1. Introduction
Turfgrass systems are an essential and highly visible component of modern European landscapes. They comprise private gardens, golf courses, athletic fields, and municipal green spaces, and provide a wide array of aesthetic, recreational, and ecological functions [
1,
2]. These functions include visual appeal, opportunities for organized sport and leisure, air purification, heat mitigation, and soil stabilization [
1,
2]. As urbanization continues, the demand for well-maintained turf areas has increased, reflecting their role in meeting recreational needs, supporting sports performance standards, and contributing to the economic value of amenity and sports sectors [
3].
This review specifically addresses the control of insect pests and turf-damaging fungal pathogens, which represent the most significant biotic threats to turf performance in European contexts. While turfgrass systems face various biotic stressors [
2,
3,
4,
5], weeds remain a notable nuisance due to their ability to outcompete desirable turf species for light, nutrients, and water, reducing vigor, uniformity, and visual quality [
5,
6,
7]. Species such as
Poa annua L. (annual bluegrass) are especially problematic in high-maintenance sites like sports fields and golf courses in Slovenia [
7,
8], with adaptability, prolific seed production, and increasing herbicide resistance highlighting the challenge [
9,
10,
11]. Although certain weed species can provide soil stabilization in unmanaged systems [
5,
7], their negative effects on playability and aesthetics outweigh these benefits in intensively managed turf. Consequently, while weeds are significant, this review focuses primarily on insect pests and turf-damaging pathogens.
Our emphasis is guided by the increasing prevalence and impact of specific pests and pathogens that cause both aesthetic and functional damage to turfgrass. The most important among these are root-feeding insect larvae such as chafer beetle larvae (Coleoptera: Scarabaeidae) and leatherjackets, larvae of
Tipula spp. L. (Diptera: Tipulidae), which compromise turf stability, create surface irregularities, and attract vertebrate predators that further disturb the turf [
1,
12,
13]. Fungal pathogens, too, present a growing challenge, particularly under changing climatic conditions and reduced pesticide availability, leading to disease outbreaks that affect sward density and appearance [
2,
7]. These organisms often act in combination with abiotic stressors and can be difficult to detect until visible damage has occurred [
2]. Consequently, understanding and managing these particular biotic threats is central to maintaining turf quality, especially as reliance on synthetic pesticides is increasingly restricted by environmental regulations and public health considerations [
14,
15].
At the same time, turfgrass systems have become flashpoints in larger debates around sustainability. Critics cite their high-water demands, input intensity, and limited biodiversity as reasons to replace them altogether [
16,
17,
18,
19,
20]. In some regions, “lawn rebate” programs even incentivize homeowners to remove turf to conserve water [
21,
22]. However, these discussions often overlook the broad ecosystem services turfgrass provides, including erosion control, carbon sequestration, temperature regulation, and allergen reduction, all services that are not easily replaced by alternative groundcovers [
23]. Importantly, environmental issues arising from turf management, such as pollution and resource use, are often magnified by poor management practices, and in the case of high-input systems, resource demands are inherent to maintaining lush, uniform turf cover in areas where it would not naturally occur [
24]. Pest control decisions in turf management are also made by a diverse group of users, including professional grounds managers, facility operators, and homeowners. This wide user base presents distinct challenges for implementing effective IPM, especially in the context of increasing economic pressures, environmental concerns, and expectations for high-quality turf performance [
5,
25,
26].
Against this backdrop, a synergistic and sustainable approach to pest management has emerged as a critical solution. This model, rooted in the principles of IPM, emphasizes the coordinated use of biological control agents, cultural practices, and minimal, targeted chemical interventions [
15,
27,
28]. Rather than relying on a single tactic, SPM promotes resilience and long-term control by combining methods that support turf health and minimize environmental impact [
15]. When implemented correctly, such an approach not only reduces pest pressures but also contributes to soil vitality, beneficial insect populations, and more sustainable urban ecosystems [
29,
30]. Its core principles include regular pest monitoring, integration of compatible control methods, timing interventions to match pest biology, minimizing chemical inputs, and fostering ecological conditions that favor natural pest suppression [
2,
3,
4,
15,
25].
This review explores the ecological foundations and applied potential of SPM in turfgrass systems, with particular emphasis on key insect pests and fungal pathogens relevant to European conditions. While the primary focus is on practices suitable for European climates, regulatory frameworks, and turfgrass species, selected international case studies have also been included to illustrate transferable principles and mechanisms of synergy. By highlighting the integration of complementary biological, cultural, and chemical tactics, this review advocates for a more adaptive and resilient approach to turf protection, one that aligns aesthetic quality and functional performance with environmental stewardship and policy imperatives. Implementing SPM offers key ecological benefits such as reduced pesticide inputs, enhanced soil biodiversity, improved turf resilience, and lower risks of environmental contamination. Operationally, it supports compliance with strict European Union pesticide-reduction directives, helps maintain consistent turf quality under variable climatic conditions, and can reduce long-term costs through improved pest suppression efficiency.
Comparable benefits have been reported in Asian contexts. For example, in Japan, the adoption of integral turf management, which combines physical and biological tactics, has reduced reliance on chemical inputs and promoted holistic turf health [
31]. In South Korea, golf course managers are increasingly applying best management practices to minimize pesticide use, reduce dependency on imported agrochemicals, and improve turfgrass performance [
32]. These examples further demonstrate the global relevance of SPM’s principles.
3. Synergistic Pest Management in European Turfgrass
The management of turfgrass in European urban green spaces, such as football fields, golf courses, and ornamental lawns, is undergoing a pivotal transition. Historically dependent on routine pesticide applications and high-input regimes, these systems are increasingly constrained by ecological concerns and regulatory mandates, most notably the European Union
Directive-2009/128-EN-EUR-Lex, n.d., [
14], targeting a 50% reduction in pesticide use and risk by 2030 [
79]. In this context, SPM has emerged as a robust framework for delivering effective pest suppression while aligning with ecological and policy imperatives.
