Effects of Photoperiod and Drought on Flowering and Growth Development of Protein-Rich Legumes under Atlantic Environments
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Sites
2.2. Data Collection
2.3. Data Analysis
3. Results
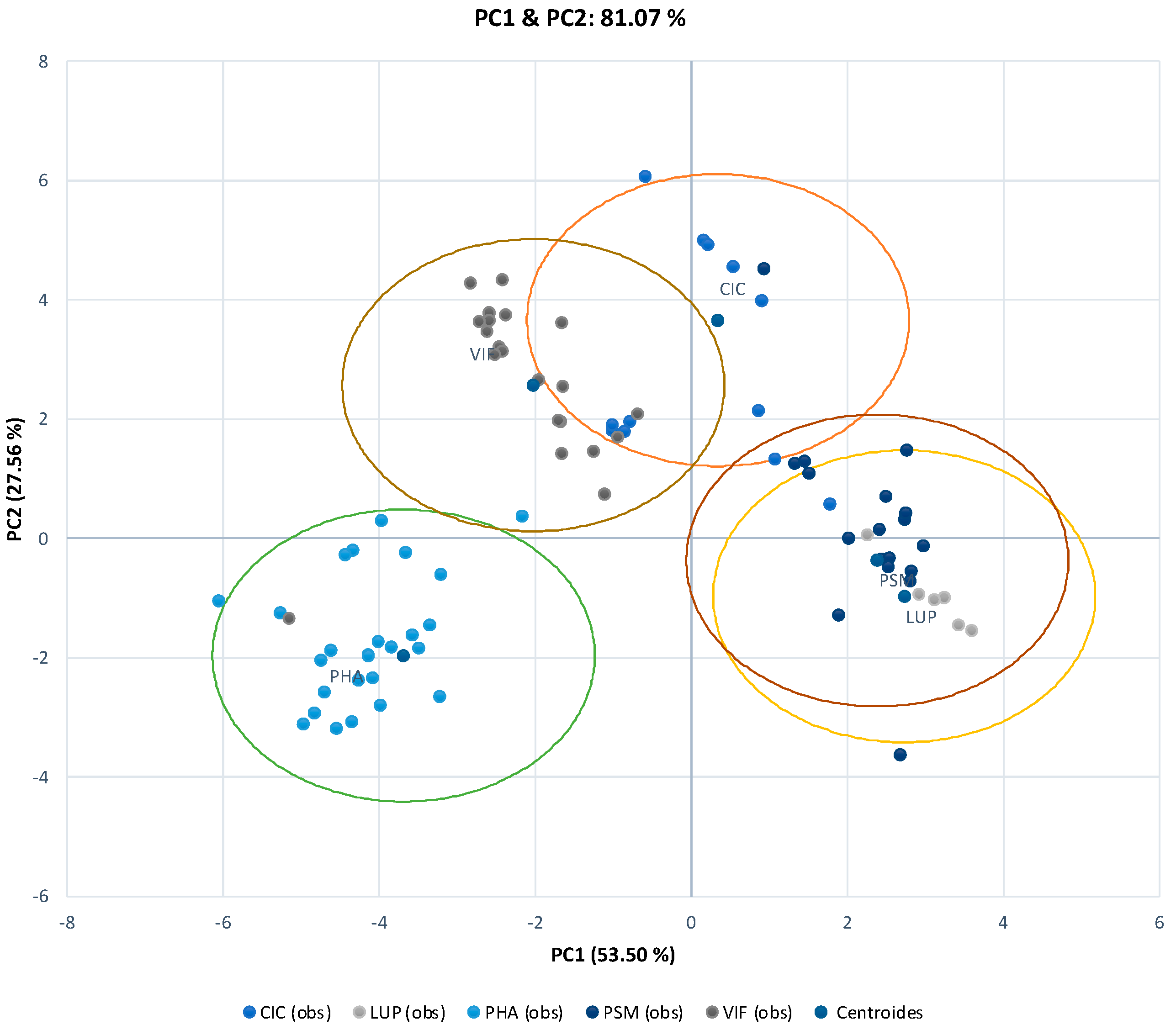
3.1. Comparison of Photoperiod Adaptation Processes of Different Legume Species Based on Seed Yield and Flowering Time
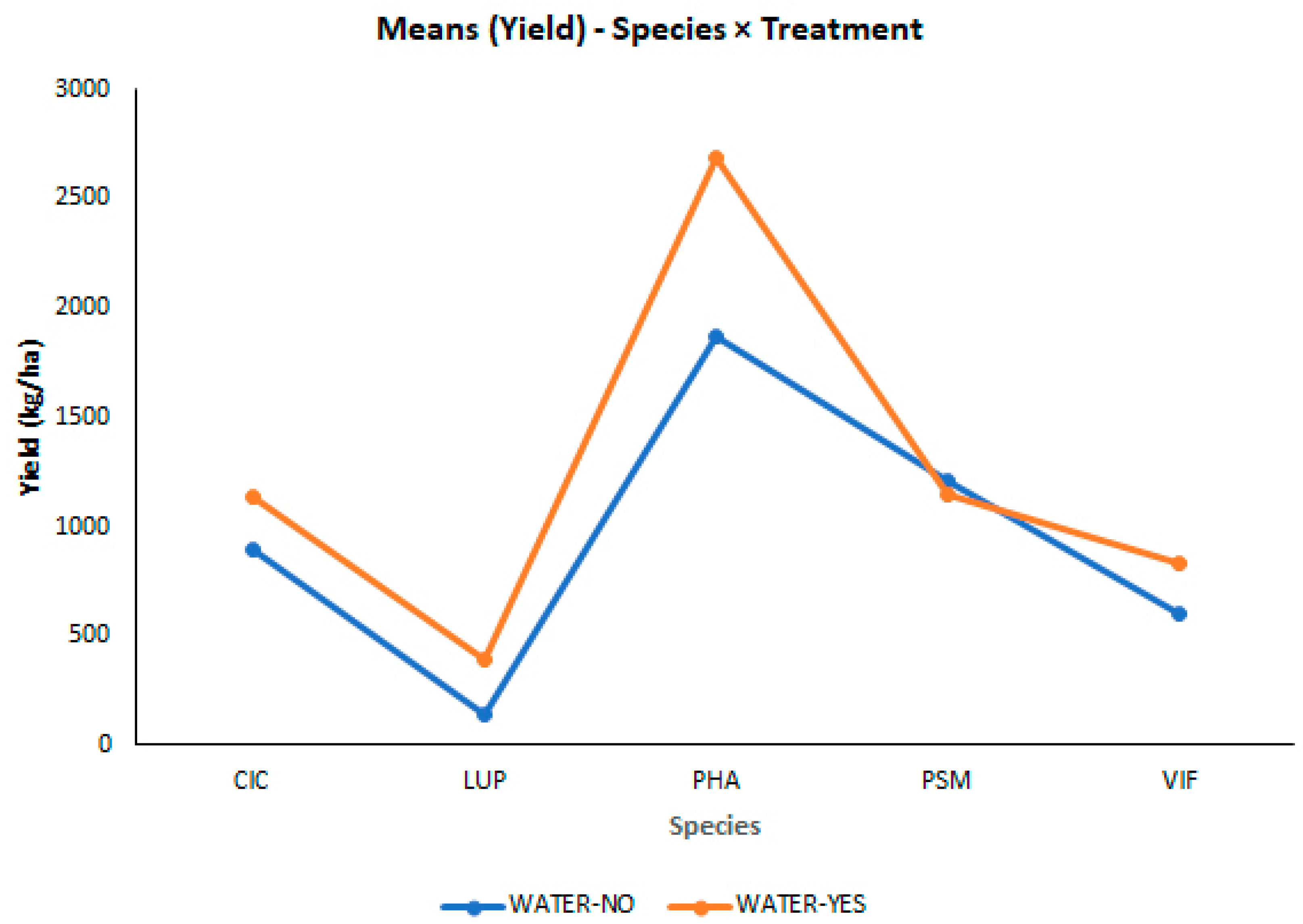
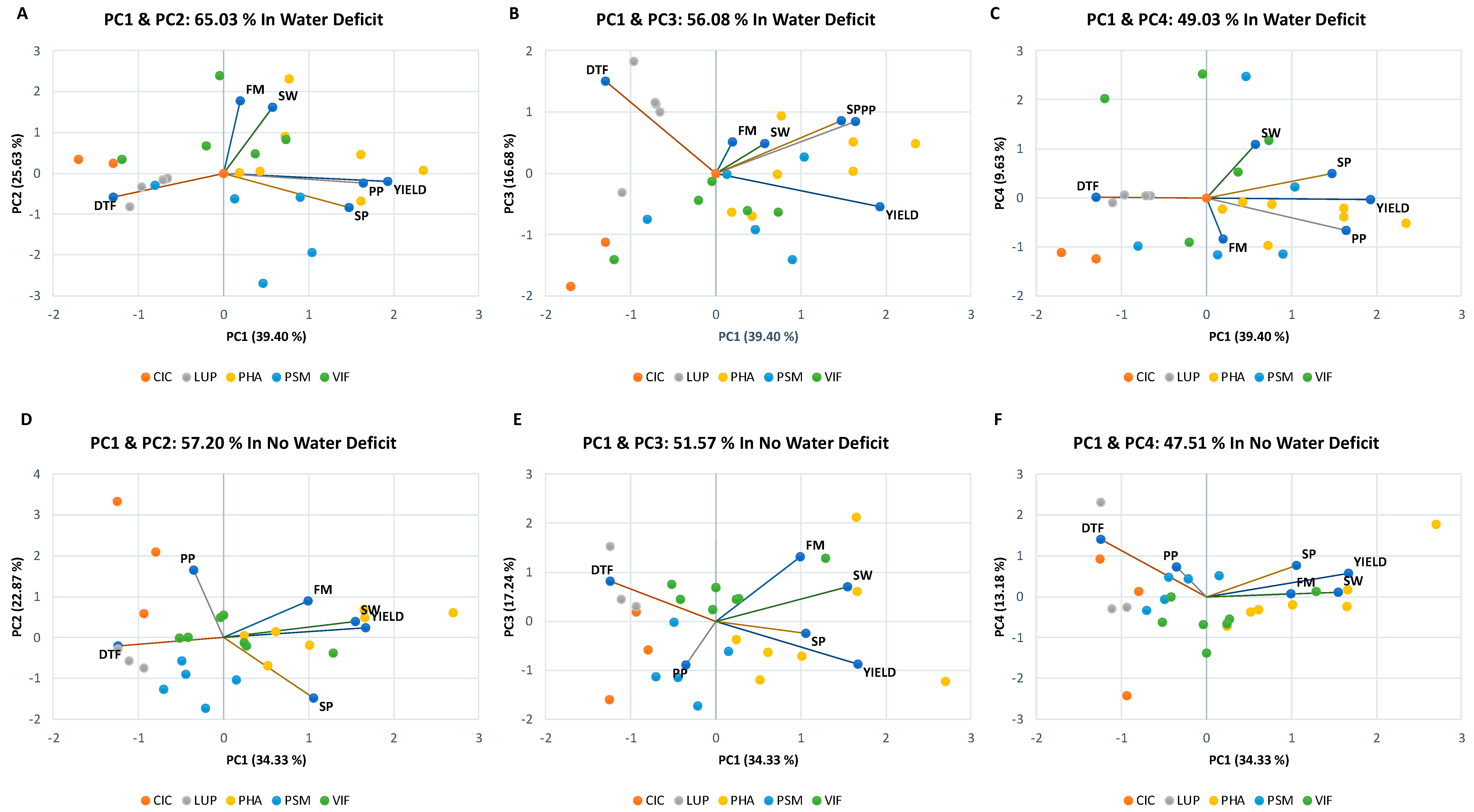
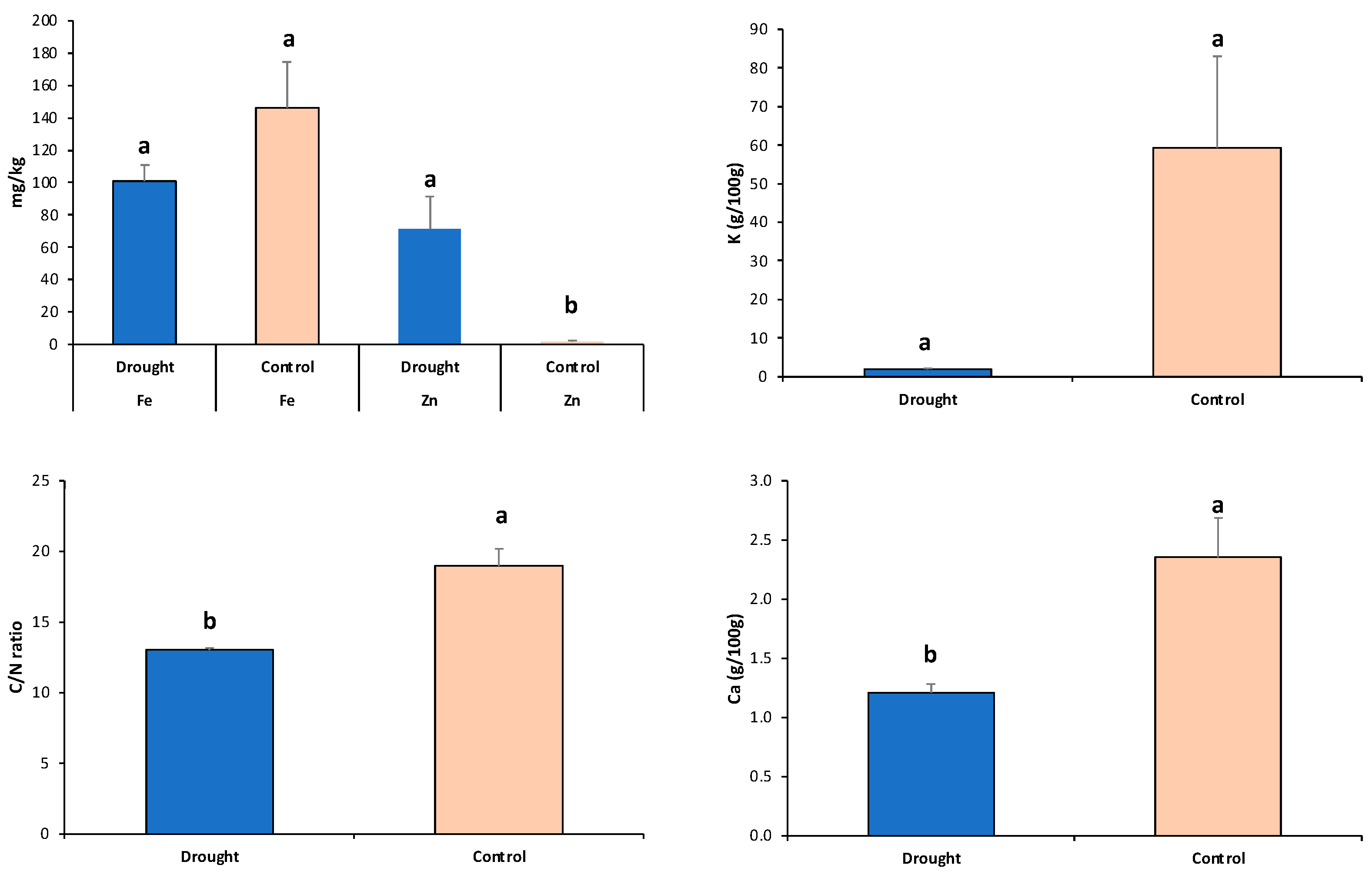
3.2. Effects of the Different Climate Manipulation Stress Levels on Agronomical Performance and Leaf Nutrient Status
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maphosa, Y.; Jideani, V.A. The Role of Legumes in Human Nutrition. In Functional Food—Improve Health through Adequate Food; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Burstin, J.; Gallardo, K.; Mir, R.R.; Varshney, R.K.; Duc, G. Improving protein content and nutrition quality. In Biology and Breeding of Food Legumes; CABI: Wallingford, UK, 2011; pp. 314–328. [Google Scholar] [CrossRef]
- Jensen, E.S.; Peoples, M.B.; Boddey, R.M.; Gresshoff, P.M.; Hauggaard-Nielsen, H.; Alves, B., Jr.; Morrison, M.J. Legumes for mitigation of climate change and the provision of feedstock for biofuels and biorefineries. A review. Agron. Sustain. Dev. 2012, 32, 329–364. [Google Scholar] [CrossRef]
- Soltani, A.; Torabi, B.; Zeinali, E.; Sarparast, R. Response of chickpea to photoperiod as a qualitative long-day plant. Asian J. Plant Sci. 2004, 6, 705–708. [Google Scholar] [CrossRef]
- Stefanova, K.T.; Buirchell, B. Multiplicative mixed models for genetic gain assessment in lupin breeding. Crop Sci. 2010, 50, 880–891. [Google Scholar] [CrossRef]
- Duc, G.; Bao, S.; Baum, M.; Redden, B.; Sadiki, M.; Suso, M.J.; Vishniakova, M.; Zong, X. Diversity maintenance and use of Vicia faba L. genetic resources. Field Crops Res. 2010, 115, 270–278. [Google Scholar] [CrossRef]
- Adhikari, K.N.; Buirchell, B.J.; Sweetingham, M.W. Length of vernalization period affects flowering time in three lupin species. Plant Breed. 2012, 131, 631–636. [Google Scholar] [CrossRef]
- Berger, J.D.; Shrestha, D.; Ludwig, C. Reproductive strategies in Mediterranean legumes: Trade-offs between phenology, seed size and vigor within and between wild and domesticated Lupinus species collected along aridity gradients. Front. Plant Sci. 2017, 8, 548. [Google Scholar] [CrossRef] [PubMed]
