Abstract
Sugarcane (Saccharum spp.) brown rust (caused by Puccinia melanocephala Syd. & P. Syd) was first reported in the United States in 1978 and is still one of the great challenges for sugarcane production. A better understanding of sugarcane genotypic variation in response to brown rust will help optimize breeding and selection strategies for disease resistance. Brown rust ratings were scaled from non-infection (0) to severe infection (4) with intervals of 0.5 and routinely recorded for genotypes in the first clonal selection stage of the Canal Point sugarcane breeding program in Florida. Data were collected from 14,272 and 12,661 genotypes and replicated check cultivars in 2012 and 2013, respectively. Mean rust rating, % infection, and severity in each family and progeny of female parent were determined, and their coefficients of variation (CV) within and among families (females) were estimated. Considerable variation exists in rust ratings among families or females. The families and female parents with high susceptibility or resistance to brown rust were identified and ranked. The findings of this study can help scientists to evaluate sugarcane crosses and parents for brown rust disease, to use desirable parents for crossing, and to improve genetic resistance to brown rust in breeding programs.
1. Introduction
Sugarcane (a complex hybrid of Saccharum spp.) is an important cash crop in Florida, USA with an annual economic impact of more than $677 million [1]. Consistent and continuous development of high-yielding sugarcane cultivars with resistance or tolerance to biotic and abiotic stresses is critical for commercial sugarcane production in South Florida [2]. The USDA-ARS Sugarcane Field Station at Canal Point (26.52° N; 80.36° W), Florida was initially established at its present site in 1920 to make sugarcane crosses and produce true seed for the Louisiana sugarcane industry. Since the 1960s, the Canal Point (CP) station has been developing sugarcane cultivars with CP prefixes for Florida under a three-party cooperative agreement among the USDA-ARS, the University of Florida, and the Florida Sugar Cane League, Inc. The CP Sugarcane Field Station also makes crosses for the USDA-ARS in Houma, Louisiana. The CP cultivars now account for more than 90% of the hectarage in Florida. In 2014, the top six major sugarcane cultivars grown in Florida were “CP 96-1252” [3], “CP 00-1101” [4],“CP 01-1372” [5], “CL 88-4730” (a cultivar of the United States Sugar Corporation), “CP 88-1762” [6], and “CP 89-2143” [7], and their percentage hectares were 16.8, 13.2, 11.1, 8.8, 8.8, and 7.7%, respectively [8].
The CP sugarcane breeding and cultivar development program (CP program) consists of six stages, namely Crossing, Seedlings, and Stages I, II, III, and IV [9]. It takes at least eight years to release a cultivar from the time a cross is made [10]. Cane yield (TCH, Mg ha−1), commercial recoverable sucrose (CRS, kg sucrose Mg−1 cane), and sucrose yield (TSH, Mg ha−1) with disease resistance are the major agronomic traits considered in advancing sugarcane clones during the selection stages. Edmé et al. [11] reported that CRS, TCH, and TSH of the Florida commercial sugarcane cultivars linearly increased by 26.0, 15.5, and 47.0%, respectively, over a 33-year period from 1968 to 2000. Underscoring the critical need for cultivar development for the Florida sugarcane industry, about 69% of the sugar yield gain in Florida was from genetic improvement attributable to the CP program [11], indicating importance of the CP program in sustaining sugarcane production in Florida.
Brown rust (Figure 1), caused by Puccinia melanocephala Syd. & P. Syd., is an important disease of sugarcane in many production areas around the world [12]. Sugarcane brown rust was first recorded in the United States in 1978. It can cause substantial losses of sugarcane growth, yields, and profits [13,14,15,16,17] and has been responsible for the withdrawal of commercial cultivars in the United States and in most sugarcane growing regions of the world [18]. Although the negative effects of brown rust on sugarcane growth and yield have been reduced by breeding and management practices, the disease is still one of most important concerns in Florida sugarcane production (Figure 1).
Currently, leaf rust diseases, including brown rust and orange rust (caused by P. kuehnii (W. Krüger) E.J. Butler), are great challenges for sustainable sugarcane production in Florida [19]. Most dominant commercial cultivars in Florida are susceptible to one or both rusts (Figure 1). The typical symptoms of sugarcane brown rust and orange rust are brown and orange colors for their spores, respectively, as their names imply. Pustules of brown rust are easier to distinguish from those of orange rust on the younger, upper leaves than on the older, lower leaves [20]. Numerous coalescing pustules of rusts cause premature tissue death on leaves. Growers use fungicides to control the negative effects of rusts on yields, but the cost of fungicide applications considerably reduce the production profits [21]. Therefore, development of rust resistant cultivars is the first priority for sustainable sugarcane production. Scientists in the CP program and in the sugarcane industry in Florida are using multiple approaches to develop new cultivars with rust resistance/tolerance and high yields. Cultivars developed in the CP program are used not only in the United States, but also in many other countries in Central America [22] and Asia where sugarcane industries use them for either breeding or commercial production. Therefore, evaluation and screening genotypes for rust resistance in the CP program are important for sustainable sugarcane production in the USA and other countries. Although Sood et al. [18] developed a whorl inoculation method to more accurately and efficiently test sugarcane genotype resistance in brown rust, it is still difficult to use the artificial inoculation test in the first clonal stage (Stage I) of the CP program because of a large number (12,000–15,000) of genotypes in this stage and because of limited resources [9]. Therefore, natural infection has been the primary means of assessing rust resistance in the Stage-I clones and further artificial inoculation tests for rust resistance are usually used in later stages (Stages II and III) of the CP program [23].

Figure 1.
(A) A photo taken on 18 March 2015 in commercial sugarcane fields with a brown rust susceptible cultivar (left) and a resistance cultivar (right) and (B) brown rust pustules on leaf abaxial surface of the susceptible sugarcane.
The selection of which sugarcane clones to be used as crossing parents is a critical decision for breeders. Knowledge and better understanding of variability in rust infection and severity among genotypes may provide useful information for sugarcane genotype advancement and for efficient use of parents in future crossing efforts for rust resistance. A study was conducted in the Stage-I fields of the CP program at the USDA-ARS Sugarcane Field Station, Canal Point, Florida. The objectives of this study were to determine variability in brown rust rating among crosses (families) based on data collected from the Stage-I clones of the CP program in 2012 and 2013 and to use the information in cross appraisal. Overall, the rust diseases (both brown and orange rusts) in the 2012 and 2013 sugarcane growing seasons in the area were the most severe in the last seven years due to favorable environment conditions for the rusts. The data collected from these two years under natural infection conditions could partly reflect genotype variation in brown rust resistance or susceptibility. In this paper, we mainly focused on sugarcane brown rust because findings of sugarcane orange rust have been reported in a current article [19].
2. Results and Discussion
2.1. Brown Rust of Check Cultivars
There were significant differences among the four check cultivars and between years in mean rating of brown rust, and the cultivar × year interaction was also highly significant (p < 0.0001) (Table 1). Except for CP 80-1743, the other three cultivars had higher mean ratings of brown rust in 2012 than in 2013. In 2012, CP 78-1628 had a significantly higher mean rating of brown rust than other cultivars. In 2013, CP 78-1628 and CP 80-1743 had higher mean brown rust ratings than CP 88-1762 and CP 89-2143. The differences in % infection of brown rust among cultivars or between years were similar to those in the mean rust rating. On the other hand, the differences in rust severity were relatively less among cultivars or between years compared with % infection (Table 1). These results are consistent to those of orange rust in sugarcane [19]. Formal statistical tests of brown rust severity were not available because CP 89-2143 and CP 88-1762 had either no plots or only one to two lots were infected.
2.2. Distribution of Brown Rust Rating
Total numbers of clones used for rust rating in Stage I of the CP program were 14,272 in 2012 and 12,661 in 2013 (Figure 2). These did not include 158 and 166 replicated check plots in 2012 and 2013, respectively. The % infections of brown rust were 34.8% in 2012 and 38.5% in 2013. Distributions of brown rust ratings based on the number of clones at each rating level are given in Figure 2. Although the peak frequency of the rust rating distribution was 2 and the overall severity value was also approximately 2 in both years, the peak value of brown rust in 2012 was greater than that in 2013. The coefficients of variation (CVs) of brown rust ratings across the infected clones in 2012 (4,969) and 2013 (4,880) were 21.2% and 28.7%, respectively. Brown rust % infection and mean rating were similar between years. The differences in severity between years were small (Figure 2).

Table 1.
Mean rating, % infection, and severity of brown rust for four check cultivars tested with sugarcane clones in the Stage-I field of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013.
| Year | Cultivar | # of plots | Mean rating | % infection | Severity |
|---|---|---|---|---|---|
| 2012 | CP 78-1628 | 40 | 1.55 a† | 77.5 a | 2.00 |
| CP 80-1743 | 40 | 0.05 b | 2.5 b | 2.00 | |
| CP 88-1762 | 40 | 0.14 b | 7.5 b | 1.83 | |
| CP 89-2143 | 38 | 0.08 b | 5.3 b | 1.50 | |
| Mean | 40 | 0.46 | 23.2 | 1.83 | |
| 2013 | CP 78-1628 | 40 | 0.34 a | 25.0 a | 1.35 |
| CP 80-1743 | 40 | 0.43 a | 20.0 a | 2.13 | |
| CP 88-1762 | 43 | 0.05 b | 2.3 b | 2.00 | |
| CP 89-2143 | 43 | 0.00 b | 0.0 b | 0.00 | |
| Mean | 42 | 0.21 | 11.8 | 1.37 |
†Data following the same letter within a year are not significant at p = 0.05.