SPM extends beyond conventional IPM by emphasizing the interactive effects of biological, cultural, and chemical controls, using their functional interplay to enhance overall efficacy [
15,
80]. This section outlines the conceptual basis of SPM in turfgrass systems, setting the stage for further exploration of its practical and ecological dimensions.
3.1. Defining the Synergistic Approach
SPM refers to the intentional co-application or temporal sequencing of pest control strategies that interact positively, achieving greater pest suppression than any individual method alone [
81]. Unlike additive IPM programs, which emphasize combination approaches, SPM focuses on
mechanistic complementarity, where interactions between different approaches produce a level of control greater than the sum of their individual effects [
15,
82]. In managed turfgrass systems, such synergy is achieved through carefully timed and compatible combinations of:
Biological agents, such as entomopathogenic nematodes (EPNs) and fungi;
Cultural practices, like aeration, irrigation scheduling, mowing, and surface rolling;
Selective chemical inputs, applied at low doses to support biological or cultural efficacy rather than override them.
These categories represent core pathways through which synergistic interactions can emerge. Biological–biological, chemical–biological, and cultural–biological combinations each offer unique potential to enhance control outcomes. The key distinction of SPM lies in its focus on these functional relationships, designing control programs not as static combinations, but as ecologically responsive systems.
This systems-based thinking is particularly relevant in European turfgrass settings, where multiple overlapping pest pressures and increasing regulatory constraints demand solutions that are not only effective, but also sustainable. In this context, SPM provides a framework for transitioning from reactive chemical management to ecologically harmonized, performance-oriented strategies [
2,
5,
6].
3.2. Ecological and Operational Benefits of SPM
SPM offers European turfgrass managers a range of ecological and operational benefits in turfgrass settings, where pesticide regulation and performance expectations often conflict. These benefits include not just improved pest suppression but also enhanced compliance, environmental protection, and turf system resilience.
3.3. Reduced Chemical Inputs and Regulatory Compliance
One of the primary advantages of SPM, compared with conventional IPM programs that often focus on additive integration of various approaches, is its emphasis on achieving true synergistic effects. This approach can further reduce chemical pesticide inputs without sacrificing turf quality, while directly supporting the European Union’s pesticide reduction goals [
79].
For example, Scandinavian golf course trials demonstrated that combining lightweight rolling with adjusted nitrogen inputs reduced
Microdochium nivale incidence by up to 50%, even when fungicide applications were halved [
69,
83]. Rolling greens 2–3 times weekly from late summer into autumn, paired with careful autumn fertilization, helped superintendents in Norway and Denmark shift from reactive spraying to a preventive, ecological approach [
66].
By modifying the growing environment through rolling, mowing, drainage, or nutrition, turf managers can suppress disease development and reduce dependency on fungicides [
15,
84].
3.4. Improved Pest Suppression Through Complementarity
Beyond reducing chemical inputs, SPM enhances turfgrass pest control by combining tactics that exploit different biological mechanisms or environmental opportunities. These synergies fall into three main categories:
3.4.1. Biological–Biological Synergies
Combining biocontrol agents can improve efficacy by targeting pests through multiple, complementary pathways. For example, applying
Metarhizium anisopliae (Mechnikov) Sorokin, four weeks before
Heterorhabditis bacteriophora Poinar, achieved over 95% mortality of
Hoplia philanthus Füssly [
85]. Similar enhancements were observed when
Beauveria brongniartii (Saccardo) Petch or
M. anisopliae were paired with nematodes against
Exomala orientalis Waterhouse,
Ectinohoplia rufipes Motschulsky, and
Coptognathus curtipennis Burmeister [
86,
87]. In greenhouse trials,
H. bacteriophora combined with either
B. bassiana (Balsamo) Vuillemin or
M. anisopliae increased larval mortality of
Cyclocephala lurida Bland [
88].
Koppenhöfer and Kaya [
89] found that
Bacillus thuringiensis subsp.
japonensis (Btj) Berliner, applied 7–14 days before nematodes, improved
Cyclocephala spp. control through stress-induced susceptibility. Similarly,
Melolontha hippocastani Fabricius larvae infected with rickettsia became 3–6 times more susceptible to nematodes, though with reduced nematode reproduction [
90].
These findings support the strategic use of staggered or co-applied biocontrols to enhance pest suppression across turf systems.
3.4.2. Chemical–Biological Synergies
Synergies also arise when entomopathogenic nematodes (EPNs) are paired with low doses of insecticides such as imidacloprid or chlorantraniliprole. These chemicals impair grub movement and grooming, increasing susceptibility to nematode infection without harming nematode viability [
91,
92]. Enhanced control of late-instar white grubs of
Popillia japonica Newman (Coleoptera: Scarabaeidae),
Cyclocephala borealis Arrow (Coleoptera: Scarabaeidae),
Cyclocephala hirta LeConte (Coleoptera: Scarabaeidae), and
Cyclocephala pasadenae Casey (Coleoptera: Scarabaeidae) has been documented using these combinations [
93,
94], and chlorantraniliprole has shown similar compatibility [
95].
However, precise timing is critical. Misaligned applications can reduce efficacy by disrupting host susceptibility or nematode function [
96]. Successful integration hinges on matching chemical exposure with biological activity windows.
3.4.3. Cultural–Biological Synergies
Cultural practices can improve biocontrol performance by modifying environmental conditions. For example,
Steinernema feltiae thrives in moist, aerated soils, a condition enhanced through irrigation and core aeration [
94,
97]. These adjustments improve nematode mobility and persistence [
98].
