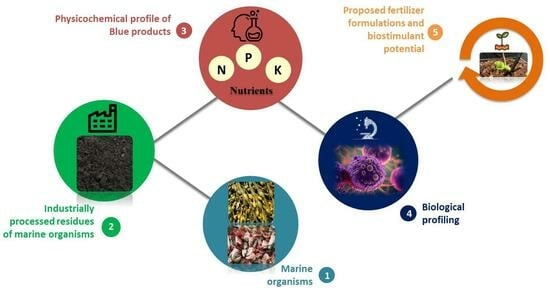
Chemical and Biological Profiling of Fish and Seaweed Residues to Be Applied for Plant Fertilization
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Methods
2.1.1. Description of the Tested Materials
2.1.2. Materials Applied in Chemical Analyses
2.2. Physical Measurements
2.2.1. pH and Conductivity
2.2.2. Total Carbon, Total Organic Carbon (TOC), and Total Nitrogen Determination
2.2.3. FT-IR Spectral Measurements
2.2.4. Microwave Digestion
2.2.5. ICP-MS Spectrometry
2.2.6. ICP-OES Analysis
2.2.7. Total Fat Analysis
2.2.8. Nitrate Determination through Ion Chromatography
2.2.9. Ammonium Determination through Derivatization Ultraviolet Spectroscopy (UV–Visible)
2.2.10. Fatty Acid Methyl Ester Analysis through GC–FID
2.2.11. Gas Chromatography–Mass Spectrometry (GC–MS)
2.3. Leaching Procedure
2.4. Extraction Procedure of HNSW, LNSW, and GFB Samples
2.5. Amino Acid Analysis
2.6. DPPH Radical Scavenging Activity
2.7. Bacterial Cell Cultures
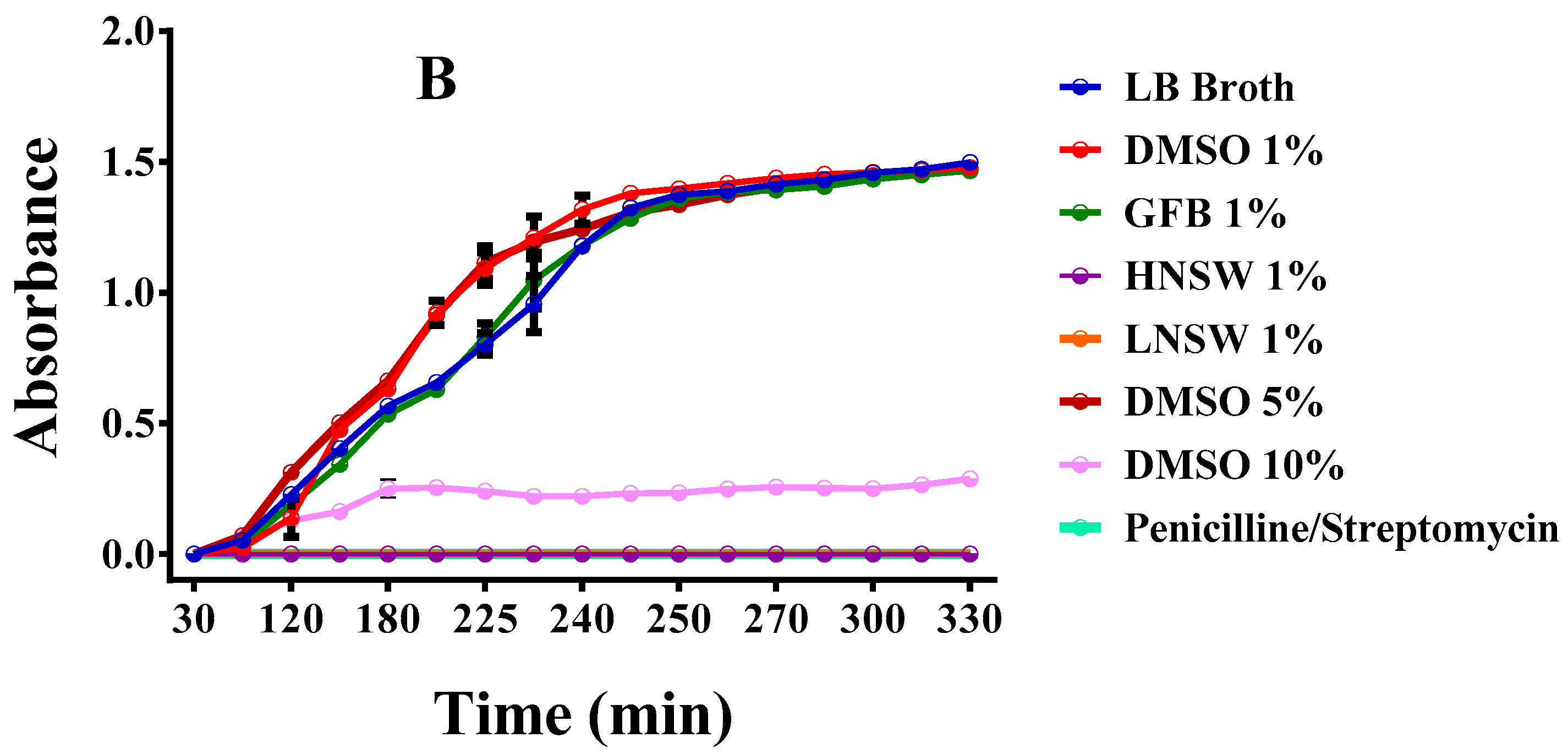
2.7.1. Growth Rate of Bacteria in Liquid Cultures
2.7.2. Determination of Zone of Inhibition (ZOI) in Solid Bacterial Cultures
2.8. Neuronal Cell Cultures
2.8.1. Cell Viability and Proliferation Studies
2.8.2. Cell Morphology Studies
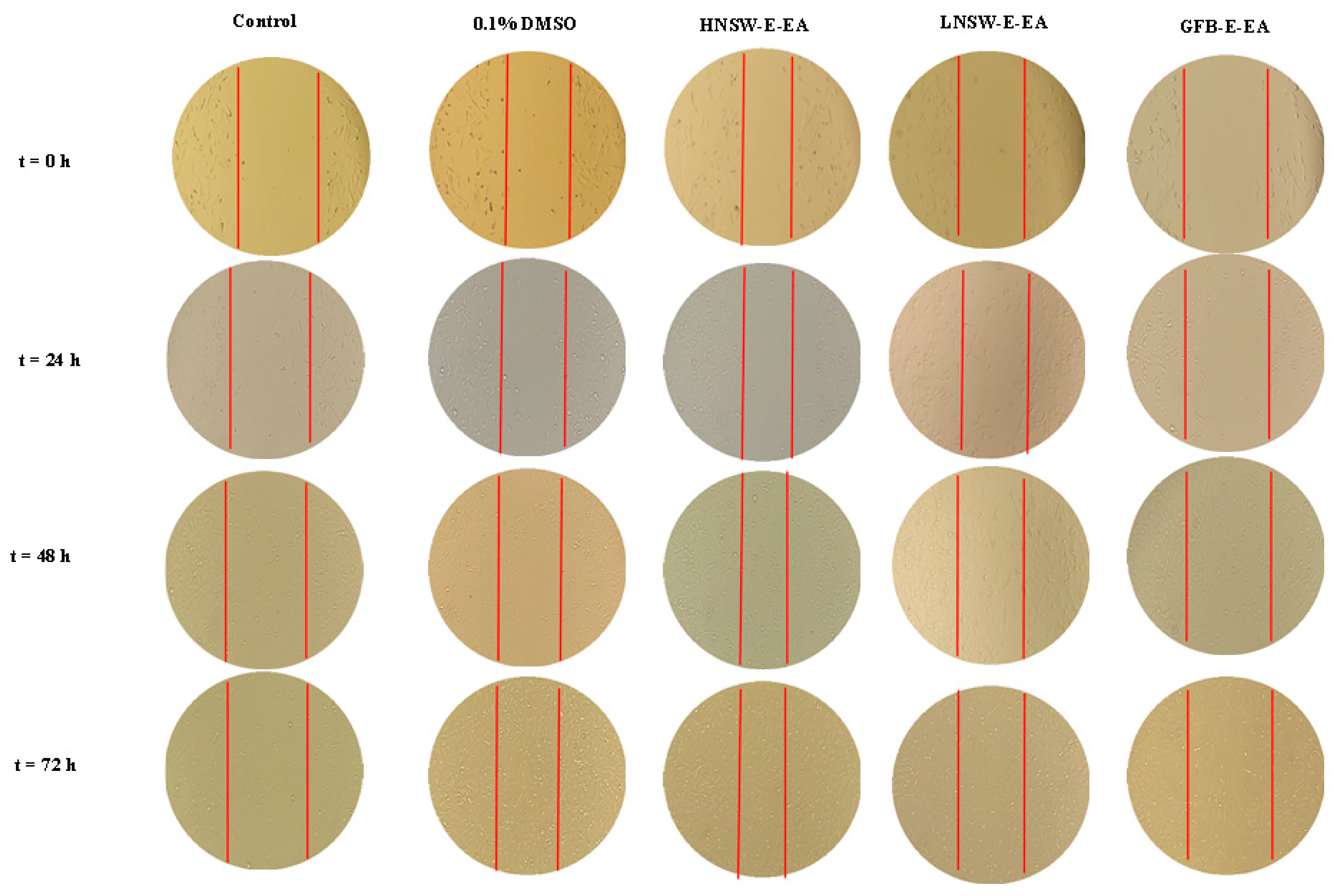
2.8.3. Cell Migration Studies
2.9. Statistical Analysis
3. Results
3.1. pH and Electrical Conductivity Measurements
3.2. FT-IR Spectra of Marine-Derived Materials
3.3. Total Carbon, Total Nitrogen and Total Organic Carbon Determination
3.4. Essential Elements and Heavy Metal Distribution
3.5. Total Fat Determination
3.6. Nitrate and Ammonium Analysis
3.7. Fatty Acid Methyl Ester (F.A.M.E.) Analysis through GC–FID
3.8. Leaching Samples
3.9. Molecular Composition of the Extracted Samples
3.10. Sample Hydrolysis and Composition
3.11. Amino Acid Analysis
3.12. DPPH Scavenging Activity
3.13. Antimicrobial Evaluation in Bacterial Cultures
3.13.1. Liquid Cultures
3.13.2. Solid Agar Cultures
3.14. Biotoxicity Investigation in Eukaryotic Cell Cultures
3.14.1. Cell Viability and Proliferation Studies
3.14.2. Cell Morphology Studies
3.14.3. Migration Studies
4. Discussion
4.1. BlueBio Waste as a Potential Ecofriendly Fertilizer in Plant Growth
4.2. Establishment of Fundamental Physicochemical Properties
4.3. Identity Formulation upon Leaching
4.4. Amino Acid Hydrolysis Supporting Fertilizer Formulations
4.5. Biological Studies
4.5.1. Bacterial Cell Cultures
4.5.2. Eukaryotic Cell Cultures
4.5.3. Antioxidant Potential
