Yield Performance of Woody Crops on Marginal Agricultural Land in Latvia, Spain and Ukraine
Abstract
:1. Introduction
2. Materials and Methods
2.1. Case Study Sites and Data Collection
2.1.1. Latvia
2.1.2. Spain
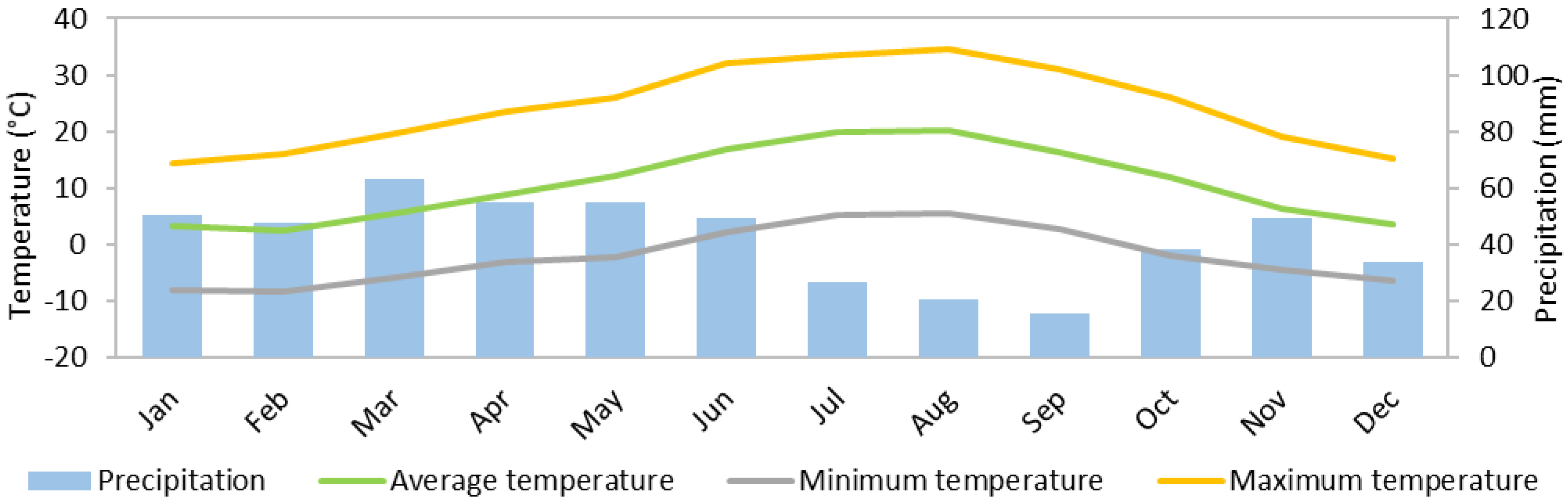
2.1.3. Ukraine
2.2. Statistical Analysis
3. Results
3.1. Latvia
3.2. Spain
3.3. Ukraine
4. Discussion
4.1. Yield Performance
4.2. Species Suitability
4.2.1. Willow and Aspen
4.2.2. Siberian Elm
4.2.3. Birch
4.2.4. Black and Gray Alder
4.3. Treatment
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Yearz | Precipitation (mm) | Mean Temperature (°C) | Maximum Temperature (°C) | Minimum Temperature (°C) |
|---|---|---|---|---|
| 2011 | 692.7 | 7.2 | 31.1 | −24.7 |
| 2012 | 935.4 | 6.0 | 31.8 | −29.6 |
| 2013 | 652.5 | 6.9 | 31.5 | −21.1 |
| 2014 | 855.5 | 7.3 | 32.3 | −18.8 |
| 2015 | 687.4 | 7.6 | 31.7 | −18.2 |
| 2016 | 894.4 | 6.9 | 31.6 | −24.4 |
| 2017 | 874.7 | 6.7 | 30.6 | −28.1 |
| 2018 | 363.9 | 7.4 | 32.9 | −23.8 |
| Annual average | 744.6 | 7.0 | 31.7 | −23.6 |
| Year | Precipitation (mm) | Mean Temperature (°C) | Maximum Temperature (°C) | Minimum Temperature (°C) | Solar Irradiance (kWh/m2) |
|---|---|---|---|---|---|
| 2009 | 107.4 | 4.8 | 18.9 | −12.8 | 98.0 |
| 2010 | 598.5 | 9.6 | 33.4 | −11.0 | 1508.0 |
| 2011 | 379.6 | 11.1 | 35.7 | −11.9 | 1629.2 |
| 2012 | 344.4 | 10.6 | 37.0 | −10.3 | 1659.6 |
| 2013 | 594.5 | 9.5 | 33.8 | −8.8 | 1547.8 |
| 2014 | 595.5 | 10.7 | 33.3 | −6.7 | 1599.6 |
| 2015 | 488.1 | 11.0 | 36.0 | −9.5 | 1605.8 |
| 2016 | 540.2 | 10.7 | 34.7 | −7.5 | 1404.5 |
| 2017 | 314.8 | 11.9 | 35.3 | −11.7 | 1654.6 |
| 2018 | 668.7 | 10.4 | 35.3 | −8.3 | 1294.5 |
| Annual average | 463.2 | 10.0 | 33.3 | −9.8 | 1400.2 |
| Year | Precipitation (mm) | Mean Temperature (°C) | Maximum Temperature (°C) | Minimum Temperature (°C) |
|---|---|---|---|---|
| 2011 | 446.0 | 8.3 | 30.8 | −17.2 |
| 2012 | 497.4 | 8.4 | 36.8 | −28.2 |
| 2013 | 618.2 | 8.6 | 29.7 | −19.0 |
| 2014 | 549.0 | 8.5 | 32.9 | −23.5 |
| 2015 | 372.0 | 9.8 | 35.2 | −18.5 |
| 2016 | 466.5 | 9.0 | 33.1 | −22.2 |
| 2017 | 538.1 | 9.0 | 33.4 | −21.6 |
| 2018 | 566.1 | 8.8 | 30.0 | −22.3 |
| 2019 | 535.8 | 9.9 | 33.1 | −12.5 |
| Annual average | 509.9 | 8.9 | 32.8 | −20.6 |


| Species | Site Condition | Planting Density, Plants ha−1 | Rotation Length, Years | * Above-Ground Biomass Yield, Dry Weight, Mg ha−1 y−1 | ** Treatment | Location | Source |
|---|---|---|---|---|---|---|---|
| Hybrid aspen (2 clones) | Poor rooting conditions—unfavorable soil texture and stoniness | 1261–2500 | 8 | 0.9–4.8 depending on clone and treatment | Fertilizer, weed control, animal prevention | Latvia | |
| Hybrid alder | 1636; | 2.0 | |||||
| Black alder | 1.3 | ||||||
| Gray alder | 1.7 | ||||||
| Birch | 1.4 | ||||||
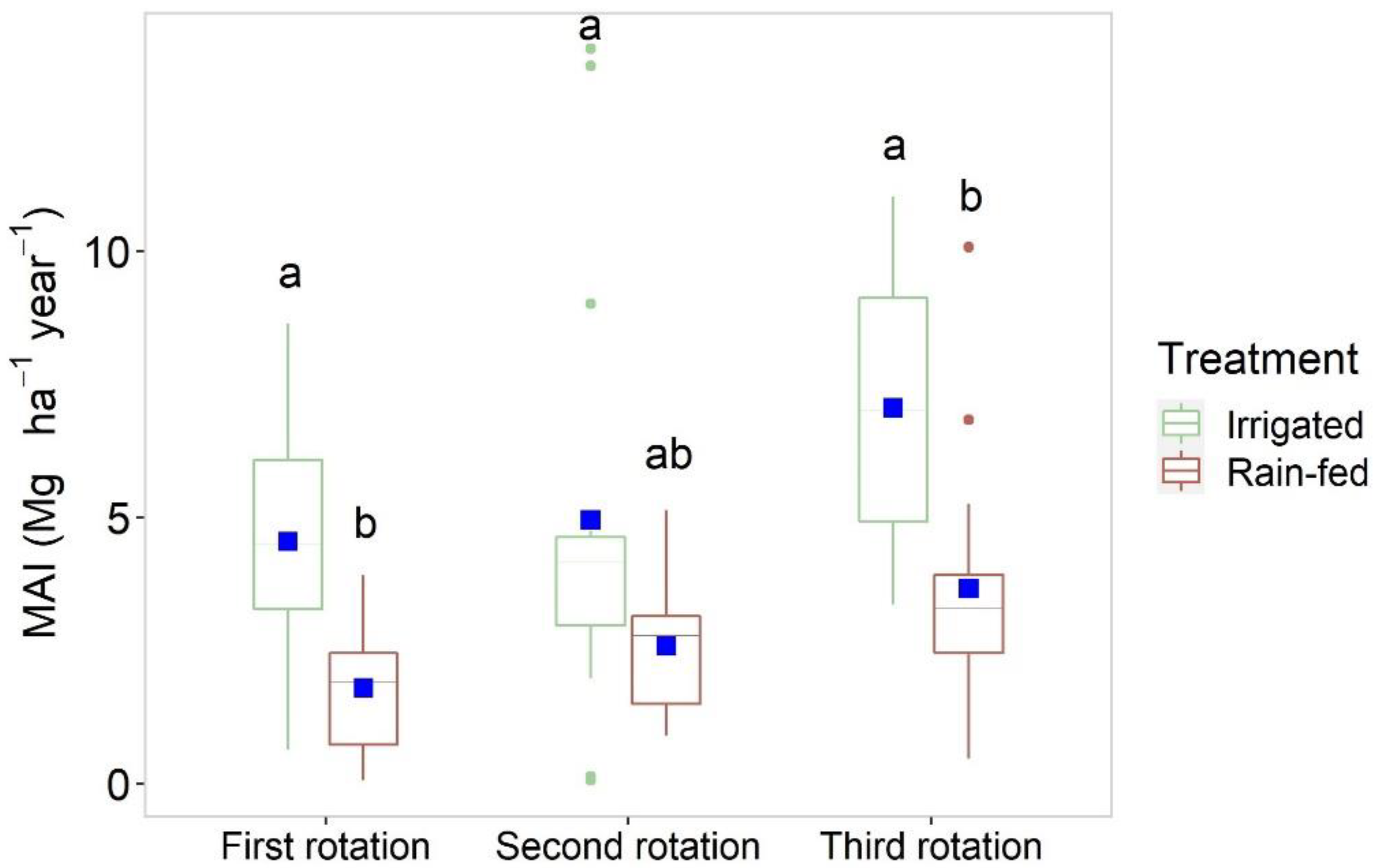
| Siberian elm | Sandy soil with unfavorable soil texture, stoniness and low soil organic carbon (<1%) | 6666 | 3 | 1.8 rain-fed and 4.5 irrigated (first rotation); 2.6 rain-fed and 4.9 irrigated (second rotation); 3.7 rain-fed and 7.1 irrigated (third rotation); | Rain-fed and irrigation | Spain | |
| Willow | Clay soil with low soil organic carbon | 20,000 | 3; | 8.4 (first rotation); 9.5 (third rotation); | Ukraine | ||
| Willow | Soil with low soil organic carbon | 20,000 | 3; | 10.8 (second rotation) | Ukraine | ||
| Willow | Soil with low soil organic carbon and high soil acidity | 20,000 | 3; | 8.6 (first rotation) | Fertilizer | Ukraine | |
| Poplar (12 clones) | Former agricultural land with sandy soil and limited drainage | 8000 | 2 | 1.5–7.2 (3.0–14.4 Mg ha−1) (first rotation) and 7.4–16.2 (14.8–32.4 Mg ha−1) (second rotation) depending on clone | Weed control | Belgium | [45,76] |
| Poplar (17 clones) | Former waste disposal site covered with a 2 m thick layer of sand, clay and rubble | 10,000 | 4 | 2.2–11.4 depending on clone | Weed control | Belgium | [75] |
| Birch | Former agricultural land, sandy soil with soil organic matter <1% and pHKCl 4.5 | 6667 (birch, maple); 20,000 (poplar, willow) | 4 | 2.6 | Belgium | [63] | |
| Maple | 1.2 | ||||||
| Poplar | 3.5 | ||||||
| Willow | 3.4 | ||||||
| Willow | Contaminated, dry, nutrient poor, sandy soils | 18,000 | 3 | 4.2–6.6 | Weed control | Belgium | [26] |
| Poplar | 1.1–1.5 | ||||||
| Willow | Former agricultural land | 14,800; 17,800; | 5 | 3.1 (15.4 Mg ha−1) control and 4.9–5.3 (24.7–26.3 Mg ha−1) irrigated | Irrigated | Estonia | [114] |
| Birch | Naturally afforested abandoned agricultural land (and 1 planted site) | 36,200 | 8 | 2.9 (22.8 Mg ha−1) | Estonia | [53] | |
| 13,900 | 2.8 (22.0 Mg ha−1) | ||||||
| 28,260 | 1.3 (10.2 Mg ha−1) | ||||||
| 3060 | 0.8 (6.0 Mg ha−1) | ||||||
| 4400 (planted) | 1.7 (13.3 Mg ha−1) (planted) | ||||||
