Sustainable Strategies for the Control of Pests in Coffee Crops
Abstract
:1. Introduction
2. Coffee Berry Borer (CBB) in Colombia
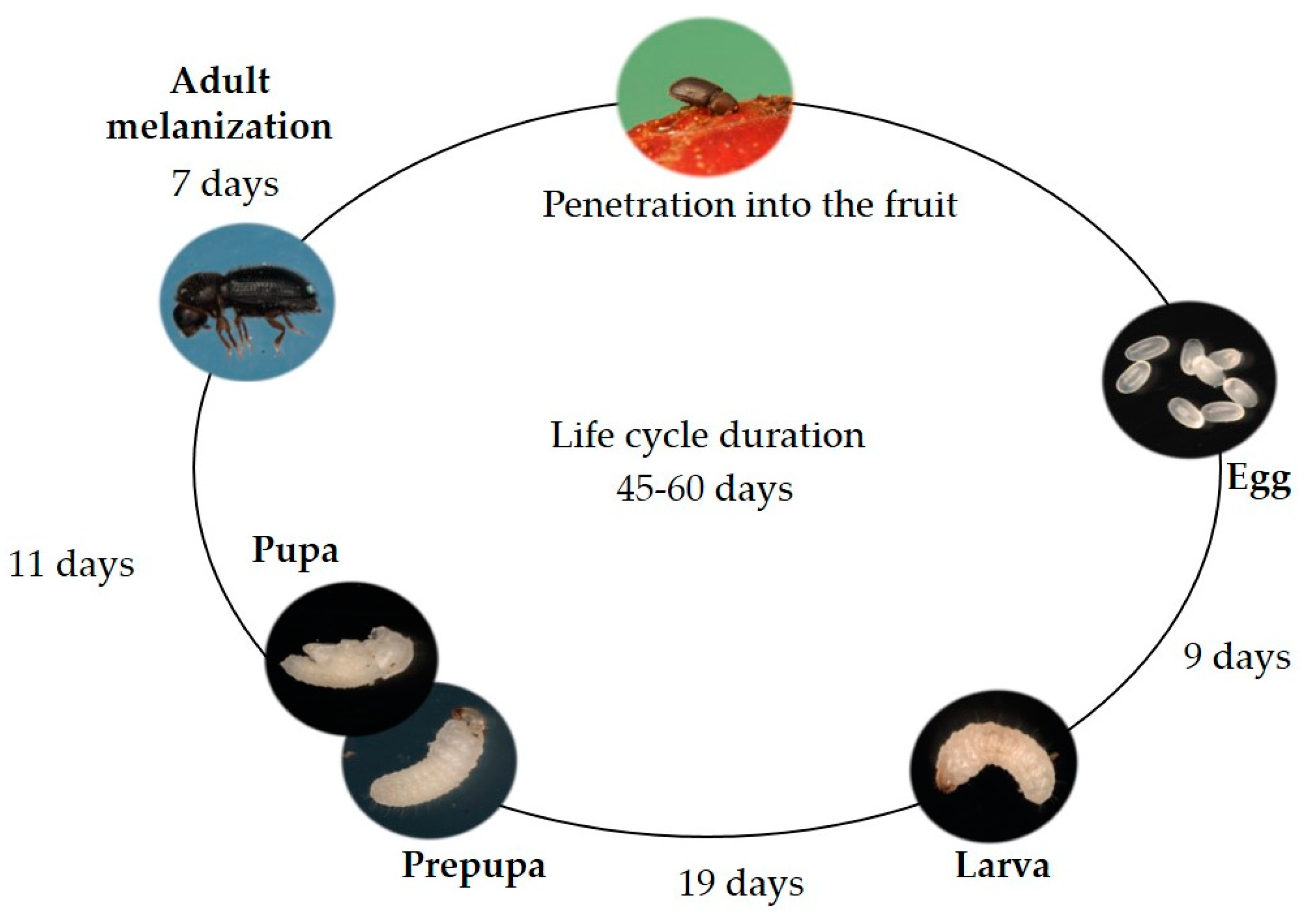
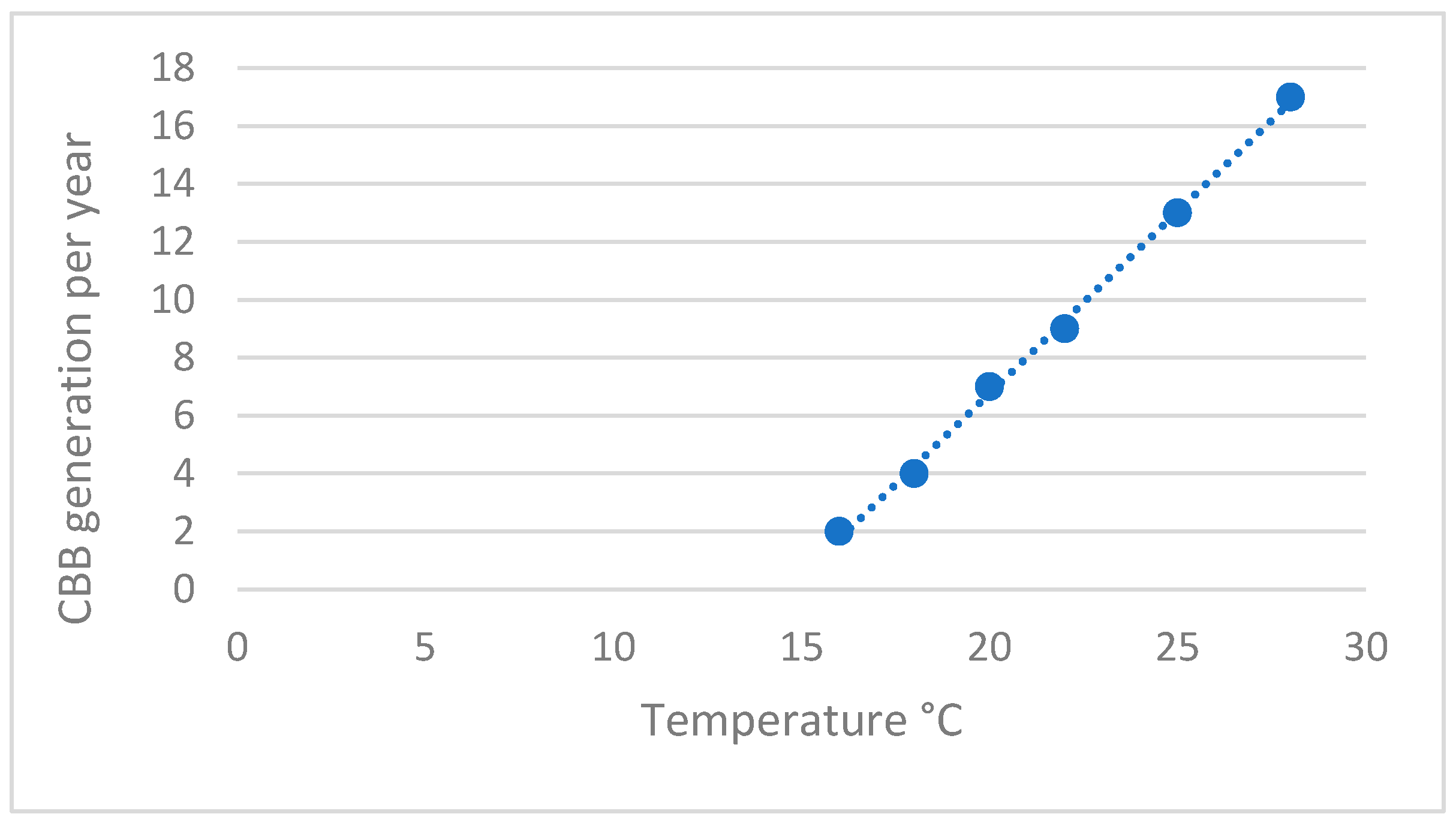
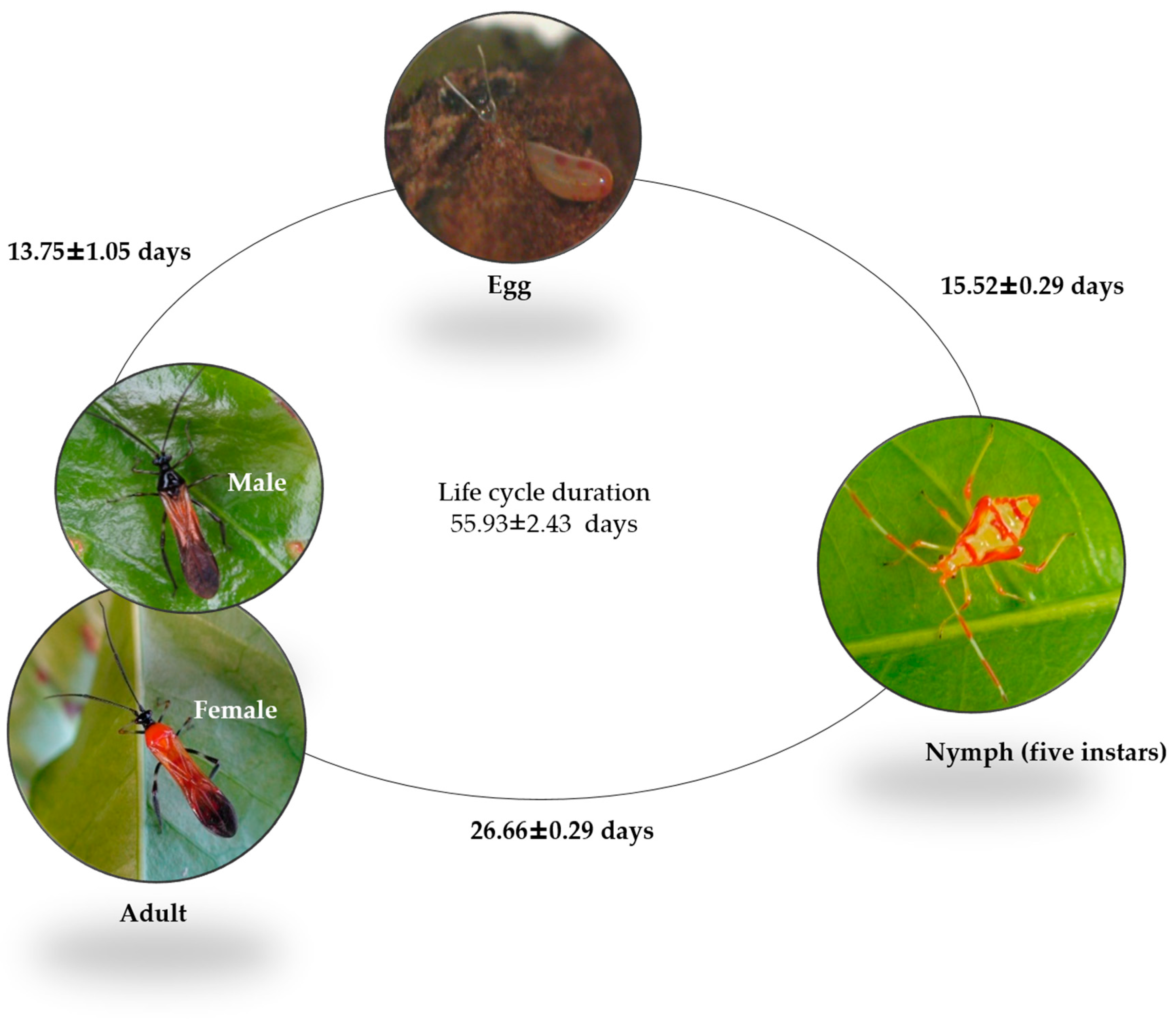
2.1. Insect Biology
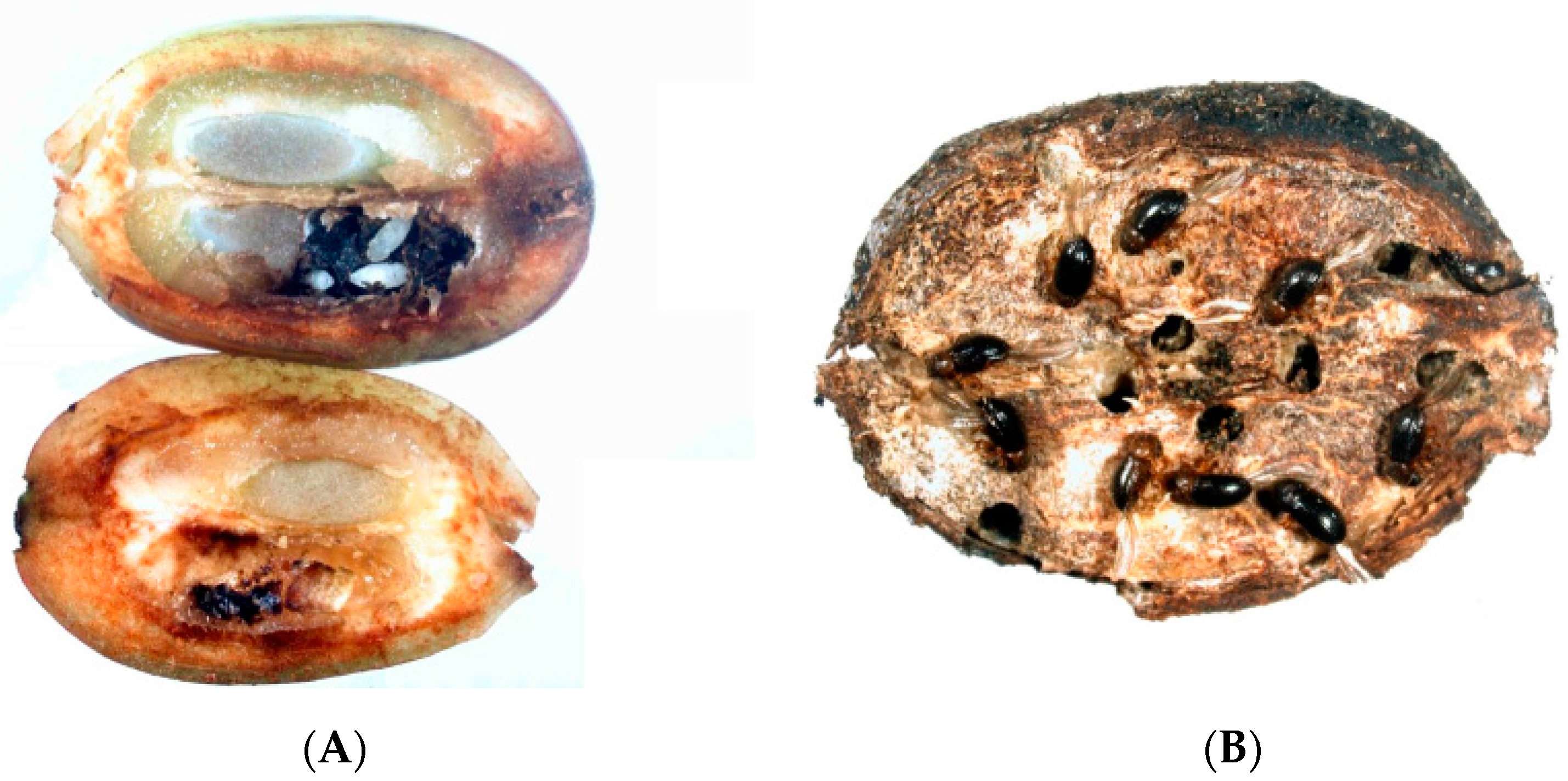
2.2. Insect Damage
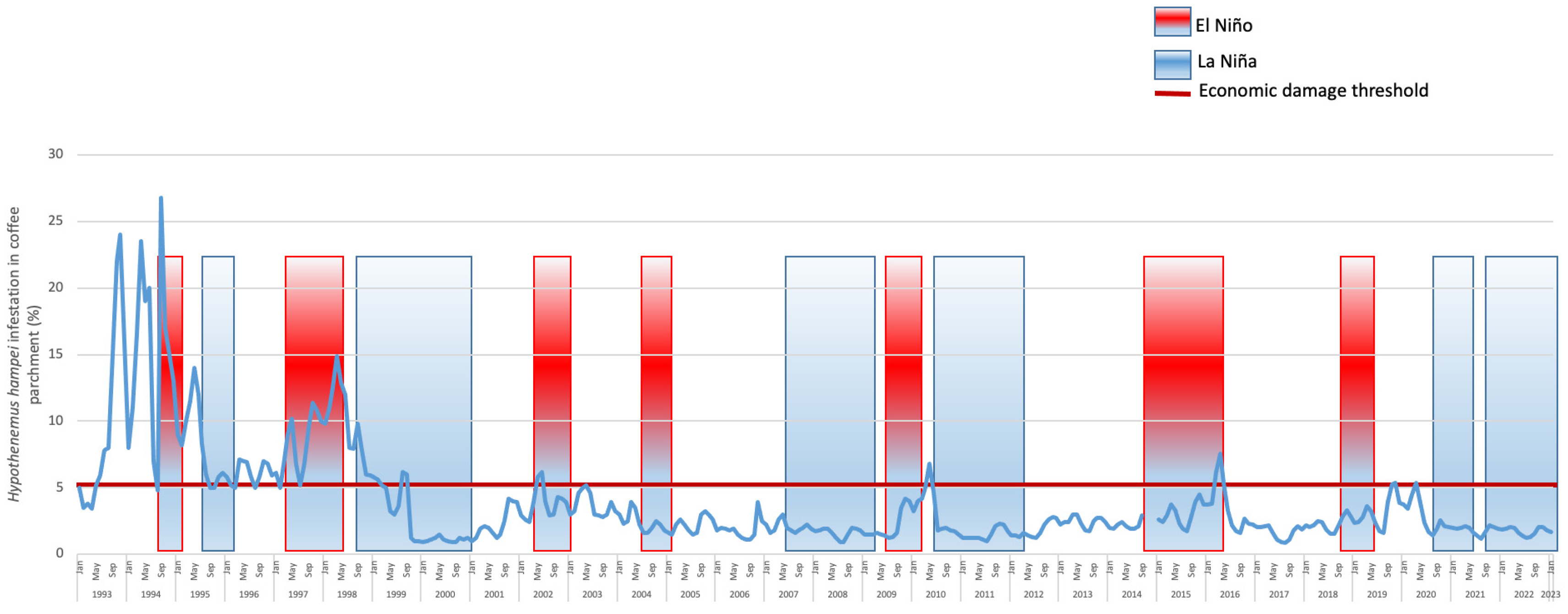
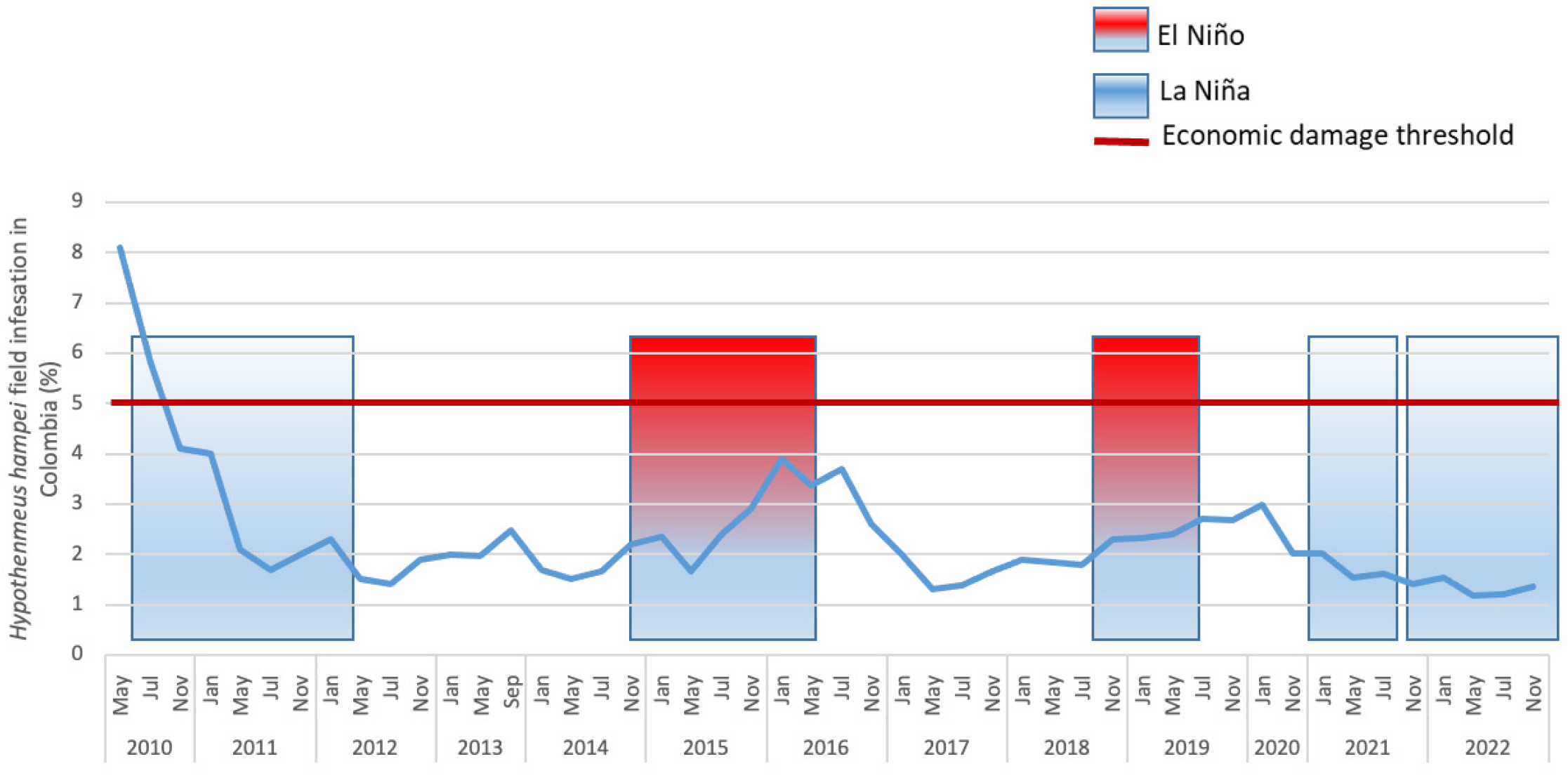
2.3. Economic Damage
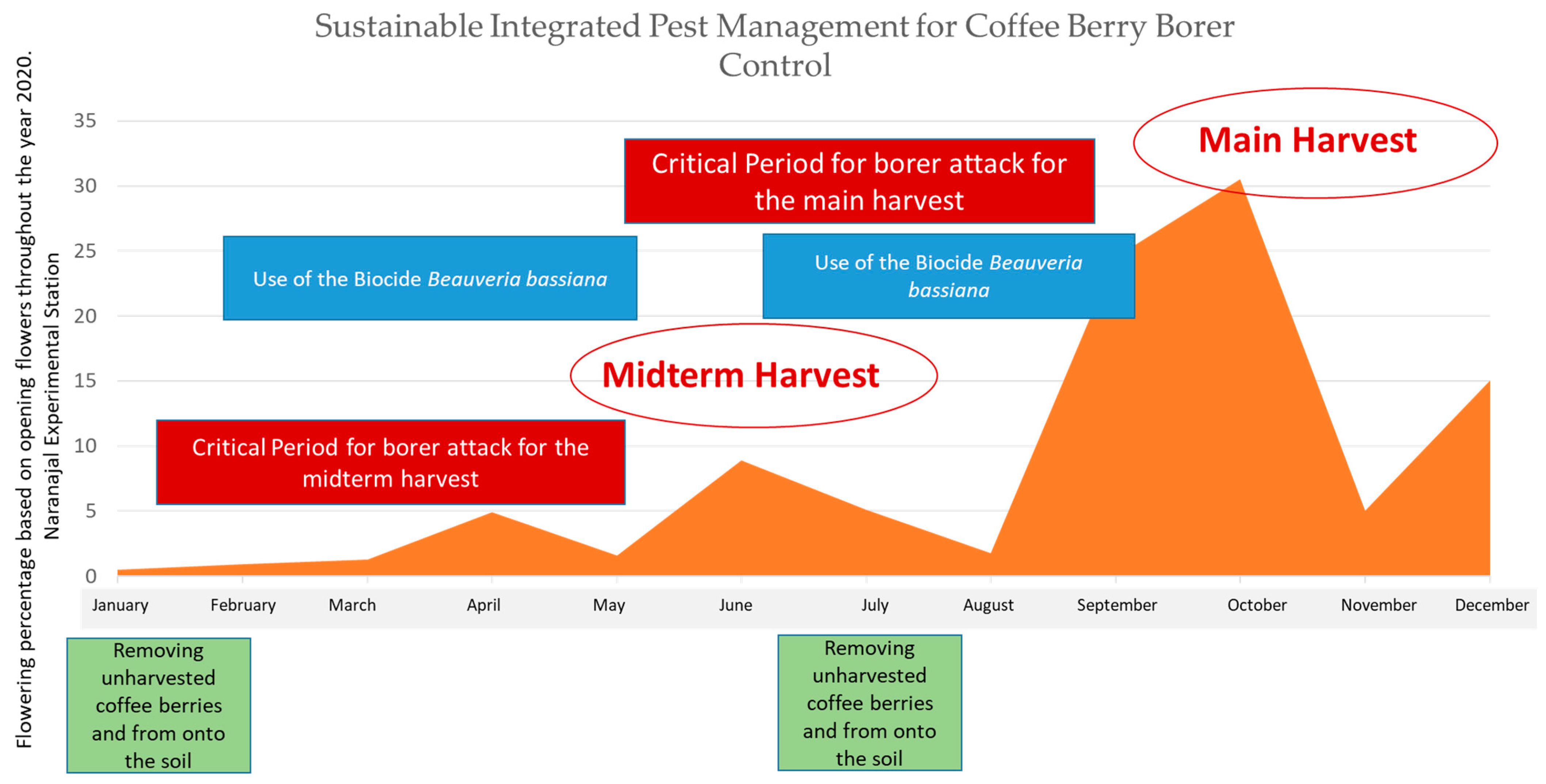
2.4. CBB Integrated Pest Management (IPM)
- Higher level of climate ambition in the European Union, 2030 and 2050.
- Supply of clean, accessible, and secure energy.
- Mobilization of the industry in the direction of a clean and circular economy.
- Efficient use of energy and resources in construction and renovation.
- Accelerate the transition to sustainable and intelligent mobility.
- “From farm to table”: a fair, healthy, and environmentally friendly food system.
- Preservation and restoration of ecosystems and biodiversity.
- Towards zero pollution in an environment without toxic substances
- –
- Flowering registration to detect the critical period when the coffee fruit stages are most susceptible to be attacked by CBB.
- –
- Evaluation of the percentage of CBB infestation in the field and the position of the insect in the coffee bean to identify the opportune moment for the use of insecticides (chemical or biological).
- –
- Identification of aggregated CBB population areas to focalize control actions.
- –
- Timely harvesting and removing unharvested coffee berries.
- –
- Use of biological controllers such as entomopathogens and parasitoids.
- –
- Characterization of coffee lots and identification of CBB aggregation areas.
- –
- Cultural control as a fundamental basis for the IPM program by means of timely harvesting and removing unharvested coffee berries twice a year after main harvesting ended.
- –
- Spraying chemical insecticides at the opportune moment and on the CBB aggregation areas solely during the critical period of the attack of the borer (after 120 days after main flowerings) and when both field infestation exceeds 2% and more than 50% of the borers were in coffee fruits entrance positions just boring the coffee berry [12].
- –
- Spraying Beauveria bassiana (Bals.-Criv.) Vuill. on CBB aggregation areas every other week during the critical period of the attack of the borer.
2.5. CBB Control Prospective
3. Monalonion velezangeli Carvalho and Costa, 1988 (Hemiptera: Miridae)
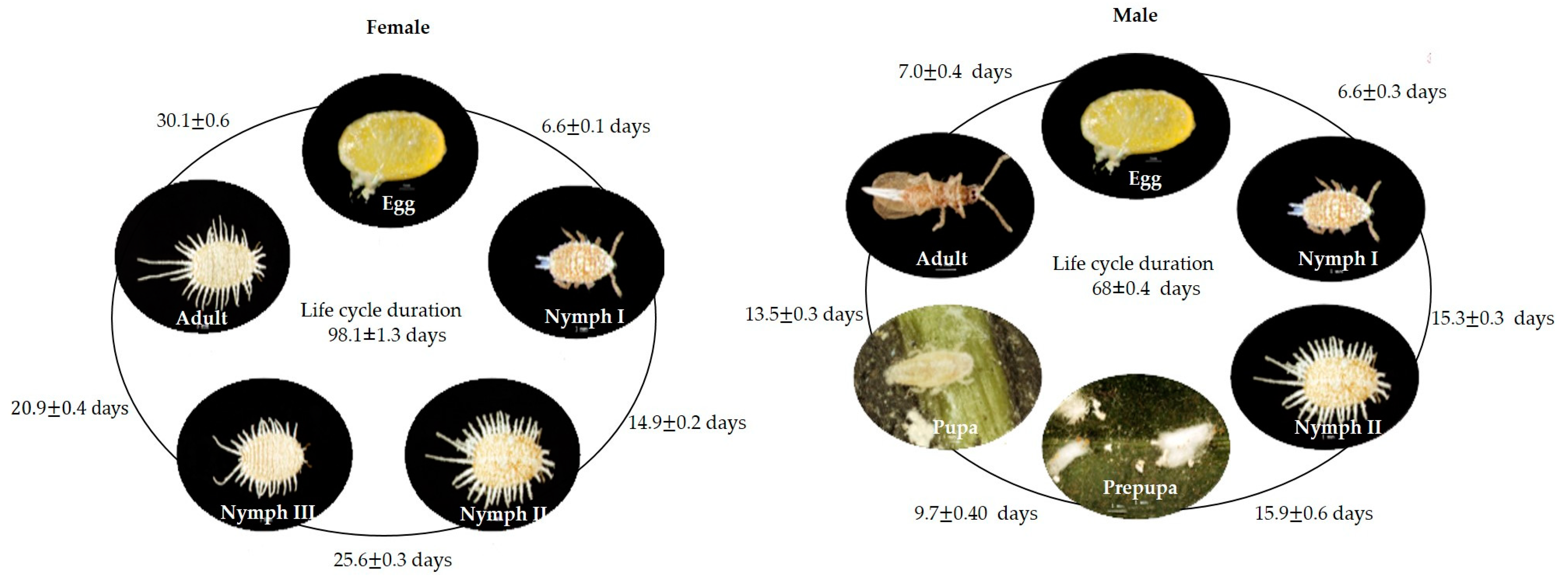
3.1. Insect Biology
3.2. Insect Damage
3.3. Economic Damage
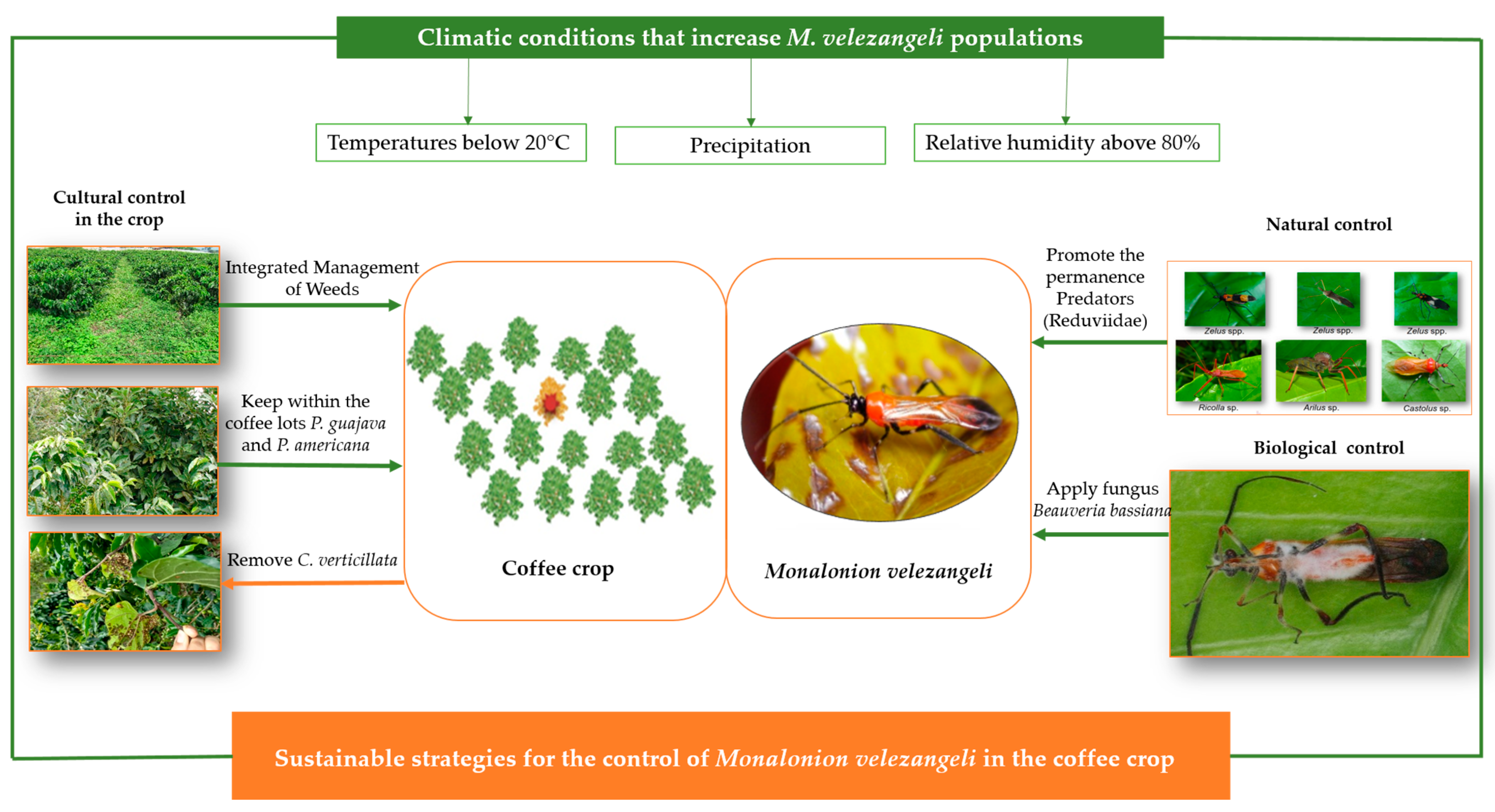
3.4. Control Strategies
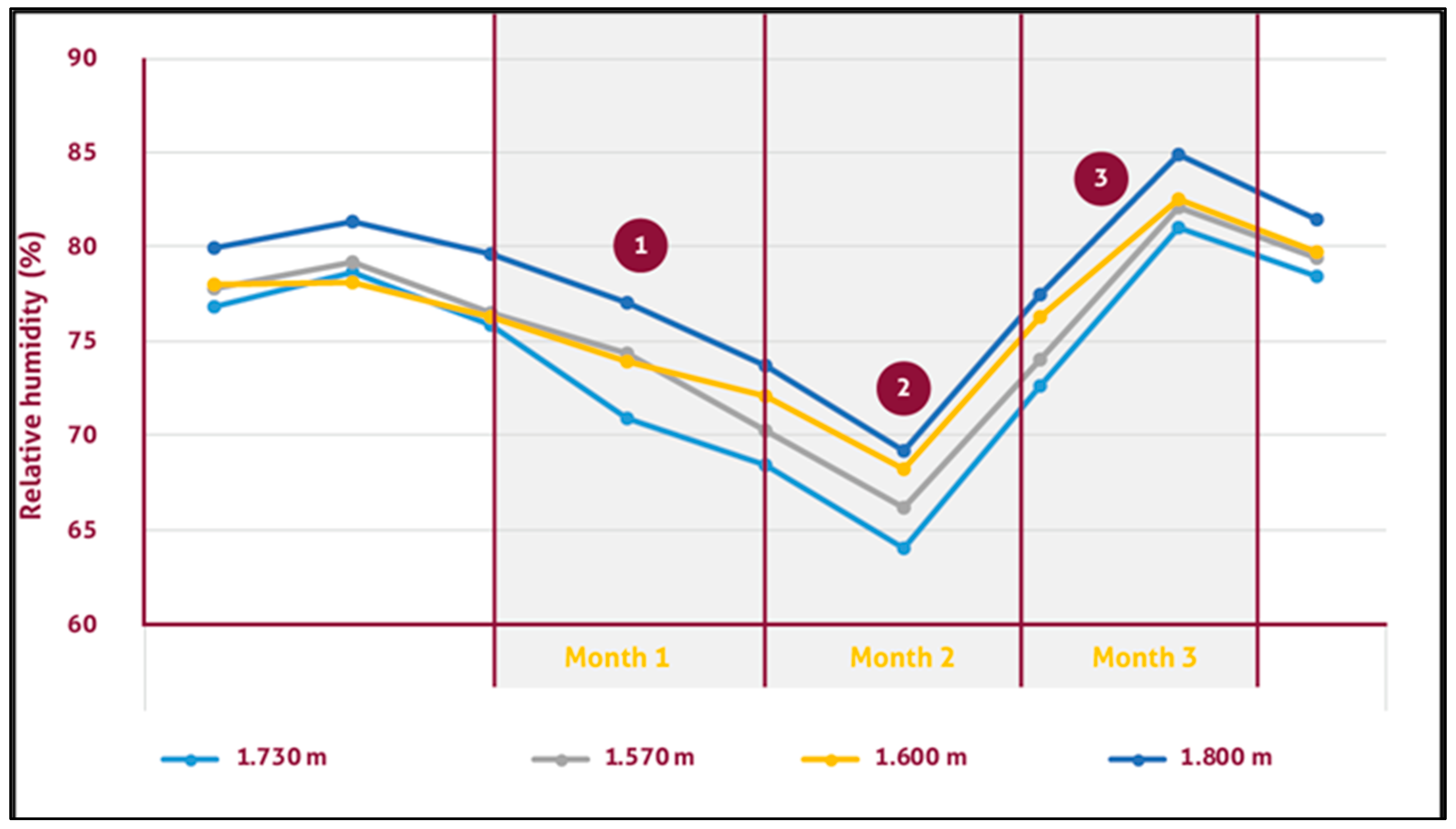
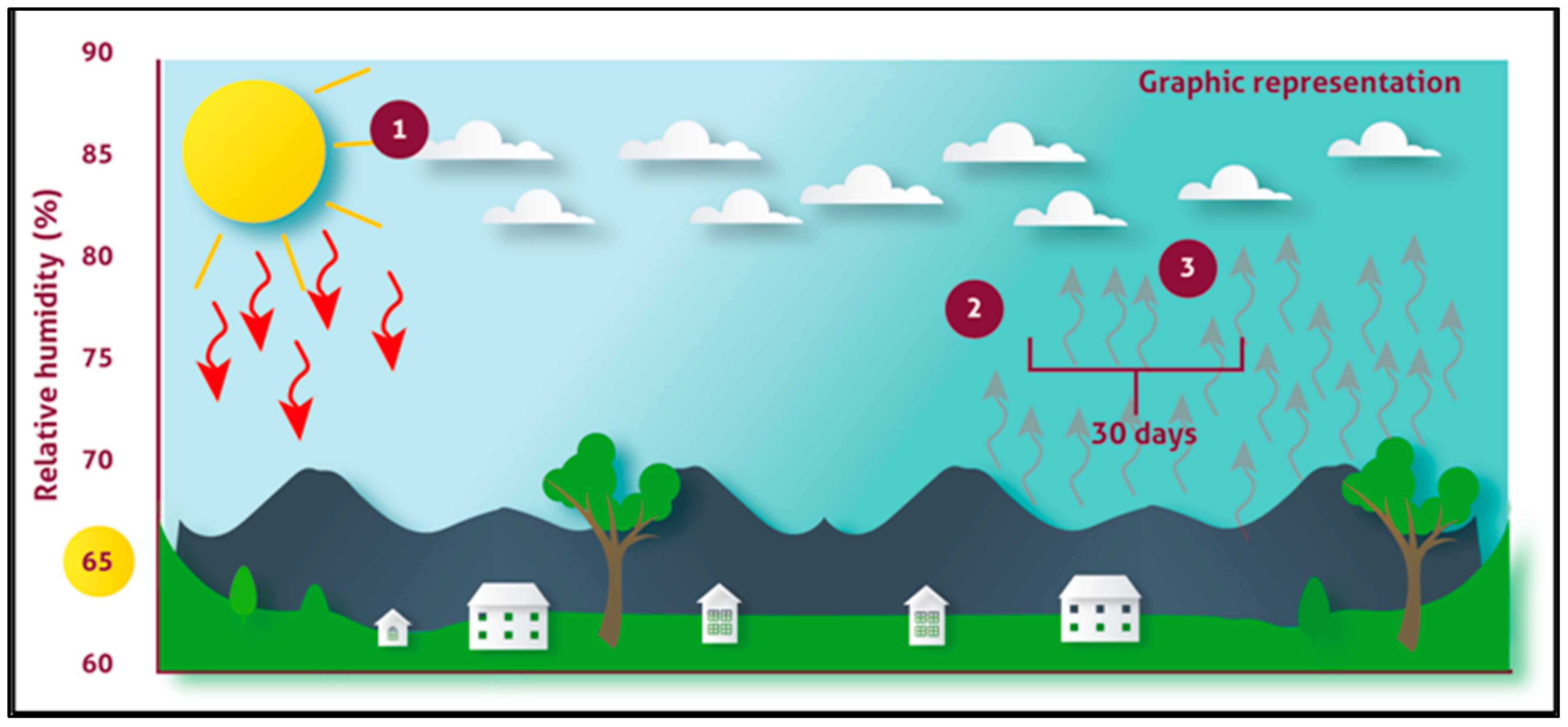
3.5. Monalonion Velezangeli and the Relationship with Weather
4. Coffee Root Mealybugs