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAE | Ascorbic Acid Equivalent |
| ARA | Arachidonic acid |
| ANOVA | Analysis of variance |
| DM | Dry matter |
| DMEM | Dulbecco’s modified Eagle’s medium |
| DMSO | Dimethylsulfoxide |
| EA | Ethyl acetate |
| EC | Electrical conductivity |
| EDTA | Ethylenediaminetetraacetic acid |
| F.A.M.E. | Fatty Acid Methyl Esters |
| GC–MS | Gas Chromatography–Mass Spectrometry |
| GFB | Ground Fish Bone |
| H | Hexane |
| HNSW | High-Nitrogen Seaweed |
| ICP-MS | Inductively Coupled Plasma Mass Spectrometry |
| ICP-OES | Inductively Coupled Plasma Optical Emission Spectrometry |
| ISTD | Internal Standard |
| LNSW | Low-Nitrogen Seaweed |
| OD | Optical Density |
| OS | Optical Density |
| ORS | Octopole Reaction System |
| PEN | Penicillin-streptomycin |
| TOC | Total Organic Carbon |
| UPLC | Ultra Performance Liquid Chromatography |
| XTT | (Sodium 3-[1-(phenylaminocarbonyl)-3,4-tetrazolium]-bis(4-methoxy-6-nitro) benzenesulfonic acid hydrate) |
| ZOI | Zone of Inhibition |
References
- Cordell, D.; White, S. Peak Phosphorus: Clarifying the key issues of a vigorous debate about long-term Phosphorus security. Sustainability 2011, 3, 2027–2049. [Google Scholar] [CrossRef]
- Al Rawashdeh, R. World peak potash: An analytical study. Resour. Policy 2020, 69, 101834. [Google Scholar] [CrossRef]
- EU 2019. Regulation (EU) 2019/1009 of the European Parliament and of the Council of 5 June 2019 Laying down Rules on the Making Available on the Market of EU Fertilising Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and Repealing Regulation (EC) No 2003/2003. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32019R1009 (accessed on 13 June 2023).
- Mącik, M.; Gryta, A.; Frąc, M. Biofertilizers in agriculture: An overview on concepts, strategies and effects on soil microorganisms. Adv. Agron. 2020, 162, 31–87. [Google Scholar] [CrossRef]
- Chaney, R.L. Food safety issues for mineral and organic fertilizers. Adv. Agron. 2012, 117, 51–116. [Google Scholar] [CrossRef]
- Halpern, M.; Bar-Tal, A.; Ofek, M.; Minz, D.; Muller, T.; Yermiyahu, U. The use of biostimulants for enhancing nutrient uptake. Adv. Agron. 2015, 130, 141–174. [Google Scholar] [CrossRef]
- Ahuja, I.; Løes, A.-K. Effect of Fish Bones and Algae Fibre as Fertilisers for Ryegrass; Report, no. 7; Norwegian Centre for Organic Agriculture (NORSØK): Tingvoll, Norway, 2019; Volume 4, Available online: https://orgprints.org/id/eprint/36439/ (accessed on 22 June 2023).