| Birch | Leveled quarry spoil | 1017 | 7 | 0.02 (0.2 Mg ha−1) | Estonia | [121] | |
| Alder | 2100 | 0.36 (2.6 Mg ha−1) | |||||
| Pine | 3042 | 0.27 (1.9 Mg ha−1) | |||||
| Willow | Restored landfill | 1000–10,500 | 3 | 10.5; 18.8–22.6 (irrigated); | Irrigation | Finland | [115] |
| Birch and willow | Naturally afforested cut-away peatland | 12,800 | 14 | 2.7–4.4 | Fertilizer | Finland | [117] |
| Hybrid aspen | Fallow agricultural land | 900 | 1 | 5.2; | Fertilizer (in second season) | Finland | [125] |
| 2 | 8.7 (17.4 Mg ha−1) control and 9.95 (19.9 Mg ha−1) fertilized | ||||||
| 3 | 7.9 (23.9 Mg ha−1) control and 9.5 (28.9 Mg ha−1) fertilized | ||||||
| Birch | Organic soils—cutaway peatlands—naturally afforested | 10–27 | 3–4 | Finland | [126] | ||
| Poplar (14 clones) | Trace element contaminated site | 7 | 3.1–8.5 | France | [82] | ||
| Poplar | Abandoned agricultural land | 7272 | 2 | 1.9 (3.7 Mg ha−1) (first rotation) 4.3 (8.6 Mg ha−1) (second rotation) | Weed control | France | [77] |
| Willow | Abandoned agricultural land | 9697 | 2 | 2.07 (4.1 Mg ha−1) (first rotation) 11.0 (21.9 Mg ha−1) (second rotation) | Weed control | France | [77] |
| Poplar (8 clones) | Disturbed, marginally fertile post-mine site | 8333 | 8 | 0.4–6.0 (3.5–46.7 Mg ha−1) | Fertilizer | Germany | [120] |
| Black locust | Post-mine site with substrate from overburden sediments dumped during opencast lignite mining and low nitrogen content | 6579; | 14 | 2.7 | Fertilizer | Germany | [71] |
| 10,929; | 3 | 1.9, 2.5 and 1.8 (first, second and third rotation) | |||||
| 9200; | 4 | 0.5 | |||||
| 8736; | 4 | - | |||||
| Willow, poplar and black locust | Land with high sand content | 6700 | 2 | 4.3, 7.7 and 9.2 (first, second and third rotation) | Italy | [57] | |
| Willow, poplar and black locust | Land with low soil organic matter | 6700 | 2 | 3.3, 12.9 and 12.2 (first, second and third rotation) | Italy | [57] | |
| Birch; Pine; | Unfavorable soil texture, limited drainage | 3300 (birch); 5000 (pine) | 8 | 0.7 (birch) and 0.3 (pine) | Weed control, animal prevention | Latvia | [97] |
| 15 | 1.9 (birch) and 3.8 (pine) | ||||||
| Aspen | Limited soil drainage, periodic flooding, low temperatures | 3300 | 8 | 0.5 | Weed control, animal prevention | Latvia | [97] |
| 15 | 4.5 | ||||||
| Spruce | Acidic soil | 3300 | 8 | 0.5–1.5 (depending on site) | Weed control, animal prevention | Latvia | [97] |
| 15 | 2.8–8.4 (depending on site) | ||||||
| Black alder | Acidic soil | 3300 | 8 | 1.3–3.3 (depending on site) | Weed control, animal prevention | Latvia | [97] |
| 15 | 2.5–15.9 (depending on site) | ||||||
| Birch | Acidic soil | 2000–3300 | 8 | 0.7–4.0 (depending on site) | Weed control, animal prevention | Latvia | [97] |
| 15 | 2.4–7.4 (depending on site) | ||||||
| Birch; Spruce; | Acidic soil, excess moisture, low P and N content, low temperatures | 3300 (spruce) | 8 | 2.1 (birch) and 1.0 (spruce) | Weed control, fertilizer, animal prevention | Latvia | [97] |
| 15 | 4.9 (birch) and 4.5 (spruce) | ||||||
| Willow | Marginal gley soils | 20,000 | 3 | 12–15 | Weed control | Northern Ireland | [56] |
| Siberian elm | Heavy black soil with a heavy clay granulometric composition | 3448–51,282 | 7 | 5.2 (first rotation) | Rain-fed | Poland | [127] |
| Willow | Poor agricultural soils (loose, sandy soil with periodical dryness) | 11,000 | 4 | 5.1–10.3 | Weed control, fertilizer | Poland | [118] |
| Poplar | 5.5–10.5 | ||||||
| Black locust | 1.6–3.7 | ||||||
| Siberian elm | Sandy soil with low organic matter content (0.92%), low nitrogen (0.03%), many gravels (39.9%) and pH 5.90 | 3333 | 3 | 1.18 rain-fed and 2.43 irrigated (first rotation) | Rain-fed and irrigation (4167 m3 ha−1y−1) | Spain | [110] |
| Siberian elm | Sandy soil with low organic matter content (0.92%), low nitrogen (0.03%), many gravels (39.9%) and pH 5.90 | 6666 | 3 | 1.63, 5.19 rain-fed and 4.93 irrigated (first rotation) | Rain-fed and irrigation (2250 m3 ha−1y−1 and 4167 m3 ha−1y−1) | Spain | [110] |
| Siberian elm | Sandy soil with unfavorable soil texture, stoniness 28% and low soil organic matter content (0.4%) | 3333 | 4 | 2.6 rain-fed and 6.0 irrigated (first rotation) | Rain-fed and irrigation (3400 m3 ha−1 y−1) | Spain | [84] |
| 6666 | 2.5 rain-fed and 6.5 irrigated (first rotation) | ||||||
| Siberian elm | Sandy clay loamy texture, pH 8.30, organic matter 4.0%, total nitrogen 0.35%, 27 ppm P (Olsen) and extreme climate | 3333 | 3 | 5.1 (first rotation) | Rain-fed | Spain | [65] |
| Siberian elm | Basic soil with an excess of calcium. Entisol orden and Xerofluvent greatgroup. | 6666 | 2 | 5.2 (first rotation) | Rain-fed | Spain | [64] |
| 3 | 13.2 (first rotation) | ||||||