4.1. Insect Biology of Puto barberi and Pseudococcus elisae
4.2. Insect Damage
4.3. Economic Damage
4.4. Control Strategy
4.5. Ecological Associations
5. Coffee Leaf Miners Leucoptera coffeella (Guerin-Meneville) (Lepidoptera: Lyonetiidae)
5.1. Insect Biology
5.2. Insect Damage
5.3. Economic Damage
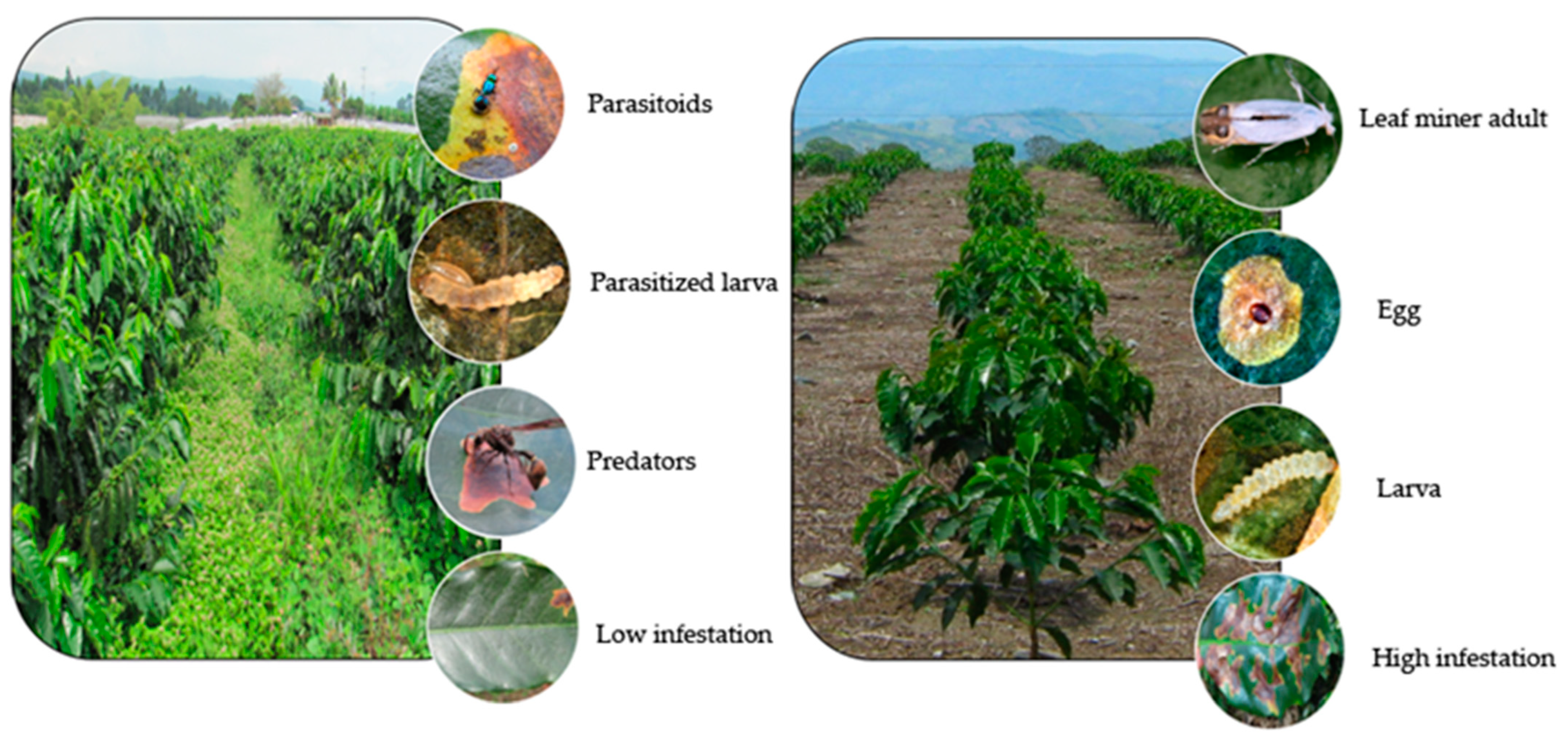
5.4. Strategies for Integrated Management Control of the Coffee Leaf Miner
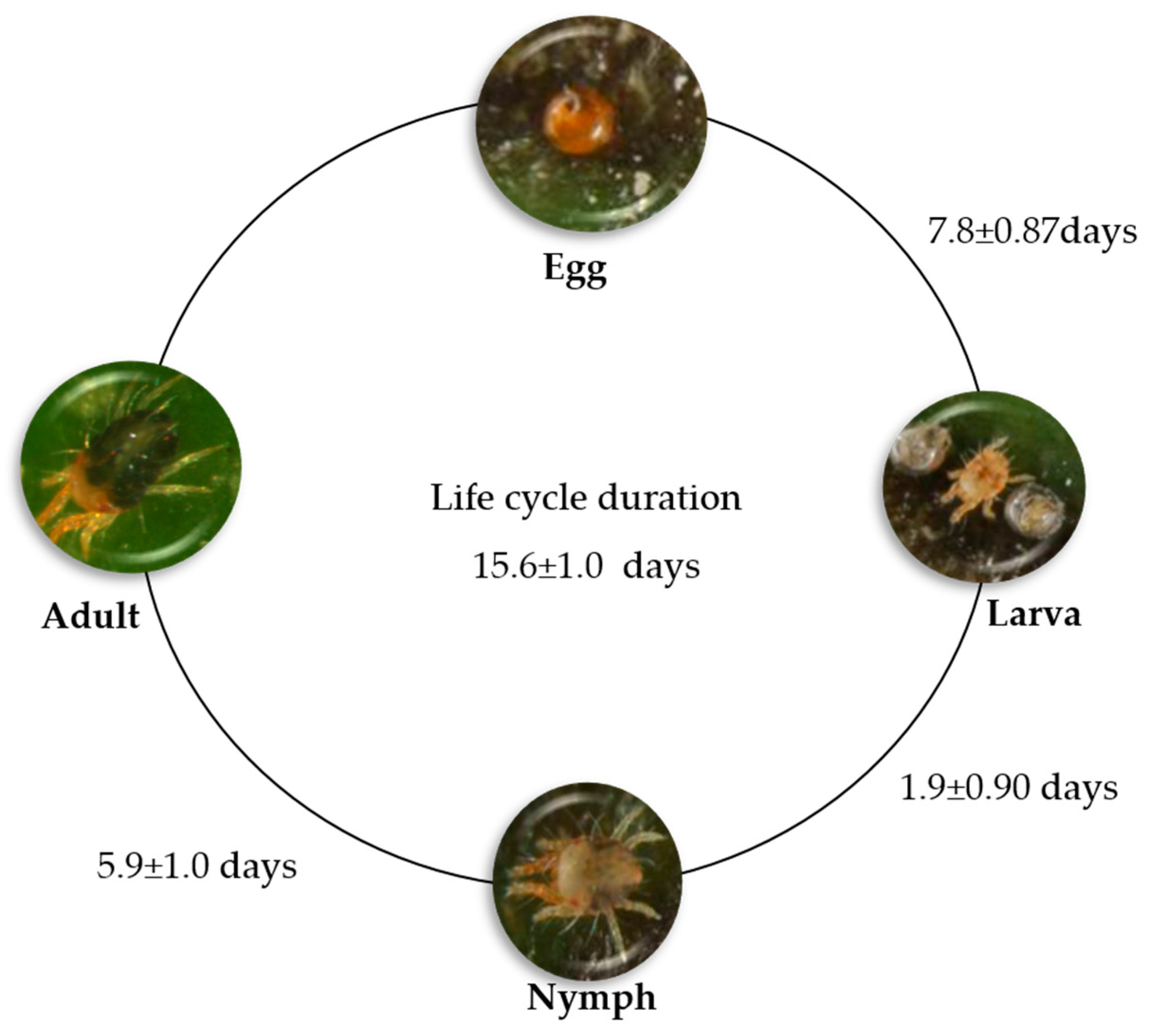
6. The Coffee Red Spider Mite Oligonychus yothersi (McGregor) (Acari: Tetranychidae)
6.1. Mite Biology
6.2. Mite Damage
6.3. Factors Affecting Red Spider Mite Survival
6.4. Effect of Volcanic Ash Deposition on the Coffee Red Spider Mite
6.5. Red Spider Mite Natural Enemies
6.6. Management Recommendations
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cafedecolombia. Available online: https://www.cafedecolombia.com/particulares/historia-del-cafe-de-colombia (accessed on 24 October 2023).
- Bustillo, P.A.E.; Castillo, H.; Villalba, D.; Morales, E.; Vélez, P. Evaluaciones de Campo con el Hongo Beauveria bassiana Para el Control de la Broca del café.; ASIC, 14e. Colloque: San Francisco, CA, USA, 1991; Volume 14, pp. 679–686. [Google Scholar]
- Bustillo, A.E.; Cardenas, R.; Villalba, D.A.; Benavides Machado, P.; Orozco, J.; Posada, F.J. Manejo Integrado de la Broca del Café: Hypothenemus hampei Ferrari en Colombia; Cenicafé: Chinchiná, Colombia, 1998; ISBN 978-958-96554-0-5. [Google Scholar]
- Bustillo, A.E. El Manejo de Cafetales y su Relación con el Control de la Broca del Café en Colombia; Cenicafé: Manizales, Colombia, 2007; pp. 16–21. [Google Scholar]
- Giraldo-Jaramillo, M.; Montoya, E.C.; Sarmiento-Herrera, N.; Quiroga Mosquera, A.; Espinosa-Osorio, J.C.; García-López, J.C.; Duque Orrego, H.; Benavides Machado, P. Vulnerabilidad de la caficultura de Antioquia a la broca del café en diferentes eventos climáticos. Av. Téc. Cenicafé 2019, 507, 1–8. [Google Scholar] [CrossRef]
- Giraldo-Jaramillo, M.; Montoya, E.C.; Sarmiento-Herrera, N.; Quiroga Mosquera, A.; Espinosa-Osorio, J.C.; García-López, J.C.; Duque Orrego, H.; Benavides Machado, P. Vulnerabilidad de la caficultura de Huila a la broca del café en diferentes eventos climáticos. Av. Téc. Cenicafé 2020, 512, 1–8. [Google Scholar] [CrossRef]
- Fetting, C. The European Green Deal; European Sustainable Development Network: Vienna, Austria, 2020; p. 22. [Google Scholar]
- Duque, H.; Baker, P.S. Devouring Profit the Socio-Economics of Coffee Berry Borer IPM; Cenicafé: Cali, Colombia, 2003; pp. 62–63. ISBN 978-958-97218-4-1. [Google Scholar]
- Benavides Machado, P.; Arévalo, H. Manejo integrado: Una estrategia para el control de La broca del café en colombia. Rev. Cenicafé 2002, 53, 39–48. [Google Scholar]
