A Novel Caffeine Oleate Formulation as an Insecticide to Control Coffee Berry Borer, Hypothenemus hampei, and Other Coffee Pests
Abstract
1. Introduction
2. Materials and Methods
2.1. Caffeine Oleate Formulation
2.2. Preventive Effect of Caffeine Oleate on CBB under Laboratory Conditions
2.3. Curative Effect of Caffeine Oleate on CBB under Laboratory Conditions
2.4. Effect of Caffeine Oleate Emulsion Components against CBB under Laboratory Conditions
2.5. Evaluation of the Preventive Effect of Caffeine Oleate on CBB in the Field
2.6. Evaluation of the Curative Effect of Caffeine Oleate on CBB in the Field
2.7. Evaluation of the Curative Effect of Caffeine Oleate on M. velezangeli under Laboratory Conditions
2.8. Evaluation of Anatomical Changes of CBB in Contact with Caffeine Oleate by Using Scanning Electron Microscopy
3. Results
3.1. Preventive Effect of Caffeine Oleate on CBB under Laboratory Conditions
3.2. Curative Effect of Caffeine Oleate on CBB under Laboratory Conditions
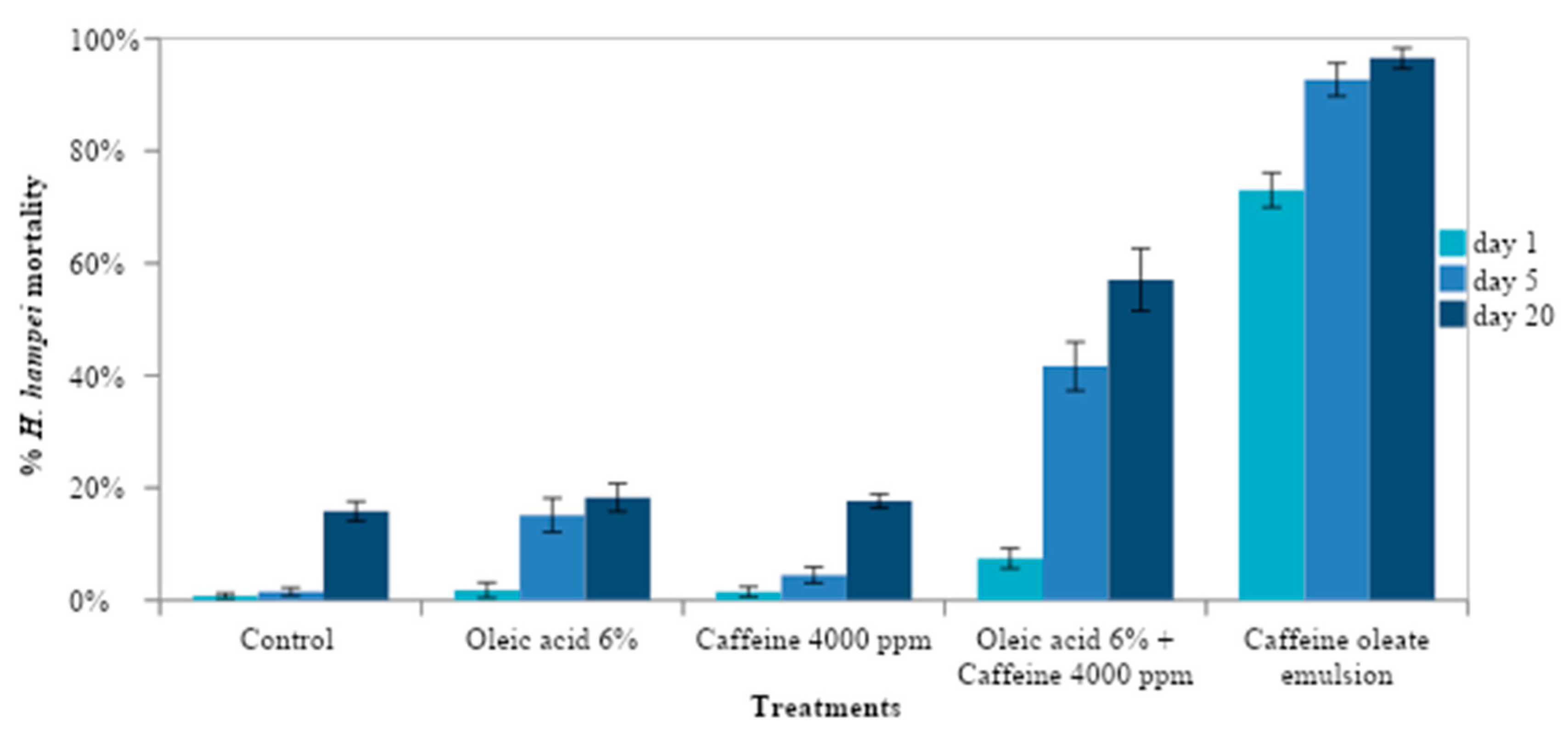
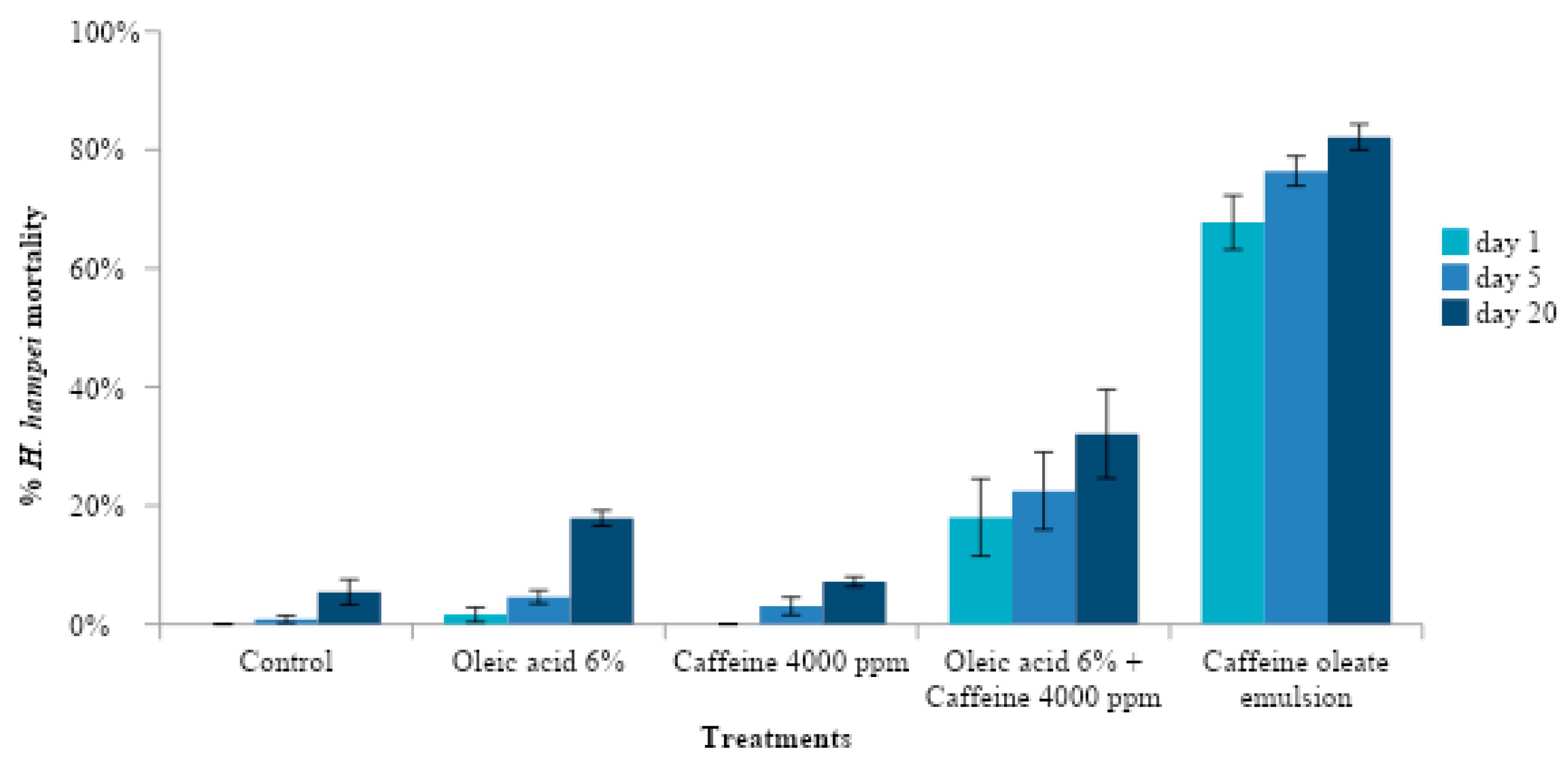
3.3. Effect of Caffeine Oleate Emulsion Components against CBB under Laboratory Conditions
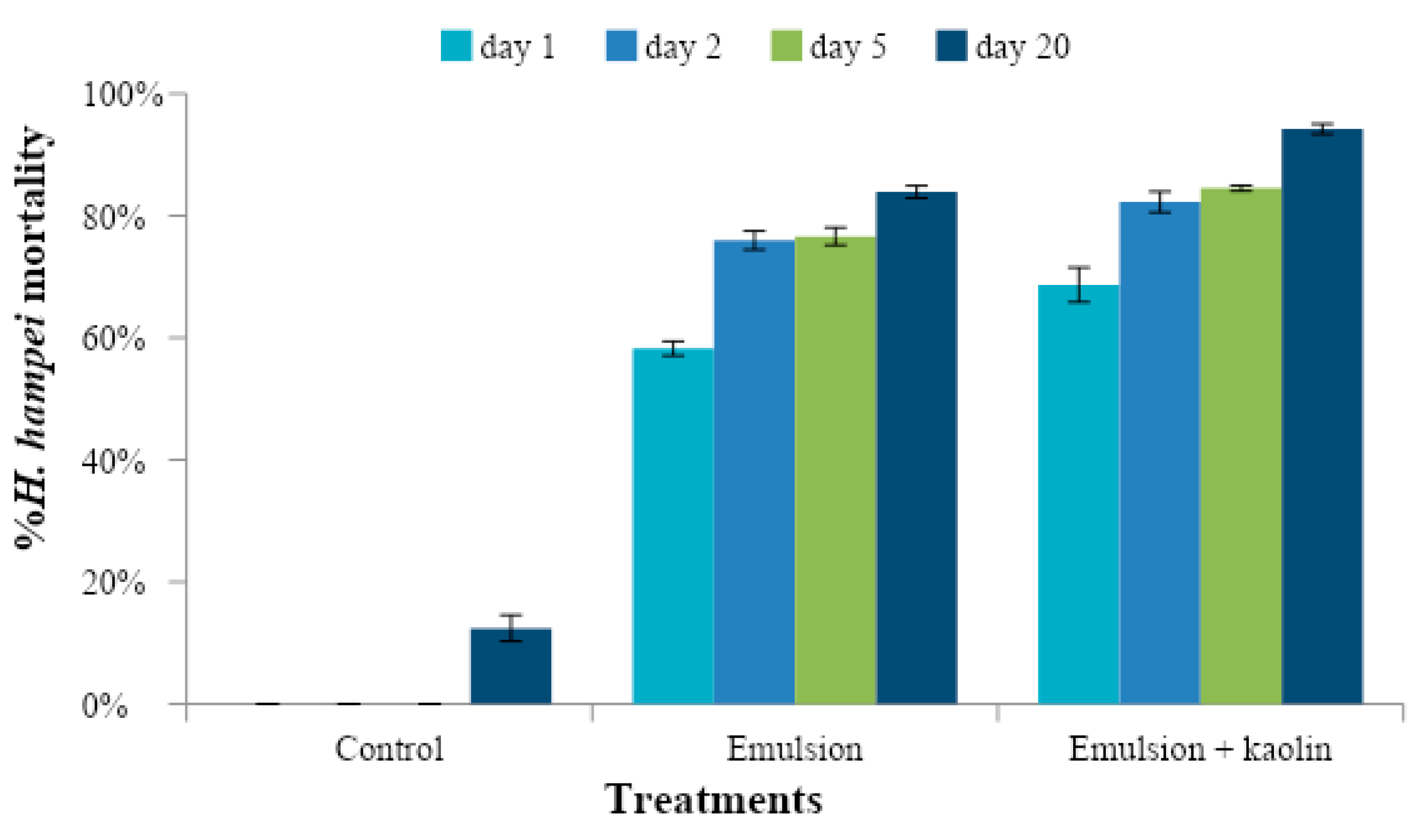
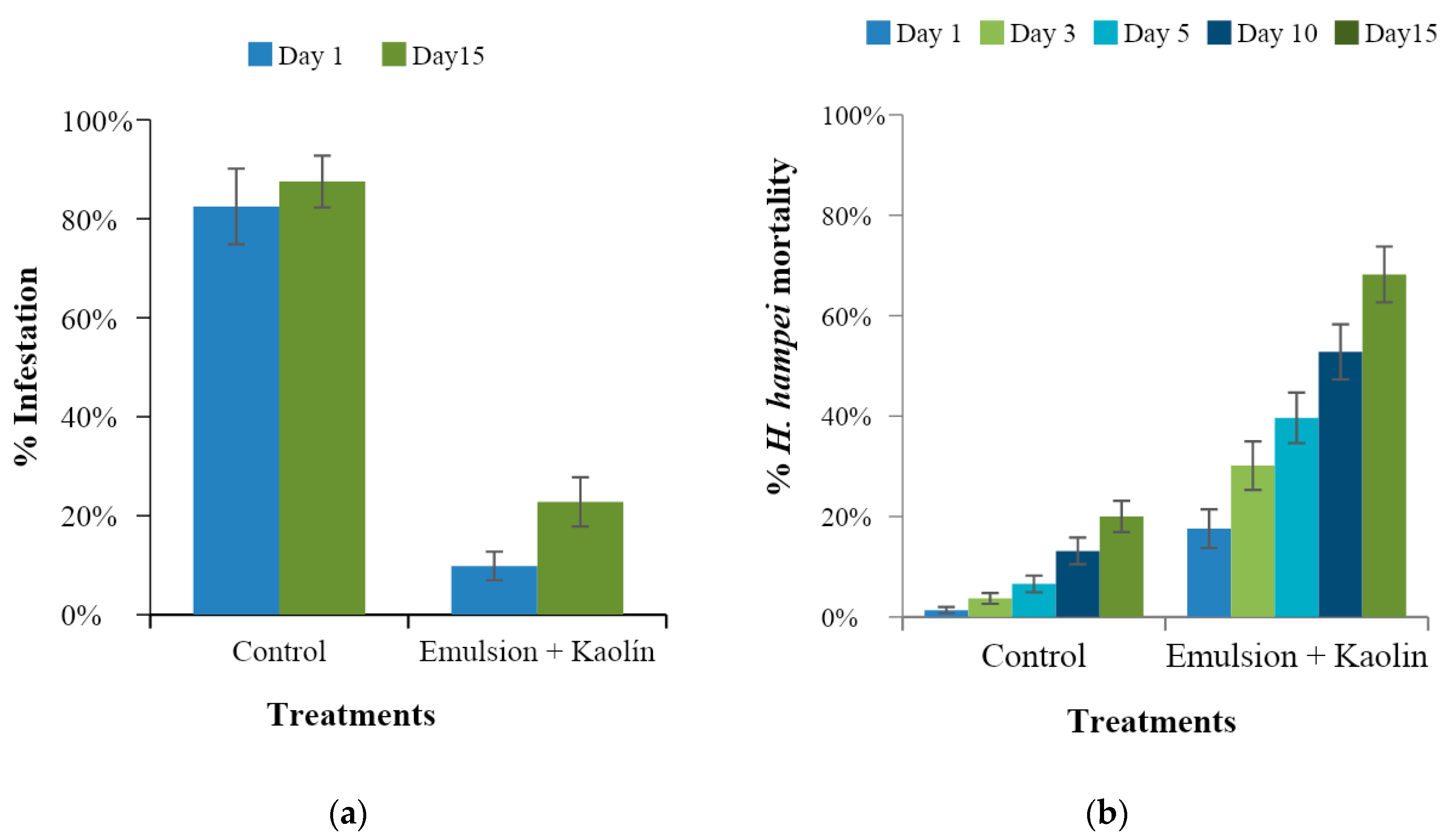
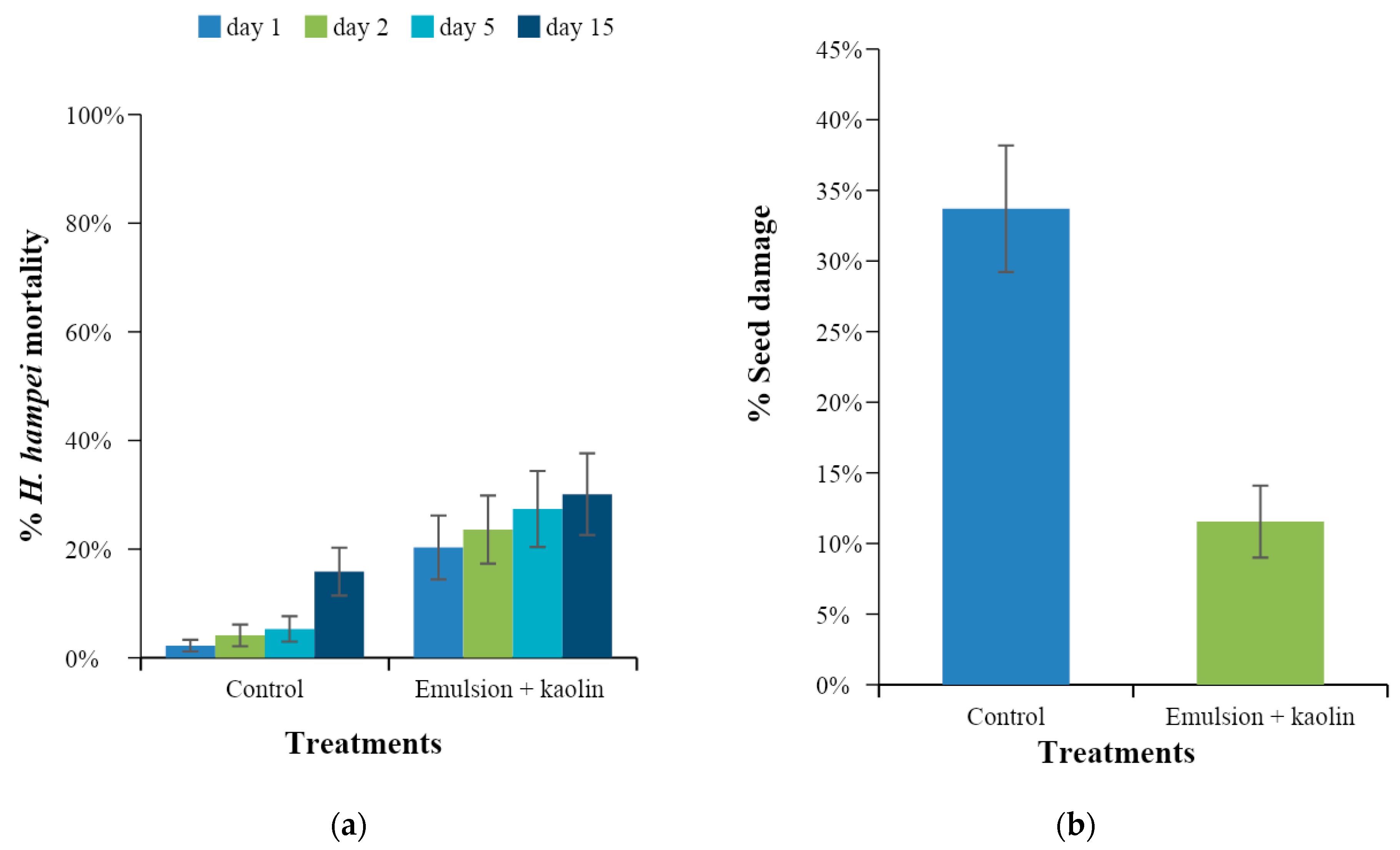
3.4. Evaluation of the Preventive Effect of Caffeine Oleate on CBB in the Field
3.5. Evaluation of the Curative Effect of Caffeine Oleate on CBB in the Field
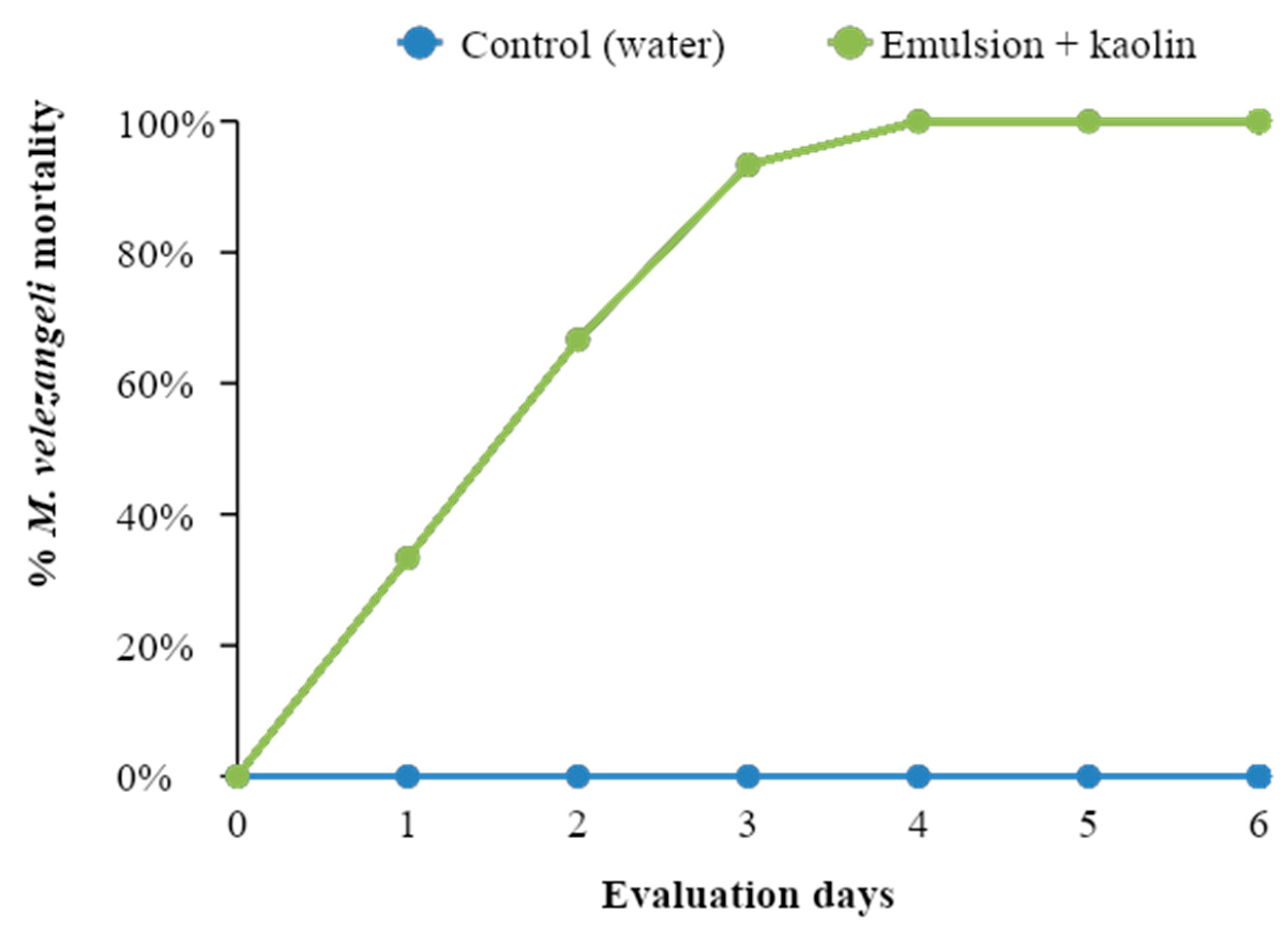
3.6. Evaluation of the Curative Effect of Caffeine Oleate on M. velezangeli under Laboratory Conditions
3.7. Evaluation of Anatomical Changes of CBB in Contact with Caffeine Oleate by Using Scanning Electron Microscopy
4. Discussion
5. Conclusions
6. Patent
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bustillo, P.A. Review of the Coffee Berry Borer, Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae), in Colombia. Rev. Colomb. Entomol. 2006, 32, 101–116. [Google Scholar] [CrossRef]
- Infante, F.; Jaramillo, J.; Castillo, A.; Vega, F. The Coffee Berry Borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae): A Short Review, with Recent Findings and Future Research Directions. Terr. Arthropod Rev. 2009, 2, 129–147. [Google Scholar] [CrossRef]
- Benavides, P.; Góngora, C.; Bustillo, A. IPM Program to Control Coffee Berry Borer Hypothenemus Hampei, with Emphasis on Highly Pathogenic Mixed Strains of Beauveria Bassiana, to Overcome Insecticide Resistance in Colombia; IntechOpen: London, UK, 2012; ISBN 978-953-307-780-2. [Google Scholar]
