Rhizoglomus intraradices Is More Prominent in Improving Soil Aggregate Distribution and Stability Than in Improving Plant Physiological Activities
Abstract
1. Introduction
2. Materials and Methods
2.1. AMF Strains
2.2. Plant Culture and Experimental Design
2.3. Variable Determinations
2.4. Data Analysis
3. Results
3.1. Effects on Plant Growth and Mycorrhizal Dependence
3.2. Effects on Spore Density, Root Mycorrhizal Colonization, and Soil Hyphal Length
3.3. Effects on Root Morphological Variables
3.4. Effects on Leaf Chlorophyll Index and Gas Exchange
3.5. Effects on Leaf and Root Sugar Concentrations
3.6. Effects on Soil Nutrient Levels
3.7. Effects on Soil Aggregate Distribution and Stability
3.8. Correlation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jie, W.G.; Hu, W.; Sun, G.; Yang, D.Y.; Qiao, W.; Wei, L.N. Effects of Rhizophagus intraradices on the main biomass and thiram residues of soybean at maturity stage. J. Food Saf. Qual. 2021, 12, 7592–7599. [Google Scholar]
- Zou, Y.N.; Wu, Q.S.; Kuča, K. Unravelling the role of arbuscular mycorrhizal fungi in mitigating the oxidative burst of plants under drought stress. Plant Biol. 2021, 23, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Q.; Cheng, S.; Aroca, R.; Zou, Y.N.; Wu, Q.S. Arbuscular mycorrhizal fungi induce flavonoid synthesis for mitigating oxidative damage of trifoliate orange under water stress. Environ. Exp. Bot. 2022, 204, 105089. [Google Scholar] [CrossRef]
- Sun, C.Y.; Yang, Y.S.; Zeeshan, M.; Qin, S.F.; Ma, J.Q.; Liu, L.; Yang, J.; Zhou, X.B.; Huang, J.H. Arbuscular mycorrhizal fungi reverse selenium stress in Zea mays seedlings by improving plant and soil characteristics. Ecotoxicol. Environ. Saf. 2021, 228, 113000. [Google Scholar] [CrossRef]
- Sun, Y. Role of arbuscular mycorrhizal fungi in plant ecosystems. Heilongjiang Agric. Sci. 2011, 8, 128–131. [Google Scholar]
- He, X.H.; Duan, Y.H.; Chen, Y.L.; Xu, M.G. A 60-year journey of mycorrhizal research in China: Past, present and future directions. Sci. China Life Sci. 2010, 53, 1374–1398. [Google Scholar] [CrossRef]
- Zhu, Y.; Liu, Y.X.; Gong, X.F.; Chen, Y.L.; Ren, A.T.; Liu, R.J.; Jin, L.; Xiong, Y.C. Symbiosis between three arbuscular mycorrhizal fungi and three host plants. Microbiol. China 2016, 43, 527–533. [Google Scholar]
- Zou, Y.-N.; Qin, Q.-Y.; Ma, W.-Y.; Zhou, L.-J.; Wu, Q.-S.; Xu, Y.-J.; Kuča, K.; Hashem, A.; Al-Arjani, A.-B.F.; Almutairi, K.F.; et al. Metabolomics reveals arbuscular mycorrhizal fungi-mediated tolerance of walnut to soil drought. BMC Plant Biol. 2023, 23, 118. [Google Scholar] [CrossRef]
- Zhang, C.X.; Tian, M.H.; Yang, S.; Du, J.Q.; He, T.Q.; Qiu, Y.P.; Zhang, X.L. Effects of arbuscular mycorrhizal fungi inoculant diversity on yield, phosphorus and potassium uptake of maize in acidic soil. Sci. Agric. Sin. 2022, 55, 2899–2910. [Google Scholar]
- Lü, L.H.; Zou, Y.N.; Wu, Q.S. Relationship between Arbuscular Mycorrhizas and Plant Growth: Improvement or Depression? In Root Biology; Giri, B., Prasad, R., Varma, A., Eds.; Soil Biology; Springer: Cham, Switzerland, 2018; Volume 52, pp. 451–464. [Google Scholar]
- Jifon, J.L.; Graham, J.H.; Drouillard, D.L.; Syvertsen, J.P. Growth depression of mycorrhizal Citrus seedlings grown at high phosphorus supply is mitigated by elevated CO2. New Phytol. 2002, 153, 133–142. [Google Scholar] [CrossRef]
