Part II—Aroma Profile of Twenty White Grapevine Varieties: A Chemotaxonomic Marker Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Vegetal Material
2.2. Climatic Conditions
2.3. Must Basic Chemical Composition
2.4. Volatile Composition
2.4.1. Chemicals
2.4.2. Varietal Volatile Compounds Based on SPME-GC-MS
2.4.3. Statistical Treatment
3. Results & Discussion
3.1. Climatic Conditions
3.2. Must Basic Chemical Composition
3.3. Volatile Composition
| Variety | Ab. | Sugar (g·L−1) | Total Acidity (g·L−1) | pH | Malic Acid (g·L−1) | Tartaric Acid (g·L−1) | MI:CO | MI:BS | T:M |
|---|---|---|---|---|---|---|---|---|---|
| ‘Agudelo’ | AG | 197.6 ± 19.1 abcde | 5.4 ± 1.1 abcde | 3.48 ± 0.24 abc | 3.0 ± 0.6 cdef | 5.1 ± 1.1 b | 3.87 ± 0.86 abc | 0.98 ± 0.36 ab | 0.58 ± 0.10 efg |
| ‘Albilla do Avia’ | AA | 231.2 ± 10.6 hi | 4.9 ± 0.8 abcd | 3.45 ± 0.17 abc | 1.0 ± 0.4 ab | 6.5 ± 0.4 cdef | 4.87 ± 1 bcd | 1.37 ± 0.28 abc | 0.15 ± 0.06 ab |
| ‘Batoca’ | BA | 199.5 ± 15.1 abcdef | 6.2 ± 1.5 cdef | 3.42 ± 0.17 abc | 3.1 ± 1.7 defg | 6.0 ± 1.1 abcde | 3.47 ± 0.84 ab | 1.03 ± 0.33 abc | 0.54 ± 0.38 defg |
| ‘Branco Lexítimo’ | BL | 224.7 ± 17.5 hi | 8.1 ± 2.2 ef | 3.19 ± 0.30 a | 3.1 ± 1.0 defg | 8 ± 1.4 f | 2.99 ± 1.12 ab | 1.11 ± 0.21 abc | 0.38 ± 0.06 abcdefg |
| ‘Blanca de Monterrei’ | BM | 176.6 ± 29.0 a | 4.5 ± 0.7 abc | 3.52 ± 0.15 abc | 1.9 ± 0.9 abcde | 4.9 ± 0.9 a | 4.20 ± 0.86 abcd | 1.13 ± 0.38 abc | 0.40 ± 0.22 abcdefg |
| ‘Dona Branca’ | DB | 209.9 ± 13.4 bcdefghi | 5.1 ± 1.1 abcd | 3.58 ± 0.27 abc | 2.9 ± 0.5 cdef | 5.4 ± 1.3 abcd | 4.33 ± 0.86 abcd | 1.12 ± 0.48 abc | 0.57 ± 0.19 defg |
| ‘Fernão Pires’ | FP | 205.0 ± 14.4 bcdefgh | 6.2 ± 1.4 cdef | 3.42 ± 0.22 abc | 3.3 ± 0.9 efg | 5.9 ± 0.7 abcde | 3.45 ± 0.68 ab | 0.98 ± 0.30 ab | 0.58 ± 0.22 defg |
| ‘Garrido Fino’ | GF | 193.1 ± 3.2 abcde | 4.9 ± 0.9 abcd | 3.48 ± 0.21 abc | 1.4 ± 0.3 abc | 6.1 ± 0.8 abcde | 4.23 ± 0.86 abcd | 1.29 ± 0.40 abc | 0.24 ± 0.08 abcd |
| ‘Godello’ | GO | 217.9 ± 14.6 efghi | 5.6 ± 1.4 bcdef | 3.50 ± 0.24 abc | 2.3 ± 1.1 bcdef | 6.6 ± 0.5 def | 4.10 ± 1.17 abc | 1.24 ± 0.39 abc | 0.36 ± 0.19 abcdef |
| ‘Italia’ | IT | 201.7 ± 20.2 abcdefg | 5.2 ± 1.70 abcd | 3.62 ± 0.34 abc | 3.1 ± 1.2 def | 5.7 ± 1.1 abcde | 4.29 ± 1.74 abcd | 1.18 ± 0.59 abc | 0.57 ± 0.32 defg |
| ‘Jarrosuelto’ | JA | 189.1 ± 0.8 abcd | 6.5 ± 1.0 cdef | 3.36 ± 0.04 abc | 3.9 ± 0.9 fg | 5.5 ± 1.0 abcde | 3.07 ± 0.48 a | 0.84 ± 0.02 a | 0.71 ± 0.03 fg |
| ‘Lado’ | LA | 229.8 ± 9.6 hi | 6.9 ± 1.7 def | 3.30 ± 0.33 ab | 3.1 ± 0.6 defg | 6.7 ± 0.4 def | 3.51 ± 0.96 ab | 1.01 ± 0.22 abc | 0.46 ± 0.10 bcdefg |
| ‘Moscatel de Bago Miúdo’ | MBM | 234.1 ± 4.9 i | 5.7 ± 1.2 bcdef | 3.48 ± 0.19 abc | 2.9 ± 1.0 cdef | 5.8 ± 0.5 abcde | 4.24 ± 0.84 abcd | 1.06 ± 0.26 abc | 0.50 ± 0.21 cdefg |
| ‘Palomino’ | PA | 188.9 ± 14.4 abcd | 3.7 ± 0.7 ab | 3.62 ± 0.15 abc | 0.4 ± 0.3 a | 5.6 ± 0.7 abcde | 5.41 ± 0.67 cd | 1.57 ± 0.46 c | 0.07 ± 0.01 a |
| ‘Pirixileira’ | PI | 184.4 ± 3.5 abc | 3.4 ± 0.5 a | 3.71 ± 0.10 c | 0.8 ± 0.3 ab | 5.2 ± 0.9 abc | 5.77 ± 0.80 d | 1.56 ± 0.40 bc | 0.16 ± 0.07 abc |
| ‘Planta Fina’ | PF | 210.9 ± 17.3 cdefghi | 5.1 ± 1.5 abcd | 3.60 ± 0.35 abc | 2.2 ± 0.0 bcde | 7.0 ± 0.6 ef | 4.38 ± 1.09 abcd | 1.47 ± 0.54 abc | 0.32 ± 0.03 abcde |
| ‘Ratiño’ | RA | 211.8 ± 17.8 defghi | 7.7 ± 1.3 f | 3.33 ± 0.37 abc | 4.7 ± 2.0 g | 6.8 ± 0.9 ef | 2.82 ± 0.38 a | 0.89 ± 0.10 a | 0.72 ± 0.41 g |
| ‘Silveiriña’ | SI | 183.0 ± 23.5 ab | 5.3 ± 0.3 abcd | 3.46 ± 0.03 abc | 1.9 ± 0.6 abcde | 6.3 ± 0.6 bcde | 3.65 ± 0.31 ab | 1.20 ± 0.19 abc | 0.31 ± 0.14 abcde |
| ‘Torrontés’ | TO | 226.8 ± 12.0 fghi | 4.4 ± 0.7 abc | 3.66 ± 0.26 abc | 1.5 ± 0.3 abcd | 6.3 ± 0.5 bcde | 5.27 ± 0.80 cd | 1.46 ± 0.33 abc | 0.24 ± 0.04 abcde |
| ‘Treixadura’ | TR | 227.1 ± 12.6 ghi | 4.8 ± 1.1 abcd | 3.69 ± 0.25 bc | 2.8 ± 0.7 cdef | 5.3 ± 0.6 abcd | 4.89 ± 1.07 bcd | 1.16 ± 0.35 abc | 0.54 ± 0.21 defg |
