Abstract
Root architecture was determined together with shoot parameters under well watered and drought conditions in the field in three soybean cultivars (A5409RG, Jackson and Prima 2000). Morphology parameters were used to classify the cultivars into different root phenotypes that could be important in conferring drought tolerance traits. A5409RG is a drought-sensitive cultivar with a shallow root phenotype and a root angle of <40°. In contrast, Jackson is a drought-escaping cultivar. It has a deep rooting phenotype with a root angle of >60°. Prima 2000 is an intermediate drought-tolerant cultivar with a root angle of 40°–60°. It has an intermediate root phenotype. Prima 2000 was the best performing cultivar under drought stress, having the greatest shoot biomass and grain yield under limited water availability. It had abundant root nodules even under drought conditions. A positive correlation was observed between nodule size, above-ground biomass and seed yield under well-watered and drought conditions. These findings demonstrate that root system phenotyping using markers that are easy-to-apply under field conditions can be used to determine genotypic differences in drought tolerance in soybean. The strong association between root and nodule parameters and whole plant productivity demonstrates the potential application of simple root phenotypic markers in screening for drought tolerance in soybean.
1. Introduction
Soybean is a vital source of vegetable protein for food and animal feed world-wide. It is predicted to become a major crop in Africa [1]. Current breeding strategies for crops such as soybean seek to identify new varieties with higher grain yields and improved nitrogen- and water-use efficiencies. Over the past decade, the use of phenomic tools to study plant phenotypes under laboratory and field conditions has attracted increasing attention, particularly with regard to making better use of “omic” approaches [1]. While considerable progress has been made recently in the development of imaging tools for the rapid phenotyping of shoots, the associated data processing and bio-informatic requirements remain challenging [2,3]. Moreover, despite intensive efforts, systems that are directly applicable in the field remain largely at the prototype stage [2,3]. Therefore, classic manual plant phenotyping techniques remain valuable tools for plant breeding [4]. Such techniques are particularly important in many developing countries, particularly in Africa, which lack the essential underpinning infrastructure required to apply more sophisticated “phenomic” approaches.
Shoot morphology characteristics are commonly used for phenotyping [5]. Shoot characteristics are generally easy to assess under field conditions, where they can often be determined simply by visual examination. Leaf movement [6] leaf flagging and shedding [7] and leaf area are commonly used in screening for drought tolerance. Decreases in leaf size and leaf expansion can also be used as measures of adaptation to drought [8]. While leaf movement traits are rarely used in selection for drought tolerance, because they are often difficult to quantify, leaf rolling and drying have been used more widely alongside molecular marker-assisted selection in crops such as in rice [9,10]. In general, plant breeders are often reluctant to apply physiological screening techniques extensively because they are regarded as expensive, time-consuming and more difficult to apply.
Roots are the first organs to perceive and respond to drought but below-ground phenotyping by screening of the root systems is rarely undertaken, particularly under field conditions. The distribution of roots, particularly those that can penetrate deeper in the soil, plays a crucial role in determining the ability of plants to capture key resources such as water and mobile nutrients like nitrate. Root architecture therefore has a profound effect on the growth and yield of crop plants. Studies on the responses of root architecture to drought have been performed on soil or on solid support media, such as hydroponics or on agar plates [11,12,13,14]. However, relatively few studies have been performed to date using root parameters to select for enhanced nitrogen use efficiency (NUE) or improved water use efficiency (WUE) in modern crop varieties.
The current inability to measure root architecture under field conditions is a major impediment to the effective application of current “phenomic” technologies in marker-assisted selection for improved root system traits. Classic root phenotyping approaches in the field, such as the analysis of soil cores and applying standard excavation techniques to determine root depth, root branching densities and root angle are still accepted as the best methods [13,15]. However, such approaches do not reveal the finer details of root architecture, anatomy (e.g., root hair densities) or function (e.g., nutrient uptake). Therefore, identifying crops with improved root architecture characteristics remains a major challenge to current plant biology, together with the development of appropriate technologies for the study of root growth in the soil, particularly under field conditions. Root architecture is greatly modified under drought stresses, which favor the production of greater numbers of longer lateral roots are root hairs to increase the total surface area for better water absorption [16]. Together with strategies that limit water loss, such as stomatal closure, leaf rolling and leaf abscission, the increase in root mass particularly deeper in the soil results in an improved plant water status that is required to support biomass production and yield. Superior root phenotypes are currently considered to be key to improved drought tolerance characteristics that allow better plant performance through more efficient water uptake in crops such as soybean [17,18,19]. Genetic variability has been demonstrated in soybean root architecture and morphology, including traits such as root angle, root diameter, length, surface area and depth [20,21]. Deeper root systems with greater root densities, particularly at depth are considered to allow better extraction of soil water [20,22]. To our knowledge, field phenotyping of root and nodule characteristics has not been widely used to select for superior root systems in soybean. The following studies were therefore undertaken to characterize drought-induced changes in soybean root architecture under field conditions in order to determine whether root traits can be linked with plant performance under drought, with a view to identifying drought-tolerant soybean cultivars. Root systems architecture was characterized in three soybean cultivars that differ in drought tolerance, under well-watered and drought conditions. The three soybean cultivars used in the followings studies are the glyphosate-resistant cultivar A5409RG Prima 2000, which is a commercial soybean cultivar grown in South Africa, and Jackson, a nominally drought-tolerant cultivar [23]. In a previous study under controlled environment conditions, Prima 2000 was shown to have better shoot growth than the other two cultivars under well watered and drought conditions [24].
2. Materials and Methods
2.1. Plant Material and Experimental Procedure
Field experiments were conducted during the 2010 cropping season (February to May 2010) at the Ukulima Root Biology Center (URBC), operated by Pennsylvania State University in Limpopo Province, South Africa (24°32.002′S, 28°07.427′E and 1237 m above sea level). The soil texture of the field was sandy according to USDA’s (2011) soil classification [25]. Soybean (Glycine max L. Merr.) cultivars grown were: A5409RG, a glyphosate resistant transgenic cultivar; Prima 2000, a commercial cultivar in South Africa; and Jackson, which is considered as a drought-tolerant soybean cultivar [24]. Before commencement of the experiment, a soil analysis for both macro- and micro-nutrients was conducted (Alpha Agric PLC soil analysis laboratory, Nylstroom, South Africa). Based on the analysis, 4 kg/ha boron, 1 kg/ha zinc sulfate and 25 kg/ha potassium sulfate were applied. Before land preparation, 3 L/ha of Roundup (Monsanto, Randburg, South Africa), a systemic, broad-spectrum herbicide, was applied to kill all weeds. The pre-emergence herbicides Unimoc EC 800 mL/ha (Meridian Agrochemical Company (Pty) Ltd., Germiston, South Africa) and Imazethaphyr 400 mL/ha (American Cyanamid Co., Wayne, NJ., US) were also sprayed before planting to control both grasses and broadleaf weeds. After planting, frequent hand-weeding was performed as needed. The nematicide oxalate 3 L/ha (SinoHarvest Agrochemical Manufacturer, Shanghai, China) was applied to prevent nematode infestation.
2.2. Planting and Experimental Layout
Experiments were conducted in a randomized complete block design with two water regimes (well-watered and water-limited drought block). Each cultivar was planted in five rows with spacing of 75 cm between rows and 10 cm between plants. Row length was 4 m and plot size was 15 m2. The central three rows were used for data collection, while the two outer rows served as borders. One seed per hole was planted at 5 cm depth using a custom built jab planter (modeled on the Alamco planter [26]) which is specially designed to plant with uniform depth.