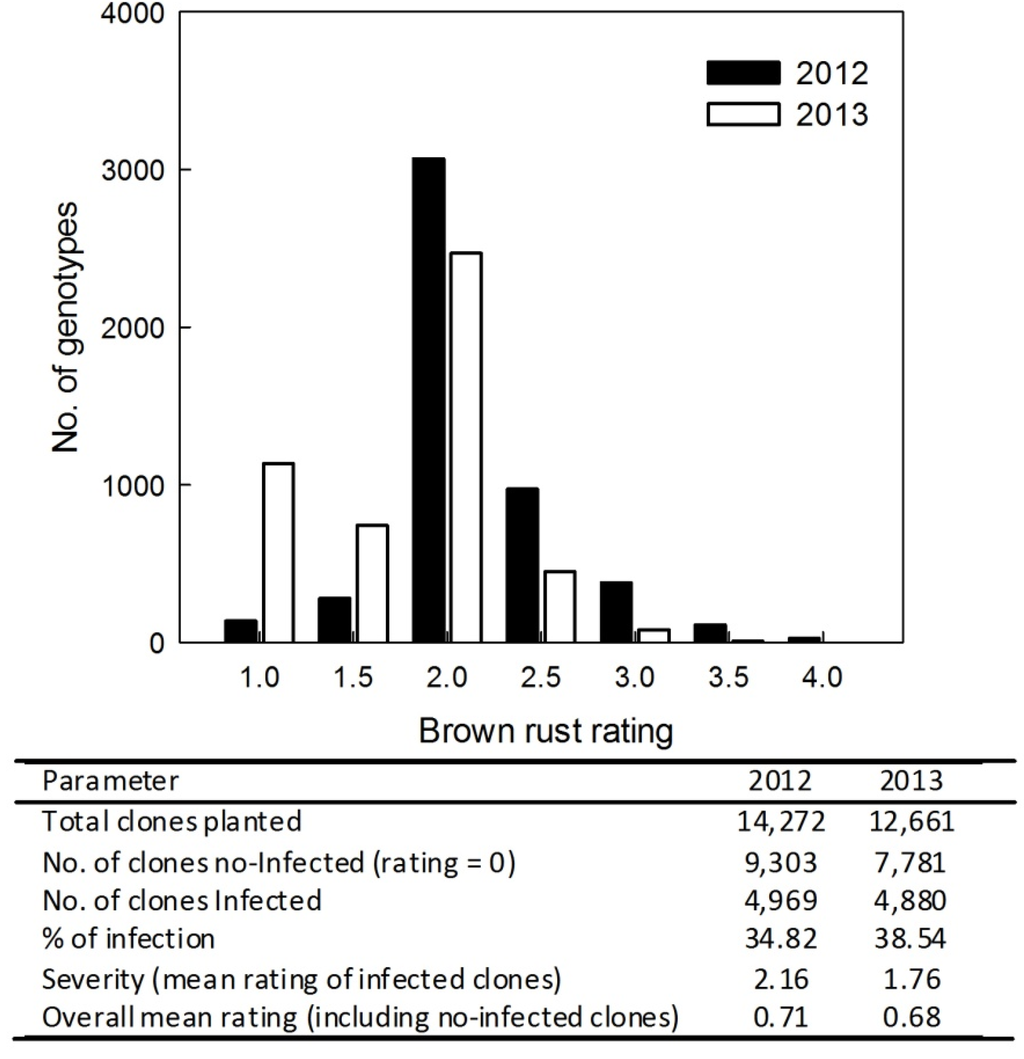
Figure 2.
Distributions of brown rust based on the number of infected clones at different rust rating levels from 1 to 4 in Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013. Note: Brown rust ratings were assessed in July; the rust rating is equal to 0 or no rust infected clones; and other important parameters are also listed in the figure.
2.3. Variability in Clonal Numbers and Brown Rust Ratings among Families
Numbers of total crosses (families) advanced to Stage I from the seedling stage by individual selection in 2012 and 2013 were 576 and 455, respectively. Clone numbers among families ranged from 1 to 214 with a mean of 25 in 2012, and from 1 to 209 with a mean of 28 in 2013, and their CVs in two years were 113% and 100%, respectively (Table 2). This substantial variability in the number of clones among families is probably associated with differences in the number of viable seeds per cross in the crossing stage, variability in the number of seedlings planted per family in the seedling stage, and variable selection rates among families in the seedling stage [19]. On a family basis, mean brown rust ratings ranged from 0.0 to 2.5 in 2012 and from 0.0 to 2.2 in 2013. Among-family CV values of brown rust in 2012 and 2013 were 62.9% and 60.3%, respectively. Within-family CVs (136%–150%) were greater than their among-family CVs. Large variability in the number of clones and in the rust ratings (Table 2) among families in Stage I of the CP program suggested that Stage-I data can be used to identify useful parental combinations and individual parents for their progeny in resistance/tolerance to rusts in the test years. As variation in sugarcane orange rust discussed by Zhao et al. [19], the greater CV for rust rating within families than among families suggests that placing more emphasis on both individual clonal evaluation and family-based evaluation in Stage I of the CP program may help improve our knowledge and ability to select genotypes with potential for eliminating the negative effects of rusts on sugarcane growth and yields.

Table 2.
Maximum, minimum, mean, standard deviation (SD), and coefficient of variation (CV) for clone number and mean brown rust ratings of 576 (2012) and 455 (2013) sugarcane families with a total of 14,272 and 12,661 clones, respectively, tested in the 2012 and 2013 Stage-I fields of the Canal Point (CP) sugarcane breeding and cultivar development program.
| Parameter | 2012 | 2013 | ||
|---|---|---|---|---|
| Clone | Rust rating | Clone | Rust rating | |
| (No. family−1) | (No. family−1) | |||
| Maximum | 214 | 2.50 | 209 | 2.50 |
| Minimum | 1 | 0.00 | 1 | 0.00 |
| Mean | 25 | 0.46 | 28 | 0.96 |
| SD | 28 | 0.49 | 28 | 0.51 |
| CV (%, among families) | 113 | 104.9 | 100 | 52.7 |
| CV (%, within families) | --- | 184.2 | --- | 119.1 |
2.4. Correlations of Mean Brown Rust Ratings with % Infection and Severity
To properly evaluate sugarcane families, at least 10 to 20 clones per family are required [24]. Therefore, the families that had ≥15 clones were used in the present study to further investigate relationships between mean rust rating and % infection or rust severity. In 576 (2012) and 455 (2013) families planted in Stage I, 295 and 263 families, respectively, had ≥15 clones. From these families with ≥15 clones, the mean brown rust ratings, % infections, and severity values for each family were calculated and the relationships between these three parameters were further determined (Figure 3). Overall, the mean ratings of brown rust among families ranged from 0.0 to 1.8; the values of % infection ranged from 0.0 to 87.0%; and the values of severity ranged from 0.0 to 3.3. Averaged across years, the CV values of the mean rating, % infection, and severity were 47.4, 44.4, and 13.6%, respectively, among families. The mean brown rust rating highly and linearly correlated with % infection (r2 = 0.91 to 0.95****), but the relationship between the mean rating and severity value was poor (r2 = 0.04 to 0.13) (Figure 3). The similar relationships between the three rust traits were also found in sugarcane orange rust [19]. Considering the variability (CV) and correlations between the three rust variables, rust severity may not be a good parameter to distinguish family differences in response to brown rust in the early selection stage of a sugarcane breeding and selection program such as the CP program.