| Significance | *** | * | ns | *** | * | * | ns | * | |
| Variety | Alcohols | Acids | Aldehydes | C6 | Thiols | Esters | Phenols |
|---|---|---|---|---|---|---|---|
| AG | 712.11 ± 224.62 ab | 514.08 ± 20.74 abc | 59.00 ± 69.85 a | 3845.38 ± 1121.59 cd | <LQ | 4.03 ± 6.98 a | 44.89 ± 27.79 f |
| AA | 490.79 ± 172.21 a | 134.53 ± 118.27 a | 57.94 ± 100.35 a | 3526.20 ± 5132.01 bcd | <LQ | 45.66 ± 58.71 ab | <LQ |
| BA | 717.24 ± 62.41 ab | 233.11 ± 173.63 ab | 35.87 ± 31.27 a | 1029.51 ± 748.36 ab | <LQ | 8.66 ± 7.61 a | 8.18 ± 14.17 abc |
| BL | 724.21 ± 144.84 ab | 628.66 ± 245.70 abc | 53.10 ± 50.97 a | 1941.89 ± 418.93 abc | 7.88 ± 8.11 ab | 64.97 ± 67.60 ab | 17.34 ± 17.79 abcd |
| BM | 743.33 ± 248.92 ab | 227.41 ± 165.33 ab | 88.33 ± 152.99 a | 1533.00 ± 344.98 abc | 3.53 ± 6.11 ab | 34.32 ± 28.18 ab | 34.01 ± 30.19 bcd |
| DB | 645.85 ± 279.15 ab | 476.24 ± 189.32 abc | 69.04 ± 35.35 a | 1302.37 ± 424.77 ab | 2.06 ± 3.57 ab | 25.22 ± 21.99 a | 32.55 ± 24.61 abcd |
| FP | 854.36 ± 190.39 ab | 223.35 ± 265.80 ab | 121.13 ± 111.07 a | 1513.36 ± 1586.81 abc | 30.51 ± 43.14 c | <LQ | 11.33 ± 8.43 abcd |
| GF | 856.00 ± 378.88 ab | 341.04 ± 215.42 abc | 46.94 ± 39.34 a | 2686.24 ± 2021.99 abcd | 20.17 ± 0.65 bc | 142.08 ± 163.96 b | 40.81 ± 39.14 cd |
| GO | 588.90 ± 68.03 ab | 673.42 ± 400.69 abc | 167.50 ± 244.82 a | 1402.35 ± 508.50 abc | 1.47 ± 2.55 ab | 17.35 ± 30.04 a | 22.70 ± 23.65 abcd |
| IT | 487.24 ± 39.64 a | 326.77 ± 250.60 abc | 28.14 ± 39.80 a | 894.69 ± 804.19 a | 5.18 ± 7.32 ab | 1.82 ± 2.58 a | 13.08 ± 11.08 abcd |
| JA | 518.45 ± 41.86 ab | 674.24 ± 625.07 abc | 89.77 ± 35.85 a | 1793.43 ± 713.65 abc | <LQ | 2.60 ± 3.67 a | 26.74 ± 13.54 abcd |
| LA | 612.69 ± 166.60 ab | 414.61 ± 230.48 abc | 84.26 ± 101.42 a | 2109.31 ± 322.15 abcd | 4.38 ± 4.77 ab | 13.63 ± 23.61 a | 5.08 ± 3.82 abc |
| MBM | 750.87 ± 377.54 ab | 318.11 ± 255.23 abc | 70.46 ± 79.10 a | 1787.66 ± 499.76 abc | 15.87 ± 27.48 abc | 17.26 ± 14.94 a | 17.68 ± 30.61 abcd |
| PA | 604.62 ± 39.64 ab | 310.10 ± 234.20 ab | 123.24 ± 117.61 a | 1848.71 ± 821.92 abc | <LQ | 47.17 ± 63.05 ab | 8.41 ± 8.08 abc |
| PI | 682.40 ± 177.29 ab | 214.59 ± 218.64 ab | 28.93 ± 43.76 a | 1433.46 ± 877.61 abc | 5.58 ± 7.64 ab | 1.64 ± 2.83 a | 17.38 ± 14.72 abcd |
| PF | 575.94 ± 83.30 ab | 926.98 ± 345.31 c | 109.40 ± 131.85 a | 1777.26 ± 907.47 abc | 2.56 ± 3.62 ab | <LQ | 23.86 ± 24.56 abcd |
| RA | 933.52 ± 512.22 b | 653.98 ± 320.71 abc | 134.13 ± 164.55 a | 4481.14 ± 879.52 d | 1.60 ± 2.77 ab | 54.75 ± 37.16 ab | 2.12 ± 3.67 ab |
| SI | 807.36 ± 103.22 ab | 765.41 ± 901.79 bc | 68.19 ± 19.34 a | 1459.19 ± 401.07 abc | 9.29 ± 3.95 ab | 124.43 ± 176.97 b | 30.20 ± 34.30 abcd |
| TO | 875.23 ± 515.68 ab | 517.17 ± 316.40 abc | 67.69 ± 50.07 a | 734.98 ± 518.34 a | 5.86 ± 8.10 ab | 1.30 ± 2.26 a | 11.01 ± 13.40 abc |
| TR | 621.25 ± 63.31 ab | 438.54 ± 441.23 abc | 50.57 ± 44.95 a | 2314.78 ± 521.47 abcd | <LQ | 24.53 ± 36.26 a | 10.02 ± 3.78 abc |
| Significance | ns | ns | ns | ns | ns | ns | ns |
| Variety | Ketones | Lactones | Terpenes | Norisoprenoids | PAHs | Sesquiterpenes | Total |
| AG | 105.75 ± 53.00 ab | 40.53 ± 10.61 abc | 17.61 ± 13.68 a | 7.96 ± 9.45 a | 82.84 ± 18.50 abc | 1.76 ± 1.70 ab | 5435.95 ± 1204.33 ab |
| AA | 61.02 ± 75.92 a | 7.95 ± 13.77 ab | 1094.80 ± 1235.11 a | 3.23 ± 5.37 a | 76.59 ± 66.77 abc | <LQ | 5498.69 ± 5481.40 ab |
| BA | 114.19 ± 59.21 ab | 18.95 ± 13.99 ab | 103.65 ± 141.33 a | 63.65 ± 92.05 b | 65.39 ± 57.35 abc | 0.39 ± 0.67 a | 2398.78 ± 761.49 a |
| BL | 100.49 ± 42.31 ab | 5.15 ± 7.16 a | 863.11 ± 568.20 a | 9.91 ± 8.71 a | 107.75 ± 56.19 bc | <LQ | 4524.45 ± 1137.93 ab |
| BM | 104.64 ± 14.85 ab | 37.18 ± 11.49 abc | 11.38 ± 4.90 a | <LQ | 79.57 ± 27.75 abc | 0.57 ± 0.98 a | 2897.28 ± 391.11 a |
