Evaluation of Soil Suppressiveness of Various Japanese Soils against the Soybean Cyst Nematode Heterodera glycines and Its Relation with the Soil Chemical and Biological Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Sample Collection
2.2. Comparison of a Growth Level of Soybean Cyst Nematodes among 15 Soils
2.2.1. DNA Extraction from Soils
2.2.2. Real-Time PCR
2.3. Soil Physicochemical and Biological Properties
2.4. Effect of the Addition of Streptomycin on the Growth of Soybean Cyst Nematode in F-Soil That Showed the Highest Suppression
2.5. Isolation and Screening of Fungi from F-Soil
2.5.1. Isolation
2.5.2. Screening
2.5.3. Phylogeny
DNA Extraction from Fungi
Sequencing
2.6. Effect of Fungal Strains on Soybean Cyst Nematode J2 and Hatching
2.6.1. Effects of Fungal Strains on J2
2.6.2. Effects on Soybean Cyst Nematode Hatching
2.7. Statistical Analysis
3. Results
3.1. Evaluate the Suppression Potential of Different Soils on Soybean Cyst Nematodes
3.2. Effect of Soil Physicochemical and Biological Properties on the Growth of Soybean Cyst Nematodes
3.3. Effect of the Addition of Streptomycin on the Growth of Soybean Cyst Nematodes
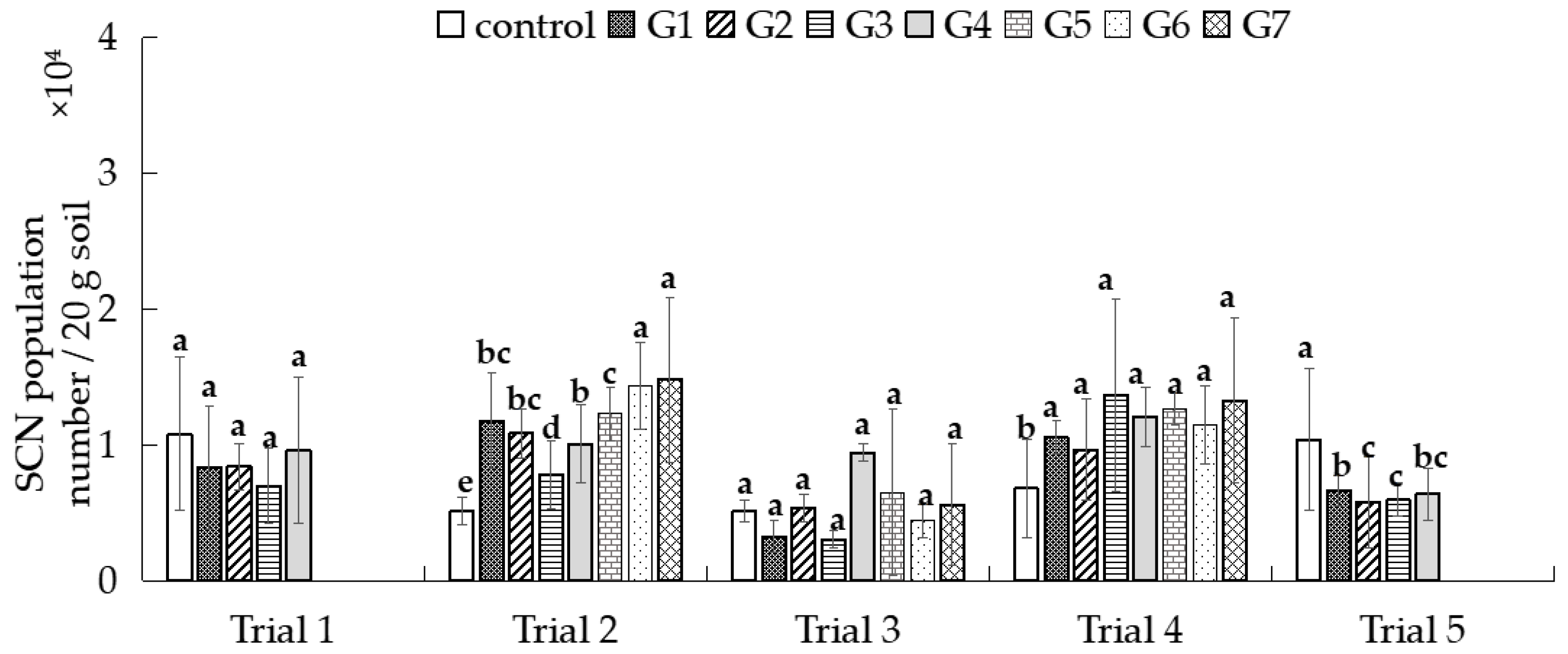
3.4. Evaluate the Suppression Potential of Different Fungal Strains on Soybean Cyst Nematodes
3.5. The Effect of Suppressive Fungi on Second-Stage Juveniles (J2)
3.6. The Effect of Suppressive Fungi on Hatching of Soybean Cyst Nematode
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shiferaw, B.; Smale, M.; Braun, H.J.; Duveiller, E.; Reynolds, M.; Muricho, G. Crops that feed the world 10. Past successes and future challenges to the role played by wheat in global food security. Food Secur. 2013, 5, 291–317. [Google Scholar] [CrossRef]
- Exposito, R.G.; de Bruijn, I.; Postma, J.; Raaijmakers, J.M. Current insights into the role of rhizosphere bacteria in disease suppressive soils. Front. Microbiol. 2017, 8, 1–7. [Google Scholar]
- Noble, R.; Coventry, E. Suppression of soil-borne plant diseases with composts: A review. Biocontrol Sci. Technol. 2005, 15, 3–20. [Google Scholar] [CrossRef]
- Anwar, S.A.; McKenry, M.V. Incidence and population density of plant-parasitic nematodes infecting vegetable crops and associated yield losses in Punjab, Pakistan. Pak. J. Zool. 2012, 44, 327–333. [Google Scholar]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Pires, R.D.L.; Goncalves, D.N.; Orue, J.P.M.; Kanashiro, W.E.S.; Rodrigues, J.F.; Machado, B.B.; Goncalves, W.N. Local descriptors for soybean disease recognition. Comput. Electron. Agric. 2016, 125, 48–55. [Google Scholar] [CrossRef]