- Constantino, L.M.; Oliveros, C.E.; Benavides Machado, P.; Gómez, J.C.; Serna, C.A.; Ramírez, C.A.; Arcila, A. Recolección de Frutos de Café Del Suelo Con Canastilla: Herramienta Para El Manejo Integrado de La Broca. Av. Téc. Cenicafé 2016, 468, 1–8. [Google Scholar]
- Benavides Machado, P.; Bustillo, A.E.; Montoya, E.C.; Cárdenas, R.; Mejía, C.G. Participación Del Control Cultural, Químico y Biológico En El Manejo de La Broca Del Café. Rev. Colomb. Entomol. 2002, 28, 161–165. [Google Scholar] [CrossRef]
- Benavides, P.; Bustillo, A.; Góngora, C. IPM Program to Control Coffee Berry Borer Hypothenemus Hampei, with Emphasis on Highly Pathogenic Mixed Strains of Beauveria Bassiana, to Overcome Insecticide Resistance in Colombia; INTECH Open Access Publisher: London, UK, 2012; ISBN 978-953-307-780-2. [Google Scholar]
- Vélez, P.E.; Benavides, M. Registro e Identificacion de Beauveria Bassiana en Hypothenemus Hampei en Ancuya, Departamento de Nariño, Colombia; Federacion Nacional de Cafeteros de Colombia: Bogotá, Colombia, 1990. [Google Scholar]
- Jaramillo, J.; Montoya, E.C.; Benavides Machado, P.; Góngora, C.E. Beauveria Bassiana y Metarhizium Anisopliae Para El Control de Broca Del Café En Frutos Del Suelo. Rev. Colomb. Entomol. 2015, 41, 95–104. [Google Scholar]
- Cruz, L.P.; Gaitan, A.L.; Gongora, C.E. Exploiting the genetic diversity of beauveria bassiana for improving the biological control of the coffee berry borer through the use of strain mixtures. Appl. Microbiol. Biotechnol. 2006, 71, 918–926. [Google Scholar] [CrossRef]
- Cárdenas-Ramírez, A.; Villalba-Guott, D.; Bustillo, A.; Montoya, E.C.; Góngora, C. Eficacia de mezclas de cepas del hongo Beauveria bassiana en el control de la broca del café. Rev. Cenicafé 2007, 58, 293–303. [Google Scholar]
- Vera, J.T.; Montoya, E.C.; Benavides, P.; Góngora, C.E. Evaluation of Beauveria bassiana (ascomycota: Hypocreales) as a control of the Coffee Berry Borer Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae) emerging from fallen, infested coffee berries on the ground. Biocontrol Sci. Technol. 2011, 21, 1–14. [Google Scholar] [CrossRef]
- Góngora, B.C.E.; Marín, M.P.; Benavides, M.P. Claves para el éxito del hongo beauveria bassiana como controlador biológico de la broca del café. Av. Téc. No 2009, 384, 2–3. [Google Scholar]
- Milner, R.J.; Lutton, G.G. Metarrhizium anisopliae: Survival of conidia in the soil. In Proceedings of the 1st International Colloquium on Invertebrate Pathology; Queen’s University Press: Kingston Ontario, ON, Canada, 1976; p. 248. [Google Scholar]
- Bustillo, A.E.; Bernal, M.G.; Benavides, P.; Chaves, B. Dynamics of Beauveria bassiana and Metarhizium anisopliae Infecting Hypothenemus hampei (Coleoptera: Scolytidae) Populations Emerging from Fallen Coffee Berries. Fla. Entomol. 1999, 82, 491. [Google Scholar] [CrossRef]
- Lara, G.J.C.; López, N.J.C.; Bustillo, P.A.E. Efecto de entomonemátodos sobre poblaciones de la broca del café, Hypothenemus hampei (Coleoptera: Scolytidae), en frutos en el suelo. Rev. Colomb. Entomol. 2004, 30, 179–185. [Google Scholar] [CrossRef]