| DB | 157.26 ± 11.47 ab | 80.85 ± 75.28 c | 472.50 ± 407.54 a | 7.74 ± 8.46 a | 76.25 ± 23.27 abc | 5.75 ± 9.96 b | 3353.68 ± 349.12 ab |
| FP | 96.42 ± 104.41 ab | 2.95 ± 4.17 a | 1794.81 ± 1034.80 a | 11.60 ± 0.85 ab | 81.36 ± 26.75 abc | 1.13 ± 0.08 ab | 4742.31 ± 1893.58 ab |
| GF | 193.63 ± 149.99 b | 37.87 ± 8.84 abc | 14.38 ± 9.94 a | 5.26 ± 2.61 a | 45.41 ± 41.34 ab | 0.40 ± 0.56 a | 4430.24 ± 1851.26 ab |
| GO | 128.26 ± 17.35 ab | 29.46 ± 30.03 abc | 11.71 ± 8.33 a | 10.22 ± 4.46 a | 64.31 ± 6.02 abc | 2.03 ± 2.37 ab | 3119.68 ± 388.29 a |
| IT | 107.26 ± 10.71 ab | 2.80 ± 3.97 a | 9169.02 ± 6950.02 c | 7.58 ± 5.42 a | 16.25 ± 22.98 a | <LQ | 11,059.83 ± 5863.60 d |
| JA | 132.56 ± 55.45 ab | 21.48 ± 20.69 abc | 31.94 ± 5.01 a | 21.75 ± 12.85 ab | 39.25 ± 11.90 ab | <LQ | 3352.21 ± 11.15 ab |
| LA | 77.30 ± 57.11 a | 34.96 ± 20.32 abc | 31.80 ± 12.07 a | 6.02 ± 5.48 a | 123.53 ± 47.06 c | <LQ | 3517.58 ± 691.65 ab |
| MBM | 115.92 ± 19.88 ab | 34.72 ± 60.14 abc | 6361.37 ± 2412.21 b | 9.74 ± 8.63 a | 64.38 ± 52.02 abc | <LQ | 9564.03 ± 3476.58 cd |
| PA | 143.73 ± 39.93 ab | 54.91 ± 71.21 abc | 113.44 ± 88.80 a | <LQ | 65.59 ± 13.88 abc | <LQ | 3319.93 ± 619.17 ab |
| PI | 130.08 ± 11.83 ab | 29.17 ± 2.92 abc | 3.66 ± 3.73 a | <LQ | 88.79 ± 9.07 bc | 1.03 ± 1.14 a | 2636.72 ± 868.91 a |
| PF | 70.13 ± 81.77 a | 40.57 ± 1.65 abc | 8.31 ± 4.70 a | 5.55 ± 3.13 a | 68.95 ± 20.34 abc | <LQ | 3609.51 ± 421.77 ab |
| RA | 134.22 ± 25.34 ab | 61.90 ± 21.30 abc | 16.02 ± 0.97 a | 44.68 ± 41.03 ab | 96.74 ± 41.11 bc | <LQ | 6614.81 ± 1750.04 bc |
| SI | 126.87 ± 79.65 ab | 66.00 ± 55.72 bc | 23.19 ± 10.84 a | 43.01 ± 63.36 ab | 87.16 ± 26.05 bc | 2.93 ± 2.82 ab | 3613.24 ± 1720.19 ab |
| TO | 134.79 ± 33.23 ab | 17.23 ± 19.44 ab | 31.51 ± 24.50 a | 1.12 ± 1.93 a | 91.40 ± 41.89 bc | 0.65 ± 1.12 a | 2489.95 ± 683.05 a |
| TR | 190.39 ± 114.70 b | 9.43 ± 12.56 ab | 98.26 ± 74.99 a | <LQ | 89.44 ± 40.23 bc | 1.16 ± 1.60 ab | 3848.38 ± 1054.21 ab |
| Significance | ns | ns | *** | ns | ns | ns | ** |
| Variety | Alcohols | Acids | Aldehydes | C6 | Thiols | Esters | Phenols |
|---|---|---|---|---|---|---|---|
| AG | 8441.40 ± 2843.02 a | 1109.42 ± 1165.58 ab | 226.71 ± 118.36 a | 1308.83 ± 702.60 a | 16.10 ± 27.89 a | 6748.22 ± 6855.12 a | 6128.44 ± 5124.32 ab |
| AA | 10,749.58 ± 747.10 a | 422.60 ± 731.96 ab | 507.66 ± 786.89 a | 1931.39 ± 1714.53 ab | <LQ | 2072.10 ± 2141.97 a | 1648.77 ± 805.69 a |
| BA | 11,342.79 ± 8324.25 a | 1235.82 ± 1658.38 abc | 170.31 ± 158.45 a | 2176.01 ± 1258.25 ab | 16.59 ± 17.85 a | 3241.36 ± 1802.72 a | 10,634.04 ± 9394.24 abc |
| BL | 8022.63 ± 3758.17 a | 1059.24 ± 1834.66 ab | 42.90 ± 37.24 a | 1983.61 ± 614.25 ab | 63.33 ± 94.70 ab | 3774.35 ± 4643.14 a | 40,810.41 ± 65026.25 c |
| BM | 11,640.42 ± 9353.88 a | 1620.68 ± 649.07 abc | 207.97 ± 206.72 a | 1864.39 ± 1842.87 ab | <LQ | 4599.40 ± 5907.16 a | 484.90 ± 82.02 a |
| DB | 14,588.54 ± 2141.92 a | 376.80 ± 456.55 ab | 75.57 ± 130.89 a | 2025.54 ± 462.22 ab | 37.81 ± 45.05 ab | 22,944.46 ± 35344.10 b | 5601.72 ± 8421.99 a |
| FP | 10,778.33 ± 5735.54 a | 1786.87 ± 2097.68 abc | 359.77 ± 340.73 a | 2591.51 ± 1404.25 ab | 25.94 ± 36.68 ab | 887.49 ± 387.96 a | 24,988.18 ± 33713.49 abc |
| GF | 6624.45 ± 6762.63 a | 1927.61 ± 876.97 abc | 78.88 ± 46.34 a | 4342.45 ± 5108.22 b | 44.38 ± 62.77 ab | 13,447.72 ± 18276.97 ab | 1575.67 ± 1127.86 a |
| GO | 8063.97 ± 2568.03 a | 1833.42 ± 1981.54 abc | 4338.09 ± 3760.32 c | 943.84 ± 655.62 a | 31.94 ± 38.81 ab | 1346.81 ± 1009.48 a | 6057.74 ± 6176.17 ab |
| IT | 7073.49 ± 4672.08 a | 3596.16 ± 2313.47 c | 1141.63 ± 1420.57 ab | 567.40 ± 65.59 a | <LQ | 4107.10 ± 739.46 a | 359.00 ± 365.25 a |
| JA | 5840.73 ± 2531.64 a | 2104.61 ± 2571.94 abc | 3155.19 ± 4346.15 bc | 1931.27 ± 1371.58 ab | 18.73 ± 26.49 a | 4802.03 ± 6495.65 a | 7641.54 ± 5763.32 abc |
| LA | 8879.90 ± 2452.78 a | 952.01 ± 940.20 ab | 92.70 ± 115.00 a | 2067.17 ± 859.20 ab | 12.52 ± 11.38 a | 3740.61 ± 3451.63 a | 4805.18 ± 5720.26 a |