- Oliveira, C.M.; Auad, A.M.; Mendes, S.M.; Frizzas, M.R. Economic Impact of Exotic Insect Pests in Brazilian Agriculture. J. Appl. Entomol. 2013, 137, 1–15. [Google Scholar] [CrossRef]
- Damon, A. A Review of the Biology and Control of the Coffee Berry Borer, Hypothenemus hampei (Coleoptera: Scolytidae). Bull. Entomol. Res. 2000, 90, 453–465. [Google Scholar] [CrossRef]
- Arcila, M.A. Insecticidas Químicos Recomendados Para el Control de la Broca del Café; Brocarta No. 49; Cenicafe: Manizales, Colombia, 2016; p. 6. [Google Scholar]
- de Oliveira, C.N.; Neves, P.M.O.J.; Kawazoe, L.S. Compatibility Between the Entomopathogenic Fungus Beauveria bassiana and Insecticides Used in Coffee Plantations. Sci. Agric. 2003, 60, 663–667. [Google Scholar] [CrossRef]
- Bustillo, P.A.E.; Cardenas, M.R.; Villalba, G.D.A.; Benavides, M.P.; Orozco, H.J.; Posada, F.F.J. Manejo Integrado de la Broca del Café: Hypothenemus Hampei Ferrari en Colombia; Cenicafe: Manizales, Colombia, 1998; ISBN 978-958-96554-0-5. [Google Scholar]
- Bustillo, P.A.E. El Manejo de Cafetales y su Relación con el Control de la Broca del Café en Colombia. Coffee Plantations Management and Its Relationship with Coffee Berry Borer Control in Colombia; Cenicafe: Manizales, Colombia, 2007. [Google Scholar]
- Navarro, L.; Gongora, C.E.; Benavides, P. Single nucleotide polymorphism detection at the Hypothenemus hampei Rdl gene by allele-specific PCR amplification with Tm-shift primers. Pestic. Biochem. Physiol. 2010, 97, 204–208. [Google Scholar] [CrossRef]
- Murillo, R.C.; Posada, F.J. Los Insectos y Otros Habitantes de Cafetales y Platanales, 1st ed.; Comite Departamental de Cafeteros del Quindío: Armenia, Colombia, 2001. [Google Scholar]
- Giraldo, J.M.; Benavides, M.P.; Villegas, G.C. Aspectos Morfológicos y Biológicos de Monalonion velezangeli Carvalho and Costa Hemiptera: Miridae en Café. Cenicafé 2010, 61, 195–205. [Google Scholar]
- Torres Jaimes, L.F.; Correa Londoño, G.A.; Cartagena Valenzuela, J.R.; Monsalve García, D.A.; Londoño Zuluaga, M.E. Relationship of Monalonion velezangeli carvalho & costa (Hemiptera: Miridae) with the Phenology of Avocado (Persea americana Mill., cv. Hass). Rev. Fac. Nac. Agron. Medellín 2012, 65, 6659–6665. [Google Scholar]
