Alleviation of Water-Deficit Stress on Seed Germination of Barley and Fenugreek in a Sandy Soil Using Superabsorbent Polymer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Soils
2.1.1. Soil Characteristics
2.1.2. Seed Germination
Final Germination Percentage (FGP)
Germination Index (GI)
+ (number of germinated seeds in the final count/days of final count).
Germination Characterization
The Water Germination Efficiency (WGE)
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Awad, A.A.M.; Rady, M.M.; Semida, W.M.; Belal, E.E.; Omran, W.M.; Al-Yasi, H.M.; Ali, E.F. Foliar nourishment with different zinc-containing forms effectively sustains carrot performance in zinc-deficient soil. Agronomy 2021, 11, 1853. [Google Scholar] [CrossRef]
- Awad, A.A.M.; El-Taib, A.B.A.; Sweed, A.A.A.; Omran, A.A.M. Nutrient contents and productivity of Triticum aestivum plants grown in clay loam soil depending on humic substances and varieties and their interactions. Agronomy 2022, 12, 705. [Google Scholar] [CrossRef]
- Abdallah, A.M. Influence of hydrogel type and concentration, and water application rate on some hydraulic properties of a sandy soil. Alex. Sci. Exch. J. 2019, 40, 349–362. [Google Scholar] [CrossRef]
- Beacham, A.M.; Vickers, L.H.; Monaghan, J.M. Vertical farming: A summary of approaches to growing skywards. J. Hortic. Sci. Biotechnol. 2019, 94, 277–283. [Google Scholar] [CrossRef]
- Elshefie, H.S.; Camele, I. Applications of Absorbent Polymers for Sustainable Plant Protection and Crop Yield. Sustainability 2021, 13, 3253. [Google Scholar] [CrossRef]
- Huttermann, A.L.; Orikiriza, J.B.; Agaba, H.Y. Application of superabsorbent polymers for improving the ecological chemistry of degraded or polluted lands. CLEAN Soil Air Water 2009, 37, 517–526. [Google Scholar] [CrossRef]
- Li, X.; He, J.Z.; Hughes, J.M.; Liu, Y.R.; Zheng, Y.M. Effects of super-absorbent polymers on a soil-wheat (Triticum aestivum L.) system in the field. Appl. Soil Ecol. 2014, 73, 58–63. [Google Scholar] [CrossRef]
- Cheng, W.-M.; Hu, X.; Wang, D.-M.; Liu, G.-H. Preparation and characteristics of corn straw-co-amps-co-a superabsorbent hydrogel. Polymers 2015, 7, 2431–2445. [Google Scholar] [CrossRef]