| MBM | 10,261.84 ± 3462.48 a | 7340.23 ± 835.67 d | 4951.25 ± 2925.32 c | 524.54 ± 908.53 a | <LQ | 2618.40 ± 4150.10 a | 2079.51 ± 1537.89 a |
| PA | 10,995.72 ± 7407.55 a | 1601.96 ± 154.98 abc | 155.76 ± 269.79 a | 2793.34 ± 3349.62 ab | 30.69 ± 27.11 ab | 8293.59 ± 6317.18 ab | 10,947.46 ± 17137.58 abc |
| PI | 7509.08 ± 5062.42 a | 1096.75 ± 652.84 ab | 247.19 ± 156.24 a | 1064.89 ± 773.05 a | 22.43 ± 38.85 a | 4263.72 ± 2694.46 a | 7369.07 ± 6303.14 ab |
| PF | 14,777.19 ± 1128.92 a | 174.76 ± 76.28 ab | 8929.35 ± 1206.32 d | 1014.23 ± 40.47 a | 105.60 ± 111.19 b | 1778.93 ± 1485.39 a | 15,443.26 ± 14,250.90 abc |
| RA | 13,988.46 ± 6347.67 a | 2475.67 ± 2016.78 bc | 352.71 ± 123.41 a | 3054.76 ± 3122.61 ab | 17.70 ± 30.66 a | 4430.71 ± 1770.87 a | 37,888.25 ± 31798.50 bc |
| SI | 9876.00 ± 871.54 a | 1270.35 ± 1454.49 abc | 122.57 ± 83.60 a | 1661.30 ± 107.67 ab | 6.88 ± 11.91 a | 5084.20 ± 3042.88 a | 7392.87 ± 6484.80 ab |
| TO | 11,178.70 ± 1489.17 a | 146.29 ± 141.42 a | 470.93 ± 554.09 a | 2884.25 ± 416.57 ab | 8.01 ± 13.88 a | 1410.91 ± 1343.37 a | 3515.08 ± 3987.11 a |
| TR | 9837.80 ± 3088.79 a | 2538.82 ± 1323.60 bc | 287.90 ± 226.44 a | 1762.90 ± 727.79 ab | 43.77 ± 53.27 ab | 4664.96 ± 3167.67 a | 6134.49 ± 4369.58 ab |
| Significance | ns | *** | *** | ns | ns | ns | ns |
| Variety | Ketones | Lactones | Terpenes | Norisoprenoids | PAHs | Sesquiterpenes | Total |
| AG | 190.80 ± 11.19 a | 9.99 ± 17.30 b | 851.14 ± 523.86 a | <LQ | 25.32 ± 33.44 abc | 41.84 ± 44.18 abc | 25,098.20 ± 8941.37 a |
| AA | 214.03 ± 109.88 a | <LQ | 10,904.56 ± 6505.24 a | 4.38 ± 7.59 ab | 128.06 ± 59.77 d | <LQ | 28,583.13 ± 8273.43 a |
| BA | 311.59 ± 190.99 ab | <LQ | 1472.98 ± 998.58 a | 5.34 ± 9.25 ab | 50.11 ± 50.22 abcd | 16.92 ± 13.91 ab | 30,673.85 ± 10,940.27 a |
| BL | 352.98 ± 268.22 ab | <LQ | 8110.17 ± 5514.22 a | <LQ | 70.71 ± 61.40 abcd | 0.83 ± 1.44 a | 64,291.16 ± 70105.08 ab |
| BM | 194.60 ± 19.30 a | <LQ | 1856.28 ± 1457.11 a | <LQ | 53.27 ± 62.62 abcd | 105.98 ± 120.60 c | 22,627.89 ± 19,109.58 a |
| DB | 228.15 ± 32.74 a | <LQ | 6776.31 ± 5167.83 a | 1.77 ± 3.07 a | 94.47 ± 19.65 bcd | 5.16 ± 8.95 a | 52,756.30 ± 36,559.87 a |
| FP | 285.79 ± 31.78 ab | <LQ | 15,127.22 ± 3121.87 a | <LQ | 107.20 ± 21.19 cd | 16.00 ± 6.92 ab | 56,954.29 ± 46,100.87 ab |
| GF | 128.93 ± 137.16 a | <LQ | 1600.09 ± 188.93 a | 139.81 ± 110.57 c | 63.45 ± 78.46 abcd | 47.88 ± 59.69 abc | 30,021.32 ± 31,594.27 a |
| GO | 190.70 ± 123.66 a | <LQ | 1435.46 ± 770.39 a | 22.63 ± 24.94 ab | 39.84 ± 42.06 abc | 8.68 ± 2.96 a | 24,313.12 ± 5657.38 a |
| IT | 173.57 ± 245.46 a | <LQ | 98,597.04 ± 99149.28 b | <LQ | 69.47 ± 98.24 abcd | 42.55 ± 60.17 abc | 115,727 ± 90,768 bc |
| JA | 181.52 ± 38.40 a | 2.33 ± 3.30 ab | 2356.76 ± 1273.43 a | <LQ | 55.02 ± 24.70 abcd | 10.45 ± 5.73 ab | 28,100.20 ± 5058.39 a |
| LA | 296.82 ± 291.33 ab | 0.98 ± 1.70 a | 1105.64 ± 452.36 a | <LQ | 54.27 ± 48.32 abcd | 2.91 ± 5.04 a | 22,010.72 ± 8129.79 a |
| MBM | 493.99 ± 196.86 ab | <LQ | 104834 ± 25440 b | <LQ | 97.91 ± 90.39 cd | 16.23 ± 28.12 ab | 13,3218 ± 25158 c |
| PA | 194.09 ± 148.05 a | <LQ | 4132.31 ± 2525.20 a | 6.68 ± 9.08 ab | 64.11 ± 51.98 abcd | 6.75 ± 6.46 a | 39,222.46 ± 22,417.89 a |
| PI | 234.56 ± 51.25 a | <LQ | 355.10 ± 158.75 a | <LQ | 37.80 ± 26.50 abc | 1.44 ± 2.49 a | 22,202.03 ± 9653.19 a |
| PF | 134.13 ± 44.75 a | <LQ | 873.30 ± 785.71 a | 46.05 ± 65.12 ab | 7.30 ± 10.32 ab | 33.71 ± 42.18 abc | 43,317.80 ± 14,879.73 a |
| RA | 249.97 ± 128.68 a | <LQ | 1081.71 ± 471.70 a | 51.46 ± 59.29 b | 98.04 ± 30.70 cd | 81.18 ± 98.43 bc | 63,770.63 ± 27,038.85 ab |
| SI | 192.19 ± 64.02 a | <LQ | 1052.09 ± 259.98 a | 30.92 ± 35.27 ab | 2.62 ± 4.54 a | 8.75 ± 8.24 a | 26,700.74 ± 2939.59 a |
