogenic archaeal diversity in paddy soil. Agric. Ecosyst. Environ. 2014, 197, 278–287. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joseph, S.; Husson, O.; Graber, E.R.; Van Zwieten, L.; Taherymoosavi, S.; Thomas, T.; Nielsen, S.; Ye, J.; Pan, G.; Chia, C.; et al. The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes. Agronomy 2015, 5, 322-340. https://doi.org/10.3390/agronomy5030322
Joseph S, Husson O, Graber ER, Van Zwieten L, Taherymoosavi S, Thomas T, Nielsen S, Ye J, Pan G, Chia C, et al. The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes. Agronomy. 2015; 5(3):322-340. https://doi.org/10.3390/agronomy5030322
Chicago/Turabian StyleJoseph, Stephen, Olivier Husson, Ellen Ruth Graber, Lukas Van Zwieten, Sara Taherymoosavi, Torsten Thomas, Shaun Nielsen, Jun Ye, Genxing Pan, Chee Chia, and et al. 2015. "The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes" Agronomy 5, no. 3: 322-340. https://doi.org/10.3390/agronomy5030322
APA StyleJoseph, S., Husson, O., Graber, E. R., Van Zwieten, L., Taherymoosavi, S., Thomas, T., Nielsen, S., Ye, J., Pan, G., Chia, C., Munroe, P., Allen, J., Lin, Y., Fan, X., & Donne, S. (2015). The Electrochemical Properties of Biochars and How They Affect Soil Redox Properties and Processes. Agronomy, 5(3), 322-340. https://doi.org/10.3390/agronomy5030322







