Fungi Parasitizing Powdery Mildew Fungi: Ampelomyces Strains as Biocontrol Agents against Powdery Mildews
Abstract
1. Introduction
2. Powdery Mildew Fungi
3. Ampelomyces, a Genus in Need of Taxonomic Revision
4. Ampelomyces as an Ecofriendly Biocontrol Agent against PM
5. Morphological and Molecular Analyses, and the Identification of Ampelomyces Strains
6. Ampelomyces Strains May Be Associated with, but Are Not Specific to, Their Host PM Species
7. Latest Results on Ampelomyces—PM Interaction
7.1. Methodological Considerations of Spray Inoculation of Ampelomyces Spores onto PM Colonies
7.2. Infection Processes of Ampelomyces Strains in PM Fungi
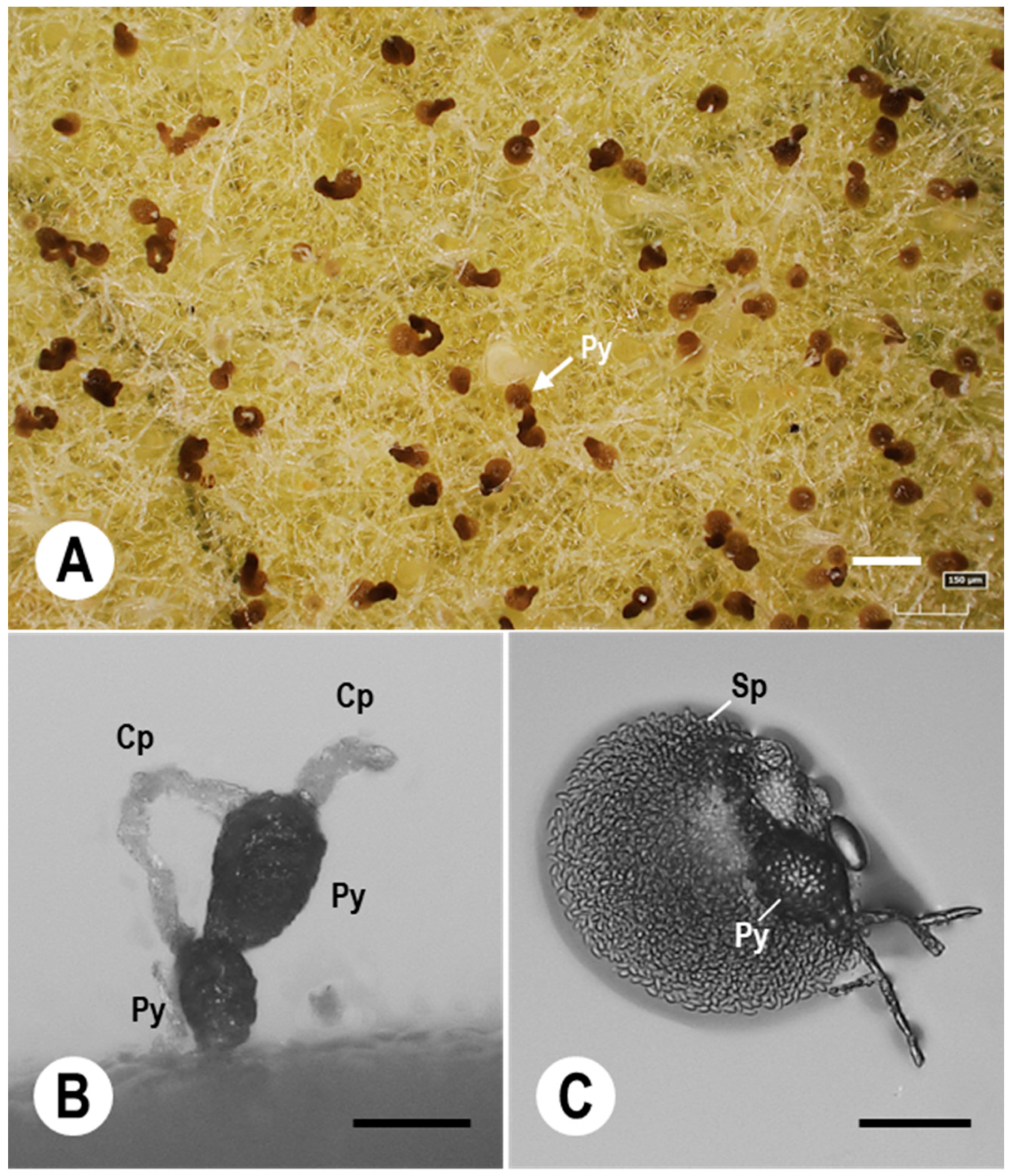
7.3. Pycnidial Development of Ampelomyces Strains in PM Fungi
7.4. Quantitative Analysis of PM Conidia Released from Ampelomyces-Parasitized PM Colonies under Greenhouse Conditions
8. Practical Application of Ampelomyces Strains as Biocontrol Agents of PM
9. Formulation and Commercialization of Ampelomyces as BCAs
10. An Ideal Spray Inoculation System for the Effective Use of Ampelomyces as a BCA
11. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kiss, L.; Vaghefi, N.; Bransgrove, K.; Dearnaley, J.D.W.; Takamatsu, S.; Tan, Y.P.; Marston, C.; Liu, S.-Y.; Jin, D.-N.; Adorada, D.L.; et al. Australia: A continent without native powdery mildews? The first comprehensive catalog indicates recent introductions and multiple host range expansion events, and leads to the re-discovery of Salmonomyces as a new lineage of the Erysiphales. Front. Microbiol. 2020, 11, 1571. [Google Scholar] [CrossRef]
- Vaghefi, N.; Kusch, S.; Németh, M.Z.; Seress, D.; Braun, U.; Takamatsu, S.; Panstruga, R.; Kiss, L. Beyond nuclear ribosomal DNA sequences: Evolution, taxonomy, and closest known saprobic relatives of powdery mildew fungi (Erysiphaceae) inferred from their first comprehensive genome-scale phylogenetic analyses. Front. Microbiol. 2022, 13, 903024. [Google Scholar] [CrossRef] [PubMed]
- Spencer, D.M. Powdery mildew of strawberries. In The Powdery Mildews; Spencer, D.M., Ed.; Academic Press: New York, NY, USA, 1978; pp. 355–358. [Google Scholar]
- Keinath, T.P.; Wintermantel, W.M.; Zitter, T.A. Compendium of Cucurbit Diseases; APS Press: St. Paul, MN, USA, 2017. [Google Scholar]
- Miyamoto, T.; Hayashi, K.; Ogawara, T. First report of the occurrence of multiple resistance to Flutianil and Pyriofenone in field isolates of Podosphaera xanthii, the causal fungus of cucumber powdery mildew. Eur. J. Plant Pathol. 2020, 156, 953–963. [Google Scholar] [CrossRef]
- Miyamoto, T.; Hayashi, K.; Okada, R.; Wari, D.; Ogawara, T. Resistance to succinate dehydrogenase inhibitors in field isolates of Podosphaera xanthii on cucumber: Monitoring, cross-resistance patterns and molecular characterization. Pestic. Biochem. Physiol. 2020, 169, 104646. [Google Scholar] [CrossRef] [PubMed]
- Vielba-Fernández, A.; Polonio, A.; Ruiz-Jiménez, L.; de Vicente, A.; Pérez-García, A.; Fernández-Ortuño, D. Fungicide resistance in powdery mildew fungi. Microorganisms 2020, 8, 1431. [Google Scholar] [CrossRef]
- Jeffries, P.; Young, T.W.K. Interfungal Parasitic Relationships; CABI Publishing: Wallingford, UK, 1994. [Google Scholar]
- Prahl, R.E.; Khan, S.; Deo, R.C. Ampelomyces mycoparasites of powdery mildews–A review. Can. J. Plant Pathol. 2023. [Google Scholar] [CrossRef]
- Kiss, L. A review of fungal antagonists of powdery mildews and their potential as biocontrol agents. Pest Manag. Sci. 2003, 59, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Kiss, L. Intracellular mycoparasites in action: Interactions between powdery mildew fungi and Ampelomyces. In Stress in Yeasts and Filamentous Fungi; Avery, S.V., Stratford, M., van West, P., Eds.; Academic Press: Cambridge, MA, USA, 2008; Volume 27, pp. 37–52. [Google Scholar]
- Hijwegen, T.; Buchenauer, H. Isolation and identification of hyperparasitic fungi associated with Erysiphaceae. Neth. J. Plant Pathol. 1984, 90, 79–84. [Google Scholar] [CrossRef]