Before initiation of drought stress, all plants were grown with adequate water supply by applying 8 mm water/day using pivot sprinkler irrigation. Plants were exposed to drought by withholding the irrigation 30 days after planting when plants were at the 3rd trifoliate leaf (V3) development stage. Plants were then grown for further 28 days without watering but were exposed to three days of rain with a total of 34 mm of rain at 7th, 19th and 26th days after drought exposure.
2.3. Soil Moisture Content
Volumetric water content of the soil was measured at the beginning of the drought experiment followed by four measurements every fifth day. Soil was sampled using a steel corer lined with a plastic tube (60 cm length and 42 mm diameter) (Giddings Machine Company Inc., Englewood, CO, USA). For sampling, four samples were taken each time from the different irrigation regimes. After determination of wet soil weight, the soil was oven-dried for 48 h at 105 °C. The volumetric water content (θv) was calculated with the formula [27]:
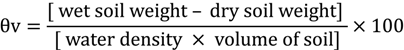
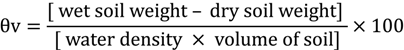
2.4. Root Architecture
Eighteen plants per cultivar (six plants per replication) for each water regime were sampled applying the root excavation technique [13]. Although it is difficult to capture all very fine lateral roots, this method is pertinent to phenotype root architecture. This technique involves acquiring the root with the whole plant and after careful washing the architectural traits are recorded. The root angle was determined by laying the whole uprooted root on an 180° protractor sketched board with the stem at 0°. The average lateral root angle on both sides was measured as root growth angle. Further root architectural measurements were carried out for the two main soybean root types (primary or tap root, and lateral or secondary root) after one month of drought exposure. Tap and lateral root thickness (diameter) was determined by multiple measurements 2 cm away from the origin of roots with an electronic digital caliper 5HA 1890 Model (Omni-Techelectronic Co. Limited, Hong Kong, China). The branching density of both tap and lateral root was determined by counting the branched roots emerged from lateral and tap roots emerging within a 2 cm root segment of either the tap root or of three randomly selected lateral roots.
2.5. Root Morphology Analysis
After one month exposure of plants to drought, three soil cores per plot were taken from each water regime for analysis of the root morphology. The steel corer lined with a plastic tube (60 cm length and 42 mm diameter) (Giddings Machine Company Inc., Englewood, CO, USA) was driven into the soil between two plants. Upon extracting the core, roots were washed out of the soil, scanned with a root scanner (Epson Perfection V 700 Photo/V 750 Pro, Seiko Epson Corporation, Nagano-ken, Japan) and the root image was analyzed with the winRHIZO 2008a program (Regent Instruments Canada Inc., Quebec, QC, Canada).
2.6. Nodule Size and Abundance Measurement
Eighteen plants per cultivar were used for root phenotyping including nodule parameters. Nodule size was rated from 1–6 by placing the multiple nodules on a board with a sketch of the diameter of nodules, i.e., 1 mm diameter scoring 1; 2 mm scoring 2 and ultimately 6 cm scoring 6. The abundance of nodules was also rated from 1–10; depending on the number of nodules with 1–10 nodules scoring 1, 11–20 scoring 2 and finally 91–100 nodules scoring 10.
2.7. Chlorophyll Content
Six plants per plot for each cultivar were sampled at the beginning and at the end of the drought experiment and chlorophyll analysis was carried out with the central, same age leaflet of the top fully expanded trifoliate leaf. Leaf chlorophyll content was measured non-destructively with the Chlorophyll Meter SPAD-502 (Spectrum Technologies Inc., Aurora, IL, USA) and the technique can be directly applied in the field. The technique instantly measures the chlorophyll content of leaves by simply clamping the meter over the leaf and obtaining a chlorophyll content reading on a scale of 0.0–99.9 [28]. The average of three individual SPAD chlorophyll meter readings (SCMR) was taken for determination of the chlorophyll content.
2.8. Biomass and Seed Yield Measurement
At flowering and mid-pod filling stage, six plants per plot were harvested for each watering regime for root phenotyping and biomass determination. Plant parts were divided into leaves, stems, and pods (at mid-pod filling stage). Dry mass of plant parts was determined after oven drying at 60 °C for 48 h (TERM-O-MAT LABOTEC, Johannesburg, South Africa). For seed yield determination of each plot, 2 rows with 3 m length (2.25 m2 area) were used and discarding a border of 0.5 m on both ends of the rows.
Yield per plot was determined by adjusting seeds to similar moisture content (10%) according to Silva and Braga [29] for common bean and Fischer and Maurer [30] for wheat. The yield per hectare (kg/ha) was finally determined based on the result obtained for yield/plot. Further, all plants from one row (3 m length) were counted and harvested independently. The seeds were manually threshed to separate the pod wall and seed. Samples were dried in an oven at 60 °C for 48 h and the dry mass was determined. pod harvest index (PHI) was calculated with the formula:
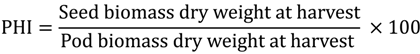
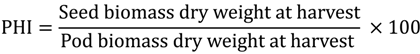
2.9. Statistical Analysis
Data were analyzed using JMP® 9.0 statistical package (SAS Institute Inc., Cary, NC, USA). Analysis of variance was used to determine the significance level, and treatment comparison to differentiate the cultivars for the measured traits using LSmeans Student’s t-test.
3. Results
3.1. Soil Moisture Content
We determined the decline in soil water content after switching off the sprinkler nozzles over the drought area with plants exposed to natural drought conditions. The measured volumetric soil water content of only 14% in our sandy soil experimental block is in agreement with the value previously reported as field capacity for this type of soil [30]. The volumetric water content of the well-watered block supplied by a sprinkler did not change significantly throughout the experimental period (Figure 1). In the drought block, which received no water, the volumetric water content gradually declined and was 7.5% after 4 weeks of drought exposure (Figure 1).
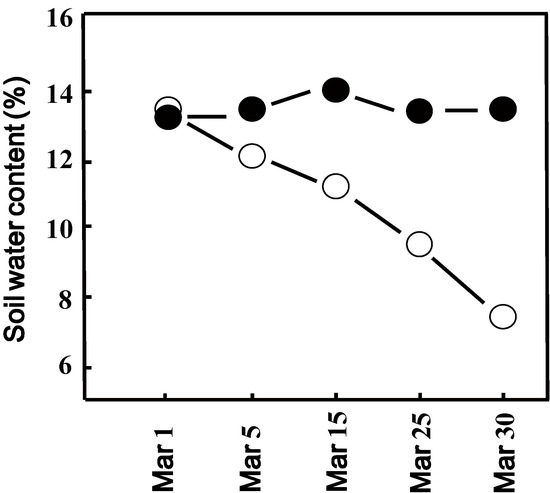
Figure 1.
Soil volumetric water content (%) of watered (closed circles) and drought (open circles) exposed experimental block. Values represent the mean of four individual soil samples per plot. Deviation of individual values was less than 5%.
Figure 1.
Soil volumetric water content (%) of watered (closed circles) and drought (open circles) exposed experimental block. Values represent the mean of four individual soil samples per plot. Deviation of individual values was less than 5%.
