Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection, Cultivation, and Preparation of Microbial Consortia for Seed Inoculation
2.2. Substrate Characterization, Soil Properties, and Fertilization
2.3. Experimental Setup and Culture Condition
2.4. Data Collection
2.5. Statistical Evaluation
3. Results
3.1. Non-Invasive Evaluation of Microbial Performance—Plant Length and Vegetation Indices
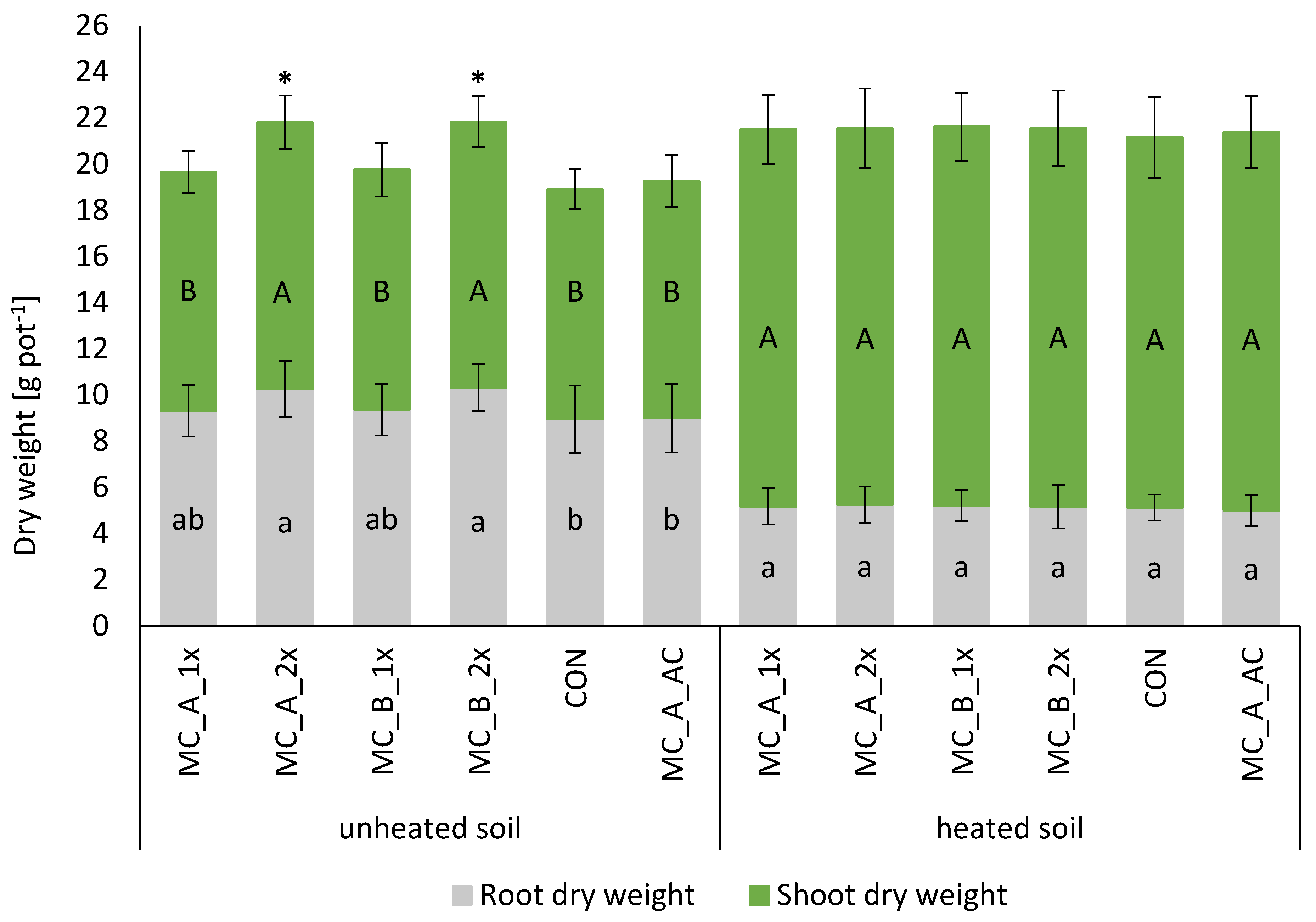
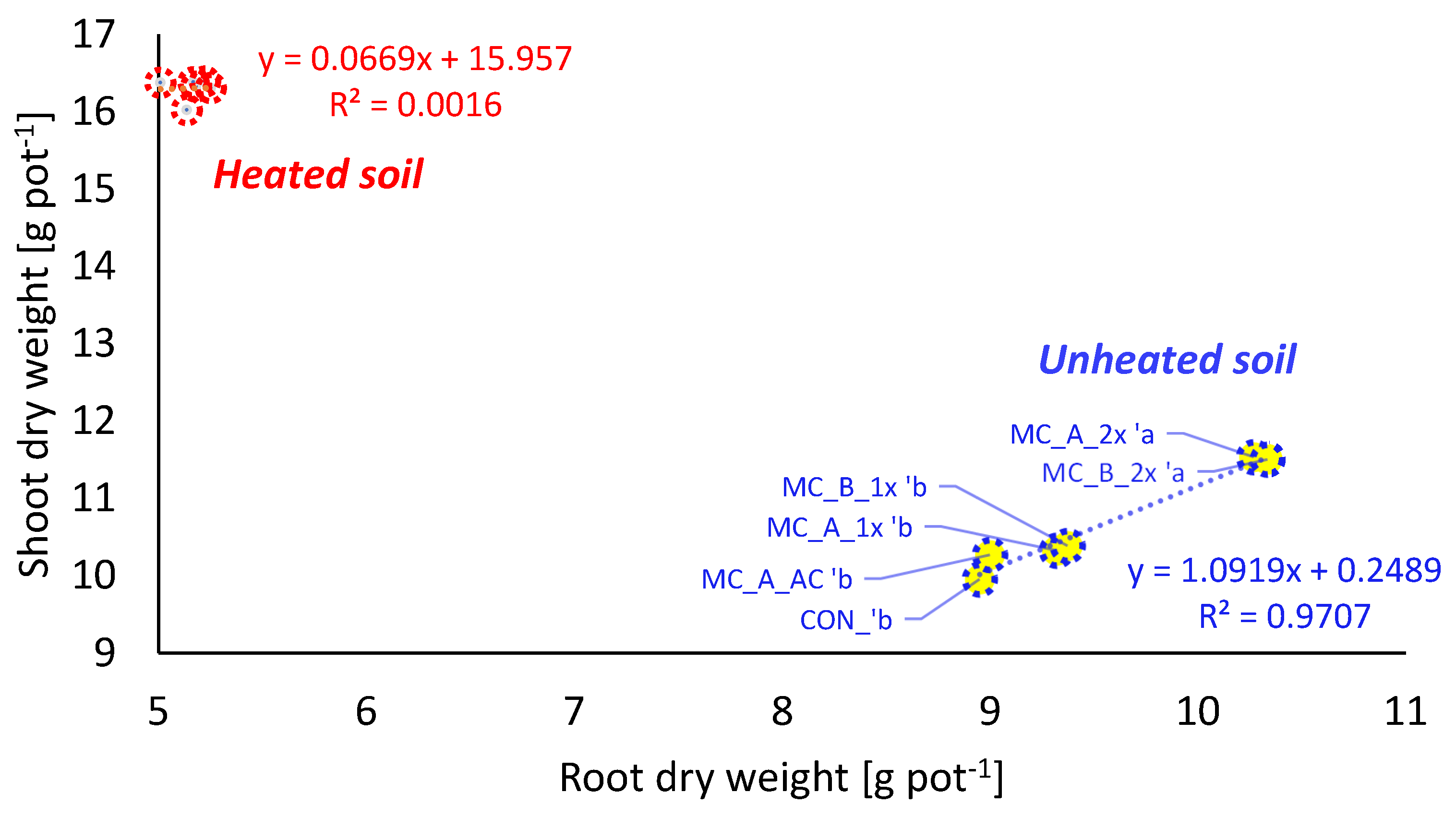
3.2. Plant Growth and Development—Shoot, Root, and Plant Biomass
3.3. Shoot Nutritional Status—Shoot Nutrient Contents and Uptake
3.4. Litterbag NIRS Analysis to Estimate Micorbial Acitivity
4. Discussion
4.1. Effects of Microbial Consortia on Crop Growth
4.2. Effects of Microbial Consortia on Litterbag Microbial Activity and Traceability
4.3. Effects of Soil Heating on Crop Growth
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schütz, L.; Gattinger, A.; Meier, M.; Müller, A.; Boller, T.; Mäder, P.; Mathimaran, N. Improving Crop Yield and Nutrient Use Efficiency via Biofertilization-A Global Meta-analysis. Front. Plant Sci. 2018, 8, 2204. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.; Garbeva, P.; Raaijmakers, J.M. The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol. Rev. 2013, 37, 634–663. [Google Scholar] [CrossRef] [PubMed]
- Kloepper, J.W.; Leong, J.; Teintze, M.; Schroth, M.N. Enhanced plant growth by siderophores produced by plant growth-promoting rhizobacteria. Nature 1980, 286, 885–886. [Google Scholar] [CrossRef]
- Bhattacharyya, P.N.; Jha, D.K. Plant growth-promoting rhizobacteria (PGPR): Emergence in agriculture. World J. Microbiol. Biotechnol. 2012, 28, 1327–1350. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Glick, B.R. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef]
- Martínez-Viveros, O.; Jorquera, M.; Crowley, D.; Gajardo, G.; Mora, M. Mechanisms and practical considerations involved in plant growth promotion by rhizobacteria. J. Soil Sci. Plant Nutr. 2010, 10, 293–319. [Google Scholar] [CrossRef]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Nasrulhaq Boyce, A. Role of Plant Growth Promoting Rhizobacteria in Agricultural Sustainability-A Review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Bradáčová, K.; Florea, A.; Bar-Tal, A.; Minz, D.; Yermiyahu, U.; Shawahna, R.; Kraut-Cohen, J.; Zolti, A.; Erel, R.; Dietel, K.; et al. Microbial Consortia versus Single-Strain Inoculants: An Advantage in PGPM-Assisted Tomato Production? Agronomy 2019, 9, 105. [Google Scholar] [CrossRef]
