1. Introduction
Since 1970, organic foods have gained interest on a global scale [
1]. Today, many farmers implement organic practices to attract consumers and increase their income. In turn, these agronomic practices promote crop rotation, biodiversity, etc., and they ban synthetic pesticides and fertilizers [
1]. However, despite the fact that organic systems produce smaller volumes of food [
2,
3], some authors have shown that organic foods are more nutritious than conventional ones, presenting higher contents of vitamin C or total antioxidants, among other things [
2,
4,
5,
6,
7,
8,
9]. In general, organic farming is considered to be more respectful of the environment than conventional farming, since it reduces the use of mineral fertilizers and pesticides, although the use of some non-synthetic pesticides, such as sulfur, copper sulphate, potassium permanganate, etc., are allowed [
10]. Currently, they have increased the number of organic farms and therefore the extent of farmland. In the EU, the total area devoted to organic farming continues to increase, covering almost 13.8 million hectares of agricultural land in 2019 [
11]. Likewise, the funds allocated to organic farming have also been increased [
12].
Worldwide, the lemon (
Citrus limon Burm.) is the third most cultivated citrus species, after orange and mandarin [
13]. Since the end of the 20th century, the consumption of citrus fruits began to increase among the world population [
14], reaching a production of more than 20 million tons in 2019 [
15]. Spain is the second lemon producer country [
16], and the two most cultivated varieties in this country are “Fino” and “Verna”, occupying almost the entire growing area destined for lemon. Lemons of the “Fino” variety are of high quality and are also used for international exports from October to February when prices are highest. “Verna” variety is harvested once the “Fino” harvest has finished and allows exports to be maintained during almost the entire period of the year [
17].
Consumers are not only looking for a visually attractive fruit (size, color, firmness, etc.), but are also looking for internal quality (flavor, volatile compounds, functional compounds, etc.) [
14,
18]. Although citrus flavor and aroma are important factors in quality, consumers buy a lemon product based mainly on their perception of product quality and value for money [
19].
In general, lemon is a fruit with an important content of bioactive compounds (vitamins, phenolic compounds, fiber, organic acids, mineral salts, etc.) responsible for beneficial health effects [
20,
21,
22]. Several authors have reported its healthy properties on various types of cancer, cardiovascular diseases, obesity, cholesterol, etc. [
20,
23,
24].
The flavor of a lemon depends on the concentration of organic acids and sugars and the relationship between them. Both are the main compounds in citric fruits. [
25,
26]. However, the main quality characteristic of citrus fruits is aroma [
27]. Most of the volatile compounds of lemon are found in the skin of the fruit presenting 85–90% of the entire essential oil fraction of the lemon [
28,
29,
30]. Citrus essential oils are used in the food industry as flavorings in beverages and alcoholic beverages, jams, jellies, sweets, ice cream and dairy products [
31,
32,
33]. It is possible to determine direct relationships between the odor or flavor of a sample and the responsible volatile compounds in two ways: (i) comparative sensory analysis, using GC-MS to detect volatiles and find associations or (ii) using GC olfactometry ports -MS to detect and identify the responsible compounds [
34].
On the other hand, the flavor, aroma, and functional quality of lemon depends on the agronomic practices, as use of rootstock, ripening, irrigation, etc., [
18,
19,
25,
31,
35,
36,
37].
Therefore, the objective of this study was to determine if the type of cultivation (organic or conventional) affects the chemical, physical and volatile composition of “Fino 49” lemon.
2. Materials and Methods
2.1. Plant Material
The assay was carried out on lemons of the “Fino 49” variety grafted on Citrus macrophylla. “Fino 49” is a clone of the “Fino” variety and is the one that represents most young plantations (less than 10 years old) in the Mediterranean area. The Fino 49 variety is very early in its production, very productive and has a tendency to bear fruit in clusters. It has stumpy trees and shorter internodes than Fino but has larger thorns. The fruits are larger and heavier (170 g) and have a higher percentage of juice, with the possibility of harvesting in September–October. The rootstock used in the test is very vigorous, tolerant to Exocortis and very resistant to Phytophotora, but it is sensitive to Tristeza and Xyloporosis. Grafted with lemon trees, it is tolerant to all serious virosis, except Xyloporosis. It presents a very good affinity with all varieties. It is resistant to limestone and salinity and is sensitive to root suffocation and very sensitive to frost. The characteristics that stand out the most are the rapid entry into production, high productivity, and early ripening, with average fruit quality. Organic growing was carried out on the “Lo Vera” orchard (38°03′24.6″ N, 0°44′25.0″ W) with an area of 10.28 ha and conventional cultivation was realized on the “Lo Lorente” orchard (38°02′21.3″ N, 0°44′51.3″ W) with an area of 9.11 ha. Both orchards were located in Alicante (Spain), at a distance of ~2 km, and the tree spacing was 6.5 × 4 m. The trees are 10 years old (both farms were planted in 2011). The climate is characterized by mild winters and hot summers, temperatures ranging between 33 and 10 °C, and light rains concentrated in spring and autumn.