- Ibiang, S.R.; Usami, T.; Sakamoto, K. Reduction of verticillium wilt in tomato by an arbuscular mycorrhizal fungus-Rhizophagus intraradices and an endophytic fungus-Penicillium pinophilum is cultivar dependent. Rhizosphere 2021, 20, 100440. [Google Scholar] [CrossRef]
- He, W.X.; Wu, Q.S.; Hashem, A.; Abd_Allah, E.F.; Muthuramalingam, P.; Al-Arjani, A.-B.F.; Zou, Y.N. Effects of symbiotic fungi on sugars and soil fertility and structure-mediated changes in plant growth of Vicia villosa. Agriculture 2022, 12, 1523. [Google Scholar] [CrossRef]
- Zhang, Y.T.; Wang, W.H.; Shen, H.; Guo, T. Influence of arbuscular mycorrhizal associations on the interspecific competition between mycorrhizal and non-mycorrhizal plants. Acta Ecol. Sin. 2012, 32, 1428–1435. [Google Scholar] [CrossRef]
- Yao, Q.; Feng, G.; Li, X.L. Variation between mycorrhizal dependency of different crops. Acta Agrono. Sin. 2000, 26, 874–878. [Google Scholar]
- Gavito, M.E.; Miller, M.H. Changes in mycorrhiza development in maize induced by crop management practices. Plant Soil 1998, 198, 185–192. [Google Scholar] [CrossRef]
- Sudová, R.; Vosátka, M. Differences in the effects of three arbuscular mycorrhizal fungal strains on P and Pb accumulation by maize plants. Plant Soil 2007, 296, 77–83. [Google Scholar] [CrossRef]
- Zhao, R.; Guo, W.; Bi, N.; Guo, J.; Wang, L.; Zhao, J.; Zhang, J. Arbuscular mycorrhizal fungi affect the growth, nutrient uptake and water status of maize (Zea mays L.) grown in two types of coal mine spoils under drought stress. Appl. Soil Ecol. 2015, 88, 41–49. [Google Scholar] [CrossRef]
- Ameer, I.; Kubar, K.A.; Ali, Q.; Ali, S.; Khan, T.; Shahzad, K.; Riaz, M.; Shah, Z.; Rajpar, I.; Ahmed, M.; et al. Land degradation resistance potential of a dry, semiarid region in relation to soil organic carbon stocks, carbon management index, and soil aggregate stability. Land Degrad. Dev. 2023, 34, 624–636. [Google Scholar] [CrossRef]
- Pihlap, E.; Steffens, M.; Kögel-Knabner, I. Initial soil aggregate formation and stabilisation in soils developed from calcareous loess. Geoderma 2021, 385, 114854. [Google Scholar] [CrossRef]
- Lehmann, A.; Zheng, W.; Ryo, M.; Soutschek, K.; Roy, J.; Rongstock, R.; Maaß, S.; Rillig, M.C. Fungal traits important for soil aggregation. Front. Microbiol. 2020, 10, 2904. [Google Scholar] [CrossRef]
- Baumert, V.L.; Vasilyeva, N.A.; Vladimirov, A.A.; Meier, I.C.; Kögel-Knabner, I.; Mueller, C.W. Root exudates induce soil macroaggregation facilitated by fungi in subsoil. Front. Environ. Sci. 2018, 6, 140. [Google Scholar] [CrossRef]
- Liu, R.C.; Meng, L.L.; Zou, Y.N.; He, X.H.; Wu, Q.S. Introduction of earthworms into mycorrhizosphere of white clover facilitates N storage in glomalin-related soil protein and contribution to soil total N. Appl. Soil Ecol. 2022, 179, 104597. [Google Scholar] [CrossRef]
- Meng, L.L.; Srivastava, A.K.; Kuča, K.; Wu, Q.S. Earthworm (Pheretima guillelmi)-mycorrhizal fungi (Funneliformis mosseae) association mediates rhizosphere responses in white clover. Appl. Soil Ecol. 2022, 172, 104371. [Google Scholar] [CrossRef]
- He, J.D.; Chi, G.G.; Zou, Y.N.; Shu, B.; Wu, Q.S.; Srivastava, A.K.; Kuča, K. Contribution of glomalin-related soil proteins to soil organic carbon in trifoliate orange. Appl. Soil Ecol. 2020, 154, 103592. [Google Scholar] [CrossRef]