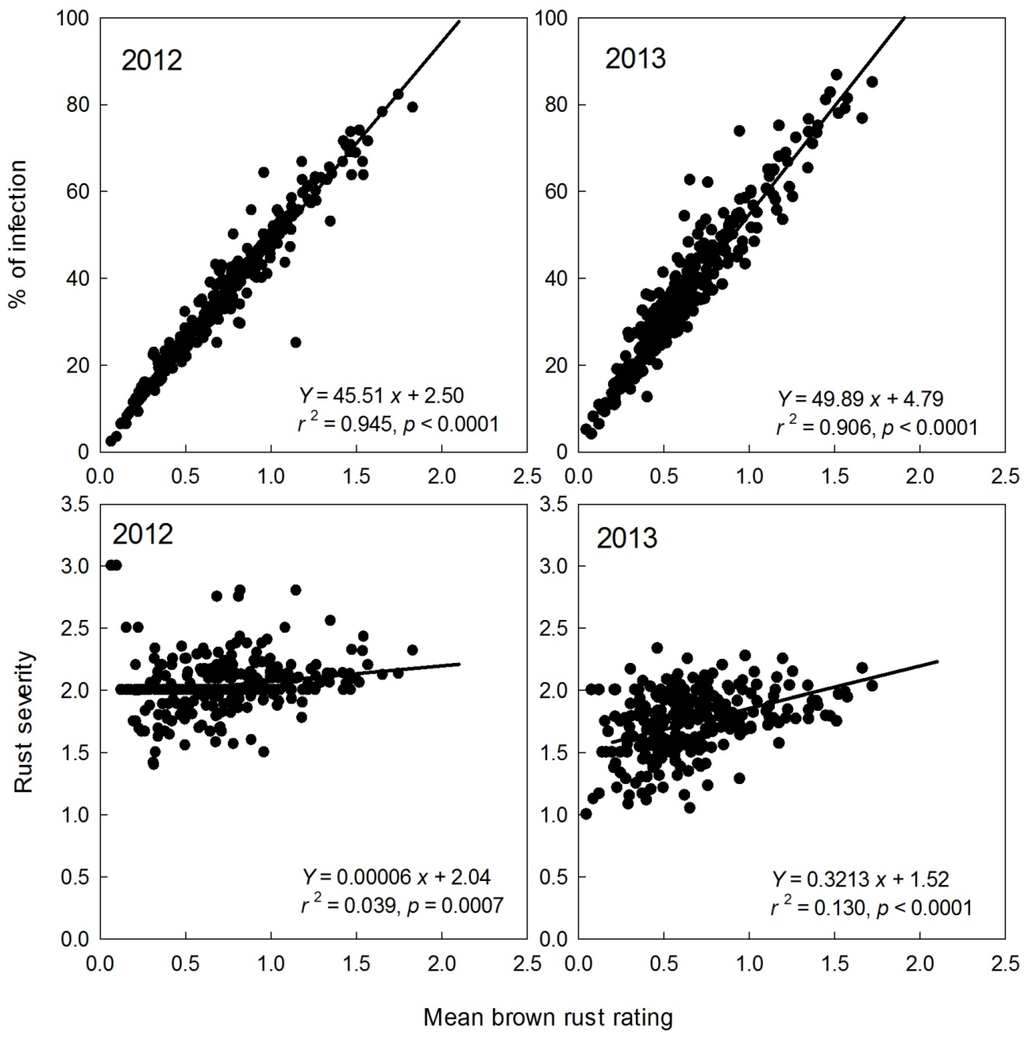
Figure 3.
Relationships between mean rust ratings and % infection or severity of brown rust for 295 and 263 families with ≥15 clones, respectively, in the 2012 and 2013 Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program.
2.5. Evaluation of Families Based on Mean Rust Rating
The families with ≥15 clones were also used to evaluate family tolerance to brown rust. The 295 and 263 families that had ≥15 clones planted in Stage I of the CP program in 2012 and 2013, respectively, were ranked based on their mean ratings of brown rust (Table 3 and Table 4). The mean brown rust ratings across families were 0.8 (ranged from 0.1 to 2.0) in 2012 and 0.7 (ranged from 0.0 to 1.7) in 2013 with CV values of 47%–48%. The top 20 families with the highest (Table 3) and 20 families with the lowest (Table 4) mean ratings of brown rust in each year were identified. The % infection and severity of brown rust for these families and their parents (females and males) are also given in Table 3 and Table 4. Overall, there was greater variability (CV) in the mean rating and in severity of brown rust within family than among families. Parents of these families could provide useful information for crossing combinations. For instance, reducing the use of parents listed in Table 3 (especially CL 90-4500, CP 95-1039, and CP 96-1252) and increasing the use of parents listed in Table 4 for crossing may improve brown rust tolerance of progeny in Stage I of the CP program.
2.6. Evaluation of Females Based on Mean Rust Ratings of Their Progeny
When data were analyzed by female parents regardless of males, there were 204 and 157 females, respectively, in 2012 and 2013. Of these females, 135 in 2012 and 113 females in 2013 had at least 15 progeny planted in the Stage-I fields. The progeny of these females were sorted by mean ratings of brown rust. The 20 females with their progeny having the greatest (Table 5) and 20 females with progeny having the lowest (Table 6) mean ratings of brown rust were further identified and ranked according to methods used by Zhao et al. [19].
There were 250 clones in 2012 with the female parent CL 90-4500 and 285 clones in 2013 with the female parent CP 92-1167. The mean brown rust ratings of these two female progeny ranked fourth and eighth, respectively (Table 5). Additionally, progeny of females CP 96-1252, CP 95-1039, and CPCL 02-8021 ranked in the top 20 in both years for their mean brown rust ratings. On the other hand, the progeny developed from some female parents had the lowest mean ratings of brown rust (Table 6). For instance, the clones with female parent CP 06-2274 or CP 88-1762 had low mean ratings of brown rust (i.e., more tolerance to brown rust) in both years.
Brown rust is one of the most devastating diseases for sugarcane production in Florida, Louisiana, and many other production areas around the world [12,16,17,18]. Using proper parents with resistance to brown rust to make crosses is important in the CP program. There was great variation in brown rust ratings among families (Table 3 and Table 4) in Stage I of the CP program. The great variation in mean ratings of brown rust among female parents was also detected (Table 5 and Table 6). Clearly, using clones listed in Table 5 as female parents increases the risk of obtaining progeny with high brown rust ratings. These clones should be limited in their use as parents in the CP program in so far as brown rust resistance is concerned. Overall, the progeny developed from female parents listed in Table 6 had the lowest mean ratings of brown rust. Increased usage of these female clones for crossing may improve resistance to brown rust of progeny in Stage I of the CP program.

Table 3.
Number of total sugarcane clones planted, mean brown rust (BR) rating, % infection, and severity and their parents for the 20 families with the highest mean BR ratings using 295 and 263 families with ≥15 clones in Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013, respectively.
| 2012 | 2013 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family | No. clone | BR rating | % infect | Severity | Female† | Male† | Family | No. clone | BR rating | % infect | Severity | Female† | Male† |
| X09-1084 | 24 | 2.02 | 79.2 | 2.55 | CL90-4500(-) | CP80-1743(+) | X10-1077 | 20 | 1.73 | 85.0 | 2.03 | CP95-1039(-) | CP06-2170(-) |
| X09-0920 | 28 | 1.84 | 82.1 | 2.24 | CL90-4500(-) | CPCL00-4027(-) | X05-0009 | 90 | 1.67 | 76.7 | 2.17 | CP97-1989(-) | CP80-1743(+) |
| X09-0864 | 32 | 1.73 | 78.1 | 2.22 | CP95-1039(-) | Poly09-23 | X09-1235 | 32 | 1.58 | 81.3 | 1.94 | CP01-2390(-) | CP04-1105(-) |
| X10-0369 | 24 | 1.71 | 66.7 | 2.56 | TCP07-4820(-) | Poly10-09 | X10-0909 | 38 | 1.57 | 78.9 | 1.98 | CL84-3152(-) | Poly10-20 |
| X10-0531 | 22 | 1.68 | 63.6 | 2.64 | CP08-2409(-) | CP02-2281(+) | X09-0184 | 36 | 1.53 | 77.8 | 1.96 | CPCL97-0393(-) | CP04-1105(-) |
| X09-1270 | 16 | 1.63 | 68.8 | 2.36 | CPCL05-1777(-) | HoCP96-540(-) | X10-0286 | 30 | 1.52 | 86.7 | 1.75 | CPCL02-8021(-) | L01-283(-) |
| X07-0943 | 23 | 1.61 | 73.9 | 2.18 | CPCL06-8007(-) | CPCL01-0271(-) | X10-0771 | 23 | 1.48 | 82.6 | 1.79 | CL84-3152(-) | CP06-2170(-) |
| X09-0181 | 21 | 1.60 | 71.4 | 2.23 | CPCL01-6755(-) | CP04-1105(-) | X10-0816 | 21 | 1.45 | 81.0 | 1.79 | CPCL01-6755(-) | CP06-2170(-) |
| X09-0186 | 21 | 1.60 | 71.4 | 2.23 | CP96-1252(-) | TCP98-4447(-) | X10-1092 | 16 | 1.41 | 75.0 | 1.88 | CP95-1039(-) | CPCL06-3470(?) |
| X09-1070 | 45 | 1.56 | 68.9 | 2.26 | CL90-4500(-) | CP02-2281(+) | X10-0603 | 15 | 1.40 | 73.3 | 1.91 | CPCL02-8021(-) | CPCL01-0271(-) |
| X10-0697 | 27 | 1.56 | 70.4 | 2.21 | TCP00-4530(-) | Poly10-15 | X10-0812 | 24 | 1.38 | 70.8 | 1.91 | CPCL02-7386(-) | CP06-2170(-) |
| X09-0796 | 34 | 1.54 | 73.5 | 2.10 | CL90-4500(-) | Poly09-21 | X10-0645 | 34 | 1.35 | 73.5 | 1.84 | CPCL02-7363(?) | CP06-2170(-) |
| X09-1232 | 17 | 1.53 | 70.6 | 2.17 | CL88-4730(-) | HoCP96-540(-) | X09-0784 | 17 | 1.35 | 76.5 | 1.77 | CP96-1252(-) | Poly09-21 |
