Sensitivities of Fall Armyworm (Spodoptera frugiperda) Populations in Different Regions of China to Four Bt Proteins
Abstract
1. Introduction
2. Materials and Methods
2.1. Purified Bt Proteins
2.2. FAW Populations
2.3. Bioassay
2.4. Data Analysis
3. Results
3.1. Sensitivity of Different FAW Populations to Vip3A Protein
3.2. Sensitivity of Different FAW Populations to Cry1F Protein
3.3. Sensitivities of Different FAW Populations to Cry1Ab Protein
3.4. Sensitivity of Different FAW Populations to Cry2Ab Protein
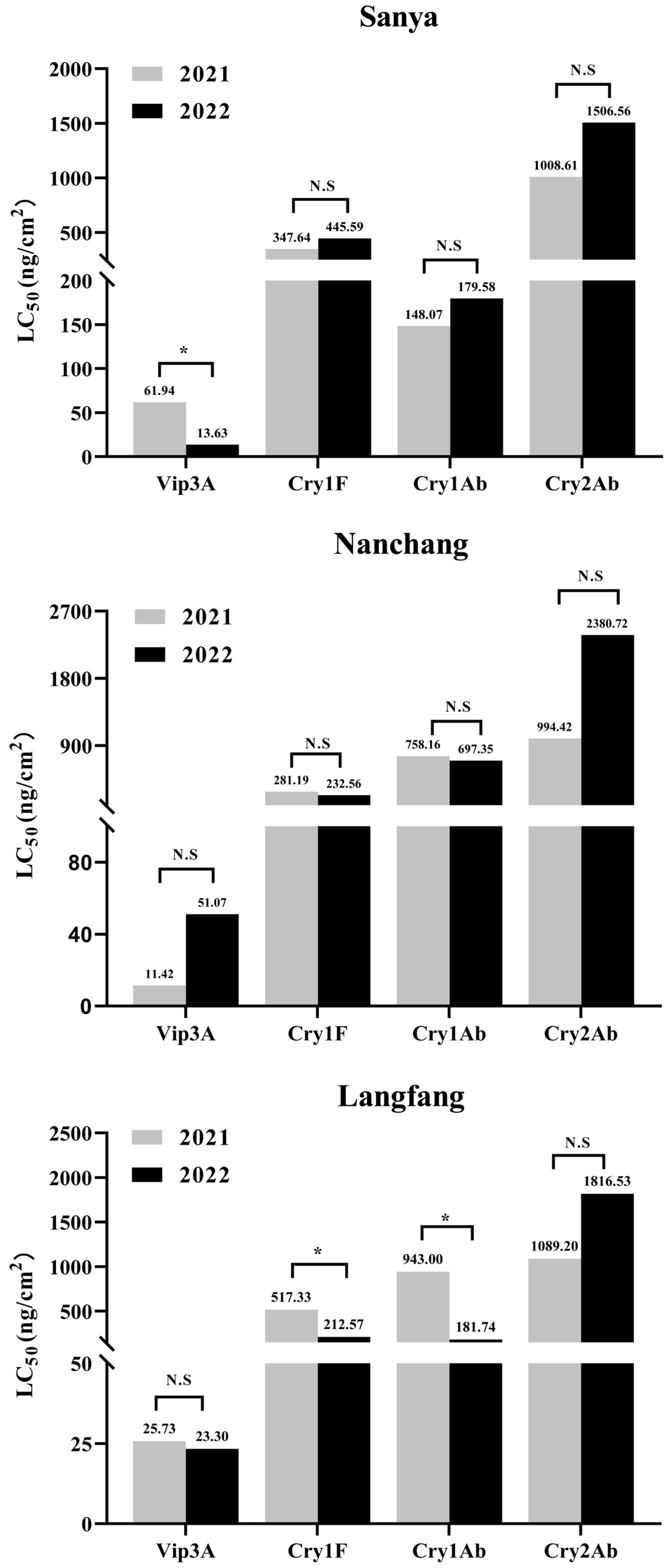
3.5. Sensitivity Comparison of the Three Populations from the Same Location Collected in Different Years
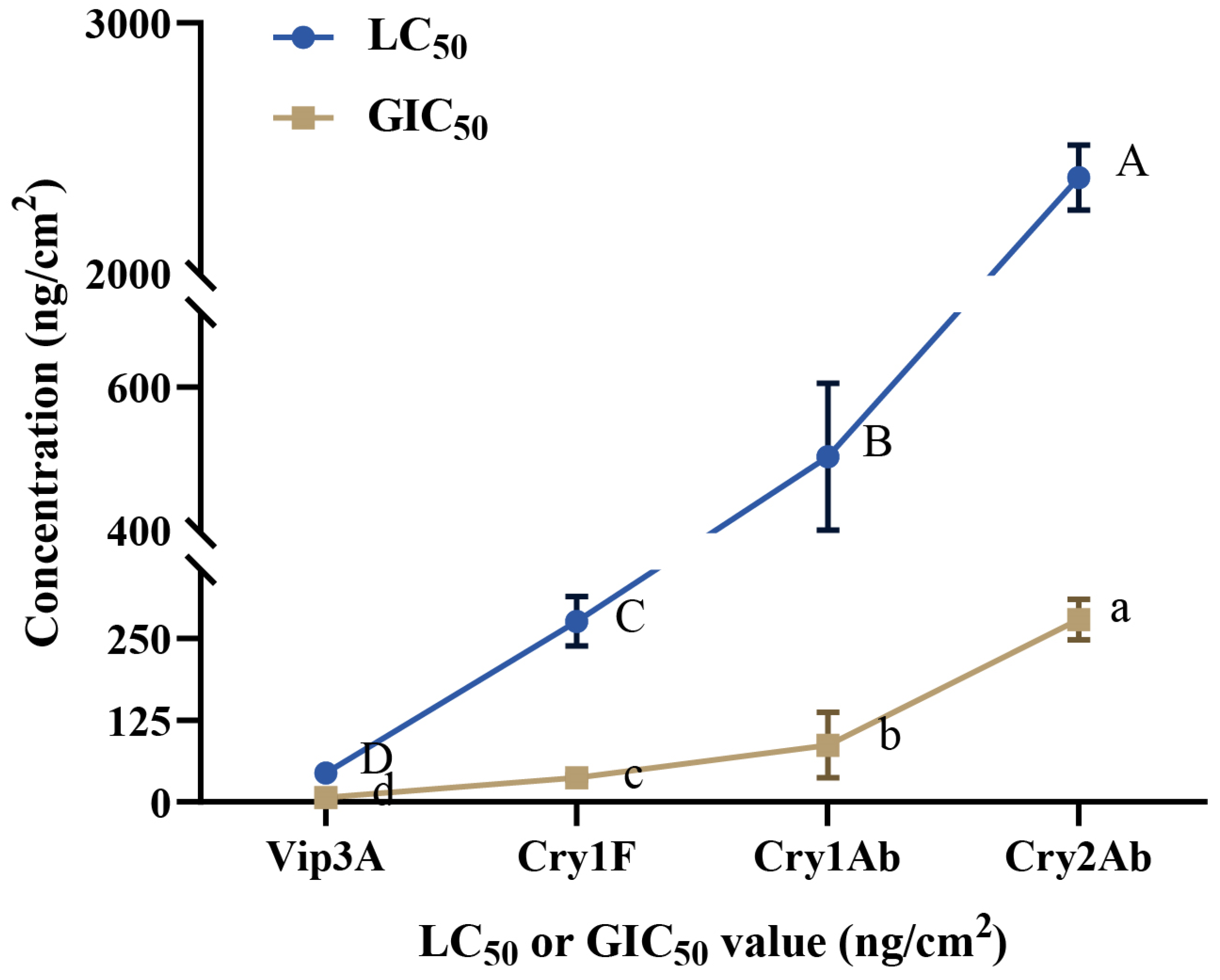
3.6. Sensitivity Comparison among the Four Bt Proteins
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Todd, E.L.; Poole, R.W. Keys and illustrations for the armyworm moths of the noctuid genus Spodoptera Guenée from the western hemisphere. Ann. Entomol. Soc. Am. 1980, 6, 722–738. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; Sousa-Silva, J.C.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef]
- FAO. Integrated Management of the Fall Armyworm on Maize; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017. [Google Scholar]
- Wu, K.M. Management strategies of fall armyworm (Spodoptera frugiperda) in China. Plant Prot. 2020, 46, 1–5. [Google Scholar]
- Zhang, L.; Jin, M.H.; Zhang, D.D.; Jiang, Y.Y.; Liu, J.; Wu, K.M.; Xiao, Y.T. Molecular identification of invasive fall armyworm Spodoptera frugiperda in Yunan province. Plant Prot. 2019, 45, 19–24. [Google Scholar]
- Van den Berg, J.; Prasanna, B.M.; Midega, C.A.O.; Ronald, P.C.; Carrière, Y.; Tabashnik, B.E. Managing fall armyworm in Africa: Can Bt maize sustainably improve control? J. Econ. Entomol. 2021, 114, 1934–1949. [Google Scholar] [CrossRef]
- Gutirrez-Moreno, R.; Mota-Sanchez, D.; Blanco, C.A.; Whalon, M.E.; Terán-Santofimio, H.; Rodriguez-Maciel, J.C.; Difonzo, C. Field-evolved resistance of the fall armyworm (Lepidoptera: Noctuidae) to synthetic insecticides in Puerto Rico and Mexico. J. Econ. Entomol. 2019, 112, 792–802. [Google Scholar] [CrossRef]
- Zhang, D.D.; Xiao, Y.T.; Xu, P.J.; Yang, X.M.; Wu, Q.L.; Wu, K.M. Insecticide resistance monitoring for the invasive populations of fall armyworm, Spodoptera frugiperda in China. J. Integr. Agric. 2021, 20, 783–791. [Google Scholar] [CrossRef]
- Buntin, G.D. Assessment of experimental Bt events against fall armyworm and corn earworm in field corn. J. Econ. Ento. 2004, 97, 259–264. [Google Scholar] [CrossRef]