- Ostrand, M.S.; DeSutter, T.M.; Daigh, A.L.M.; Limb, R.F.; Steele, D.D. Superabsorbent polymer characteristics, properties, and applications. Agrosyst. Geosci. Environ. 2020, 3, e20074. [Google Scholar] [CrossRef]
- Justs, J.; Wyrzykowski, M.; Winnefeld, F.; Bajare, D.; Lura, P. Influence of superabsorbent polymers on hydration of cement pastes with low water-to-binder ratio. J. Therm. Anal. Calorim. 2014, 115, 425–432. [Google Scholar] [CrossRef]
- Calo, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Palacios-Romero, A.; Rodriguez-Laguna, R.; Razo-Zarate, R.; Meza-Rangel, J.; Prieto-Garcia, F.; Hernandez-Flores, M.D. Survival of plants of pinus leiophylla schiede ex schltdl. & charm., by adding water reservoirs at transplanting in a greenhouse. Rev. Chapingo Ser. Cienc. For. Ambient. 2017, 23, 35–45. [Google Scholar]
- Song, X.; Zhu, C.; Fan, D.; Mi, Y.; Li, X.; Fu, R.; Duan, Z.; Wang, Y.; Feng, R. A novel human-like collagen hydrogel scaffold with porous structure and sponge-like properties. Polymers 2017, 9, 638. [Google Scholar] [CrossRef]
- Lejcus, K.; Dabrowska, J.; Grzybowska-Pietras, J.; Garlikowski, D.; Lejcus, I.; Pawlowski, A.; Spitalniak, M. Optimisation of operational parameters for nonwoven sheaths of water absorbing geocomposites in unsaturated soil conditions. Fibres Text. East. Eur. 2016, 24, 110–116. [Google Scholar] [CrossRef]
- El Hady, O.A.; Tayel, M.Y.; Lofty, A.A. Super gel as a soil conditioner II—Its effects on plant growth, enzyme activity, water use efficiency and nutrient uptake. Acta Hortic. 1981, 12, 257–265. [Google Scholar] [CrossRef]
- Abrisham, E.S.; Jafari, M.; Tavili, A.; Rabii, A.; Chahoki, M.A.Z.; Zare, S.; Egan, T.; Yazdanshenas, H.; Ghasemian, D.; Tahmoures, M. Effects of a super absorbent polymer on soil properties and plant growth for use in land reclamation. Arid Land Res. Manag. 2018, 32, 407–420. [Google Scholar] [CrossRef]
- Bai, W.; Zhang, H.; Liu, B.; Wu, Y.; Song, J.Q. Effects of super-absorbent polymers on the physical and chemical properties of soil following different wetting and drying cycles. Soil Use Manag. 2010, 26, 253–260. [Google Scholar] [CrossRef]
