Methodology for Olive Fruit Quality Assessment by Means of a Low-Cost Multispectral Device
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Reference Analyses
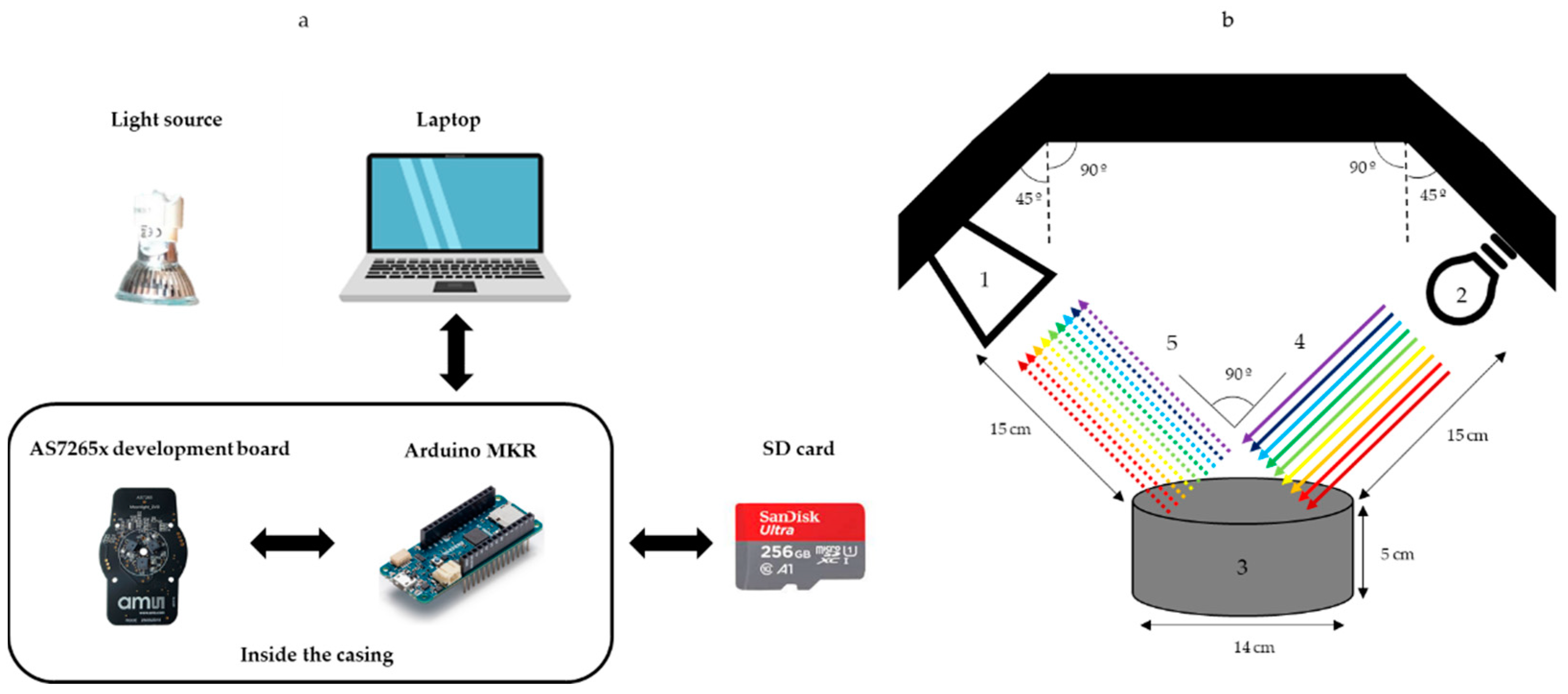
2.3. Spectral System
- Spectral sensor: the AS7265x development board (Figure 1), based on the AS7265x smart spectral sensor family (AMS, AG, Premstätten, Austria), was used. The sensor is composed of three chips, and each of them have six independent on-device optical filters whose spectral response is defined at a range between 410 nm and 940 nm, with full width at half maximum (FWHM) of 20 nm. The combination of the three sensors results in an 18-channel multispectral sensor.
- Light source: a 35 W dichroic halogen bulb, which offers a broadband spectrum allowing for accurate reflectance measurements, was employed. Halogen lamps have a wider spectral range of emission than that of LEDs, which enabled taking advantage of the sensor capabilities in the NIR domain. Moreover, using a relatively high power reduced the influence of ambient light interference, as the magnitude of the reflectance signal from the olive samples is considerably higher when compared to background and ambient light. Notwithstanding, an acquisition chamber was used to isolate the spectral measurement procedure to minimize signal noise.