| X09-0949 | 44 | 1.52 | 63.6 | 2.39 | CL90-4500(-) | CP06-2664(-) | X09-1146 | 27 | 1.28 | 66.7 | 1.92 | CP80-1827(-) | MaleMix09G |
| X09-0893 | 33 | 1.50 | 66.7 | 2.25 | CP97-1387(-) | Poly09-24 | X09-0864 | 18 | 1.28 | 72.2 | 1.77 | CP95-1039(-) | Poly09-23 |
| X09-0784 | 36 | 1.49 | 63.9 | 2.33 | CP96-1252(-) | Poly09-21 | X10-0569 | 29 | 1.26 | 58.6 | 2.15 | CP89-2377(-) | Poly10-12 |
| X09-1228 | 26 | 1.44 | 65.4 | 2.33 | CL90-4500(-) | HoCP96-540(-) | X10-0479 | 23 | 1.24 | 60.9 | 2.04 | CP08-1773(-) | CPCL06-3332(+) |
| X09-1234 | 24 | 1.44 | 62.5 | 2.30 | CP95-1039(-) | CP04-1105(-) | X10-0691 | 24 | 1.23 | 66.7 | 1.84 | CPCL96-2061(-) | Poly10-15 |
| X10-0375 | 15 | 1.43 | 60.0 | 2.39 | CP99-1893(-) | Poly10-09 | X11-0497 | 16 | 1.22 | 68.8 | 1.77 | CP97-1777(-) | TCP04-4709(-) |
| X07-1099 | 17 | 1.38 | 52.9 | 2.61 | CPCL99-1371(-) | Mix07J | X11-0539 | 15 | 1.20 | 53.3 | 2.25 | CP02-2281(+) | CR1009(?) |
| Mean‡ | 26 (42) | 1.59 (0.78) | 69 (36) | 2.33 (2.19) | Mean‡ | 27 (43) | 1.40 (0.67) | 73 (38) | 1.92 (1.75) | ||||
| Max | 45 (214) | 2.02 (2.02) | 82 (82) | 2.64 (3.00) | Max | 90 (209) | 1.73 (1.73) | 87 (87) | 2.25 (3.25) | ||||
| Min | 15 (15) | 1.38 (0.07) | 53 (2.3) | 2.10 (1.58) | Min | 15 (15) | 1.20 (0.00) | 53 (0.0) | 1.75 (0.00) | ||||
| CV (A) ¶ | 33 (71) | 10 (47) | 10 (45) | 7 (10) | CV(A) ¶ | 60 (66) | 11 (48) | 12 (44) | 8 (15) | ||||
| CV (W) ¶ | 73 (158) | 18 (17) | CV(W) ¶ | 67 (151) | 21 (26) | ||||||||
† The “+”, “-”, and “?” in the parenthesis of each parent indicate presence, absence, and non-availability of the Bru1 gene, respectively; ‡ The first values of mean, maximum (Max), minimum (Min), and CV are calculated based on the top highest 20 families and the second values within parentheses are based on all 279 (2012) or 265 (2013) families with ≥15 clones planted; ¶ The CV(A) and CV(W) represent among- and within-family CVs, respectively.

Table 4.
Number of total sugarcane clones planted, mean brown rust (BR) rating, % infection, and severity and their parents for the 20 families with the lowest mean BR ratings using 295 and 263 families with ≥15 clones in Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013, respectively.
| 2012 | 2013 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family | No. clone | BR rating | % infect | Severity | Female† | Male† | Family | No. clone | BR rating | % infect | Severity | Female† | Male† |
| X09-0783 | 44 | 0.28 | 13.6 | 2.08 | CP88-1762(+) | Poly09-21 | X10-1194 | 28 | 0.25 | 14.3 | 1.75 | CPCL99-2574(-) | Poly10-22 |
| X09-0634 | 41 | 0.28 | 14.6 | 1.92 | CPCL05-1108(?) | Poly09-19 | X10-0347 | 18 | 0.25 | 16.7 | 1.50 | CPCL99-2103(-) | CP06-2664(-) |
| X07-0933 | 30 | 0.27 | 13.3 | 2.00 | CPCL99-1371(-) | CL89-5189(-) | X10-0709 | 16 | 0.25 | 18.8 | 1.33 | CP06-2664(-) | CL95-5255(+) |
| X09-0978 | 17 | 0.26 | 11.8 | 2.25 | CPCL02-8001(-) | Poly09-25 | X10-0360 | 54 | 0.24 | 13.0 | 1.86 | CPCL06-3458(?) | CP88-1762(+) |
| X09-0386 | 16 | 0.25 | 12.5 | 2.00 | CP02-1143(+) | Poly09-13 | X10-0136 | 37 | 0.23 | 18.9 | 1.21 | CP06-2397(+) | Poly10-03 |
| X09-1117 | 81 | 0.25 | 14.8 | 1.67 | CPCL00-0458(-) | MaleMix09E | X09-1034 | 18 | 0.22 | 11.1 | 2.00 | CPCL99-2103(+) | CL88-4730(-) |
| X09-0874 | 33 | 0.24 | 12.1 | 2.00 | CPCL05-1108(?) | Poly09-23 | X10-0536 | 70 | 0.22 | 15.7 | 1.41 | CP05-1730(?) | Poly10-10 |
| X09-1114 | 96 | 0.23 | 13.5 | 1.69 | CP02-2454(+) | MaleMix09E | X10-0374 | 56 | 0.21 | 10.7 | 2.00 | CP88-1762(+) | Poly10-09 |
| X10-0351 | 22 | 0.23 | 9.1 | 2.50 | CP06-2406(?) | CP06-2042(+) | X10-0467 | 26 | 0.21 | 15.4 | 1.38 | CPCL02-7386(-) | CP06-2335(+) |
| X10-0378 | 47 | 0.22 | 10.6 | 2.10 | CL89-5189(-) | Poly10-09 | X09-0511 | 15 | 0.20 | 13.3 | 1.50 | CP02-2065(+) | Poly09-16 |
| X09-1133 | 52 | 0.22 | 9.6 | 2.30 | CP89-1509(+) | MaleMix09F | X10-0543 | 25 | 0.18 | 12.0 | 1.50 | CP06-3040(-) | Poly10-11 |
| X07-1118 | 71 | 0.21 | 11.3 | 1.88 | CP04-2166(+) | CPCL00-6131(-) | X10-1109 | 28 | 0.18 | 10.7 | 1.67 | CPCL06-3458(?) | Mix10-06 |
| X07-0837 | 33 | 0.20 | 9.1 | 2.17 | CP03-1401(-) | Poly07-04 | X09-1239 | 18 | 0.17 | 11.1 | 1.50 | CL89-5189(-) | CP96-1252(-) |
| X10-0535 | 23 | 0.17 | 8.7 | 2.00 | CP02-1554(-) | Poly10-10 | X10-0693 | 22 | 0.16 | 9.1 | 1.75 | CP05-1730(?) | Poly10-15 |
| X07-1280 | 95 | 0.16 | 6.3 | 2.58 | CP00-1630(+) | Poly07-16 | X10-0318 | 21 | 0.14 | 9.5 | 1.50 | CP07-1860(?) | CP97-1777(-) |
| X09-1150 | 25 | 0.16 | 8.0 | 2.00 | CP05-1518(+) | CP88-1762(+) | X10-0895 | 28 | 0.13 | 10.7 | 1.17 | CPCL96-4974(-) | Poly10-19 |
| X09-1188 | 31 | 0.13 | 6.5 | 2.00 | CP05-1518(+) | CP80-1743(+) | X10-0176 | 16 | 0.13 | 6.3 | 2.00 | CP06-2897(-) | CL88-2747(-) |
| X10-0360 | 16 | 0.13 | 6.3 | 2.00 | CPCL06-3458(?) | CP88-1762(+) | X10-0272 | 50 | 0.09 | 8.0 | 1.13 | CP06-2274(-) | Poly10-05 |
| X09-1269 | 30 | 0.10 | 3.3 | 3.00 | CPCL05-1306(?) | HoCP96-540(-) | X10-0152 | 25 | 0.08 | 4.0 | 2.00 | CP08-2003(?) | CP88-1762(+) |
| X09-1236 | 44 | 0.07 | 2.3 | 3.00 | CP03-1160(-) | CP04-1105(-) | X10-0389 | 17 | 0.00 | 0.0 | 0.00 | CPCL02-6848(+) | Poly10-09 |
| Mean‡ | 42(42) | 0.20(0.78) | 10(36) | 2.16(2.17) | Mean‡ | 29(43) | 0.18(0.67) | 12(38) | 1.59(1.75) | ||||
| Max | 96(214) | 0.28(2.02) | 16(82) | 3.00(3.00) | Max | 70(209) | 0.25(1.73) | 19(87) | 2.00(3.25) | ||||
| Min | 16(15) | 0.07(0.07) | 2.3(2.3) | 1.67(1.58) | Min | 15(15) | 0.00(0.00) | 0.0(0.0) | 0.00(0.00) | ||||
| CV(A) ¶ | 59(71) | 31(47) | 37(45) | 17(10) | CV(A) ¶ | 55(66) | 38(48) | 41(44) | 18(15) | ||||
| CV(W) ¶ | 337(158) | 14(17) | CV(W) ¶ | 299(151) | 28(26) | ||||||||
† The “+”, “-”, and “?” in the parenthesis of each parent indicate presence, absence, and non-availability of the Bru1 gene, respectively; ‡ The first values of mean, maximum (Max), minimum (Min), and CV are calculated based on the lowest 20 families and the second values within parentheses are based on all 279 (2012) or 265 (2013) families with ≥15 clones planted; ¶The CV(A) and CV(W) represent among- and within-family CVs, respectively.

