Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Dosage Optimisation
2.3. Field Experiment
2.4. Growth, Yield and Water Status Measures
2.5. Protein, Carbohydrate, and Mineral Determinations from the Maize Flour
2.6. Statistical Procedures
3. Results
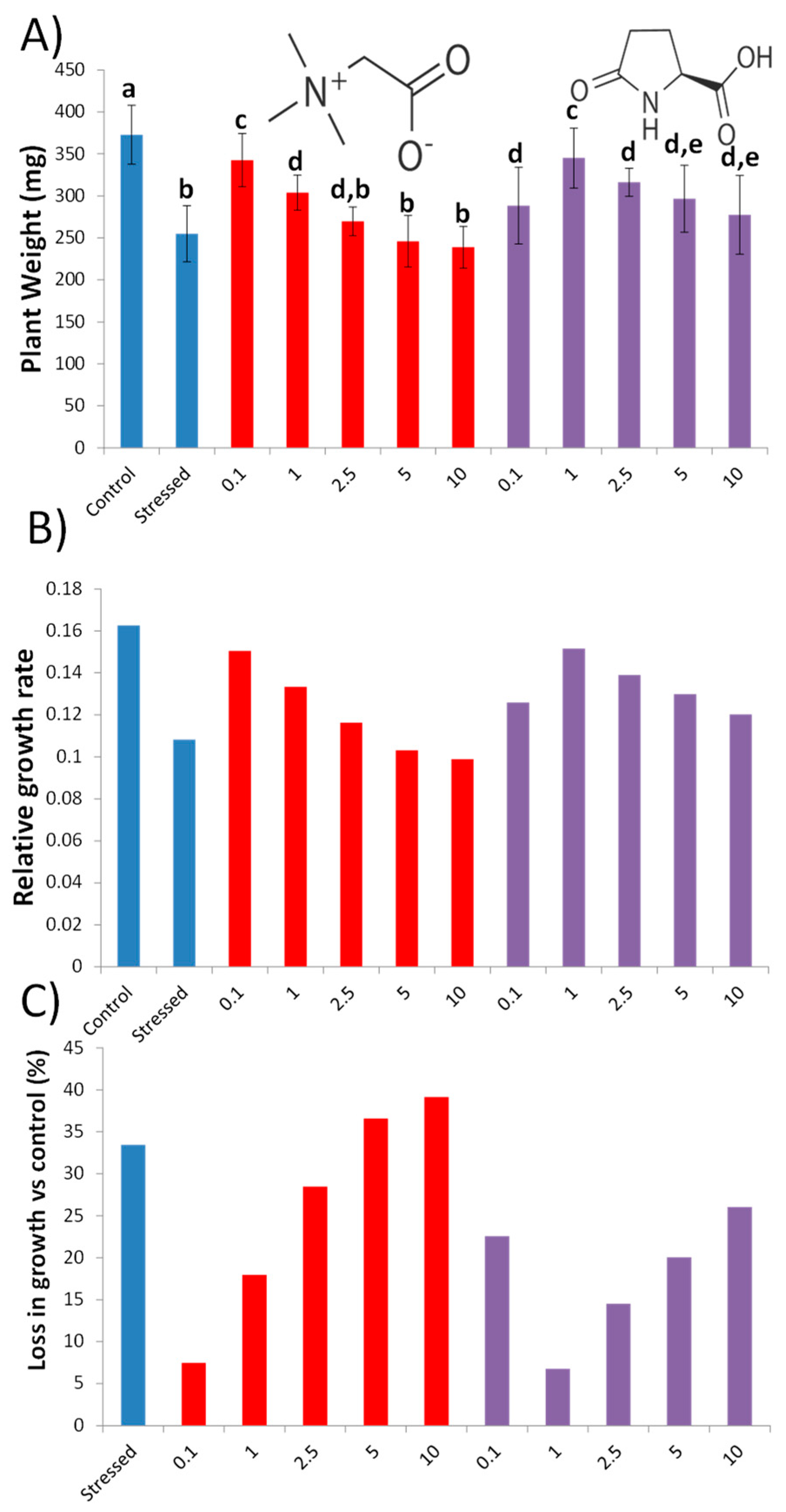