Other practices, such as mowing height adjustment, rolling, and thatch reduction, can suppress disease while supporting microbial activity. For instance, rolling combined with reduced nitrogen applications halved
Microdochium nivale incidence, even with lower fungicide input [
99].
Soil health improvements through composting, aeration, or organic amendments promote beneficial organisms and increase turf resilience [
100]. These approaches reflect a systems-level design where management fosters ecological conditions unfavorable to pests but supportive of biocontrol agents.
Together, these synergies increase control reliability and buffer the variability often associated with biological tools under field conditions. The integration of biological, chemical, and cultural tactics, when properly sequenced and site-adapted, forms the backbone of robust, sustainable SPM programs.
3.5. Ecosystem Services and Turf Resilience in Synergistic Pest Management
Beyond direct pest suppression, SPM enhances soil health, biodiversity, and turf resilience, critical components of long-term sustainability in turfgrass systems.
By reducing broad-spectrum pesticide use and integrating organic inputs and biological controls, SPM fosters soil microbial diversity, improves nutrient cycling, and promotes natural antagonism of pathogens and pests [
3,
29]. Practices such as compost topdressing and reduced fungicide programs support beneficial microbial communities that, in turn, strengthen root development and create self-reinforcing disease suppression feedbacks [
29].
Low-input systems also encourage invertebrate biodiversity. Earthworms, ground beetles, and other decomposers contribute to thatch degradation, aeration, and indirect pest control, while habitat features like clover roughs and wildflower strips support pollinators and broader biodiversity targets [
23]. Some biological control agents, such as
Beauveria brogniartii, can even establish permanently after repeated applications, providing persistent biological control on a long-term basis [
100], and endophyte-enhanced cultivars offer durable, pest-resistant turf with minimal external inputs [
76].
These ecological enhancements translate into greater physiological and visual turf resilience. Integrating cultural practices, such as adaptive mowing, aeration, and ecologically informed irrigation, with resilient cultivars fosters denser, stress-tolerant turf that rebounds more effectively from biotic and abiotic pressures [
3,
6]. Endophyte-containing grasses not only deter insect feeding but also maintain visual quality under drought, supporting uniformity in color and texture across seasons [
29].
Eco-functional turf designs, those prioritizing multifunctionality and resource efficiency, have demonstrated visual stability across environmental gradients. Historical turf concepts adapted for modern low-input settings uphold both cultural and ecological values [
101]. Precision irrigation and nutrient scheduling, guided by ecological feedback, further reduce stress and enhance canopy uniformity, even under variable climatic conditions [
102].
Ultimately, SPM contributes significantly to aesthetic and functional outcomes by preventing pest-induced thinning and promoting rapid regrowth. This ensures more consistent visual quality throughout the season, an essential goal for high-demand turf settings such as sports fields, golf courses, and urban green spaces.
3.6. Challenges to Implementation in European Turfgrass
While SPM offers clear ecological and operational benefits, its adoption in European turfgrass systems faces interconnected environmental, economic, technical, and cultural challenges. Environmental constraints include variable climatic conditions, restrictions on pesticide use, and the sensitivity of biological agents such as EPNs and fungi to soil temperature, soil moisture, ultraviolet exposure, and pH [
15,
79,
82,
84,
94]. Compacted or heavily trafficked soils, which are common in sports turf and other intensively managed surfaces, can limit nematode mobility and reduce their capacity to locate hosts. Certain fungicides or fertilizers may also disrupt beneficial microbial activity, complicating integration with standard turf management practices and reducing the reliability of biological control under field conditions [
82,
84].
Economic barriers are also significant. Many biological products have higher upfront costs than conventional pesticides and may require repeated applications to achieve effective suppression. The successful use of these products often depends on skilled personnel who can manage environmental requirements, adjust timing, and ensure compatibility between control measures. These labor and training demands represent a particular challenge for municipalities and public institutions that operate under strict budget constraints [
3,
83].
Technical complexity further limits adoption. Effective SPM requires accurate pest diagnostics, detailed knowledge of pest life cycles, and careful timing to coincide with susceptible pest stages. It also requires compatibility assessments to avoid counterproductive combinations, such as the simultaneous use of incompatible chemical and biological products [
29,
83]. In turfgrass systems, the lack of widely accessible, turf-specific decision-support tools across Europe makes it more difficult for managers to optimize these integrated strategies.
Regarding product availability, several biological control agents, particularly nematode-based products for chafer grub control, are already formulated specifically for turfgrass applications [
79,
84]. Conversely, many fungal products have been developed and optimized for intensive semi-protected crops such as soft fruits, tomatoes, and ornamentals, which are often grown under glasshouse or polytunnel conditions. Broad-acre agriculture has comparatively few biocontrol options, largely due to economic considerations, as the main cost constraints are associated with fuel and large-scale application, making many biologicals less competitive [
79,
84]. While some turf biocontrol products remain unoptimised for the cool-season conditions typical of much of Europe, this limitation is not primarily due to regulatory fragmentation. In most cases, the registration process for approved products is relatively straightforward. The greater challenge lies in expanding labeled uses of registered products, increasing the number of validation trials in turf-specific environments, and strengthening collaboration between scientists, industry partners, and regulatory agencies to accelerate the adaptation of existing agents for turfgrass pest and disease management [
79,
84].
Cultural barriers also influence adoption. Turf managers, particularly in high-performance venues such as golf courses and sports stadiums, are often under pressure to maintain visually uniform, high-quality turf. The slower or less predictable results of biological or cultural interventions may not align with these expectations [
15,
103]. Traditional management standards, which prioritize flawless visual presentation, can conflict with approaches that promote greater ecological diversity or accept minor variations in sward appearance. At the same time, ecological values such as maintaining pollinator habitats, supporting soil biodiversity, and reducing chemical inputs must be balanced against these aesthetic expectations. This tension can delay adoption, even when the long-term environmental and operational benefits of SPM are clear [
15,
103].