- Moloșag, A.; Pârvulescu, O.C.; Ion, V.A.; Asănică, A.C.; Soane, R.; Moț, A.; Dobrin, A.; Frîncu, M.; Løes, A.-K.; Cabell, J.; et al. Effects of marine residue-derived fertilizers on strawberry growth, nutrient content, fruit yield and quality. Agronomy 2023, 13, 1221. [Google Scholar] [CrossRef]
- Matos, G.S.; Pereira, S.G.; Genisheva, Z.A.; Gomes, A.M.; Teixeira, J.A.; Rocha, C.M.R. Advances in extraction methods to recover added-value compounds from seaweeds: Sustainability and Functionality. Foods 2021, 10, 516. [Google Scholar] [CrossRef]
- Gibilisco, P.E.; Lancelotti, J.L.; Negrin, V.L.; Idaszkin, Y.L. Composting of seaweed waste: Evaluation on the growth of Sarcocornia perennis. J. Environ. Manag. 2020, 274, 111193. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Mohamed, A.A.; Mohamed, H.I.; Ramadan, K.M.A.; Barqawi, A.A.; Mansour, A.T. Phytochemical and potential properties of peaweeds and their recent applications: A Review. Mar. Drugs 2022, 20, 342. [Google Scholar] [CrossRef]
- Hrólfsdóttir, A.Þ.; Arason, S.; Sveinsdóttir, H.I.; Gudjónsdóttir, M. Added value of Ascophyllum nodosum side stream utilization during seaweed meal processing. Mar. Drugs 2022, 20, 340. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Je, J.-G.; An, H.; Baek, K.; Lee, J.M.; Yim, M.-J.; Ko, S.-C.; Kim, J.-Y.; Oh, G.-W.; Kang, M.-C.; et al. Isolation and characterization of efficient active compounds using high-performance centrifugal partition chromatography (CPC) from anti-inflammatory activity fraction of Ecklonia maxima in South Africa. Mar. Drugs 2022, 20, 471. [Google Scholar] [CrossRef]
- Illera-Vives, M.; Labandeira, S.S.; Brito, L.M.; López-Fabal, A.; López-Mosquera, M.E. Evaluation of compost from seaweed and fish waste as a fertilizer for horticultural use. Sci. Hortic. 2015, 186, 101–107. [Google Scholar] [CrossRef]
- Ahuja, I.; Dauksas, E.; Remme, J.F.; Richardsen, R.; Løes, A.-K. Fish and fish waste-based fertilizers in organic farming—With status in Norway: A review. Waste Manag. 2020, 115, 95–112. [Google Scholar] [CrossRef] [PubMed]
- López-Mosquera, M.E.; Fernández-Lema, E.; Villares, R.; Corral, R.; Alonso, B.; Blanco, C. Composting fish waste and seaweed to produce a fertilizer for use in organic agriculture. Proc. Environ. Sci. 2011, 9, 113–117. [Google Scholar] [CrossRef]
- Løes, A.-K.; Ahuja, I.; de Boer, A.; Rittl, T. Fertilisation Effects of Marine-Derived Residual Materials on Agricultural Crops; Report, no. 13; Norwegian Centre for Organic Agriculture (NORSØK): Tingvoll, Norway, 2022; Volume 7, Available online: https://orgprints.org/id/eprint/45330/ (accessed on 22 June 2023).