- Jarvis, W.R.; Shaw, L.A.; Traquair, J.A. Factors affecting antagonism of cucumber powdery mildew by Stephanoascus flocculosus and S. rugulosus. Mycol. Res. 1989, 92, 162–165. [Google Scholar] [CrossRef]
- Verhaar, M.A.; Hijwegen, T.; Zadoks, J.C. Glasshouse experiments on biocontrol of cucumber powdery mildew (Sphaerotheca fuliginea) by the mycoparasites Verticillium lecanii and Sporothrix rugulosa. Biol. Cont. 1996, 6, 353–360. [Google Scholar] [CrossRef]
- Hoch, H.C.; Provvidenti, R. Mycoparasitic relationships: Cytology of the Sphaerotheca fuliginea-Tilletiopsis sp. interaction. Phytopathology 1979, 69, 359–362. [Google Scholar] [CrossRef]
- Hijwegen, T. Biological control of cucumber powdery mildew by Tilletiopsis minor. Neth. J. Plant Pathol. 1986, 92, 93–95. [Google Scholar] [CrossRef]
- Cesati, V. Ampelomyces quisqualis Ces. Bot. Ztg. 1852, 10, 301–302. [Google Scholar]
- Yarwood, C.E. An overwintering pycnidial stage of Cicinnobolus. Mycologia 1939, 31, 420–422. [Google Scholar] [CrossRef]
- Jarvis, W.R.; Slingsby, K. The control of powdery mildew of greenhouse cucumber by water sprays and Ampelomyces quisqualis. Plant Dis. Reptr. 1977, 61, 728–730. [Google Scholar]
- Sundheim, L. Control of cucumber powdery mildew by the hyperparasite Ampelomyces quisquialis and fungicides. Plant Pathol. 1982, 31, 209–214. [Google Scholar] [CrossRef]
- Kiss, L. Natural occurrence of Ampelomyces intracellular mycoparasites in mycelia of powdery mildew fungi. New Phytol. 1998, 140, 709–714. [Google Scholar] [CrossRef]
- Falk, S.P.; Gadoury, D.M.; Cortesi, P.; Pearson, R.C.; Seem, R.C. Parasitism of Uncinula necator cleistothecia by the mycoparasite Ampelomyces quisqualis. Phytopathology 1995, 85, 794–800. [Google Scholar] [CrossRef]
- Huth, L.; Ash, G.J.; Idnurm, A.; Kiss, L.; Vaghefi, N. The “bipartite” structure of the first genome of Ampelomyces quisqualis, a common hyperparasite and biocontrol agent of powdery mildews, may point to its evolutionary origin from plant pathogenic fungi. Genome Biol. Evol. 2021, 13, evab182. [Google Scholar] [CrossRef]
- Kiss, L.; Russell, J.C.; Szentiványi, O.; Xu, X.; Jeffries, P. Biology and biocontrol potential of Ampelomyces mycoparasites, natural antagonists of powdery mildew fungi. Biocont. Sci. Technol. 2004, 14, 635–651. [Google Scholar] [CrossRef]
- Manjunatha, L.; Singh, S.; Ravikumara, B.M.; Narasa Reddy, G.; Senthikumar, M. Ampelomyces. In Beneficial Microbes in Agro-ecology; Amaresan, N., Senthil Kumar, M., Annapuma, K., Kumar, K., Sankaranarayanan, A., Eds.; Academic Press: Amsterdam, The Netherlands, 2020; Chapter 44; pp. 833–860. [Google Scholar] [CrossRef]
- Matsuda, Y.; Kashimoto, K.; Takikawa, Y.; Aikami, R.; Nonomura, T.; Toyoda, H. Occurrence of new powdery mildew on greenhouse tomato cultivars. J. Gen. Plant Pathol. 2001, 67, 294–298. [Google Scholar] [CrossRef]
- White, J.F., Jr.; Johnston, S.A.; Wang, C.L.; Chin, C.K. First report of powdery mildew in greenhouse-grown tomatoes in New Jersey. Plant Dis. 1997, 81, 227. [Google Scholar] [CrossRef] [PubMed]
- Braun, U.; Cook, R.T.A. Taxonomic Manual of the Erysiphales (Powdery Mildews); CBS-KNAW Fungal Biodiversity Centre: Utrecht, The Netherlands, 2012. [Google Scholar]
- Yarwood, C.E. Powdery mildews. Bot. Rev. 1957, 23, 235–301. [Google Scholar] [CrossRef]
- Braun, U. A monograph of the Erysiphales (powdery mildews). Beih. Nova Hedwig. 1987, 89, 1–700. [Google Scholar]
- Rallos, L.E.E.; Baudoin, A.B. Co-occurrence of two allelic variants of CYP51 in Erysiphe necator and their correlation with over-expression for DMI resistance. PLoS ONE 2016, 11, e0148025. [Google Scholar] [CrossRef]
- Pintye, A.; Németh, M.Z.; Molnár, O.; Horváth, Á.; Spitzmüller, Z.; Szalóki, N.; Pal, K.; Váczy, K.; Kovács, G. Improved DNA extraction and quantitative real-time PCR for genotyping Erysiphe necator and detecting the DMI fungicide resistance marker A495T, using single ascocarps. Phytopathol. Medit. 2020, 59, 97–106. [Google Scholar] [CrossRef]