- Lei, A.Q.; Li, Q.S.; Li, Y.; Zou, Y.N.; Wu, Q.S. Effects of symbiotic fungi on fruit quality and soil characteristics of Lane Late navel orange. J. Huazhong Agric Univ. 2022, 41, 77–83. [Google Scholar]
- Cheng, X.F.; Xie, M.M.; Li, Y.; Liu, B.Y.; Liu, C.Y.; Wu, Q.S.; Kuča, K. Effects of field inoculation with arbuscular mycorrhizal fungi and endophytic fungi on fruit quality and soil properties of Newhall navel orange. Appl. Soil Ecol. 2022, 170, 104308. [Google Scholar] [CrossRef]
- Liu, M.; Li, Y.; Mao, Z.J.; Liu, L. Effects of salt and temperature on Tagetes erecta seed germination. Pratacult. Sci. 2014, 31, 1104–1109. [Google Scholar]
- Kamran, M.; Ahmad, S.; Ahmad, I.; Hussain, I.; Meng, X.; Zhang, X.; Javed, T.; Ullah, M.; Ding, R.; Xu, P.; et al. Paclobutrazol application favors yield improvement of maize under semiarid regions by delaying leaf senescence and regulating photosynthetic capacity and antioxidant system during grain-filling Stage. Agronomy 2020, 10, 187. [Google Scholar] [CrossRef]
- Xie, M.M.; Chen, S.M.; Zou, Y.N.; Srivastava, A.K.; Rahman, M.M.; Wu, Q.S.; Kuča, K. Effects of Rhizophagus intraradices and Rhizobium trifolii on growth and N assimilation of white clover. Plant Growth Regul. 2021, 93, 311–318. [Google Scholar] [CrossRef]
- Li, Y.X.; Zhang, T.B.; Du, H.L.; Che, Q.Q. The allelopathy of five pine needle extracts on Trifolium repens L. J. Nucl. Agr. Sci. 2020, 34, 1606–1612. [Google Scholar]
- Wang, Y.S.; Zhang, S.B.; Yin, X.F.; Liu, J.B.; Wu, F.X. Isolation and identification of arbuscular mycorrhizal fungi from mainland China. Microbiol. China 2016, 43, 2154–2165. [Google Scholar]
- An, Z.Q.; Hendrix, J.W.; Hershman, D.E.; Henson, G.T. Evaluation of the “most probable number” (MPN) and wet-sieving methods for determining soil-borne populations of endogonaceous mycorrhizal fungi. Mycologia 1990, 82, 576–581. [Google Scholar] [CrossRef]
- Asghari, H.R.; Marschner, P.; Smith, S.E.; Smith, F.A. Growth response of Atriplex nummularia to inoculation with arbuscular mycorrhizal fungi at different salinity levels. Plant Soil 2005, 273, 245–256. [Google Scholar] [CrossRef]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161. [Google Scholar] [CrossRef]
- Liang, S.M.; Zhang, F.; Zou, Y.N.; Kuča, K.; Wu, Q.S. Metabolomics analysis reveals drought responses of trifoliate orange by arbuscular mycorrhizal fungi with a focus on terpenoid profile. Front. Plant Sci. 2021, 12, 740524. [Google Scholar] [CrossRef] [PubMed]
- Ames, R.N.; Bethlenfalvay, G.J. Mycorrhizal fungi and the integration of plant and soil nutrient dynamics. J. Plant Nutr. 1987, 10, 1313–1321. [Google Scholar] [CrossRef]
- Wu, Q.S.; Peng, Y.H.; Zou, Y.N.; Liu, C.Y. Exogenous polyamines affect mycorrhizal development of Glomus mosseae colonized citrus (Citrus tangerine) seedlings. ScienceAsia 2010, 36, 254–258. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Qi, R.M.; Li, J.; Lin, Z.A.; Li, Z.J.; Li, Y.T.; Yang, X.D.; Zhang, J.J.; Zhao, B.Q. Temperature effects on soil organic carbon, soil labile organic carbon fractions, and soil enzyme activities under long-term fertilization regimes. Appl. Soil Ecol. 2016, 102, 36–45. [Google Scholar] [CrossRef]