- Løes, A.-K.; Grønmyr, F.; Pommeresche, R.; Rittl, T. Algae Fibre for Soil Improvement (FIMO); Report, no. 8; Norwegian Centre for Organic Agriculture (NORSØK): Tingvoll, Norway, 2022; Volume 7, Available online: https://orgprints.org/id/eprint/44040/ (accessed on 22 June 2023).
- Løes, A.-K. Benefits and challenges of marine-derived fertilisers. In Proceedings of the RELACS Webinar Series, Frick, Switzerland, March–April 2021; Norwegian Centre for Organic Agriculture (NORSØK): Tingvoll, Norway. Available online: https://orgprints.org/id/eprint/44770/ (accessed on 22 June 2023).
- Amino Acids in Feeds. Performic Acid Oxidation with Acid Hydrolysis-Sodium Metabisulfite Method, AOAC 1997 994.12. In Official Methods of Analysis of AOAC INTERNATIONAL, 21st ed.; AOAC INTERNATIONAL: Rockville, MD, USA, 2019; Method 994.12. [CrossRef]
- Mourtzinos, I.; Konteles, S.; Kalogeropoulos, N.; Karathanos, V.T. Thermal oxidation of vanillin affects its antioxidant and antimicrobial properties. Food Chem. 2009, 114, 791–797. [Google Scholar] [CrossRef]
- Wang, Y.; McGivern, D.R.; Cheng, L.; Li, G.; Lemon, S.M.; Niu, J.; Su, L.; Reszka-Blanco, N.J. Ribavirin contributes to hepatitis C Virus suppression by augmenting pDC activation and type 1 IFN Production. PLoS ONE 2015, 10, e0135232. [Google Scholar] [CrossRef]
- Chaveroux, C.; Bruhat, A.; Carraro, V.; Jousse, C.; Averous, J.; Maurin, A.C.; Parry, L.; Mesclon, F.; Muranishi, Y.; Cordelier, P.; et al. Regulating the expression of therapeutic transgenes by controlled intake of dietary essential amino acids. Nat. Biotechnol. 2016, 34, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Martinotti, S.; Ranzato, E. Scratch wound healing assay. Methods Mol. Biol. 2020, 2109, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Gangidi, R.R.; Proctor, A.; Meullenet, J.F. Milled rice surface lipid measurement by diffuse reflectance fourier transform infrared spectroscopy (DRIFTS). J. Am. Oil. Chem. Soc. 2002, 79, 7–12. [Google Scholar] [CrossRef]
- Thomas, A.S.; Saravanakumar, R.; Gupta, P.V. Evaluation of cytotoxic activity of protein extracts from the leaves of morinda pubescens on human cancer cell lines. Rev. Bras. Farmacogn. 2017, 27, 99–104. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, M.; Alalawy, A.I.; Almutairi, F.M.; Al-Duais, M.A.; Wang, J.; Salama, E.S. Identification and characterization of marine seaweeds for biocompounds production. Environ. Technol. Innov. 2021, 24, 101848. [Google Scholar] [CrossRef]
- Yip, W.H.; Joe, L.S.; Mustapha, W.A.W.; Maskat, M.Y.; Said, M. Characterisation and stability of pigments extracted from sargassum binderi obtained from Semporna. Sabah. Sains Malays. 2014, 43, 1345–1354. [Google Scholar]
- Løes, A.-K.; Ahlin, J.P.; Ahuja, I.; Krogstad, T.; Smevoll, S.; Waag, H. Effects of formic acid preservation of fishbones on the extractability of ammonium lactate–acetate soluble calcium, phosphorus, magnesium, and potassium. Waste Biomass Valorization 2022, 13, 3547–3559. [Google Scholar] [CrossRef]