6. Synergistic Implementation Matrix and Seasonal Calendar
Bridging the gap between SPM theory and on-the-ground turfgrass application requires tools that are both systematic and practical. To support this, we propose two complementary components within a hybrid framework that integrates strategic planning with seasonal precision for Central Europe and other similar regions with the same seasonal progression. The first component is a strategic implementation matrix (
Table 1,
Table 2 and
Table 3), which outlines targeted interventions for each major pest group. This includes tools such as monitoring and diagnostic methods, biological control agents, cultural practices, and selective chemical inputs, all matched to specific seasonal periods. The matrix translates the core principles of SPM into a sequenced, pest-specific action plan that can be directly applied in the field.
While the matrices highlight promising combinations, not all integrated approaches produce additive or enhanced effects. Some pairings of
Beauveria bassiana or
Metarhizium anisopliae with other tactics have reduced fungal sporulation or nematode infectivity under certain conditions [
87,
88,
121]. Timing mismatches between entomopathogenic nematodes and other interventions can also limit effectiveness [
85,
86], and certain pesticides may impair nematode viability and host-finding ability [
134]. These examples stress the need for compatibility testing, correct sequencing, and context-specific trials to ensure that SPM strategies remain both effective and adaptable.
The chemical options listed in the matrices are drawn from published studies that have demonstrated significant efficacy in turfgrass systems worldwide. Their inclusion illustrates the potential role of selective chemical inputs within an SPM framework. Several of these active substances, including imidacloprid and chlorantraniliprole, are either not registered for turfgrass use or are prohibited for soil application within the European Union. This status is indicated in the tables using data from the Slovenian registration database FITO-INFO [
137], maintained by the Administration for Food Safety, Veterinary Sector, and Plant Protection of Slovenia, and the official plant protection portal [
138]. In practice, the framework should be adapted to each region by substituting locally registered and permitted products with similar modes of action, thereby maintaining both legislative compliance and adherence to SPM principles.
Table 1.
Seasonal SPM Tasks for Tipula spp. Management.
Table 1.
Seasonal SPM Tasks for Tipula spp. Management.
| Season | Monitoring & Diagnostics | Biological Control | Cultural Practices | Selective Chemicals |
|---|
| Early Spring | Scout for larvae in moist, low-cut areas. Use soil sampling and soap flushes to estimate density. Spec for damage thresholds. EPN persistence. | Apply S. feltie if thresholds exceeded at 10–15 °C in moist soils [138]. | Light rolling; raise mowing height. | Avoid applications. |
| Late Spring | Assess treatment results. Plan for reseeding or overseeding by late summer. | Apply S. feltie if thresholds exceeded at 10–15 °C in moist soils [138]. | Core aeration to improve soil health. | / |
| Early Summer | / | Apply EPNs only if the 2nd generation is confirmed (rare in the European Union). | Adjust irrigation to support roots and prevent waterlogging. | / |
| Mid Summer | Monitor turf for stress symptoms. | / | Reduce mowing frequency. | Spot treat if damage persists and EPNs fail (e.g., tefluthrin 1, lambda-cyhalothrin 1); not approved for turf in Slovenia [137]. |
| Late Summer | / | / | Overseed with endophyte | / |
| Autumn | Monitor adult crane fly emergence. | / | / | Apply selective insecticide if needed. Only spot-treat. Not approved for turf in Slovenia [137] |
| Winter | Map larval hotspots from fall emergence. Plan early spring control strategies. | / | Limit mowing and traffic to protect dormant roots. | / |
Table 2.
Seasonal SPM Tasks for Chafer Grub Management.
Table 2.
Seasonal SPM Tasks for Chafer Grub Management.
| Season | Monitoring & Diagnostics | Biological Control | Cultural Practices | Selective Chemicals |
|---|
| Early Spring | Soil sampling to detect overwintering grubs; forecast emergence and development stages using heat-sum models. | / | Avoid intensive cultivation; initiate moderate irrigation | / |
| Late Spring | Monitor adult beetles with pheromone traps to time larval hatch. | / | Core aerate to reduce compaction; support root and EPN function. | / |
| Early Summer | Track young larval presence post adult flights. | Apply EPNs (e.g., Heterorhabditis bacteriophora) targeting early-instar larvae under moist soil conditions [138]. | Maintain moderate mowing height; ensure adequate irrigation that supports both EPN survival and turf vigor. | Apply insecticide (e.g., tefluthrin 1, lambda-cyhalothrin 1) at a low dose 1–2 weeks before EPNs for synergistic priming. Both are registered against chafer grubs but not approved for turf use in Slovenia [137]. |
| Mid Summer | Assess infestation persistence, especially in previously affected areas. | Apply EPFs (e.g., Beauveria bassiana or Metarhizium anisopliae) if late-instar grubs persist [137]. | Reduce mowing frequency; perform surface recovery if needed (spot overseeding where heavy grubs feeding have thinned the canopy) | Spot treat only if thresholds breached and biologicals are ineffective. Not approved for turf in Slovenia [137] |
| Late Summer | Evaluate grub mortality from earlier biological or chemical inputs; flag persistent zones. | / | Aerate, irrigate, and overseed with endophyte-enhanced cultivars. | / |
| Autumn | Post-treatment larval counts; map infestation patterns. | / | Minimize stress; topdress or amend soils if needed. | In some regions, apply reduced-risk insecticide for late hatch; avoid overlap with biocontrols. Only spot-treatments, not approved for turf in Slovenia [137]. |
| Winter | Review seasonal data; prepare maps for next year’s applications. | / | Limit foot traffic; amend nutrient plans based on soil tests. | / |
Table 3.