Table 5.
Number of total clones planted and mean brown rust (BR) rating for the 20 females in which their progeny had the highest mean BR ratings using 135 and 113 females with ≥15 progeny clones in Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013, respectively.
| 2012 | 2013 | ||||
|---|---|---|---|---|---|
| Female† | No. clone | Mean BR rating | Female† | No. clone | Mean BR rating |
| US90-0018 (-) | 19 | 2.05 | CP97-1989 (-) | 95 | 1.61 |
| TCP07-4820 (?) | 24 | 1.71 | CPCL97-0393 (-) | 38 | 1.55 |
| CPCL05-1777 (-) | 16 | 1.63 | CP95-1039 (-) | 95 | 1.26 |
| CL90-4500 (-) | 250 | 1.54 | CPCL96-2061 (-) | 24 | 1.23 |
| CP97-1387 (-) | 33 | 1.50 | CPCL95-2287 (-) | 17 | 1.15 |
| CP99-1893 (-) | 15 | 1.43 | CP80-1827 (-) | 48 | 1.08 |
| TCP00-4530 (-) | 53 | 1.39 | CP08-2398 (-) | 36 | 1.08 |
| TCP97-4416 (-) | 39 | 1.37 | CP92-1167 (-) | 285 | 1.03 |
| CPCL01-6755 (-) | 63 | 1.34 | CL88-4730 (-) | 91 | 1.00 |
| CP04-1566 (-) | 17 | 1.29 | TCP04-4688 (-) | 18 | 1.00 |
| TCP07-4806 (?) | 19 | 1.26 | CPCL06-8004 (-) | 113 | 0.97 |
| CP96-1252 (-) | 192 | 1.25 | CL84-3152 (-) | 630 | 0.97 |
| CP06-3098 (-) | 20 | 1.23 | CP96-1252 (-) | 243 | 0.93 |
| CPCL06-8007 (-) | 46 | 1.20 | CPCL02-8021 (-) | 158 | 0.92 |
| CPCL99-1777 (-) | 29 | 1.17 | CP00-1301 (-) | 28 | 0.91 |
| CPCL02-8021 (-) | 248 | 1.14 | CPCL02-7363 (?) | 90 | 0.90 |
| CP95-1039 (-) | 304 | 1.14 | CP01-2390 (-) | 204 | 0.88 |
| CP06-2657 (?) | 40 | 1.13 | CP06-2214 (+) | 252 | 0.86 |
| CP08-2409 (-) | 58 | 1.11 | CL87-1630 (-) | 61 | 0.85 |
| CP78-1628 (-) | 54 | 1.11 | CP08-1965 (-) | 128 | 0.85 |
| Mean‡ | 77 (102) | 1.35 (0.67) | Mean‡ | 133 (108) | 1.05 (0.64) |
| Max | 304 (639) | 2.05 (2.05) | Max | 630 (630) | 1.61 (1.61) |
| Min | 15 (15) | 1.11 (0.14) | Min | 17 (16) | 0.85 (0.00) |
| CV (A) ¶ | 118 (109) | 18 (44) | CV (A) ¶ | 108 (109) | 21 (43) |
| CV (W) ¶ | 87 (156) | CV (W) ¶ | 95 (152) | ||
† The “+”, “-”, and “?” in the parenthesis of each female parent indicate presence, absence, and non-availability of the Bru1 gene, respectively; ‡ The first values of mean, maximum (Max), minimum (Min), and coefficient of variation (CV) are calculated based on mean brown rust ratings of top 20 female parents and the second values within parentheses are based on all 136 (2012) or 113 (2013) female parents with ≥15 clones planted; ¶ The CV (A) and CV (W) represent among- and within-family CVs, respectively.

Table 6.
Number of total clones planted and mean brown rust (BR) rating for the 20 females in which their progeny had the lowest mean BR ratings using 135 and 113 females with ≥15 progeny clones in Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program in 2012 and 2013, respectively.
| 2012 | 2013 | ||||
|---|---|---|---|---|---|
| Female† | No. clone | Mean BR rating | Female† | No. clone | Mean BR rating |
| CPCL00-1373 (-) | 58 | 0.42 | CP99-1896 (-) | 48 | 0.40 |
| CPCL02-7406 (+) | 70 | 0.42 | CL87-2882 (-) | 36 | 0.38 |
| CP02-2454 (+) | 228 | 0.42 | CPCL02-8001 (-) | 35 | 0.37 |
| CP04-1367 (+) | 122 | 0.41 | CP06-1730 (?) | 50 | 0.37 |
| CP89-1509 (+) | 95 | 0.41 | CP88-1762 (+) | 96 | 0.33 |
| CP05-1466 (+) | 52 | 0.40 | CL87-2282 (?) | 20 | 0.33 |
| CP06-2274 (-) | 53 | 0.40 | CPCL02-0843 (+) | 20 | 0.33 |
| CPCL05-1009 (-) | 18 | 0.39 | CPCL02-7610 (+) | 49 | 0.31 |
| HoCP04-856 (-) | 16 | 0.38 | CPCL02-7080 (-) | 85 | 0.30 |
| CL88-2747 (-) | 103 | 0.36 | CL94-0150 (-) | 43 | 0.29 |
| CP01-1178 (-) | 38 | 0.36 | CPCL06-3470 (?) | 16 | 0.28 |
| CP03-1912 (+) | 64 | 0.35 | CP05-1678 (-) | 72 | 0.28 |
| CP00-2164 (+) | 51 | 0.34 | CPCL99-2574 (-) | 28 | 0.25 |
| CPCL00-0458 (-) | 138 | 0.31 | CP06-2274 (-) | 89 | 0.25 |
| CPCL02-6334 (+) | 151 | 0.29 | CPCL99-2103 (-) | 36 | 0.24 |
| CP88-1762 (+) | 44 | 0.28 | CP05-1730 (?) | 92 | 0.21 |
| CP00-1630 (+) | 138 | 0.21 | CP06-3040 (-) | 25 | 0.18 |
| CP03-1160 (-) | 72 | 0.18 | CPCL96-4974 (-) | 28 | 0.13 |
| CP02-1554 (-) | 23 | 0.17 | CP08-2003 (?) | 25 | 0.08 |
| CP05-1518 (+) | 56 | 0.14 | CPCL02-6848 (+) | 19 | 0.00 |
| Mean‡ | 80 (102) | 0.33 (0.67) | Mean‡ | 46 (108) | 0.26 (0.64) |
| Max | 228 (639) | 0.42 (2.05) | Max | 96 (630) | 0.40 (1.61) |
| Min | 16 (15) | 0.14 (0.14) | Min | 16 (16) | 0.00 (0.00) |
| CV (A) ¶ | 67 (109) | 27 (44) | CV (A)¶ | 59 (109) | 39 (43) |
| CV (W) ¶ | 246 (156) | CV (W) ¶ | 245 (152) | ||
† The “+”, “-”, and “?” in the parenthesis of each female parent indicate presence, absence, and non-availability of the Bru1 gene, respectively; ‡ The first values of mean, maximum (Max), minimum (Min), and coefficients of variation (CV) are calculated based on mean brown rust ratings of the lowest 20 female parents and the second values within parentheses are based on all 136 (2012) or 113 (2013) female parents with ≥15 clones planted; ¶ The CV (A) and CV (W) represent among- and within-family CVs, respectively.
2.7. Bru1 in Parental Clones and Rust Rating in Their Progeny
Bru1 is a major gene for resistance to brown rust of sugarcane [25]. It is important for sugarcane breeders to develop a database of parental clones using agronomic and physiological traits and molecular markers for improving disease resistance and for sustaining yields and profits [19]. Phylogenetic analysis of sugarcane rusts based on rDNA sequences and phylogenetic relationships of sugarcane rust fungi have been reported [26,27]. The sugarcane brown rust resistance gene (Bru1) was recently found to be prevalent in 86% of brown rust-resistant clones in a sample of 380 modern cultivars and breeding materials covering worldwide diversity [28]. Therefore, the opportunity exists to utilize Bru1 in marker-assisted breeding and selection in order to improve brown rust resistance in sugarcane [29]. Bru1 has been used as a marker to identify if sugarcane genotypes are potentially resistant to brown rust [25,30,31]. Since 2009, this marker has also been utilized in the CP program to direct breeding strategies for brown rust resistance [29]. The information of Bru1 for parents (available on the CP database) used in this study is given in Table 3, Table 4, Table 5, and Table 6. In general, dominant parents of the top 20 crosses that had the highest brown rust ratings (Table 3), or female parents that had high probability to produce progeny with high brown rust ratings (Table 5) showed absence of Bru1. However, parents of the crosses with the lowest brown rust ratings (Table 4) or female parents that had a high probability to produce progeny with the lowest brown rust ratings (Table 6) did not always show the presence of Bru1. For instance, the cross (family) X09-1236 in 2012 had the lowest mean brown rust rating among 295 families, but Bru1 was not detected in its parents CP03-1160 and CP04-1105 (Table 4). These results suggested that other genes may also be involved in the either durable or non-durable resistance of sugarcane plants to brown rust in addition to Bru1 [23] as reported by other studies [32,33,34,35]. Racedo et al. [34] recently found that the predominant source of resistance to brown rust in the sugarcane breeding program at Tucuman, Argentina would be a resistance source independent of the Bru1 gene. They suggested that it is necessary to characterize both genetic diversity of the pathogen and the alternative sources of resistance in order to improve brown rust disease management.