- Siebert, M.; Leonard, S.; Stewart, J. Evaluation of corn hybrids expressing Cry1F, cry1A.105, Cry2Ab2, Cry34Ab1/Cry35Ab1, and Cry3Bb1 against southern United States insect pests. Econ. Entomol. 2012, 105, 1825–1834. [Google Scholar] [CrossRef]
- Storer, N.P.; Babcock, J.M.; Schlenz, M.; Meade, T.; Thompson, G.D.; Bing, J.W.; Huckaba, R.M. Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J. Econ. Entomol. 2010, 103, 1031–1038. [Google Scholar] [CrossRef]
- Buntin, G.D. Corn expressing Cry1Ab or Cry1F endotoxin for fall armyworm and corn earworm management in field corn for grain production. FLA Entomol. 2008, 91, 523–530. [Google Scholar]
- Bernardi, O. Biological activity of Bt proteins expressed in different structures of transgenic corn against Spodoptera frugiperda. Ciênc. Rural 2016, 46, 1019–1024. [Google Scholar] [CrossRef]
- Omoto, C.; Bernardi, O.; Salmeron, E.; Sorgatto, R.J.; Dourado, P.M.; Crivellari, A.; Carvalho, R.A.; Willse, A.; Martinelli, S.; Head, G.P. Field-evolved resistance to Cry1Ab maize by Spodoptera frugiperda in Brazil. Pest Manag. Sci. 2016, 72, 1727–1736. [Google Scholar] [CrossRef]
- Yang, F.; Kerens, D.L.; Head, G.P.; Price, P.; Huang, F. Cross-Resistance to purified Bt proteins, Bt corn and Bt cotton in a Cry2Ab2-corn resistant strain of Spodoptera frugiperda. Pest Manag. Sci. 2017, 73, 2495–2503. [Google Scholar] [CrossRef]
- Storer, N.P. Status of resistance to Bt maize in Spodoptera frugiperda: Lessons from Puerto Rico. Invertebr. Pathol. 2012, 110, 294–300. [Google Scholar] [CrossRef]
- Farias, J.R.; Andow, D.A.; Horikoshi, R.J.; Sorgatto, R.J.; Fresia, P.; Dos Santos, A.C.; Omoto, C. Field-Evolved resistance to Cry1F maize by Spodoptera frugiperda (Lepidoptera: Noctuidae) in Brazil. Crop Prot. 2014, 64, 150–158. [Google Scholar] [CrossRef]
- Chandrasena, D.I.; Signorini, A.M.; Abratti, G.; Storer, N.P.; Olaciegui, M.L.; Alves, A.P.; Pilcher, C.D. Characterization of field-evolved resistance to Bacillus thuringiensis-derived Cry1F δ-Endotoxin in Spodoptera frugiperda populations from Argentina. Pest Manag. Sci. 2018, 74, 746–754. [Google Scholar] [CrossRef]
- Li, G.P.; Reifig, D.; Miao, J.; Gould, F.; Huang, F.N.; Feng, H.Q. Frequency of Cry1F non-recessive resistance alleles in North Carolina field populations of Spodoptera frugiperda (Lepidoptera: Noctuidae). PLoS ONE 2016, 11, e0154492. [Google Scholar] [CrossRef]
- Fatoretto, J.C.; Michel, A.P.; Filho, M.C.S.; Silva, N. Adaptive potential of fall armyworm (Lepidoptera: Noctuidae) limits bt trait durability in Brazil. J. Integr. Pest Manag. 2017, 8, 17. [Google Scholar] [CrossRef]