- Cheng, H.Q.; Giri, B.; Wu, Q.S.; Zou, Y.N.; Kuča, K. Arbuscular mycorrhizal fungi mitigate drought stress in citrus by modulating root microenvironment. Arch. Agron. Soil Sci. 2022, 68, 1217–1228. [Google Scholar] [CrossRef]
- Liu, L.Z.; Li, P.J.; Gong, Z.Q.; Zhang, Y.L. Effects of arbuscular mycorrhizal fungi isolated from mining area on the enhancement of Cd uptake in marigold plants. Microbiol. China 2011, 38, 575–582. [Google Scholar]
- Wang, S.Y.; Wei, H.; Chen, K.Y.; Dong, Q.; Ji, B.M.; Zhang, J. Practical methods for arbuscular mycorhizal fungal spore densty, hyphal density and colonization rate of AMF. Bio-Protoc. J. 2021, 101, e2104253. [Google Scholar]
- Du, J.Q. Effects of arbuscular mycorrhizal fungi inoculation on rhizosphere microenvironment of different Greening plants. Jiangsu Agr. Sci. 2017, 45, 149–152. [Google Scholar]
- Zhang, Y.Q. Studies on Root-Soil Systems of Several Grain Crops. Ph.D. Thesis, Shanxi Agricutural University, Taigu, China, 2005. [Google Scholar]
- Li, Y.Y.; Wang, X.J.; Dou, C.Y.; Lin, S.S.; Luo, Q.Y.; Cui, H.J.; Sun, L.; Jin, L. Effects of four host plants and different cultivation densities on the propagation of arbuscular mycorrhizal fungi. Acta Pratacult. Sin. 2013, 22, 128–135. [Google Scholar]
- Liu, R.C.; Yang, L.; Zou, Y.N.; Wu, Q.S. Root-associated endophytic fungi modulate endogenous auxin and cytokinin levels to improve plant biomass and root morphology of trifoliate orange. Hortic. Plant J. 2022. [Google Scholar] [CrossRef]
- Shao, Y.D.; Hu, X.C.; Wu, Q.S.; Yang, T.Y.; Srivastava, A.K.; Zhang, D.J.; Gao, X.B.; Kuča, K. Mycorrhizas promote P acquisition of tea plants through changes in root morphology and P transporter gene expression. South Afr. J. Bot. 2021, 137, 455–462. [Google Scholar] [CrossRef]
- Qu, M.H.; Li, S.; Yu, Y.C.; Zhang, J.C. Effects of arbuscular mycorrhizal fungi on the root morphology of Zenia insignis in karst soil habitat. Sci. Soil Water Conserv. 2021, 19, 106–114. [Google Scholar]
- Zhang, F.; Zou, Y.N.; Wu, Q.S.; Kuča, K. Arbuscular mycorrhizas modulate root polyamine metabolism to enhance drought tolerance of trifoliate orange. Environ. Exp. Bot. 2020, 171, 103962. [Google Scholar] [CrossRef]
- Frosi, G.; Barros, V.A.; Oliveira, M.T.; Santos, M.; Ramos, D.G.; Maia, L.C.; Santos, M.G. Arbuscular mycorrhizal fungi and foliar phosphorus inorganic supply alleviate salt stress effects in physiological attributes, but only arbuscular mycorrhizal fungi increase biomass in woody species of a semiarid environment. Tree Physiol. 2018, 38, 25–36. [Google Scholar] [CrossRef]
- Sonal, M.; Mahaveer, P.S.; Anjana, J. Improved photosynthetic efficacy of maize (Zea mays) plants with arbuscular mycorrhizal fungi (AMF) under high temperature stress. J. Photochem. Photobiol. B 2018, 180, 149–154. [Google Scholar]
- Chandrasekaran, M.; Chanratana, M.; Kim, K.; Seshadri, S.; Sa, T. Impact of arbuscular mycorrhizal fungi on photosynthesis, water status, and gas exchange of plants under salt stress-a meta-analysis. Front. Plant Sci. 2019, 10, 457. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, N.; Antolin, M.C.; Sánchez-Díaz, M. Gas exchange is related to the hormone balance in mycorrhizal or nitrogen-fixing alfalfa subjected to drought. Physiol. Plant. 1997, 100, 989–997. [Google Scholar] [CrossRef]