2.8. Relationships between Brown and Orange Rusts
Genotypic variation in sugarcane orange rust resistance/tolerance at family and female parent levels for the Stage I clones of the CP program has been reported [19]. However, it is unclear if there are any correlations between brown rust and orange rust ratings across genotypes. Relationships between mean ratings of brown rust and orange rust across a large number of families, females or males were determined in this study (Figure 4) based on mean values of rust ratings across families, females, and males that had ≥15 progeny clones in the 2012 and 2013 Stage I of the CP program. Overall, there were the trends of negative relationships between brown and orange rust ratings of sugarcane progeny clones across families, females, and males. Most of the linear relationships were significant even through r2 values ranged 0.02 to 0.30 because of the large number of samples (Figure 4). These relationships were stronger in 2013 (p < 0.0001) than in 2012 (p = 0.211–0.0012) (Figure 4). The results suggested that sugarcane genotypes, which are resistant to brown rust disease, may be susceptible to orange rust. Therefore, caution must be taken for the reversible relationships between brown and orange rusts when using parents to make crosses for rust resistance. Some parents may produce progeny with resistance to one rust, but be highly susceptible to the other rust. For instance, the progeny of CP 88-1762 was resistant to brown rust (Table 6), but was highly susceptible to orange rust [19].
Multiple approaches have been and are being used for eliminating the negative effects of brown and orange rusts on yields, quality, and profits of sugarcane. These include standard breeding, molecular marker assisted selection, disease monitoring, adjustment of management practices, fungicide applications, and selection of rust resistant cultivars in the breed program by intensive rust screening [23]. Studies have shown that parents producing progeny with a high frequency of transgressive segregates for agronomic traits should provide the best opportunity for sugarcane breeders to select clones superior to their parents [36]. In addition to directly evaluating parental lines for brown rust and other diseases, therefore, parental evaluation based on their progeny performance for rust resistance and other agronomic traits in the early clonal stage of a sugarcane breeding program should help optimize parental selection and crossing combinations. Studies have suggested that family selection is effective in improving sugarcane populations in early selection stages [36,37,38,39], because it can identify those families that harbor the highest proportion of desirable clones and makes it possible to focus on selection for superior clones [36]. Availability of family data for rust diseases and agronomic traits helps sugarcane breeders improve crossing combinations for developing genotypes with resistance to rusts and high yields.
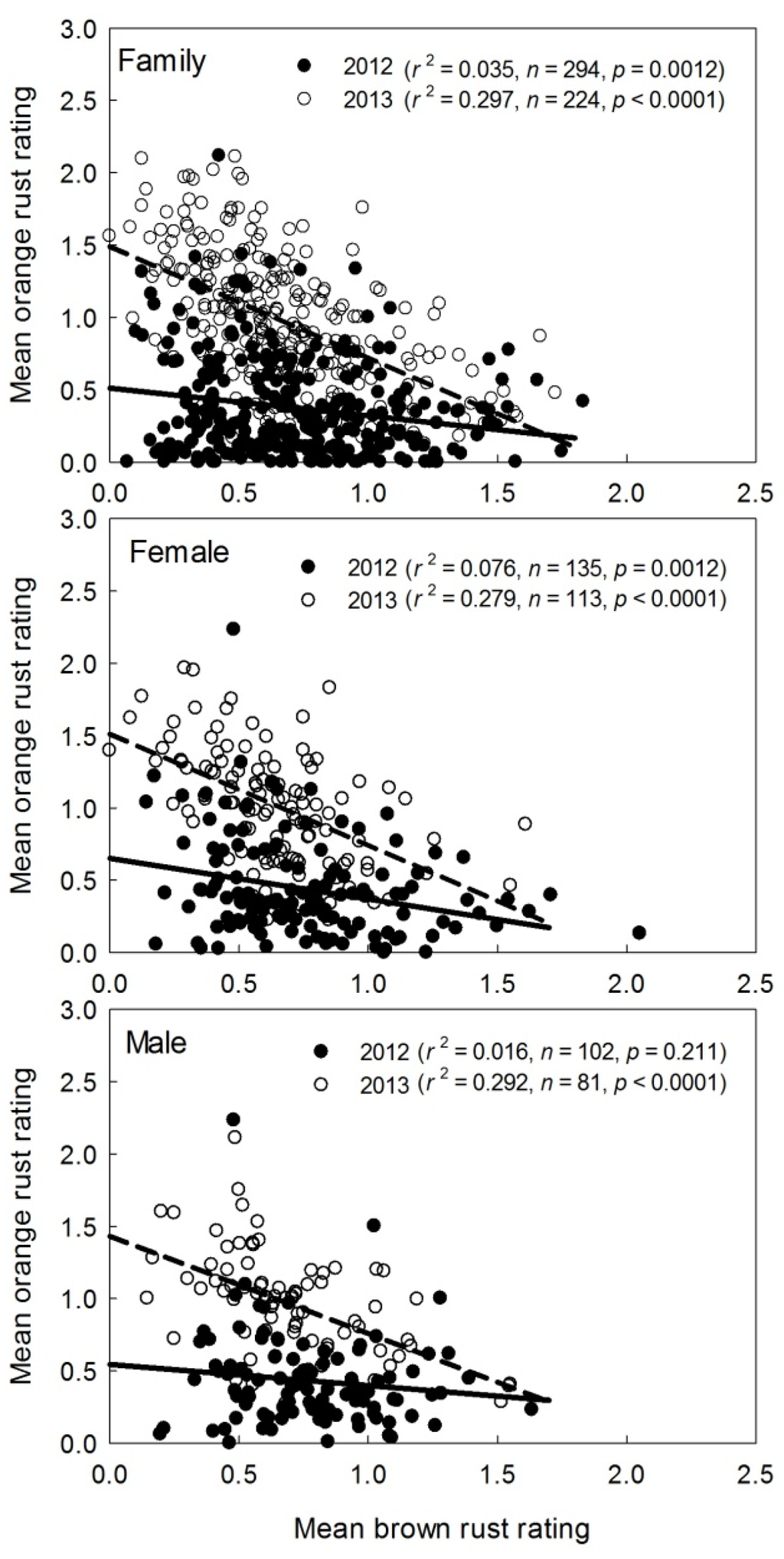
Figure 4.
Relationships between mean brown rust ratings and mean orange rust ratings of clones based on the crossing families, females, and males those had ≥15 progeny clones in the 2012 and 2013 Stage I of the Canal Point (CP) sugarcane breeding and cultivar development program.
3. Materials and Methods
3.1. Plant Culture
A total of 14,272 genotypes (stools, each stool came from a true seedling plant) in 2012 and 12,661 genotypes in 2013 were visually selected at mature from fields of the 2011 and 2012 seedling stages. One stalk was cut in each of all these selected genotypes and planted in single-row plots in the Stage I fields in January of 2012 and 2013, respectively. To facilitate stalk transport and planting, two to five stalks (each stalk came from a true seed) in the seedling fields were bundled and labeled by family (i.e., cross) prior to advancing them to Stage I [9]. These bundles were randomly distributed in the Stage-I fields. One stalk was placed in each plot and cut into two sections (each approximately 0.9 m long). The two sections were placed in the center of the plot as double lines of cane. The plot length was 2.4 m, with 1.5 m between-row spacing. There was a 1.5-m gap between adjacent clones within a row to allow scientists to recognize individual clones during disease rating, growth vigor evaluation, and selection. Four commercial cultivars, “CP 78-1628” [40], “CP 80-1743” [41], CP 88-1762, and CP 89-2143, were used as checks each year and randomly planted in a pattern of one check plot in approximately every 80 to 100 field plots. Approximately 40 replicated plots for each check cultivar were planted each year in the Stage-I fields. There was a 4.5 m alley every eight rows to facilitate field maintenance and genotype selection.
3.2. Data Collection of Brown Rust Disease
Although the peak season is in March to June for brown rust in most sugarcane production fields in Florida, the optimal plant age for brown rust development has been reported to be between 4 and 6 months [42,43]. The Stage-I planting dates of the CP program are routinely from late January to early February, therefore, brown rust and orange rust symptoms were evaluated in July to August in 2012 and 2013. Brown rust symptoms were differentiated from that of orange rust using the 20× pocket magnifying glass in the field, and positive identifications of inconclusive symptoms were made by observing spore morphology under a microscope in the laboratory. Rust diseases were recorded using a scale from 0 (no rust infection) to 4 (most severe rust infection) with intervals of 0.5. The 0 to 4 scale levels were defined as: 0 = no rust, 1 = one to a few pustules, 2 = patching presented, 3 = patching widespread up into the upper canopy with some lower leaf death, and 4 = massive amounts of rust pustules with heavy lower leaf death. In general, the plants with rust ratings of 0 to 1 were considered as resistant or tolerant, with rating 2 were considered as moderately susceptible, and with ratings of 3 and 4 were considered as susceptible and highly susceptible, respectively.