- Bradáčová, K.; Kandeler, E.; Berger, N.; Ludewig, U.; Neumann, G. Microbial consortia inoculants stimulate early growth of maize depending on nitrogen and phosphorus supply. Plant Soil Environ. 2020, 66, 105–112. [Google Scholar] [CrossRef]
- Di Benedetto, N.A.; Campaniello, D.; Bevilacqua, A.; Cataldi, M.P.; Sinigaglia, M.; Flagella, Z.; Corbo, M.R. Isolation, Screening, and Characterization of Plant-Growth-Promoting Bacteria from Durum Wheat Rhizosphere to Improve N and P Nutrient Use Efficiency. Microorganisms 2019, 7, 541. [Google Scholar] [CrossRef] [PubMed]
- Lavakush; Yadav, J.; Verma, J.P.; Jaiswal, D.K.; Kumar, A. Evaluation of PGPR and different concentration of phosphorus level on plant growth, yield and nutrient content of rice (Oryza sativa). Ecol. Eng. 2014, 62, 123–128. [Google Scholar] [CrossRef]
- Çakmakçı, R.; Erat, M.; Erdoğan, Ü.; Dönmez, M.F. The influence of plant growth–promoting rhizobacteria on growth and enzyme activities in wheat and spinach plants. J. Plant Nutr. Soil Sci. 2007, 170, 288–295. [Google Scholar] [CrossRef]
- Javaid, A.; Bajwa, R.; Anjum, T. Effect of heat-sterilization and EM (effective microorganisms) application on wheat (Triticum aestivum L.) grown in organic-amended sandy loam soil. Cereal Res. Commun. 2008, 36, 489–499. [Google Scholar] [CrossRef]
- Schenck zu Schweinsberg-Mickan, M.; Müller, T. Impact of effective microorganisms and other biofertilizers on soil microbial characteristics, organic-matter decomposition, and plant growth. J. Plant Nutr. Soil Sci. 2009, 172, 704–712. [Google Scholar] [CrossRef]
- Mayer, J.; Scheid, S.; Widmer, F.; Fließbach, A.; Oberholzer, H.-R. How effective are ‘Effective microorganisms® (EM)’? Results from a field study in temperate climate. Appl. Soil Ecol. 2010, 46, 230–239. [Google Scholar] [CrossRef]
- Mpanga, I.K.; Gomez-Genao, N.; Moradtalab, N.; Wanke, D.; Chrobaczek, V.; Ahmed, A.; Windisch, S.; Geistlinger, J.; Hafiz, F.B.; Walker, F.; et al. The role of N form supply for PGPM-host plant interactions in maize. J. Plant Nutr. Soil Sci. 2019, 182, 908–920. [Google Scholar] [CrossRef]
- Bashan, Y. Inoculants of plant growth-promoting bacteria for use in agriculture. Biotechnol. Adv. 1998, 16, 729–770. [Google Scholar] [CrossRef]
- Thomloudi, E.-E.; Tsalgatidou, P.C.; Douka, D.; Spantidos, T.-N.; Dimou, M.; Venieraki, A.; Katinakis, P. Multistrain versus single-strain plant growth promoting microbial inoculants-The compatibility issue. Hell. Plant Prot. J. 2019, 12, 61–77. [Google Scholar] [CrossRef]