- Flores, F.; Hybl, M.; Knudsen, J.; Marget, P.; Muel, F.; Nadal, S.; Narits, L.; Raffiot, B.; Sass, O.; Solis, I.; et al. Adaptation of spring faba bean types across European climates. Field Crops Res. 2013, 145, 1–9. [Google Scholar] [CrossRef]
- Annicchiarico, P.; Barrett, B.; Brummer, E.C.; Julier, B.; Marshall, A.H. Achievements and challenges in improving temperate perennial forage legumes. Crit. Rev. Plant Sci. 2015, 34, 327–380. [Google Scholar] [CrossRef]
- Daba, K.; Warkentin, T.D.; Bueckert, R.; Todd, C.D.; Tar’an, B. Determination of Photoperiod-Sensitive Phase in Chickpea (Cicer arietinum L.). Front. Plant Sci. 2016, 7, 478. [Google Scholar] [CrossRef]
- Książkiewicz, M.; Nazzicari, N.; Yang, H.; Nelson, M.N.; Renshaw, D.; Rychel-Bielska, S.; Ferrari, B.; Carelli, M.; Tomaszewska, M.; Stawiński, S.; et al. A high-density consensus linkage map of white lupin highlights synteny with narrow-leafed lupin and provides markers tagging key agronomic traits. Sci. Rep. 2017, 7, 15335. [Google Scholar] [CrossRef]
- Cowling, W.A. Genetic diversity in narrow-leafed lupin breeding after the domestication bottleneck. In The Lupin Genome; Springer: Berlin/Heidelberg, Germany, 2020; pp. 1–17. [Google Scholar] [CrossRef]
- Dutta, A.; Trivedi, A.; Nath, C.P.; Gupta, D.S.; Hazra, K.K. A comprehensive review on grain legumes as climate-smart crops: Challenges and prospects. Environ. Chall. 2022, 7, 100479. [Google Scholar] [CrossRef]
- White, J.W.; Laing, D.R. Photoperiod response of flowering in diverse genotypes of common bean (Phaseolus vulgaris). Field Crops Res. 1989, 22, 113–128. [Google Scholar] [CrossRef]
- Roeber, V.M.; Schmülling, T.; Cortleven, A. The photoperiod: Handling and causing stress in plants. Front. Plant Sci. 2022, 12, 781988. [Google Scholar] [CrossRef]
- Sita, K.; Sehgal, A.; HanumanthaRao, B.; Nair, R.M.; Prasad, P.V.V.; Kumar, S.; Gaur, P.M.; Farooq, M.; Siddique, K.H.M.; Varshney, R.K.; et al. Food Legumes and Rising Temperatures: Effects, Adaptive Functional Mechanisms Specific to Reproductive Growth Stage and Strategies to Improve Heat Tolerance. Front. Plant Sci. 2017, 8, 1658. [Google Scholar] [CrossRef] [PubMed]
- Vadez, V.; Berger, J.D.; Warkentin, T.; Asseng, S.; Ratnakumar, P.; Rao, K.P.C.; Gaur, P.M.; Munier-Jolain, N.; Larmure, A.; Voisin, A.-S.; et al. Adapting grain legumes to climatic changes: A review. Agron. Sustain. Dev. 2012, 32, 31–44. [Google Scholar] [CrossRef]
- Cernay, C.; Ben-Ari, T.; Pelzer, E.; Meynard, J.M.; Makowski, D. Estimating variability in grain legume yields across Europe and the Americas. Sci. Rep. 2015, 5, 11171. [Google Scholar] [CrossRef]