Seasonal SPM Tasks for Major Turfgrass Fungal Pathogens.
Table 3.
Seasonal SPM Tasks for Major Turfgrass Fungal Pathogens.
| Season | Monitoring & Diagnostics | Biological Control | Cultural Practices | Selective Chemicals |
|---|
| Early Spring | Assess for Microdochium nivale; map susceptible zones (poorly drained, snow-covered) | / | Light rolling; avoid heavy mowing; calibrate irrigation to prevent early-season saturation. | Apply preventive QoI or SDHI fungicides (e.g., pyraclostrobin, boscalid) against Microdochium nivale and Red Thread when risk is high. In Slovenia, also registered: prothioconazole and tebuconazole (non-selective—apply at low doses or as spot treatments to protect beneficial fungi). Time treatments before wet/cold spells [137]. |
| Late Spring | Scout for early signs of Dollar Spot, Clarireedia spp. (shaded and humid microclimates) | / | Core aerate compacted or thatchy areas to reduce Gaeumannomyces and Clarireedia risk. Maintain balanced N and K to avoid turf stress and reduce Dollar Spot and Anthracnose. | / |
| Early Summer | Monitor disease symptoms; adjust based on canopy humidity. | Apply compost teas, Trichoderma spp., or Pythium oligandrum or other soil microbial stimulants to suppress Take-All Patch and enhance microbial competition in the rhizosphere [137]. | Optimize mowing height; reduce canopy humidity. | Use localized SDHI or DMI fungicides (e.g., boscalid, prothioconazole) for visible Dollar Spot or Anthracnose. Avoid broad-spectrum sprays to protect beneficial fungi [137]. |
| Mid Summer | Inspect for Red Thread, Anthracnose, Dollar Spot; use thresholds. | / | Minimize mowing and nitrogen during heat or drought to limit Anthracnose. | Use spot fungicides only when pressure is high; rotate modes of action to prevent resistance. |
| Late Summer | Evaluate disease progression; assess turf quality. | Reapply biological inoculants like Trichoderma or mycorrhizal blends to outcompete pathogens like Gaeumannomyces [137]. | Overseed with endophyte-enhanced, disease-resistant cultivars; aerate and amend soil organically. | Continue selective fungicide applications for Clarireedia and Colletotrichum spp. if symptom progression threatens turf quality. |
| Autumn | Conduct disease mapping; test soil. | / | Raise mowing height; topdress to protect crowns from winter damage. | Apply preventive fungicides for Microdochium nivale before winter if the risk is high (e.g., cold, moist conditions or past occurrence). |
| Winter | Review disease and input logs to plan ahead. | / | Limit foot traffic and mechanical damage. | / |
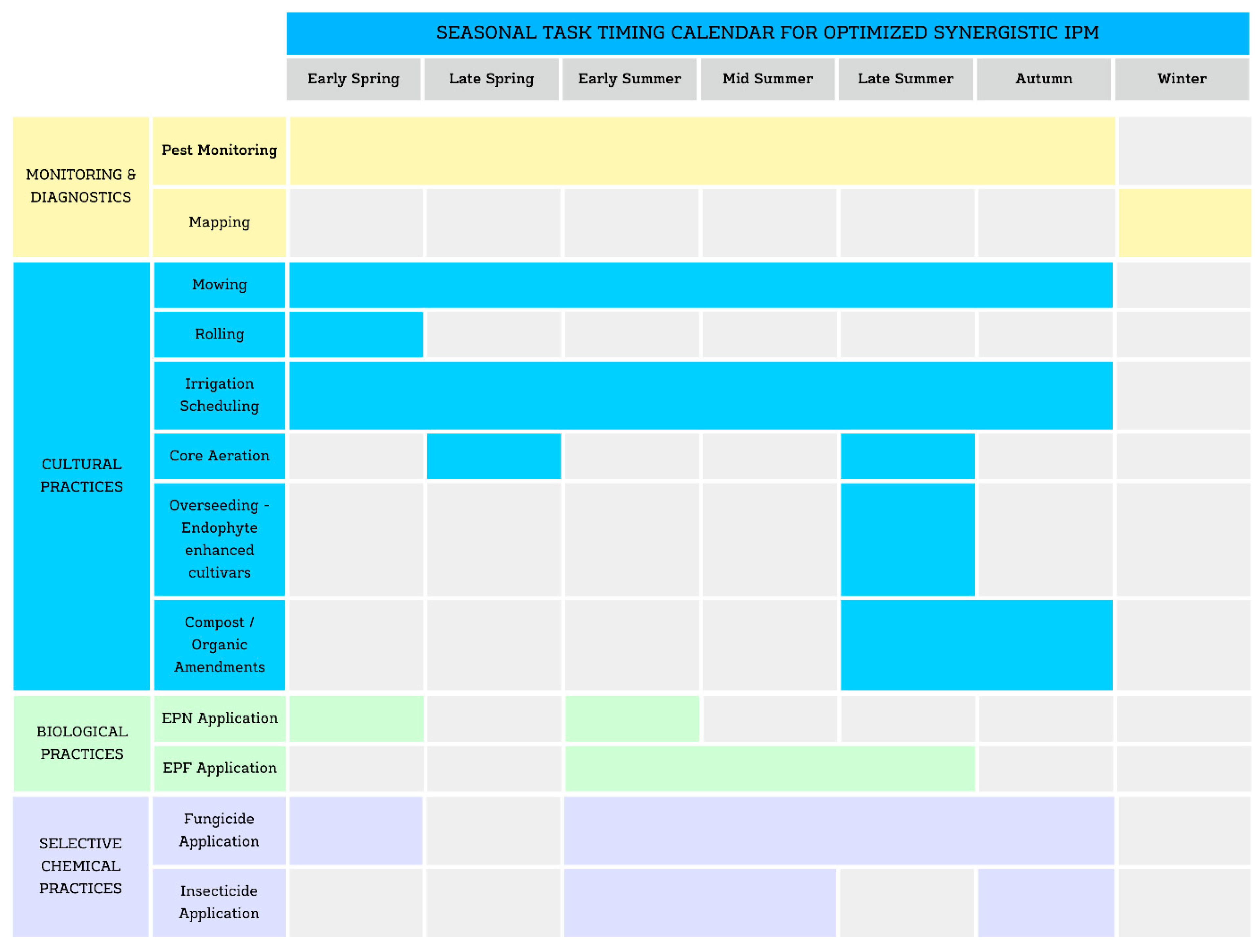
The second component is a seasonal task calendar (
Figure 1), which visualizes how these interventions can be coordinated throughout the turfgrass growing season in Central Europe and other similar climates. By aligning control tactics with pest biology, soil conditions, and operational windows, the calendar provides an intuitive overview that reinforces both timing and integration of the proposed control measures.