Additionally, all clones were visually evaluated for other diseases [leaf scald (Xanthomonas albilineans), smut (Sporisorium scitamineum), and mosaic] and plant vigor and agronomic traits [9] at the same time and in early September. A subjective plant vigor rating was determined for individual clones. All clones with high vigor rating or better than check cultivars in vigor rating, acceptable rust resistance (rust rating ≤1), and no other disease symptoms were further assessed for Brix value, an indicator of juice sucrose concentration, in early November. Approximately the 1,500 best clones with the largest vigor rate × Brix products were advanced to Stage II of the CP program. Recently, Zhao et al. [9] reported the details of vigor rating, Brix, and the Stage-I selection strategies as well as orange rust evaluation in the CP program [19]. Therefore, we mainly focused on brown rust on the basis of families and female parents in this paper. The information of sugarcane brown rust resistance gene (Bru1) for parents of the Stage-I clones was searched from a database of the CP program to investigate the relationships between Bru1 of parents and the rust resistance of their progeny. Three variables of mean rust rating, percentage of rust infection, and rust severity were used to determine variation of rust diseases among families (or female parents based on their progeny). These variables were defined and estimated using the following formulas [19]:
3.3. Data Analysis
The four check cultivars were randomly planted in the Stage-I field and each had approximately 40 replicated plots (Table 1) each year. Therefore, the MIXED procedure of SAS [44] was used to test the effects of check cultivar, year, and their interaction on the mean ratings of brown rust and on the % infection. If the hypothesis of equal means between the check cultivars was rejected by the F test, the trait means were separated with the least significant difference (LSD) at p = 0.05. The LSD values were calculated with the standard error (SE) values generated by the Diff option in the SAS MIXED procedure.
For the Stage-I clones, their parental combinations in the Crossing stage varied annually. Thus, the Stage-I data were analyzed separately for each year. Brown rust rating distributions were determined by pooling data across all clones within a year. Data of brown rust were analyzed for each family and female parent. Data of male parents were not analyzed because many of the progeny in the 2012 and 2013 Stage I were developed from poly crosses (i.e., where a female tassel received pollen from several different male tassels). Thus, the specific males were unknown for all poly crosses. For families and female parents that had ≥15 progeny clones planted in Stage I, the mean ratings and coefficients of variation (CVs) were determined for brown rust to assess variability. The means and CVs were calculated using PROC MEANS of SAS [44]. Then, the CV values of among-families (females) were obtained based on their means and the CVs of within-families (females) were estimated by averaged CVs of individual families (females). Coefficients of variation for brown rust in each family were calculated from the individual clonal values of rust rating from all clones within a family or female according to Zhao et al. [9]. The within-family or within-female CV for each parameter was estimated by calculating the overall mean CV of all individual family (female) CVs for that trait. The among-family CVs were estimated using the mean (rather than individual clone) values of each family (female). For example, to calculate the among-family CV of 20 families for brown rust, we would have calculated the CV based on the standard deviation (SD) and overall mean from the 20 mean rust values of each of the 20 families (i.e., CV = SD ÷ Mean × 100). The variability among- and within-families was described using respective CVs. The top 20 families that were most susceptible or most tolerant to brown rust in each year were further determined by ranking their mean rust ratings. The top 20 females in which their progeny were most susceptible or most tolerant to brown rust in each year were also determined based on mean ratings of brown rust.
4. Conclusions
Analyses of brown rust data collected from large numbers of individual clones in Stage I of the CP program in 2012 and 2013 revealed that there was great variation in rust ratings among genotypes and among families. Our data indicated that using brown rust rating data along with individual selection data on plant vigor and stalk juice Brix [9] and orange rust rating [19] could be useful to evaluate family performance in the first clonal stage of the CP program. The among- and within-family variability in these agronomic and disease resistance traits would improve our parental selection and optimize crosses among selected parents which should result in progeny with improved rust resistance and yield potential. Additional caution must be taken for the apparent inverse relationships between brown and orange rust resistance. These findings are useful not only for the local CP sugarcane breeding program, but also for others which import CP varieties around the world.
Acknowledgments
Authors thank Philip Aria for his valuable technical assistance and skilled data management. We also appreciate Tom Abbott and all other staff at the USDA-ARS Sugarcane Field Station for assistance on planting, field management, and harvest.
Author Contributions
Duli Zhao contributed to conception and design of the experiment, analyzed and interpreted data, wrote the manuscript and integrated all ideas and comments from other authors and peer reviewers. R. Wayne Davidson and Miguel Baltazar coordinated in rust data collection and interpretation. Jack C. Comstock made contributions to the original ideas of the experiment and disease data quality. Per McCord and Sushma Sood contributed to identification and interpretation of the brown rust resistance gene, Bru1.
Conflict of Interest
The authors declare no conflict of interest.
References
- USDA-NASS. Available online: http://usda.mannlib.cornell.edu/usda/current/CropValuSu/ CropValuSu-02-14-2014.pdf (accessed on 4 November 2014).
- Zhao, D.; Glaz, B.; Comstock, J.C. Sugarcane response to water deficit stress during early growth on organic and sand soils. Am. J. Agric. Biol. Sci. 2010, 5, 403–414. [Google Scholar] [CrossRef]
- Edmé, S.J.; Tai, P.Y.P.; Glaz, B.; Gilbert, R.A.; Miller, J.D.; Davidson, J.O.; Dunckelman, J.W.; Comstock, J.C. Registration of ‘CP 96-1252’ sugarcane. Crop Sci. 2005, 45, 423. [Google Scholar] [CrossRef]
- Gilbert, R.A.; Comstock, J.C.; Glaz, B.; Edmé, S.J.; Davidson, R.W.; Glynn, N.C.; Miller, J.D.; Tai, P.Y.P. Registration of ‘CP 00-1101’ sugarcane. J. Plant Reg. 2008, 2, 95–101. [Google Scholar] [CrossRef]
- Edmé, S.J.; Davidson, R.W.; Gilbert, R.A.; Comstock, J.C.; Glynn, N.C.; Glaz, B.; del Blanco, I.A.; Miller, J.D.; Tai, P.Y.P. Registration of ‘CP 01-1372’ sugarcane. J. Plant Reg. 2009, 3, 150–157. [Google Scholar] [CrossRef]
- Tai, P.Y.P.; Shine, J.M., Jr.; Deren, C.W.; Glaz, B.; Miller, J.D.; Comstock, J.C. Registration of ‘CP 88-1762’ sugarcane. Crop Sci. 1997, 37, 1388. [Google Scholar] [CrossRef]
- Glaz, B.; Miller, J.D.; Deren, C.W.; Tai, P.Y.P.; Shine, J.M., Jr.; Comstock, J.C. Registration of ‘CP 89-2143’ sugarcane. Crop Sci. 2000, 40, 577. [Google Scholar] [CrossRef]
- Rice, R.; Baucum, L.; Davidson, W. Sugarcane variety census: Florida 2014. Sugar J. 2015, 78, 8–16. [Google Scholar]
- Zhao, D.; Comstock, J.C.; Glaz, B.; Edmé, S.J.; Glynn, N.C.; Del Blanco, I.A.; Gilbert, R.A.; Davidson, R.W.; Chen, C.Y. Vigor rating and Brix for first clonal selection stage of the Canal Point sugarcane cultivar development program. J. Crop Improv. 2012, 26, 60–75. [Google Scholar] [CrossRef]
- Tai, P.Y.P.; Miller, J.D. Family performance at early stages of selection and frequency of superior clones from crosses among Canal Point cultivars of sugarcane. J. Am. Soc. Sugar Cane Technol. 1989, 9, 62–70. [Google Scholar]
- Edmé, S.J.; Miller, J.D.; Glaz, B.; Tai, P.Y.P.; Comstock, J.C. Genetic contributions to yield gains in the Florida sugarcane industry across 33 years. Crop Sci. 2005, 45, 92–97. [Google Scholar] [CrossRef]
- Raid, R.N.; Comstock, J.C. Common rust. In A Guide to Sugarcane Diseases; Rott, P., Bailey, R.A., Comstock, J.C., Croft, B.J., Saumtally, A.S., Eds.; Centre de Cooperation Internationale en Recherche Agronomique pour le Developpement (CIRAD) and International Society of Sugar Cane Technologists (ISSCT): Montpellier, France, 2000; pp. 85–89. [Google Scholar]
- Comstock, J.C.; Raid, R.N. Sugarcane common rust. In Current Trends in Sugarcane Pathology; Bhargava, K.S., Rao, G.P., Gillaspie, A.G., Jr., Upadhyaya, P.P., Filino, A.B., Agnihotri, V.P., Chen, C.T., Eds.; International Books and Periodicals Supply Service: Delhi, India, 1994; pp. 1–10. [Google Scholar]
- Comstock, J.C.; Shine, J.M., Jr.; Raid, R.N. Effect of rust on sugarcane growth and biomass. Plant Dis. 1992, 76, 175–177. [Google Scholar] [CrossRef]
- Comstock, J.C.; Shine, J.M., Jr.; Raid, R.N. Effect of early rust infection on subsequent sugarcane growth. Sugar Cane 1992, 4, 7–9. [Google Scholar]
- Hoy, J. Brown rust is coming back—What can you do to prevent loss? Sugar Bull. 2012, 90, 19–21. [Google Scholar]
- Hoy, J.W.; Hollier, C.A. Effect of brown rust on yield of sugarcane in Louisiana. Plant Dis. 2009, 93, 1171–1174. [Google Scholar] [CrossRef]
- Sood, S.G.; Comstock, J.C.; Glynn, N.C. Leaf whorl inoculation method for screening sugarcane rust resistance. Plant Dis. 2009, 93, 1335–1340. [Google Scholar] [CrossRef]
- Zhao, D.; Davidson, R.W.; Baltazar, M.; Comstock, J.C. Field evaluation of sugarcane orange rust for first clonal stage of the CP cultivar development program. Am. J. Agric. Biol. Sci. 2015, 10, 1–11. [Google Scholar]
- Rott, P.; Sood, S.; Comstock, J.C.; Raid, R.N.; Glynn, N.C.; Gilbert, R.A.; Sandhu, H.S. Sugarcane orange rust. EDIS. Available online: http://edis.ifas.ufl.edu/sc099 (accessed on 20 July 2015).