| Siberian elm | Sandy loam texture, low organic matter content (0.75%), nitrogen 0.08% and pH 5.87 | 6666 | 2.5 | 3.46 (first rotation) | Rain-fed | Spain | [66] |
| 2 | 1.9 (first rotation) | ||||||
| Poplar | Sandy soil with low organic matter content in semi-arid climatic conditions | 10,000 | 3 (for 9 years) | 12 | Fertilizer | Spain | [58] |
| Willow | 9 | ||||||
| Black locust | 7 | ||||||
| Sycamore | 3 | ||||||
| Willow (3 clones) | Former mining area | 9876; 14,815; | 5 | 0.3, 0.7, 1.7 (1,.3, 3.6, and 8.6 Mg ha−1) depending on clone, 0.2, 1.1, 1.3 (1.1, 5.4, 6.6 Mg ha−1) depending on treatment, 0.8, 1.0 (4.0 and 5.2 Mg ha−1) depending on density | Fertilizer, weed control | Spain | [69] |
| Poplar | Degraded soils | 5000 | 3 | 12.3–17.9 (36.9–53.8 Mg ha−1) | Fertilizer, irrigation | Spain | [78] |
| Eucalyptus | 14.7–18.3 (44.2–55.0 Mg ha−1) | ||||||
| Paulownia | Degraded, acidic soils | 5000 | 3 | 1.1–1.7 (3.3–5.1 Mg ha−1) | Fertilizer, irrigation | Spain | [78] |
| Eucalyptus | 13.5–19.7 (40.4–59.2 Mg ha−1) |
References
- Ustaoglu, E.; Collier, M.J. Farmland abandonment in Europe: An overview of drivers, consequences, and assessment of the sustainability implications. Environ. Rev. 2018, 26, 396–416. [Google Scholar] [CrossRef]
- Perpiña Castillo, C.; Kavalov, B.; Diogo, V.; Jacobs-Crisioni, C.; Batista e Silva, F.; Lavalle, C. Agricultural Land Abandonment in the EU within 2015–2030. In JRC Report 113718; European Commission: Ispra, Italy, 2018. [Google Scholar]
- Abolina, E.; Luzadis, V.A. Abandoned agricultural land and its potential for short rotation woody crops in Latvia. Land use policy 2015, 49, 435–445. [Google Scholar] [CrossRef]
- Shengfa, L.; Xiubin, L. Global understanding of farmland abandonment: A review and prospects. J. Geogr. Sci. 2017, 27, 1123–1150. [Google Scholar] [CrossRef]
- Ruskule, A.; Nikodemus, O.; Kasparinska, Z.; Kasparinskis, R.; Brūmelis, G. Patterns of afforestation on abandoned agriculture land in Latvia. Agrofor. Syst. 2012, 85, 215–231. [Google Scholar] [CrossRef]
- Rey Benayas, J.M. Restoring forests after land abandonment. In Forest Restoration in Landscapes; Springer: New York, NY, USA, 2005; pp. 356–360. [Google Scholar] [CrossRef]
- Munroe, D.K.; van Berkel, D.B.; Verburg, P.H.; Olson, J.L. Alternative trajectories of land abandonment: Causes, consequences and research challenges. Curr. Opin. Environ. Sustain. 2013, 5, 471–476. [Google Scholar] [CrossRef]
- Lange, L.; Connor, K.O.; Arason, S.; Bundgård-Jørgensen, U.; Canalis, A.; Carrez, D.; Gallagher, J.; Gøtke, N.; Huyghe, C.; Jarry, B.; et al. Developing a Sustainable and Circular Bio-Based Economy in EU: By Partnering Across Sectors, Upscaling and Using New Knowledge Faster, and For the Benefit of Climate, Environment & Biodiversity, and People & Business. Front. Bioeng. Biotechnol. 2021, 8, 619066. [Google Scholar] [CrossRef]
- Lindegaard, K.N.; Adams, P.W.R.; Holley, M.; Lamley, A.; Henriksson, A.; Larsson, S.; von Engelbrechten, H.G.; Esteban Lopez, G.; Pisarek, M. Short rotation plantations policy history in Europe: Lessons from the past and recommendations for the future. Food Energy Secur. 2016, 5, 125–152. [Google Scholar] [CrossRef]
- Mandley, S.J.; Daioglou, V.; Junginger, H.M.; van Vuuren, D.P.; Wicke, B. EU bioenergy development to 2050. Renew. Sustain. Energy Rev. 2020, 127, 109858. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Renewables 2020 Global Status Report; United Nations Environment Programme: Nairobi, Republic of Kenya, 2020; ISBN 978-3-948393-00-7. [Google Scholar]
- Guidi, W.; Piccioni, E.; Bonari, E. Evapotranspiration and crop coefficient of poplar and willow short-rotation coppice used as vegetation filter. Bioresour. Technol. 2008, 99, 4832–4840. [Google Scholar] [CrossRef]
- Bloemen, J.; Fichot, R.; Horemans, J.A.; Broeckx, L.S.; Verlinden, M.S.; Zenone, T.; Ceulemans, R. Water use of a multigenotype poplar short-rotation coppice from tree to stand scale. GCB Bioenergy 2017, 9, 370–384. [Google Scholar] [CrossRef] [Green Version]
- Xi, B.; Clothier, B.; Coleman, M.; Duan, J.; Hu, W.; Li, D.; Di, N.; Liu, Y.; Fu, J.; Li, J.; et al. Irrigation management in poplar (Populus spp.) plantations: A review. For. Ecol. Manag. 2021, 494, 119330. [Google Scholar] [CrossRef]
- Geyer, W.A. Biomass production in the Central Great Plains USA under various coppice regimes. Biomass Bioenergy 2006, 30, 778–783. [Google Scholar] [CrossRef]
- Quinn, L.D.; Straker, K.C.; Guo, J.; Kim, S.; Thapa, S.; Kling, G.; Lee, D.K.; Voigt, T.B. Stress-Tolerant Feedstocks for Sustainable Bioenergy Production on Marginal Land. Bioenergy Res. 2015, 8, 1081–1100. [Google Scholar] [CrossRef] [Green Version]
- Nocentini, A.; Monti, A. Land-use change from poplar to switchgrass and giant reed increases soil organic carbon. Agron. Sustain. Dev. 2017, 37, 23. [Google Scholar] [CrossRef]