3.2. Below-Ground Phenotypic Analysis
3.2.1. Root Architectural Traits
To study the response of roots in order to quantify root phenotypes of the three cultivars under well-watered and drought conditions, we first measured the root angle, root diameter and also root branching of field-grown plants. Although measurement of the root angle can be easily performed directly in the field, the technique requires extracting the root to examine the root system (Figure 2 and Figure 3). The root angle was significantly different (p < 0.01) among the three cultivars, regardless of the water regime applied, with 24°, 66°, and 53° for A5409RG (shallow root system), Jackson (deep root system), and Prima 2000 (intermediate root system), respectively (Table 1A; Figure 3A,B). The drought-tolerant cultivar Jackson had a significantly larger (p < 0.05) tap root diameter than the two other cultivars under both water regimes (Table 1A). In contrast, the lateral root diameter (thickness), as measured with an electronic digital caliper, was significantly larger (p < 0.05) in roots of cultivar A-5409RG than in Jackson and Prima 2000 regardless of the water regime (Table 1A). Drought did not greatly change root angle, tap and lateral root diameter in the three cultivars with the highest increase in root angle (10.7%) in A5409RG and the highest decrease in lateral root diameter (16%) in cultivar Jackson due to drought (Table 1A). Drought treatment significantly increased (p < 0.05) the root branching density, which was determined by counting the branched roots of both tap and lateral roots. Branching density differed among cultivars and tap root branching increased under drought by 53% in cultivar Jackson and 57% in cultivar Prima 2000 with an even higher increase in lateral root branching of 76% in Jackson and 67% in Prima 2000. This branching increase in both cultivars was significantly greater (p < 0.05) than for the more drought-sensitive cultivar A5409RG, which had only a 29% and 42% increase in tap root branching and lateral root branching, respectively (Table 1B).
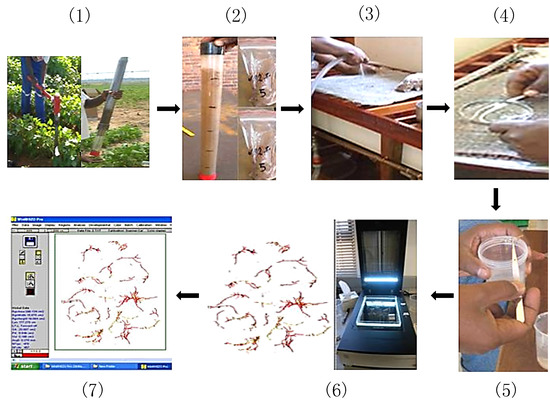
Figure 2.
Steps used in the analysis of root morphology. Step 1: soil coring; Step 2: dividing soil samples; Step 3: washing of roots; Step 4: separating roots from soil; Step 5: preserving roots in 25% ethanol; Step 6: scanning roots using root scanner Epson Perfection and step 7 analyzing the scanned root images using Winrhizo software.
Figure 2.
Steps used in the analysis of root morphology. Step 1: soil coring; Step 2: dividing soil samples; Step 3: washing of roots; Step 4: separating roots from soil; Step 5: preserving roots in 25% ethanol; Step 6: scanning roots using root scanner Epson Perfection and step 7 analyzing the scanned root images using Winrhizo software.
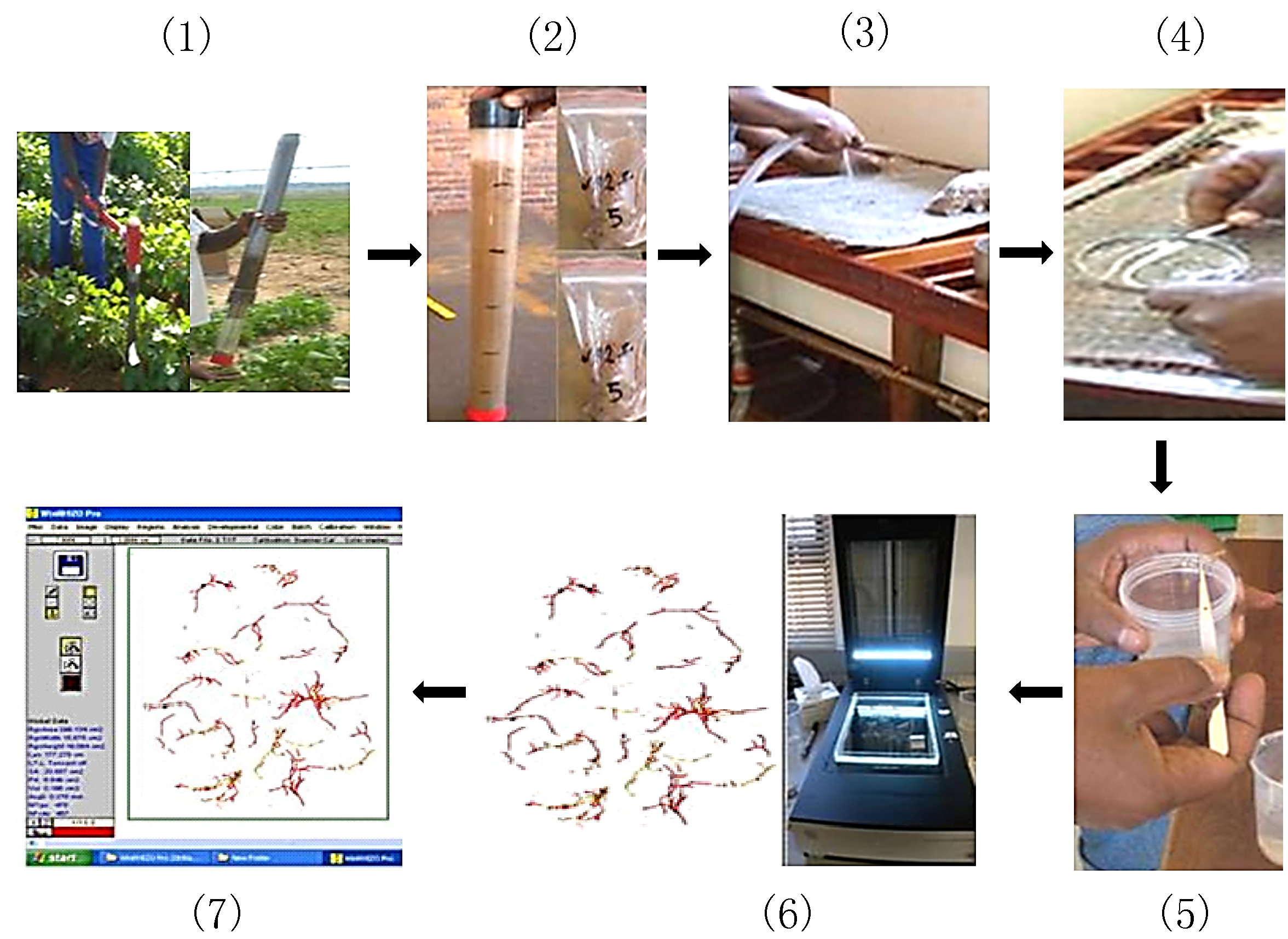

Figure 3.
Images of the isolated and cleaned soybean roots together with root architectural angle measurement with a 180° protractor sketched board. (A) Root angle measurement of cultivar Prima 2000 representing an intermediate root angle genotype and (B) root angle measurement of cultivar A5409RG, which represents the shallow root angle genotype.
Figure 3.
Images of the isolated and cleaned soybean roots together with root architectural angle measurement with a 180° protractor sketched board. (A) Root angle measurement of cultivar Prima 2000 representing an intermediate root angle genotype and (B) root angle measurement of cultivar A5409RG, which represents the shallow root angle genotype.

Table 1.