- Amede, T.; Schubert, S.; Stahr, K. Mechanisms of drought resistance in grain legumes I: Osmotic adjustment. SINET Ethiop. J. Sci. 2003, 26, 37–46. [Google Scholar] [CrossRef]
- Ashraf, M.F.M.R.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Alessandri, A.; De Felice, M.; Zeng, N.; Mariotti, A.; Pan, Y.; Cherchi, A.; Lee, J.-Y.; Wang, B.; Ha, K.-J.; Ruti, P.; et al. Robust assessment of the expansion and retreat of Mediterranean climate in the 21st century. Sci. Rep. 2014, 4, 7211. [Google Scholar] [CrossRef]
- Ergon, A.; Seddaiu, G.; Korhonen, P.; Virkajärvi, P.; Bellocchi, G.; Jørgensen, M.; Østrem, L.; Reheul, D.; Volaire, F. How can forage production in Nordic and Mediterranean Europe adapt to the challenges and opportunities arising from climate change. Eur. J. Agron. 2018, 92, 97–106. [Google Scholar] [CrossRef]
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of climate change on crops adaptation and strategies to tackle its outcome: A review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Dijkstra, F.A. Drought effect on plant nitrogen and phosphorus: A meta-analysis. New Phytol. 2014, 204, 924–931. [Google Scholar] [CrossRef]
- Myers, S.S.; Zanobetti, A.; Kloog, I.; Huybers, P.; Leakey, A.D.B.; Bloom, A.J.; Carlisle, E.; Dietterich, L.H.; Fitzgerald, G.; Hasegawa, T.; et al. Increasing CO2 threatens human nutrition. Nature 2014, 510, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.; Iqbal, M.A.; Li, C.; Iqbal, A.; Abbas, R.N. Overviewing Drought and Heat Stress Amelioration—From Plant Responses to Microbe-Mediated Mitigation. Sustainability 2023, 15, 1671. [Google Scholar] [CrossRef]
- Smith, M.R.; Myers, S.S. Impact of anthropogenic CO2 emissions on global human nutrition. Nat. Clim. Chang. 2018, 8, 834–839. [Google Scholar] [CrossRef]
- Weyant, C.; Brandeau, M.L.; Burke, M.; Lobell, D.B.; Bendavid, E.; Basu, S. Anticipated burden and mitigation of carbon-dioxide-induced nutritional deficiencies and related diseases: A simulation modeling study. PLoS Med. 2018, 15, e1002586. [Google Scholar] [CrossRef] [PubMed]
- Demers, D.A.; Gosselin, A. Growing greenhouse tomato and sweet pepper under supplemental lighting: Optimal photoperiod, negative effects of long photoperiod and their causes. In Proceedings of the IV International ISHS Symposium on Artificial Lighting 580, Quebec, QC, Canada, 7–9 November 2000; pp. 83–88. [Google Scholar] [CrossRef]
- Kong, Y.; Zheng, Y. Response of growth, yield, and quality of edible-podded snow peas to supplemental LED lighting during winter greenhouse production. Can. J. Plant Sci. 2018, 99, 676–687. [Google Scholar] [CrossRef]