- Akhter, J.; Mahmood, K.; Malik, K.; Mardan, A.; Ahmad, M.; Iqbal, M. Effects of hydrogel amendment on water storage of sandy loam and loam soils and seedling growth of barley, wheat and chickpea. Plant Soil Environ. 2004, 50, 463–469. [Google Scholar] [CrossRef]
- Akelah, A. Functionalized Polymeric Materials in Agriculture and the Food Industry; Springer US: New York, NY, USA, 2013. [Google Scholar]
- Dehkordi, K. Evaluation of two types of superabsorbent polymer on soil water and some soilmicrobial properties. Paddy Water Environ. 2018, 16, 143–152. [Google Scholar] [CrossRef]
- Yun, K.K.; Kim, K.K.; Choi, W. Hygral behavior of superabsorbent polymers with various particle sizes and crosslinking densities. Polymers 2017, 9, 600. [Google Scholar] [CrossRef]
- Thombare, N.; Mishra, S.; Siddiqui, M.; Jha, U.; Singh, D.; Mahajan, G.R. Design and development of guar gum based novel, superabsorbent and moisture retaining hydrogels for agricultural applications. Carbohydr. Polym. 2018, 185, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Durian, D.J. Effect of hydrogel particle additives on water-accessible pore structure of sandy soils: A custom pressure plate apparatus and capillary bundle model. Phys. Rev. E 2013, 87, 053013. [Google Scholar] [CrossRef] [PubMed]
- Gokavi, N.; Rudragouda; Mote, K.; Mukharib, D.S.; Manjunath, A.N.; Raghuramulu, Y. Performance of hydrogel on seed germination and growth of young coffee seedlings in nursery. J. Pharmacogn. Phytochem. 2018, 7, 1364–1366. [Google Scholar]
- Liang, R.; Liu, M.; Wu, L. Controlled release NPK compound fertilizer with the function of water retention. React. Funct. Polym. 2007, 67, 769–779. [Google Scholar] [CrossRef]
- De Barros, A.F.; Pimentel, L.D.; Araujo, E.F. Superabsorbent polymer application in seeds and planting furrow: It will be a new opportunity for rainfed agriculture. Semin. CIencias Agrar. 2017, 38, 1703–1714. [Google Scholar] [CrossRef]
- Guilherme, M.R.; Aouada, F.A.; Fajardo, A.R.; Martins, A.F.; Paulino, A.T.; Davi, M.F.; Rubira, A.F.; Muniz, E.C. Superabsorbent hydrogels based on polysaccharides for application in agriculture as soil conditioner and nutrient carrier: A review. Eur. Polym. J. 2015, 72, 365–385. [Google Scholar] [CrossRef]