- Controller board: the communication between the spectral sensor and a computer was implemented using an Arduino MKR board (Arduino LLC, Monza, Italy). A custom software was developed for the configuration of the capturing parameters (exposure time and gain). The software awaits user input to capture a sample spectrum. When capturing is triggered, the Arduino board sends the command to the sensor and gathers data. Then, the acquired data are sent to a computer and stored in an SD card for further analysis.
System Components and Cost
2.4. Methodology
2.4.1. Multispectral Signal Capture
2.4.2. Data Pre-Processing
2.4.3. Reference Parameter Modeling by Means of Multispectral Data
2.5. Criteria for Model Performance Evaluation
3. Results
3.1. Quality Condition of Samples
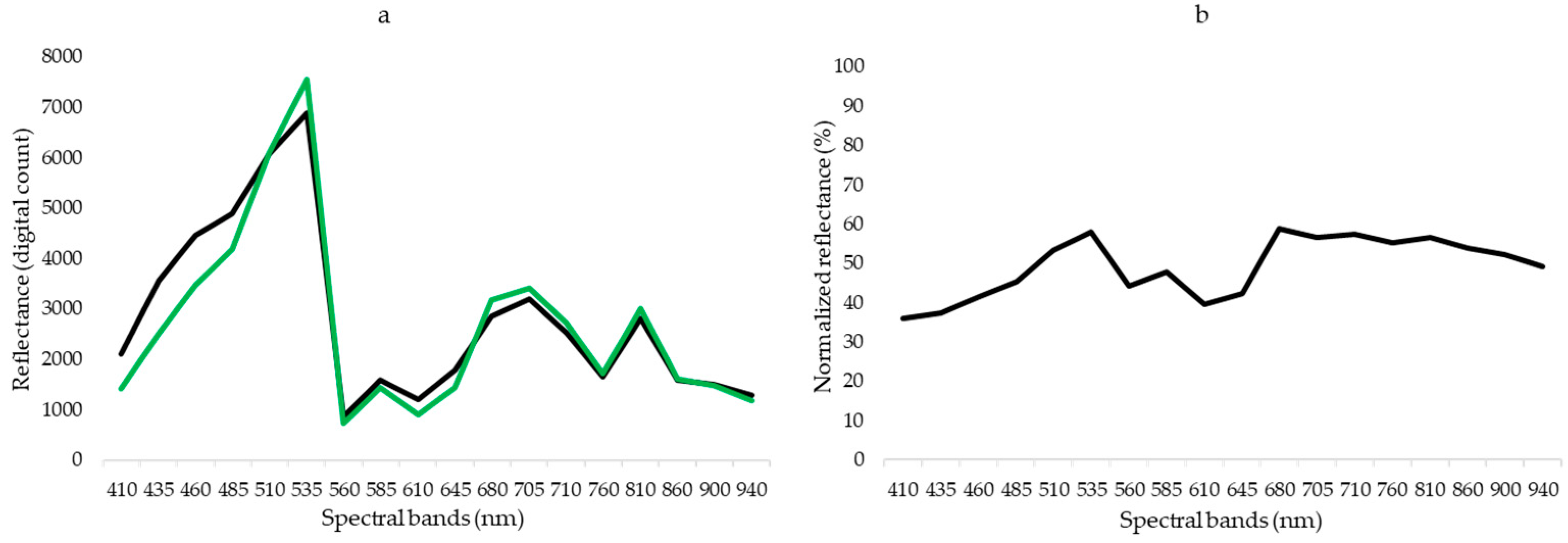
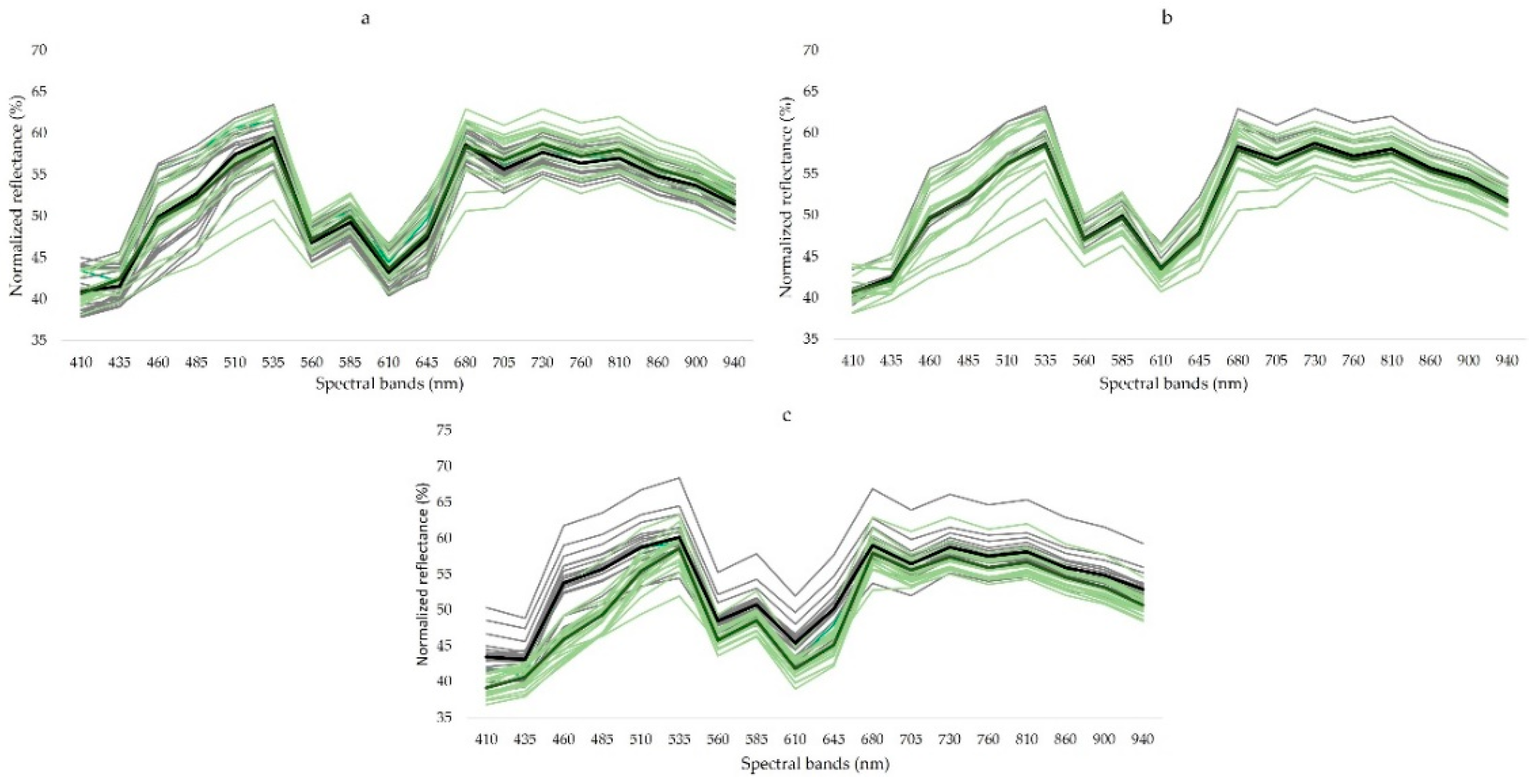
3.2. Spectral Signature of Samples
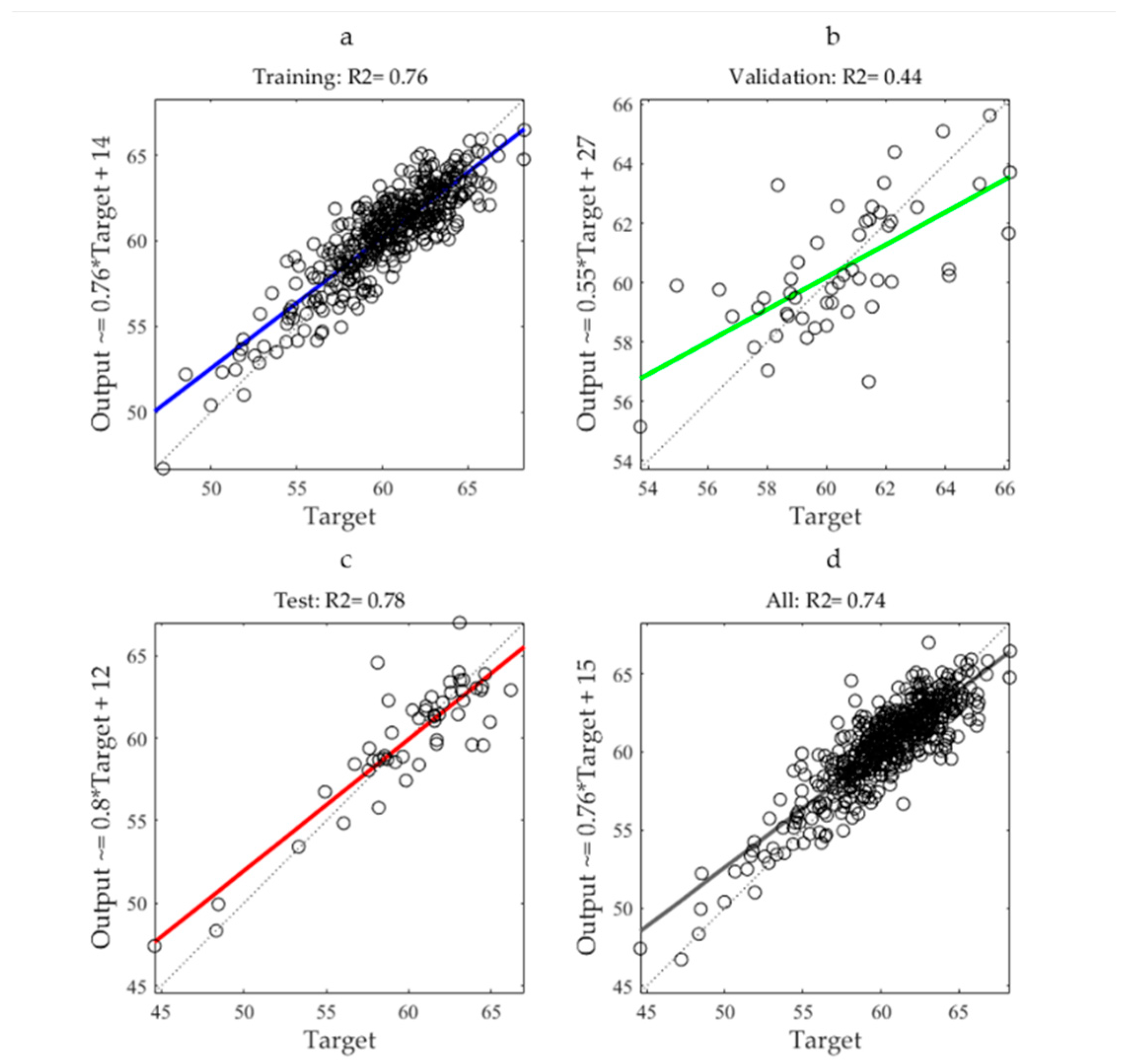
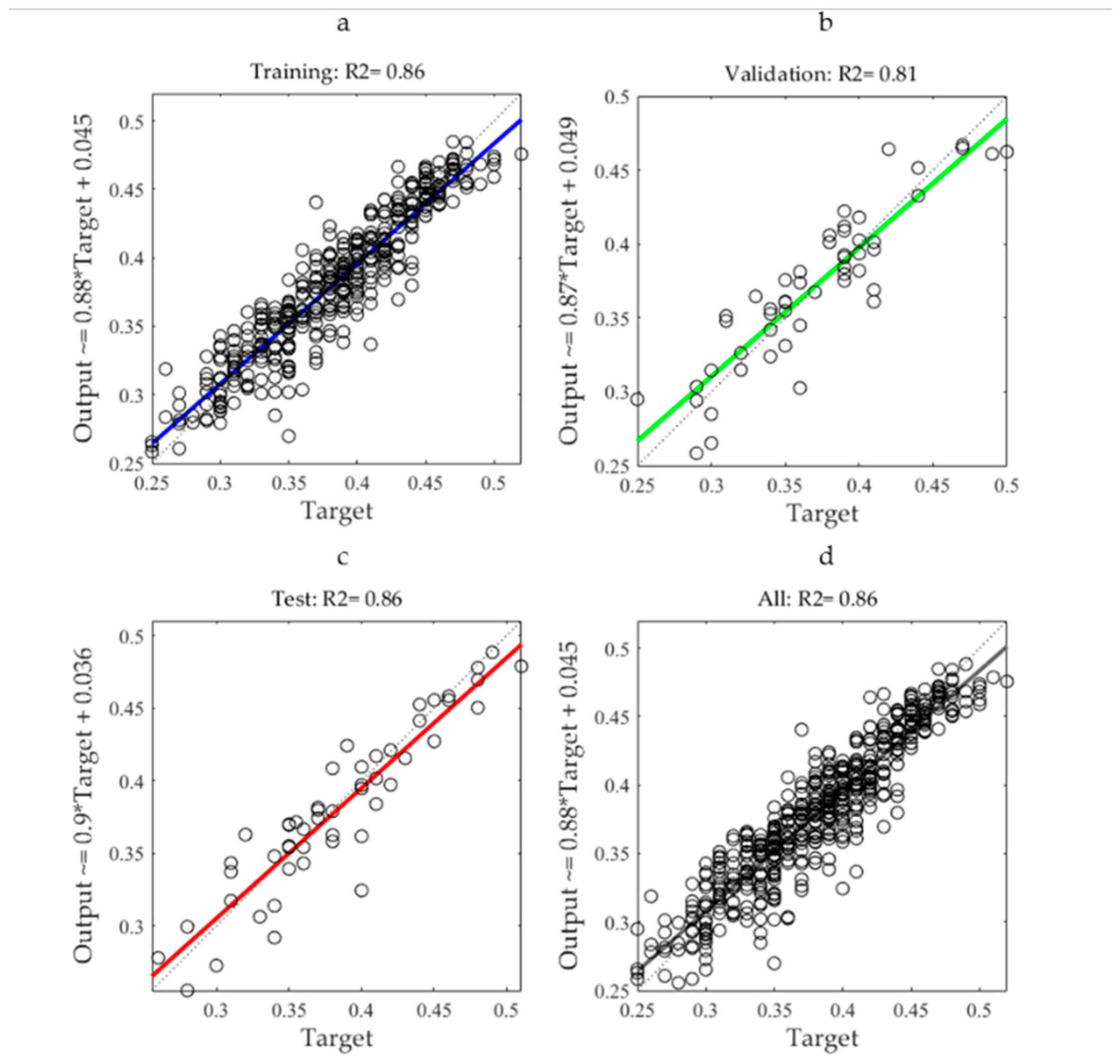
3.3. Performance of Estimation Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Available online: https://www.fao.org/faostat/en/#data/QV (accessed on 12 January 2022).