- Tabacchioni, S.; Passato, S.; Ambrosino, P.; Huang, L.; Caldara, M.; Cantale, C.; Hett, J.; Del Fiore, A.; Fiore, A.; Schlüter, A.; et al. Identification of Beneficial Microbial Consortia and Bioactive Compounds with Potential as Plant Biostimulants for a Sustainable Agriculture. Microorganisms 2021, 9, 426. [Google Scholar] [CrossRef] [PubMed]
- Vishwakarma, K.; Kumar, N.; Shandilya, C.; Mohapatra, S.; Bhayana, S.; Varma, A. Revisiting Plant-Microbe Interactions and Microbial Consortia Application for Enhancing Sustainable Agriculture: A Review. Front. Microbiol. 2020, 11, 560406. [Google Scholar] [CrossRef] [PubMed]
- Bargaz, A.; Lyamlouli, K.; Chtouki, M.; Zeroual, Y.; Dhiba, D. Soil Microbial Resources for Improving Fertilizers Efficiency in an Integrated Plant Nutrient Management System. Front. Microbiol. 2018, 9, 1606. [Google Scholar] [CrossRef]
- Woo, S.L.; Pepe, O. Microbial Consortia: Promising Probiotics as Plant Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 1801. [Google Scholar] [CrossRef]
- Pérez-de-Luque, A.; Tille, S.; Johnson, I.; Pascual-Pardo, D.; Ton, J.; Cameron, D.D. The interactive effects of arbuscular mycorrhiza and plant growth-promoting rhizobacteria synergistically enhance host plant defences against pathogens. Sci. Rep. 2017, 7, 16409. [Google Scholar] [CrossRef]
- Masoero, G.; Oggiano, P.; Migliorini, P.; Taran, A.; Nuti, M.; Giovannetti, G. Litterbag-NIRS to Forecast Yield: A Horticultural Case with Biofertilizer Effectors. J. Soil Sci. Plant Nutr. 2021, 22, 186–200. [Google Scholar] [CrossRef]
- Baldi, E.; Toselli, M.; Masoero, G.; Nuti, M. Organic and Symbiotic Fertilization of Tomato Plants Monitored by Litterbag-NIRS and Foliar-NIRS Rapid Spectroscopic Methods. JAR 2020, 3, 9–26. [Google Scholar] [CrossRef]
- Volpato, S.; Masoero, G.; Giovannetti, G.; Nuti, M. Arbuscular Mycorrhizal Biofertilizers Sources in the Potato (Solanum Tuberosum) Plant show Interactions with Cultivars on Yield and Litter-bags Spectral Features. JAR 2020, 2, 9–17. [Google Scholar] [CrossRef]
- Volpato, S.; Masoero, G.; Mazzinelli, G.; Balconi, C.; Locatelli, S.; Lanzanova, C.; Ardigò, A.; Giovannetti, G.; Nuti, M. Spectroscopic and Foliar pH Model for Yield Prediction in a Symbiotic Corn Production. JAR 2019, 2, 1–18. [Google Scholar] [CrossRef][Green Version]