- Meng, L.L.; Liu, R.C.; Yang, L.; Zou, Y.N.; Srivastava, A.K.; Kuča, K.; Hashem, A.; Abd_Allah, E.F.; Giri, B.; Wu, Q.S. The change in fatty acids and sugars reveals the association between trifoliate orange and endophytic fungi. J. Fungi 2021, 7, 716. [Google Scholar] [CrossRef] [PubMed]
- Salmeron-Santiago, I.A.; Martínez-Trujillo, M.; Valdez-Alarcón, J.J.; Pedraza-Santos, M.E.; Santoyo, G.; Pozo, M.J.; Chávez-Bárcenas, A.T. An updated review on the modulation of carbon partitioning and allocation in arbuscular mycorrhizal plants. Microorganisms 2021, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, V.; Shamshiri, M.H.; Alaei, H.; Salehi, H. The role of inoculum identity for growth, photosynthesis, and chlorophyll fluorescence of Zinnia plants by arbuscular mycorrhizal fungi under varying water regimes. Photosynthetica 2019, 57, 409–419. [Google Scholar] [CrossRef]
- Cao, M.A.; Liu, R.C.; Xiao, Z.Y.; Hashem, A.; Abd_Allah, E.F.; Alsayed, M.F.; Harsonowati, W.; Wu, Q.S. Symbiotic fungi alter the acquisition of phosphorus in Camellia oleifera through regulating root architecture, plant phosphate transporter gene expressions and soil phosphatase activities. J. Fungi 2022, 8, 800. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.L.; Wang, K.; Wang, J. Effect of different inoculation treat-ments on AM fungal communities and the sustainability of soil remedi-ation in Daliuta coal mining subsidence area in Northwest China. Appl. Soil Ecol. 2018, 132, 107–113. [Google Scholar] [CrossRef]
- Wang, Y.J.; He, X.H.; Meng, L.L.; Zou, Y.N.; Wu, Q.S. Extraradical mycorrhizal hyphae promote soil carbon sequestration through difficultly extractable glomalin-related soil protein in response to soil water stress. Microb. Ecol. 2022. [Google Scholar] [CrossRef]
- Godbold, D.L.; Hoosbeek, M.; Lukac, M.; Cotrufo, M.F.; Janssens, I.; Ceulemans, R.J.; Polle, A.; Velthorst, E.J.; Scarascia-Mugnozza, G.; De Angelis, P.; et al. Mycorrhizal hyphal turnover as a dominant process for carbon input into soil organic matter. Plant Soil 2006, 281, 15–24. [Google Scholar] [CrossRef]
- Ren, A.T.; Mickan, B.S.; Li, J.Y.; Zhou, R.; Zhang, X.C.; Ma, M.S.; Wesly, K.; Xiong, Y.C. Soil labile organic carbon sequestration is tightly correlated with the abundance and diversity of arbuscular mycorrhizal fungi in semiarid maize fields. Land Degrad. Dev. 2021, 32, 1224–1236. [Google Scholar] [CrossRef]
- Holátko, J.; Brtnický, M.; Kučerík, J.; Kotianová, M.; Elbl, J.; Kintl, A.; Kynický, J.; Benada, O.; Datta, R.; Jansa, J. Glomalin-truths, myths, and the future of this elusive soil glycoprotein. Soil Biol. Biochem. 2021, 153, 108116. [Google Scholar] [CrossRef]
- Adam, W.G.; Richard, E.F.; Fran, L.W.; Andrew, R.S.R.; Peter, L.; Kai-Uwe, E.; Tom, Z.R.; Robert, I.R.B. Glomalin-related soil protein contains non-mycorrhizal-related heat-stable proteins, lipids and humic materials. Soil Biol. Biochem. 2011, 43, 702–717. [Google Scholar]
- Wu, Q.S.; Cao, M.Q.; Zou, Y.N.; He, X.H. Direct and indirect effects of glomalin, mycorrhizal hyphae, and roots on aggregate stability in rhizosphere of trifoliate orange. Sci. Rep. 2014, 4, 5823. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.N.; Srivastava, A.K.; Wu, Q.S.; Huang, Y.M. Glomalin-related soil protein and water relations in mycorrhizal citrus (Citrus tangerina) during soil water deficit. Arch. Agron. Soil Sci. 2014, 60, 1103–1114. [Google Scholar] [CrossRef]