| TO | 379.17 ± 328.66 ab | <LQ | 8603.15 ± 13,645.97 a | <LQ | 57.88 ± 6.36 abcd | 18.35 ± 16.62 ab | 28,672.74 ± 17,653.21 a |
| TR | 710.18 ± 762.90 b | <LQ | 2544.15 ± 1016.31 a | <LQ | 36.56 ± 35.14 abc | 2.73 ± 2.37 a | 28,564.26 ± 2535.54 a |
| Significance | ns | ns | *** | ** | ns | ns | ** |
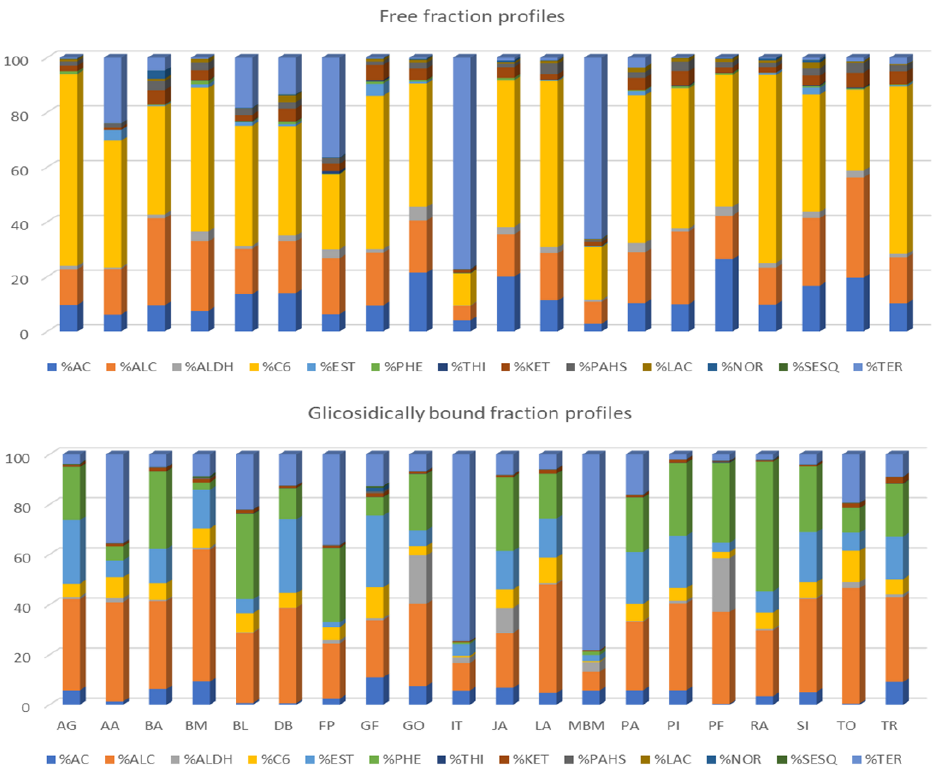
3.3.1. Free Volatile Fraction
3.3.2. Glycosidically Bound Volatile Fraction
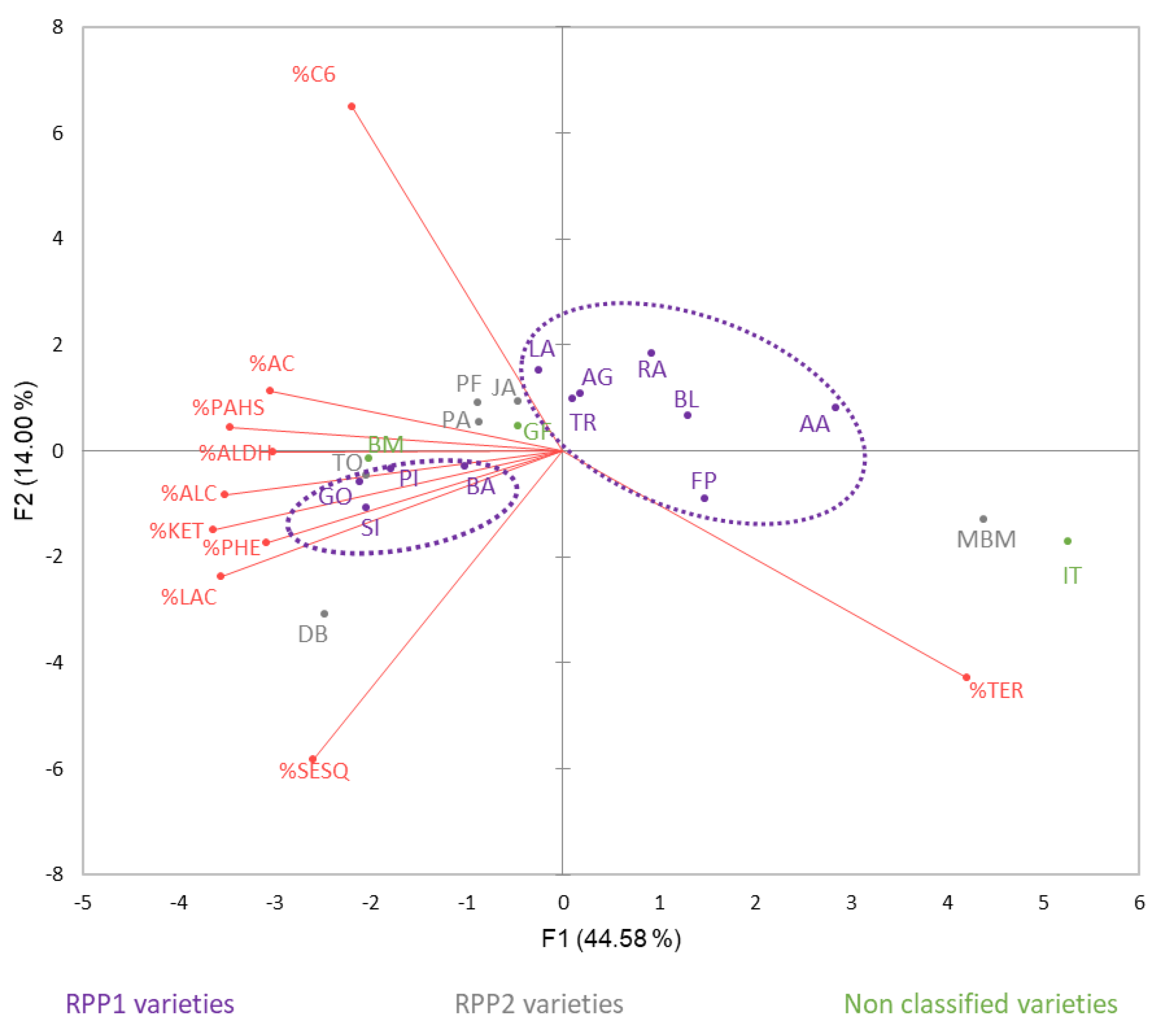
3.3.3. Variety Classification: Aromatic Relationship PCAs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Resco, P.; Iglesias, A.; Bardaji, I.; Sotés, V. Exploring adaptation choices for grapevine regions in Spain. Reg. Environ. Chang. 2016, 16, 979–993. [Google Scholar] [CrossRef]
- Vilanova, M.; Genisheva, Z.; Masa, A.; Oliveira, J.M. Correlation between volatile composition and sensory properties in Spanish Albariño wines. Microchem. J. 2010, 95, 240–246. [Google Scholar] [CrossRef]
- Vilanova, M.; Campo, E.; Escudero, A.; Graña, M.; Masa, A.; Cacho, J. Volatile composition and sensory properties of Vitis vinifera red cultivars from Northwest Spain: Correlation between sensory and instrumental analysis. Anal. Chim. Acta 2012, 720, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Vilanova, M.; Oliveira, J.M.; Rivas, R.; Alonso, J.C.; Martínez-Zapater, J.M.; Ibáñez, J.; Cacho, J. Primera parte: El sector vitivinícola gallego en el siglo XXI. El triunfo de las variedades de cultivo tradicional. In El Potencial Aromático de las Variedades de Vid Cultivadas en Galicia; Xunta de Galicia-Consellería de Medio Rural: Santiago de Compostela, Spain, 2017; Available online: https://libraria.xunta.gal/sites/default/files/downloads/publicacion/libro_vid_i_castellano.pdf (accessed on 10 December 2022).
- Rocha, S.M.; Coutinho, P.; Coelho, E.; Barros, A.S.; Delgadillo, I.; Coimbra, M.A. Relationships between the varietal volatile composition of the musts and white wine aroma quality. A four-year feasibility study. LWT -Food Sci. Technol. 2010, 43, 1508–1516. [Google Scholar] [CrossRef]
- Genisheva, Z.; Oliveira, J.M. Monoterpenic characterization of white cultivars from Vinhos Verdes appellation of origin (North Portugal). J. Inst. Brew. 2009, 115, 308–317. [Google Scholar] [CrossRef]
- Vineyard Enquises. Ministerio de Agricultura, Pesca y Alimentación. (MAPA). Available online: https://www.mapa.gob.es/es/estadistica/temas/estadisticas-agrarias/agricultura/encuestas-de-vinedo/ (accessed on 2 November 2022).
- Estratexia de Dinamización Económica, Territorial e Turística das Comarcas Vitivinícolas de Galicia. Available online: https://mediorural.xunta.gal/sites/default/files/publicacions/portada/ESTRATEXIA-DINAMIZACION_V9_1_sep.pdf (accessed on 2 November 2022).
- Vierra, G. Pretenders at the Table—Are table wines no longer food friendly? Wine Bus. Mon. 2004, 11, 17–21. [Google Scholar]
- Antolín, M.C.; Toledo, M.; Pascual, I.; Irigoyen, J.J.; Goicoechea, N. The Exploitation of Local Vitis vinifera L. Biodiversity as a Valuable Tool to Cope with Climate Change Maintaining Berry Quality. Plants 2021, 10, 71. [Google Scholar] [CrossRef]
- Carbonell-Bejerano, P.; de Carvalho, L.C.; Eiras Dias, J.E.; Martinez-Zapater, J.M.; Amancio, S. Chapter 15: Exploiting Vitis genetic diversity to manage with stress. In Grapevine in a Changing Environment: A Molecular and Ecophysiological Perspective; Gerós, H., Chaves, M.M., Gil, H.M., Delrot, S., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; pp. 347–380. [Google Scholar] [CrossRef]
- Lacombe, T. Contribution á L’étude de L’histoire Evolutive de la Vigne Cultivée (Vitis vinifera L.) par L’analyse de la Diversité Génétique Neuter et de Genes D’intérêt, Centre International d’Etudes Supérieures en Sciences Agronomiques, Montpellier SupAgro, 18 December 2012. Available online: https://www.supagro.fr/theses/extranet/12-0040_Lacombe.pdf (accessed on 1 November 2022).