- Sohaib, M.; Zahir, Z.A.; Khan, M.Y.; Ans, M.; Asghar, H.N.; Yasin, S.; Al-Barakah, F.N.I. Comparative evaluation of different carrier-based multi-strain bacterial formulations to mitigate the salt stress in wheat. Saudi J. Biol. Sci. 2020, 27, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Miles, A.A.; Misra, S.S.; Irwin, J.O. The estimation of the bactericidal power of the blood. J. Hyg. 1938, 38, 732–749. [Google Scholar] [CrossRef] [PubMed]
- Verband Deutscher Landwirtschaftlicher Untersuchungs-und Forschungsanstalten. Handbuch der Landwirtschaftlichen Versuchs-und Untersuchungsmethodik: Methodenbuch Band I Die Untersuchung von Böden, 4th ed.; VDLUFA: Darmstadt, Germany, 1991. [Google Scholar]
- Quilliam, R.S.; Glanville, H.C.; Wade, S.C.; Jones, D.L. Life in the ‘charosphere’–Does biochar in agricultural soil provide a significant habitat for microorganisms? Soil Biol. Biochem. 2013, 65, 287–293. [Google Scholar] [CrossRef]
- Khan, M.S.; Zaidi, A. Synergistic Effects of the Inoculation with Plant Growth-Promoting Rhizobacteria and an Arbuscular Mycorrhizal Fungus on the Performance of Wheat. Turk. J. Agric. For. 2007, 6, 355–362. [Google Scholar]
- Baldi, E.; Gioacchini, P.; Montecchio, D.; Mocali, S.; Antonielli, L.; Masoero, G.; Toselli, M. Effect of Biofertilizers Application on Soil Biodiversity and Litter Degradation in a Commercial Apricot Orchard. Agronomy 2021, 11, 1116. [Google Scholar] [CrossRef]
- Rouse, J.; Haas, H.; Schell, J.; Deering, D. Monitoring Vegetation Systems in the Great Plains with ERTS. In Proceedings of the Third ERTS Symposium, NASA SP-351, Washington, DC, USA, 10–14 December 1973; pp. 309–317. [Google Scholar]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing Carotenoid Content in Plant Leaves with Reflectance Spectroscopy. Photochem. Photobiol. 2002, 75, 272. [Google Scholar] [CrossRef]
- Gamon, J.A.; Peñuelas, J.; Field, C.B. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ. 1992, 41, 35–44. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Anvironment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 16 February 2022).
- Chrominski, K.; Tkacz, M. Comparison of outlier detection methods in biomedical data. J. Med. Inform. Technol. 2010, 16, 89–94. [Google Scholar]
- Russel, L. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. R Package Version 1.4.8. 2020. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 16 February 2022).
- Campbell, R.C. Reference Sufficiency Ranges for Plant Analysis in the Southern Region of the United States; Southern Cooperative Series Bulletin #394; North Carolina Department of Agriculture and Consumer Services Agronomic Division: Raleigh, NC, USA, 2009; ISBN 1581613946.