- Lerch-Olson, E.R.; Robertson, A.E. Effect of co-inoculations with Pythium and Fusarium species on seedling disease development of soybean. Can. J. Plant Pathol. 2020, 42, 408–418. [Google Scholar] [CrossRef]
- Clevinger, E.M.; Biyashev, R.; Lerch-Olson, E.; Yu, H.P.; Quigley, C.; Song, Q.J.; Dorrance, A.E.; Robertson, A.E.; Maroof, M.A.S. Identification of quantitative disease resistance loci toward four Pythium species in soybean. Front. Plant Sci. 2021, 12, 2–13. [Google Scholar] [CrossRef]
- Arjoune, Y.; Sugunaraj, N.; Peri, S.; Nair, S.V.; Skurdal, A.; Ranganathan, P.; Johnson, B. Soybean cyst nematode detection and management: A review. Plant Methods 2022, 18, 6–12. [Google Scholar] [CrossRef]
- Fox, C.M.; Kim, K.S.; Cregan, P.B.; Hill, C.B.; Hartman, G.L.; Diers, B.W. Inheritance of soybean aphid resistance in 21 soybean plant introductions. Theor. Appl. Genet. 2014, 127, 43–50. [Google Scholar] [CrossRef]
- Hu, W.M.; Strom, N.B.; Haarith, D.; Chen, S.Y.; Bushley, K.E. Seasonal variation and crop sequences shape the structure of bacterial communities in cysts of soybean cyst nematode. Front. Microbiol. 2019, 10, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.H.; Thurlow, D.L.; Eason, J.T. Influence of tillage and crop-rotation on yields of corn, soybean, and wheat. Agron. J. 1988, 80, 76–80. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M.; Askary, T.H. Fungal and bacterial nematicides in integrated nematode management strategies. Egypt. J. Biol. Pest Control 2018, 28, 7–18. [Google Scholar] [CrossRef]
- Hajji-Hedfi, L.; Hlaoua, W.; Al-Judaibi, A.A.; Rhouma, A.; Horrigue-Raouani, N.; Abdel-Azeem, A.M. Comparative effectiveness of filamentous fungi in biocontrol of Meloidogyne javanica and activated defense mechanisms on tomato. J. Fungi 2023, 9, 37. [Google Scholar] [CrossRef] [PubMed]
- Toyota, K.; Ritz, K.; Kuninaga, S.; Kimura, M. Impact of fumigation with metam sodium upon soil microbial community structure in two Japanese soils. Soil Sci. Plant Nutr. 1999, 45, 207–223. [Google Scholar] [CrossRef]
- Deacon, J.W. The nature and practice of biological control of plant pathogens. Nature 1984, 309, 732. [Google Scholar] [CrossRef]
- Becker, D.M.; Kinkel, L.L.; Schottel, J.L. Evidence for interspecies communication and its potential role in pathogen suppression in a naturally occurring disease suppressive soil. Can. J. Microbiol. 1997, 43, 985–990. [Google Scholar] [CrossRef]
- Young, C.C.; Cheng, K.T.; Waller, G.R. Phenolic-compounds in conducive and suppressive soils on clubroot disease of crucifers. Soil Biol. Biochem. 1991, 23, 1183–1189. [Google Scholar] [CrossRef]