A comparison of root traits in three field grown soybean cultivars: A5409RG, Jackson and Prima 2000, under well-watered conditions and after one of month drought stress.

(A) Root angle, tap and lateral root diameter (thickness)
| Cultivar | Root Growth Angle | Tap Root Diameter (mm) | Lateral Root Diameter (mm) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| WW | D | (%) | WW | D | (%) | WW | D | (%) | |
| A-5409RG | 23.3 ± 1.6c | 25.8 ± 2.3c | 10.7 | 2.5 ± 0.1c | 2.3 ± 0.4c | −8.0 | 4.1 ± 0.3a | 4.2 ± 0.1a | 2.4 |
| Jackson | 64.2 ± 2.3a | 68.3 ± 1.6a | 6.3 | 4.5 ± 0.5a | 4.3 ± 0.2a | −4.4 | 2.5 ± 0.2b | 2.1 ± 1.1b | −16.0 |
| Prima 2000 | 51.7 ± 3.3b | 54.2 ± 2.9b | 4.8 | 3.8 ± 0.1b | 3.6 ± 0.5b | −5.2 | 2.5 ± 0.1b | 2.2 ± 0.2b | −12.0 |
| Significance | ** | ** | ** | ** | ** | ** | |||

(B) Tap and lateral root branching densities
| Cultivar | Tap Root Branching Density | Lateral Root Branching Density | ||||
|---|---|---|---|---|---|---|
| WW | D | (%) | WW | D | (%) | |
| A5409RG | 4.4 ± 0.3 | 5.7 ± 0.4b | 29 | 3.4 ± 0.3b | 4.8 ± 0.3b | 42 |
| Jackson | 5.0 ± 0.2 | 7.7 ± 0.6a | 53 | 4.2 ± 0.2ab | 7.3 ± 0.2a | 76 |
| Prima 2000 | 4.7 ± 0.3 | 7.3 ± 0.3a | 57 | 4.5 ± 0.4a | 7.5 ± 0.2a | 67 |
| Significance | ns | * | ns | ** | ||
WW represents well-watered and D drought treated cultivars. Significance level was determined using ANOVA (** p < 0.01, * p < 0.05, and ns p > 0.05) and difference between treatment means was determined using the LS means Student’s t-test. Means followed by the same letter within the column are not significantly different. The result is the mean ± SEM of six plants per plot exposed to 28 days of drought (%) represents percentage change of well-watered samples due to drought treatment.
3.2.2. Root Morphology
We measured root morphology reflected in parameters of root length, root area and volume as well as number of root tips and average root diameter as easily measurable phenotypic characteristics for plant performance. Measurement of these characteristics required excavating the plant from the soil for examination. Under well-watered conditions, we found no significant difference (p > 0.05) among the three cultivars. The root morphology phenotype, except for average root diameter, changed however when plants were exposed to drought. A considerable decrease in root length, root surface and root volume was observed in the drought-sensitive cultivar A5409RG (Table 2). In contrast, root length, root surface area, root volume and also number of root tips cultivars greatly increased under drought in Jackson and Prima 2000 (Table 2). However, Prima 2000 outperformed the other two cultivars with the longest roots, higher root area and volume, as well as the highest number of root tips and highest root diameter under drought (Table 2), which has previously already been reported by our group as a result from a preliminary study [31].

Table 2.
A comparison of root morphology traits in three soybean cultivars (A5409RG, Jackson and Prima 2000) under well-watered conditions and drought.
| Cultivars | Root Length (cm) | Surface Area (cm2) | Root Volume (cm3) | Root Tip Number | Root Diameter (mm) |
|---|---|---|---|---|---|
| Well-watered | |||||
| A-5409RG | 56.6 ± 10.0 | 8.3 ± 1.3 | 0.10 ± 0.01 | 180.6 ± 21.4 | 0.49 ± 0.03 |
| Jackson | 56.9 ± 11.7 | 8.0 ± 1.5 | 0.09 ± 0.02 | 190.5 ± 24.9 | 0.46 ± 0.04 |
| Prima 2000 | 51.5 ± 11.4 | 6.5 ± 1.4 | 0.07 ± 0.02 | 168.7 ± 24.2 | 0.43 ± 0.04 |
| Significance | ns | ns | ns | ns | ns |
| Drought | |||||
| A-5409RG | 41.0 ± 0.4b | 4.7 ± 2.31b | 0.05 ± 0.03b | 177.8 ± 58.1b | 0.40 ± 0.07 |
| Jackson | 98.6 ± 0.4a | 11.0 ± 1.8a | 0.12 ± 0.02a | 301.4 ± 44.1ab | 0.48 ± 0.05 |
| Prima 2000 | 120.5 ± 0.4a | 15.4 ± 1.8a | 0.16 ± 0.02a | 377.8 ± 45.9a | 0.54 ± 0.05 |
| Significance | ** | ** | ** | * | ns |
Significance level was determined using ANOVA (** p < 0.01, * p < 0.05, and ns p > 0.05) and difference between treatment means was determined using the LS means Student’s t-test. Means followed by the same letter within the column are not significantly different. The result is the mean ± SEM of six replicates for each core samples, and for each treatment using soil cores up to 60 cm soil depth. Data were obtained using the procedures outlined in Figure 2.
3.2.3. Root Nodules
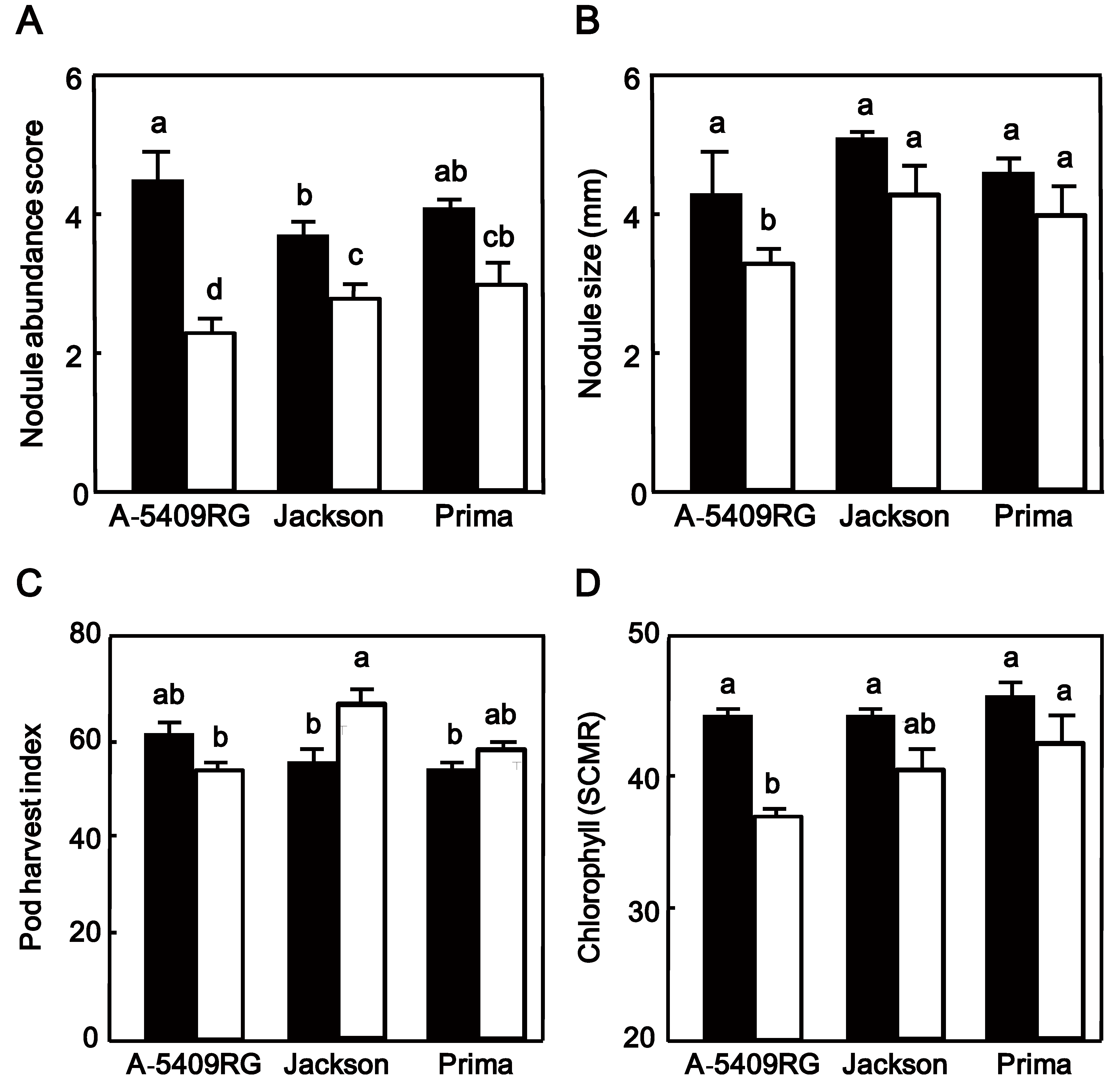
The cultivar A-5409RG had under well-watered conditions the highest nodule abundance of all three cultivars (Figure 4A), but under drought cultivar Prima had more nodules than the two other cultivars (Figure 4A). Nodule size of the three cultivars was further not significantly different under well-watered conditions (Figure 4B) but differed among cultivars in drought condition with Jackson, known to have a prolonged N2 fixation during the early stages of drought [17,26], and also Prima 2000 having bigger nodules than A-5409RG (Figure 4B).