- Fehr, W.R.; Caviness, C.E. Stages of soybean development. Iowa Coop. Ext. Serv. Iowa Agric. Home Econ. Exp. Stn. Spec. Rep. 1977, 80, 11. [Google Scholar]
- Guilioni, L.; Wery, J.; Tardieu, F. Heat stress-induced abortion of buds and flowers in pea: Is sensitivity linked to organ age or to relations between reproductive organs? Ann. Bot. 1997, 80, 159–168. [Google Scholar] [CrossRef]
- Kaushal, N.; Awasthi, R.; Gupta, K.; Gaur, P.; Siddique, K.H.; Nayyar, H. Heat-stress-induced reproductive failures in chickpea (Cicer arietinum) are associated with impaired sucrose metabolism in leaves and anthers. Funct. Plant Biol. 2013, 40, 1334–1349. [Google Scholar] [CrossRef]
- Kumari, V.; Roy, A.; Vijayan, R.; Banerjee, P.; Verma, V.; Nalia, A.; Pramanik, M.; Mukherjee, B.; Ghosh, A.; Reja, H.; et al. Drought and Heat Stress in Cool-Season Food Legumes in Sub-Tropical Regions: Consequences, Adaptation, and Mitigation Strategies. Plants 2021, 10, 1038. [Google Scholar] [CrossRef] [PubMed]
- El Nadi, A.H. Water relations of beans I. Effects of water stress on growth and flowering. Exp. Agric. 1969, 5, 195–207. [Google Scholar] [CrossRef]
- Hailemichael, G.; Catalina, A.; González, M.R.; Martin, P. Relationships between water status, leaf chlorophyll content and photosynthetic performance in Tempranillo vineyards. South Afr. J. Enol. Vitic. 2016, 37, 149–156. [Google Scholar] [CrossRef]
- Zivcak, M.; Brestic, M.; Balatova, Z.; Drevenakova, P.; Olsovska, K.; Kalaji, H.M.; Yang, X.; Allakhverdiev, S.I. Photosynthetic electron transport and specific photoprotective responses in wheat leaves under drought stress. Photosynth. Res. 2013, 117, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Garg, B.K. Nutrient uptake and management under drought: Nutrient-moisture interaction. Curr. Agric. 2003, 27, 1–8. [Google Scholar]
- Hu, Y.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Barber, S.A. Soil Nutrient Bioavailability: A Mechanistic Approach; John Wiley & Sons: Hoboken, NJ, USA, 1995. [Google Scholar]
- Himelblau, E.; Amasino, R.M. Nutrients mobilized from leaves of Arabidopsis thaliana during leaf senescence. J. Plant Physiol. 2001, 158, 1317–1323. [Google Scholar] [CrossRef]
- Sankaran, R.P.; Grusak, M.A. Whole shoot mineral partitioning and accumulation in pea (Pisum sativum). Front. Plant Sci. 2014, 5, 149. [Google Scholar] [CrossRef]
- Maillard, A.; Diquélou, S.; Billard, V.; Laîné, P.; Garnica, M.; Eprudent, M.; Garcia-Mina, J.-M.; Eyvin, J.-C.; Ourry, A. Leaf mineral nutrient remobilization during leaf senescence and modulation by nutrient deficiency. Front. Plant Sci. 2015, 6, 317. [Google Scholar] [CrossRef]