- Castro, A.M.; Tapias, J.; Ortiz, A.; Benavides, P.; Góngora, C.E. Identification of Attractant and Repellent Plants to Coffee Berry Borer, Hypothenemus hampei. Entomol. Exp. Appl. 2017, 164, 120–130. [Google Scholar] [CrossRef]
- Góngora, C.E.; Tapias, J.; Jaramillo, J.; Medina, R.; Gonzalez, S.; Casanova, H.; Ortiz, A.; Benavides, P. Evaluation of Terpene-Volatile Compounds Repellent to the Coffee Berry Borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae). J. Chem. Ecol. 2020, 46, 881–890. [Google Scholar] [CrossRef]
- Castro, A.M.; Benavides, P.; Góngora, C.E. Push-Pull Strategy for Handling the Coffee Berry Borer. Trends Entomol. 2018, 14, 51–61. [Google Scholar]
- Benavides, P.; Gil, Z.N.; Escobar, L.E.; Navarro-Escalante, L.; Follett, P.; Diaz-Soltero, H. Pilot Testing of an Area-Wide Biological Control Strategy against the Coffee Berry Borer in Colombia Using African Parasitoids. Insects 2023, 14, 865. [Google Scholar] [CrossRef]
- Cárdenas, R.; Posada, F.J. Los Insectos y Otros Habitantes de Cafetales y Platanales; Cenicafé: Manizales, Colombia, 2001. [Google Scholar]
- Carvalho, J.C.M.; Costa, L.A.A. Mirídeos Neotropicais. CCXCVII: Duas Novas Espécies Do Gênero Monalonion Herrich-Schaffer (Hemiptera). Rev. Bras. Biol. 1988, 48, 893–896. [Google Scholar]
- Ramírez, H.J.; Gil-Palacio, Z.; Benavides Machado, P.; Bustillo, A.E. Estudio de Los Insectos Asociados a Un Nuevo Disturbio En Café Denominado Chamusquina. Rev. Cienc. Agríc. 2007, 24, 65–73. [Google Scholar]
- Laiton-Jiménez, L.A.; López Franco, F.; Montoya, E.C.; Benavides Machado, P. Bases para establecer una alerta temprana de monalonion velezangeli (Hemiptera: Miridae) en café en el huila. Rev. Cenicafé 2020, 71, 7–20. [Google Scholar] [CrossRef]
- Giraldo-Jaramillo, M.; Benavides Machado, P.; Villegas-García, C. Aspectos Morfológicos y Biológicos de Monalonion Velezangeli Carvalho & Costa (Hemiptera: Miridae) En Café. Rev. Cenicafé 2010, 61, 195–205. [Google Scholar]
- Ocampo Flórez, V.; Durán Prieto, J.; Albornoz, M.; Forero, D. New Plant Associations for Monalonion Velezangeli (Hemiptera: Miridae) in Green Urban Areas of Bogotá (Colombia). Acta Biológica Colomb. 2018, 23, 205–208. [Google Scholar] [CrossRef]
- Wheeler, A.J.R. Plant Bugs (Miridae) as Plant Pests. In Heteroptera of Economic Importance; Schaefer, C.W., Panizzi, A.R., Eds.; CRC Press LLC: Dordrecht, The Netherlands, 2000; pp. 37–83. [Google Scholar]
- Benavides Machado, P.; Gil-Palacio, Z.; Constantino, L.M.; Villegas García, C.; Giraldo-Jaramillo, M. Plagas Del Café: Broca, Minador, Cochinillas Harinosas, Arañita Roja y Monalonion. In Manual del Cafetero Colombiano: Investigación y Tecnología para la Sostenibilidad de la Caficultura; Cenicafé: Manizales, Colombia, 2013; Volume 2, pp. 215–260. [Google Scholar]