- Wen, Z.; Conville, J.; Matthews, P.; Hootman, T.; Himes, J.; Wong, S.; Huang, F.; Ni, X.; Chen, J.S.; Bramlett, M. More than 10 years after commercialization, Vip3A-expressing MIR162 remains highly efficacious in controlling major lepidopteran maize pests: Laboratory resistance selection versus field reality. Pestic. Biochem. Physiol. 2023, 192, 105385. [Google Scholar] [CrossRef]
- Bernardi, O.; Bernardi, D.; Ribeiro, R.S.; Okuma, D.M.; Salmeron, E.; Fatoretto, J.; Medeiros, F.C.L.; Burd, T.; Omoto, C. Frequency of resistance to Vip3Aa20 toxin from Bacillus thuringiensis in Spodoptera frugiperda (Lepidoptera: Noctuidae) populations in Brazil. Crop Prot. 2015, 76, 7–14. [Google Scholar] [CrossRef]
- Zhang, D.D.; Wu, K.M. The bioassay of Chinese domestic Bt-Cry1Ab and Bt-(Cry1Ab+Vip3Aa) maize against the fall armyworm (Spodoptera frugiperda). Plant Prot. 2019, 45, 54–60. [Google Scholar]
- Liang, J.G.; Zhang, D.D.; Li, D.Y.; Zhao, S.Y.; Wang, C.Y.; Xiao, Y.T.; Xu, D.; Yang, Y.Z.; Li, G.P.; Wang, L.L.; et al. Expression profiles of Cry1Ab protein and its insecticidal efficacy against the invasive Fall Armyworm for Chinese domestic GM maize DBN9936. J. Integr. Agric. 2021, 20, 792–803. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, D.; Zhao, S.; Wu, K. Susceptibilities of the invasive fall armyworm (Spodoptera frugiperda) to the insecticidal proteins of Bt maize in China. Toxins 2022, 14, 507. [Google Scholar] [CrossRef]
- Hu, J.; Ding, D.; Bai, S.Y.; Jing, T.X.; Yang, Y.Z. Effect of two transgenic maize on population increase of Spodoptera frugiperda. J. Yangzhou Univ. Agric. Life Sci. Ed. 2023, 44, 75–81. [Google Scholar]
- Tabashnik, B.E.; Mota-Scanchez, D.; Whalon, M.E.; Hollingworth, R.M.; Carrière, Y. Defining terms for proactive management of resistance to Bt crops and pesticides. J. Econ. Entomol. 2014, 107, 496–507. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Carrière, Y. Surge in insect resistance to transgenic crops and prospects for sustainability. Nat. Biotechnol. 2017, 35, 926–935. [Google Scholar] [CrossRef]
- Yue, D.R. Quality Zoning and Industrial Layout of Corn in China; China Agriculture Press: Beijing, China, 2004. [Google Scholar]
- Liang, G.M.; Tan, W.J.; Guo, Y.Y. Improvement of artificial feeding technology of cotton bollworm. Plant Prot. 1999, 25, 16–18. [Google Scholar]
- Robertson, J.L.; Preisler, H.K.; Russell, R.M. PoloPlus, Probit and Logit Analysis: User’s Guide, version 1.0, LeOra Software: Petaluma, CA, USA, 2003.