- Wolkovich, E.M.; de Cortázar-Atauri, I.G.; Morales-Castilla, I.; Nicholas, K.A.; Lacombe, T. From Pinot to Xinomavro in the world’s future wine-growing regions. Nat. Clim. Chang. 2018, 8, 29–37. [Google Scholar] [CrossRef]
- García-Muñoz, S.; Muñoz-Organero, G.; Fernández-Fernández, E.; Cabello, F. Sensory characterisation and factors influencing quality of wines made from 18 minor varieties (Vitis vinifera L.). Food Qual. Prefer. 2014, 32, 241–252. [Google Scholar] [CrossRef]
- Álvarez, M.G.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gándara, J. Relationships between Godello white wine sensory properties and its aromatic fingerprinting obtained by GC–MS. Food Chem. 2011, 129, 890–898. [Google Scholar] [CrossRef]
- Duchêne, E.; Huard, F.; Dumas, V.; Scheneider, C.; Merdinoglu, D. The challenge of adapting grapevine varieties to climate change. Clim. Res. 2010, 41, 193–204. [Google Scholar] [CrossRef]
- Duchêne, E.; Butterlin, G.; Dumas, V.; Merdinoglu, D. Towards the adaptation of grapevine varieties to climate change: QTLs and candidate genes for developmental stages. Theor. Appl. Genet. 2012, 124, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Belliveau, S.; Smit, B.; Bradshaw, B. Multiple exposures, and dynamic vulnerability: Evidence from the grape industry in the Okanagan Valley, Canada. Glob. Environ. Chang. 2006, 16, 364–378. [Google Scholar] [CrossRef]
- Petronilho, S.; Lopez, R.; Ferreira, V.; Coimbra, M.A.; Rocha, S.M. Revealing the Usefulness of Aroma Networks to Explain Wine Aroma Properties: A Case Study of Portuguese Wines. Molecules 2020, 25, 272. [Google Scholar] [CrossRef]
- Vilanova, M.; Escudero, A.; Graña, M.; Cacho, J. Volatile composition and sensory properties of North West Spain white wines. Food Res. Int. 2013, 54, 562–568. [Google Scholar] [CrossRef]
- Wason, E.; Hunter, M. Genetic variation in plant volatile emission does not result in differential attraction of natural enemies in the field. Oecologia 2014, 174, 479–491. [Google Scholar] [CrossRef]
- Yang, S.; Meng, Z.; Fan, J.; Yan, L.; Yang, Y.; Zhao, Z. Evaluation of the volatile profiles in pulp of 85 apple cultivars (Malus domestica) by HS–SPME combined with GC–MS. J. Food Meas. Charact. 2021, 15, 4215–4225. [Google Scholar] [CrossRef]
- Bouzas-Cid, Y.; Trigo-Córdoba, E.; Falqué, E.; Orriols, I.; Mirás-Avalos, J.M. Influence of supplementary irrigation on the amino acid and volatile composition of Godello wines from the Ribeiro Designation of Origin. Food Res. Int. 2018, 111, 715–723. [Google Scholar] [CrossRef]
- Alem, H.; Rigou, P.; Schneider, R.; Ojeda, H.; Torregrosa, L. Impact of agronomic practices on grape aroma composition: A review. J. Sci. Food Agric. 2019, 99, 975–985. [Google Scholar] [CrossRef]
- Coletta, A.; Toci, A.T.; Pati, S.; Ferrara, G.; Grieco, F.; Tufariello, M.; Crupi, P. Effect of Soil Management and Training System on Negroamaro Wine Aroma. Foods 2021, 10, 454. [Google Scholar] [CrossRef]
- Šikuten, I.; Štambuk, P.; Tomaz, I.; Marchal, C.; Kontić, J.K.; Lacombe, T.; Maletić, E.; Preiner, D. Discrimination of genetic and geographical groups of grape varieties (Vitis vinifera L.) based on their volatile organic compounds. Front. Plant Sci. 2022, 13, 942148. [Google Scholar] [CrossRef] [PubMed]
- Chitarrini, G.; Lazazzara, V.; Lubes, G.; Agnolet, S.; Valls, J.; von Lutz, H.; Brunner, K.; Lozano, L.; Guerra, W.; Ciesa, F.; et al. Volatile profiles of 47 monovarietal cloudy apple juices from commercial, old, red-fleshed and scab-resistant apple cultivars. Eur. Food Res. Technol. 2021, 247, 2739–2749. [Google Scholar] [CrossRef]
- Beghè, D.; Cirlini, M.; Beneventi, E.; Miroslav, Č.; Tatjana, P.; Ganino, T.; Petruccelli, R.; Dall’Asta, C. Volatile profile of Italian and Montenegrine pomegranate juices for geographical origin classification. Eur. Food Res. Technol. 2021, 247, 211–220. [Google Scholar] [CrossRef]
- Laucou, V.; Launay, A.; Bacilieri, R.; Lacombe, T.; Adam-Blondon, A.F.; Berard, A.; Chauveau, A.; de Andrés, M.T.; Hausmann, L.; Ibáñez, J.; et al. Extended diversity analysis of cultivated grapevine Vitis vinifera with 10 K genome-wide SNPs. PLoS ONE 2018, 13, e0192540. [Google Scholar] [CrossRef] [PubMed]
- Cunha, J.; Ibáñez, J.; Teixeira-Santos, M.; Brazão, J.; Fevereiro, P.; Martínez-Zapater, J.M.; Eiras-Dias, J.E. Genetic Relationships Among Portuguese Cultivated and Wild Vitis vinifera L. Germplasm. Front. Plant Sci. 2020, 11, 127. [Google Scholar] [CrossRef]
- Díaz-Losada, E.; Salgado, A.T.; Ramos-Cabrer, A.M.; Río Segade, S.; Diéguez, S.C.; Pereira-Lorenzo, S. Twenty microsatellites (SSRs) reveal two main origins of variability in grapevine cultivars from Northwestern Spain. Vitis 2010, 49, 55–62. [Google Scholar] [CrossRef]
- Díaz-Losada, E.; Salgado, A.T.; Ramos-Cabrer, A.M.; Pereira-Lorenzo, S. Determination of genetic relationships of Albariño and Loureira cultivars with the Caíño group by microsatellites. Am. J. Enol. Viticult. 2011, 62, 3. [Google Scholar] [CrossRef]
- Díaz-Losada, E.; Fernández, I.O.; Martínez, F.R.; Salgado, A.T.; Cabrer, A.R.; Lorenzo, S.P. A Colección de Vides da Estación de Viticultura e Enoloxía de Galicia; Xunta de Galicia-Consellería do Medio Rural: Santiago de Compostela, Spain. 2011. Available online: https://libraria.xunta.gal/sites/default/files/documents/11-0247_0.pdf (accessed on 15 December 2022).