- Gine, A.; Carrasquilla, M.; Martinez-Alonso, M.; Gaju, N.; Sorribas, F.J. Characterization of soil suppressiveness to root-knot nematodes in organic horticulture in plastic greenhouse. Front. Plant Sci. 2016, 7, 4–8. [Google Scholar] [CrossRef]
- AzconAguilar, C.; Barea, J.M. Arbuscular mycorrhizas and biological control of soil-borne plant pathogens—An overview of the mechanisms involved. Mycorrhiza 1996, 6, 457–464. [Google Scholar]
- Powlson, D.S.; Gregory, P.J.; Whalley, W.R.; Quinton, J.N.; Hopkins, D.W.; Whitmore, A.P.; Hirsch, P.R.; Goulding, K.W.T. Soil management in relation to sustainable agriculture and ecosystem services. Food Policy 2011, 36, S72–S87. [Google Scholar] [CrossRef]
- Seong, J.; Shin, J.; Kim, K.; Cho, B.K. Microbial production of nematicidal agents for controlling plant-parasitic nematodes. Process Biochem. 2021, 108, 69–79. [Google Scholar] [CrossRef]
- Lu, W.W.; Zha, Q.Z.; Zhang, H.L.; Chen, H.Y.H.; Yu, J.; Tu, F.; Ruan, H.H. Changes in soil microbial communities and priming effects induced by rice straw pyrogenic organic matter produced at two temperatures. Geoderma 2021, 400, 2–10. [Google Scholar] [CrossRef]
- Nour, S.M.; Lawrence, J.R.; Zhu, H.; Swerhone, G.D.W.; Welsh, M.; Welacky, T.W.; Topp, E. Bacteria associated with cysts of the soybean cyst nematode (Heterodera glycines). Appl. Environ. Microbiol. 2003, 69, 607–615. [Google Scholar] [CrossRef]
- Hu, W.M.; Chen, S.Y.; Liu, X.Z. Effect of temperature treatment on survival of Heterodera glycines and its associated fungi and bacteria. Nematology 2016, 18, 845–855. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Borneman, J.; Becker, J.O. Identifying microorganisms involved in specific pathogen suppression in soil. Annu. Rev. Phytopathol. 2007, 45, 153–172. [Google Scholar] [PubMed]
- Richardson, A.E.; Simpson, R.J. Soil microorganisms mediating phosphorus availability. Plant Physiol. 2011, 156, 989–996. [Google Scholar] [CrossRef]
- Weller, D.M.; Raaijmakers, J.M.; Gardener, B.B.M.; Thomashow, L.S. Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu. Rev. Phytopathol. 2002, 40, 309–318. [Google Scholar] [CrossRef]
- Lai, H.L.; Gao, F.Y.; Su, H.; Zheng, P.; Li, Y.Y.; Yao, H.Y. Nitrogen distribution and soil microbial community characteristics in a legume-cereal intercropping system: A review. Agronomy 2022, 12, 1900. [Google Scholar] [CrossRef]
- Ma, R.; Liu, X.Z.; Jian, H.; Li, S.D. Detection of Hirsutella spp. and Pasteuria sp. parasitizing second-stage juveniles of Heterodera glycines in soybean fields in China. Biol. Control 2005, 33, 223–229. [Google Scholar] [CrossRef]
- Hu, W.M.; Samac, D.A.; Liu, X.Z.; Chen, S.Y. Microbial communities in the cysts of soybean cyst nematode affected by tillage and biocide in a suppressive soil. Appl. Soil Ecol. 2017, 119, 396–406. [Google Scholar] [CrossRef]