- Zhang, L.; Liu, B.; Zheng, W.; Liu, C.; Zhang, D.; Zhao, S.; Li, Z.; Xu, P.; Wilson, K.; Withers, A.; et al. Genetic structure and insecticide resistance characteristics of fall armyworm populations invading China. Mol. Ecol. Resour. 2020, 20, 1682–1696. [Google Scholar] [CrossRef]
- Huang, F.N.; Qureshi, J.A.; Meagher, R.L., Jr.; Reisig, D.D.; Head, G.P.; Andow, D.A.; Ni, X.Z.; Kerns, D.; Buntin, G.D.; Niu, Y.; et al. Cry1F resistance in fall armyworm Spodoptera frugiperda: Single gene versus pyramided Bt maize. PLoS ONE 2014, 9, e112958. [Google Scholar] [CrossRef]
- Leite, N.A.; Mendes, S.M.; Santos-Amaya, O.F.; Santos, C.A.; Teixeira, T.P.M.; Guedes, R.N.C.; Pereira, E.J.G. Rapid selection and characterization of Cry1F resistance in a Brazilian strain of fall armyworm. Entomol. Exp. Appl. 2016, 158, 236–247. [Google Scholar] [CrossRef]
- Flagel, L.; Lee, Y.W.; Wanjugi, H.; Swarup, S.; Brown, A.; Wang, J.; Kraft, E.; Greenplate, J.; Simmons, J.; Adams, N.; et al. Mutational disruption of the ABCC2 gene in fall armyworm, Spodoptera frugiperda, confers resistance to the Cry1Fa and Cry1A.105 insecticidal proteins. Sci. Rep. 2018, 8, 7255. [Google Scholar] [CrossRef]
- Figueiredo, C.S.; Lemes, A.R.N.; Sebastião, I.; Desidério, J.A. Synergism of the Bacillus thuringiensis Cry1, Cry2, and Vip3 proteins in Spodoptera frugiperda control. Appl. Biochem. Biotechnol. 2019, 188, 798–809. [Google Scholar] [CrossRef]
- Rivero-Borja, M.; Rodríguez-Maciel, J.C.; Urzúa Gutiérrez, J.A.; Silva-Aguayo, G.; Chandrasena, D.I.; Felix-Bermudez, N.C.; Storer, N.P. Baseline of susceptibility to the Cry1F protein in Mexican populations of fall armyworm. J. Econ. Entomol. 2020, 113, 390–398. [Google Scholar] [CrossRef]
- Abdelgaffar, H.; Perera, O.P.; Jurat-Fuentes, J.L. ABC transpoter mutations in Cry1F-resistant Spodoptera frugiperda do not result in altered susceptibility to selected samll molecule pesticides. Pest Manag. Sci. 2021, 77, 949–955. [Google Scholar] [CrossRef]
- Xu, T.T.; Wang, Y.Q.; Hu, F.; Bi, S.J.; Hu, B.J.; Wang, Z.Y.; Xu, L.N. Susceptibility of different geographical populations of fall armyworm Spodoptera frugiperda in Anhui Province to Bt proteins. J. Plant Prot. 2022, 49, 1521–1527. [Google Scholar]
- Siegfried, B.D.; Spencer, T.; Nearman, J. Baseline susceptibility of the corn earworm (Lepidoptera: Noctuidae) to the Cry1Ab toxin from Bacillus thuringiensis. J. Econ. Entomol. 2000, 93, 1265–1268. [Google Scholar] [CrossRef]