- López-Granados, F.; Jurado-Expósito, M.; Álamo, S.; García-Torres, L. Leaf nutrient spatial variability and site-specific fertilization maps within olive (Olea europaea L.) orchards. Eur. J. Agron. 2004, 21, 209–222. [Google Scholar] [CrossRef]
- Gunstone, F. Vegetable Oils in Food Technology: Composition, Properties and Uses, 2nd ed.; Gunstone, F., Ed.; Wiley-Blackwell: Oxford, UK, 2011; ISBN 9781444339918. [Google Scholar]
- Barranco Navero, D.; Fernandez Escobar, R.; Rallo Romero, L. El Cultivo del Olivo, 7th ed.; Mundi-Prensa Libros: Madrid, Spain, 2017. [Google Scholar]
- IOC (International Olive Council). Trade Standard Applying to Olive Oils and Olive Pomace Oils. 2019. Available online: https://www.internationaloliveoil.org/wp-content/uploads/2019/12/trade-standard-REV-14-Eng.pdf (accessed on 12 January 2022).
- Sánchez, A.G.; Martos, N.R.; Ballesteros, E. Estudio comparative de distintas técnicas analíticas (espectroscopía de NIR y RMN y extracción mediante Soxhlet) para la determinación del contenido graso y de humedad en aceitunas y orujo de Jaén. Grasas y Aceites 2005, 56, 220–227. [Google Scholar]