- Rodas, C.A.; Serna, R.; Hurley, B.P.; Bolaños, M.D.; Granados, G.M.; Wingfield, M.J. Three New and Important Insect Pests Recorded for the First Time in Colombian Plantations. South. For. J. For. Sci. 2014, 76, 245–252. [Google Scholar] [CrossRef]
- Montilla Pérez, J.; Londoño Zuluaga, M.E.; Monsalve García, D.A.; Correa Londoño, G.A. Evaluación de Insecticidas para el Manejo de Monalonion velezangeli, Carvalho & Costa (Hemiptera: Miridae) en Aguacate. Rev. Fac. Nac. Agron. Medellín 2014, 67, 7141–7150. [Google Scholar]
- Pillmoor, J.B.; Wright, K.; Terry, A.S. Natural Products as a Source of Agrochemicals and Leads for Chemical Synthesis. Pestic. Sci. 1993, 39, 131–140. [Google Scholar] [CrossRef]
- Céspedes, C.L.; Calderón, J.S.; Lina, L.; Aranda, E. Growth inhibitory effects on fall armyworm Spodoptera frugiperda of some limonoids isolated from Cedrela spp. (Meliaceae). J. Agric. Food Chem. 2000, 48, 1903–1908. [Google Scholar] [CrossRef] [PubMed]
- Sparks, T.C.; Hahn, D.R.; Garizi, N.V. Natural products, their derivatives, mimics and synthetic equivalents: Role in agrochemical discovery. Pest Manag. Sci. 2017, 73, 700–715. [Google Scholar] [CrossRef] [PubMed]
- Lorsbach, B.A.; Sparks, T.C.; Cicchillo, R.M.; Garizi, N.V.; Hahn, D.R.; Meyer, K.G. Natural Products: A Strategic Lead Generation Approach in Crop Protection Discovery. Pest Manag. Sci. 2019, 75, 2301–2309. [Google Scholar] [CrossRef]
- Athayde, M.L.; Coelho, G.C.; Schenkel, E.P. Caffeine and Theobromine in Epicuticular Wax of Ilex Paraguariensis A. St.-Hil. Phytochemistry 2000, 55, 853–857. [Google Scholar] [CrossRef]
- Moon, S.J.; Köttgen, M.; Jiao, Y.; Xu, H.; Montell, C.A. Taste Receptor Required for the Caffeine Response In Vivo. Curr. Biol. 2006, 16, 1812–1817. [Google Scholar] [CrossRef]
- Petroski, R.J.; Stanley, D.W. Natural Compounds for Pest and Weed Control. J. Agric. Food Chem. 2009, 57, 8171–8179. [Google Scholar] [CrossRef]
- Uefuji, H.; Tatsumi, Y.; Morimoto, M.; Kaothien-Nakayama, P.; Ogita, S.; Sano, H. Caffeine Production in Tobacco Plants by Simultaneous Expression of Three Coffee N-Methyltrasferases and Its Potential as a Pest Repellant. Plant Mol. Biol. 2005, 59, 221–227. [Google Scholar] [CrossRef]
- Alshuaib, W.B.; Mathew, M.V. Caffeine Modulates Potassium Currents in Drosophila Neurons. Int. J. Dev. Neurosci. Off. J. Int. Soc. Dev. Neurosci. 2006, 24, 249–253. [Google Scholar] [CrossRef]
- Kim, Y.S.; Uefuji, H.; Ogita, S.; Sano, H. Transgenic Tobacco Plants Producing Caffeine: A Potential New Strategy for Insect Pest Control. Transgenic Res. 2006, 15, 667–672. [Google Scholar] [CrossRef]