- He, K.; Wang, Z.; Wen, L.; Bai, S.; Ma, X.; Yao, Z. Determination of baseline susceptibility to Cry1Ab protein for Asian corn borer (Lep., Crambidae). J. Appl. Entomol. 2005, 129, 407–412. [Google Scholar] [CrossRef]
- Yang, F.; Morsello, S.; Head, G.P.; Sansone, C.; Huang, F.; Gilreath, R.T.; Kerns, D.L. F2 screen, ingeritance and cross-resistance of field derived Vip3A resistance in Spodoptera frugiperda collected from Louisiana USA. Pest Manag. Sci. 2018, 74, 1769–1778. [Google Scholar] [CrossRef]
- Li, G.P.; Ji, T.J.; Sun, X.X.; Jiang, Y.Y.; Wu, K.M.; Feng, H.Q. Susceptibility evaluation of invaded Spodoptera frugiperda population in Yunnan province to five Bt proteins. Plant Prot. 2019, 45, 15–20. [Google Scholar]
- Ingber, D.A.; Mason, C.E.; Flexner, L. Cry1 Bt susceptibilities of fall armyworm (Lepidoptera: Noctuidae) host strains. J. Econ. Entomol. 2018, 111, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Luttrell, R.G.; Wan, L.; Knighten, K. Variation in susceptibility of Noctuid (Lepidoptera) larvae attacking cotton and soybean to purified endotoxin proteins and commercial formulations of Bacillus thuringiensis. J. Econ. Entomol. 1999, 92, 21–32. [Google Scholar] [CrossRef]
- Archer, T.L.; Patrick, C.; Schuster, G.; Cronholm, G.; Bynum, E.D., Jr.; Morrison, W.P. Ear and shank damage by corn borers and corn earworms to four events of Bacillus thuriengiensis transgenic maize. Crop Prot. 2001, 20, 139–144. [Google Scholar] [CrossRef]
- Buntin, G.D.; Lee, R.D.; Willson, D.M.; McPherson, R.M. Evaluation of yieldgard transgenic resistance for control of fall armywom and corn earworm (Lepidoptera: Noctuidae) on corn. FLA Entomol. 2001, 84, 37–42. [Google Scholar] [CrossRef]
- Buntin, G.D. Corn expressing CrylAb endotoxin for management of fall armywom and corn earwrorm (Lepidoptera: Noctuidae) in silage production. J. Entomol. Sci. 2010, 45, 283–295. [Google Scholar]
- Zhang, J.F.; Jin, M.H.; Yang, Y.C.; Liu, L.L.; Yang, Y.B.; Gómez, I.; Bravo, A.; Soberón, M.; Xiao, Y.T.; Liu, K.Y. The cadherin protein is not involved in susceptibility to Bacillus thuringiensis Cry1Ab or Cry1Fa toxins in Spodoptera frugiperda. Toxins 2020, 12, 375. [Google Scholar] [CrossRef]
- Guan, F.; Zhang, J.; Shen, H.; Wang, X.; Padovan, A.; Walsh, T.K.; Tay, W.T.; Gordon, K.H.J.; James, W.; Czepak, C.; et al. Wholegenome sequencing to detect mutations associated with resistance to insecticides and Bt proteins in Spodoptera frugiperda. Insect Sci. 2021, 28, 627–638. [Google Scholar] [CrossRef]
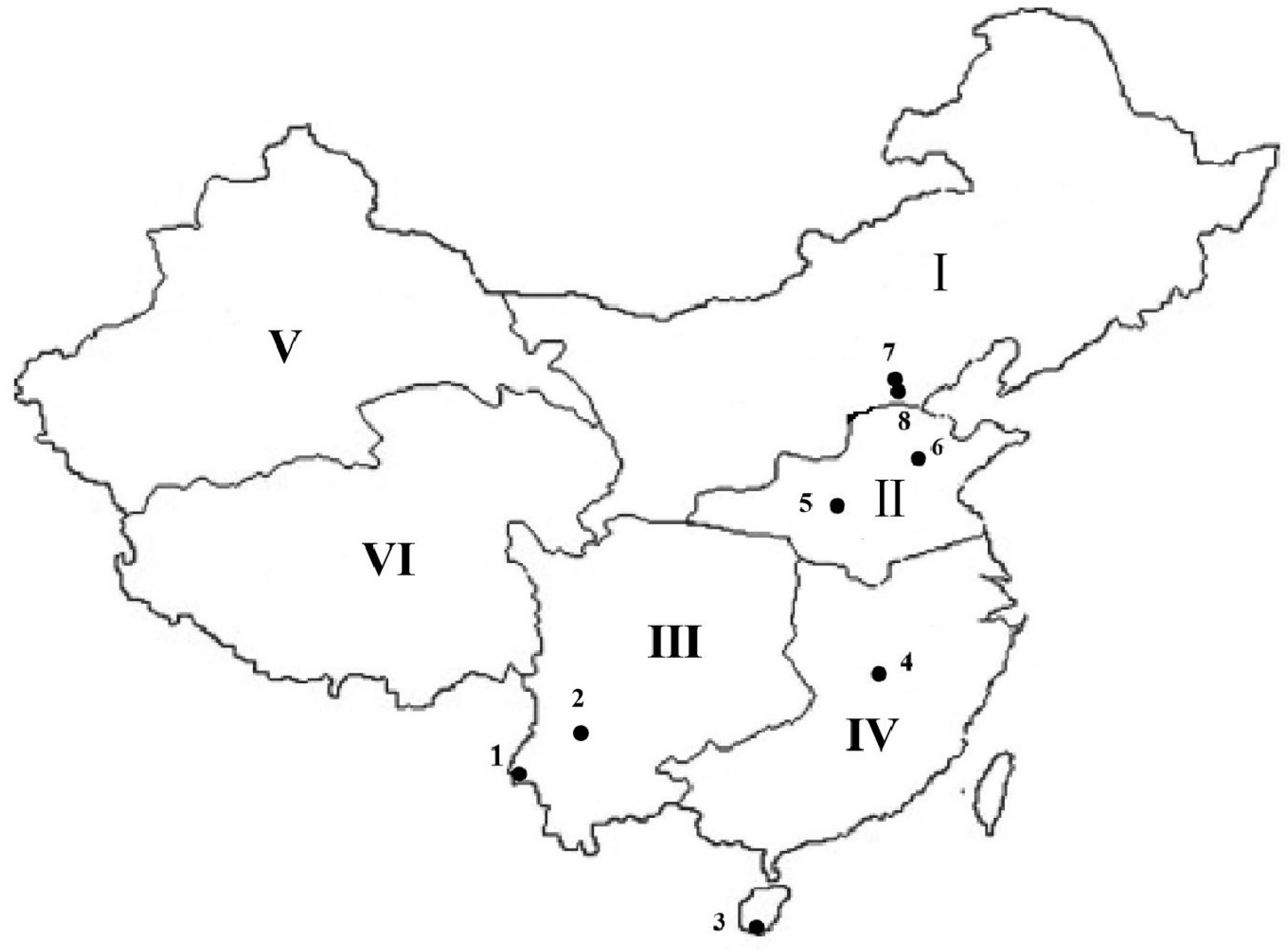
| Location | Coordinates | Map Ref. No. | Collection Date | No. of Larvae Collected | ||