The organic farming orchard has a clay-textured soil (sand 29.55%, silt 30.00% and clay 40.45%), with an organic matter content of 1.40%. The nitrate content is 62.20 mg kg−1, the Olsen assimilable phosphorus content is 15.8 mg kg−1 and potassium in the aqueous extract is 1.07 meq L−1. The cation exchange capacity is 14.70 meq 100 g−1. The C/N ratio is 7.84 and the field capacity is 27.60 (% dry soil). The conductivity of the 1:2 aqueous extract at 25 °C is 1.21 dS m−1.
The conventional orchard has a loam-clay-loam texture soil (sand 12.05%, silt 52.50% and clay 35.45%), with an organic matter content of <0.65 %. The nitrate content is 88.10 mg kg−1, the Olsen assimilable phosphorus content is 5.37 mg kg−1 and potassium in the aqueous extract is 0.39 meq L−1. The cation exchange capacity is 10.40 meq 100 g−1. The C/N ratio is 5.93 and the field capacity is 28.40 (% dry soil). The conductivity of the 1:2 aqueous extract at 25 °C is 0.31 dS m−1.
As for the water used, for the orchard destined for organic farming, it had an electrical conductivity of 1.16 dS m−1 and a pH of 8.4. The cation content expressed in mmol L−1 was 1.74 for calcium; potassium 0.21; magnesium 1.87 and sodium 4.31. The anion content expressed in mmol L−1 was <0.02 for nitrates; phosphates <0.01; sulfates 2.44; bicarbonates 3.00 and chlorides 4.37.
The water of the conventional orchard had an electrical conductivity of 1.32 dS m−1 and a pH of 8.1. The cation content expressed in mmol L−1 was 2.12 for calcium; potassium 0.27; magnesium 1.91 and sodium 4.83. The anion content expressed in mmol L−1 was <0.02 for nitrates; phosphates 0.05; sulfates 2.98; bicarbonates 3.39 and chlorides 4.54.
Treatments to control pests and diseases at the organic and conventional orchards are in
Table 1.
Regarding fertilization, the fertilization formula in the two farms was as follows: 257 kg ha−1 of N, 47 kg ha−1 of P2O5 and 160 kg ha−1 of K2O. The fertilizers used in the organic orchard were:
Solublack H 87 (70% humic acids and 15% fulvic acids).
Brotolim eco N-AA (6% N + 12% AA + 30% MO).
Ourpizca (CaO 6%).
Solured Mn-2.5.
Solured Zn-2.6.
Unicquel (Iron chelate 6%).
Haifa sop bio (K2O 53% + SO3 40%).
Agrosol fluid 2-4-6 + AA.
Copper shuttle (Cu 6.13%).
The fertilizers used in the conventional production were:
Brotolim primavera + Ca + Mg (10-2-5 + 3.5 CaO + 1.5 MgO).
Unicquel (Iron chelate 6%).
Novatec fluid engorde (7,5-2,5-6,8).
Brotolim Ca AC (CaO 8%).
Brotolim engorde + Ca (8-2-6 + 2,5 CaO).
Lumbusol (organic matter 30%).
The sampling of the fruits took place in the second half of October 2021. For each orchard (organic and conventional) 100 lemons from 10 trees (10 fruits per tree) were hand-harvested (from the middle part and the entire perimeter area of each tree) at physiological maturity, and immediately transported to the laboratory. Once in the laboratory, a selection of the 75 most homogeneous fruits (size, shape, and color) was made and 50 of these were separated for the physical-chemical, organic acids, sugars, and volatile compounds determinations, and the other 25 fruits were used for the sensory analysis.