Together, the strategic matrix and seasonal calendar ensure clarity in planning across diverse turf systems such as golf courses, football fields, and public lawns. This dual-tool format offers a pragmatic structure for implementing SPM, enhancing decision-making, improving treatment efficiency, and supporting long-term ecological balance [
139].
6.1. SPM Matrix for Tipula spp. (Craneflies/Leatherjackets) Control
Among turfgrass insect pests,
Tipula spp. present distinctive challenges due to their soil-dwelling larvae and preference for cool, moist environments. Monitoring should combine soil sampling with soap flushes and visual inspection in moist, low-cut areas where larvae are most active [
61,
62,
64]. For many turfgrass systems, traditional action thresholds for initiating control are around 80–120 larvae/m
2, though site-specific conditions and turf use may warrant adjustments [
60,
61,
64]. Where EPNs are applied, persistence should be assessed through follow-up soil sampling to verify establishment and inform supplementary measures. The following
Table 1 outlines a seasonally structured SPM strategy that combines cultural, biological, and selective chemical practices to manage populations effectively and sustainably.
This pest-specific framework (
Table 1) illustrates how precise timing and the integration of compatible practices can enhance the effectiveness of
Tipula spp. control while minimizing ecological disruption. Tailored to European turfgrass systems, the framework aligns interventions with local pest phenology and environmental conditions. Incorporating persistence checks for biological agents and adhering to defined damage thresholds can improve decision-making and reduce unnecessary inputs. Emphasis on monitoring thresholds, soil moisture, and temperature cues enables targeted use of biological controls and selective inputs. By reducing chemical dependency and trying to foster long-term turf resilience, this approach could offer a practical, regionally adapted solution for sustainable
Tipula management in Europe, as supported by recent studies (e.g., [
61,
62,
64,
65]).
6.2. SPM Matrix for Chafer Grubs (Scarabaeidae larvae) Control
Chafer grubs, larvae of Scarabaidae beetles such as
Melolontha melolontha,
Amphimallon solstitiale,
Phyllopertha horticola, and the invasive
Popillia japonica, pose substantial threats to turfgrass through root feeding and canopy thinning. Effective, sustainable management depends on a seasonally aligned strategy that integrates monitoring, cultural adjustments, biological agents, and targeted chemical priming [
139]. Young larval presence after adult flights can be tracked by combining pheromone trap counts with follow-up soil sampling in known oviposition areas, typically 2–3 weeks after peak adult activity [
57,
59,
88]. Early detection is essential as EPN efficacy is highest against first-instar larvae under adequate soil moisture [
85,
89]. The following table outlines a pest-specific, SPM framework that adapts interventions to grub phenology and environmental windows.
This seasonal framework (
Table 2) also shows how aligning control efforts with larval vulnerability and favorable soil conditions enhances the effectiveness of chafer grub management. Tailored to European turfgrass systems, it emphasizes the integration of entomopathogenic nematodes, fungal agents, and reduced-risk inputs alongside soil health practices. Incorporating pheromone-based adult monitoring with timed soil inspections for early instars ensures interventions are optimally placed in the pest life cycle, improving reliability while avoiding unnecessary inputs [
57,
59,
85,
89]. By coordinating these tactics seasonally, the approach could offer a practical, ecologically informed roadmap for sustainable and resilient white grub control in Europe, as supported by various studies (e.g., [
34,
43,
47,
51,
57,
59,
85,
88,
89,
140,
141]).
6.3. SPM Matrix for Major Turfgrass Fungal Pathogens
In temperate Central European climates such as Slovenia’s, turfgrass fungal pathogens, including
Microdochium nivale,
Clarireedia spp.,
Laetisaria fuciformis,
Gaeumannomyces graminis var.
avenae, and
Colletotrichum spp., all pose persistent threats to both aesthetic and functional turf quality [
6,
68]. Cool, wet springs and fluctuating summer conditions create extended windows of disease pressure, particularly on intensively managed surfaces like golf greens, sports pitches, and ornamental lawns [
6,
68]. The following table outlines a seasonally structured, SPM plan, tailored for Central European growing conditions, integrating cultural practices, biological enhancements, and judicious fungicide use to achieve durable pathogen suppression.
By combining timely diagnostics, well-timed fungicide applications, and biologically supportive soil management, this seasonally structured SPM approach, as presented in
Table 3, provides effective control of key turfgrass fungal pathogens under Central European conditions. The strategy supports disease reduction while maintaining turf function and aligns with core principles of IPM relevant to Slovenia and similar temperate regions. By coordinating these tactics seasonally, the approach could offer a practical, ecologically informed roadmap for sustainable and resilient disease control in Europe, as supported by different studies (e.g., [
7,
67,
74,
78,
99,
114,
128,
142,
143]).