- Jönsson, M.; Allahgholi, L.; Sardari, R.R.R.; Hreggviðsson, G.O.; Karlsson, E.N. Extraction and modification of macroalgal polysaccharides for current and next-generation applications. Molecules 2020, 25, 930. [Google Scholar] [CrossRef] [PubMed]
- Ragunathan, V.; Pandurangan, J.; Ramakrishnan, T. Gas chromatography-mass spectrometry analysis of methanol extracts from marine red seaweed Gracilaria corticata. Pharmacogn. J. 2019, 11, 547–554. [Google Scholar] [CrossRef]
- Shobier, A.H.; Abdel Ghani, S.A.; Barakat, K.M. GC/MS spectroscopic approach and antifungal potential of bioactive extracts produced by marine macroalgae. Egypt. J. Aquat. Res. 2016, 42, 289–299. [Google Scholar] [CrossRef]
- Yamamoto, M.; Baldermann, S.; Yoshikawa, K.; Fujita, A.; Mase, N.; Watanabe, N. Determination of volatile compounds in four commercial samples of Japanese green algae using solid phase microextraction gas chromatography mass spectrometry. Sci. World J. 2014, 2014, 289780. [Google Scholar] [CrossRef]
- Machado, M.; Machado, S.; Pimentel, F.B.; Freitas, V.; Alves, R.C.; Oliveira, M.B.P.P. Amino acid profile and protein quality assessment of macroalgae produced in an integrated multi-trophic aquaculture system. Foods 2020, 9, 1382. [Google Scholar] [CrossRef]
- Lourenco, S.O.; Barbarino, E.; De-Paula, J.C.; Pereira, L.O.d.S.; Marquez, U.M.L. Amino acid composition, protein content and calculation of nitrogen-to-protein conversion factors for 19 tropical seaweeds. Phycol. Res. 2002, 50, 233–241. [Google Scholar] [CrossRef]
- Kauanova, S.; Urazbayev, A.; Vorobjev, I. The frequent sampling of wound scratch assay reveals the “opportunity” window for quantitative evaluation of cell motility-impeding Drugs. Front. Cell Dev. Biol. 2021, 9, 640972. [Google Scholar] [CrossRef]
- Savvas, D. Hydroponics: A modern technology supporting the application of integrated crop management in greenhouse. J. Food Agric. Environ. 2003, 1, 80–86. [Google Scholar]
- Sambo, P.; Nicoletto, C.; Giro, A.; Pii, Y.; Valentinuzzi, F.; Mimmo, T.; Lugli, P.; Orzes, G.; Mazzetto, F.; Astolfi, S.; et al. Hydroponic solutions for soilless production systems: Issues and opportunities in a smart agriculture perspective. Front. Plant Sci. 2019, 10, 923. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; van Iersel, M.W. Sodium chloride effects on growth, morphology, and physiology of Chrysanthemum (Chrysanthemum × morifolium). Hort Sci. Horts 2008, 43, 1888–1891. [Google Scholar] [CrossRef]
- Flowers, T.J.; Munns, R.; Colmer, T.D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann. Bot. 2015, 115, 419–431. [Google Scholar] [CrossRef]
- Huang, Z.; Wang, C.; Feng, Q.; Liou, R.-M.; Lin, Y.-F.; Qiao, J.; Lu, Y.; Chang, Y. The mechanisms of sodium chloride stress mitigation by salt-tolerant plant growth promoting rhizobacteria in wheat. Agronomy 2022, 12, 543. [Google Scholar] [CrossRef]
- Malík, M.; Velechovský, J.; Praus, L.; Janatová, A.; Kahánková, Z.; Klouček, P.; Tlustoš, P. amino acid supplementation as a biostimulant in medical cannabis (Cannabis sativa L.) plant nutrition. Front. Plant Sci. 2022, 13, 868350. [Google Scholar] [CrossRef] [PubMed]