2.2. Physical and Chemical Analysis
A toal of fifty lemon fruits from each grow system were selected to determine color, pH, size, total soluble solids (TSS), total titratable acidy, and weight. For the determination of the lemons’ weight (FW), a Mettler balance model AG204 scale (Mettler Toledo, Barcelona, Spain) was used. A digital caliper (model 500-197-20, 150 mm; Mitutoyo Corp., Aurora, IL, USA) was used for the determination of the size of each fruit: peel thickness (PT), polar diameter (PD), and equatorial diameter (ED). The lemon’s color was measured in peel and juice. Peel color was measured in CIE
L*
a*
b* coordinates (
L*,
a*,
b*) along the equatorial axis of each fruit (the same 50 used for the measurement of size and weight) using a calibrated Minolta CRC-200 (Minolta Camera Co. Osaka, Japan) spectrophotometer, with illuminated D65 and an observer of 10°. The results were expressed according to the CIE
L*
a*
b* system (CIE, 1931). The mean values for coordinates
L* (lightness),
a* (red-green),
b* (blue-yellow),
C* (Chroma) and h (tone) and with them, the corresponding citrus color index (CCI = 1000
a*/
L*
b*) using the equation proposed by Jiménez-Cuesta, et al. [
38] were calculated. Color parameters in CIE
L*
a*
b* were also measured on the lemon juice placed in a liquid accessory CR-A70. Total soluble solids (TSS) content (°Brix) was measured using a digital Atago refractometer (model N-20; Atago, Bellevue, WA, USA) at 20 °C. Total titratable acidy and pH were determined by acid–base potentiometer (877 Titrino plus, Metrohm ion analyses CH9101, Herisau, Switzerland), using 0.1 mol L
−1 NaOH up to a pH of 8.1; results were expressed as g of citric acid L
−1. Measurements were determined in triplicate.
2.3. Organic Acids and Sugars
Organic acids and sugars were identified and quantified according to Hernández, et al. [
39] with some modifications. The lemon juices were prepared by hand-squeezing in a commercial juicer. Then, the juices were centrifuged at 15,000×
g for 20 min (Sigma 3–18 K; Sigma, Osterode am Harz, Germany), filtered through a 0.45 μm Millipore filter, and injected (10 µL) into a high-performance liquid chromatograph (HPLC) system (HP Series 1100, Hewlett-Packard, Wilmington DE, USA). Organic acids were separated on a Supelcogel C-610H column (30 cm × 7.8 mm) fitted with a Supelcoguard column (5 cm × 4.6 mm) (Supelco, Inc., Bellefonte, PA, USA) and a diode array detector (210 nm) (HPLC, Waldbronn, Germany) was used for the detection. For the determination of sugars, the same HPLC system and conditions were used, but detection was performed with a refractive index detector (HPLC, Waldbronn, Germany). Standard curves of pure organic acids and sugars were used for quantification. Concentrations of both organic acids and sugars were expressed as g 100 mL
−1 of juice. Determinations of sugars and organic acids were conducted in triplicate.
2.4. Volatile Compounds
The volatile compounds of the lemon juice and essential oil obtained from the peel were analyzed. The determination of volatile compounds of the essential oil was carried out following the method described by Aguilar-Hernández, Sánchez-Bravo, Hernández, Carbonell-Barrachina, Pastor-Pérez and Legua [
18]. The extraction of the volatile compounds of the samples of lemon juice was carried out using the headspace solid-phase micro-extraction (HS-SPME) method. For the extraction, a SPME 50/30 mm DVB/CAR/PDMS (Divinylbenzene/Carboxen/Polydimethylsiloxane) fiber (Supelco) was used, whose exposure time was 40 min at a temperature of 45 °C and with constant agitation (500 rpm) using a magnetic stirrer (IKA C-MAG HS 4, IKA-Werke GmbH & Co. KG, Staufen, Germany).
A chromatograph Shimadzu GC2030 (Shimadzu Scientific Instruments, Inc., Columbia, MD, USA) with an SLB-5 MS column (Teknokroma, Barcelona, Spain) was used to separate the volatile compounds. The column had a length of 30 m, an internal diameter of 0.25 mm, and a film thickness of 0.25 μm. For the identification of compounds, the chromatograph was coupled with a Shimadzu TQ8040 NX mass spectrometer detector. The parameters of the mass spectrometer were: (i) mass range 45–400 m/z, (ii) scan speed 5000 amu/s, (iii) event time of 0.200 s, and (iv) electronic impact of 70 eV. Helium was used as a carrier gas, with a split ratio of 1:10, a purge flow of 6 mL min−1, and a total column flow of 0.6 mL min−1. The temperature of the detector was 300 °C, and the temperature of the injector was 230 °C. The total desorption time of the sample in the injection port was 3 min.