- Chen, S.Y.; Liu, X.Z. Control of the soybean cyst nematode by the fungi Hirsutella rhossiliensis and Hirsutella minnesotensis in greenhouse studies. Biol. Control 2005, 32, 208–219. [Google Scholar] [CrossRef]
- Siddiqui, Z.A.; Mahmood, I. Role of bacteria in the management of plant parasitic nematodes: A review. Bioresour. Technol. 1999, 69, 167–179. [Google Scholar] [CrossRef]
- Goto, K.; Min, Y.Y.; Sato, E.; Toyota, K. A multiplex real-time PCR assay for the simultaneous quantification of the major plant-parasitic nematodes in Japan. Nematology 2011, 13, 713–720. [Google Scholar]
- Shirai, S.; Toyota, K. Optimisation of a species-specific primer set to quantify the soybean cyst nematode, Heterodera glycines, in soil using real-time PCR. Nematology 2019, 21, 1037–1042. [Google Scholar] [CrossRef]
- Brown, J.H.; Vaz, J.E.; Benzo, Z.; Mejias, C. A comparison of extraction and suspension methods for determining exchangeable potassium in soils. Appl. Clay Sci. 1999, 14, 245–255. [Google Scholar] [CrossRef]
- Tran, T.S.; Giroux, M.; Guilbeault, J.; Audesse, P. Evaluation of Mehlich-iii extractant to estimate the available P in Quebec soils. Commun. Soil Sci. Plant Anal. 1990, 21, 1–28. [Google Scholar] [CrossRef]
- Wang, Y.X.; Chikamatsu, S.; Gegen, T.; Sawada, K.; Toyota, K.; Riya, S.; Hosomi, M. Application of biogas digestate with rice straw mitigates nitrate leaching potential and suppresses root-knot nematode (Meloidogyne incognita). Agronomy 2019, 9, 227. [Google Scholar] [CrossRef]
- Nguyen, S.V.; Nguyen, P.K.T.; Araki, M.; Perry, R.N.; Tran, L.B.; Chau, K.M.; Min, Y.Y.; Toyota, K. Seasonal variation and vertical distribution of nematode communities and their relation to soil chemical property and rice productivity under triple rice cultivation in the Mekong delta, Vietnam. Nematology 2021, 23, 33–46. [Google Scholar] [CrossRef]
- Martin, J.P. Citation Classic—Use of acid, rose-bengal, and streptomycin in the plate method for estimating soil fungi. Curr. Contents/Agric. Biol. Environ. Sci. 1980, 20, 1–10. [Google Scholar]
- Zhou, G.; Whong, W.Z.; Ong, T.; Chen, B. Development of a fungus-specific PCR assay for detecting low-level fungi in an indoor environment. Mol. Cell. Probes 2000, 14, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Warnke, S.A.; Chen, S.Y.; Wyse, D.L.; Johnson, G.A.; Porter, P.M. Effect of rotation crops on hatch, viability and development of Heterodera glycines. Nematology 2008, 10, 869–882. [Google Scholar]
- Min, Y.Y.; Toyota, K.; Sato, E. A novel nematode diagnostic method using the direct quantification of major plant-parasitic nematodes in soil by real-time PCR. Nematology 2012, 14, 265–276. [Google Scholar] [CrossRef]