- Apostolopoulou, A.A.; Köhn, S.; Stehle, B.; Lutz, M.; Wüst, A.; Mazija, L.; Rist, A.; Galizia, C.G.; Lüdke, A.; Thum, A.S. Caffeine Taste Signaling in Drosophila Larvae. Front. Cell. Neurosci. 2016, 10, 193. [Google Scholar] [CrossRef]
- Hall, L.M.; Ren, D.; Feng, G.; Eberl, D.F.; Dubald, M.; Yang, M.; Hannan, F.; Kousky, C.T.; Zheng, W. Calcium Channel as a New Potential Target for Insecticides. In Molecular Action of Insecticides on Ion Channels; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 1995; Volume 591, pp. 162–172. ISBN 978-0-8412-3165-8. [Google Scholar]
- Bloomquist, J.R. Ion Channels as Targets for Insecticides. Annu. Rev. Entomol. 1996, 41, 163–190. [Google Scholar] [CrossRef]
- Schmeller, T.; Wink, M. Utilization of Alkaloids in Modern Medicine. In Alkaloids; Springer: Boston, MA, USA, 1998. [Google Scholar]
- Barone, J.J.; Roberts, H.R. Caffeine Consumption. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 1996, 34, 119–129. [Google Scholar] [CrossRef]
- FDA. Spilling the Beans: How Much Caffeine Is Too Much? 2021. Available online: http://www.fda.gov/consumers/consumer-updates/spilling-beans-how-much-caffeine-too-much (accessed on 9 October 2021).
- Heckman, M.A.; Weil, J.; Gonzalez de Mejia, E. Caffeine (1,3,7-Trimethylxanthine) in Foods: A Comprehensive Review on Consumption, Functionality, Safety, and Regulatory Matters. J. Food Sci. 2010, 75, R77–R87. [Google Scholar] [CrossRef]
- Nathanson, J.A. Caffeine and Related Methylxanthines: Possible Naturally Occurring Pesticides. Science 1984, 226, 184–187. [Google Scholar] [CrossRef]
- Kumar, N.S.; Hewavitharanage, P.; Adikaram, N.K.B. Attack on Tea by Xyleborus Fornicatus: Inhibition of the Symbiote, Monacrosporium Ambrosium, by Caffeine. Phytochemistry 1995, 40, 1113–1116. [Google Scholar] [CrossRef]
- Hewavitharanage, P.; Karunaratne, S.; Kumar, N.S. Effect of Caffeine on Shot-Hole Borer Beetle (Xyleborusfornicatus) of Tea (Camellia sinensis). Phytochemistry 1999, 51, 35–41. [Google Scholar] [CrossRef]
- Aneja, M.; Gianfagna, T. Induction and Accumulation of Caffeine in Young, Actively Growing Leaves of Cocoa (Theobroma cacao L.) by Wounding or Infection with Crinipellis perniciosa. Physiol. Mol. Plant Pathol. 2001, 59, 13–16. [Google Scholar] [CrossRef]