- Mafa-Attoye, T.G.; Thevathasan, N.V.; Dunfield, K.E. Indications of shifting microbial communities associated with growing biomass crops on marginal lands in Southern Ontario. Agrofor. Syst. 2020, 94, 735–746. [Google Scholar] [CrossRef] [Green Version]
- Kahle, P.; Janssen, M. Impact of short-rotation coppice with poplar and willow on soil physical properties. J. Plant Nutr. Soil Sci. 2020, 183, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Georgiadis, P.; Vesterdal, L.; Stupak, I.; Raulund-Rasmussen, K. Accumulation of soil organic carbon after cropland conversion to short-rotation willow and poplar. GCB Bioenergy 2017, 9, 1390–1401. [Google Scholar] [CrossRef]
- Bardos, R.P.; Bone, B.; Andersson-Sköld, Y.; Suer, P.; Track, T.; Wagelmans, M. Crop-based systems for sustainable risk-based land management for economically marginal damaged land. Remediation 2011, 21, 11–33. [Google Scholar] [CrossRef]
- Finzi, A.C.; Canham, C.D.; Van Breeman, N. Erratum: Canopy tree-soil interactions within temperate forests: Species effects on pH and cations (Ecological Applications 8 (447–454)). Ecol. Appl. 1998, 8, 905. [Google Scholar] [CrossRef]
- Kang, S.; Post, W.M.; Nichols, J.A.; Wang, D.; West, T.O.; Bandaru, V.; Izaurralde, R.C. Marginal Lands: Concept, Assessment and Management. J. Agric. Sci. 2013, 5, 129–139. [Google Scholar] [CrossRef] [Green Version]
- McKay, H. (Ed.) Short Rotation Forestry: Review of Growth and Environmental Impacts; Forest Research: Surrey, UK, 2011; Volume 2, ISBN 978-0-85538-827-0. [Google Scholar]
- Mertens, J.; Van Nevel, L.; De Schrijver, A.; Piesschaert, F.; Oosterbaan, A.; Tack, F.M.G.; Verheyen, K. Tree species effect on the redistribution of soil metals. Environ. Pollut. 2007, 149, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Ruttens, A.; Boulet, J.; Weyens, N.; Smeets, K.; Adriaensen, K.; Meers, E.; van Slycken, S.; Tack, F.; Meiresonne, L.; Thewys, T.; et al. Short rotation coppice culture of willows and poplars as energy crops on metal contaminated agricultural soils. Int. J. Phytoremediation 2011, 13, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Thiry, Y.; Colle, C.; Yoschenko, V.; Levchuk, S.; Van Hees, M.; Hurtevent, P.; Kashparov, V. Impact of Scots pine (Pinus sylvestris L.) plantings on long term 137Cs and 90Sr recycling from a waste burial site in the Chernobyl Red Forest. J. Environ. Radioact. 2009, 100, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Elbersen, B.; van Eupen, M.; Mantel, S.; Alexopoulou, E.; Zanghou, B.; Boogaard, H.; Carrasco, J.; Ceccarelli, T.; Ciria, C.S.; Ciria, P.; et al. Mapping Marginal land potentially available for industrial crops in Europe. In Proceedings of the European Biomass Conference and Exhibition Proceedings 26thEUBCE, Copenagen, Denmark, 14–17 May 2018; p. 71. [Google Scholar]
- Emery, I.; Mueller, S.; Qin, Z.; Dunn, J.B. Evaluating the Potential of Marginal Land for Cellulosic Feedstock Production and Carbon Sequestration in the United States. Environ. Sci. Technol. 2017, 51, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Gerwin, W.; Repmann, F.; Galatsidas, S.; Vlachaki, D.; Gounaris, N.; Baumgarten, W.; Volkmann, C.; Keramitzis, D.; Kiourtsis, F.; Freese, D. Assessment and quantification of marginal lands for biomass production in Europe using soil-quality indicators. Soil 2018, 4, 267–290. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.; Post, W.; Wang, D.; Nichols, J.; Bandaru, V.; West, T. Hierarchical marginal land assessment for land use planning. Land Use Policy 2013, 30, 106–113. [Google Scholar] [CrossRef]
- Lewis, S.M.; Kelly, M. Mapping the potential for biofuel production on marginal lands: Differences in definitions, data and models across scales. ISPRS Int. J. Geo-Information 2014, 3, 430–459. [Google Scholar] [CrossRef] [Green Version]
- Mellor, P.; Lord, R.A.; João, E.; Thomas, R.; Hursthouse, A. Identifying non-agricultural marginal lands as a route to sustainable bioenergy provision—A review and holistic definition. Renew. Sustain. Energy Rev. 2021, 135, 110220. [Google Scholar] [CrossRef]
- Shortall, O.K. ‘Marginal land’ for energy crops: Exploring definitions and embedded assumptions. Energy Policy 2013, 62, 19–27. [Google Scholar] [CrossRef]
- Turley, D.; Taylor, M.; Laybourn, R.; Hughes, J.; Kilpatrick, J.; Procter, C.; Wilson, L.; Edgington, P. Assessment of the availability of ‘marginal’ and ‘idle’ land for bioenergy crop production in England and Wales. Res. Proj. Final Rep. 2010, 86. [Google Scholar]
- MAGIC-MAPS. Available online: https://iiasa-spatial.maps.arcgis.com/apps/webappviewer/index.html?id=270aa7d778c245228fe82dc826cbd703 (accessed on 10 February 2022).