- Bradáčová, K.; Sittinger, M.; Tietz, K.; Neuhäuser, B.; Kandeler, E.; Berger, N.; Ludewig, U.; Neumann, G. Maize Inoculation with Microbial Consortia: Contrasting Effects on Rhizosphere Activities, Nutrient Acquisition and Early Growth in Different Soils. Microorganisms 2019, 7, 329. [Google Scholar] [CrossRef]
- Cataldi, M.P.; Heuer, S.; Mauchline, T.H.; Wilkinson, M.D.; Masters-Clark, E.; Di Benedetto, N.A.; Corbo, M.R.; Flagella, Z. Effect of Plant Growth Promoting Bacteria on the Growth of Wheat Seedlings Subjected to Phosphate Starvation. Agronomy 2020, 10, 978. [Google Scholar] [CrossRef]
- Kumar, V.; Behl, R.K.; Narula, N. Establishment of phosphate-solubilizing strains of Azotobacter chroococcum in the rhizosphere and their effect on wheat cultivars under greenhouse conditions. Microbiol. Res. 2001, 156, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Egamberdiyeva, D. Plant-growth-promoting rhizobacteria isolated from a Calcisol in a semi-arid region of Uzbekistan: Biochemical characterization and effectiveness. J. Plant Nutr. Soil Sci. 2005, 168, 94–99. [Google Scholar] [CrossRef]
- Nguyen, M.L.; Glaes, J.; Spaepen, S.; Bodson, B.; Du Jardin, P.; Delaplace, P. Biostimulant effects of Bacillus strains on wheat from in vitro towards field conditions are modulated by nitrogen supply. J. Plant Nutr. Soil Sci. 2019, 182, 325–334. [Google Scholar] [CrossRef]
- Abbasi, M.K.; Sharif, S.; Kazmi, M.; Sultan, T.; Aslam, M. Isolation of plant growth promoting rhizobacteria from wheat rhizosphere and their effect on improving growth, yield and nutrient uptake of plants. Plant Biosyst. 2011, 145, 159–168. [Google Scholar] [CrossRef]
- Vacheron, J.; Desbrosses, G.; Bouffaud, M.-L.; Touraine, B.; Moënne-Loccoz, Y.; Muller, D.; Legendre, L.; Wisniewski-Dyé, F.; Prigent-Combaret, C. Plant growth-promoting rhizobacteria and root system functioning. Front. Plant Sci. 2013, 4, 356. [Google Scholar] [CrossRef] [PubMed]
- Kapulnik, Y.; Okon, Y.; Henis, Y. Changes in root morphology of wheat caused by Azospirillum inoculation. Can. J. Microbiol. 1985, 31, 881–887. [Google Scholar] [CrossRef]
- Brimecombe, M.J.; De Leij, F.; Lynch, J.M. Effect of introduced Pseudomonas fluorescens strains on the uptake of nitrogen by wheat from 15N-enriched organic residues. World J. Microbiol. Biotechnol. 1999, 15, 417–423. [Google Scholar] [CrossRef]
- Kudoyarova, G.R.; Vysotskaya, L.B.; Arkhipova, T.N.; Kuzmina, L.Y.; Galimsyanova, N.F.; Sidorova, L.V.; Gabbasova, I.M.; Melentiev, A.I.; Veselov, S.Y. Effect of auxin producing and phosphate solubilizing bacteria on mobility of soil phosphorus, growth rate, and P acquisition by wheat plants. Acta Physiol. Plant. 2017, 39, 253. [Google Scholar] [CrossRef]
- Viscardi, S.; Ventorino, V.; Duran, P.; Maggio, A.; De Pascale, S.; Mora, M.; Pepe, O. Assessment of plant growth promoting activities and abiotic stress tolerance of Azotobacter chroococcum strains for a potential use in sustainable agriculture. J. Soil Sci. Plant Nutr. 2016, 3, 848–863. [Google Scholar] [CrossRef]
- Wani, S.A.; Chand, S.; Wani, M.A.; Ramzan, M.; Hakeem, K.R. Azotobacter chroococcum–A Potential Biofertilizer in Agriculture: An Overview. In Soil Science: Agricultural and Environmental Prospectives; Hakeem, K.R., Akhtar, J., Sabir, M., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 333–348. ISBN 978-3-319-34449-2. [Google Scholar]
- Ferreira, C.M.H.; López-Rayo, S.; Lucena, J.J.; Soares, E.V.; Soares, H.M.V.M. Evaluation of the Efficacy of Two New Biotechnological-Based Freeze-Dried Fertilizers for Sustainable Fe Deficiency Correction of Soybean Plants Grown in Calcareous Soils. Front. Plant Sci. 2019, 10, 1335. [Google Scholar] [CrossRef]
- Noar, J.D.; Bruno-Bárcena, J.M. Azotobacter vinelandii: The source of 100 years of discoveries and many more to come. Microbiology 2018, 164, 421–436. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Narula, N. Solubilization of inorganic phosphates and growth emergence of wheat as affected by Azotobacter chroococcum mutants. Biol. Fertil. Soils 1999, 28, 301–305. [Google Scholar] [CrossRef]