- Hollingsworth, R.G.; Armstrong, J.W.; Campbell, E. Caffeine as a Repellent for Slugs and Snails. Nature 2002, 417, 915–916. [Google Scholar] [CrossRef] [PubMed]
- Laranja, A.T.; Manzato, A.J.; de Campos Bicudo, H.E.M. Caffeine Effect on Mortality and Oviposition in Successive Generations of Aedes Aegypti. Rev. Saude Publica 2006, 40, 1112–1117. [Google Scholar] [CrossRef]
- Russell, D.W.; Fernández-Caldas, E.; Swanson, M.C.; Seleznick, M.J.; Trudeau, W.L.; Lockey, R.F. Caffeine, a Naturally Occurring Acaricide. J. Allergy Clin. Immunol. 1991, 87, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Araque, P.C.H.; Ortiz, C.; Henao, B.; Peláez, C. Insecticidal Activity of Caffeine Aqueous Solutions and Caffeine Oleate Emulsions against Drosophila melanogaster and Hypothenemus hampei. J. Agric. Food. Chem. 2007, 55, 6918–6922. [Google Scholar] [CrossRef]
- Filho, O.G.; Mazzafera, P. Caffeine Does Not Protect Coffee Against the Leaf Miner Perileucoptera Coffeella. J. Chem. Ecol. 2000, 26, 1447–1464. [Google Scholar] [CrossRef]
- Shao, E.-S.; Lin, G.-F.; Liu, S.; Ma, X.-L.; Chen, M.-F.; Lin, L.; Wu, S.-Q.; Sha, L.; Liu, Z.-X.; Hu, X.-H.; et al. Identification of Transcripts Involved in Digestion, Detoxification and Immune Response from Transcriptome of Empoasca Vitis (Hemiptera: Cicadellidae) Nymphs. Genomics 2017, 109, 58–66. [Google Scholar] [CrossRef]
- van den Bosch, T.J.M.; Welte, C.U. Detoxifying Symbionts in Agriculturally Important Pest Insects. Microb. Biotechnol. 2017, 10, 531–540. [Google Scholar] [CrossRef]
- Guerreiro Filho, O.; Mazzafera, P. Caffeine and Resistance of Coffee to the Berry Borer Hypothenemus hampei (Coleoptera: Scolytidae). J. Agric. Food Chem. 2003, 51, 6987–6991. [Google Scholar] [CrossRef]
- Ceja-Navarro, J.A.; Vega, F.E.; Karaoz, U.; Hao, Z.; Jenkins, S.; Lim, H.C.; Kosina, P.; Infante, F.; Northen, T.R.; Brodie, E.L. Gut Microbiota Mediate Caffeine Detoxification in the Primary Insect Pest of Coffee. Nat. Commun. 2015, 6, 7618. [Google Scholar] [CrossRef]
- Glenn, D.M.; Puterka, G.J.; vanderZwet, T.; Byers, R.E.; Feldhake, C. Hydrophobic Particle Films: A New Paradigm for Suppression of Arthropod Pests and Plant Diseases. J. Econ. Entomol. 1999, 92, 759–771. [Google Scholar] [CrossRef]
- Hernández, M.B.D.; Garzo, E.I.; Duque, M.; González, P.; Castiel, A.F. Partículas de caolín: Efecto sobre la mortalidad y desarrollo de “Trichoplusia ni” Hubner. Bol. Sanid. Veg. Plagas 2002, 28, 177–184. [Google Scholar]