- Tolba, S.; Egan, S.; Kallifidas, D.; Wellington, E.M.H. Distribution of streptomycin resistance and biosynthesis genes in streptomycetes recovered from different soil sites. FEMS Microbiol. Ecol. 2002, 42, 269–276. [Google Scholar] [CrossRef][Green Version]
- Meyer, S.L.F.; Roberts, D.P. Combinations of biocontrol agents for management of plant-parasitic nematodes and soilborne plant-pathogenic fungi. J. Nematol. 2002, 34, 1–8. [Google Scholar]
- Agusti-Brisach, C.; Cabral, A.; Gonzalez-Dominguez, E.; Perez-Sierra, A.; Leon, M.; Abad-Campos, P.; Garcia-Jimenez, J.; Oliveira, H.; Armengol, J. Characterization of Cylindrodendrum, Dactylonectria and Ilyonectria isolates associated with loquat decline in Spain, with description of Cylindrodendrum alicantinum sp nov. Eur. J. Plant Pathol. 2016, 145, 103–118. [Google Scholar] [CrossRef]
- Iqbal, M.; Dubey, M.; McEwan, K.; Menzel, U.; Franko, M.A.; Viketoft, M.; Jensen, D.F.; Karlsson, M. Evaluation of Clonostachys rosea for control of plant- parasitic nematodes in soil and in roots of carrot and wheat. Phytopathology 2018, 108, 52–59. [Google Scholar] [CrossRef]
- Zuckerman, B.M.; Dicklor, M.B.; Matheny, M.; Acosta, N. Control of plant-parasitic nematodes by a nematicidal strain of Aspergillus niger. J. Chem. Ecol. 1994, 20, 33–43. [Google Scholar] [CrossRef]
- Arastehfar, A.; Carvalho, A.; Houbraken, J.; Lombardi, L.; Garcia-Rubio, R.; Jenks, J.D.; Rivero-Menendez, O.; Aljohani, R.; Jacobsen, I.D.; Berman, J.; et al. Aspergillus fumigatus and aspergillosis: From basics to clinics. Stud. Mycol. 2021, 100, 10–35. [Google Scholar] [CrossRef]
- Garbeva, P.; van Veen, J.A.; van Elsas, J.D. Assessment of the diversity, and antagonism towards Rhizoctonia solani AG3, of pseudomonas species in soil from different agricultural regimes. FEMS Microbiol. Ecol. 2004, 47, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Piombo, E.; Vetukuri, R.R.; Sundararajan, P.; Kushwaha, S.; Jensen, D.F.; Karlsson, M.; Dubey, M. Comparative small RNA and degradome sequencing provides insights into antagonistic interactions in the biocontrol fungus Clonostachys rosea. Appl. Environ. Microbiol. 2022, 88, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Cristobal-Alejo, J.; Diaz-Braga, A.; Herrera-Parra, E.; Heredia, G.; Medina-Baizabal, I.L.; Canto-Canche, B.; Tzec-Sima, M.; Gamboa-Angulo, M. Clonostachys rosea selected by nematicidal screening and its efficacy against Meloidogyne incognita in a greenhouse. Biocontrol Sci. Technol. 2021, 31, 1283–1297. [Google Scholar] [CrossRef]