- Price, A.H.; Hendry, G.A.F. Iron-catalysed oxygen radical formation and its possible contribution to drought damage in nine native grasses and three cereals. Plant Cell Environ. 1991, 14, 477–484. [Google Scholar] [CrossRef]
- Gadallah MAA Effects of indole-3-acetic acid and zinc on the growth, osmotic potential and soluble carbon and nitrogen components of soybean plants growing under water deficit. J. Arid. Environ. 2000, 44, 451–467. [CrossRef]
- Iuchi, S.; Kobayashi, M.; Taji, T.; Naramoto, M.; Seki, M.; Kato, T.; Tabata, S.; Kakubari, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J. 2001, 27, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhang, X.; Wang, W.; Wang, Y.; Ming, F. The suppression of WRKY44 by GIGANTEA-miR172 pathway is involved in drought response of Arabidopsis thaliana. PLoS ONE 2013, 8, e73541. [Google Scholar] [CrossRef] [PubMed]
- Fitter, A.H.; Fitter, R.S.R.; Harris, I.T.B.; Williamson, M.H. Relationships between first flowering date and temperature in the flora of a locality in central England. Funct. Ecol. 1995, 9, 55–60. [Google Scholar] [CrossRef]
- Prasad, P.V.; Boote, K.J.; Allen, L.H., Jr.; Thomas, J.M. Effects of elevated temperature and carbon dioxide on seed-set and yield of kidney bean (Phaseolus vulgaris L.). Glob. Change Biol. 2002, 8, 710–721. [Google Scholar] [CrossRef]
- Rainey, K.M.; Griffiths, P.D. Inheritance of heat tolerance during reproductive development in snap bean (Phaseolus vulgaris L.). J. Am. Soc. Hortic. Sci. 2005, 130, 700–706. [Google Scholar] [CrossRef]
- Krishnamurthy, L.; Gaur, P.M.; Basu, P.S.; Chaturvedi, S.K.; Tripathi, S.; Vadez, V.; Rathore, A.; Varshney, R.K.; Gowda, C.L.L. Large genetic variation for heat tolerance in the reference collection of chickpea (Cicer arietinum L.) germplasm. Plant Genet. Resour. 2011, 9, 59–69. [Google Scholar] [CrossRef]
- Kumar, S.; Thakur, P.; Kaushal, N.; Malik, J.A.; Gaur, P.; Nayyar, H. Effect of varying high temperatures during reproductive growth on reproductive function, oxidative stress and seed yield in chickpea genotypes differing in heat sensitivity. Arch. Agron. Soil Sci. 2013, 59, 823–843. [Google Scholar] [CrossRef]
- Kumari, V.V.; Banerjee, P.; Verma, V.C.; Sukumaran, S.; Chandran, M.A.S.; Gopinath, K.A.; Venkatesh, G.; Yadav, S.K.; Singh, V.K.; Awasthi, N.K. Plant nutrition: An effective way to alleviate abiotic stress in agricultural crops. Int. J. Mol. Sci. 2022, 23, 8519. [Google Scholar] [CrossRef]