- Huggenberger, F.; Collins, M.A.; Skylakakis, G. Decreased sensitivity of Sphaerotheca fuliginea to fenarimol and other ergosterol-biosynthesis inhibitors. Crop Prot. 1984, 3, 137–149. [Google Scholar] [CrossRef]
- McGrath, M.T.; Shishkoff, N. Resistance to triadimefon and benomyl: Dynamics and impact on managing cucurbit powdery mildew. Plant Dis. 2001, 85, 147–154. [Google Scholar] [CrossRef]
- McGrath, M.T.; Shishkoff, N. Frist report of the cucurbit powdery mildew fungus (Podosphaera xanthii) resistant to strobilurin fungicides in the United States. Plant Dis. 2003, 87, 1007. [Google Scholar] [CrossRef]
- Lebeda, A.; Sedláková, B. Fungicide resistance in population of cucurbit powdery mildew. J. Plant Pathol. 2008, 90, S2.142. [Google Scholar]
- Petit, A.N.; Fontaine, F.; Vatsa, P.; Clément, C.; Vaillant-Gaveau, N. Fungicide impacts on photosynthesis in crop plants. Photosyn. Res. 2012, 111, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Geiger, F.; Bengtsson, J.; Berendse, F.; Weisser, W.W.; Emmerson, M.; Morales, M.B.; Ceryngier, P.; Liira, J.; Tscharntke, T.; Winqvist, C.; et al. Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl. Ecol. 2010, 11, 97–105. [Google Scholar] [CrossRef]
- De Bary, A. Eurotium, Erysiphe, Cicinnobolus: Nebst Bemerkungen über die Geschlechtsorgane der Ascomyceten. In Beitrage zur Morphologie und Physiologie der Pilze; De Bary, A., Woronin, M., Eds.; Verlag von C. Winter: Frankfurt, Germany, 1870; pp. 1–95. [Google Scholar]
- Rogers, D. On Cicinobolus. Mycologia 1959, 51, 96–98. [Google Scholar] [CrossRef]
- Kiss, L.; Nakasone, K.K. Ribosomal DNA internal transcribed spacer sequences do not support the species status of Ampelomyces quisqualis, a hyperparasite of powdery mildew fungi. Curr. Genet. 1998, 33, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Yang, J.; Kovács, G.M.; Szentiványi, O.; Li, B.; Xu, X.; Kiss, L. Genetic diversity of Ampelomyces mycoparasites isolated from different powdery mildew species in China inferred from analyses of rDNA ITS sequences. Fungal Div. 2007, 24, 225–240. [Google Scholar]
- Pintye, A.; Bereczky, Z.; Kovács, G.M.; Nagy, L.G.; Xu, X.; Legler, S.E.; Váczy, Z.; Váczy, K.Z.; Caffi, T.; Rossi, V.; et al. No indication of strict host associations in a widespread mycoparasite: Grapevine powdery mildew (Erysiphe necator) is attacked by phylogenetically distant Ampelomyces strains in the field. Phytopathology 2012, 102, 707–716. [Google Scholar] [CrossRef]
- Park, M.-J.; Choi, Y.-J.; Hong, S.-B.; Shin, H.-D. Genetic variability and mycohost association of Ampelomyces quisqualis isolates inferred from phylogenetic analyses of ITS rDNA and actin gene sequences. Fungal Biol. 2010, 114, 235–247. [Google Scholar] [CrossRef]
- Prahl, R.E.; Khan, S.; Deo, R.C. The role of internal transcribed spacer 2 secondary structures in classifying mycoparasitic Ampelomyces. PLoS ONE 2021, 16, e0253772. [Google Scholar] [CrossRef]
- Legler, S.E.; Pintye, A.; Caffi, T.; Gulyás, S.; Bohár, G.; Rossi, V.; Kiss, L. Sporulation rate in culture and mycoparasitic activity, but not mycohost specificity, are the key factors for selecting Ampelomyces strains for biocontrol of grapevine powdery mildew (Erysiphe necator). Eur. J. Plant Pathol. 2016, 144, 723–736. [Google Scholar] [CrossRef]
- Kiss, L. Genetic diversity in Ampelomyces isolates, hyperparasites of powdery mildew fungi, inferred from RFLP analysis of the rDNA ITS region. Mycol. Res. 1997, 101, 1073–1080. [Google Scholar] [CrossRef]
- Hino, I.; Kato, H. Cicinnoboli parasitic on mildew fungi. Bull. Miyazaki Coll. Agric. For. 1929, 1, 91–98. [Google Scholar]
- Emmons, C.W. Cicinnobolus cesatii, a study in host-parasite relationships. Bull. Torrey Bot. Club 1930, 57, 421–439. [Google Scholar] [CrossRef]
- Blumer, S. Die Erysiphaceen Mitteleuropas mit besonderer Berücksichtigung der Schweiz. Beitr. Zur Kryptogamenflora Schweiz 1933, 7, 1–483. [Google Scholar]
- Clare, B.G. Ampelomyces quisqualis (Cicinnobolus cesatii) on Queensland Erysiphaceae. Univ. Qld. Pap. 1964, 4, 147–149. [Google Scholar]