- Cai, L.N.; Xu, S.N.; Lu, T.; Lin, D.Q.; Yao, S.J. Salt-tolerant and thermostable mechanisms of an endoglucanase from marine Aspergillus niger. Bioresour. Bioprocess. 2022, 9, 8–13. [Google Scholar] [CrossRef]
- Joubert, D.A.; Slaughter, A.R.; Kemp, G.; Becker, J.V.W.; Krooshof, G.H.; Bergmann, C.; Benen, J.; Pretorius, I.S.; Vivier, M.A. The grapevine polygalacturonase-inhibiting protein (VvPGIP1) reduces Botrytis cinerea susceptibility in transgenic tobacco and differentially inhibits fungal polygalacturonases. Transgenic Res. 2006, 15, 687–702. [Google Scholar] [CrossRef]
- Faraji, F.; Golmohammadzadeh, R.; Rashchi, F.; Alimardani, N. Fungal bioleaching of WPCBs using Aspergillus niger: Observation, optimization and kinetics. J. Environ. Manag. 2018, 217, 775–787. [Google Scholar] [CrossRef]
- Yeon, J.; Park, A.R.; Kang, M.; Nguyen, V.T.; Lee, Y.; Kim, H.M.; Park, H.W.; Ha, P.; Koo, Y.; Kim, J.C. Control of root-knot nematodes on tomato by eliciting resistance through Aspergillus niger-derived oxalic acid. J. Pest Sci. 2022, 96, 1287–1297. [Google Scholar] [CrossRef]
- Shende, R.; Wong, S.S.W.; Meitei, H.T.; Lal, G.; Madan, T.; Aimanianda, V.; Pal, J.K.; Sahu, A. Protective role of host complement system in Aspergillus fumigatus infection. Front. Immunol. 2022, 13, 5–11. [Google Scholar] [CrossRef]
- Brito, V.D.; Achimon, F.; Zunino, M.P.; Zygadlo, J.A.; Pizzolitto, R.P. Fungal diversity and mycotoxins detected in maize stored in silo-bags: A review. J. Sci. Food Agric. 2022, 102, 2640–2650. [Google Scholar] [CrossRef]
- Kong, L.A.; Shi, X.; Chen, D.; Yang, N.; Yin, C.F.; Yang, J.; Wang, G.F.; Huang, W.K.; Peng, H.; Peng, D.L.; et al. Host-induced silencing of a nematode chitin synthase gene enhances resistance of soybeans to both pathogenic Heterodera glycines and Fusarium oxysporum. Plant Biotechnol. J. 2022, 20, 809–811. [Google Scholar] [CrossRef]
- Di, X.T.; Takken, F.L.W.; Tintor, N. How phytohormones shape interactions between plants and the soil-borne fungus Fusarium oxysporum. Front. Plant Sci. 2016, 7, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Bowers, J.H.; Locke, J.C. Effect of botanical extracts on the population density of Fusarium oxysporum in soil and control of Fusarium wilt in the greenhouse. Plant Dis. 2000, 84, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, L.M.; Smith, L.J.; Aitken, E.A.B. Identification and characterization of non-pathogenic Fusarium oxysporum capable of increasing and decreasing Fusarium wilt severity. Mycol. Res. 2006, 110, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Bärenstrauch, M.; Mann, S.; Jacquemin, C.; Bibi, S.; Sylla, O.K.; Baudouin, E.; Buisson, D.; Prado, S.; Kunz, C. Molecular crosstalk between the endophyte Paraconiothyrium variabile and the phytopathogen Fusarium oxysporum—Modulation of lipoxygenase activity and beauvericin production during the interaction. Fungal Genet. Biol. 2020, 139, 7–13. [Google Scholar] [CrossRef]