- Góngora, C.E.; Laiton, J.L.A.; Gil, Z.N.; Benavides, P. Evaluación de Beauveria Bassiana Para El Control de Monalonion Velezangeli (Hemiptera: Miridae) En El Cultivo Del Café. Rev. Colomb. Entomol. 2020, 46, e7685. [Google Scholar] [CrossRef]
- Li, Q.S.; Liu, Q.X.; Deng, W.X. Effects of temperature and humidity on the laboratory population of Adelphocoris lineolatus geoze. Acta Ecol. Sin. 1994, 14, 312–317. [Google Scholar]
- Mani, M.; Shivaraju, C. Economic Importance. In Mealybugs and their Management in Agricultural and Horticultural Crops; Mani, M., Shivaraju, C., Eds.; Springer India: New Delhi, India, 2016; pp. 131–140. ISBN 978-81-322-2675-8. [Google Scholar]
- Mansour, R.; Grissa-Lebdi, K.; Suma, P.; Mazzeo, G.; Russo, A. Key Scale Insects (Hemiptera: Coccoidea) of High Economic Importance in a Mediterranean Area: Host Plants, Bio-Ecological Characteristics, Natural Enemies and Pest Management Strategies—A Review. Plant Prot. Sci. 2017, 53, 1–14. [Google Scholar] [CrossRef]
- Caballero, A.; Ramos-Portilla, A.A.; Suárez-González, D.; Serna, F.; Gil, Z.N.; Benavides, P. Los Insectos Escama (Hemiptera: Coccomorpha) de Raíces de Café (Coffea arabica L.) En Colombia, Con Registros de Hormigas (Hymenoptera: Formicidae) En Asociación. Cienc. Tecnol. Agropecu. 2019, 20, 69–92. [Google Scholar] [CrossRef]
- Caballero, A.; Pacheco da Silva, V.C.; Kaydan, M.B.; Rueda-Ramirez, D.; Kondo, T.; Ramos-Portilla, A.A.; Duarte Gómez, W. First Report of Pseudococcus viburni (Hemiptera: Pseudococcidae) in Colombia: Morphometric and Molecular Analysis, with Notes on Morphological Variation in Specimens from Brazil and Colombia. Acta Zool. 2023, 104, 309–322. [Google Scholar] [CrossRef]
- Gil-Palacio, Z.; Caballero, A.; Ramos, A.A.; Arcila-Moreno, A.; Benavides Machado, P. Diagnóstico de Las Cochinillas de Las Raíces Del Café En Ocho Departamentos Cafeteros de Colombia. Av. Téc. Cenicafé 2021, 524, 1–8. [Google Scholar] [CrossRef]
- Santa-Cecília, L.V.C.; Souza, B.; Prado, E.; Souza, J.C.; Fornazier, M.J. Cochonilhas-Farinhentas Em Cafeeiros: Reconhecimento e Controle CT008. Circ. Téc. 2007, 8, 1–4. [Google Scholar]
- Villegas-García, C.; Peña, H.D.; Muñoz, R.I.; Martinez, H.E.; Benavides Machado, P. Aspectos Del Ciclo de Vida de Puto Barberi Cockerell (Hemiptera: Putoidae). Rev. Cenicafé 2013, 64, 31–41. [Google Scholar]
- Gil-Palacio, Z.; Carmona, L.A.; Navarro, L.; Arcila, A.; Benavides Machado, P. Ciclos de Vida, Descripción de Daños y Comportamiento de Las Cochinillas de Las Raíces Del Café En Colombia. In Proceedings of the Memorias 49 Congreso Socolen, Sociedad Colombiana de Entomología, Bogotá, Colombia, 6 July 2022; pp. 91–93. [Google Scholar]