- Belsare, S.W.; Moniz, L.; Deo, V.B. The hyperparasite Ampelomyces quisqualis Ces. from Maharashtra State, India. Biovigyanam 1980, 6, 173–176. [Google Scholar]
- Hanlin, R.T.; Tortolero, O. Brasiliomyces, a new host for Ampelomyces. Mycotaxon 1984, 21, 459–462. [Google Scholar]
- Nagy, S.G.; Vajna, L. Ampelomyces fajok elöfordulása lisztharmatgombákban Magyarországon. Mikológiai Közlemények 1990, 29, 103–112. (In Hungarian) [Google Scholar]
- Puzanova, L.A. Distribution of hyperparasites from the genus Ampelomyces Ces. ex Schlecht. on the powdery mildew fungi in northern Caucasus and their importance in disease control. Mikol. I Fitopatol. 1991, 25, 438–442. (In Russian) [Google Scholar]
- Tsay, J.G.; Tung, B. Ampelomyces quisqualis Ces. ex Schlecht., a hyperparasite of the asparagus bean powdery mildew pathogen Erysiphe polygoni in Taiwan. Trans. Mycol. Soc. R.O.C. 1991, 6, 55–58. [Google Scholar]
- Paulech, C.; Herrera, S.; Fornet, E. Phytopathogenic micromycetes of the family Erysiphaceae distributed in Cuba. Ceska Mykol. 1993, 46, 303–314. (In Slovakian) [Google Scholar]
- Shin, H.-D. Isolation and identification of hyperparasites against powdery mildew fungi in Korea. Kor. J. Mycol. 1994, 22, 355–365. (In Korean) [Google Scholar]
- Kiss, L. Graminicolous powdery mildew fungi as new natural hosts of Ampelomyces mycoparasites. Can. J. Bot. 1997, 75, 680–683. [Google Scholar] [CrossRef]
- Ranković, B. Hyperparasites of the genus Ampelomyces on powdery mildew fungi in Serbia. Mycopathologia 1997, 139, 157–164. [Google Scholar] [CrossRef]
- Németh, M.Z.; Mizuno, Y.; Kobayashi, H.; Seress, D.; Shishido, N.; Kimura, Y.; Takamatsu, S.; Suzuki, T.; Takikawa, Y.; Kakutani, K.; et al. Ampelomyces strains isolated from diverse powdery mildew hosts in Japan: Their phylogeny and mycoparasitic activity, including timing and quantifying mycoparasitism of Pseudoidium neolycopersici on tomato. PLoS ONE 2021, 16, e0251444. [Google Scholar] [CrossRef] [PubMed]
- Németh, M.Z.; Pintye, A.; Horváth, Á.N.; Vági, P.; Kovács, G.M.; Gorfer, M.; Kiss, L. Green fluorescent protein transformation sheds more light on a widespread mycoparasitic interaction. Phytopathology 2019, 109, 1404–1416. [Google Scholar] [CrossRef] [PubMed]
- Riess, H. Uber Byssocystis textilis. Hedwigia 1852, 1, 23. [Google Scholar]
- Yarwood, C.E. Ampelomyces quisqualis on clover mildew. Phytopathology 1932, 22, 31. [Google Scholar]
- Siozios, S.; Tosi, L.; Ferrarini, A.; Ferrari, A.; Tononi, P.; Bellin, D.; Maurhofer, M.; Gessler, C.; Delledonne, M.; Pertot, I. Transcriptional reprogramming of the mycoparasitic fungus Ampelomyces quisqualis during the powdery mildew host-induced germination. Phytopathology 2015, 105, 199–209. [Google Scholar] [CrossRef]
- Sivakumar, T.; Balabaskar, P.; Renganathan, P.; Sanjeevkumar, K. To evaluate the bio-efficacy and phytotoxicty of powder formulation of Biodewcon (Ampelomyces quisqualis 2.00% Wp) against powdery mildew (Sphaerotheca fuliginea) in cucumber crop. Plant Arch. 2020, 20, 3811–3815. [Google Scholar]
- Németh, M.Z.; Li, G.; Seress, D.; Pintye, A.; Molnár, O.; Kovács, G.M.; Kiss, L.; Gorfer, M. What is the role of the nitrate reductase (euknr) gene in fungi that live in nitrate-free environments? A targeted gene knock-out study in Ampelomyces mycoparasites. Fungal Biol. 2021, 125, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Sundheim, L.; Krekling, T. Host-parasite relationships of the hyperparasite Ampelomyces quisqualis and its powdery mildew host Sphaerotheca fuliginea. Phytopath. Z. 1982, 104, 202–210. [Google Scholar] [CrossRef]
- Hashioka, Y.; Nakai, Y. Ultrastructure of pycnidial development and mycoparasitism of Ampelomyces quisqualis parasitic on Erysiphales. Trans. Mycol. Soc. Jpn. 1980, 21, 329–338. [Google Scholar]
- Angeli, D.; Puopolo, G.; Maurhofer, M.; Gessler, C.; Pertot, I. Is the mycoparasitic activity of Ampelomyces quisqualis biocontrol strains related to phylogeny and hydrolytic enzyme production? Biol. Cont. 2012, 63, 348–358. [Google Scholar] [CrossRef]