- Mpanga, I.K.; Nkebiwe, P.M.; Kuhlmann, M.; Cozzolino, V.; Piccolo, A.; Geistlinger, J.; Berger, N.; Ludewig, U.; Neumann, G. The Form of N Supply Determines Plant Growth Promotion by P-Solubilizing Microorganisms in Maize. Microorganisms 2019, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Vinci, G.; Cozzolino, V.; Mazzei, P.; Monda, H.; Spaccini, R.; Piccolo, A. An alternative to mineral phosphorus fertilizers: The combined effects of Trichoderma harzianum and compost on Zea mays, as revealed by 1H NMR and GC-MS metabolomics. PLoS ONE 2018, 13, e0209664. [Google Scholar] [CrossRef] [PubMed]
- Wani, S.; Chand, S.; Ali, T. Potential Use of Azotobacter chroococcum in Crop Production: An Overview. Curr. Agric. Res. 2013, 1, 35–38. [Google Scholar] [CrossRef]
- Kizilkaya, R. Nitrogen fixation capacity of Azotobacter spp. strains isolated from soils in different ecosystems and relationship between them and the microbiological properties of soils. J. Environ. Biol. 2009, 30, 73–82. [Google Scholar] [PubMed]
- Kapp Junior, C.; Guimarães, A.M.; Caires, E.F. Use of active canopy sensors to discriminate wheat response to nitrogen fertilization under no-tillage. Eng. Agric. 2016, 36, 886–894. [Google Scholar] [CrossRef][Green Version]
- Dobbelaere, S.; Croonenborghs, A.; Thys, A.; Ptacek, D.; Okon, Y.; Vanderleyden, J. Effect of inoculation with wild type Azospirillum brasilense and A. irakense strains on development and nitrogen uptake of spring wheat and grain maize. Biol. Fertil. Soils 2002, 36, 284–297. [Google Scholar] [CrossRef]
- Narula, N.; Kumar, V.; Singh, B.; Bhatia, R.; Lakshminarayana, K. Impact of Biofertilizers on grain yield in spring wheat under varying fertility conditions and wheat-cotton rotation. Arch. Agron. Soil Sci. 2005, 51, 79–89. [Google Scholar] [CrossRef]
- Ozturk, A.; Caglar, O.; Sahin, F. Yield response of wheat and barley to inoculation of plant growth promoting rhizobacteria at various levels of nitrogen fertilization. J. Plant Nutr. Soil Sci. 2003, 166, 262–266. [Google Scholar] [CrossRef]
- Shaharoona, B.; Naveed, M.; Arshad, M.; Zahir, Z.A. Fertilizer-dependent efficiency of Pseudomonads for improving growth, yield, and nutrient use efficiency of wheat (Triticum aestivum L.). Appl. Microbiol. Biotechnol. 2008, 79, 147–155. [Google Scholar] [CrossRef] [PubMed]
- van Veen, J.A.; van Overbeek, L.S.; van Elsas, J.D. Fate and activity of microorganisms introduced into soil. Microbiol. Mol. Biol. Rev. 1997, 61, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Malowany, S.N.; Newton, J.D. Studies on stream sterilization of soils; some effects on physical, chemical, and biological properties. Can. J. Res. 1947, 25, 189–208. [Google Scholar] [CrossRef] [PubMed]
- Roux-Michollet, D.; Czarnes, S.; Adam, B.; Berry, D.; Commeaux, C.; Guillaumaud, N.; Le Roux, X.; Clays-Josserand, A. Effects of steam disinfestation on community structure, abundance and activity of heterotrophic, denitrifying and nitrifying bacteria in an organic farming soil. Soil Biol. Biochem. 2008, 40, 1836–1845. [Google Scholar] [CrossRef]
- Gelsomino, A.; Petrovičová, B.; Zaffina, F.; Peruzzi, A. Chemical and microbial properties in a greenhouse loamy soil after steam disinfestation alone or combined with CaO addition. Soil Biol. Biochem. 2010, 42, 1091–1100. [Google Scholar] [CrossRef]
- Kitur, B.K.; Frye, W.W. Effects of Heating on Soil Chemical Properties and Growth and Nutrient Composition of Corn and Millet. Soil Sci. Soc. Am. J. 1983, 47, 91–94. [Google Scholar] [CrossRef]
- Endlweber, K.; Scheu, S. Establishing arbuscular mycorrhiza-free soil: A comparison of six methods and their effects on nutrient mobilization. Appl. Soil Ecol. 2006, 34, 276–279. [Google Scholar] [CrossRef]
- Marschner, H.; Kirkby, E.A.; Cakmak, I. Effect of mineral nutritional status on shoot-root partitioning of photoassimilates and cycling of mineral nutrients. J. Exp. Bot. 1996, 47, 1255–1263. [Google Scholar] [CrossRef]