- Sarkar, B.; Basak, B.B.; Sarkar, S.; Mandal, S. Adaptive Soil Management: From Theory to Practices; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Stegmann, R.; Lotter, S.; King, L.; Hopping, W. Fate of an Absorbent Gelling Material for Hygiene Paper Products in Landfill and Composting. Waste Manag. Res. 1993, 11, 155–170. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Nuzzaci, M.; Logozzo, G.; Gioia, T.; Camele, I. Biological investigations on the role of hydrogel formulations containing bioactive natural agents against some common phytopathogens of Phaseolus vulgaris L. and seed germination. J. Biol. Res. 2020, 3, 114–122. [Google Scholar] [CrossRef]
- Abdallah, A.M. The effect of hydrogel particle size on water retention properties and availability under water stress. Int. Soil Water Conserv. Res. 2019, 7, 275–285. [Google Scholar] [CrossRef]
- Hejduk, S.; Baker, S.W.; Spring, C.A. Evaluation of the effects of incorporation rate and depth of water-retentive amendment materials in sports turf constructions. Acta Agric. Scand. Sect. B Soil Plant Sci. 2012, 62, 155–164. [Google Scholar] [CrossRef]
- Wroblewska, K.; Debicz, R.; Babelewski, P. The influence of water sorbing geocomposite and pine bark mulching on growth and flowering of some perennial species. Acta Sci. Pol. Hortorum Cultus 2012, 11, 203–216. [Google Scholar]
- Ismail, H.; Irani, M.; Ahmad, Z. Starch-based hydrogels: Present status and applications. Int. J. Polym. Mater. 2013, 62, 411–420. [Google Scholar] [CrossRef]
- Pattanaaik, S.K.; Singh, B.; Wangchu, L.; Debnath, P.; Hazarika, B.N.; Pandey, A.K. Effect of hydrogel on water and nutrient management of Citrus limon. Int. J. Agric. Innov. Res. 2015, 3, 1656–1659. [Google Scholar]
- Willenborg, C.J.; Gulden, R.H.; Johnson, E.N.; Shirtliffe, S.J. Germination characteristics of polymer-coated canola (Brassica napus L.) seeds subjected to moisture stress at different temperatures. Agron. J. 2004, 96, 786–791. [Google Scholar] [CrossRef]
- Akhter, F.N.; Kabir, G.; Mannan, M.A.; Shaheen, N.N. Aging Effect of Wheat and Barley Seeds Upon Germination Mitotic Index and Chromosomal Damage. Med. J. Islam. World Acad. Sci. 1992, 5, 44–48. [Google Scholar]