- Díaz-Losada, E.; Salgado, A.T.; Ramos-Cabrer, A.M.; Díaz-Hernández, B.; Pereira-Lorenzo, S. Genetic and geographical structure in grapevines from northwestern Spain. Ann. Appl. Biol. 2012, 161, 24–35. [Google Scholar] [CrossRef]
- Díaz-Losada, E.; Cortés-Diéguez, S.; Rodríguez-Torres, I.; Mirás-Avalos, J.M.; Orriols-Fernández, I.; Pereira-Lorenzo, S. Characterization of the nearly extinct ‘Albilla’ cultivar from Galicia and its relationships with other Spanish ‘Albillos’. J. Int. Sci. Vigne Vin. 2013, 47, 261–268. [Google Scholar] [CrossRef]
- Díaz-Fernández, Á.; Díaz-Losada, E.; Cortés-Diéguez, S. Approach to the Chemotaxonomic Characterization of Traditional Cultivation Grape Varieties through Their Varietal Aroma Profile. Foods 2022, 11, 1427. [Google Scholar] [CrossRef]
- Díaz-Fernández, Á.; Díaz-Losada, E.; Cortés-Diéguez, S. Diversity among Traditional Minority Red Grape Varieties According to Their Aromatic Profile. Agronomy 2022, 12, 1799. [Google Scholar] [CrossRef]
- Díaz-Fernández, Á.; Díaz-Losada, E.; Moreno, D.; Valdés Sánchez, M.E. Anthocyanin profile of Galician endangered varieties. A tool for varietal selection. Food Res. Int. 2022, 154, 110983. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Fernández, Á.; Díaz-Losada, E.; Vázquez-Arias, A.; Pujol, A.P.; Cardona, D.M.; Valdés-Sánchez, M.E. Non-Anthocyanin Compounds in Minority Red Grapevine Varieties Traditionally Cultivated in Galicia (Northwest Iberian Peninsula), Analysis of Flavanols, Flavonols, and Phenolic Acids. Plants 2023, 12, 4. [Google Scholar] [CrossRef]
- Ferreira, V.; Lopez, R. The Actual and Potential Aroma of Winemaking Grapes. Biomolecules 2019, 9, 818. [Google Scholar] [CrossRef] [PubMed]
- Vitis International Variety Catalogue VIVC. Available online: https://www.vivc.de/ (accessed on 5 November 2022).
- Torres, R.; Ferrara, G.; Soto, F.; López, J.A.; Sanchez, F.; Mazzeo, A.; Pérez-Pastor, A.; Domingo, R. Effects of soil and climate in a table grape vineyard with cover crops. Irrigation management using sensors networks. Ciênc. Téc. Vitiviníc. 2017, 32, 72–81. [Google Scholar] [CrossRef]
- Huglin, P. Nouveau mode d’évaluation des possibilités héliothermiques d’en milieu viticole. Comptes. Rendus. Acad. Agric. Fr. 1978, 64, 1117–1126. [Google Scholar]
- Tonietto, J.; Carbonneau, A. A multicriteria climatic classification system for grape growing regions worldwide. Agric. For. Meteolo. 2004, 124, 81–97. [Google Scholar] [CrossRef]
- Togores, J.H. Chapter 3: Vendimia. Recepción de Uva en la Bodega. In Tomo I: Tratado de Enología, 2nd ed.; Mundi-Prensa: Madrid, Spain, 2010. [Google Scholar]
- Bazán, D.L.; del Río, P.G.; Domínguez, J.M.; Cortés-Diéguez, S.; Mejuto, J.C.; Pérez-Guerra, N. The Chemical, Microbiological and Volatile Composition of Kefir-Like Beverages Produced from Red Table Grape Juice in Repeated 24-h Fed-Batch Subcultures. Foods 2022, 11, 3117. [Google Scholar] [CrossRef]
- Masa, A.; Vilanova, M. Flavonoid and aromatic characterisation of cv. Albarín blanco (Vitis vinifera L.). Food Chem. 2008, 107, 273–281. [Google Scholar] [CrossRef]
- Pedroza, M.A.; Zalacain, A.; Lara, J.F.; Salinas, M.R. Global grape aroma potential and its individual analysis by SBSE-GC-MS. Food Res. Int. 2010, 43, 1003–1008. [Google Scholar] [CrossRef]
- Marais, J. Terpenes in the Aroma of Grapes and Wines: A Review. S. Afr. J. Enol. Vitic. 1983, 4, 49–60. [Google Scholar] [CrossRef]
- Yu, F.; Utsumi, R. Diversity, regulation, and genetic manipulation of plant mono- and sesquiterpenoid biosynthesis. Cell Mol. Life Sci. 2009, 66, 3043–3052. [Google Scholar] [CrossRef] [PubMed]
- Conde, C.; Silva, P.; Fontes, N.; Dias, A.; Tavares, R.; Sousa, M.; Agasse, A.; Delrot, S.; Gerós, H. Biochemical changes throughout Grape Berry development and fruit and wine quality. Food 2007, 1, 1–22. Available online: https://www.researchgate.net/publication/228555202_Biochemical_changes_throughout_Grape_Berry_development_and_fruit_and_wine_quality (accessed on 5 December 2022).
- Zellner, B.D.A.; Casilli, A.; Dugo, P.; Dugo, G.; Mondello, L. Odour fingerprint acquisition by means of comprehensive two-dimensional gas chromatography-olfactometry and comprehensive two-dimensional gas chromatography/mass spectrometry. J. Chromatogr. A 2007, 1141, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Arciola, C.R.; Barbieri, R.; Silva, A.S.; Nabavi, S.F.; Sokeng, A.J.T.; Izadi, M.; Jafari, N.J.; Suntar, I.; Daglia, M.; et al. Update on monoterpenes as antimicrobial agents: A particular focus on p-Cymene. Materials 2017, 10, 947. [Google Scholar] [CrossRef] [PubMed]
- Flavornet. Available online: https://www.flavornet.org/flavornet.html (accessed on 12 December 2022).
- May, B.; Wust, M. Sesquiterpene profiles of different grape varieties. In Advances and Challenges in Flavor Chemistry & Biology; Hofmann, T., Meyerhof, W., Schieberle, P., Eds.; Deutsche Forschungsanstalt für Lebensmittelchemie: Freising, Germany, 2011; pp. 323–327. [Google Scholar]
- Petronilho, S.; Coimbra, M.A.; Rocha, S.M. A critical review on extraction techniques and gas chromatography-based determination of grapevine derived sesquiterpene. Anal. Chim. Acta 2014, 846, 8–35. [Google Scholar] [CrossRef]
- Kalua, C.M.; Boss, P.K. Comparison of major volatile compounds from Riesling and Cabernet Sauvignon grapes (Vitis vinifera L.) from fruitset to harvest. Aust. J. Grape Wine Res. 2010, 16, 337–348. [Google Scholar] [CrossRef]
- Todorova, I.T.; Batovska, D.I.; Parushev, S.P.; Djakova, G.J.; Popov, S.S. Surface metabolic composition of berries collected from 13 Bulgarian grapevine plants. Nat. Prod. Res. 2010, 24, 935–941. [Google Scholar] [CrossRef]
- Pineau, B.; Barbe, J.C.; Van Leeuwen, C.; Dubordieu, D. Which impact for β-damascenone on red wines aroma? J. Agric. Food Chem. 2007, 55, 4103–4108. [Google Scholar] [CrossRef]
- Tomasino, E.; Bolman, S. The potential effect of β-Ionone and β-Damascenone on sensory perception of Pinot Noir wine aroma. Molecules 2021, 26, 1288. [Google Scholar] [CrossRef]
- González-Barreiro, C.; Rial-Otero, R.; Cancho-Grande, B.; Simal-Gándara, J. Wine aroma compounds in grapes: A critical review. Crit. Rev. Food Sci. Nutr. 2015, 55, 202–218. [Google Scholar] [CrossRef]
- Slaghenaufi, D.; Peruch, E.; De Cosmi, M.; Nouvelet, L.; Ugliano, M. Volatile and phenolic composition of monovarietal red wines of Valpolicella appellations. OENO One 2021, 55, 279–294. [Google Scholar] [CrossRef]
- Oliveira, J.M.; Faria, M.; Sá, F.; Barros, F.; Araújo, I.M. C6-alcohols as varietal markers for assessment of wine origin. Anal. Chim. Acta 2006, 563, 300–309. [Google Scholar] [CrossRef]
- López-Tamames, E.; Carro-Mariño, N.; Gunata, Y.Z.; Sapis, C.; Baumes, R.; Bayonove, C. Potential aroma in several varieties of Spanish grapes. J. Agric. Food Chem. 1997, 45, 1729–1735. [Google Scholar] [CrossRef]
- Canosa, P.; Oliveira, J.M.; Masa, A.; Vilanova, M. Study of the Volatile and Glycosidically Bound Compounds of Minority Vitis vinifera Red Cultivars from NW Spain. J. Inst. Brew. 2011, 117, 462–471. [Google Scholar] [CrossRef]
- The Good Scent Company. Available online: https://www.thegoodscentcompany.com (accessed on 5 January 2023).