- Tapias, I.L.J.; Martinez, D.C.P.; Benavides, M.P.; Gongora, B.C.E. Método de laboratorio para evaluar el efecto de insecticidas sobre la broca del café. Laboratory method to evaluate the effect of insecticides on coffee berry borer. Cenicafé 2017, 68, 76–89. [Google Scholar]
- Góngora, C.E.; Tapias, J.; Martínez, C.P.; Benavides, P. Methodology to Test Control Agents and Insecticides Against the Coffee Berry Borer Hypothenemus hampei. J. Vis. Exp. Jove 2022, 181, e63694. [Google Scholar] [CrossRef]
- Luo, L.; Lane, M.E. Topical and Transdermal Delivery of Caffeine. Int. J. Pharm. 2015, 490, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Tavagnacco, L.; Brady, J.W.; Bruni, F.; Callear, S.; Ricci, M.A.; Saboungi, M.L.; Cesàro, A. Hydration of Caffeine at High Temperature by Neutron Scattering and Simulation Studies. J. Phys. Chem. 2015, 119, 13294–13301. [Google Scholar] [CrossRef]
- Shaw, P.J.; Cirelli, C.; Greenspan, R.J.; Tononi, G. Correlates of Sleep and Waking in Drosophila Melanogaster. Science 2000, 287, 1834–1837. [Google Scholar] [CrossRef]
- Trask, A.V.; Motherwell, W.D.S.; Jones, W. Pharmaceutical Cocrystallization: Engineering a Remedy for Caffeine Hydration. Cryst. Growth Des. 2005, 5, 1013–1021. [Google Scholar] [CrossRef]
- Cook, D.; Regnier, Z.R. Complexes of N -methylxanthines with carboxylic acid. Can. J. Chem. 1968, 46, 3055–3059. [Google Scholar] [CrossRef]
- Green, P.G.; Guy, R.H.; Hadgraft, J. In Vitro and In Vivo Enhancement of Skin Permeation with Oleic and Lauric Acids. Int. J. Pharm. 1988, 48, 103–111. [Google Scholar] [CrossRef]
- Cooper, E.R. Increased Skin Permeability for Lipophilic Molecules. J. Pharm. Sci. 1984, 73, 1153–1156. [Google Scholar] [CrossRef]
- Aungst, B.J.J.; Rogers, N.; Shefter, E. Enhancement of Naloxone Penetration through Human Skin in Vitro Using Fatty Acids, Fatty Alcohols, Surfactants, Sulfoxides and Amides. Int. J. Pharm. 1986, 33, 225–234. [Google Scholar] [CrossRef]
- Green, P.G.; Hadgraft, J. Facilitated Transfer of Cationic Drugs across a Lipoidal Membrane by Oleic Acid and Lauric Acid. Int. J. Pharm. 1987, 37, 251–255. [Google Scholar] [CrossRef]
- Fisone, G.; Borgkvist, A.; Usiello, A. Caffeine as a Psychomotor Stimulant: Mechanism of Action. Cell. Mol. Life Sci. CMLS 2004, 61, 857–872. [Google Scholar] [CrossRef]
- Calvo, I.A.; Gabrielli, N.; Iglesias-Baena, I.; García-Santamarina, S.; Hoe, K.-L.; Kim, D.U.; Sansó, M.; Zuin, A.; Pérez, P.; Ayté, J.; et al. Genome-Wide Screen of Genes Required for Caffeine Tolerance in Fission Yeast. PLoS ONE 2009, 4, e6619. [Google Scholar] [CrossRef]