- Xu, P.; Liang, L.Z.; Dong, X.Y.; Shen, R.F. Effect of arbuscular mycorrhizal fungi on aggregate stability of a clay soil inoculating with two different host plants. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2014, 65, 23–29. [Google Scholar] [CrossRef]
- Zhang, H.S.; Zhou, M.X.; Zai, X.M.; Zhao, F.G.; Qin, P. Spatio-temporal dynamics of arbuscular mycorrhizal fungi and soil organic carbon in coastal saline soil of China. Sci. Rep. 2020, 10, 9781. [Google Scholar] [CrossRef]

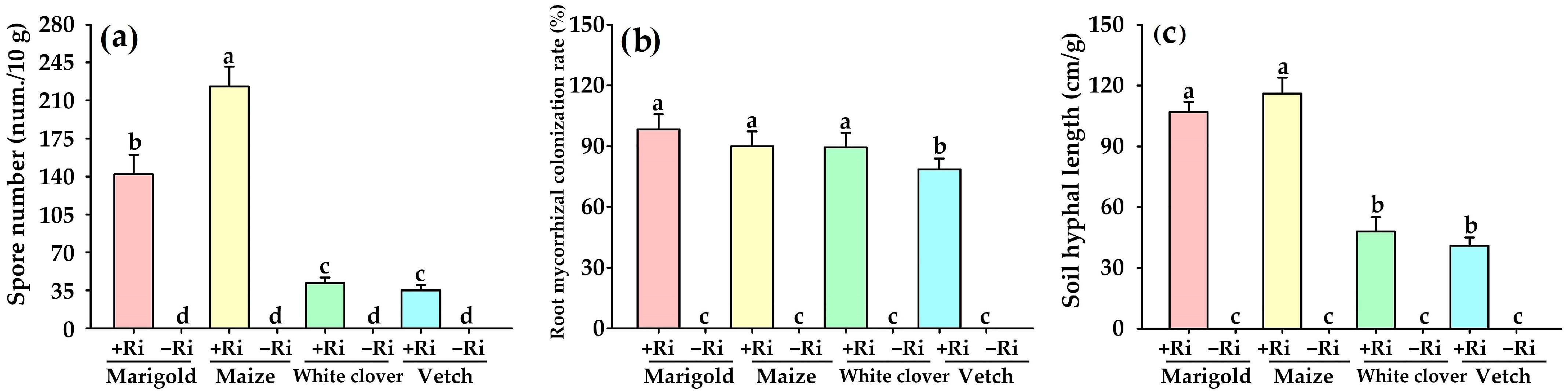
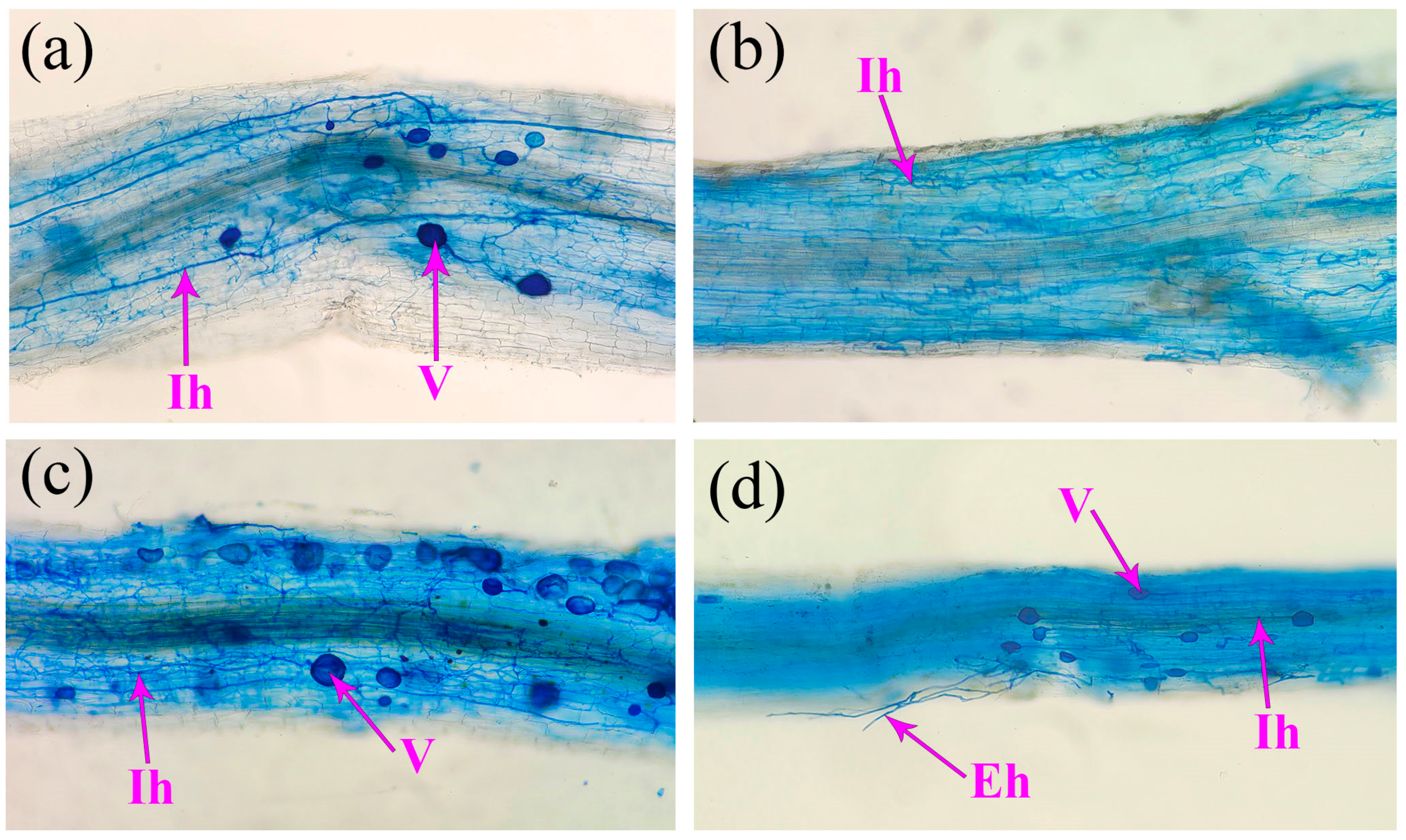
| Plants | AMF | Plant Height (cm) | Stem Diameter (mm) | Leaf Number (num./Plant) | Biomass (g/Plant) | Mycorrhizal Dependence (%) | |
|---|---|---|---|---|---|---|---|
| Shoot | Root | ||||||
| Marigold | +Ri | 35.43 ± 2.41 d | 5.04 ± 0.16 b | 15.8 ± 2.2 a | 0.331 ± 0.062 b | 1.288 ± 0.466 b | 88.87 ± 8.31 a |
| −Ri | 18.30 ± 1.46 e | 2.41 ± 0.32 c | 9.3 ± 1.3 cd | 0.185 ± 0.047 bc | 0.540 ± 0.108 c | ||
| Maize | +Ri | 48.75 ± 2.93 b | 6.13 ± 0.34 a | 10.8 ± 1.0 bc | 1.190 ± 0.322 a | 4.960 ± 0.277 a | −11.47 ± 2.88 d |
| −Ri | 59.58 ± 4.43 a | 4.72 ± 0.28 b | 11.0 ± 0.8 bc | 1.140 ± 0.151 a | 4.798 ± 0.584 a | ||
| White clover | +Ri | 17.98 ± 0.79 f | 1.04 ± 0.15 d | 8.3 ± 0.5 d | 0.047 ± 0.005 c | 0.106 ± 0.008 d | 48.19 ± 5.00 b |
| −Ri | 14.45 ± 1.02 e | 0.50 ± 0.18 e | 3.5 ± 0.6 e | 0.018 ± 0.013 c | 0.039 ± 0.011 d | ||
| Vetch | +Ri | 40.83 ± 1.12 c | 1.03 ± 0.21 d | 12.5 ± 0.6 b | 0.025 ± 0.010 c | 0.051 ± 0.009 d | 29.23 ± 6.22 c |
| −Ri | 33.45 ± 2.73 d | 0.66 ± 0.10 e | 8.0 ± 1.6 d | 0.004 ± 0.001 c | 0.045 ± 0.019 d | ||
| Significance | |||||||
| Ri | ** | ** | ** | ** | ** | ** | |
| Plants | ** | ** | ** | NS | * | ** | |
| Interaction | ** | ** | ** | NS | NS | ** | |
| Plants | AMF | Total Length (cm) | Project Area (cm2) | Surface Area (cm2) | Diameter (mm) | Volume (cm3) |
|---|---|---|---|---|---|---|
| Marigold | +Ri | 197.28 ± 16.25 b | 10.85 ± 1.65 b | 18.00 ± 1.75 b | 0.76 ± 0.10 a | 2.795 ± 1.063 b |
| −Ri | 80.97 ± 10.37 c | 6.56 ± 0.31 c | 10.37 ± 1.32 cd | 0.41 ± 0.04 e | 1.083 ± 0.215 c | |
| Maize | +Ri | 270.54 ± 6.24 a | 12.92 ± 0.45 a | 20.99 ± 0.19 a | 0.69 ± 0.09 a | 4.044 ± 0.521 a |
| −Ri | 268.93 ± 11.73 a | 12.87 ± 0.80 a | 20.92 ± 0.62 a | 0.61 ± 0.02 bc | 3.578 ± 0.710 a | |
| White clover | +Ri | 30.81 ± 9.29 d | 2.94 ± 0.84 d | 10.43 ± 0.60 cd | 0.44 ± 0.07 de | 0.230 ± 0.071 d |
| −Ri | 24.54 ± 7.69 d | 3.46 ± 0.76 d | 7.06 ± 1.43 e | 0.26 ± 0.03 f | 0.124 ± 0.018 d | |
| Vetch | +Ri | 80.16 ± 18.85 c | 6.95 ± 1.87 c | 11.18 ± 2.41 c | 0.43 ± 0.05 de | 0.335 ± 0.192 d |
| −Ri | 40.98 ± 6.76 d | 4.52 ± 0.59 d | 9.04 ± 0.43 d | 0.53 ± 0.09 cd | 0.281 ± 0.126 d | |
| Significance | ||||||
| Ri | ** | ** | ** | ** | ** | |
| Plants | ** | ** | ** | ** | ** | |
| Interaction | ** | ** | ** | ** | ** | |
| Plants | AMF | Chlorophyll Index | Net Photosynthetic Rate (μmol/m2/s) | Stomatal Conductance (μmol/m2/s) | Transpiration Rate (mmol/m2/s) |
|---|---|---|---|---|---|
| Marigold | +Ri | 18.46 ± 2.44 b | 1.80 ± 0.15 d | 0.05 ± 0.03 c | 0.97 ± 0.44 c |
| −Ri | 14.23 ± 1.89 c | 1.49 ± 0.51 d | 0.04 ± 0.01 c | 0.78 ± 0.17 c | |