6.4. Registered Biological Control Products for Tipula spp. and Scarabaeidae in Slovenia
Table 4 presents biological control products currently registered in Slovenia for the management of
Tipula spp. (leatherjackets) and Scarabaeidae (chafer grubs) in turfgrass systems. Product listings are based on the FITO-INFO database, maintained by the Administration for Food Safety, Veterinary Sector, and Plant Protection of Slovenia [
137], and the official plant protection portal [
138]. The table consolidates entomopathogenic nematodes and other biological agents with official registration for use against these target pests, with details on organism, product, target, stage, and optimal soil conditions to support SPM planning.
Details on all available biological and reduced-risk products for turfgrass fungal disease management are already provided in
Table 3, which focuses on pathogen control.
These registered biological control agents can be incorporated into the seasonal SPM frameworks outlined in
Table 1 and
Table 2, ensuring applications are timed to pest vulnerability and environmental conditions. Data are from the FITO-INFO database and Slovenian plant protection guidelines [
137,
138].
6.5. SPM Task Calendar for Major Turfgrass Pests
The seasonal task calendar (
Figure 1) serves as a practical, time-based guide that translates the strategic implementation matrix into a chronological sequence of actions. It outlines seasonally timed interventions targeting key turf pests using integrated combinations of monitoring, biological, cultural, and selective chemical tactics. Tasks are aligned with pest biology and prevailing environmental conditions, with emphasis on compatibility and timing to achieve synergistic effects.
The calendar captures essential activities such as aeration, mowing, irrigation scheduling, and the application of biological agents (e.g.,
Steinernema spp.,
Heterorhabditis spp.,
Beauveria spp.,
Metarhizium spp.) [
84,
86,
100], alongside selective, well-timed chemical inputs. Its structure shows how these actions can be sequenced or layered. For example, matching nematode applications to optimal soil moisture and larval stages [
51,
140], or scheduling fungicide treatments to avoid interference with beneficial organisms [
114].
The purpose of the seasonal task calendar is to provide turf managers with an intuitive, visual tool that links what to do with when to do it. This improves the precision and consistency of interventions, facilitates proactive rather than reactive management, and ensures that control measures work together rather than in isolation [
104,
144]. By embedding timing and integration into a single framework, the calendar helps maintain turf function and aesthetics while supporting the ecological and operational goals of SPM.
Pest and pathogen complexes differ between regions, and in Slovenia, there are currently no registered chemical pesticides for turf-infesting insect groups. Their inclusion in the calendar is therefore for completeness within a whole-based SPM approach, enabling adaptation for regions where such products are available. In these cases, implementation is guided by local authorizations, pest monitoring data, and region-specific best practices. Official decision-support resources, such as Slovenia’s FITO-INFO system [
137] and the national Biotic Plant Protection guidelines [
138], help ensure compliance, effective timing, and science-based integration of biological and other compatible measures. Successes in Japan and South Korea illustrate that the approach can deliver benefits outside Europe [
31,
32].
7. Selected Case Studies of Synergistic IPM Applications
IPM increasingly capitalizes on synergistic interactions between biological, cultural, and chemical tactics to enhance pest suppression while reducing reliance on intensive chemical inputs [
15,
25]. When used strategically, these combinations often result in greater efficacy than any single method alone, particularly in complex systems like turfgrass, where environmental variability and pest resilience challenge traditional control measures [
15,
103].
This section presents selected case studies across multiple domains, including entomopathogenic nematodes, fungal and bacterial biocontrol agents, reduced-risk fungicides, and cultural practices, each targeting a representative pest or pathogen group relevant to turfgrass systems. The aim is to illustrate the principles and outcomes of interactions reported in the literature, rather than to prescribe specific treatments for all regions.
In this review, the term “synergistic” is used only when the original study statistically demonstrated a greater-than-additive effect. In some published studies, claimed synergy may in fact be additive or inconclusive. This reflects inconsistencies in how synergy is defined or tested in the wider IPM literature and does not represent a misinterpretation in the present review. Where statistical confirmation was lacking, we have described the interaction as “combined”, “additive”, or “improved” rather than “synergistic.” This distinction is applied consistently throughout the case studies (
Section 7) and the implementation frameworks to ensure accuracy.
Where chemical components are involved, their regulatory status may differ between regions, and use should comply with local legislation. The examples, drawn from laboratory, greenhouse, and field research, highlight how application timing, formulation compatibility, and ecological context influence results.
While the central focus remains on different synergies in turfgrass-specific IPM systems, the limited availability of published synergy studies, particularly for European white grub species such as
Melolontha melolontha and
Amphimallon solstitiale, and for fungal pathogens like
Laetisaria fuciformis and
Colletotrichum spp., necessitated the inclusion of select case studies from horticultural and agricultural systems. These were chosen based on their relevance to specific pest groups of interest and their potential applicability to turfgrass contexts. Collectively, they offer transferable insights into integrated strategies that optimize pest and disease suppression through synergistic approaches. It should be noted that while this section discusses a broad range of agents and combinations for completeness, the practical implementation framework provided in
Section 6 focuses on products and strategies currently available and permitted in the European Union.
Table 5 outlines documented synergistic, additive, and combined interactions targeting
Tipula paludosa, the European crane fly, using combinations of biological, cultural, and chemical controls. Results from lab, greenhouse, and field studies indicate enhanced larval mortality through strategies such as timing optimization, dual entomopathogen applications, and environmental manipulation (e.g., mowing height). Positive effects were most consistently observed in early instar larvae, particularly at lower temperatures and in biologically diverse formulations (
Table 5). However, outcomes varied depending on environmental context, suggesting field scalability remains complex.