|---|---|---|---|---|---|---|
| Production Region | Province | City | ||||
| Southwest Hilly Corn Region | Yunnan | Dehong | 24.01° N, 97.82° E | 1 | August 2021 | 120 |
| Dali | 25.23° N, 100.24° E | 2 | May 2022 | 183 | ||
| Southern Hilly Corn Region | Hainan | Sanya | 18.38° N, 109.20° E | 3 | July 2021, July 2022 | 159, 188 |
| Jiangxi | Nanchang | 28.33° N, 115.56° E | 4 | August 2021, August 2022 | 239 | |
| Huang–Huai–Hai Summer Corn Region | Henan | Yuanyan | 35.06° N, 113.96° E | 5 | August 2021 | 130 |
| Shandong | Jinan | 36.39° N, 117.01° E | 6 | August 2021 | 218 | |
| Northern Spring Corn Region | Hebei | Langfang | 39.53° N, 116.70° E | 7 | September 2021, July 2022 | 76, 65 |
| Beijing | Tongzhou | 39.90° N, 116.65° E | 8 | September 2022 | 138 | |
| Population | N * | LC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 | GIC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 |
|---|---|---|---|---|---|---|---|
| Dehong2021 | 720 | 88.33 (41.64–139.19) a | 2.42 ± 0.32 | 19.11 | 2.36 (1.21–3.79) de | 0.85 ± 0.08 | 7.08 |
| Dali2022 | 720 | 46.52 (24.54–79.45) abc | 1.88 ± 0.18 | 28.26 | 7.05 (4.12–10.54) abc | 1.26 ± 0.08 | 19.96 |
| Sanya2021 | 720 | 61.94 (40.34–86.91) ab | 2.49 ± 0.25 | 14.38 | 4.91 (0.37–13.34) abcde | 0.78 ± 0.07 | 56.65 |
| Sanya2022 | 720 | 13.63 (7.75–19.05) e | 2.70 ± 0.35 | 10.09 | 13.66 (8.27–21.40) ab | 1.64 ± 0.10 | 41.14 |
| Nangchan2021 | 720 | 11.42 (4.59–21.30) ce | 1.28 ± 0.10 | 33.03 | 7.37 (4.70–10.49) abc | 1.07 ± 0.08 | 14.91 |
| Nanchang2022 | 720 | 51.07 (20.81–77.24) abc | 3.91 ± 0.55 | 24.44 | 1.94 (0.40–4.25) de | 0.88 ± 0.08 | 22.08 |
| Yuanyan2021 | 720 | 68.07(39.44–96.71) ab | 2.25 ± 0.30 | 10.42 | 2.09 (0.98–3.51) de | 1.24 ± 0.09 | 7.28 |
| Jinan2021 | 720 | 20.12 (9.62–33.40) cde | 1.96 ± 0.17 | 29.46 | 14.86 (9.16–22.53) a | 1.43 ± 0.09 | 30.96 |
| Tongzhou2022 | 720 | 73.83 (43.91–122.63) a | 1.07 ± 0.13 | 7.19 | 14.03 (6.35–25.03) ac | 0.66 ± 0.06 | 17.06 |
| Langfang2021 | 720 | 25.73 (11.10–42.78) bcde | 1.70 ± 0.17 | 20.71 | 1.43 (0.77–2.21) e | 0.96 ± 0.10 | 5.75 |
| Langfang2022 | 720 | 23.30 (7.26–40.11) cde | 1.75 ± 0.23 | 14.33 | 5.06 (2.80–7.76) cd | 0.98 ± 0.07 | 12.11 |
| Population | N * | LC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 | GIC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 |
|---|---|---|---|---|---|---|---|
| Dehong2021 | 720 | 262.11 (109.78–469.77) abc | 1.16 ± 0.14 | 8.77 | 138.97 (26.27–316.84) a | 0.81 ± 0.14 | 35.41 |
| Dali2022 | 720 | 174.71 (96.42–289.03) bc | 1.19 ± 0.09 | 22.90 | 3.24 (0.25–10.61) bc | 0.65 ± 0.08 | 10.59 |