- Ipek, A.; Kaya, M.D.; Gürbüz, B. Effects of seed age and GA3 application on germination of fenugreek (Trigonella foenum-graecum L.) and cumin (Cuminum cyminum L.) seeds. J. Agric. Sci. 2008, 14, 57–61. [Google Scholar]
- Romanenko, V.A. Computation of the Autumn Soil Moisture Using a Universal Relationship for a Large Area; Ukrainian Hydrometeorological Research Institute: Kiev, Ukraine, 1961. [Google Scholar]
- Xu, C.Y.; Singh, V.P. Dependence of evaporation on meteorological variables at different time-scales and intercomparison of estimation methods. Hydrol. Process. 1998, 12, 429–442. [Google Scholar] [CrossRef]
- Jackson, M.L. Soil Chemical Analysis; Prentice Hall. Inc.: Englewood Cliffs, NJ, USA, 1985; Volume 498, pp. 183–204. [Google Scholar]
- Gee, G.W.; Bauder, J.W. Particle Size Analysis. In Methods of Soil Analysis; Part 1; Wiley: Hoboken, NJ, USA, 1986; pp. 383–409. [Google Scholar]
- Saxton, K.E.; Rawls, W.J.; Romberger, J.S.; Papendick, R.I. Estimating generalized soil-water characteristics from texture. Soil Sci. Soc. Am. J. 1986, 50, 1031–1036. [Google Scholar] [CrossRef]
- Association of Official Seed Analysts. Seed Vigor Testing Handbook, 1st ed.; AOSA: Albuquerque, NM, USA, 1983. [Google Scholar]
- Zahedifar, M.; Zohrabi, S. Germination and seedling characteristics of drought-stressed corn seed as influenced by seed priming with potassium nano-chelate and sulfate fertilizers. Acta Agric. Slov. 2016, 107, 113–128. [Google Scholar] [CrossRef]
- Javaid, M.M.; Florentine, S.; Ali, H.H.; Weller, S. Effect of environmental factors on the germination and emergence of Salvia verbenaca L. cultivars (verbenaca and vernalis): An invasive species in semi-arid and arid rangeland regions. PLoS ONE 2018, 13, e0194319. [Google Scholar] [CrossRef]
- Snedecor, W.G.; Cochran, G.W. Statistical Methods, 7th ed.; The Iowa State University Press: Ames, IA, USA, 1980. [Google Scholar]
- Seleiman, M.F.; Al-Suhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; Abdul-Wajid, H.H.; Battaglia, M.L. Drought Stress Impacts on Plants and Different Approaches to Alleviate Its Adverse Effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef] [PubMed]