- Ragni, L.; Berardinelli, A.; Cevoli, C.; Valli, E. Assessment of the water content in extra virgin olive oils by Time Domain Reflectometry (TDR) and Partial Least Squares (PLS) regression methods. J. Food Eng. 2012, 111, 66–72. [Google Scholar] [CrossRef]
- Verrelst, J.; Malenovský, Z.; Van der Tol, C.; Camps-Valls, G.; Gastellu-Etchegorry, J.P.; Lewis, P.; North, P.; Moreno, J. Quantifying Vegetation Biophysical Variables from Imaging Spectroscopy Data: A Review on Retrieval Methods. Surv. Geophys. 2019, 40, 589–629. [Google Scholar] [CrossRef]
- Noguera, M.; Aquino, A.; Ponce, J.M.; Cordeiro, A.; Silvestre, J.; Calderón, R.; Marcelo, M. da E.; Pedro Jordão; Andújar, J.M. Nutritional status assessment of olive crops by means of the analysis and modelling of multispectral images taken with UAVs. Biosyst. Eng. 2021, 211, 1–18. [Google Scholar] [CrossRef]
- Fazari, A.; Pellicer-Valero, O.J.; Gómez-Sanchıs, J.; Bernardi, B.; Cubero, S.; Benalia, S.; Zimbalatti, G.; Blasco, J. Application of deep convolutional neural networks for the detection of anthracnose in olives using VIS/NIR hyperspectral images. Comput. Electron. Agric. 2021, 187, 106252. [Google Scholar] [CrossRef]