- Abdelkader, T.S.; Chang, S.N.; Kim, T.H.; Song, J.; Kim, D.S.; Park, J.H. Exposure Time to Caffeine Affects Heartbeat and Cell Damage-Related Gene Expression of Zebrafish Danio Rerio Embryos at Early Developmental Stages. J. Appl. Toxicol. 2013, 33, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, L.; Xia, S.; Feng, C.; Guo, A. Relationship between Visual Learning/Memory Ability and Brain CAMP Level in Drosophila. Sci. China C Life Sci. 1998, 41, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Byers, D.; Davis, R.L.; Kiger, J.A. Defect in Cyclic AMP Phosphodiesterase Due to the Dunce Mutation of Learning in Drosophila Melanogaster. Nature 1981, 289, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Mustard, J.A. The Buzz on Caffeine in Invertebrates: Effects on Behavior and Molecular Mechanisms. Cell. Mol. Life Sci. 2014, 71, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Ebbinghaus-Kintscher, U.; Luemmen, P.; Lobitz, N.; Schulte, T.; Funke, C.; Fischer, R.; Masaki, T.; Yasokawa, N.; Tohnishi, M. Phthalic Acid Diamides Activate Ryanodine-Sensitive Ca2+ Release Channels in Insects. Cell Calcium 2006, 39, 21–33. [Google Scholar] [CrossRef]






| Code | Treatment | Caffeine | Oleic Acid | Kaolin |
|---|---|---|---|---|
| T1 | Control (Water) | |||
| T2 | Emulsion | 4000 μg mL−1 | 6% (p/p) | |
| T3 | Emulsion + kaolin | 4000 μg mL−1 | 6% (p/p) | 0.1% |
| Treatment | Caffeine | Oleic Acid | Tween | Kaolin |
|---|---|---|---|---|
| T1. Control (Water) | ||||
| T2. Oleic acid 6% | - | 6% (p/p) | 0.3% | - |
| T3. Caffeine 4000 μg mL−1 | 4000 μg mL−1 | 6% (p/p) | 0.3% | - |
| T4. Oleic Acid 6% + Caffeine 4000 μg mL−1 | 4000 μg mL−1 | 6% (p/p) | 0.3% | - |
| T5. Caffeine oleate emulsion (Emulsion + kaolin) | 4000 μg mL−1 | 6% (p/p) | 0.3% | 0.1% |
| Treatment | Experimental Unit | Mortality (%) | Healthy Seed (%) | ||||
|---|---|---|---|---|---|---|---|
| Average | Sd | Average | Sd | ||||
| Control (water) | 15 | 12.4 | 8.3 | 37.0 | 6.3 | ||
| Emulsion | 15 | 83.9 | *b | 3.9 | 89.0 | *b | 6.0 |
| Emulsion + kaolin | 15 | 94.2 | *a | 3.2 | 9.42 | *a | 3.7 |
| Treatment | Experimental Unit | Mortality (%) | Healthy Seed (%) | ||||
|---|---|---|---|---|---|---|---|
| Average | Sd | Average | Sd | ||||
| Control (water) | 9 | 11.1 | 3.0 | 57.3 | 3.9 | ||
| Emulsion | 9 | 46.8 | *b | 6.6 | 79.2 | *b | 8.6 |