- Copeland, L.O.; McDonald, M.F. Principles of Seed Science and Technology; Springer Science & Business Media: New York, NY, USA, 2012; ISBN 978-1-4615-1619-4. [Google Scholar]
- Moghadam, H.R.T. Superabsorbent polymer mitigates deleterious effects of arsenic in wheat. Rhizosphere 2017, 3, 40–43. [Google Scholar] [CrossRef]
- Hadas, A. Germination and Seedling Establishment. In Encyclopedia of Soils in the Environment; Hillel, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 130–137. [Google Scholar] [CrossRef]
- Obroucheva, N.V.; Sinkevich, I.A.; Lityagina, S.V.; Novikova, G.V. Water relations in germinating seeds. Russ. J. Plant Physiol. 2017, 64, 625–633. [Google Scholar] [CrossRef]
- Vijayaprabhakar, A.; Vijayakumar, E.; Krishnaveni, T.R.S.; Jayanthi, C. Influence of soil and weather factors on the germination of dry sown rain-fed crops on the semi-arid tracts of Tamil Nadu, India. Appl. Ecol. Environ. Res. 2021, 19, 1203–1220. [Google Scholar] [CrossRef]
- Wuest, S.B.; Lutcher, L.K. Soil water Potential Requirement for Germination of Winter Wheat. Soil Sci. Soc. Am. J. 2012, 77, 279–283. [Google Scholar] [CrossRef]
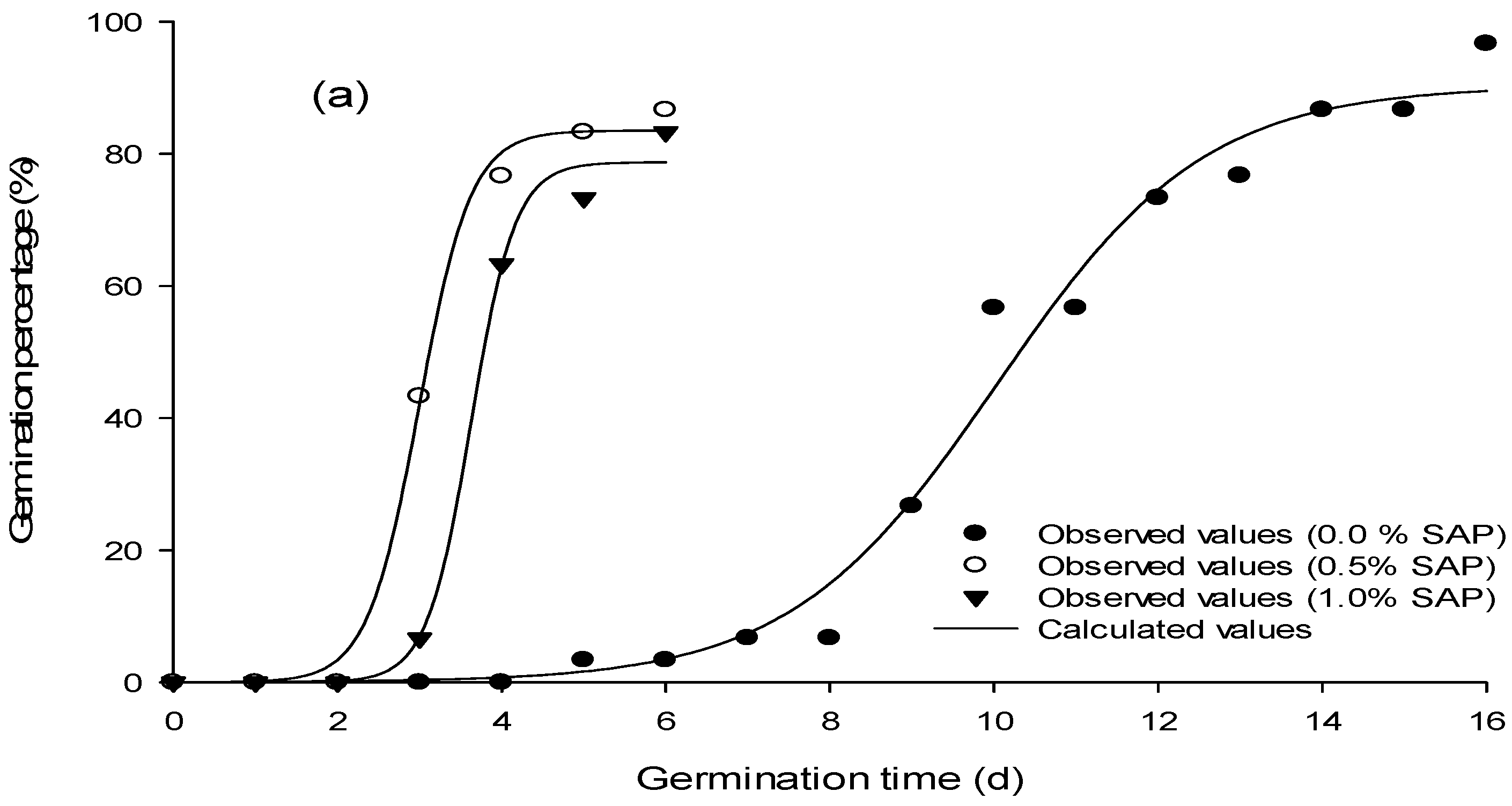
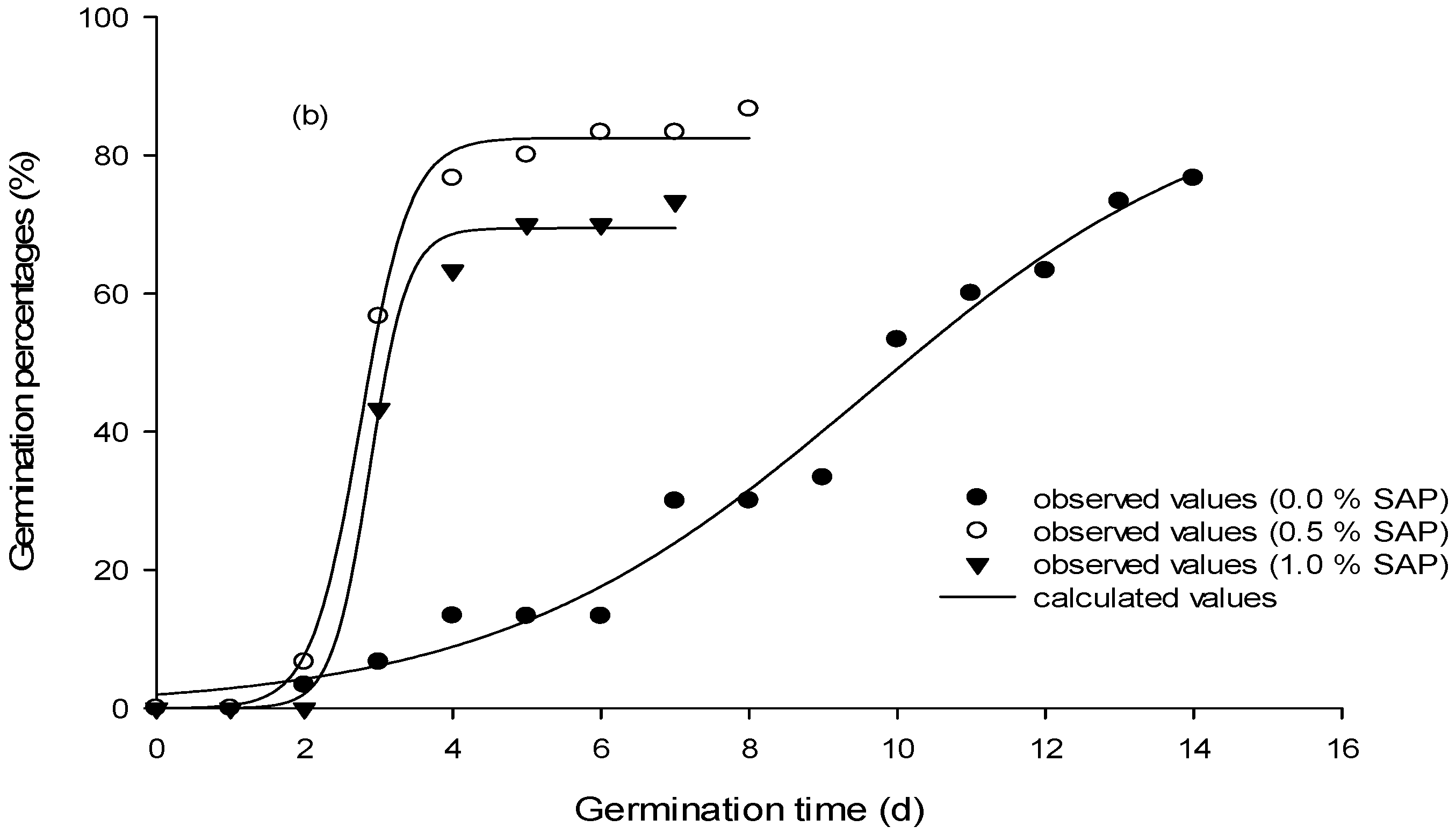

| SAP (0.0%) | SAP (0.5%) | SAP (1.0%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| * TFG | ** AW | TBG | AW | * TFG | ** AW | TBG | AW | * TFG | ** AW | TBG | AW |
| day | cm | day | cm | day | cm | day | cm | day | cm | day | cm |
| 0 | 2.01 | 0 | 2.01 | 0 | 2.01 | 0 | 2.01 | 0 | 2.01 | 0 | 2.01 |
| 7 | 0.68 | 7 | 0.68 | 7 | 0.68 | 6 | 0.58 | 7 | 0.68 | 6 | 0.58 |
| 10 | 0.71 | 10 | 0.71 | 8 | 0.24 | -- | -- | -- | -- | -- | -- |
| 14 | 0.22 | 15 | 0.27 | -- | -- | -- | -- | -- | -- | -- | -- |
| -- | -- | 16 | 0.05 | -- | -- | -- | -- | -- | -- | -- | -- |
| Month | ANT | ADT | ARH | AWS | AP |
|---|---|---|---|---|---|
| (°C) | (%) | (m·s−1) | (mm·day−1) | ||
| November | 9.07 | 30.54 | 40.63 | 2.96 | 0.00 |
| December | 6.18 | 27.93 | 42.97 | 2.67 | 0.00 |
| January | 3.13 | 27.25 | 44.78 | 3.01 | 0.00 |
| February | 2.57 | 28.33 | 39.47 | 3.36 | 0.00 |
| Proprieties | Unit | Value |
|---|---|---|
| pH | ---- | 7.73 |
| EC | dS·m−1 | 1.097 |
| Total CaCO3 | % | 2.63 |
| Sand | % | 93.2 |