| Maize | +Ri | 15.38 ± 1.84 c | 8.26 ± 0.64 b | 0.27 ± 0.05 a | 5.14 ± 0.80 a |
| −Ri | 12.78 ± 1.68 cd | 7.70 ± 1.27 bc | 0.16 ± 0.07 b | 3.57 ± 1.22 b | |
| White clover | +Ri | 30.13 ± 2.05 a | 9.73 ± 1.22 a | 0.22 ± 0.06 a | 3.80 ± 1.16 b |
| −Ri | 20.96 ± 2.81 b | 6.86 ± 0.51 c | 0.14 ± 0.04 b | 3.05 ± 0.64 b | |
| Vetch | +Ri | 13.70 ± 2.14 cd | 2.35 ± 0.25 d | 0.05 ± 0.02 c | 1.01 ± 0.39 c |
| −Ri | 10.90 ± 0.53 d | 2.13 ± 0.60 d | 0.04 ± 0.01 c | 0.99 ± 0.24 c | |
| Significance | |||||
| Ri | ** | ** | ** | ** | |
| Plants | ** | ** | ** | * | |
| Interaction | * | ** | * | NS | |
| Plants | AMF | Sucrose (mg/g) | Fructose (mg/g) | Glucose (mg/g) | |||
|---|---|---|---|---|---|---|---|
| Leaf | Root | Leaf | Root | Leaf | Root | ||
| Marigold | +Ri | 32.04 ± 1.27 d | 54.91 ± 8.08 e | 42.61 ± 9.23 b | 90.30 ± 5.45 bc | 39.79 ± 1.68 a | 16.12 ± 1.41 c |
| −Ri | 33.59 ± 5.78 d | 46.33 ± 9.07 ef | 38.18 ± 1.60 b | 79.02 ± 4.83 c | 27.17 ± 4.95 b | 12.82 ± 2.57 c | |
| Maize | +Ri | 56.11 ± 4.30 a | 141.17 ± 12.03 a | 63.30 ± 8.75 a | 124.88 ± 9.42 a | 22.19 ± 3.87 bc | 25.81 ± 3.37 a |
| −Ri | 40.25 ± 5.13 c | 101.24 ± 4.21 b | 57.90 ± 11.48 a | 101.37 ± 18.15 b | 20.51 ± 3.94 c | 21.75 ± 3.30 b | |
| White clover | +Ri | 49.46 ± 4.08 b | 62.60 ± 5.94 cd | 34.51 ± 4.28 bc | 30.50 ± 2.98 d | 22.55 ± 4.01 bc | 26.06 ± 0.70 a |
| −Ri | 58.12 ± 2.63 a | 40.27 ± 3.91 f | 27.33 ± 0.76 cd | 23.86 ± 1.66 d | 24.54 ± 5.08 bc | 23.26 ± 3.47 ab | |
| Vetch | +Ri | 59.92 ± 3.99 a | 67.60 ± 9.73 c | 35.25 ± 5.91 bc | 30.77 ± 4.14 d | 26.82 ± 5.29 bc | 15.11 ± 1.15 c |
| −Ri | 38.12 ± 5.22 cd | 45.71 ± 8.17 ef | 21.98 ± 2.38 d | 26.28 ± 2.70 d | 40.41 ± 1.07 a | 15.64 ± 2.30 c | |
| Significance | |||||||
| Ri | ** | ** | ** | ** | ** | ** | |
| Plants | ** | ** | ** | ** | NS | * | |
| Interaction | ** | ** | NS | NS | ** | NS | |
| Plants | AMF | NH4+-N (mg/kg) | NO3−-N (mg/kg) | Olsen-P (mg/kg) | Available K (mg/kg) | EE-GRSP (mg/g) | DE-GRSP (mg/g) | T-GRSP (mg/g) | SOC (mg/g) |
|---|---|---|---|---|---|---|---|---|---|
| Marigold | +Ri | 64.9 ± 5.6 a | 115.6 ± 29.5 a | 91.1 ± 15.8 d | 134.6 ± 18.7 a | 2.12 ± 0.19 abc | 5.13 ± 0.67 a | 7.24 ± 0.76 a | 45.89 ± 5.15 a |
| −Ri | 61.5 ± 3.9 a | 97.1 ± 17.5 ab | 66.3 ± 15.4 d | 79.5 ± 8.1 c | 2.06 ± 0.05 abc | 2.98 ± 1.26 c | 5.04 ± 1.26 d | 36.91 ± 3.82 bcd | |
| Maize | +Ri | 61.8 ± 2.3 a | 111.4 ± 15.7 ab | 92.6 ± 9.0 d | 92.7 ± 3.6 c | 2.36 ± 0.47 a | 4.12 ± 0.15 b | 6.49 ± 0.35 ab | 43.89 ± 6.52 ab |
| −Ri | 59.2 ± 3.3 a | 89.0 ± 14.9 b | 74.9 ± 19.5 d | 74.9 ± 10.9 c | 1.81 ± 0.03 c | 3.82 ± 0.21 b | 5.64 ± 0.21 bcd | 29.93 ± 5.15 edf | |
| White clover | +Ri | 35.4 ± 3.7 c | 52.0 ± 5.1 c | 323.0 ± 22.9 b | 50.8 ± 5.9 d | 1.94 ± 0.05 bc | 4.01 ± 0.06 b | 5.95 ± 0.08 bc | 39.90 ± 6.52 abc |
| −Ri | 35.8 ± 4.7 c | 50.4 ± 8.8 c | 269.9 ± 36.9 c | 85.3 ± 7.9 c | 2.01 ± 0.09 bc | 3.59 ± 0.07 bc | 5.61 ± 0.09 bcd | 25.94 ± 5.15 f | |
| Vetch | +Ri | 44.1 ± 7.3 b | 52.4 ± 6.6 c | 371.8 ± 34.1 a | 90.5 ± 5.9 c | 2.19 ± 0.17 ab | 4.90 ± 0.32 a | 7.09 ± 0.41 a | 28.93 ± 3.82 ef |
| −Ri | 41.9 ± 1.6 bc | 49.8 ± 2.0 c | 364.3 ± 17.6 a | 114.4 ± 17.8 b | 2.18 ± 0.03 ab | 3.34 ± 0.15 bc | 5.52 ± 0.17 cd | 34.92 ± 2.00 cde | |
| Significance | |||||||||
| Ri | ** | ** | ** | ** | NS | NS | NS | ** | |