| Emulsion + kaolin | 9 | 77.8 | *a | 3.7 | 90.0 | *a | 2.9 |
| Treatment | Experimental Unit | Mortality (%) | Healthy Seed (%) | ||||
|---|---|---|---|---|---|---|---|
| Average | Sd | Average | Sd | ||||
| T1. Control (Water) | 5 | 15.6 | c | 3.9 | 47.0 | c | 4.6 |
| T2. Oleic acid 6% | 5 | 18.3 | c | 5.6 | 53.0 | c | 5.1 |
| T3. Caffeine 4000 μg mL−1 | 5 | 17.7 | c | 2.8 | 53.7 | c | 6.8 |
| T4. Oleic Acid 6% + caffeine 4000 μg mL−1 | 5 | 57.1 | b | 2.3 | 83.7 | b | 3.2 |
| T5. Caffeine oleate emulsion (emulsion + kaolin) | 5 | 96.5 | a | 4.1 | 95.7 | a | 6.0 |
| Treatment | Experimental Unit | Mortality (%) | Healthy Seed (%) | ||||
|---|---|---|---|---|---|---|---|
| Average | Sd | Average | Sd | ||||
| T1. Control (Water) | 5 | 5.4 | c | 4.7 | 64.2 | b | 7.4 |
| T2. Oleic acid 6% | 5 | 17.9 | c | 2.3 | 66.7 | b | 5.2 |
| T3. Caffeine 4000 μg mL−1 | 5 | 7.2 | bc | 1.8 | 66.3 | b | 2.2 |
| T4. Oleic Acid 6% + caffeine 4000 μg mL−1 | 5 | 32.1 | b | 16.7 | 71.2 | b | 8.4 |
| T5. Caffeine oleate emulsion (emulsion + kaolin) | 5 | 82.2 | a | 4.8 | 91.0 | a | 3.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Góngora, C.E.; Tapias, J.; Jaramillo, J.; Medina, R.; González, S.; Restrepo, T.; Casanova, H.; Benavides, P. A Novel Caffeine Oleate Formulation as an Insecticide to Control Coffee Berry Borer, Hypothenemus hampei, and Other Coffee Pests. Agronomy 2023, 13, 1554. https://doi.org/10.3390/agronomy13061554
Góngora CE, Tapias J, Jaramillo J, Medina R, González S, Restrepo T, Casanova H, Benavides P. A Novel Caffeine Oleate Formulation as an Insecticide to Control Coffee Berry Borer, Hypothenemus hampei, and Other Coffee Pests. Agronomy. 2023; 13(6):1554. https://doi.org/10.3390/agronomy13061554
Chicago/Turabian StyleGóngora, Carmenza E., Johanna Tapias, Jorge Jaramillo, Rubén Medina, Sebastián González, Tatiana Restrepo, Herley Casanova, and Pablo Benavides. 2023. "A Novel Caffeine Oleate Formulation as an Insecticide to Control Coffee Berry Borer, Hypothenemus hampei, and Other Coffee Pests" Agronomy 13, no. 6: 1554. https://doi.org/10.3390/agronomy13061554
APA StyleGóngora, C. E., Tapias, J., Jaramillo, J., Medina, R., González, S., Restrepo, T., Casanova, H., & Benavides, P. (2023). A Novel Caffeine Oleate Formulation as an Insecticide to Control Coffee Berry Borer, Hypothenemus hampei, and Other Coffee Pests. Agronomy, 13(6), 1554. https://doi.org/10.3390/agronomy13061554