- Yu, Q.; Xie, B.J.; Zhang, Y.; Zhou, H.Y.; Pan, S.Y. Study on Aroma Components in Fruit from Three Different Satsuma Mandarin Varieties. Agric. Sci. China 2007, 6, 1487–1493. [Google Scholar] [CrossRef]
- IARC Working Group of the Evaluation of Carcinogenic Risks to Humans. Some Chemicals Present in Industrial and Consumer Products, Food and Drinking-Water; International Agency for Research on Cancer: Lyon, France, 2013. Available online: https://www.ncbi.nlm.nih.gov/books/NBK373195/ (accessed on 10 January 2023).
- Sasaki, K.; Takase, H.; Tanzawa, F.; Kobayashi, H.; Saito, H.; Matsuo, H.; Takata, R. Identification of Furaneol Glucopyranoside, the Precursor of Strawberry-like Aroma, Furaneol, in Muscat Bailey A. Am. J. Enol. Vitic. 2014, 66, 91–94. [Google Scholar] [CrossRef]
- Cerreti, M.; Esti, M.; Benucci, I.; Liburdi, K.; de Simone, C.; Ferranti, P. Thiol precursors in grape. Aust. J. Grape Wine Res. 2015, 21, 411–416. [Google Scholar] [CrossRef]
- Mariano, A.P.; Ramos, A.L.; de Oliveira Júnior, A.H.; García, Y.M.; de Paula, A.C.; Silva, M.R.; Augusti, R.; de Araújo, R.L.; Melo, J.O. Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp. Molecules 2022, 27, 935. [Google Scholar] [CrossRef]
- Chen, X.Y.; Dou, Y.X.; Luo, D.D.; Zhang, Z.B.; Li, C.L.; Zeng, H.F.; Su, Z.R.; Xie, J.H.; Lai, X.P.; Li, Y.C. β-Patchoulene from patchouli oil protects against LPS-induced acute lung injury via suppressing NF-κB and activating Nrf2 pathways. Int. Immunopharmacol. 2017, 50, 270–278. [Google Scholar] [CrossRef]
- Welke, J.E.; Manfroi, V.; Zanus, M.; Lazarotto, M.; Zini, C.A. Characterization of the volatile profile of Brazilian Merlot wines through comprehensive two-dimensional gas chromatography time-of-flight mass spectrometric detection. J. Chromatogr. A 2012, 1226, 124–139. [Google Scholar] [CrossRef]
- Burdock, G.A. Fenaroli’s Handbook of Flavour Ingredients, 6th ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2010. [Google Scholar]
- Weyerstahl, P.; Meisel, T.; Mewes, K.; Negahdari, S. Struktur und Geruch, XIII. Synthese und olfaktorische Eigenschaften von Megastigmatrienon-Analoga. Liebigs Ann. Der Chem. 1991, 1, 19–25. [Google Scholar] [CrossRef]
- García-Carpintero, E.G.; Sánchez-Palomo, E.; Gallego, M.A.G.; González-Viñas, M.A. Volatile and sensory characterization of red wines from cv. Moravia Agria minority grape variety cultivated in La Mancha region over five consecutive vintages. Food Res. Int. 2011, 44, 1549–1560. [Google Scholar] [CrossRef]
- Peinado, R.A.; Moreno, J.; Bueno, J.E.; Moreno, J.A.; Mauricio, J.C. Comparative study of aromatic compounds in two young white wines subjected to prefermentative cryomaceration. Food Chem. 2004, 84, 585–590. [Google Scholar] [CrossRef]
- Poitou, X.; Redon, P.; Pons, A.; Bruez, E.; Delière, L.; Marchal, A.; Cholet, C.; Geny-Denis, L.; Darriet, P. Methyl salicylate, a grape and wine chemical marker and sensory contributor in wines elaborated from grapes affected or not by cryptogrammic diseases. Food Chem. 2021, 360, 130120. [Google Scholar] [CrossRef] [PubMed]
- Culbert, J.A.; Jiang, W.; Ristic, R.; Puglisi, C.J.; Nixon, E.C.; Shi, H.; Wilkinson, K.L. Glycosylation of Volatile Phenols in Grapes following Pre-Harvest (On-Vine) vs. Post-Harvest (Off-Vine) Exposure to Smoke. Molecules 2021, 26, 5277. [Google Scholar] [CrossRef] [PubMed]
- Vararu, F.; Moreno-Garcia, J.; Cotea, V.V.; Moreno, J. Grape musts differentiation based on selected aroma compounds using SBSE-GC-MS and statistical analysis. Vitis 2015, 54, 97–105. [Google Scholar] [CrossRef]
- Pérez-Navarro, J.; Izquierdo-Cañas, P.M.; Mena-Morales, A.; Chacón-Vozediano, J.L.; Martínez-Gascueña, J.; García-Romero, E.; Hermosín-Gutiérrez, I.; Gómez-Alonso, S. Comprehensive Chemical and Sensory Assessment of Wines Made from White Grapes of Vitis vinifera Cultivars Albillo Dorado and Montonera del Casar: A Comparative Study with Airén. Foods 2020, 9, 1282. [Google Scholar] [CrossRef]
- Dimitrovska, M.; Bocevska, M.; Dimitrovski, D.; Murkovic, M. Anthocyanin composition of Vranec, Cabernet Sauvignon, Merlot and Pinot Noir grapes as indicator of their varietal differentiation. Eur. Food Res. Technol. 2011, 232, 591–600. [Google Scholar] [CrossRef]
- Mattivi, F.; Guzzon, F.; Vrhowsek, U.; Stefanini, M.; Velasco, R. Metabolite profiling of grape: Flavonols and anthocyanins. J. Agric. Food Chem. 2006, 54, 7692–7702. [Google Scholar] [CrossRef]
- Pomar, F.; Novo, M.; Masa, A. Varietal differences among the anthocyanin profiles of 50 red table grapes cultivars studied by high performance liquid chromatography. J. Chromatogr. A 2005, 1094, 34–41. [Google Scholar] [CrossRef]
- Yang, C.; Wang, Y.; Liang, Z.; Fan, P.; Wu, B.; Yang, L.; Wang, Y.; Li, S. Volatiles of grape berries evaluated at the germplasm level by headspace-SPME with GC–MS. Food Chem. 2009, 114, 1106–1114. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Fernández, Á.; Díaz-Losada, E.; Domínguez, J.M.; Cortés-Diéguez, S. Part II—Aroma Profile of Twenty White Grapevine Varieties: A Chemotaxonomic Marker Approach. Agronomy 2023, 13, 1168. https://doi.org/10.3390/agronomy13041168
Díaz-Fernández Á, Díaz-Losada E, Domínguez JM, Cortés-Diéguez S. Part II—Aroma Profile of Twenty White Grapevine Varieties: A Chemotaxonomic Marker Approach. Agronomy. 2023; 13(4):1168. https://doi.org/10.3390/agronomy13041168
Chicago/Turabian StyleDíaz-Fernández, Ángela, Emilia Díaz-Losada, José Manuel Domínguez, and Sandra Cortés-Diéguez. 2023. "Part II—Aroma Profile of Twenty White Grapevine Varieties: A Chemotaxonomic Marker Approach" Agronomy 13, no. 4: 1168. https://doi.org/10.3390/agronomy13041168
APA StyleDíaz-Fernández, Á., Díaz-Losada, E., Domínguez, J. M., & Cortés-Diéguez, S. (2023). Part II—Aroma Profile of Twenty White Grapevine Varieties: A Chemotaxonomic Marker Approach. Agronomy, 13(4), 1168. https://doi.org/10.3390/agronomy13041168