| Plants | NS | * | NS | NS | NS | ** | ** | ** | |
| Interaction | NS | NS | * | ** | * | ** | * | ** | |
| Plants | AMF | WSA (%) | MWD (mm) | |||
|---|---|---|---|---|---|---|
| 2–4 mm | 1–2 mm | 0.5–1 mm | 0.25–0.5 mm | |||
| Marigold | +Ri | 5.00 ± 0.81 a | 4.16 ± 0.96 a | 5.41 ± 0.87 b | 16.25 ± 1.79 a | 31.41 ± 1.99 a |
| −Ri | 3.33 ± 0.60 b | 1.83 ± 0.19 b | 3.75 ± 0.41 cd | 13.00 ± 0.66 bc | 20.44 ± 1.66 c | |
| Maize | +Ri | 3.58 ± 0.50 b | 4.08 ± 0.73 a | 6.91 ± 0.31 a | 10.67 ± 0.72 cd | 26.06 ± 1.46 b |
| −Ri | 2.16 ± 0.33 c | 2.66 ± 0.60 b | 5.41 ± 0.56 b | 13.42 ± 0.68 b | 19.60 ± 1.32 cd | |
| White clover | +Ri | 4.75 ± 0.95 a | 4.08 ± 0.68 a | 6.66 ± 0.98 a | 14.00 ± 1.24 b | 30.63 ± 2.95 a |
| −Ri | 2.25 ± 0.31 c | 2.58 ± 0.31 b | 3.91 ± 0.68 c | 9.25 ± 1.10 d | 17.03 ± 1.86 d | |
| Vetch | +Ri | 4.66 ± 0.72 a | 3.91 ± 0.63 a | 6.58 ± 0.99 a | 12.08 ± 1.77 bc | 29.34 ± 2.92 a |
| −Ri | 1.33 ± 0.27 c | 2.25 ± 0.31 b | 2.75 ± 0.56 d | 12.25 ± 2.87 bc | 14.03 ± 1.43 e | |
| Significance | ||||||
| Ri | ** | NS | ** | ** | ** | |
| Plants | ** | ** | ** | * | ** | |
| Interaction | * | NS | * | ** | ** | |
| Root Colonization | Soil Hyphal Length | Soil Spore Density | SOC | ||
|---|---|---|---|---|---|
| Root colonization | 1.00 | 0.82 ** | 0.72 ** | 0.54 ** | |
| Soil NH4+-N | 0.12 | 0.35 * | 0.47 ** | 0.46 ** | |
| Soil NO3−-N | 0.25 | 0.47 ** | 0.56 ** | 0.58 ** | |
| Soil Olsen-P | −0.07 | −0.31 | −0.40 * | −0.43 * | |
| Soil available K | 0.11 | 0.31 | 0.33 | 0.28 | |
| SOC | 0.54 ** | 0.63 ** | 0.63 ** | 1.00 | |
| WSA | 2–4 mm | 0.82 ** | 0.58 ** | 0.42 * | 0.40 * |
| 1–2 mm | 0.86 ** | 0.71 ** | 0.64 ** | 0.42 * | |
| 0.5–1 mm | 0.74 ** | 0.56 ** | 0.51 ** | 0.16 | |
| 0.25–0.5 mm | 0.30 | 0.21 | 0.06 | 0.30 | |
| MWD | 0.91 ** | 0.68 ** | 0.53 ** | 0.43 * | |
| EE-GRSP | 0.26 | 0.51 ** | 0.53 ** | 0.36 * | |
| DE-GRSP | 0.67 ** | 0.57 ** | 0.44 * | 0.23 | |
| T-GRSP | 0.69 ** | 0.67 ** | 0.55 ** | 0.30 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, W.-J.; Zou, Y.-N.; Hashem, A.; Avila-Quezada, G.D.; Abd_Allah, E.F.; Wu, Q.-S. Rhizoglomus intraradices Is More Prominent in Improving Soil Aggregate Distribution and Stability Than in Improving Plant Physiological Activities. Agronomy 2023, 13, 1427. https://doi.org/10.3390/agronomy13051427
Wu W-J, Zou Y-N, Hashem A, Avila-Quezada GD, Abd_Allah EF, Wu Q-S. Rhizoglomus intraradices Is More Prominent in Improving Soil Aggregate Distribution and Stability Than in Improving Plant Physiological Activities. Agronomy. 2023; 13(5):1427. https://doi.org/10.3390/agronomy13051427
Chicago/Turabian StyleWu, Wei-Jia, Ying-Ning Zou, Abeer Hashem, Graciela Dolores Avila-Quezada, Elsayed Fathi Abd_Allah, and Qiang-Sheng Wu. 2023. "Rhizoglomus intraradices Is More Prominent in Improving Soil Aggregate Distribution and Stability Than in Improving Plant Physiological Activities" Agronomy 13, no. 5: 1427. https://doi.org/10.3390/agronomy13051427
APA StyleWu, W.-J., Zou, Y.-N., Hashem, A., Avila-Quezada, G. D., Abd_Allah, E. F., & Wu, Q.-S. (2023). Rhizoglomus intraradices Is More Prominent in Improving Soil Aggregate Distribution and Stability Than in Improving Plant Physiological Activities. Agronomy, 13(5), 1427. https://doi.org/10.3390/agronomy13051427