- Rodrigues-Silva, N.; de Oliveira Campos, S.; de Sá Farias, E.; de Souza, T.C.; Martins, J.C.; Picanço, M.C. Relative Importance of Natural Enemies and Abiotic Factors as Sources of Regulation of Mealybugs (Hemiptera: Pseudococcidae) in Brazilian Coffee Plantations: Identification of Potential Biological Control Agent of the Mealybug. Ann. Appl. Biol. 2017, 171, 303–315. [Google Scholar] [CrossRef]
- Balakrishnan, M.M.; Sreedharan, K.; Venkatesha, M.G.; Bhat, P.K. Observations on Ferrisia virgata (Ckll.) (Homoptera: Pseudococcidae) and Its Natural Enemies on Coffee, with New Records of Predators and Host Plants. J. Coffee Res. India 1991, 21, 11–19. [Google Scholar]
- Gil-Palacio, Z.; Constantino, L.M.; Benavides Machado, P. Enemigos Naturales de Cochinillas Harinosas (Hemiptera: Coccomorpha: Pseudococcidae y Putoidae) Asociados a Las Raíces Del Café En Colombia. Rev. Colomb. Entomol. 2023, 49, 1–16. [Google Scholar] [CrossRef]
- Shylesha, A.N.; Mani, M. Natural Enemies of Mealybugs. In Mealybugs and Their Management in Agricultural and Horticultural Crops; Mani, M., Shivaraju, C., Eds.; Springer India: New Delhi, India, 2016; pp. 149–171. ISBN 978-81-322-2675-8. [Google Scholar]
- Moore, D. Agents Used for Biological Control of Mealybugs (Pseudococcidae). Biocontrol News Inf. 1988, 9, 209–225. [Google Scholar]
- Góngora, C.E.; Gil-Palacio, Z. Control Biológico de Cochinillas de Las Raíces Del Café Con Hongos Entomopatógenos. Rev. Cenicafé 2020, 71, 53–65. [Google Scholar] [CrossRef]
- Gil Palacio, Z.; Arcila-Moreno, A.; Márquez, J.S.; Ospina-Penagos, C.; Mejía, S.E.; Benavides Machado, P. Validación Del Manejo de Neochavesia Caldasiae (Balachowsky, 1957) Hemiptera: Rhizoecidae En Fredonia, Antioquia. Rev. Cenicafé 2022, 73, e73106. [Google Scholar] [CrossRef]
- Villegas-García, C.; Zabala, G.A.; Ramos, A.A.; Benavides Machado, P. Identificación y hábitos de cochinillas harinosas asociadas a raíces del café en Quindío. Rev. Cenicafé 2009, 60, 362–373. [Google Scholar]
- Raghoonundon, B.; Raspé, O.; Thongklang, N.; Hyde, K.D. Phlebopus (Boletales, Boletinellaceae), a Peculiar Bolete Genus with Widely Consumed Edible Species and Potential for Economic Development in Tropical Countries. Food Biosci. 2021, 41, 100962. [Google Scholar] [CrossRef]
- Infante, F.; Armbrecht, I.; Constantino, L.M.; Benavides, P. Coffee Pests. In Forest Microbiology; Elsevier: Amsterdam, The Netherlands, 2023; Volume 3, pp. 213–225. ISBN 978-0-443-18694-3. [Google Scholar]
- Bustillo, A.E. El minador de la hoja del cafeto, Leucoptera coffeellum (Lepidoptera: Lyonetiidae). In Los Insectos y su Manejo en la Caficultura Colombiana; Cenicafé: Manizales, Colombia, 2008; pp. 360–363. ISBN 97895898193. [Google Scholar]
- Souza, J.C.; Reis, P.R.; Rigitano, R.L.O. Bicho Mineiro Do Cafeeiro: Biologia, Danos e Manejo Integrado. Bol. Téc. EPAMIG 1998, 54, 1–48. [Google Scholar]