- Bista, D.R.; Heckathorn, S.A.; Jayawardena, D.M.; Mishra, S.; Boldt, J.K. Effects of drought on nutrient uptake and the levels of nutrient-uptake proteins in roots of drought-sensitive and-tolerant grasses. Plants 2018, 7, 28. [Google Scholar] [CrossRef]
- Rouphael, Y.; Cardarelli, M.; Schwarz, D.; Franken, P.; Colla, G. Effects of drought on nutrient uptake and assimilation in vegetable crops. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Springer: Berlin/Heidelberg, Germany, 2012; pp. 171–195. [Google Scholar] [CrossRef]
- Peck, A.W.; McDonald, G.K. Adequate zinc nutrition alleviates the adverse effects of heat stress in bread wheat. Plant Soil 2010, 337, 355–374. [Google Scholar] [CrossRef]
- Umair Hassan, M.; Aamer, M.; Umer Chattha, M.; Haiying, T.; Shahzad, B.; Barbanti, L.; Nawaz, M.; Rasheed, A.; Afzal, A.; Liu, Y.; et al. The critical role of zinc in plants facing the drought stress. Agriculture 2020, 10, 396. [Google Scholar] [CrossRef]
- Galluzzi, G.; Seyoum, A.; Halewood, M.; López Noriega, I.; Welch, E.W. The role of genetic resources in breeding for climate change: The case of public breeding programmes in eighteen developing countries. Plants 2020, 9, 1129. [Google Scholar] [CrossRef] [PubMed]



| Location | Site | ENV a | Coordinates | Altitude (masl) b | Soil pH | Rainfall (mm) c | Temperature (°C) c | Relative Humidity (%) d | Time Frame | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Latitude | Longitude | Min | Max | Average | Min | Max | Average | |||||||
| Pontevedra | Salcedo | ENV1 | 42.4068011 | −8.6416949 | 35 | 5.7 | 707 | 3.3 | 26.6 | 13.4 | 62 | 92 | 80 | November 2021 to June 2022 |
| Orense | Xunqueira de Ambia | ENV2 | 42.148440 | −7.725078 | 618 | 5.0 | 118 | 2.9 | 33.3 | 17.2 | 60 | 89 | 70 | June 2021 to September 2021 |
| Pontevedra | Salcedo | ENV3 | 42.4069364 | −8.6426684 | 30 | 6.3 | 327 | 7.6 | 32.0 | 17.9 | 58 | 92 | 78 | March 2022 to August 2022 |
| Orense | Corga, Sandias | ENV4 | 42.132144 | −7.729865 | 619 | 4.5 | 154 | 1.1 | 34.2 | 16.8 | 55 | 90 | 73 | April 2022 to August 2022 |
| Species/Environments | Average Daily Temperature (°C) | Mean Photoperiod (Hours) | Accumulated Photoperiod (Hours) | Photothermal Units | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VE to R1 a | R1 to R5 | R5 to R7 | VE to R1 a | R1 to R5 | R5 to R7 | VE to R1 a | R1 to R5 | R5 to R7 | VE to R1 a | R1 to R5 | R5 to R7 | |
| Environment 1 | ||||||||||||
| PHA | 11.56 | 8.82 | 11.25 | 9.27 | 9.61 | 11.04 | 509.85 | 211.31 | 463.47 | 107.16 | 84.72 | 124.14 |