- Jiang, D.K. Chemical control of sugarcane rust P. melanocephala. Rep. Taiwan Sugar Res. Inst. 1985, 108, 25–34. [Google Scholar]
- Machado, G.R., Jr. The spread of CP varieties, Canal Point, Florida, USA in other countries. Sugar J. 2013, 77, 20–21. [Google Scholar]
- Zhao, D.; Comstock, J.C.; Glaz, B.; Edmé, S.J.; Davidson, R.W.; Gilbert, R.A.; Glynn, N.C.; Sood, S.; Sandhu, H.; McMorkle, K.; et al. Registration of ‘CP 05-1526’ sugarcane. J. Plant Reg. 2013, 7, 305–311. [Google Scholar] [CrossRef]
- Wang, L.P.; Jackson, P.A.; Lu, X.; Fan, Y.H.; Foreman, J.W.; Chen, X.K.; Deng, H.H.; Fu, C.; Ma, L.; Aitken, K.S. Evaluation of sugarcane × Saccharum spontaneum progeny for biomass composition and yield components. Crop Sci. 2008, 48, 951–961. [Google Scholar] [CrossRef]
- Asnaghi, C.; Roques, D.; Ruffel, S.; Kaye, C.; Hoarau, J.Y.; Telismart, H.; Girard, J.C.; Raboin, L.M.; Risterucci, A.M.; Grivet, L.; D’Hont, A. Targeted mapping of a sugarcane rust resistance gene Bru1 using bulked segregant analysis and AFLP markers. Theor. Appl. Genet. 2004, 108, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Virtudazo, E.V.; Nakamura, H.; Kakishima, M. Phylogenetic analysis of sugarcane rusts based on sequences of ITS, 5.8 S rDNA and D1/D2 regions of LSU rDNA. J. Gen. Plant Pathol. 2001, 67, 28–36. [Google Scholar] [CrossRef]
- Dixon, L.J.; Castlebury, L.A.; Aime, M.C.; Glynn, N.C.; Comstock, J.C. Phylogenetic relationships of sugarcane rust fungi. Mycol. Progress. 2010, 9, 459–468. [Google Scholar] [CrossRef]
- Costet, L.; Cunff, L.L.; Royaert, S.; Raboin, L.-M.; Hervouet, C.; Toubi, L.; Telismart, H.; Garsmeur, O.; Rousselle, Y.; Pauquet, J.; Nibouche, S.; Glaszmann, J.-C.; Hoarau, J.-Y.; D’Hont, A. Haplotype structure around Bru1 reveals a narrow genetic basis for brown rust resistance in modern sugarcane cultivars. Theor. Appl. Genet. 2012, 125, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Glynn, N.C.; Laborde, C.; Davidson, R.W.; Irey, M.S.; Glaz, B.; D’Hont, A.; Comstock, J.C. Utilization of a major brown rust resistance gen in sugarcane breeding. Mol. Breeding. 2013, 31, 323–331. [Google Scholar] [CrossRef]
- Asnaghi, C.; Paulet, F.; Kaye, C.; Grivet, L.; Glaszmann, J.C.; D’Hont, A. Application of synteny across the Poaceae to determine the map location of a rust resistance gene of sugarcane. Theor. Appl. Genet. 2000, 10, 962–969. [Google Scholar] [CrossRef]
- Cunff, L.L.; Garsmeur, O.; Raboin, L.M.; Pauquet, J.; Telismart, H.; Selvi, A.; Grivet, L.; Philippe, R.; Begum, D.; Deu, M.; Costet, L.; Wing, R.; Glaszmann, J.C.; D’Hont, A. Diploid/polyploid syntenic shuttle mapping and haplotype-specific chromosome walking toward a rust resistance gene (Bru1) in highly polyploid sugarcane (2n ~ 12x ~ 115). Genetics 2008, 180, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Raboin, L.-M.; Oliveira, K.M.; Lecunff, L.; Telismart, H.; Roques, D.; Butterfield, M.; Hoarau, J.-Y.; D‘Hont, A. Genetic mapping in sugarcane, a high polyploid, using bi-parental progeny: identification of a gene controlling stalk colour and a new rust resistance gene. Theor. Appl. Genet. 2006, 112, 1382–1391. [Google Scholar] [CrossRef] [PubMed]
- Hoy, J.W.; Avellaneda, M.C.; Bombecini, J. Variability in Puccinia melanocephala pathogenicity and resistance in sugarcane cultivars. Plant Dis. 2014, 98, 1728–1732. [Google Scholar] [CrossRef]
- Racedo, J.; Perera, M.F.; Bertani, R.; Funes, C.; Victoria González, V.; Cuenya, M.I.; D’Hont, A.; Welin, B.; Castagnaro, A.P. Bru1 gene and potential alternative sources of resistance to sugarcane brown rust disease. Euphytica. 2013, 191, 429–436. [Google Scholar] [CrossRef]
- Parco, A.S.; Avellaneda, M.C.; Hale, A.H.; Hoy, J.W.; Kimbeng, C.A.; Pontif, M.J.; Gravois, K.A.; Baisakh, N. Frequency and distribution of the brown rust resistance gene Bru1 and implications for the Louisiana sugarcane breeding programme. Plant Breeding 2014, 133, 654–659. [Google Scholar] [CrossRef]
- Shanthi, R.M.; Bhagyalakshmi, K.V.; Hemaprabha, G.; Alarmelu, S.; Nagarajan, R. Relative performance of the sugarcane families in early selection stages. Sugar Tech. 2008, 10, 114–118. [Google Scholar] [CrossRef]
- Chang, Y.S.; Milligan, S.B. Estimating the potential of sugarcane families to produce elite genotypes using univariate cross prediction methods. Theor. Appl. Genet. 1992, 84, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Cox, M.C.; Hogarth, D.M. The effectiveness of family selection in early stages of sugarcane improvement program. In Focused Plant Improvement: Towards Responsible and Sustainable Agriculture; Proceedings Tenth Australian Plant Breeding Conference, Gold Coast, Australia, April 1993; Imrie, B.C., Hacker, J.B., Eds.; Vol. 2, pp. 53–54.
- McRae, T.A.; Hogarth, D.M.; Foreman, J.F.; Braithwaite, M. Selection sugarcane families in the Burdekin district. In Focused Plant Improvement: Towards Responsible and Sustainable Agriculture; Proceedings Tenth Australian Plant Breeding Conference, Gold Coast, Australia, April 1993; Imrie, B.C., Hacker, J.B., Eds.; Vol.1, pp. 77–82.
- Tai, P.Y.P.; Miller, J.D.; Glaz, B.; Deren, C.W.; Shine, J.M. Registration of ‘CP 78-1628’ sugarcane. Crop Sci. 1991, 31, 236. [Google Scholar] [CrossRef]
- Deren, C.W.; Glaz, B.; Tai, P.Y.P.; Miller, J.D.; Shine, J.M., Jr. Registration of ‘CP 80-1743’ sugarcane. Crop Sci. 1991, 31, 235–236. [Google Scholar] [CrossRef]
- Comstock, J.C.; Ferreira, S.A. Sugarcane rust: Factors affecting infection and symptoms development. Proc. XIX Int. Soc. Sugar Cane Technol. (ISSCT) Congress. 1986, 19, 402–410. [Google Scholar]
- Victoria, J.; Moreno, C.; Cassalett, C. Genotype-environment interaction and its effect on sugar cane rust incidence. Sugar Cane 1990, 4, 13–17. [Google Scholar]
- SAS. SAS System for Windows Release 9.3, SAS Inst.: Cary, NC, USA, 2010.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).