- Nantes, J.F.D.; Parra, J.R.P. Avaliação de Danos Causados Por Perileucoptera coffeella (Guérin—Méneville, 1842) (Lepidoptera-Lyonetiidae), Em Três Variedades de Café (Coffea Spp.). Solo 1977, 69, 26–29. [Google Scholar]
- Paliz, V.; Mendoza, J. Plagas Del Cafeto. In Manual del Cultivo del Café; Sotomayor, H.I., Ed.; INIAP: Quevedo, Ecuador, 1993; pp. 144–166. [Google Scholar]
- Ramírez, V.H.; Gaitán, A.L.; Benavides Machado, P.; Constantino, L.M.; Gil, Z.N.; Sadeghian, S.; González, H. Recomendaciones Para La Reducción Del Riesgo En La Caficultura de Colombia Ante Un Eventoclimático de El Niño. Av. Téc. Cenicafé 2014, 1–12. [Google Scholar]
- Parra, J.R.P. Biologia Comparada de Perileucoptera coffeella (Guérin-Menéville, 1842) (Lepidoptera, Lyonetiidae) Visando Ao Seu Zoneamento Ecológico No Estado de São. Rev. Bras. Entomol. 1985, 29, 45–76. [Google Scholar]
- Lomelí-Flores, J.R.; Barrera, J.F.; Bernal, J.S. Impacts of Weather, Shade Cover and Elevation on Coffee Leafminer Leucoptera coffeella (Lepidoptera: Lyonetiidae) Population Dynamics and Natural Enemies. Crop Prot. 2010, 29, 1039–1048. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nichols, C.I. Biodiversidad y Manejo de Plagas. Perspectivas Agroecológicas; Icaria Editorial S.A.: Barcelona, Spain, 2007; ISBN 978-84-7426-764-8. [Google Scholar]
- Waller, J.M.; Bigger, M.; Hillocks, R.J. Coffee Pests, Diseases and Their Management.; CABI: Wallingford, UK, 2007. [Google Scholar]
- Constantino, L.M.; Flórez, J.C.; Benavides, P.; Bacca, T. Minador de Las Hojas Del Cafeto: Una Plaga Potencial Por Efectos Delcambio Climático. Av. Téc. Cenicafé 2011, 1–12. [Google Scholar] [CrossRef]
- David-Rueda, G.; Constantino, C.L.M.; Montoya, E.C.; Ortega, M.O.E.; Gil, Z.N.; Benavides-Machado, P. Diagnóstico de Leucoptera coffeella (Lepidoptera: Lyonetiidae) y Sus Parasitoides En El Departamento de Antioquia, Colombia. Rev. Colomb. Entomol. 2016, 42, 4–11. [Google Scholar] [CrossRef]
- Francke, W.; Tóth, M.; Szöcs, G.; Krieg, W.; Ernest, H.; Buschmann, E. Identifizierung and Synthese von Dimethylalkanen Als Sexuallockstoffe Weiblicher Miniermotten (Lyonetiidae). Z. Für Naturforsch. C 1988, 43, 787–789. [Google Scholar] [CrossRef]
- Bacca, T.; Lima, E.R.; Picanco, M.C.; Guedes, R.N.C.; Viana, J.H.M. Optimum Spacing of Pheromone Traps for Monitoring the Coffee Leaf Miner Leucoptera coffeella. Entomol. Exp. Appl. 2006, 119, 39–45. [Google Scholar] [CrossRef]
- Ambrogi, B.; Lima, E.; Sousa-Souto, L. Efficacy of Mating Disruption for Control of the Coffee Leaf Miner. BioAssay 2006, 1, 1–5. [Google Scholar] [CrossRef]
- Guerreiro, F.O. Coffee Leaf Miner Resistance. Braz. J. Plant Physiol. 2006, 18, 109–117. [Google Scholar] [CrossRef]
- Perthuis, B.; Pradon, J.L.; Montagnon, C.; Dufour, M.; Leroy, T. Stable Resistance against the Leaf Miner Leucoptera coffeella Expressed by Genetically Transformed Coffea Canephora in a Pluriannual Field Experiment in French Guiana. Euphytica 2005, 144, 321–329. [Google Scholar] [CrossRef]