| PSM | 11.25 | 14.90 | 18.74 | 10.83 | 13.32 | 14.71 | 1386.24 | 666.00 | 235.28 | 121.84 | 198.47 | 275.57 |
| LUP | 11.33 | 15.53 | 19.35 | 10.99 | 13.47 | 14.76 | 1527.61 | 511.86 | 516.60 | 124.52 | 209.19 | 285.61 |
| CIC | 12.49 | 19.62 | 21.44 | 11.95 | 14.84 | 14.68 | 2210.75 | 341.21 | 748.68 | 149.26 | 291.06 | 314.74 |
| VIF | 10.88 | 12.65 | 17.90 | 9.96 | 12.01 | 14.27 | 946.20 | 744.31 | 542.07 | 108.36 | 151.86 | 255.34 |
| Average | 11.50 | 14.30 | 17.74 | 10.60 | 12.65 | 13.89 | 1316.13 | 494.94 | 501.22 | 122.23 | 187.06 | 251.08 |
| Environment 2 | ||||||||||||
| PHA | 21.81 | 21.94 | 17.00 | 14.83 | 14.34 | 12.67 | 192.73 | 286.80 | 810.88 | 323.33 | 314.62 | 215.39 |
| PSM | 22.04 | 21.55 | 17.85 | 14.81 | 14.29 | 12.81 | 236.96 | 300.09 | 627.45 | 326.41 | 307.95 | 228.57 |
| LUP b | ||||||||||||
| CIC | 22.39 | 22.06 | 17.29 | 14.80 | 14.02 | 12.52 | 251.52 | 336.36 | 575.92 | 331.26 | 309.17 | 216.47 |
| VIF | 22.11 | 22.12 | 16.43 | 14.76 | 13.97 | 12.29 | 265.59 | 307.34 | 737.40 | 330.36 | 309.02 | 201.92 |
| Average | 22.09 | 21.92 | 17.14 | 14.80 | 14.15 | 12.57 | 236.70 | 307.65 | 687.91 | 327.84 | 310.19 | 215.59 |
| Environment 3 | ||||||||||||
| PHA | 15.84 | 18.72 | 19.97 | 14.11 | 15.03 | 15.11 | 423.15 | 270.45 | 256.87 | 315.81 | 281.27 | 301.75 |
| PSM | 13.50 | 18.87 | 19.40 | 12.43 | 14.71 | 15.15 | 944.30 | 279.49 | 196.95 | 278.20 | 277.58 | 293.91 |
| LUP | 16.60 | 21.11 | 22.30 | 13.64 | 14.80 | 14.41 | 1268.52 | 384.67 | 115.28 | 305.40 | 312.32 | 321.34 |
| CIC | 15.30 | 19.45 | 19.85 | 13.31 | 14.85 | 15.18 | 838.22 | 460.20 | 242.88 | 297.90 | 288.74 | 301.32 |
| VIF | 15.30 | 19.89 | 20.56 | 13.32 | 14.86 | 15.17 | 865.80 | 371.38 | 424.76 | 298.23 | 295.47 | 311.90 |
| Average | 15.31 | 19.61 | 20.42 | 13.36 | 14.85 | 15.00 | 868.00 | 353.24 | 247.35 | 299.11 | 291.07 | 306.04 |
| Environment 4 | ||||||||||||
| PHA | 18.11 | 18.91 | 21.61 | 14.81 | 14.88 | 14.42 | 473.92 | 371.88 | 245.14 | 331.60 | 281.29 | 311.62 |
| PSM | 13.45 | 16.96 | 18.98 | 13.87 | 14.77 | 14.87 | 374.36 | 398.79 | 475.84 | 310.44 | 250.50 | 282.23 |
| LUP c | ||||||||||||
| CIC c | ||||||||||||
| VIF | 12.19 | 16.02 | 18.83 | 13.78 | 14.65 | 14.84 | 261.82 | 395.55 | 593.40 | 167.98 | 234.69 | 279.34 |
| Average | 14.58 | 17.30 | 19.81 | 14.15 | 14.77 | 14.71 | 370.03 | 388.74 | 438.13 | 270.00 | 255.49 | 291.06 |
| Source | DF a | Mean Squares | F-Value | P > F |
|---|---|---|---|---|
| Environment (E) | 1 | 41,820,448.87 | 222.93 | <0.0001 |
| Genotype (Species) | 66 | 7,203,185.01 | 38.40 | <0.0001 |