- Elbersen, B.; van Eupen, M.; Verzandvoort, S.; Boogaard, H.; Mucher, S.; Cicarreli, T.; Elbersen, W.; Mantel, S.; Bai, Z.; Iqbal, Y.; et al. Methodological Approaches to Identify and Map Marginal Land Suitable for Industrial Crops in Europe; EU Horiz. 2020; MAGIC; GA-No. 727698; Wageningen University & Research: Wageningen, The Netherlands, 2018. [Google Scholar]
- FAO. The State of Food and Agriculture: Food Aid for Food Security? FAO: Rome, Italy, 2006. [Google Scholar]
- Liepiņš, J. Latvijas Kokaudžu Biomasas Un Oglekļa Uzkrājuma Novērtēšanas Metodes Forest Stand Biomass and Carbon Stock Estimates in Latvia; Latvijas Lauksaimniecības Universitāte: Jelgava, Latvia, 2020. [Google Scholar]
- R Core Team. R: A Language and Environment; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Johansson, T. Biomass production of hybrid aspen growing on former farm land in Sweden. J. For. Res. 2013, 24, 237–246. [Google Scholar] [CrossRef]
- Keoleian, G.A.; Volk, T.A. Renewable energy from willow biomass crops: Life cycle energy, environmental and economic performance. CRC. Crit. Rev. Plant Sci. 2005, 24, 385–406. [Google Scholar] [CrossRef]
- Rosso, L.; Facciotto, G.; Bergante, S.; Vietto, L.; Nervo, G. Selection and testing of Populus alba and Salix spp. as bioenergy feedstock: Preliminary results. Appl. Energy 2013, 102, 87–92. [Google Scholar] [CrossRef]
- Tullus, A.; Tullus, H.; Vares, A.; Kanal, A. Early growth of hybrid aspen (Populus × wettsteinii Hämet-Ahti) plantations on former agricultural lands in Estonia. For. Ecol. Manag. 2007, 245, 118–129. [Google Scholar] [CrossRef]
- Verlinden, M.S.; Broeckx, L.S.; Van den Bulcke, J.; Van Acker, J.; Ceulemans, R. Comparative study of biomass determinants of 12 poplar (Populus) genotypes in a high-density short-rotation culture. For. Ecol. Manag. 2013, 307, 101–111. [Google Scholar] [CrossRef]
- Kauter, D.; Lewandowski, I.; Claupein, W. Quantity and quality of harvestable biomass from Populus short rotation coppice for solid fuel use—A review of the physiological basis and management influences. Biomass Bioenergy 2003, 24, 411–427. [Google Scholar] [CrossRef]
- Tullus, A.; Rytter, L.; Tullus, T.; Weih, M.; Tullus, H. Short-rotation forestry with hybrid aspen (Populus tremula L. × P. tremuloides Michx.) in Northern Europe. Scand. J. For. Res. 2012, 27, 10–29. [Google Scholar] [CrossRef]
- Daugaviete, M.; Bārdulis, A.; Daugavietis, U.; Lazdiņa, D.; Bārdule, A. Potential of Producing Wood Biomass in Short-Rotation Grey Alder (Alnus Incana Moench) Plantations on Agricultural Lands. In Proceedings of the 25th NJF Congress, Riga, Latvia, 16–18 June 2015; pp. 394–399. [Google Scholar]
- Liesebach, M.; Von Wuehlisch, G.; Muhs, H.J. Aspen for short-rotation coppice plantations on agricultural sites in Germany: Effects of spacing and rotation time on growth and biomass production of aspen progenies. For. Ecol. Manag. 1999, 121, 25–39. [Google Scholar] [CrossRef]
- Tullus, A.; Lukason, O.; Vares, A.; Padari, A.; Lutter, R.; Tullus, T.; Karoles, K.; Tullus, H. Economics of Hybrid Aspen (Populus tremula L. × P. tremuloides Michx.) and Silver Birch (Betula pendula Roth.) Plantations on Abandoned Agricultural Lands in Estonia. Balt. For. 2012, 18, 288–298. [Google Scholar]
- Johansson, T. Biomass equations for determining fractions of European aspen growing on abandoned farmland and some practical implications. Biomass Bioenergy 1999, 17, 471–480. [Google Scholar] [CrossRef]
- Oliveira, N.; Pérez-Cruzado, C.; Cañellas, I.; Rodríguez-Soalleiro, R.; Sixto, H. Poplar short rotation coppice plantations under mediterranean conditions: The case of Spain. Forests 2020, 11, 1352. [Google Scholar] [CrossRef]
- Uri, V.; Vares, A.; Tullus, H.; Kanal, A. Above-ground biomass production and nutrient accumulation in young stands of silver birch on abandoned agricultural land. Biomass Bioenergy 2007, 31, 195–204. [Google Scholar] [CrossRef]
- Bergkvist, P.; Ledin, S. Stem biomass yields at different planting designs and spacings in willow coppice systems. Biomass Bioenergy 1998, 14, 149–156. [Google Scholar] [CrossRef]
- Kellomäki, S.; Kilpelaïnen, A.; Alam, A. Forest bioenergy production: Management, carbon sequestration and adaptation. In Forest BioEnergy Production: Management, Carbon Sequestration and Adaptation; Springer: New York, NY, USA, 2013; p. 268. [Google Scholar] [CrossRef]
- McElroy, G.; Dawson, W.M. Biomass from Short-rotation Coppice Willow on Marginal Land. Biomass 1986, 10, 225–240. [Google Scholar] [CrossRef]
- Amaducci, S.; Facciotto, G.; Bergante, S.; Perego, A.; Serra, P.; Ferrarini, A.; Chimento, C. Biomass production and energy balance of herbaceous and woody crops on marginal soils in the Po Valley. GCB Bioenergy 2017, 9, 31–45. [Google Scholar] [CrossRef] [Green Version]
- Fernández, M.J.; Barro, R.; Pérez, J.; Ciria, P. Production and composition of biomass from short rotation coppice in marginal land: A 9-year study. Biomass Bioenergy 2020, 134, 105478. [Google Scholar] [CrossRef]
- Kopp, R.F.; Abrahamson, L.P.; White, E.H.; Volk, T.A.; Nowak, C.A.; Fillhart, R.C. Willow biomass production during ten successive annual harvests. Biomass Bioenergy 2001, 20, 1–7. [Google Scholar] [CrossRef]
- Di Nasso, N.O.; Guidi, W.; Ragaglini, G.; Tozzini, C.; Bonari, E. Biomass production and energy balance of a 12-year-old short-rotation coppice poplar stand under different cutting cycles. GCB Bioenergy 2010, 2, 89–97. [Google Scholar] [CrossRef]
- Don, A.; Osborne, B.; Hastings, A.; Skiba, U.; Carter, M.S.; Drewer, J.; Flessa, H.; Freibauer, A.; Hyvönen, N.; Jones, M.B.; et al. Land-use change to bioenergy production in Europe: Implications for the greenhouse gas balance and soil carbon. GCB Bioenergy 2012, 4, 372–391. [Google Scholar] [CrossRef] [Green Version]
- European Environmental Agency (EEA). Estimating the Environmentally Compatible Bioenergy Potential from Agriculture; European Environmental Agency (EEA): Copenhagen, Denmark, 2007.