- Orozco, H.J.; Duque, M.C.; Mesa, N.C. Efecto de La Temperatura Sobre La Tabla de Vida de Oligonychus Yothersi En Coffea Arabica. Rev. Cenicafé 1990, 41, 5–18. [Google Scholar]
- Giraldo-Jaramillo, M.; Galindo Leva, L.A.; Benavides Machado Pablo, P.; Forero, D. Aprenda a conocer las chinches depredadoras de plagas del café. Av. Técnicos Cenicafé. 2011, 412, 1–8. [Google Scholar] [CrossRef]
- Wille, A.; Fuentes, G. Efecto de la ceniza del Volcán: Irazú (Costa Rica) en algunos insectos. Rev. Biol. Trop. 1975, 23, 165–175. [Google Scholar] [PubMed]
- Gil-Palacio, Z.N.; Constantino, L.M.; Martínez, H.; Benavides, P. Aprenda a Manejar La Arañita Roja Del Café. Avances Técnicos Cenicafé. Av. Téc. Cenicafé 2013, 436, 1–8. [Google Scholar]
- Huerta, G. Como Afecta La Ceniza Volcánica a Las Abejas y a La Actividad Apicola. Presencia 2011, 54, 27–29. [Google Scholar]
- Rojas Barrantes, M.; Ramírez Valerio, D.; Constantino, L.M. Brote de Oligonychus Yothersi En Cafetales de Costa Rica Por La Ceniza Del Volcán Poás Durante El Año 2017. Agron. Costarric. 2020, 44, 113–122. [Google Scholar] [CrossRef]
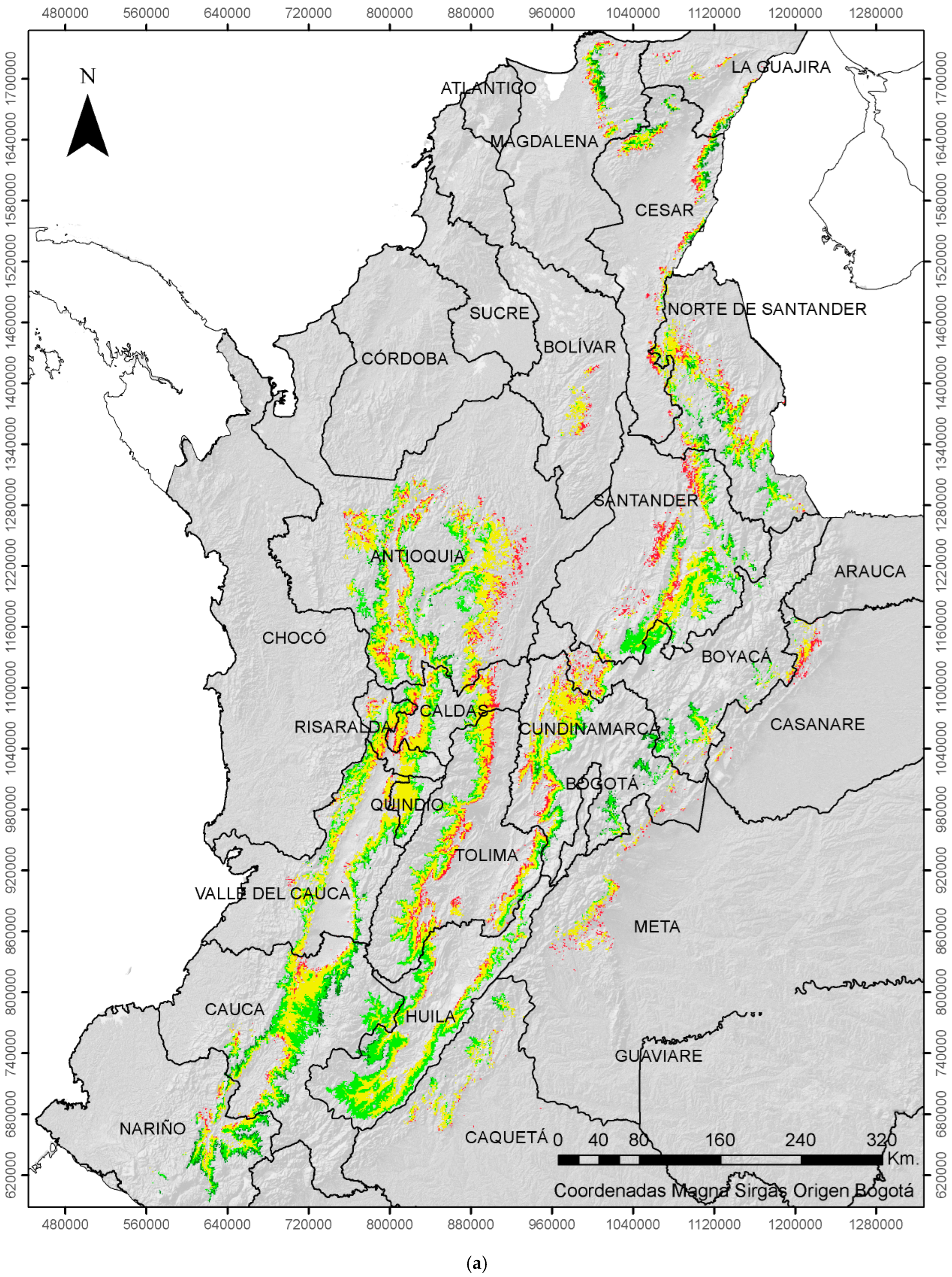
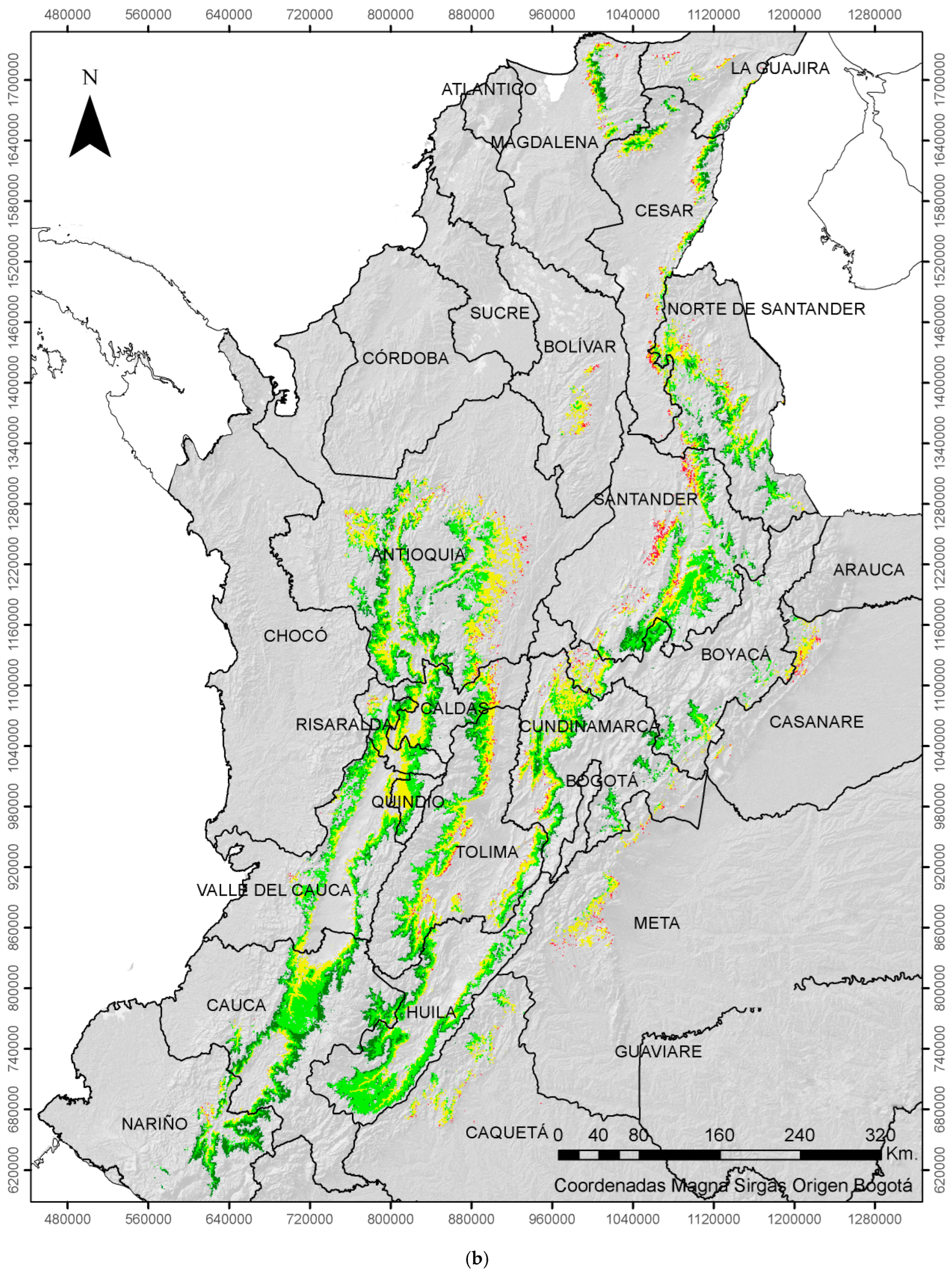
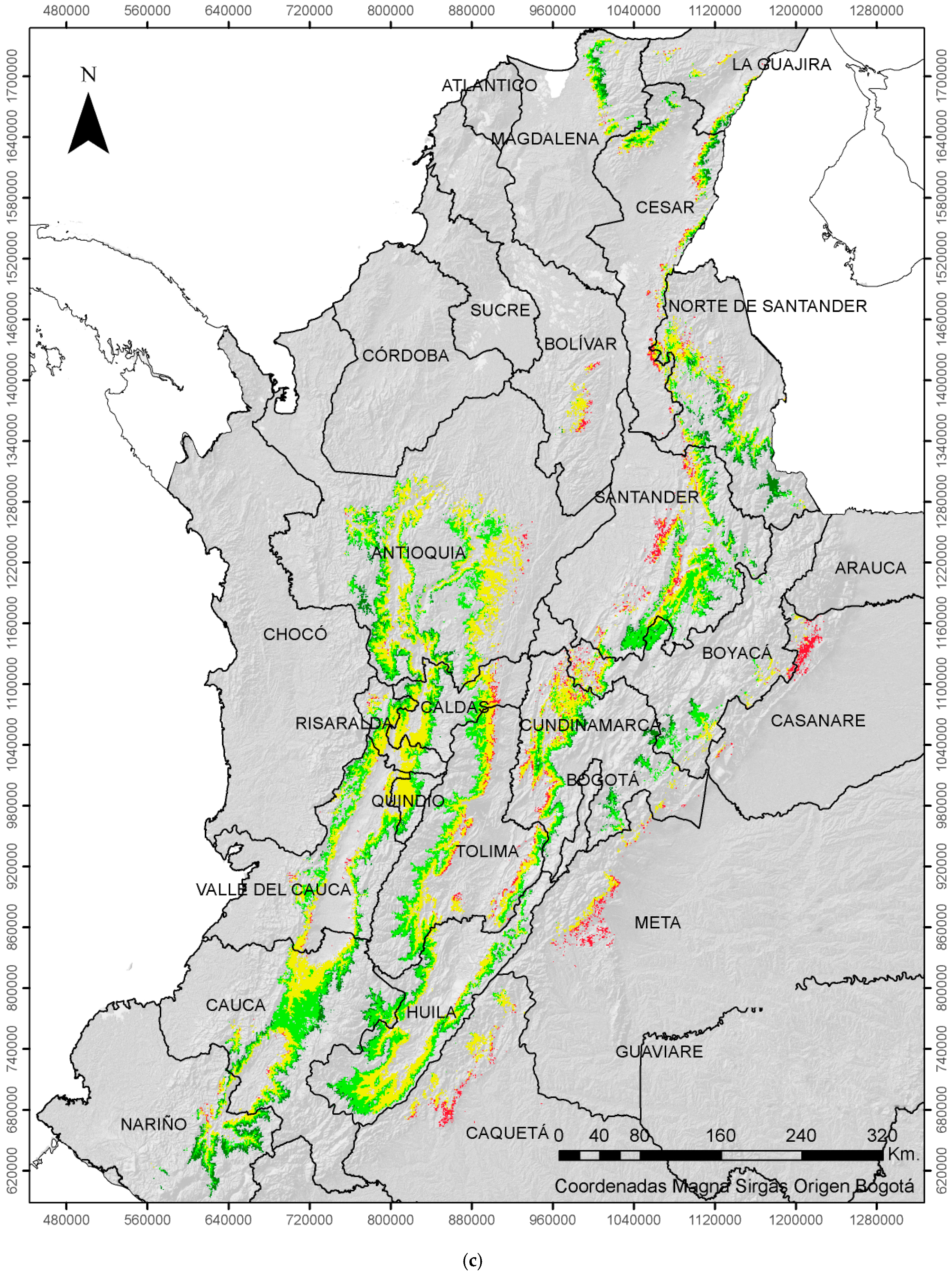
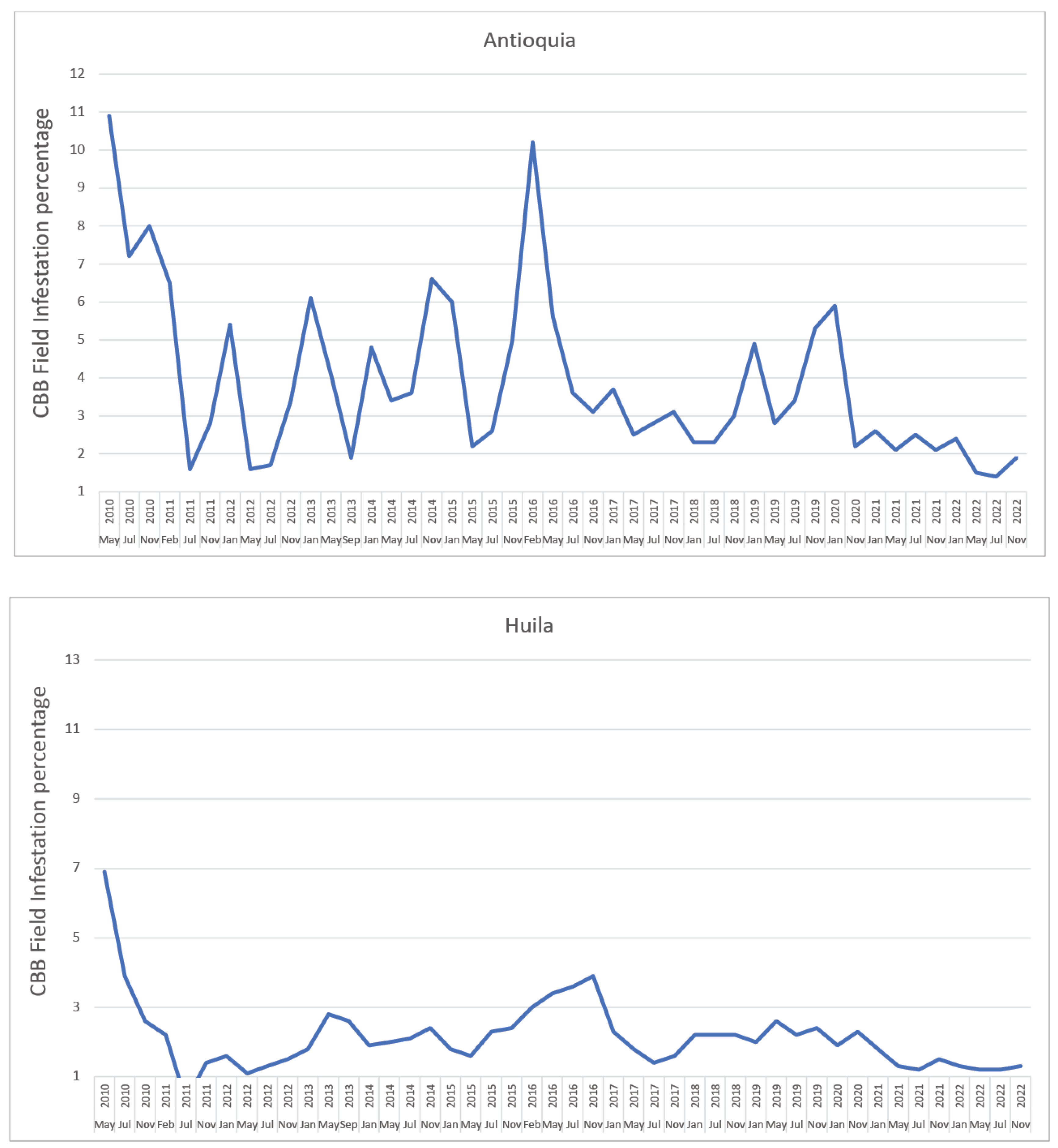




| ENSO | Coffee Growing Area (%) | |||
|---|---|---|---|---|
| EVENT | Very Low | Low | Moderate | High |
| Neutral | 11 | 40 | 43 | 6 |
| La Niña | 20 | 45 | 32 | 3 |
| El Niño | 6 | 34 | 49 | 11 |
| ENSO | Coffee Growing Area (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| EVENT | Very Low | Low | Moderate | High | ||||
| Locality | Antioquia | Huila | Antioquia | Huila | Antioquia | Huila | Antioquia | Huila |
| Neutral | 9.1 | 7.8 | 39.0 | 49.9 | 50.9 | 42.1 | 1.0 | 0.2 |
| La Niña | 17.7 | 20.8 | 41.1 | 65.0 | 38.9 | 14.1 | 2.3 | 0.1 |
| El Niño | 4.0 | 2.1 | 28.7 | 57.0 | 55.3 | 39.0 | 12.0 | 1.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Góngora, C.E.; Gil, Z.N.; Constantino, L.M.; Benavides, P. Sustainable Strategies for the Control of Pests in Coffee Crops. Agronomy 2023, 13, 2940. https://doi.org/10.3390/agronomy13122940
Góngora CE, Gil ZN, Constantino LM, Benavides P. Sustainable Strategies for the Control of Pests in Coffee Crops. Agronomy. 2023; 13(12):2940. https://doi.org/10.3390/agronomy13122940
Chicago/Turabian StyleGóngora, Carmenza E., Zulma Nancy Gil, Luis Miguel Constantino, and Pablo Benavides. 2023. "Sustainable Strategies for the Control of Pests in Coffee Crops" Agronomy 13, no. 12: 2940. https://doi.org/10.3390/agronomy13122940
APA StyleGóngora, C. E., Gil, Z. N., Constantino, L. M., & Benavides, P. (2023). Sustainable Strategies for the Control of Pests in Coffee Crops. Agronomy, 13(12), 2940. https://doi.org/10.3390/agronomy13122940