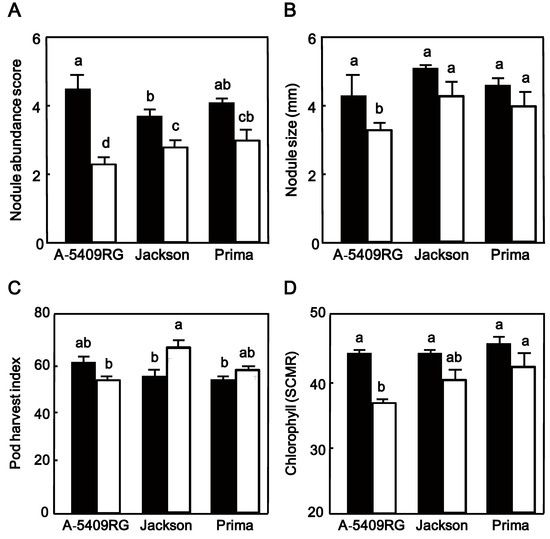
Figure 4.
(A) Nodule abundance; (B) nodule size; (C) pod harvest index and (D) leaf chlorophyll content (SCMR) of plants from field-grown soybean cultivarsA-5409RG Jackson and Prima 2000 either exposed to watered (closed bars) or drought (open bars) conditions. Data shown for nodule abundance and size represent the mean ± SEM of six plants per plot after exposure for one month to either well-watered or drought conditions. Data shown for pod harvest index are the mean ± SEM of plants from a single row per plot and data for chlorophyll represent the mean ± SEM of six plants per plot with chlorophyll measurements replicated three times. Means followed by the same letter on bars are not-significantly different tested by LS means Student’s t-test (p = 0.05).
Figure 4.
(A) Nodule abundance; (B) nodule size; (C) pod harvest index and (D) leaf chlorophyll content (SCMR) of plants from field-grown soybean cultivarsA-5409RG Jackson and Prima 2000 either exposed to watered (closed bars) or drought (open bars) conditions. Data shown for nodule abundance and size represent the mean ± SEM of six plants per plot after exposure for one month to either well-watered or drought conditions. Data shown for pod harvest index are the mean ± SEM of plants from a single row per plot and data for chlorophyll represent the mean ± SEM of six plants per plot with chlorophyll measurements replicated three times. Means followed by the same letter on bars are not-significantly different tested by LS means Student’s t-test (p = 0.05).
3.3. Above-Ground Phenotypic Analysis
3.3.1. Pod Harvest Index (PHI), Seed Yield and Chlorophyll Content
We also measured some plant performance characteristics. The three soybean cultivars were not significantly (p > 0.05) different for PHI when they were well-watered (Figure 4C). Exposure to drought significantly (p < 0.05) increased the PHI in Prima 2000 and Jackson but not in A5409RG, with greater increase in Jackson than in Prima 2000 (Figure 4C). Drought reduced seed yield in all cultivars with Prima 2000 out-performing the other two cultivars. Prima 2000 had significantly greater (p < 0.05) seed yield (2.3 t/ha) than the two other cultivars (Jackson 1.7 t/ha and A5409RG 1.8 t/ha) (Table 3). We also found a significant correlation between nodule size and seed yield under well-watered condition (R2 = 0.45; p = 0.046) and also under drought (R2 = 0.592; p = 0.015).
Chlorophyll meter readings (SCMR) of all cultivars were not significantly different at the beginning of drought exposure (45 SCMR). Drought reduced SCMR in leaves of all cultivars, but Prima 2000 outperformed the two other cultivars and A5409RG had the lowest (p < 0.05) SCMR (Figure 4D).

Table 3.
A comparison of shoot performance in three soybean cultivars (A5409RG, Jackson and Prima 2000) under well-watered and drought conditions. Biomass at flowering (F), mid pod filling stage (MP) and seed yield traits.
| Cultivar | Leaf Dry Mass F (g) | Stem Dry Mass F (g) | Total Biomass F (g) | Leaf Dry Mass MP (g) | Stem Dry Mass MP (g) | Pod Dry Mass MP (g) | Total Biomass MP (g) | Seed Yield (t/ha) |
|---|---|---|---|---|---|---|---|---|
| Well-watered | ||||||||
| A5409RG | 6.6 ± 0.3ab | 5.8 ± 0.2ab | 12.4 ± 0.2b | 28.7 ± 0.4a | 29.0 ± 0.4a | 13.3 ± 0.5a | 71.0 ± 0.7a | 4.0 ± 0.08a |
| Jackson | 5.9 ± 0.1b | 5.1 ± 0.2b | 11.0 ± 0.4c | 22.9 ± 0.3b | 25.1 ± 0.3b | 10.4 ± 0.3b | 58.4 ± 0.6b | 2.0 ± 0.10 |
| Prima 2000 | 7.3 ± 0.2a | 6.4 ± 0.1a | 13.7 ± 0.3a | 29.9 ± 1.2a | 28.2 ± 1.2a | 13.9 ± 0.4a | 71.9 ± 1.7a | 4.4 ± 0.10 |
| Significance | ** | ** | ** | ** | ** | ** | ** | ** |
| Drought | ||||||||
| A5409RG | 3.8 ± 0.1b | 3.6 ± 0.1b | 7.4 ± 0.1b | 23.1 ± 0.5b | 24.4 ± 0.5ab | 7.9 ± 0.4c | 55.4 ± 1.0b | 1.8 ± 0.02b |
| Jackson | 4.0 ± 0.9b | 4.2 ± 0.2b | 8.2 ± 0.2b | 21.7 ± 0.2b | 21.2 ± 0.4b | 9.4 ± 0.1b | 52.2 ± 0.5b | 1.7 ± 0.02b |
| Prima 2000 | 6.0 ± 0.2a | 4.9 ± 0.2a | 10.9 ± 0.3a | 26.5 ± 0.6a | 26.2 ± 2.1a | 11.2 ± 0.3a | 63.8 ± 2.6a | 2.3 ± 0.03a |
| Significance | ** | ** | ** | ** | * | ** | ** | ** |
Data represent the mean ± SEM of three replications under both well watered and drought conditions. Biomass at flowering and mid pod filling stage was taken on six representative individual plants per plot. Different letters within a column denote a significant difference (p < 0.05).