- Rotem, Y.; Yarden, O.; Sztejnberg, A. The mycoparasite Ampelomyces quisqualis expresses exgA encoding and exo-β-1,3-glucanase in culture and during mycoparasitism. Phytopathology 1999, 89, 631–638. [Google Scholar] [CrossRef]
- Philipp, W.D. Extracellular enzymes and nutritional physiology of Ampelomyces quisqualis Ces., hyperparasite of powdery mildew, in vitro. J. Phytopathol. 1985, 114, 274–283. [Google Scholar] [CrossRef]
- Vaidya, S.; Thakur, V.S. Ampelomyces quisqualis Ces.–A mycoparasite of apple powdery mildew in western Himalayas. Indian Phytopathol. 2005, 58, 250–251. [Google Scholar]
- Szentiványi, O.; Kiss, L.; Russell, J.C.; Kovács, G.M.; Varga, K.; Jankovics, T.; Lesemann, S.; Xu, X.-M.; Jeffries, P. Ampelomyces mycoparasites from apple powdery mildew identified as a distinct group based on single-stranded conformation polymorphism analysis of the rDNA ITS region. Mycol. Res. 2005, 109, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Liyanage, K.K.; Khan, S.; Brooks, S.; Mortimer, P.E.; Karunarathna, S.C.; Xu, J.; Hyde, K.D. Morpho-molecular characterization of two Ampelomyces spp. (Pleosporales) strains mycoparasites of powdery mildew of Hevea brasiliensis. Front. Microbiol. 2018, 9, 1–10. [Google Scholar] [CrossRef]
- Angeli, D.; Maurhofer, M.; Gessler, C.; Pertot, I. Existence of different physiological forms within genetically diverse strains of Ampelomyces quisqualis. Phytoparasitica 2012, 40, 37–51. [Google Scholar] [CrossRef]
- Kiss, L.; Pintye, A.; Kovács, G.M.; Jankovics, T.; Fontaine, M.C.; Harvey, N.; Xu, X.; Nicot, P.C.; Bardin, M.; Shykoff, J.A.; et al. Temporal isolation explains host-related genetic differentiation in a group of widespread mycoparasitic fungi. Mol. Ecol. 2011, 20, 1492–1507. [Google Scholar] [CrossRef]
- Pintye, A.; Ropars, J.; Harvey, N.; Shin, H.-D.; Leyronas, C.; Nicot, P.C.; Giraud, T.; Kiss, L. Host phenology and geography as drivers of differentiation in generalist fungal mycoparasites. PLoS ONE 2015, 10, e0120703. [Google Scholar] [CrossRef]
- Sztejnberg, A.; Galper, S.; Mazar, S.; Lisker, N. Ampelomyces quisqualis for biological and integrated control of powdery mildews in Israel. J. Phytopathol. 1989, 124, 285–295. [Google Scholar] [CrossRef]
- Paratt, S.R.; Laine, A.-L. Pathogen dynamics under both bottom-up host resistance and top-down hyperparasite attack. J. Appl. Ecol. 2018, 55, 2976–2985. [Google Scholar] [CrossRef] [PubMed]
- Philipp, W.-D.; Crüger, G. Parasitismus von Ampelomyces quisqualis auf echten Mehltaupilzenal an Gurken und anderen Gemüsearten. Z. Pflanzenkrankh. Pflanzenschutz 1979, 86, 129–142. [Google Scholar]
- Falk, S.P.; Gadoury, D.M.; Pearson, R.C.; Seem, R.C. Partial control of grape powdery mildew by the mycoparasite Ampelomyces quisqualis. Plant Dis. 1995, 79, 483–490. [Google Scholar] [CrossRef]
- Mairovich, G.; Marder, J.B.; Shtienberg, D.; Sztejnberg, A. Studies of biological control of powdery mildew in cereals by the hyperparasite Ampelomyces quisqualis. Phytoparasitica 1996, 24, 157–158. [Google Scholar]
- Szentiványi, O.; Kiss, L. Overwintering of Ampelomyces mycoparasites on apple trees and other plants infected with powdery mildews. Plant Pathol. 2003, 52, 737–746. [Google Scholar] [CrossRef]
- Poulin, R.; Krasnov, B.R.; Mouillot, D. Host specificity in phylogenetic and geographic space. Trends Parasitol. 2011, 27, 355–361. [Google Scholar] [CrossRef]
- Sztejnberg, A. Biological control of powdery mildews by Ampelomyces quisqualis. Phytopathology 1979, 69, 1047. [Google Scholar]
- Sundheim, L. Ampelomyces quisqualis, a hyperparasitic fungus in biological control of powdery mildews on greenhouse cucumber. Acta Hortic. 1984, 156, 229–236. [Google Scholar] [CrossRef]
- Philipp, W.-D.; Grauer, U.; Grossmann, F. Ergänzende Untersuchungen zur biologischen und integrierten Bekämpfung von Gurkenmehltau unter Glass durch Ampelomyces quisqualis. Z. Pflanzenkrankh. Pflanzenschutz 1984, 91, 438–443. [Google Scholar]