- Vande Walle, I.; Van Camp, N.; Van de Casteele, L.; Verheyen, K.; Lemeur, R. Short-rotation forestry of birch, maple, poplar and willow in Flanders (Belgium) I-Biomass production after 4 years of tree growth. Biomass Bioenergy 2007, 31, 267–275. [Google Scholar] [CrossRef]
- Sanz, M.; Curt, M.D.; Plaza, A.; García-Müller, M.; Fernández, J. Assessment of siberian elm coppicing cycle. In Proceedings of the 19th European Biomass Conference, Berlin, Germany, 6–10 June 2011; pp. 601–605. [Google Scholar]
- Fernández, J.; Iriarte, L.; Sanz, M.; Curt, M.D. Preliminary study of Siberian elm (Ulmus Pumila L.) as an energy crop in a continental-Mediterranean Climate. In Proceedings of the 17th European Biomass Conference & Exhibition—From Research to Industry and Markets, Hamburg, Germany, 29 June–3 July 2009; pp. 148–153. [Google Scholar]
- Sanchéz, J.; Sanz, M.; Curt, M.D.; Fernández, J.; Mosquera, F. Influence of planting season on Siberian elm yield and economic prospects. In Proceedings of the 23rd European Biomass Conference and Exhibition, Vienna, Austria, 1–4 June 2015; pp. 198–204. [Google Scholar]
- Geyer, W.A.; Argent, R.M.; Walawender, W.P. Biomass properties and gasification behavior of 7 year-old Siberian elm. Wood Fiber Sci. 1987, 19, 176–182. [Google Scholar]
- Geyer, W.A. Influence of environmental factors on woody biomass productivity in the Central Great Plains, USA. Biomass Bioenergy 1993, 4, 333–337. [Google Scholar] [CrossRef]
- Castaño-Díaz, M.; Barrio-Anta, M.; Afif-Khouri, E.; Cámara-Obregón, A. Willow short rotation coppice trial in a former mining area in Northern Spain: Effects of clone, fertilization and planting density on yield after five years. Forests 2018, 9, 154. [Google Scholar] [CrossRef] [Green Version]
- Kuznetsova, T.; Rosenvald, K.; Ostonen, I.; Helmisaari, H.S.; Mandre, M.; Lõhmus, K. Survival of black alder (Alnus glutinosa L.), silver birch (Betula pendula Roth.) and Scots pine (Pinus sylvestris L.) seedlings in a reclaimed oil shale mining area. Ecol. Eng. 2010, 36, 495–502. [Google Scholar] [CrossRef]
- Grünewald, H.; Böhm, C.; Quinkenstein, A.; Grundmann, P.; Eberts, J.; von Wühlisch, G. Robinia pseudoacacia L.: A lesser known tree species for biomass production. Bioenergy Res. 2009, 2, 123–133. [Google Scholar] [CrossRef]
- Hansen, E.A. Poplar woody biomass yields: A look to the future. Biomass Bioenergy 1991, 1, 1–7. [Google Scholar] [CrossRef]
- Stolarski, M.J.; Niksa, D.; Krzyżaniak, M.; Tworkowski, J.; Szczukowski, S. Willow productivity from small- and large-scale experimental plantations in Poland from 2000 to 2017. Renew. Sustain. Energy Rev. 2019, 101, 461–475. [Google Scholar] [CrossRef]
- Fang, S.; Liu, Y.; Yue, J.; Tian, Y.; Xu, X. Assessments of growth performance, crown structure, stem form and wood property of introduced poplar clones: Results from a long-term field experiment at a lowland site. For. Ecol. Manag. 2021, 479, 118586. [Google Scholar] [CrossRef]
- Laureysens, I.; Bogaert, J.; Blust, R.; Ceulemans, R. Biomass production of 17 poplar clones in a short-rotation coppice culture on a waste disposal site and its relation to soil characteristics. For. Ecol. Manag. 2004, 187, 295–309. [Google Scholar] [CrossRef]
- Verlinden, M.S.; Broeckx, L.S.; Ceulemans, R. First vs. second rotation of a poplar short rotation coppice: Above-ground biomass productivity and shoot dynamics. Biomass Bioenergy 2015, 73, 174–185. [Google Scholar] [CrossRef] [Green Version]
- Guénon, R.; Bastien, J.C.; Thiébeau, P.; Bodineau, G.; Bertrand, I. Carbon and nutrient dynamics in short-rotation coppice of poplar and willow in a converted marginal land, a case study in central France. Nutr. Cycl. Agroecosyst. 2016, 106, 293–309. [Google Scholar] [CrossRef]
- Madejón, P.; Alaejos, J.; García-Álbala, J.; Fernández, M.; Madejón, E. Three-year study of fast-growing trees in degraded soils amended with composts: Effects on soil fertility and productivity. J. Environ. Manag. 2016, 169, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Pulford, I.D.; Watson, C. Phytoremediation of heavy metal-contaminated land by trees—A review. Environ. Int. 2003, 29, 529–540. [Google Scholar] [CrossRef]
- Jensen, J.K.; Holm, P.E.; Nejrup, J.; Larsen, M.B.; Borggaard, O.K. The potential of willow for remediation of heavy metal polluted calcareous urban soils. Environ. Pollut. 2009, 157, 931–937. [Google Scholar] [CrossRef]
- Glenz, C.; Schlaepfer, R.; Iorgulescu, I.; Kienast, F. Flooding tolerance of Central European tree and shrub species. For. Ecol. Manag. 2006, 235, 1–13. [Google Scholar] [CrossRef]
- Chalot, M.; Girardclos, O.; Ciadamidaro, L.; Zappelini, C.; Yung, L.; Durand, A.; Pfendler, S.; Lamy, I.; Driget, V.; Blaudez, D. Poplar rotation coppice at a trace element-contaminated phytomanagement site: A 10-year study revealing biomass production, element export and impact on extractable elements. Sci. Total Environ. 2020, 699, 134260. [Google Scholar] [CrossRef]
- Ericsson, K.; Rosenqvist, H.; Nilsson, L.J. Energy crop production costs in the EU. Biomass Bioenergy 2009, 33, 1577–1586. [Google Scholar] [CrossRef] [Green Version]
- Sanz, M.; Pérez, J.; Carrasco, J.E.; Ciria, P. Biomass yield of Siberian elm under different crop conditions on marginal agricultural land. In Proceedings of the 28th European Biomass Conference & Exhibition, Marseille, France, 6–9 July 2020; pp. 238–241. [Google Scholar]
- Martynov, V.V.; Nikulina, T.V. Population surge of zigzag elm sawfly (Aproceros leucopoda (Takeuchi, 1939): Hymenoptera: Argidae) in the Northern Cis-Azov Region. Russ. J. Biol. Invasions 2017, 8, 135–142. [Google Scholar] [CrossRef]
- Oswalt, C.M.; Oswalt, S.N. Chapter 8: Invasive Plants on Forest Land in the United States. In General Technical Report SRS 207; USDA-Forest Service, Southern Research Station: Asheville, NC, USA, 2013; pp. 123–134. [Google Scholar]
- Lakicevic, M.; Reynolds, K.M.; Orlovic, S.; Kolarov, R. Measuring dendrofloristic diversity in urban parks in Novi Sad (Serbia). Trees, For. People 2022, 8, 100239. [Google Scholar] [CrossRef]
- Brunet, J.; Zalapa, J.E.; Pecori, F.; Santini, A. Hybridization and introgression between the exotic Siberian elm, Ulmus pumila, and the native Field elm, U. minor, in Italy. Biol. Invasions 2013, 15, 2717–2730. [Google Scholar] [CrossRef]
- BOE. Real Decreto 1628/2011, de 14 de Noviembre, por el que se Regula el Listado y Catálogo Español de Especies Exóticas Invasoras. 2011. Available online: https://www.boe.es/buscar/doc.php?id=BOE-A-2011-19398 (accessed on 31 March 2022).