3.3.2. Biomass and Plant Maturation
We also determined the biomass of different above-ground plant parts. Regardless of the water regime and biomass measurement stage, the three cultivars differed significantly (p < 0.05) for their leaf, stem and total biomass (Table 3). Prima 2000, however, was again best-performing with significantly (p < 0.05) greater dry mass (leaf, stem, and pod) than the two other cultivars. Drought caused a significant reduction of total biomass in all three cultivars at the flowering stage with 21% and 25% biomass reduction in Prima 2000 and Jackson, respectively but with 40% biomass reduction in A5409RG. Stem diameter under well-watered conditions was higher for Prima 2000 and A5409RG than for Jackson and drought treatment did not significantly change stem diameter. Although we found a positive association of chlorophyll reading with seed yield, this association was not significant.
Drought exposure also caused faster plant maturation (Table 4). Jackson matured faster under drought (81 days) and well-watered conditions (90 days) than the two other cultivars which matured under well-watered conditions after115–118 days and after 98–101 days under drought (Table 4). Under well-watered conditions, the three cultivars were also similar in their biomass accumulation (leaf and stem) per day, but biomass accumulation was greater for Jackson and Prima 2000 under drought conditions (Table 4).

Table 4.
A comparison of time to maturity and biomass accumulation in soybean cultivars A5409RG, Jackson and Prima 2000.
| Cultivar | Maturity (days) | Leaf (g/day) | Stem (g/day) |
|---|---|---|---|
| Well-watered | |||
| A5409RG | 101 | 0.24 | 0.25 |
| Jackson | 90 | 0.25 | 0.28 |
| Prima 2000 | 98 | 0.26 | 0.25 |
| Drought | |||
| A5409RG | 118 | 0.23 | 0.24 |
| Jackson | 81 | 0.27 | 0.26 |
| Prima 2000 | 115 | 0.27 | 0.27 |
Calculation was carried out by dividing either the mean seed yield or biomass at mid-pod filling (leaf, stem and pod) by days to maturity for each cultivar.
4. Discussion
The absence of suitable technologies to determine root architecture accurately under field conditions is a major impediment to the genetic improvement of root traits associated with drought tolerance and enhanced WUE. Analyzing root architecture in the manner performed in the present study therefore remains what can be described as the current “gold standard”, for such measurements. The results presented here demonstrate that root phenotyping can be a useful and simple method for the selection of drought-tolerant cultivars of important crops such as soybean under field conditions. These findings are in line with observations using a soybean collection that matched root and shoot architecture traits as well as other parameters such as carbon assimilation, and nutrient and water uptake to harvestable grain yields during drought [20]. Moreover, the drought tolerance characteristics reported here are consistent with previous observations of relative drought tolerance in the Jackson, Prima 2000 and A5409RG cultivars made under controlled environment conditions [24,31]. Although the study described here compared relatively few cultivars, the observed phenotypic variations may serve as a first step to establishing reference cultivars for different root phenotypes that can be applied in such studies in the future.
Root architecture and nodule sustainability are important parameters associated with sustained plant performance under drought. Jackson, which is a drought-escaping cultivar with early flowering and deep root phenotype, has longer and deeper roots that contribute to better water uptake [20,22,23], performed well under drought conditions. However, Prima 2000, which has an “intermediate” root system, was the most drought-tolerant cultivar under the well watered and drought conditions imposed in these studies. Prima 2000 had a greater shoot biomass and yield than the other two cultivars under drought. The A5409RG type of root system is classifed as “shallow” [20]. A shallow root system may be less beneficial in reaching deeper water resources during drought. In support of this hypothesis, our results demonstrate that A5409RG was the most drought-sensitive cultivar.
Prima 2000 was able to maintain more nodules than the other two cultivars under drought conditions. This finding suggests that in addition to root system architecture, the ability to form and sustain root nodules may also be an important trait underpinning shoot productivity under drought. This conclusion is also supported at least in part by studies showing a direct relationship of the QTL for nitrogen use efficiency and root architecture [32,33]. Jackson and Prima 2000 had larger nodules than A5409RG under drought conditions. Large nodules have a greater phloem supply of carbohydrates and water than smaller nodules [34,35]. Drought can adversely affect the volumetric flow in the phloem phylum decreasing the relative water content of the nodules leading to a smaller nodule size [36]. The ability to maintain the water supply to the nodules is therefore an important trait that would facilitate higher rates of symbiotic nitrogen fixation (SNF). A positive correlation was observed between nodule size and seed yield. Moreover, this correlation was largely independent of water availability. While more work is required to understand the mechanistic relationships between nodule size and SFN in Jackson and Prima 2000 occurring during drought, the findings of this study suggest that nodule size is a useful phenotypic trait that might be used for the selection for high seed yield under field conditions.
Root architectures that maximize water uptake can only be an advantage under drought if they are accompanied by appropriate shoot characteristics associated with high yields. High grain yields fundamentally depend on the net photosynthesis by the leaf canopy over the growing season, as well as partitioning of photosynthetic products to harvested organs [37]. The data presented here show that Prima 2000 had the greatest shoot biomass and the highest seed yield under drought conditions. The lower yield of Jackson may be related to the shorter period of vegetative growth and hence photo-assimilate acquisition, and/or a lower ability to partition assimilated carbohydrates to the grain, or both. The seed yield and leaf biomass accumulation data reported here for plants grown under drought indicate show that Jackson and Prima 2000 are able to make better use of assimilates than A5409RG. Given the overall performance of Prima 2000 and Jackson under drought conditions, we consider that these cultivars could serve as parents in future studies seeking to improve seed yield during either late stage (Jackson) or intermittent (Prima 2000) drought exposures.
We have used PHI as a selection criterion for drought tolerance in soybean. PHI appears to be a good selection trait for the translocation of photo-assimilates in common beans, where an association between PHI and grain yield was reported [38,39]. Unlike harvest index determinations, which require a large amount of biomass, PHI determinations are easier to perform and they are hence more useful in breeding programs in which hundreds of germplasm accessions have to be screened [38,39]. The non-destructive chlorophyll measurements made using the SPAD 502 Plus Chlorophyll Meter provide a semi-quantitative indicator of the level of leaf greenness. While this technique is often used in field phenotyping studies, the data reported here suggest that great care must be exercised in data interpretation because the parameters measured in this way are not directly correlated with either biomass or seed yield. These findings support similar observations in dry bean [40]. The usefulness of this method in predicting seed yield is therefore uncertain.