- Sztejnberg, A.; Mazar, S. Biocontrol of cucumber and carrot powdery mildew by Ampelomyces quisqualis. Phytopathology 1985, 75, 1301–1302. [Google Scholar]
- Dik, A.J.; Verhaar, M.A.; Bélanger, R.R. Comparison of three biological control agents against cucumber powdery mildew (Sphaerotheca fuliginea) in semi-commercial-scale glasshouse trials. Eur. J. Plant Pathol. 1998, 104, 413–423. [Google Scholar] [CrossRef]
- Shishkoff, N.; McGrath, M.T. AQ10 biofungicide combined with chemical fungicides or AddQ spray adjuvant for control of cucurbit powdery mildew in detached leaf culture. Plant Dis. 2002, 86, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Angeli, D.; Saharan, K.; Segarra, G.; Sicher, C.; Pertot, I. Production of Ampelomyces quisqualis conidia in submerged fermentation and improvements in the formulation for increased shelf-life. Crop Prot. 2017, 97, 135–144. [Google Scholar] [CrossRef]
- Sztejnberg, A.; Mazar, S. Studies on the hyperparasite Ampelomyces quisqualis and preliminary trials on biological control of powdery mildew. Phytoparasitica 1983, 11, 219–220. [Google Scholar]
- Romero, D.; Rivera, M.E.; Cazorla, F.M.; De Vicente, A.; Pérez-García, A. Effect of mycoparasitic fungi on the development of Sphaerotheca fusca in melon leaves. Mycol. Res. 2003, 107, 64–71. [Google Scholar] [CrossRef]
- Kimura, Y.; Németh, M.Z.; Numano, K.; Mitao, A.; Shirakawa, T.; Seress, D.; Takikawa, Y.; Kakutani, K.; Matsuda, Y.; Kiss, L.; et al. Hyperparasitic fungi against melon powdery mildew pathogens: Quantitative analysis of conidia released from single colonies of Podosphaera xanthii parasitized by Ampelomyces. Agronomy 2023, 13, 1204. [Google Scholar] [CrossRef]
- Hofstein, R.; Fridlender, B. Development of production, formulation and delivery systems. In Brighton Crop Protection Conference, Pest and Diseases; British Crop Protection Council: Farnham, UK, 1994; Volume 3, pp. 1273–1280. [Google Scholar]
- Hofstein, R.; Daoust, R.A.; Aeschlimann, J.P. Constrains to the development of biofungicides: The example of “AQ10”, a new product for controlling powdery mildews. Entomophaga 1996, 41, 455–460. [Google Scholar] [CrossRef]
- Pasini, C.; D’Aquila, F.; Curir, P.; Gullino, M.L. Effectiveness of antifungal compounds against rose powdery mildew (Sphaerotheca pannosa var. rosae) in glasshouses. Crop Prot. 1997, 16, 251–256. [Google Scholar] [CrossRef]
- Pertot, I.; Zasso, R.; Amsalem, L.; Baldessari, M.; Angeli, G.; Elad, Y. Integrating biocontrol agents in strawberry powdery mildew control strategies in high tunnel growing systems. Crop Prot. 2008, 27, 622–631. [Google Scholar] [CrossRef]
- Gautam, A.K.; Avasthi, S. Ampelomyces quisqualis Ces.–A mycoparasite of Euphorbia hirta powdery mildew in Himachal Pradesh, India. J. Phytopathol. Pest Manag. 2016, 3, 64–70. [Google Scholar]
- Parratt, S.R.; Barrès, B.; Penczykowski, R.M.; Laine, A.L. Local adaptation at higher trophic levels: Contrasting hyperparasite-pathogen infection dynamics in the field and laboratory. Mol. Ecol. 2017, 26, 1964–1979. [Google Scholar] [CrossRef] [PubMed]
- Carbó, A.; Teixidó, N.; Usall, J.; Solsona, C.; Torres, R. Formulated Ampelomyces quisqualis CPA-9 applied on zucchini leaves: Influence of abiotic factors and powdery mildew mycoparasitization. Eur. J. Plant Pathol. 2021, 161, 37–48. [Google Scholar] [CrossRef]
- Philipp, W.-D.; Beuther, E.; Hermann, D.; Klinkert, F.; Oberwalder, C.; Schmidtke, M.; Straub, B. Zur Formulierung des Mehltauhyperparasiten Ampelomyces quisqualis Ces. Z. Pflanzenkrankh. Pflanzenschutz 1990, 97, 120–132. [Google Scholar]
- Gu, Y.H.; Ko, W.H. Water agarose medium for studying factors affecting germination of conidia of Ampelomyces quisqualis. Mycol. Res. 1997, 101, 422–424. [Google Scholar] [CrossRef]
- Suzuki, T.; Murakami, T.; Takizumi, Y.; Ishimaru, H.; Kudo, D.; Takikawa, Y.; Matsuda, Y.; Kakutani, K.; Bai, Y.; Nonomura, T. Trichomes: Interaction sites of tomato leaves with biotrophic powdery mildew pathogens. Eur. J. Plant Pathol. 2018, 150, 115–125. [Google Scholar] [CrossRef]