| Genotype × Enviroment (GE) | 22 | 4,140,047.50 | 22.07 | <0.0001 |
| Error | 193 | 187,596.42 |
| Source | DF a | kg ha−1 | ||
|---|---|---|---|---|
| Mean Square | F Value | Pr > F | ||
| Environment (ENV) | 1 | 636,106.84 | 1.56 | 0.213 |
| Treatment (T) | 1 | 4,767,266.8 | 11.71 | 0.001 |
| Genotype (G) | 33 | 3,926,123.94 | 9.65 | <0.0001 |
| G × T | 17 | 2,642,056.29 | 6.49 | <0.0001 |
| G × ENV | 9 | 4,315,768.3 | 10.6 | <0.0001 |
| ENV × T | 1 | 25,982.76 | 0.06 | 0.801 |
| Error | 205 | 407,027.98 | ||
| Species*Treatment | Mean Yield (kg ha−1) | ±SE | ||
| PHA*YES | 2686.99a | 156.178 | ||
| PHA*NO | 1866.60b | 165.427 | ||
| PSM*NO | 1204.72a | 169.615 | ||
| PSM*YES | 1142.77a | 151.321 | ||
| CIC*YES | 1137.38a | 273.497 | ||
| CIC*NO | 890.33b | 173.215 | ||
| VIF*YES | 833.79a | 203.852 | ||
| VIF*NO | 606.51a | 256.905 | ||
| LUP*YES | 391.04a | 136.855 | ||
| LUP*NO | 145.51b | 79.046 |
| Parameters | Drought | Control | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DTF | C/N | Ca | Fe | K | Zn | DTF | C/N | Ca | Fe | K | Zn | |
| Yield | −0.651 | 0.362 | 0.357 | 0.314 | 0.362 | −0.289 | −0.433 | 0.694 | 0.512 | 0.205 | −0.002 | −0.436 |
| DTF | −0.525 | −0.492 | −0.467 | −0.523 | 0.406 | −0.212 | −0.226 | −0.040 | −0.097 | 0.297 | ||
| C/N | 0.987 | 0.867 | 1.000 | −0.797 | 0.472 | 0.125 | 0.017 | −0.745 | ||||
| Ca | 0.814 | 0.988 | −0.834 | −0.029 | −0.001 | −0.613 | ||||||
| Fe | 0.862 | −0.390 | 0.905 | −0.342 | ||||||||
| K | −0.803 | −0.481 | ||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, A.M.; Pesqueira, A.M.; García, L.; Santalla, M. Effects of Photoperiod and Drought on Flowering and Growth Development of Protein-Rich Legumes under Atlantic Environments. Agronomy 2023, 13, 1025. https://doi.org/10.3390/agronomy13041025
González AM, Pesqueira AM, García L, Santalla M. Effects of Photoperiod and Drought on Flowering and Growth Development of Protein-Rich Legumes under Atlantic Environments. Agronomy. 2023; 13(4):1025. https://doi.org/10.3390/agronomy13041025
Chicago/Turabian StyleGonzález, Ana M., Ana M. Pesqueira, Lucio García, and Marta Santalla. 2023. "Effects of Photoperiod and Drought on Flowering and Growth Development of Protein-Rich Legumes under Atlantic Environments" Agronomy 13, no. 4: 1025. https://doi.org/10.3390/agronomy13041025
APA StyleGonzález, A. M., Pesqueira, A. M., García, L., & Santalla, M. (2023). Effects of Photoperiod and Drought on Flowering and Growth Development of Protein-Rich Legumes under Atlantic Environments. Agronomy, 13(4), 1025. https://doi.org/10.3390/agronomy13041025