- Brouwer, R. Nutritive influences on the distribution of dry matter in the plant. NJAS 1962, 10, 399–408. [Google Scholar] [CrossRef]
- Ericsson, T. Growth and shoot: Root ratio of seedlings in relation to nutrient availability. Plant Soil 1995, 168–169, 205–214. [Google Scholar] [CrossRef]
- Jager, G.; van der Boon, J.; Rauw, G. The influence of soil steaming on some properties of the soil and on the growth and heading of winter glasshouse lettuce. II. The reaction of the crop. NJAS 1969, 17, 241–245. [Google Scholar] [CrossRef]
- Sonneveld, C. Changes in Chemical Properties of Soil Caused by Steam Sterilization. In Soil Disinfestation; Mulder, D., Ed.; Elsevier: Amsterdam, The Netherlands, 1979; pp. 39–50. ISBN 9780444416926. [Google Scholar]
| Microbial Strain | MC 1 | Properties [22] | Pellet (I) CFU 2 mL−1 or Spores mL−1 | Zeolite (II) CFU g−1 or Spores g−1 |
|---|---|---|---|---|
| Azotobacter chroococcum LS132 | A | N2-fixation | 3.00 × 1010 | 2.80 × 107 |
| Bacillus licheniformis PS141 | A | IAA 3 production | 3.20 × 109 | 1.80 × 106 |
| Komagataella pastoris PP59 | A | PGP 4 | 1.50 × 1010 | 1.80 × 105 |
| Paraburkholderia tropica MDIIIAzo225 | A | N2-fixation | 5.00 × 109 | 2.80 × 103 |
| Pseudomonasgranadensis A23/T3c | A | PGP | 2.50 × 1011 | 4.80 × 106 |
| Trichoderma harzianum TH01 * | A | PGP | 2.20 × 1010 | 5.00 × 107 |
| Azotobacter vinelandii DSM 2289 | B | N2-fixation, siderophore production | 1.10 × 1011 | 1.90 × 107 |
| Bacillusamyloliquefaciens LMG 9814 | B | Alpha-glucosidase, alpha-amylase, and isoamylase production | 5.00 × 109 | 3.95 × 106 |
| Bacillussp. BV84 | B | Biocontrol/PGP | 1.00 × 109 | 4.00 × 106 |
| Pseudomonas fluorescens DR54 | B | Biocontrol | 1.06 × 1011 | 5.00 × 106 |
| Rahnella aquatilis BB23/T4d | B | PGP | 8.00 × 1011 | 7.00 × 105 |
| pH | P | K | Mg | Na | Fe | Cu | Mn | Zn | B | Ca | Mo | TN 3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Value 1 | 6.7 | 100 | 200 | 120 | 8 | 270 | 5.4 | 430 | 13 | 0.41 | 1731 | <0.04 | 0.12 |
| Level 2 | C | C | D | E | - | - | E | E | E | C | - | A | - |
| Analysis of Plant Length and Different Vegetation Indices of Wheat Plants | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Main Effects | Microbial Inoculation | Soil Treatment | Trial Replication 5 | ||||||||
| Parameter | DAS 1 | MC_A_1x | MC_A_2x | MC_B_1x | MC_B_2x | CON | MC_A_AC | Unheated Soil | Heated Soil | Trial 1 | Trial 2 |
| Plant length (cm) | 28 | 43.7 | 44.0 | 43.7 | 44.0 | 43.4 | 42.9 | 43.8 | 43.4 | 43.1 b | 44.1 a |
| 42 | 53.6 | 54.8 | 53.6 | 54.3 | 53.3 | 53.8 | 53.4 b | 54.3 a | 53.3 b | 54.4 a | |
| 56 | 60.1 | 60.5 | 61.1 | 61.0 | 58.7 | 59.2 | 58.8 b | 61.4 a | 58.8 b | 61.3 a | |
| NDVI 2 | 28 | 0.574 | 0.575 | 0.571 | 0.561 | 0.568 | 0.574 | 0.555 b | 0.586 a | 0.566 b | 0.575 a |
| 42 | 0.601 | 0.601 | 0.593 | 0.603 | 0.595 | 0.592 | 0.589 b | 0.606 a | 0.607 a | 0.589 b | |
| 56 | 0.611 | 0.609 | 0.605 | 0.606 | 0.609 | 0.608 | 0.601 b | 0.615 a | 0.611 a | 0.604 b | |
| PRI 3 | 28 | 0.031 | 0.032 | 0.032 | 0.030 | 0.030 | 0.030 | 0.030 b | 0.032 a | 0.039 a | 0.023 b |
| 42 | 0.031 | 0.032 | 0.031 | 0.032 | 0.032 | 0.031 | 0.032 | 0.031 | 0.020 b | 0.043 a | |
| 56 | 0.022 | 0.022 | 0.022 | 0.022 | 0.023 | 0.023 | 0.023 | 0.022 | 0.025 a | 0.021 b | |
| CRI 4 | 28 | 2.701 | 2.718 | 2.701 | 2.628 | 2.687 | 2.711 | 2.649 b | 2.734 a | 2.436 b | 2.946 a |
| 42 | 2.756 | 2.779 | 2.735 | 2.766 | 2.773 | 2.714 | 2.782 a | 2.726 b | 2.915 a | 2.592 b | |
| 56 | 3.093 | 3.029 | 3.033 | 3.041 | 3.083 | 3.057 | 3.134 a | 2.978 b | 3.103 a | 3.009 b | |
| Wheat Shoot Nutrient Uptake (mg pot−1) | ||||||
|---|---|---|---|---|---|---|
| N Uptake | P Uptake | K Uptake | ||||
| Parameters | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 |
| MC_A_1x | 354.9 bc | 376.5 abc | 44.6 | 35.4 | 746.0 | 722.8 a |
| MC_A_2x | 385.1 a | 400.0 a | 47.9 | 39.4 | 782.3 | 742.5 a |