- Beccaccioli, M.; Pucci, N.; Salustri, M.; Scortichini, M.; Zaccaria, M.; Momeni, B.; Loreti, S.; Reverberi, M.; Scala, V. Fungal and bacterial oxylipins are signals for intra- and inter-cellular communication within plant disease. Front. Plant Sci. 2022, 13, 2–12. [Google Scholar] [CrossRef]
- Lee, H.S.; Kang, J.W.; Kim, B.H.; Park, S.G.; Lee, C. Statistical optimization of culture conditions for the production of enniatins H, I, and MK1688 by Fusarium oxysporum KFCC 11363P. J. Biosci. Bioeng. 2011, 111, 279–285. [Google Scholar] [CrossRef]
- Larkin, R.P.; Hopkins, D.L.; Martin, F.N. Suppression of Fusarium wilt of watermelon by nonpathogenic Fusarium oxysporum and other microorganisms recovered from a disease-suppressive soil. Phytopathology 1996, 86, 812–819. [Google Scholar] [CrossRef]
- Goettel, M.S.; Koike, M.; Kim, J.J.; Aiuchi, D.; Shinya, R.; Brodeur, J. Potential of Lecanicillium spp. for management of insects, nematodes and plant diseases. J. Invertebr. Pathol. 2008, 98, 256–261. [Google Scholar] [CrossRef]
- Chen, S.Y.; Chen, F.J. Fungal parasitism of Heterodera glycines eggs as influenced by egg age and pre-colonization of cysts by other fungi. J. Nematol. 2003, 35, 271–277. [Google Scholar]



| Soil Samples | Exchangeable K (g/kg) | Available PO4-P (mg/kg) | Total C (g/kg) | Total N (g/kg) | C/N | pH (H2O) | Basal Soil Respiration (mg/kg/h) | Aboveground Dry Weight (g) | Belowground Dry Weight (g) | SPAD Value |
|---|---|---|---|---|---|---|---|---|---|---|
| Figure 1A | ||||||||||
| A | 0.25 | 18.0 | 104 | 14.3 | 7.30 | 5.3 | 2.60 | 1.45 | 0.46 | 51.4 |
| B | 0.26 | 93.0 | 95.0 | 6.40 | 14.8 | 5.2 | 1.50 | 1.03 | 0.57 | 47.8 |
| C | 0.52 | 121 | 105 | 7.96 | 13.2 | 6.3 | 4.30 | 0.85 | 0.41 | 46.9 |
| D | 0.35 | 47.2 | 104 | 7.53 | 13.8 | 5.5 | 1.90 | 1.16 | 0.72 | 47.1 |
| E | 0.27 | 224 | 85.3 | 7.69 | 11.1 | 4.9 | 3.00 | 1.17 | 0.43 | 44.1 |
| F | 2.09 | 233 | 38.0 | 4.46 | 8.50 | 7.4 | 1.90 | 0.48 | 0.17 | 47.8 |
| G | 1.21 | 236 | 63.6 | 6.04 | 10.5 | 7.4 | 2.70 | 1.00 | 0.38 | 47.3 |
| Figure 1B | ||||||||||
| H | 0.27 | 70.8 | 57.1 | 4.95 | 11.5 | 7.1 | 1.70 | 1.00 | 0.16 | 45.0 |
| I | 0.20 | 52.8 | 34.6 | 3.74 | 9.30 | 7.3 | 1.21 | 0.84 | 0.18 | 41.6 |
| J | 0.09 | 90.2 | 33.7 | 2.92 | 11.5 | 6.4 | 1.60 | 1.13 | 0.15 | 45.9 |
| K | 0.24 | 17.8 | 55.5 | 5.02 | 11.1 | 6.4 | 0.98 | 0.87 | 0.09 | 43.1 |
| L | 0.11 | 182 | 41.5 | 3.56 | 11.7 | 6.1 | 1.18 | 1.00 | 0.15 | 40.8 |