Several studies show that combining biological, cultural, and chemical tactics can improve
Tipula spp. control. However, field results are inconsistent. Laboratory synergies, such as
S. carpocapsae +
Bti, often fail outdoors due to fluctuating soil temperature, desiccation, or UV stress [
62,
64,
65,
82,
146]. Similar losses in efficacy have been reported in forestry and agricultural trials when environmental conditions reduced persistence of nematodes or bacteria [
132,
148]. Some combinations also produce neutral or antagonistic effects, for example, when nematodes are applied with incompatible chemicals or entomopathogenic fungi [
132,
133,
134]. In the EU, many chemical substances are not approved for turfgrass [
14,
137,
138], requiring substitution with registered products and site-specific testing before adoption.
Table 6 compiles synergistic, additive, and combined interactions targeting Scarabaeidae larvae (white grubs), primarily in turfgrass and related horticultural systems. Numerous studies report that entomopathogenic nematodes (EPNs) such as
Heterorhabditis bacteriophora exhibit enhanced larvicidal efficacy when combined with fungal pathogens (
Metarhizium anisopliae,
Beauveria bassiana) or insecticides such as imidacloprid or chlorantraniliprole (
Table 6). Environmental manipulations, including increased mowing height and soil aeration, further improved EPN efficacy by optimizing soil microclimate and larval contact (
Table 6). These studies underscore the potential for integrated, cost-effective grub control, especially under moisture-sensitive or warm soil conditions. However, SPM studies specifically targeting European chafer species, such as
Melolontha melolontha or
Amphimallon solstitiale, remain scarce, limiting region-specific conclusions.
Biological–biological and chemical–biological combinations often improve grub mortality in trials [
85,
89,
91,
93,
149]. Yet, success is less certain in European turf. Cool soils, low moisture, and late-instar larvae can reduce control [
34,
57,
84]. Some trials, including those in agricultural crops, found no improvement over single treatments when timing or soil conditions were suboptimal. Many effective chemicals, such as neonicotinoids, are banned for turfgrass in the EU [
14,
137,
138]. Product availability also varies between member states [
79,
84]. Under these limits, cultural–biological approaches and region-specific biological strains become more important [
47,
85].
Table 7 shows synergistic, additive, and enhanced combinations employed against prominent turfgrass fungal pathogens, including
Rhizoctonia solani,
Fusarium spp.,
Microdochium nivale,
Clarireedia spp., and
Gaeumannomyces graminis var.
avenae. Strategies include integrated use of microbial biocontrol agents (e.g.,
Trichoderma,
Bacillus subtilis,
Pseudomonas) with organic amendments, reduced-risk fungicides, and cultural practices like rolling and topdressing
Table 7). Enhanced pathogen suppression and improved turf quality were commonly observed, particularly when treatments stimulated beneficial soil microbial communities or triggered plant defense responses (
Table 7). Despite this progress, no synergistic studies were found for key turf pathogens
Laetisaria fuciformis and
Colletotrichum spp., indicating a clear research gap in fungal IPM under turf conditions.
Cultural–biological–chemical combinations, such as rolling plus fungicide or
Trichoderma +
Bacillus subtilis, can suppress diseases [
67,
114,
156,
160,
162]. However, results are not always reliable. In turf and in crop systems such as cereals and vegetables, some fungicides have reduced beneficial fungi when applied too close to biocontrol agents [
84,
114]. No synergy studies exist for
Laetisaria fuciformis or
Colletotrichum spp., leaving a gap in fungal IPM research [
6,
68]. EU rules limit the fungicide classes and microbial products available [
14,
137,
138]. Programs, therefore, require careful sequencing, legal compliance, and more region-specific trials to confirm compatibility.
8. Conclusions and Future Directions
The shift toward sustainable pest management in European turfgrass systems mirrors broader environmental, regulatory, and societal changes shaping the future of urban green infrastructure. As regulatory frameworks tighten and expectations for low-input yet high-performance turf intensify, practitioners across sports fields, golf courses, and residential lawns are turning to SPM. These strategies, which combine biological, cultural, and, when contextually justified, chemical controls, have demonstrated clear advantages over single-mode interventions.
This review underscores that synergy, particularly biological–biological and biological–cultural interactions, offers consistent improvements in pest suppression, ecological resilience, and soil health. Field and laboratory studies confirm that integrating entomopathogenic nematodes with microbial agents (e.g., Bti, Metarhizium spp.) or enhancing their activity through cultural interventions like aeration, mowing height, and irrigation management, amplifies control efficacy against key pests such as Tipula spp. and Scarabaeidae larvae. Moreover, although chemical–cultural synergies remain less explored in European turf systems due to regulatory constraints, selected studies reveal their strong potential when designed with ecological precision. Tactics such as reduced fungicide inputs paired with rolling or targeted nitrogen application have shown not only improved disease suppression but also enhanced turf quality.
Yet, broad implementation of these SPM systems remains limited. Key barriers include the lack of regionally adapted biocontrol strains, product registration inconsistencies across European Union member states, and technical complexity in coordinating multi-modal strategies. Turfgrass managers may also face logistical hurdles and knowledge gaps, particularly when transitioning from legacy chemical programs to ecologically integrated solutions.
Addressing these challenges demands coordinated investment across research, practitioner training, regulatory adaptation, and commercial product development. Future priorities include expanding region-specific field trials, accelerating approval pathways for compatible biocontrol formulations, and embedding SPM principles within national turf management guidelines and certification schemes.
Looking ahead, climate variability, biodiversity objectives, and increasing public scrutiny of chemical inputs will only intensify the demand for resilient, multi-functional turf systems. SPM, when operationalized with precision and supported by institutional frameworks, offers a coherent and forward-looking foundation. By leveraging ecological interactions and aligning control timing with pest phenology and soil dynamics, it is possible to achieve sustainable, high-performance turfgrass management that aligns with both environmental mandates and professional performance standards.