| Sanya2021 | 720 | 347.64 (196.91–547.35) ab | 1.38 ± 0.12 | 14.50 | 2.50 (0.14–9.38) bc | 0.53 ± 0.07 | 10.52 |
| Sanya2022 | 720 | 445.59 (75.93–1017.25) abc | 1.03 ± 0.14 | 22.46 | 67.65 (35.92–108.02) a | 0.82 ± 0.07 | 13.59 |
| Nangchan2021 | 720 | 281.19 (185.73–382.71) abc | 1.61 ± 0.19 | 4.18 | 82.83 (41.98–137.16) a | 1.21 ± 0.08 | 33.45 |
| Nanchang2022 | 720 | 232.56 (61.70–551.02) abc | 0.93 ± 0.09 | 28.42 | 2.35 (0.25–7.86) c | 0.65 ± 0.08 | 6.39 |
| Yuanyan2021 | 720 | 141.23 (51.50–283.01) bc | 1.13 ± 0.09 | 33.16 | 17.05 (7.54–29.55) abc | 0.76 ± 0.07 | 7.57 |
| Jinan2021 | 720 | 111.21 (49.42–196.63) c | 1.22 ± 0.11 | 18.66 | 42.73 (8.91–80.74) ab | 1.07 ± 0.08 | 55.98 |
| Tongzhou2022 | 720 | 308.56 (209.88–406.54) ab | 2.24 ± 0.26 | 7.84 | 27.90 (7.66–57.27) abc | 0.77 ± 0.07 | 20.52 |
| Langfang2021 | 720 | 517.33 (379.99–699.84) a | 1.38 ± 0.11 | 3.87 | 17.22 (9.94–25.94) b | 0.86 ± 0.08 | 5.83 |
| Langfang2022 | 720 | 212.57 (122.11–336.49) bc | 1.08 ± 0.09 | 10.04 | 8.09 (3.59–14.23) bc | 0.77 ± 0.08 | 5.30 |
| Population | N * | LC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 | GIC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 |
|---|---|---|---|---|---|---|---|
| Dehong2021 | 720 | 541.85 (273.04–1059.28) abc | 0.96 ± 0.11 | 17.40 | 2.59 (0.25–8.88) b | 0.36 ± 0.06 | 4.88 |
| Dali2022 | 720 | 434.79 (300.40–594.88) bc | 2.18 ± 0.19 | 16.43 | 14.22 (3.45–31.35) b | 0.71 ± 0.07 | 14.31 |
| Sanya2021 | 720 | 148.07 (68.38–255.29) de | 1.13 ± 0.12 | 8.82 | 12.56 (3.28–27.40) b | 0.65 ± 0.07 | 10.83 |
| Sanya2022 | 720 | 179.58 (127.54–240.41) e | 1.82 ± 0.26 | 3.28 | 1.58 (0.038–7.65) b | 0.42 ± 0.68 | 4.83 |
| Nangchan2021 | 720 | 758.16 (496.30–1039.59) ab | 1.40 ± 0.19 | 4.11 | 4.85 (0.10–34.08) b | 0.31 ± 0.07 | 9.12 |
| Nanchang2022 | 720 | 697.35 (492.16–926.19) ab | 1.56 ± 0.23 | 1.52 | 11.77 (1.33–33.58) b | 0.51 ± 0.08 | 6.66 |
| Yuanyan2021 | 720 | 135.76 (71.85–212.63) e | 1.11 ± 0.11 | 8.40 | 10.34 (0.99–35.52) b | 0.69 ± 0.10 | 4.88 |
| Jinan2021 | 720 | 401.30 (240.87–642.05) bcd | 1.61 ± 0.12 | 28.36 | 31.72 (5.87–77.57) b | 0.70 ± 0.08 | 12.41 |
| Tongzhou2022 | 720 | 1108.47 (780.35–1438.36) a | 2.15 ± 0.32 | 8.08 | 464.86 (323.66–635.77) a | 0.90 ± 0.07 | 8.38 |
| Langfang2021 | 720 | 943.00 (457.67–1474.74) ab | 1.61 ± 0.12 | 17.64 | 372.56 (203.35–602.15) a | 1.26 ± 0.08 | 33.53 |
| Langfang2022 | 720 | 181.74 (32.08–406.36) cde | 1.12 ± 0.14 | 18.46 | 28.36 (0.10–134.23) b | 0.60 ± 0.06 | 28.46 |