| MC_B_1x | 376.5 ab | 388.1 ab | 46.4 | 38.4 | 788.7 | 722.0 a |
| MC_B_2x | 366.9 abc | 398.9 ab | 45.0 | 40.8 | 810.8 | 755.6 a |
| CON | 344.5 c | 340.1 c | 47.1 | 38.4 | 774.7 | 639.5 b |
| MC_A_AC | 346.1 bc | 352.3 bc | 48.6 | 42.6 | 806.4 | 694.0 ab |
| Unheated soil | 223.1 b | 207.9 b | 34.8 b | 28.6 b | 557.4 b | 528.4 b |
| Heated soil | 501.6 a | 544.1 a | 58.3 a | 49.7 a | 1012.2 a | 897.1 a |
| LS Means for Microbial Consortia (MC) Treatments | Contrasts | Experimental Effects | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AK 1 | T 2 | Variables 3 | Hay | Decay | MC_ A_1x | MC_ A_2x | MC_ B_1x | MC_ B_2x | CON | MC_ A_AC | MC 5 vs. CON | MC_A_AC vs. CON | Trial 2 vs. GM 6 in % | Heated vs. GM 6 in % |
| ADL | r | Acid Detergent Lignin | 13.03 | −41% | 7.80 | 7.69 | 8.13 | 7.56 | 7.11 | 7.74 | 0.68 * | 0.63 * | −5.0% | −1.7% |
| CF | r | Crude Fiber | 21.54 | −38% | 13.48 | 13.43 | 14.26 | 12.87 | 12.02 | 13.76 | 1.49 | 1.74 | −10%* | −3.3% |
| ADF | r | Acid Detergent Fiber | 37.96 | −22% | 31.00 | 29.13 | 31.51 | 28.90 | 27.80 | 29.75 | 2.33 * | 1.95 | −3.2% | −1.2% |
| NDFD | r | NDF Digestibility | 67.52 | −17% | 55.67 | 55.46 | 56.90 | 55.38 | 54.83 | 56.55 | 1.02 | 1.72 * | 1.4%* | −0.9% |
| DNDF | r | Digestible NDF | 30.40 | −17% | 25.31 | 25.23 | 25.90 | 25.22 | 24.89 | 25.71 | 0.52 | 0.82 * | 1.3%* | −0.9% |
| Cell | r | Cellulose | 24.44 | −10% | 23.19 | 21.44 | 23.38 | 21.34 | 20.69 | 22.01 | 1.65 * | 1.32 | −2.6% | −1.1% |
| E | r | Gross Energy 4 | 16.95 | −6% | 16.00 | 15.97 | 16.06 | 15.98 | 15.88 | 15.98 | 0.12 * | 0.09 | −0.3% | −0.1% |
| EE | r | Ether Extract | 2.98 | −5% | 2.83 | 2.82 | 2.82 | 2.81 | 2.84 | 2.83 | −0.02 | −0.01 | −0.5% | −0.1% |
| NDF | k | Neutral Detergent Fiber | 45.02 | 1% | 45.46 | 45.48 | 45.52 | 45.54 | 45.40 | 45.47 | 0.10 * | 0.07 | 0.0% | 0.0% |
| NFE | k | N-Free Extract | 45.92 | 6% | 48.53 | 48.65 | 48.28 | 48.60 | 48.70 | 48.41 | −0.19 | −0.30 | 0.7%* | 0.2% * |
| IVTD | k | Total Digestibility | 67.58 | 20% | 80.77 | 81.66 | 80.92 | 81.76 | 81.50 | 81.28 | −0.22 | −0.22 | 0.2% | 0.1% * |
| INDF | k | Indigestible NDF | 14.62 | 37% | 20.15 | 20.25 | 19.62 | 20.32 | 20.51 | 19.75 | −0.42 | −0.75 * | −1.8%* | 1.2% * |
| CP | k | Crude Protein | 7.88 | 65% | 12.64 | 13.27 | 12.60 | 13.20 | 13.34 | 12.99 | −0.41 | −0.34 | 1.0% | 0.5% |
| CMI | k | Crop Mat. Index | 0.45 | 120% | 0.98 | 0.98 | 1.01 | 0.96 | 1.00 | 0.96 | −0.01 | −0.04 | −1.7% | 1.5%* |
| Hemi | k | Hemicellulose | 7.06 | 124% | 14.46 | 16.35 | 14.01 | 16.64 | 17.60 | 15.72 | −2.2 * | −1.88 | 6.1% | 2.5% |
| Ash | k | Ash | 6.05 | 201% | 17.80 | 18.33 | 17.44 | 18.62 | 19.10 | 18.15 | −1.1 * | −0.95 | 3.6% | 1.3% * |
| r | m | r_Fingerprinting | 4.94 | 5.46 | 4.48 | 5.59 | 6.09 | 5.06 | −0.97 * | −1.03 * | 5.4%* | 4.8% * | ||
| k | m | k_Fingerprinting | 5.78 | 6.30 | 5.60 | 6.38 | 6.57 | 6.02 | −0.55 * | −0.55 | 3.5% | 2.2% * | ||
| r-k | m | r-to-k Difference | −0.83 | −0.84 | −1.12 | −0.79 | −0.48 | −0.96 | −0.42 * | −0.48 * | −106%* | −145%* | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hett, J.; Neuhoff, D.; Döring, T.F.; Masoero, G.; Ercole, E.; Bevivino, A. Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity. Agronomy 2022, 12, 899. https://doi.org/10.3390/agronomy12040899
Hett J, Neuhoff D, Döring TF, Masoero G, Ercole E, Bevivino A. Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity. Agronomy. 2022; 12(4):899. https://doi.org/10.3390/agronomy12040899
Chicago/Turabian StyleHett, Jonas, Daniel Neuhoff, Thomas F. Döring, Giorgio Masoero, Enrico Ercole, and Annamaria Bevivino. 2022. "Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity" Agronomy 12, no. 4: 899. https://doi.org/10.3390/agronomy12040899
APA StyleHett, J., Neuhoff, D., Döring, T. F., Masoero, G., Ercole, E., & Bevivino, A. (2022). Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity. Agronomy, 12(4), 899. https://doi.org/10.3390/agronomy12040899