- León, L.; Rallo, L.; Garrido, A. Análisis de aceituna intacta mediante espectroscopia en el Infrarrojo Cercano (NIRS): Una herramienta de utilidad en programas de mejora de olivo. Grasas y Aceites 2003, 54, 41–47. [Google Scholar] [CrossRef]
- Kavdir, I.; Buyukcan, M.B.; Lu, R.; Kocabiyik, H.; Seker, M. Prediction of olive quality using FT-NIR spectroscopy in reflectance and transmittance modes. Biosyst. Eng. 2009, 103, 304–312. [Google Scholar] [CrossRef]
- Cayuela, J.A.; García, J.M.; Caliani, N. NIR prediction of fruit moisture, free acidity and oil content in intact olives. Grasas y Aceites 2009, 60, 194–202. [Google Scholar] [CrossRef]
- Cayuela, J.A.; Camino, M. del C.P. Prediction of quality of intact olives by near infrared spectroscopy. Eur. J. Lipid Sci. Technol. 2010, 112, 1209–1217. [Google Scholar] [CrossRef]
- Salguero-Chaparro, L.; Baeten, V.; Fernández-Pierna, J.A.; Peña-Rodríguez, F. Near infrared spectroscopy (NIRS) for on-line determination of quality parameters in intact olives. Food Chem. 2013, 139, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Salguero-Chaparro, L.; Peña-Rodríguez, F. On-line versus off-line NIRS analysis of intact olives. LWT Food Sci. Technol. 2014, 56, 363–369. [Google Scholar] [CrossRef]
- Gracia, A.; León, L. Non-destructive assessment of olive fruit ripening by portable near infrared spectroscopy. Grasas y Aceites 2011, 62, 268–274. [Google Scholar]
- Bellincontro, A.; Taticchi, A.; Servili, M.; Esposto, S.; Farinelli, D.; Mencarelli, F. Feasible application of a portable NIR-AOTF tool for on-field prediction of phenolic compounds during the ripening of olives for oil production. J. Agric. Food Chem. 2012, 60, 2665–2673. [Google Scholar] [CrossRef]
- Bellincontro, A.; Caruso, G.; Mencarelli, F.; Gucci, R. Oil accumulation in intact olive fruits measured by near infrared spectroscopy-acousto-optically tunable filter. J. Sci. Food Agric. 2013, 93, 1259–1265. [Google Scholar] [CrossRef]
- Fernández-Espinosa, A.J. Combining PLS regression with portable NIR spectroscopy to on-line monitor quality parameters in intact olives for determining optimal harvesting time. Talanta 2016, 148, 216–228. [Google Scholar] [CrossRef]
- FOSS. Analytical Solutions for Food Quality Improvement and Control. Available online: https://www.fossanalytics.com/en (accessed on 21 April 2021).