- Speer, E.O. Ampelomyces cesati (Fungi, Sphaeropsidales). Taxon 1978, 27, 549–562. [Google Scholar] [CrossRef]
- Kiss, L.; Pintye, A.; Zséli, G.; Jankovics, T.; Szentiványi, O.; Hafez, Y.M.; Cook, R.T.A. Microcyclic conidiogenesis in powdery mildews and its association with intracellular parasitism by Ampelomyces. Eur. J. Plant Pathol. 2010, 126, 445–451. [Google Scholar] [CrossRef]
- Zeng, L.F.; Jiang, Y.C. An investigation of parasitic fungi on Erysiphe spp. RoPP 1986, 65, 419. [Google Scholar]
- Machida, K.; Trifonov, L.S.; Ayer, W.A.; Lu, Z.-X.; Laroche, A.; Huang, H.C.; Cheng, K.J.; Zantige, J.L. 3(2H)-Benzofuranones and chromanes from liquid cultures of the mycoparasitic fungus Coniothyrium minitans. Phytochemistry 2001, 58, 173–177. [Google Scholar] [CrossRef] [PubMed]
- McQuilken, M.P.; Gemmell, J.; Whipps, J.M. Some nutritional factors affecting production of biomass and antifungal metabolites of Coniothyrium minitans. Biocont. Sci. Technol. 2002, 12, 443–454. [Google Scholar] [CrossRef]
- McQuilken, M.P.; Gemmell, J.; Hill, R.A.; Whipps, J.M. Production of macrosphelide A by the mycoparasite Coniothyrium minitans. FEMS Microbiol. Lett. 2003, 219, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Beuther, E.; Philipp, W.D.; Grossmann, F. Untersuchungen zum hyperparasitismus von Ampelomyces quisqualis auf gurkenmehltau (Sphaerotheca fuliginea). Phytopath. Z. 1981, 101, 265–270. [Google Scholar] [CrossRef]
- Aylor, D.E. The role of intermittent wind in the dispersal of fungal pathogens. Annu. Rev. Phytopathol. 1990, 28, 73–92. [Google Scholar] [CrossRef]
- Brown, J.K.M.; Hovmøller, M.S. Aerial dispersal of pathogens on the global and continental scales and its impact on plant disease. Science 2002, 297, 537–541. [Google Scholar] [CrossRef]
- Nonomura, T.; Matsuda, Y.; Yamashita, S.; Akahoshi, H.; Takikawa, Y.; Kakutani, K.; Toyoda, H. Natural woody plant, Mallotus japonicus, as an ecological partner to transfer different pathotypic conidia of Oidium neolycopersici to greenhouse tomatoes. Plant Protect. Sci. 2013, 49, S33–S40. [Google Scholar] [CrossRef]
- Suzuki, T.; Iwasaki, S.; Hisazumi, H.; Miyamoto, A.; Ogami, H.; Takikawa, Y.; Kakutani, K.; Matsuda, Y.; Nonomura, T. Inhibitory effects of blue light-emitting diode irradiation on Podosphaera xanthii conidial release and infection of melon seedlings. Agriculture 2022, 12, 198. [Google Scholar] [CrossRef]
- Rouse, D.I.; MacKenzie, D.R.; Nelson, R.R. Density dependent sporulation of Erysiphe graminis f. sp. tritici. Phytopathology 1984, 74, 1176–1180. [Google Scholar] [CrossRef]
- Bushnell, W.R. The role of powdery mildew research in understanding host-parasite interaction: Past, present, and future. In The Powdery Mildews: A Comprehensive Treatise; Bélanger, R.R., Bushnell, W.R., Dik, A.J., Carver, T.L.W., Eds.; APS Press: St. Paul, MN, USA, 2002; pp. 1–12. [Google Scholar]
- Suzuki, T.; Nakamura, R.; Takagi, N.; Takikawa, Y.; Kakutani, K.; Matsuda, Y.; Matsui, K.; Nonomura, T. Quantitative analysis of the lifelong production of conidia released from single colonies of Podosphaera xanthii on melon leaves using electrostatic techniques. Austral. Plant Pathol. 2019, 48, 297–307. [Google Scholar] [CrossRef]
- Ayabe, S.; Kimura, Y.; Umei, N.; Takikawa, Y.; Kakutani, K.; Matsuda, Y.; Nonomura, T. Real-time collection of conidia released from living single colonies of Podosphaera aphanis on strawberry leaves under natural conditions with electrostatic techniques. Plants 2022, 11, 3453. [Google Scholar] [CrossRef]
- Kiss, L. The role of hyperparasites in host plant–parasitic fungi relationships. In Biotic Interactions in Plant–Pathogen Associations; Jeger, M.J., Spence, N.J., Eds.; CABI Publishing: Wallingford, UK, 2001; pp. 227–236. [Google Scholar]