- Romero, L.C.; Ángeles Aroca, M.; Laureano-Marín, A.M.; Moreno, I.; García, I.; Gotor, C. Cysteine and cysteine-related signaling pathways in Arabidopsis thaliana. Molec. Plant 2014, 7, 264–276. [Google Scholar] [CrossRef]
- Haag, A.F.; Kerscher, B.; Dall’Angelo, S.; Sani, M.; Longhi, R.; Baloban, M.; Wilson, H.M.; Mergaert, P.; Zanda, M.; Ferguson, G.P. Role of cysteine residues and disulfide bonds in the activity of a legume root nodule-specific, cysteine rich peptide. J. Biol. Chem. 2012, 287, 10791–10798. [Google Scholar] [CrossRef]
- Richau, K.H.; Kaschani, F.; Verdoes, M.; Pansuriya, T.C.; Niessen, S.; Stuber, K.; Colby, T.; Overkleeft, H.S.; Bogyo, M.; Van der Hoorn, R.A. Subclassification and biochemical analysis of plant papain-like cysteine proteases displays subfamily-specific characteristics. Plant Physiol. 2012, 158, 1583–1599. [Google Scholar] [CrossRef] [PubMed]
- Madende, M.; Hayes, M. Fish by-product use as biostimulants: An overview of the current state of the art, including relevant legislation and regulations within the EU and USA. Molecules 2020, 25, 1122. [Google Scholar] [CrossRef]
- Parađiković, N.; Teklić, T.; Zeljković, S.; Lisjak, M.; Špoljarević, M. Biostimulants research in some horticultural plant species—A review. Food Energy Secur. 2019, 8, e00162. [Google Scholar] [CrossRef]
- Rouphael, Y.; Cardarelli, M.; Bonini, P.; Colla, G. Synergistic action of a microbial-based biostimulant and a plant derived-protein hydrolysate enhances lettuce tolerance to alkalinity and salinity. Front Plant Sci. 2017, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Beavers, W.N.; Monteith, A.J.; Amarnath, V.; Mernaugh, R.L.; Roberts, L.J., 2nd; Chazin, W.J.; Davies, S.S.; Skaar, E.P. Arachidonic acid kills staphylococcus aureus through a lipid peroxidation mechanism. mBio 2019, 10, e01333-19. [Google Scholar] [CrossRef]
- Herndon, J.L.; Peters, R.E.; Hofer, R.N.; Simmons, T.B.; Symes, S.J.; Giles, D.K. Exogenous polyunsaturated fatty acids (PUFAs) promote changes in growth, phospholipid composition, membrane permeability and virulence phenotypes in Escherichia coli. BMC Microbiol. 2020, 20, 305. [Google Scholar] [CrossRef]
- Jeon, W.B.; Park, B.H.; Choi, S.K.; Lee, K.M.; Park, J.K. Functional enhancement of neuronal cell behaviors and differentiation by elastin-mimetic recombinant protein presenting Arg-Gly-Asp peptides. BMC Biotechnol. 2012, 12, 61. [Google Scholar] [CrossRef] [PubMed]
- Feles, S.; Overath, C.; Reichardt, S.; Diegeler, S.; Schmitz, C.; Kronenberg, J.; Baumstark-Khan, C.; Hemmersbach, R.; Hellweg, C.E.; Liemersdorf, C. Streamlining culture conditions for the neuroblastoma cell line SH-SY5Y: A prerequisite for functional studies. Methods Protoc. 2022, 5, 58. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D.C.; van der Zwan, T.; Richards, A.; Little, H.; Coaker, G.L.; Bostock, R.M. The oomycete microbe-associated molecular pattern, arachidonic acid, and an ascophyllum nodosum-derived plant biostimulant induce defense metabolome remodeling in tomato. Phytopathology 2023, 113, 1084–1092. [Google Scholar] [CrossRef]