| Population | N * | LC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 | GIC50 (95% FL, ng/cm2) § | Slope ± SE | χ2 |
|---|---|---|---|---|---|---|---|
| Dehong2021 | 720 | 2781.24 (1557.35–7184.05) abc | 1.27 ± 0.17 | 18.35 | 296.41 (178.72–440.95) bc | 0.85 ± 0.07 | 10.69 |
| Dali2022 | 720 | 5492.50 (4087.30–9106.10) a | 1.86 ± 0.34 | 5.72 | 304.57 (221.58–419.87) bc | 1.19 ± 0.09 | 15.45 |
| Sanya2021 | 720 | 1008.61 (54.11–1357.48) de | 2.02 ± 0.29 | 7.46 | 117.94 (82.22–161.32) de | 0.89 ± 0.06 | 8.16 |
| Sanya2022 | 720 | 1506.56 (1231.76–1776.26) cde | 2.95 ± 0.39 | 5.23 | 67.33 (48.86–88.39) de | 1.14 ± 0.08 | 7.77 |
| Nangchan2021 | 720 | 994.42 (725.19–1290.45) e | 2.86 ± 0.28 | 14.33 | 96.18 (67.49–118.71) de | 1.16 ± 0.08 | 8.42 |
| Nanchang2022 | 720 | 2380.72 (1033.92–4711.16) abcde | 1.81 ± 0.36 | 15.22 | 490.15 (326.75–693.32) b | 0.76 ± 0.06 | 7.74 |
| Yuanyan2021 | 720 | 4489.60 (2067.40–22081.00) ab | 1.02 ± 0.10 | 25.67 | 1266.07 (877.72–1817.75) a | 1.27 ± 0.07 | 30.75 |
| Jinan2021 | 720 | 3231.40 (1905.90–7364.20) ab | 1.10 ± 0.10 | 14.55 | 224.54 (52.33–493.20) bcd | 0.75 ± 0.06 | 35.06 |
| Tongzhou2022 | 720 | 1409.79 (990.00–1871.47) cde | 1.59 ± 0.26 | 6.99 | 152.95 (60.22–277.95) ce | 0.75 ± 0.07 | 15.36 |
| Langfang2021 | 720 | 1089.20 (382.21–2194.60) cde | 1.19 ± 0.17 | 19.49 | 25.01 (6.42–54.09) df | 0.55 ± 0.06 | 12.08 |
| Langfang2022 | 720 | 1816.53 (1350.92–2782.32) bcd | 2.17 ± 0.39 | 6.86 | 24.17 (13.35–37.12) f | 0.77 ± 0.07 | 8.47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Y.; Huang, C.; Chen, Y.; Han, L.; Xie, J.; Chen, X. Sensitivities of Fall Armyworm (Spodoptera frugiperda) Populations in Different Regions of China to Four Bt Proteins. Agronomy 2023, 13, 2415. https://doi.org/10.3390/agronomy13092415
Zhou Y, Huang C, Chen Y, Han L, Xie J, Chen X. Sensitivities of Fall Armyworm (Spodoptera frugiperda) Populations in Different Regions of China to Four Bt Proteins. Agronomy. 2023; 13(9):2415. https://doi.org/10.3390/agronomy13092415
Chicago/Turabian StyleZhou, Yanfang, Chunmeng Huang, Yi Chen, Lanzhi Han, Jiajian Xie, and Xiuping Chen. 2023. "Sensitivities of Fall Armyworm (Spodoptera frugiperda) Populations in Different Regions of China to Four Bt Proteins" Agronomy 13, no. 9: 2415. https://doi.org/10.3390/agronomy13092415
APA StyleZhou, Y., Huang, C., Chen, Y., Han, L., Xie, J., & Chen, X. (2023). Sensitivities of Fall Armyworm (Spodoptera frugiperda) Populations in Different Regions of China to Four Bt Proteins. Agronomy, 13(9), 2415. https://doi.org/10.3390/agronomy13092415