In our studies, the root crown had to be extracted from the soil for phenotyping [41]. Such excavation approaches are labor-intensive and tend to destroy a significant amount of root system information. While high-throughput field-based root phenotyping systems that allow simultaneous measurements of parameters such as WUE, root depth and density are currently not available for field studies, non-destructive methods based on root imaging techniques are currently being developed [42]. Current field-phenotyping platforms focus on shoot growth, gas exchange characteristics and the status of the canopies of large numbers of plants, which are monitored by sensors placed on tractors, robots or flying platforms [42]. In contrast, high throughput lab-based root system phenotyping platforms that are currently used are not highly accurate often because they use methods that have a relatively low resolution [42]. A support vector machine method using single top view digital images of excavated maize roots was recently used to classify more than 99% of the roots in 235 maize genotypes [43]. In addition, transparent tubes (mini-rhizotrons) installed vertically, horizontally, or at various angles in the field have been used to measure root elongation rates, root densities and surface areas, root numbers and root lengths at different soil depths throughout the growing season [44,45]. In these systems, the growing roots are imaged around the outside walls of the tubes with cameras inserted throughout the tube length. Tomographic measurements of the root system in situ using X-rays (micro-Computed Tomography; μCT) is also being applied to study the role of roots in drought adaptation [45]. This approach can be used to visualize three-dimensional root architecture in natural habitats [46]. However, until such advanced root growth imaging tools are widely available for use under field conditions; destructive root measurements remain the simplest available methods suitable for selection protocols.
5. Conclusions
In summary, the results presented here confirm the importance of root system architecture in drought tolerance to soybean. While manual phenotyping tools are labor-intensive and time consuming, they are still of great value particularly in developing countries, where high throughput phenotypic screening systems using imaging technologies are currently unavailable. The data reported here apply to light soil types from which roots can be easily removed. The three cultivars studied here in field experiments show similar relative drought tolerance characteristics and performance to those determined in controlled environment studies, with Prima 2000 being the most drought tolerant cultivar and A5409RG being the least drought tolerant cultivar. The observed correlation between nodule size and seed yield demonstrates that nodule parameters have considerable potential as phenotypic markers for drought-tolerance screening in soybean. We conclude that nodule characteristics also have potential as targets for molecular marker development.
Acknowledgments
This work was funded by Tropical legume II (TL II) through International Centre of Tropical Agricultural (CIAT) Cali, Colombia, the CIAT-Africa or Pan Africa Bean Research Alliance (PABRA), Uganda, and the Howard G Buffett Foundation. We also gratefully acknowledge the financial support given by FP7-PIRSES-GA-2008-230830 (LEGIM; Berhanu Fenta, Christine Foyer and Karl Kunert) and PIIF-GA-2011-299347 (Soylife; Karl Kunert.). We thank Curtis Markle Frederick, for his support at URBC.
Author Contributions
Berhanu Fenta carried out all experimental work and prepared the first draft of the article. Karl Kunert and Stephen Beebe designed the experiments and oversaw the experimental work of Berhanu Fenta at University of Pretoria. Jonathan Lynch, James Burridge, and Katy Barlow provided underpinning methodologies in root phenotyping and oversaw the field trial at Ukulima Root Biology Centre (URBC). Christine H. Foyer helped design the experiments, and wrote the final version of the manuscript together with Karl Kunert.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sinclair, T.R.; Marrou, H.; Soltani, A.; Valdez, V. Soybean production in Africa. Glob. Food Biol. 2014. [Google Scholar] [CrossRef]
- Furbank, R.T.; Tester, M. Phenomics—Technologies to relieve the phenotyping bottleneck. Trends Plant Sci. 2011, 16, 635–644. [Google Scholar] [CrossRef]
- Araus, J.L.; Cairns, J.E. Field high-throughput phenotyping: The new crop breeding frontier. Trends Plant Sci. 2013, 19, 1–62. [Google Scholar]
- Beebe, S.E.; Rao, I.M.; Blair, M.W.; Acosta-Gallegos, J.A. Phenotyping common beans for adaptation to drought. Front Physiol. 2013, 4. [Google Scholar] [CrossRef]
- Manavalan, L.P.; Guttikonda, S.K.; Phan Tran, L.S.; Nguyen, H.T. Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol. 2009, 50, 1260–1276. [Google Scholar]
- Pastenes, C.; Pimentel, P.; Lillo, J. Leaf movements and photoinhibition in relation to water stress in field-grown beans. J. Exp. Bot. 2005, 56, 425–433. [Google Scholar] [CrossRef]
- Acosta Gallegos, J.A. Selection of Common Bean (Phaseolus vulgaris L.) Genotypes with Enhanced Drought Tolerance and Biological Nitrogen Fixation. Ph.D. Thesis, Michigan State University, East Lansing, MI, USA, 1988. [Google Scholar]
- Blum, A. Drought resistance, water-use efficiency, and yield potential—Are they compatible, dissonant, or mutually exclusive? A. J. Agric. Res. 2005, 56, 1159–1168. [Google Scholar] [CrossRef]
- Kanagaraj, P.; Prince, K.S.J.; Sheeba, J.A.; Biji, K.R.; Paul, S.B.; Senthil, A.; Babu, R.C. Microsatellite markers linked to drought resistance in rice. Curr. Sci. 2010, 98, 836–839. [Google Scholar]
- Salunkhe, A.S.; Poornima, R.; Prince, K.S.J.; Kanagaraj, P.; Sheeba, J.A.; Amudha, K.; Suji, K.K.; Senthil, A.; Babu, R.C. Fine mapping QTL for drought resistance traits in rice using bulk segregant analysis. Mol. Biotechnol. 2011, 49, 90–95. [Google Scholar] [CrossRef]
- Manavalan, L.P.; Guttikonda, S.K.; Nguyen, V.T.; Shannon, J.G.; Nguyen, H.T. Evaluation of diverse soybean germplasm for root growth and architecture. Plant Soil 2010, 330, 503–514. [Google Scholar] [CrossRef]
- Suji, K.K.; Prince, K.; Mankhar, P.S.; Kanagaraj, P.; Poornima, R.; Amutha, K.; Kavitha, S.; Biji, K.R.; Gomez, S.M.; Babu, R.C. Evaluation of rice near iso-genic lines with root QTLs for plant production and root traits in rainfed target populations of environment. Field Crop Res. 2012, 137, 89–96. [Google Scholar]
- Trachsel, S.; Kaeppler, S.M.; Brown, K.M.; Lynch, J.P. Shovelomics: High throughput phenotyping of maize root architecture in the field. Plant Soil 2011, 341, 75–87. [Google Scholar] [CrossRef]
- Prince, S.J.; Mutava, R.N.; Pegoraro, C.; Oliveira, A.C.D.; Nguyen, H.T. Root characters. In Genomics and Breeding for Climate Resilient Crops; Kole, C., Ed.; Springer: Berlin, Germany, 2013; pp. 67–131. [Google Scholar]
- Nielsen, K.L.; Lynch, J.P.; Weiss, H.N. Fractal geometry of bean root systems: Correlations between spatial and fractal dimension. Am. J. Bot. 1997, 84, 26–33. [Google Scholar] [CrossRef]
- Osmont, K.S.; Sibout, R.; Hardtke, C.S. Hidden branches: Developments in root system architecture. Annu. Rev. Plant Biol. 2007, 58, 93–113. [Google Scholar] [CrossRef]
- Lopes, M.S.; Araus, J.L.; van Heerden, P.D.R.; Foyer, C.H. Enhancing drought tolerance in C4 crops. J. Exp. Bot. 2011, 62, 3135–3153. [Google Scholar] [CrossRef]
- Matsui, T.; Singh, B.B. Root characteristics in cowpea related to drought tolerance at the seedling stage. Exp. Agric. 2003, 39, 29–38. [Google Scholar] [CrossRef]
- Agbicodo, E.M.; Fatokun, C.A.; Muranaka, S.; Visser, R.G.F.; van der Linden, C.G. Breeding drought tolerant cowpea: Constraints, accomplishments, and future prospects. Euphytica 2009, 167, 353–370. [Google Scholar] [CrossRef]
- Zhao, J.; Fu, J.; Liao, H.; He, Y.; Nian, H.; Hu, Y.; Qiu, L.; Dong, Y.; Yan, X. Characterization of root architecture in an applied core collection for phosphorus efficiency of soybean germplasm. Chin. Sci. Bull. 2004, 49, 1611–1620. [Google Scholar] [CrossRef]
- Ao, J.; Fu, J.; Tian, J.; Yan, X.; Liao, H. Genetic variability for root morph-architecture traits and root growth dynamics as related to phosphorus efficiency in soybean. Funct. Plant Biol. 2010, 37, 304–312. [Google Scholar]
- Garay, A.F.; Wilhelm, W.W. Root system characteristics of two soybean isolines undergoing water stress conditions. Agron. J. 1982, 75, 973–977. [Google Scholar] [CrossRef]
- Serraj, R.; Bona, S.; Purcell, L.C.; Sinclair, T.R. Nitrogen accumulation and nodule activity of field-grown Jackson soybean in response to water deficits. Field Crops Res. 1997, 52, 109–116. [Google Scholar] [CrossRef]
- Fenta, B.A.; Driscoll, S.P.; Kunert, K.J.; Foyer, C.H. Characterization of drought-tolerance traits in nodulated soya beans: The importance of maintaining photosynthesis and shoot biomass under drought-induced limitations on nitrogen Metabolism. J. Agric. Crop Sci. 2011, 198, 92–103. [Google Scholar]
- USDA, 2011. Available online: http://soils.usda.gov/ (accessed on 13 February 2014).