- Trang, N.M.; Duy, T.K.; Huyen, T.T.N.; Danh, L.V.Q.; Dinh, A. An investigation into the use of a low-Cost NIR integrated circuit spectrometer to measure chlorophyll content index. Int. J. Innov. Technol. Explor. Eng. 2019, 8, 35–38. [Google Scholar]
- Moinard, S.; Brunel, G.; Ducanchez, A.; Crestey, T.; Rousseau, J.; Tisseyre, B. Testing the potential of a new low-cost multispectral sensor for decision support in agriculture. In Precision Agriculture ´21; Wageningen Academic Publishers: Wageningen, The Netherlands, 2021; pp. 411–418. [Google Scholar]
- Verrelst, J.; Muñoz, J.; Alonso, L.; Delegido, J.; Rivera, J.P.; Camps-Valls, G.; Moreno, J. Machine learning regression algorithms for biophysical parameter retrieval: Opportunities for Sentinel-2 and -3. Remote Sens. Environ. 2012, 118, 127–139. [Google Scholar] [CrossRef]
- Kalacska, M.; Lalonde, M.; Moore, T.R. Estimation of foliar chlorophyll and nitrogen content in an ombrotrophic bog from hyperspectral data: Scaling from leaf to image. Remote Sens. Environ. 2015, 169, 270–279. [Google Scholar] [CrossRef]
- Wang, F.; Huang, J.; Wang, Y.; Liu, Z.; Peng, D.; Cao, F. Monitoring nitrogen concentration of oilseed rape from hyperspectral data using radial basis function. Int. J. Digit. Earth 2013, 6, 550–562. [Google Scholar] [CrossRef]
- Huang, Z.; Turner, B.J.; Dury, S.J.; Wallis, I.R.; Foley, W.J. Estimating foliage nitrogen concentration from HYMAP data using continuum removal analysis. Remote Sens. Environ. 2004, 93, 18–29. [Google Scholar] [CrossRef]
- Jensen, R.R.; Hardin, P.J.; Hardin, A.J. Estimating urban leaf area index (LAI) of individual trees with hyperspectral data. Photogramm. Eng. Remote Sensing 2012, 78, 495–504. [Google Scholar] [CrossRef]
- García-Martínez, H.; Flores-Magdaleno, H.; Ascencio-Hernández, R.; Khalil-Gardezi, A.; Tijerina-Chávez, L.; Mancilla-Villa, O.R.; Vázquez-Peña, M.A. Corn Grain Yield Estimation from Vegetation Indices, Canopy Cover, Plant Density, and a Neural Network Using Multispectral and RGB Images Acquired with Unmanned Aerial Vehicles. Agriculture 2020, 10, 277. [Google Scholar] [CrossRef]
- Liu, W.Y.; Pan, J. A hyperspectral assessment model for leaf chlorophyll content of Pinus massoniana based on neural network. Chinese J. Appl. Ecol. 2017, 28, 1128–1136. [Google Scholar]
- Zipori, I.; Bustan, A.; Kerem, Z.; Dag, A. Olive paste oil content on a dry weight basis (OPDW): an indicator for optimal harvesting time in modern olive orchards. Grasas y Aceites 2016, 67, e137. [Google Scholar]
- Gracia, P.; Sánchez-Gimeno, A.C.; Benito, M.; Oria, R.; Lasa, J.M. Short communication. Harvest time in hedgerow ‘Arbequina’ olive orchards in areas with early frosts. Span. J. Agric. Res. 2012, 10, 179–182. [Google Scholar] [CrossRef]
- Abenoza, M.; Lasa, J.M.; Benito, M.; Oria, R.; Sánchez Gimeno, A.C. The evolution of Arbequina olive oil quality during ripening in a commercial super-high density orchard in north-east Spain. Riv. Ital. Delle Sostanze Grasse 2015, 93, 83–92. [Google Scholar]
- Haberman, A.; Dag, A.; Shtern, N.; Zipori, I.; Erel, R.; Ben-Gal, A.; Yermiyahu, U. Significance of proper nitrogen fertilization for olive productivity in intensive cultivation. Sci. Hortic. 2019, 246, 710–717. [Google Scholar] [CrossRef]
- Haberman, A.; Dag, A.; Shtern, N.; Zipori, I.; Erel, R.; Ben-Gal, A.; Yermiyahu, U. Long-Term Impact of Potassium Fertilization on Soil and Productivity in Intensive Olive Cultivation. Agronomy 2019, 9, 525. [Google Scholar] [CrossRef]
- Ben-Gal, A.; Ron, Y.; Yermiyahu, U.; Zipori, I.; Naoum, S.; Dag, A. Evaluation of regulated deficit irrigation strategies for oil olives: A case study for two modern Israeli cultivars. Agric. Water Manag. 2021, 245, 106577. [Google Scholar] [CrossRef]
- Williams, P.C. Handbook of Near Infrared Analysis, 2nd ed.; Burns, D.A., Ciurczak, E.W., Eds.; Marcel Dekker: New York, NY, USA, 2001. [Google Scholar]