- Daoust, R.A.; Hofstein, R. Ampelomyces quisqualis, a new biofungicide to control powdery mildew in grapes. In Brighton Crop Protection Conference, Pest and Diseases; British Crop Protection Council: Farnham, UK, 1996; Volume 1, pp. 33–40. [Google Scholar]
- Sundheim, L.; Tronsmo, A. Hyperparasites in biological control. In Biocontrol of Plant Diseases; Mukerji, K.G., Garg, K.L., Eds.; CRC Press: Boca Raton, FL, USA, 1988; Volume 1, pp. 53–69. [Google Scholar]
- Kiss, L. How dangerous is the use of fungal biocontrol agents to nontarget organisms? New Phytol. 2004, 163, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Paulitz, T.C.; Bélanger, R.R. Biological control in greenhouse systems. Annu. Rev. Phytopathol. 2001, 39, 103–133. [Google Scholar] [CrossRef] [PubMed]
- Bélanger, R.R.; Labbé, C. Control of powdery mildews without chemicals: Prophylactic and biological alternatives for horticultural crops. In The Powdery Mildews: A Comprehensive Treatise; Bélanger, R.R., Bushnell, W.R., Dik, A.J., Carver, T.L.W., Eds.; APS Press: St. Paul, MN, USA, 2002; pp. 256–267. [Google Scholar]
- Kaur, L.; Gupta, B.; Sharma, I.M.; Joshi, A.K. Eco-friendly management of powdery mildew of mango through biocontrol agents. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 392–396. [Google Scholar] [CrossRef]
- Philipp, W.-D.; Hellstern, A. Biologische Mehltaubekämpfung mit Ampelomyces quisqualis bei reduzierter Luftfeuchtigkeit. Z. Pflanzenkrankh. Pflanzenschutz 1986, 93, 384–391. [Google Scholar]
- Verhaar, M.A.; Kerssies, A.; Hijwegen, T. Effect of RH on mycoparasitism of rose powdery mildew with and without treatments with mycoparasites. Z. Pflanzenkrankh. Pflanzenschutz 1999, 106, 158–165. [Google Scholar]
- Epton, H.A.S.; Hamed El Nil, Y.F. Improvement in the hyperparasitic activity of Ampelomyces quisqualis in the biocontrol of powdery mildew of cucumber. IOBC/WPRS Bull. 1993, 16, 86–89. [Google Scholar]
- Hijwegen, T. Biological control of cucumber powdery mildew with Tilletiopsis minor under greenhouse conditions. Neth. J. Plant Pathol. 1992, 98, 221–225. [Google Scholar] [CrossRef]
- McGrath, M.T.; Shishkoff, N. Evaluation of biocompatible products for managing cucurbit powdery mildew. Crop Prot. 1999, 18, 471–478. [Google Scholar] [CrossRef]
- Sztejnberg, A.; Galper, S.; Lisker, N. Conditions for pycnidial production and spore formation by Ampelomyces quisqualis. Can. J. Microbiol. 1990, 36, 193–198. [Google Scholar] [CrossRef]
- Gilardi, G.; Manker, D.C.; Garibaldi, A.; Gullino, M.L. Efficacy of the biocontrol agents Bacillus subtilis and Ampelomyces quisqualis applied in combination with fungicides against powdery mildew of zucchini. J. Plant Dis. Prot. 2008, 115, 208–213. [Google Scholar] [CrossRef]
- Hijwegen, T. Effect of seventeen fungicolous fungi on sporulation of cucumber powdery mildew. Neth. J. Plant Pathol. 1988, 94, 185–190. [Google Scholar] [CrossRef]
- Carbó, A.; Torres, R.; Usall, J.; Ballesta, J.; Teixidó, N. Biocontrol potential of Ampelomyces quisqualis strain CPA-9 against powdery mildew: Conidia production in liquid medium and efficacy on zucchini leaves. Sci. Hortic. 2020, 267, 109337. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Németh, M.Z.; Seress, D.; Nonomura, T. Fungi Parasitizing Powdery Mildew Fungi: Ampelomyces Strains as Biocontrol Agents against Powdery Mildews. Agronomy 2023, 13, 1991. https://doi.org/10.3390/agronomy13081991
Németh MZ, Seress D, Nonomura T. Fungi Parasitizing Powdery Mildew Fungi: Ampelomyces Strains as Biocontrol Agents against Powdery Mildews. Agronomy. 2023; 13(8):1991. https://doi.org/10.3390/agronomy13081991
Chicago/Turabian StyleNémeth, Márk Z., Diána Seress, and Teruo Nonomura. 2023. "Fungi Parasitizing Powdery Mildew Fungi: Ampelomyces Strains as Biocontrol Agents against Powdery Mildews" Agronomy 13, no. 8: 1991. https://doi.org/10.3390/agronomy13081991
APA StyleNémeth, M. Z., Seress, D., & Nonomura, T. (2023). Fungi Parasitizing Powdery Mildew Fungi: Ampelomyces Strains as Biocontrol Agents against Powdery Mildews. Agronomy, 13(8), 1991. https://doi.org/10.3390/agronomy13081991