- Dulamsuren, C.; Hauck, M.; Nyambayar, S.; Bader, M.; Osokhjargal, D.; Oyungerel, S.; Leuschner, C. Performance of Siberian elm (Ulmus pumila) on steppe slopes of the northern Mongolian mountain taiga: Drought stress and herbivory in mature trees. Environ. Exp. Bot. 2009, 66, 18–24. [Google Scholar] [CrossRef]
- Alesso, S.P.; Tapias, R.; Alaejos, J.; Fernández, M. Biomass Yield and Economic, Energy and Carbon Balances of Ulmus pumila L., Robinia pseudoacacia L. and Populus × euroamericana (Dode) Guinier Short-Rotation Coppices on Degraded Lands under Mediterranean Climate. Forests 2021, 12, 1337. [Google Scholar] [CrossRef]
- Monedero, E.; Hernández, J.J.; Collado, R. Combustion-related properties of poplar, willow and black locust to be used as fuels in power plants. Energies 2017, 10, 997. [Google Scholar] [CrossRef] [Green Version]
- Sixto, H.; Cañellas, I.; van Arendonk, J.; Ciria, P.; Camps, F.; Sánchez, M.; Sánchez-González, M. Growth potential of different species and genotypes for biomass production in short rotation in Mediterranean environments. For. Ecol. Manag. 2015, 354, 291–299. [Google Scholar] [CrossRef]
- Vítková, M.; Tonika, J.; Müllerová, J. Black locust-Successful invader of a wide range of soil conditions. Sci. Total Environ. 2015, 505, 315–328. [Google Scholar] [CrossRef]
- De Marco, A.; Arena, C.; Giordano, M.; Virzo De Santo, A. Impact of the invasive tree black locust on soil properties of Mediterranean stone pine-holm oak forests. Plant Soil 2013, 372, 473–486. [Google Scholar] [CrossRef]
- Hytönen, J.; Aro, L.; Jylhä, P. Biomass production and carbon sequestration of dense downy birch stands on cutaway peatlands. Scand. J. For. Res. 2018, 33, 764–771. [Google Scholar] [CrossRef]
- Daugaviete, M.; Bambe, B.; Lazdiņš, A.; Lazdiņa, D. Plantāciju Mežu Augšanas Gaita, Produktivitāte un Ietekme uz Vidi; LVMI Silava: Salaspils, Latvia, 2017; p. 470. [Google Scholar]
- Dubois, H.; Verkasalo, E.; Claessens, H. Potential of birch (betula pendula roth and b. pubescens ehrh.) for forestry and forest-based industry sector within the changing climatic and socio-economic context ofwestern Europe. Forests 2020, 11, 336. [Google Scholar] [CrossRef] [Green Version]
- Christersson, L.; Sennerby-Forsse, L.; Zsuffa, L. The role and significance of woody biomass plantations in Swedish agriculture. For. Chron. 1993, 69, 687–693. [Google Scholar] [CrossRef]
- Claessens, H.; Oosterbaan, A.; Savill, P.; Rondeux, J. A review of the characteristics of black alder (Alnus glutinosa (L.) Gaertn.) and their implications for silvicultural practices. Forestry 2010, 83, 163–175. [Google Scholar] [CrossRef] [Green Version]
- Vurdu, H. Anatomical Characteristics of Stem, Branch and Root Wood in European Black Alder (Alnus glutinosa L. Gaertn.); Iowa State University: Ames, Iowa, 1977. [Google Scholar]
- Giardina, C.P.; Huffman, S.; Binkley, D.; Caldwell, B.A. Alders increase soil phosphorus availability in a Douglas-fir plantation. Can. J. For. Res. 1995, 25, 1652–1657. [Google Scholar] [CrossRef]
- Myrold, D.D.; Huss-Danell, K. Alder and lupine enhance nitrogen cycling in a degraded forest soil in Northern Sweden. Plant Soil 2003, 254, 47–56. [Google Scholar] [CrossRef]
- Uri, V.; Tullus, H.; Lo, K. Biomass production and nutrien accumulation in short-rotation grey alder. For. Ecol. Manag. 2002, 161, 169–179. [Google Scholar] [CrossRef]
- Yrjälä, K.; Zheng, H. Renewable energy from woody biomass of poplar and willow src coupled to biochar production. Handb. Environ. Chem. 2021, 99, 133–150. [Google Scholar] [CrossRef]
- Sevel, L.; Ingerslev, M.; Nord-Larsen, T.; Jørgensen, U.; Holm, P.E.; Schelde, K.; Raulund-Rasmussen, K. Fertilization of SRC Willow, II: Leaching and Element Balances. Bioenergy Res. 2014, 7, 338–352. [Google Scholar] [CrossRef]
- Böhlenius, H.; Nilsson, U.; Salk, C. Liming increases early growth of poplars on forest sites with low soil pH. Biomass Bioenergy 2020, 138, 105572. [Google Scholar] [CrossRef]
- Lebrun, M.; Miard, F.; Nandillon, R.; Hattab-Hambli, N.; Scippa, G.S.; Bourgerie, S.; Morabito, D. Eco-restoration of a mine technosol according to biochar particle size and dose application: Study of soil physico-chemical properties and phytostabilization capacities of Salix viminalis. J. Soils Sediments 2018, 18, 2188–2202. [Google Scholar] [CrossRef]
- Callesen, I.; Raulund-Rasmussen, K.; Jrgensen, B.B.; Kvist-Johannsen, V. Growth of beech, oak, and four conifer species along a soil fertility gradient. Balt. For. 2006, 12, 14–23. [Google Scholar]
- Pérez, I.; Pérez, J.; Carrasco, J.; Ciria, P. Siberian elm responses to different culture conditions under short rotation forestry in Mediterranean areas. Turkish J. Agric. For. 2014, 38, 652–662. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.M.; Roychowdhury, R.; Fujita, M. Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef]
- Park, G.E.; Lee, D.K.; Kim, K.W.; Batkhuu, N.O.; Tsogtbaatar, J.; Zhu, J.J.; Jin, Y.; Park, P.S.; Hyun, J.O.; Kim, H.S. Morphological characteristics and water-use efficiency of siberian elm trees (Ulmus pumila L.) within arid regions of northeast asia. Forests 2016, 7, 280. [Google Scholar] [CrossRef] [Green Version]
- Bista, D.R.; Heckathorn, S.A.; Jayawardena, D.M.; Mishra, S.; Boldt, J.K. Effects of drought on nutrient uptake and the levels of nutrient-uptake proteins in roots of drought-sensitive and -tolerant grasses. Plants 2018, 7, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holm, B.; Heinsoo, K. Municipal wastewater application to Short Rotation Coppice of willows—Treatment efficiency and clone response in Estonian case study. Biomass Bioenergy 2013, 57, 126–135. [Google Scholar] [CrossRef]