| M | 0.21 | 48.6 | 38.8 | 4.63 | 8.40 | 5.8 | 1.79 | 1.25 | 0.24 | 44.4 |
| N | 0.09 | 55.8 | 30.9 | 3.62 | 8.60 | 6.1 | 0.86 | 0.86 | 0.13 | 41.2 |
| O | 0.02 | 15.5 | 69.4 | 6.66 | 10.4 | 4.7 | 0.55 | 1.52 | 0.31 | 45.7 |
| Soil Samples | Exchangeable K (g/kg) | Available PO4-P (mg/kg) | Total C (g/kg) | Total N (g/kg) | C/N | pH (H2O) | Basal Soil Respiration (mg/kg/h) | Aboveground Dry Weight (g) | Belowground Dry Weight (g) | SPAD Value |
|---|---|---|---|---|---|---|---|---|---|---|
| Figure 1 A | 0.30 | 0.17 | 0.19 | 0.83 | 0.24 | 0.35 | 0.66 | ** | ** | 0.86 |
| Figure 1 B | 0.23 | 0.41 | 0.29 | 0.17 | 0.58 | 0.75 | 0.48 | 0.59 | 0.88 | 0.37 |
| Closest Species | Cylindrodendrum alicantinum | Clonostachys rosea | A spergillus fumigatus | F usarium oxysporum | A spergillus niger | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| f0 | h | a2 | a27 | a17 | a18 | a20 | a23 | a25 | a8 | a10 | a21 | a22 | a26 | |
| Trial 6 | 30 * | 34 * | - | - | - | - | - | - | - | - | - | - | - | - |
| Trial 7 | 71 * | 52 * | - | - | - | - | - | - | - | - | - | - | - | - |
| Trial 8 | - | - | 31 * | - | - | - | - | - | - | 50 * | - | - | - | - |
| Trial 9 | - | - | - | 24 * | 25 * | 49 * | 39 * | 49 * | 31 * | - | 21 * | 34 * | 16 * | 31 * |
| Trial 10 | - | - | 65 * | 64 * | 8 | 36 * | 15 * | 34 * | 79 * | 60 * | 7 | 22 * | 58 * | 72 * |
| Trial 11 | 16 * | - | 36 * | - | 74 * | 56 * | - | - | 18 * | - | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Wu, J.; Perry, R.N.; Toyota, K. Evaluation of Soil Suppressiveness of Various Japanese Soils against the Soybean Cyst Nematode Heterodera glycines and Its Relation with the Soil Chemical and Biological Properties. Agronomy 2023, 13, 2826. https://doi.org/10.3390/agronomy13112826
Yang Y, Wu J, Perry RN, Toyota K. Evaluation of Soil Suppressiveness of Various Japanese Soils against the Soybean Cyst Nematode Heterodera glycines and Its Relation with the Soil Chemical and Biological Properties. Agronomy. 2023; 13(11):2826. https://doi.org/10.3390/agronomy13112826
Chicago/Turabian StyleYang, Yanyan, Junnan Wu, Roland N. Perry, and Koki Toyota. 2023. "Evaluation of Soil Suppressiveness of Various Japanese Soils against the Soybean Cyst Nematode Heterodera glycines and Its Relation with the Soil Chemical and Biological Properties" Agronomy 13, no. 11: 2826. https://doi.org/10.3390/agronomy13112826
APA StyleYang, Y., Wu, J., Perry, R. N., & Toyota, K. (2023). Evaluation of Soil Suppressiveness of Various Japanese Soils against the Soybean Cyst Nematode Heterodera glycines and Its Relation with the Soil Chemical and Biological Properties. Agronomy, 13(11), 2826. https://doi.org/10.3390/agronomy13112826