- Maes, W.H.; Steppe, K. Perspectives for Remote Sensing with Unmanned Aerial Vehicles in Precision Agriculture. Trends Plant Sci. 2019, 24, 152–164. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Lecourt, J.; Bishop, G. Advances in Non-Destructive Early Assessment of Fruit Ripeness towards Defining Optimal Time of Harvest and Yield Prediction—A Review. Plants 2018, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Casale, M.; Simonetti, R. Review: Near infrared spectroscopy for analysingolive oils. J. Near Infrared Spectrosc. 2014, 22, 59–80. [Google Scholar] [CrossRef]
- Guzmán, E.; Baeten, V.; Pierna, J.A.F.; García-Mesa, J.A. A portable Raman sensor for the rapid discrimination of olives according to fruit quality. Talanta 2012, 93, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.B.; Blasco, J.; Zude-Sasse, M.; Sun, X. Visible-NIR ‘point’ spectroscopy in postharvest fruit and vegetable assessment: The science behind three decades of commercial use. Postharvest Biol. Technol. 2020, 168, 111246. [Google Scholar] [CrossRef]
- Di Nonno, S.; Ulber, R. Portuino—A Novel Portable Low-Cost Arduino-Based Photo- and Fluorimeter. SSRN Electron. J. 2022, 12. [Google Scholar] [CrossRef]
- Poh, J.J.; Wu, W.L.; Goh, N.W.J.; Tan, S.M.X.; Gan, S.K.E. Spectrophotometer on-the-go: The development of a 2-in-1 UV–Vis portable Arduino-based spectrophotometer. Sensors Actuators A Phys. 2021, 325, 112698. [Google Scholar] [CrossRef]
- Noguera, M.; Millán, B.; Pérez-Paredes, J.J.; Ponce, J.M.; Aquino, A.; Andújar, J.M. A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring. Remote Sens. 2020, 12, 723. [Google Scholar] [CrossRef]
- Westad, F.; Schmidt, A.; Kermit, M. Incorporating chemical band-assignment in near infrared spectroscopy regression models. J. Near Infrared Spectrosc. 2008, 16, 265–273. [Google Scholar] [CrossRef]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]


| Description | Approx. Cost (€) |
|---|---|
| AS7265x development board | 150 |
| Arduino MKR | 24 |
| Light source | 3 |
| Other components (PTFE disc, PLA for device enclosure, etc.) | 10 |
| Total | 187 |
| Range | Mean | SD | |
|---|---|---|---|
| Moisture (%) | 44.58–68.29 | 60.40 | 3.26 |
| Acidity (%) | 0.25–0.52 | 0.38 | 0.06 |
| Fat content (%) | 8.92–24.43 | 16.32 | 2.45 |
| Moisture (%) | Acidity (%) | Fat Content (%) | |
|---|---|---|---|
| R2 | 0.78 | 0.86 | 0.62 |
| RMSEP | 3.31 | 5.83 | 10.44 |
| This Work | Fernández-Espinosa (2016) [20] | Salguero-Chaparro et al., (2013) [15] | Cayuela et al., (2009) [13] | |||||
|---|---|---|---|---|---|---|---|---|
| Chemometric | ANN | PCA-PLS | PCA-PLS | PCA-PLS | ||||
| Range | 410–940 | 1000–2300 | 380–1690 | 1100–2300 | ||||
| Statistics | R2 | RMSEP | R2 | RMSEP | R2 | RMSEP | R2 | RMSEP |
| Moisture | 0.78 | 3.31 | 0.88 | 4.98 | 0.88 | 3.3 | 0.88 | 1.52 |
| Acidity | 0.86 | 5.83 | 0.83 | 38.8 | 0.72 | 2.7 | 0.79 | 0.05 |
| Fat content | 0.62 | 10.44 | 0.76 | 20 | 0.79 | 2.36 | 0.72 | 7.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noguera, M.; Millan, B.; Aquino, A.; Andújar, J.M. Methodology for Olive Fruit Quality Assessment by Means of a Low-Cost Multispectral Device. Agronomy 2022, 12, 979. https://doi.org/10.3390/agronomy12050979
Noguera M, Millan B, Aquino A, Andújar JM. Methodology for Olive Fruit Quality Assessment by Means of a Low-Cost Multispectral Device. Agronomy. 2022; 12(5):979. https://doi.org/10.3390/agronomy12050979
Chicago/Turabian StyleNoguera, Miguel, Borja Millan, Arturo Aquino, and José Manuel Andújar. 2022. "Methodology for Olive Fruit Quality Assessment by Means of a Low-Cost Multispectral Device" Agronomy 12, no. 5: 979. https://doi.org/10.3390/agronomy12050979
APA StyleNoguera, M., Millan, B., Aquino, A., & Andújar, J. M. (2022). Methodology for Olive Fruit Quality Assessment by Means of a Low-Cost Multispectral Device. Agronomy, 12(5), 979. https://doi.org/10.3390/agronomy12050979