- Ettala, M.O. Short-rotation tree plantations at sanitary landfills. Top. Catal. 1988, 6, 291–302. [Google Scholar] [CrossRef]
- Kjøller, R.; Cruz-paredes, C.; Clemmensen, K.E. Soil Biological Communities and Ecosystem Resilience; Springer: Berlin/Heidelberg, Germany, 2017; pp. 223–252. [Google Scholar] [CrossRef]
- Hytönen, J.; Kaunisto, S. Effect of fertilization on the biomass production of coppiced mixed birch and willow stands on a cut-away peatland. Biomass Bioenergy 1999, 17, 455–469. [Google Scholar] [CrossRef]
- Stolarski, M.J.; Krzyżaniak, M.; Szczukowski, S.; Tworkowski, J.; Załuski, D.; Bieniek, A.; Gołaszewski, J. Effect of Increased Soil Fertility on the Yield and Energy Value of Short-Rotation Woody Crops. Bioenergy Res. 2015, 8, 1136–1147. [Google Scholar] [CrossRef] [Green Version]
- Salam, M.M.A.; Mohsin, M.; Pulkkinen, P.; Pelkonen, P.; Pappinen, A. Effects of soil amendments on the growth response and phytoextraction capability of a willow variety (S. viminalis × S. schwerinii × S. dasyclados) grown in contaminated soils. Ecotoxicol. Environ. Saf. 2019, 171, 753–770. [Google Scholar] [CrossRef]
- Bungart, R.; Hüttl, R.F. Growth dynamics and biomass accumulation of 8-year-old hybrid poplar clones in a short-rotation plantation on a clayey-sandy mining substrate with respect to plant nutrition and water budget. Eur. J. For. Res. 2004, 123, 105–115. [Google Scholar] [CrossRef]
- Kuznetsova, T.; Lukjanova, A.; Mandre, M.; Lõhmus, K. Aboveground biomass and nutrient accumulation dynamics in young black alder, silver birch and Scots pine plantations on reclaimed oil shale mining areas in Estonia. For. Ecol. Manag. 2011, 262, 56–64. [Google Scholar] [CrossRef]
- Löf, M.; Bolte, A.; Jacobs, D.F.; Jensen, A.M. Nurse Trees as a Forest Restoration Tool for Mixed Plantations: Effects on Competing Vegetation and Performance in Target Tree Species. Restor. Ecol. 2014, 22, 758–765. [Google Scholar] [CrossRef]
- Joffre, R.; Rambal, S. Linked references are available on JSTOR for this article: How tree cover influences the water balance of mediterranean rangelands. Ecology 1993, 74, 570–582. [Google Scholar] [CrossRef]
- Al-Namazi, A.A.; Bonser, S.P. Plant strategies in extremely stressful environments: Are the effects of nurse plants positive on all understory species? J. Plant Interact. 2020, 15, 233–240. [Google Scholar] [CrossRef]
- Hytönen, J. Biomass, nutrient content and energy yield of short-rotation hybrid aspen (P. tremula x P. tremuloides) coppice. For. Ecol. Manag. 2018, 413, 21–31. [Google Scholar] [CrossRef]
- Jylhä, P.; Hytönen, J.; Ahtikoski, A. Profitability of short-rotation biomass production on downy birch stands on cut-away peatlands in northern Finland. Biomass Bioenergy 2015, 75, 272–281. [Google Scholar] [CrossRef]
- Berbeć, A.K.; Matyka, M. Planting density effects on grow rate, biometric parameters, and biomass calorific value of selected trees cultivated as src. Agriculture 2020, 10, 583. [Google Scholar] [CrossRef]



| Country | Leading Marginality Factors | Affected Area (km2) | Affected Area of Total Utilized Agricultural Area (%) |
|---|---|---|---|
| Latvia | Adverse climate | 8980 | 30 |
| Excessive wetness | 3602 | 12 | |
| Limitations in rooting | 1475 | 5 | |
| Total: | 12,161 | 41 | |
| Spain | Adverse climate | 77,490 | 23 |
| Limitations in rooting | 76,179 | 22 | |
| Low soil fertility | 33,166 | 10 | |
| Total: | 148,496 | 44 | |
| Ukraine | Low soil fertility | 37,000 | 9 |
| Adverse climate | 30,000 | 7 | |
| Limitations in rooting | 29,100 | 7 | |
| Total: | 133,920 | 31 |
| Country | Location | Establishment Year | Marginality | Density, Plants ha−1 | Species | Treatment |
|---|---|---|---|---|---|---|
| Latvia | 56.69 N, 25.14 E | 2011 | Poor rooting conditions—unfavorable soil texture and stoniness | 2500; 1273; 1227; | Hybrid aspen | Control; Wood ash; Sewage sludge; Digestate |
| 1636 | Gray alder | Control; Wood ash; Sewage sludge | ||||
| 1636 | Black alder | |||||
| 1636 | Hybrid alder | |||||
| 1636 | Birch | |||||
| Spain | 41.36 N, 2.30 W | 2009 | Unfavorable soil texture, stoniness and soil organic carbon < 1% | 6666 | Siberian elm | Rain-fed; Irrigated |
| Ukraine | 48.99 N, 27.46 E | 2016 | Soil organic carbon < 1%, soil pH < 5 | 20,000 | Willow | Fertilizer (N60) |
| 2013 | Soil organic carbon < 1% | Control | ||||
| 2016; 2011; | Clay soil (clay content > 50%), soil organic carbon < 1% | |||||
| 2011; 2013; 2016; | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Celma, S.; Sanz, M.; Ciria, P.; Maliarenko, O.; Prysiazhniuk, O.; Daugaviete, M.; Lazdina, D.; von Cossel, M. Yield Performance of Woody Crops on Marginal Agricultural Land in Latvia, Spain and Ukraine. Agronomy 2022, 12, 908. https://doi.org/10.3390/agronomy12040908
Celma S, Sanz M, Ciria P, Maliarenko O, Prysiazhniuk O, Daugaviete M, Lazdina D, von Cossel M. Yield Performance of Woody Crops on Marginal Agricultural Land in Latvia, Spain and Ukraine. Agronomy. 2022; 12(4):908. https://doi.org/10.3390/agronomy12040908
Chicago/Turabian StyleCelma, Santa, Marina Sanz, Pilar Ciria, Oksana Maliarenko, Oleh Prysiazhniuk, Mudrite Daugaviete, Dagnija Lazdina, and Moritz von Cossel. 2022. "Yield Performance of Woody Crops on Marginal Agricultural Land in Latvia, Spain and Ukraine" Agronomy 12, no. 4: 908. https://doi.org/10.3390/agronomy12040908
APA StyleCelma, S., Sanz, M., Ciria, P., Maliarenko, O., Prysiazhniuk, O., Daugaviete, M., Lazdina, D., & von Cossel, M. (2022). Yield Performance of Woody Crops on Marginal Agricultural Land in Latvia, Spain and Ukraine. Agronomy, 12(4), 908. https://doi.org/10.3390/agronomy12040908