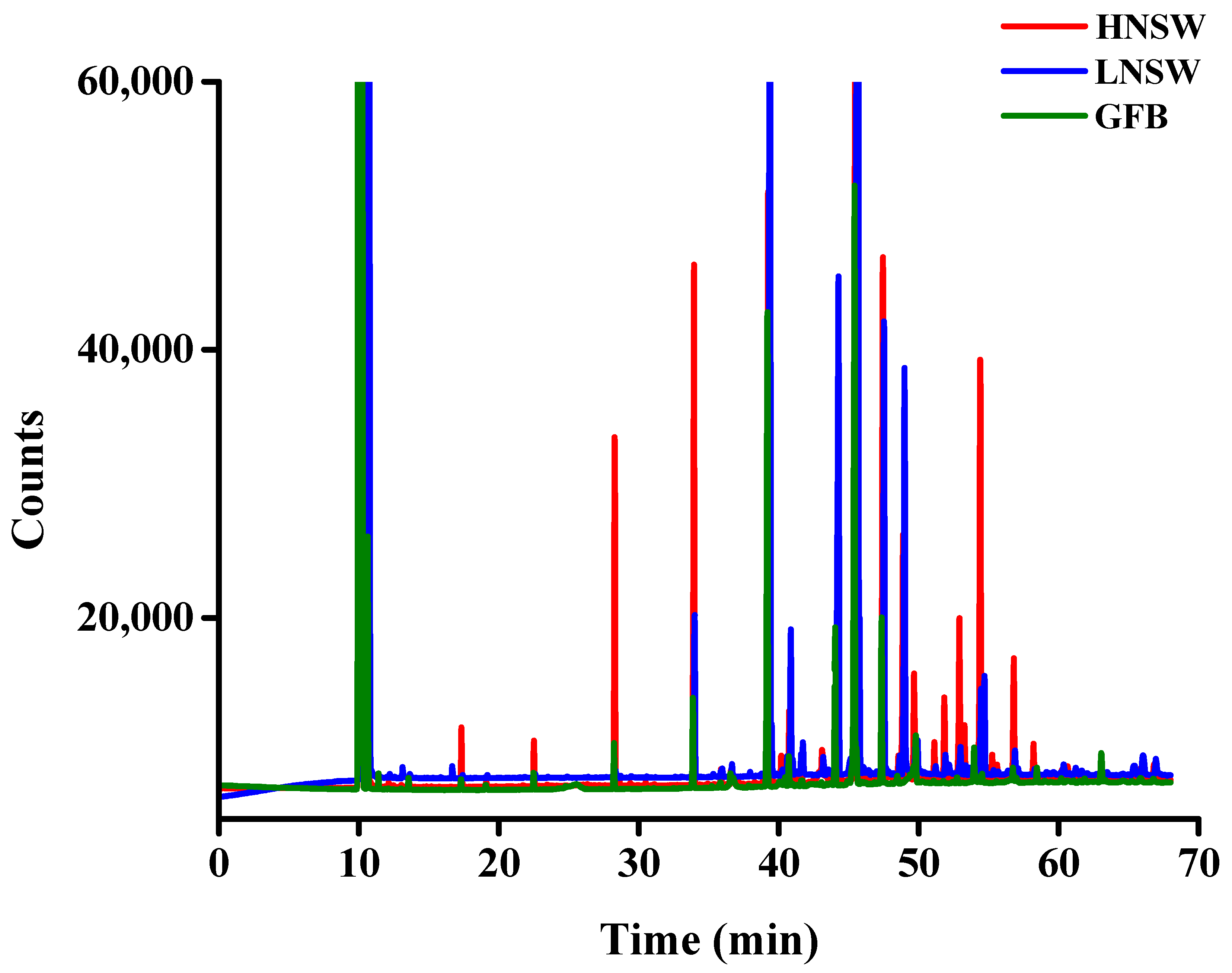





| Parameter | HNSW | LNSW | GFB |
|---|---|---|---|
| pH | 9.5 ± 0.2 | 10.3 ± 0.2 | 7.1 ± 0.2 |
| EC (μS/cm) | 58,000 ± 2175 | 31,750 ± 1190 | 8500 ± 319 |
| Total C (% w/w) | 12.1 ± 0.2 | 12.1 ± 0.2 | 7.9 ± 0.1 |
| Total Organic C (% w/w) | 6.5 ± 0.1 | 6.2 ± 0.1 | 6.1 ± 0.1 |
| Total Nitrogen (% w/w) | 0.42 ± 0.05 | 0.16 ± 0.02 | 2.05 ± 0.23 |
| Dry matter (%) | 29.4 ± 1.5 | 28.8 ± 1.4 | 24.5 ± 1.2 |
| Element (Concentration) | HNSW | LNSW | GFB | |
|---|---|---|---|---|
| Heavy metals | Pb (mg/kg) | <0.1 | <0.1 | <0.1 |
| Cd (mg/kg) | 0.20 ± 0.02 | 0.30 ± 0.03 | <0.05 | |
| Ni (mg/kg) | 0.70 ± 0.12 | 1.5 ± 0.3 | 0.30 ± 0.05 | |
| Cr (mg/kg) | 1.2 ± 0.2 | 1.3 ± 0.2 | 0.6 ± 0.1 | |
| As (mg/kg) | 7.1 ± 1.0 | 4.5 ± 0.6 | 1.7 ± 0.2 | |
| Nutrients | K (% w/w) | 2.35 ± 0.10 | 1.52 ± 0.09 | 0.22 ± 0.01 |
| Ca (% w/w) | 1.05 ± 0.17 | 2.22 ± 0.37 | 2.06 ± 0.34 | |
| Mg (% w/w) | 0.55 ± 0.10 | 0.32 ± 0.06 | 0.05 ±0.01 | |
| Na (% w/w) | 0.45 ± 0.08 | 0.34 ± 0.06 | 0.27 ± 0.05 | |
| P (% w/w) | 0.07 ± 0.01 | 0.08 ± 0.02 | 1.20 ± 0.24 | |
| Fe (mg/kg) | 29.5 ± 6.6 | 28.3 ± 6.3 | 8.5 ± 1.9 | |
| Mn (mg/kg) | 13.2 ± 1.7 | 7.9 ± 1.0 | <0.5 | |
| Cu (mg/kg) | 1.3 ± 0.3 | 1.0 ± 0.2 | <1.0 | |
| Zn (mg/kg) | 21.5 ± 3.1 | 12.2 ± 1.8 | 12 ± 1.7 | |
| Se (mg/kg) | <1.0 | <1.0 | <1.0 | |
| Si (mg/kg) | 215.8 ± 42.7 | 83.8 ± 16.6 | 63.7 ± 12.6 | |
| Al (mg/kg) | 37.4 ± 5.6 | 19.5 ± 2.9 | 3.6 ± 0.5 | |
| Co (mg/kg) | <0.5 | <0.5 | <0.5 | |
| Mo (mg/kg) | <0.5 | <0.5 | <0.5 | |
| B (mg/kg) | 43.1 ± 9.7 | 23.6 ± 5.3 | 10.1 ± 2.3 | |
| S (% w/w) | 0.31 ± 0.04 | 0.26 ± 0.04 | 0.16 ± 0.02 | |
| DM | Dry matter (%) | 29.4 ± 1.5 | 28.8 ± 1.4 | 24.5 ± 1.2 |
| Parameter | HNSW Dry Basis | HNSW-L Dry Basis | LNSW Dry Basis | LNSW-L Dry Basis |
|---|---|---|---|---|
| EC (μS/cm) | 76,243 ± 2859 | 1940 ± 73 | 143,389 ± 5377 | 1720 ± 65 |
| K (% w/w) | 8.16 ± 0.47 | 2.78 ± 0.16 | 5.17 ± 0.30 | 1.60 ± 0.09 |
| Ca (% w/w) | 4.11 ± 0.64 | 3.65 ± 0.60 | 9.16 ± 1.51 | 7.55 ± 1.25 |
| Mg (% w/w) | 1.91 ± 0.36 | 2.23 ± 0.42 | 1.09 ± 0.21 | 0.97 ± 0.18 |
| Na (% w/w) | 1.56 ± 0.27 | 0.60 ± 0.11 | 1.16 ± 0.20 | 0.41 ± 0.07 |
| P (% w/w) | 0.24 ± 0.05 | 0.24 ± 0.05 | 0.27 ± 0.05 | 0.27 ± 0.05 |
| Fe (mg/kg) | 102.4 ± 22.9 | 79.5 ± 17.8 | 96.3 ± 21.6 | 75.2 ± 16.8 |
| Mn (mg/kg) | 45.8 ± 6.0 | 52.0 ± 6.8 | 26.9 ± 3.5 | 29.9 ± 3.9 |
| Cu (mg/kg) | 4.5 ± 1.1 | 4.2 ± 1.0 | 3.4 ± 0.8 | 3.4 ± 0.8 |
| Zn (mg/kg) | 74.7 ± 10.8 | 79.9 ± 11.5 | 41.5 ± 6.0 | 40.0 ± 5.8 |
| Total C (% w/w) | 42.0 ± 0.6 | 39.9 ± 0.6 | 41.2 ± 0.6 | 37.2 ± 0.5 |
| Total N (% w/w) | 1.46 ± 0.16 | 1.34 ± 0.15 | 0.54 ± 0.06 | 0.44 ± 0.05 |
| No | Amino Acid | HNSW | LNSW | GFB |
|---|---|---|---|---|
| 1 | His | 0.18 ± 0.01 | 0.04 ± 0.01 | 5.74 ± 0.79 |
| 2 | Ser | 0.55 ± 0.04 | 0.06 ± 0.00 | 32.77 ± 3.61 |
| 3 | Arg | 0.35 ± 0.01 | 0.04 ± 0.00 | 44.35 ± 4.85 |
| 4 | Gly | 2.86 ± 0.02 | 0.73 ± 0.02 | 73.55 ± 8.14 |
| 5 | Asp | 3.58 ± 0.04 | 0.94 ± 0.03 | 56.26 ± 7.83 |
| 6 | Glu | 5.96 ± 0.04 | 1.28 ± 0.04 | 84.52 ± 10.64 |