The oven program was the following:
Initial temperature of 80 °C.
Ramp of 3 °C min−1 from up to 170 °C.
Increase of 25 °C min−1 up to 300 °C, and maintained for 50 s.
A commercial alkane standard mixture (Sigma-Aldrich, Steinheim, Germany) was used to calculate the retention indexes (Kovat’s index). NIST 17 Mass Spectral and Retention Index libraries were used for the identification of compounds. Only compounds with spectral similarity >90% and with a deviation of less than 10 units of linear retention similarity were considered as correct hits. The analysis of the volatile compounds of the lemon samples was run in triplicate.
2.5. Sensory Analysis
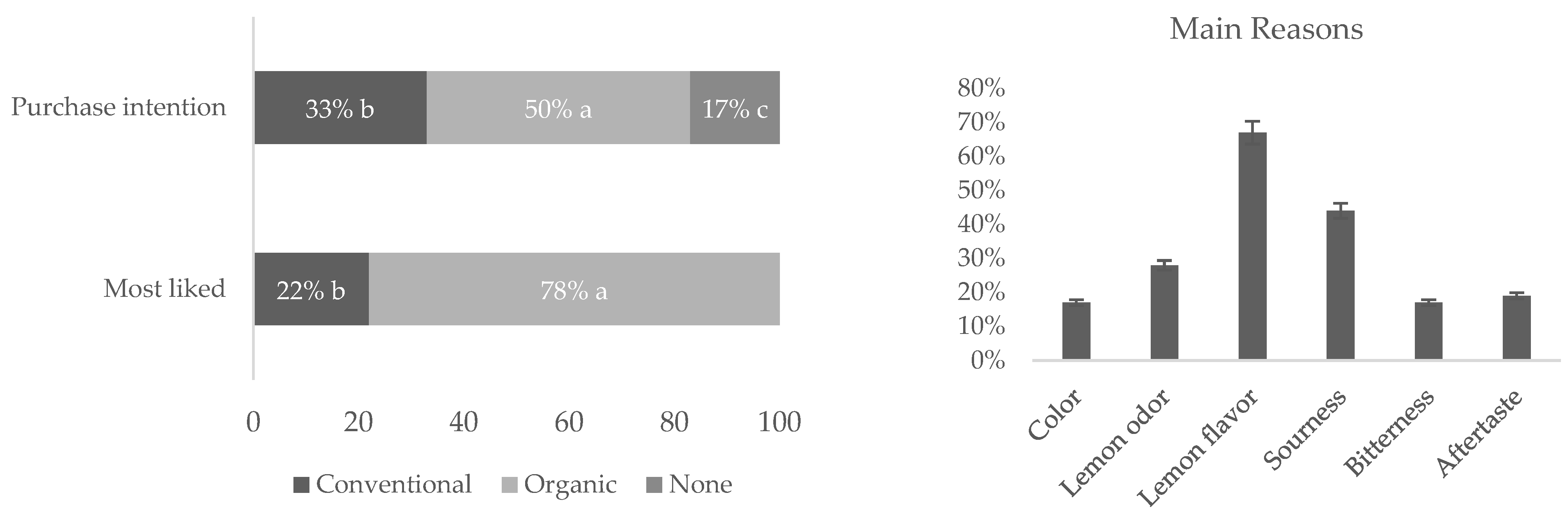
A consumer’s study was carried out with 100 participants. The juice samples were diluted (1 mL in 25 mL of water) for analysis. All samples were served at a temperature of 8 °C and encoded using three aleatory digit codes. Between each of the samples, osmotic water, and crackers (unsalted) were used to clean the palate. The test room had a temperature of 22 °C and a combination of natural and non-natural (fluorescent) light. Consumers were asked about color, lemon odor, lemon flavor, sourness, bitterness, and aftertaste. In addition, consumers indicated their “global” satisfaction and purchase intention for the samples under study. Demographic data were also collected. A 9-point scale was used, where 9 = like extremely and 1 = dislike extremely.
2.6. Statistical Analysis
A one-way analysis of variance (ANOVA) and Tukey’s multiple range test were performed for the analysis of the results. XLSTAT software (2016.02.27444 version, Addinsoft: New York, NY, USA) was used for the statistical treatment of the data. The confidence interval was 95% and the significant difference was defined as p < 0.05. All of the determinations were run in triplicate.