| Silt | % | 2.5 |
| Clay | % | 4.3 |
| Texture | ---- | Sand |
| SC | m3/m3 | 0.35 |
| FC | m3/m3 | 0.12 |
| WP | m3/m3 | 0.05 |
| Ca2+ | meqL−1 | 6.00 |
| Mg2+ | meqL−1 | 2.33 |
| K+ | meqL−1 | 0.86 |
| Na+ | meqL−1 | 1.78 |
| CO32− | meqL−1 | 0.00 |
| HCO3− | meqL−1 | 3.33 |
| Cl− | meqL−1 | 3.00 |
| SO4− | meqL−1 | 4.64 |
| Saturation capacity of SAP | g water/g SAP | 231.5 |
| Levels of SAP (%) | Barley | Fenugreek | ||
|---|---|---|---|---|
| FGP % | GI (Day−1) | FGP % | GI (Day−1) | |
| 0.0 | 96.67 | 0.93 | 76.67 | 1.11 |
| 0.5 | 86.67 | 2.47 | 86.67 | 2.66 |
| 1.0 | 86.67 | 2.01 | 80.00 | 2.46 |
| LSD (0.05) | ns | 0.76 | ns | 1.29 |
| SAP Levels (%) | Barley | Fenugreek | ||||
|---|---|---|---|---|---|---|
| T50 (Days) * | Tmax (Days) * | WGE (cm−1) ** | T50 (Days) * | Tmax (Days) * | WGE (cm−1) | |
| 0.0 | 10.0 | 16 | 27.76 | 8.82 | 14 | 23.07 |
| 0.5 | 3.0 | 6 | 30.04 | 2.79 | 8 | 29.90 |
| 1.0 | 3.6 | 6 | 31.49 | 2.85 | 7 | 28.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, R.R.S.; Nassar, I.N.; Ghallab, A.; Ali, E.F.; Alqubaie, A.I.; Rady, M.M.; Awad, A.A.M. Alleviation of Water-Deficit Stress on Seed Germination of Barley and Fenugreek in a Sandy Soil Using Superabsorbent Polymer. Agronomy 2023, 13, 2324. https://doi.org/10.3390/agronomy13092324
Ali RRS, Nassar IN, Ghallab A, Ali EF, Alqubaie AI, Rady MM, Awad AAM. Alleviation of Water-Deficit Stress on Seed Germination of Barley and Fenugreek in a Sandy Soil Using Superabsorbent Polymer. Agronomy. 2023; 13(9):2324. https://doi.org/10.3390/agronomy13092324
Chicago/Turabian StyleAli, Rehab R. S., Ibrahim N. Nassar, Ahmed Ghallab, Esmat F. Ali, Ahmad I. Alqubaie, Mostafa M. Rady, and Ahmed A. M. Awad. 2023. "Alleviation of Water-Deficit Stress on Seed Germination of Barley and Fenugreek in a Sandy Soil Using Superabsorbent Polymer" Agronomy 13, no. 9: 2324. https://doi.org/10.3390/agronomy13092324
APA StyleAli, R. R. S., Nassar, I. N., Ghallab, A., Ali, E. F., Alqubaie, A. I., Rady, M. M., & Awad, A. A. M. (2023). Alleviation of Water-Deficit Stress on Seed Germination of Barley and Fenugreek in a Sandy Soil Using Superabsorbent Polymer. Agronomy, 13(9), 2324. https://doi.org/10.3390/agronomy13092324