- Hand Operated Planters. Available online: http://www.almaco.com/products/productDetail.cfm?ProductID=33 (accessed on 13 February 2014).
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 14th ed.; Pearson-Prentice Hall: Upper Saddle River, NJ, USA, 2008; p. 990. [Google Scholar]
- SPAD 502 Plus Chlorophyll Meter. Available online: http://www.specmeters.com/nutrient-management/chlorophyll-meters/spad/spad502p/ (accessed on 18 December 2013).
- Silva, H.C.; Braga, G.L. Effect of soaking and cooking on the oligosaccharide content of dry beans (Phaseolus vulgaris L.). J. Food Sci. 1982, 47, 924–925. [Google Scholar] [CrossRef]
- Fischer, R.; Maurer, R. Drought resistance in spring wheat cultivars. I. Grain yield responses. Aust. J. Agric. Res. 1978, 29, 897–912. [Google Scholar] [CrossRef]
- Fenta, B.A.; Schlüter, U.; Marquez Garcia, B.; DuPlessis, M.; Foyer, C.H.; Kunert, K.J. Identification and Application of Phenotypic and Molecular Markers for Abiotic Stress Tolerance in Soybean. In Soybean—Genetics and Novel Techniques for Yield Enhancement; Krezhova, D., Ed.; InTech: Rijeka, Croatia, 2011; Volume 9; pp. 181–200. [Google Scholar]
- Sall, K.; Sinclair, T.R. Soybean genotypic differences in sensitivity of symbiotic nitrogen fixation to soil dehydration. Plant Soil 1991, 133, 31–37. [Google Scholar] [CrossRef]
- Coque, M.; Martin, A.; Veyrieras, J.B.; Hirel, B.; Gallais, A. Genetic variation for N-remobilization and postsilking N-uptake in a set of maize recombinant inbred lines. 3. QTL detection and coincidences. Theor. Appl. Genet. 2008, 117, 729–747. [Google Scholar] [CrossRef]
- Garnett, T.; Conn, V.; Kaiser, B.N. Root based approaches to improving nitrogen use efficiency in plants. Plant Cell Environ. 2009, 32, 1272–1283. [Google Scholar] [CrossRef]
- King, C.A.; Purcell, L.C. Soybean nodule size and relationship to nitrogen fixation response to water deficit. Crop Sci. 2001, 41, 1099–1107. [Google Scholar] [CrossRef]
- Walsh, K.B.; Canny, M.J.; Layzell, D.B. Vascular transport and soybean nodule function: II. A role for phloem supply in product export. Plant Cell Environ. 1989, 12, 713–723. [Google Scholar] [CrossRef]
- Mayers, J.D.; Lawn, R.J.; Byth, D.E. Adaptation of soybean (Glycine max (L.) Merrill) to the dry season of the tropics. II. Effects of genotype and environment on biomass and seed yield. Aust. J. Agric. Res. 1991, 42, 517–530. [Google Scholar] [CrossRef]
- Beebe, S.; Rao, I.M.; Blair, M.W.; Butare, L. Breeding for abiotic stress tolerance in common bean: Present and future challenges. In Proceedings of the 14th Australian Plant Breeding & 11th SABRAO Conference, Brisbane, Australia, 10–14 August 2009.
- Rao, I.M.; Beebe, S.; Ricaurte, J.; Cajiao, C.; Polania, J.; Garcia, R. Phenotypic evaluation of drought resistance in advanced lines of common bean (Phaseolus vulgaris L.). In Proceedings of the ASA-CSSA-SSSA International Annual Meeting, New Orleans, LA, USA, 4–8 November 2007.
- Guler, S.; Ozcelik, H. Relationships between leaf chlorophyll and yield related characters of dry bean (Phaseolus vulgaris L.). Asian J. Plant Sci. 2007, 6, 700–703. [Google Scholar] [CrossRef]
- Lynch, J.P. Root phenes for enhanced soil exploration and phosphorus acquisition: Tools for Future Crops. Plant Physiol. 2011, 156, 1041–1049. [Google Scholar] [CrossRef]
- Zhu, J.; Ingram, P.A.; Benfey, P.N.; Elich, T. From lab to field, new approaches to phenotyping root system architecture. Curr. Opin. Plant Biol. 2011, 14, 310–317. [Google Scholar] [CrossRef]
- Zhong, D.; Novais, J.; Grift, T.E.; Bohn, M.; Han, J. Maize root complexity analysis using a Support Vector Machine method. Comput. Electron. Agric. 2009, 69, 46–50. [Google Scholar] [CrossRef]
- Eberbach, P.L.; Hoffmann, J.; Moroni, S.J.; Wade, L.J.; Weston, L.A. Rhizo-lysimetry: Facilities for the simultaneous study of root behavior and resource use by agricultural crop and pasture systems. Plant Methods 2013, 9. [Google Scholar] [CrossRef]
- Mooney, S.J.; Pridmore, T.P.; Helliwell, J.; Bennett, M.J. Developing X-ray computed tomography to non-invasively image 3-D root systems architecture in soil. Plant Soil 2012, 352, 1–22. [Google Scholar] [CrossRef]
- Mairhofer, S.; Zappala, S.; Tracy, S.; Sturrock, C.; Bennett, M.J.; Jon Mooney, S.; Pridmore, T.P. Recovering complete plant root system architectures from soil via X-ray μ-Computed Tomography. Plant Methods 2013, 9. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