| 7 | Thr | 0.60 ± 0.00 | 0.07 ± 0.00 | 25.27 ± 3.00 |
| 8 | Ala | 3.59 ± 0.03 | 1.02 ± 0.06 | 48.68 ± 5.70 |
| 9 | Pro | 1.86 ± 0.01 | 0.48 ± 0.02 | 39.38 ± 4.64 |
| 10 | Cys | - | - | 2.04 ± 0.23 |
| 11 | Lys | 1.70 ± 0.04 | 0.45 ± 0.04 | 51.71 ± 3.70 |
| 12 | Tyr | 1.51 ± 0.09 | 0.41 ± 0.01 | 26.77 ± 2.26 |
| 13 | Met | 0.71 ± 0.01 | 0.15 ± 0.01 | 17.04 ± 1.77 |
| 14 | Val | 2.50 ± 0.02 | 0.63 ± 0.03 | 25.29 ± 3.20 |
| 15 | ILe | 1.88 ± 0.02 | 0.38 ± 0.02 | 20.88 ± 2.81 |
| 16 | Leu | 3.77 ± 0.01 | 0.91 ± 0.04 | 39.71 ± 4.95 |
| 17 | Phe | 2.28 ± 0.04 | 0.59 ± 0.03 | 22.07 ± 2.43 |
| Extract | Control | HNSW-E-EA | LNSW-E-EA | |||
|---|---|---|---|---|---|---|
| Species | 16% | 80% | 100% | 80% | 100% | |
| E. coli | 27.0 | n.e.z. | n.e.z. | |||
| 28.0 | n.e.z. | n.e.z. | ||||
| 27.0 | n.e.z. | n.e.z. | ||||
| 28.0 | n.e.z. | n.e.z. | ||||
| 28.0 | n.e.z. | n.e.z. | ||||
| Average | 27.6 | n.c.v. | n.c.v. | |||
| SD | 0.5 | n.c.v. | n.c.v. | |||
| S. aureus | 15.0 | 9.0 | 11.0 | 15.0 | 9.0 | |
| 16.0 | 10.0 | 12.0 | 16.0 | 10.0 | ||
| 16.0 | 10.0 | 11.0 | 16.0 | 10.0 | ||
| 15.0 | 8.0 | 12.0 | 15.0 | 8.0 | ||
| 16.0 | 9.0 | 10.0 | 16.0 | 9.0 | ||
| Average | 15.6 | 9.3 | 11.2 | 15.6 | 9.3 | |
| SD | 0.5 | 0.8 | 0.8 | 0.5 | 0.8 | |
| Concentration (ngextract/g Dry Material/mL DMSO) | Migration Speed (mm/h) | |
|---|---|---|
| Control | 0.01130 ± 0.001 | |
| 0.1% DMSO | 0.00881 ± 0.002 | |
| HNSW-E-EA | 28.0 × 102 | 0.00797 ± 0.001 |
| 28.0 | 0.01100 ± 0.001 | |
| 28.0 × 10−2 | 0.00898 ± 0.001 | |
| LNSW-E-EA | 24.6 × 102 | 0.00839± 0.001 |
| 24.6 | 0.01190 ± 0.001 | |
| 24.6 × 10−2 | 0.00881 ± 0.002 | |
| GFB-E-EA | 64.8 × 102 | 0.00840 ± 0.001 |
| 64.8 | 0.00750 ± 0.001 | |
| 64.8 × 10−2 | 0.01250 ± 0.002 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maroulis, M.; Matsia, S.; Lazopoulos, G.; Pârvulescu, O.C.; Ion, V.A.; Bujor, O.-C.; Cabell, J.; Løes, A.-K.; Salifoglou, A. Chemical and Biological Profiling of Fish and Seaweed Residues to Be Applied for Plant Fertilization. Agronomy 2023, 13, 2258. https://doi.org/10.3390/agronomy13092258
Maroulis M, Matsia S, Lazopoulos G, Pârvulescu OC, Ion VA, Bujor O-C, Cabell J, Løes A-K, Salifoglou A. Chemical and Biological Profiling of Fish and Seaweed Residues to Be Applied for Plant Fertilization. Agronomy. 2023; 13(9):2258. https://doi.org/10.3390/agronomy13092258
Chicago/Turabian StyleMaroulis, Marios, Sevasti Matsia, Georgios Lazopoulos, Oana Cristina Pârvulescu, Violeta Alexandra Ion, Oana-Crina Bujor, Joshua Cabell, Anne-Kristin Løes, and Athanasios Salifoglou. 2023. "Chemical and Biological Profiling of Fish and Seaweed Residues to Be Applied for Plant Fertilization" Agronomy 13, no. 9: 2258. https://doi.org/10.3390/agronomy13092258
APA StyleMaroulis, M., Matsia, S., Lazopoulos, G., Pârvulescu, O. C., Ion, V. A., Bujor, O.-C., Cabell, J., Løes, A.-K., & Salifoglou, A. (2023). Chemical and Biological Profiling of Fish and Seaweed Residues to Be Applied for Plant Fertilization. Agronomy, 13(9), 2258. https://doi.org/10.3390/agronomy13092258