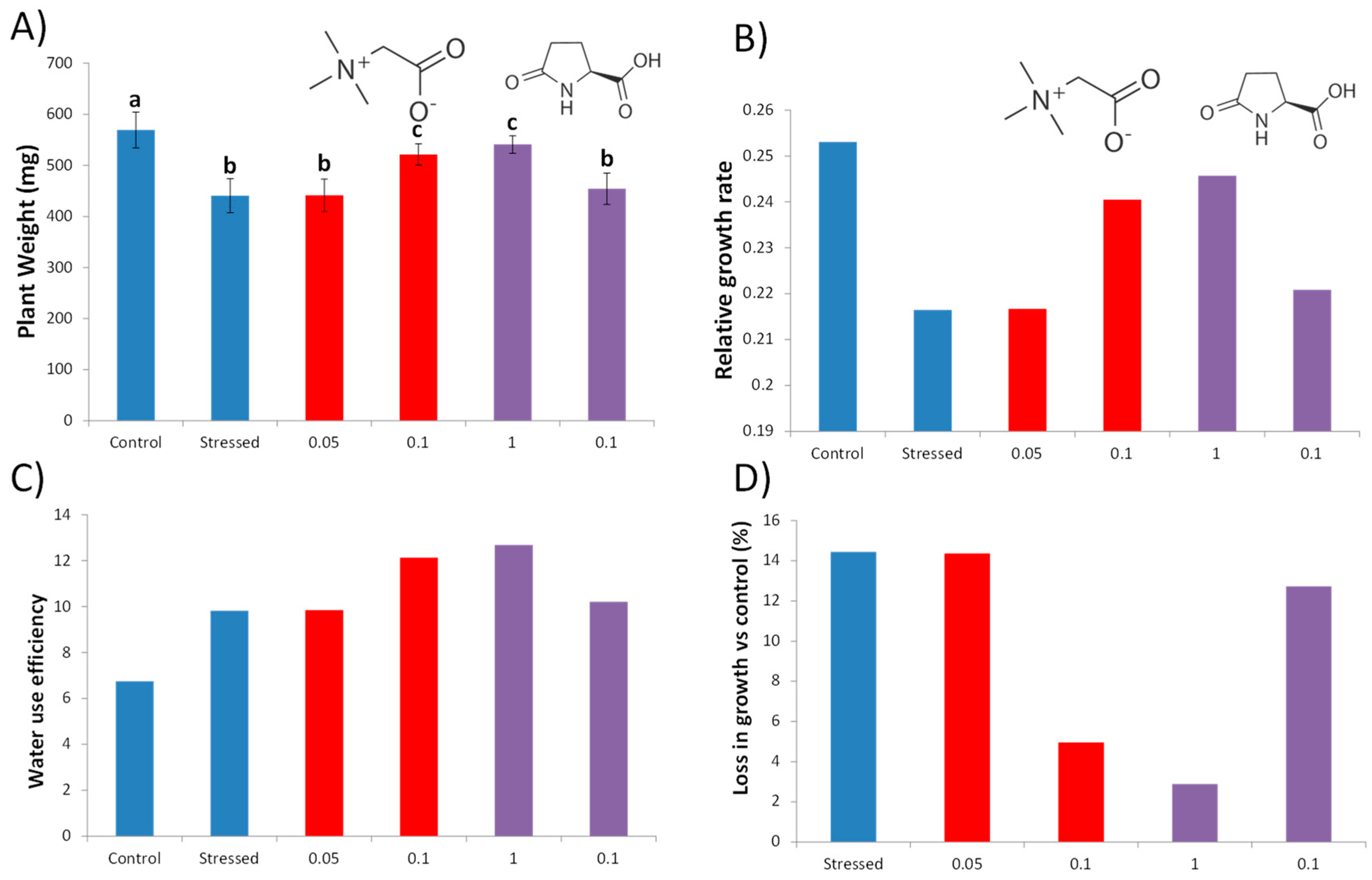
3.1. Glycine Betaine and L-Pyroglutamic Acid Improve Drought Tolerance under Water Deficit Stress
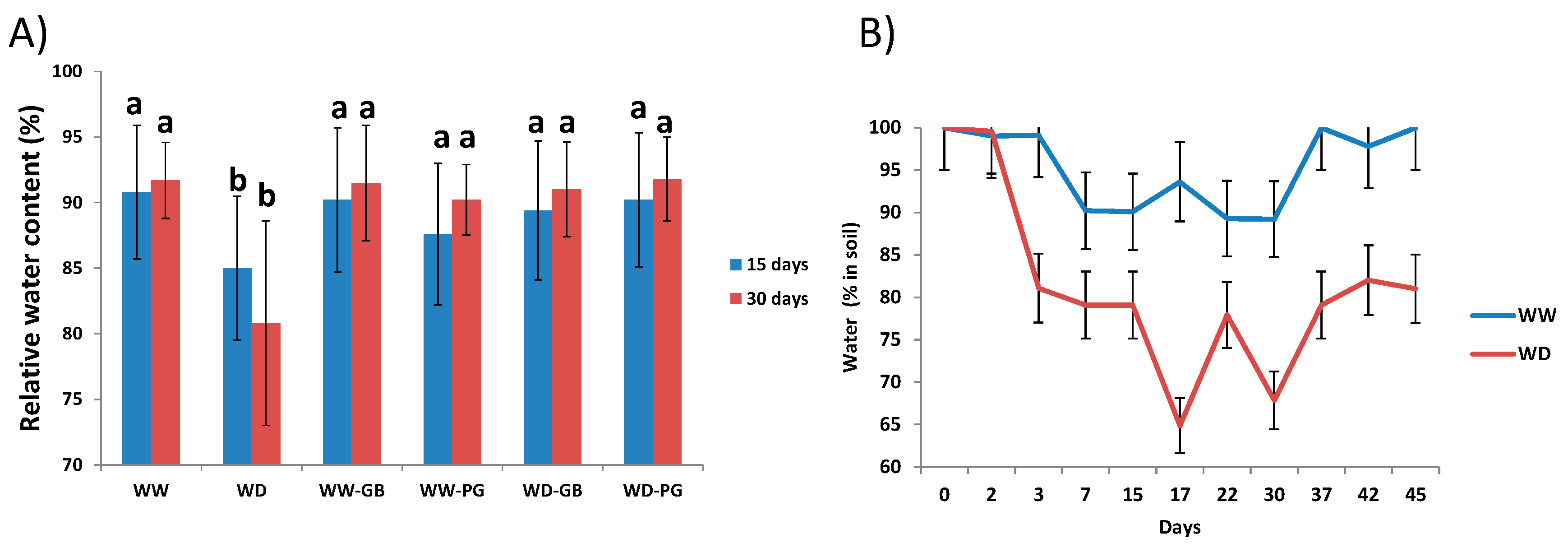
3.2. Biostimulants Are Capable of Ameliorating Negative Effects Caused by 20% Less Watering under Field Conditions
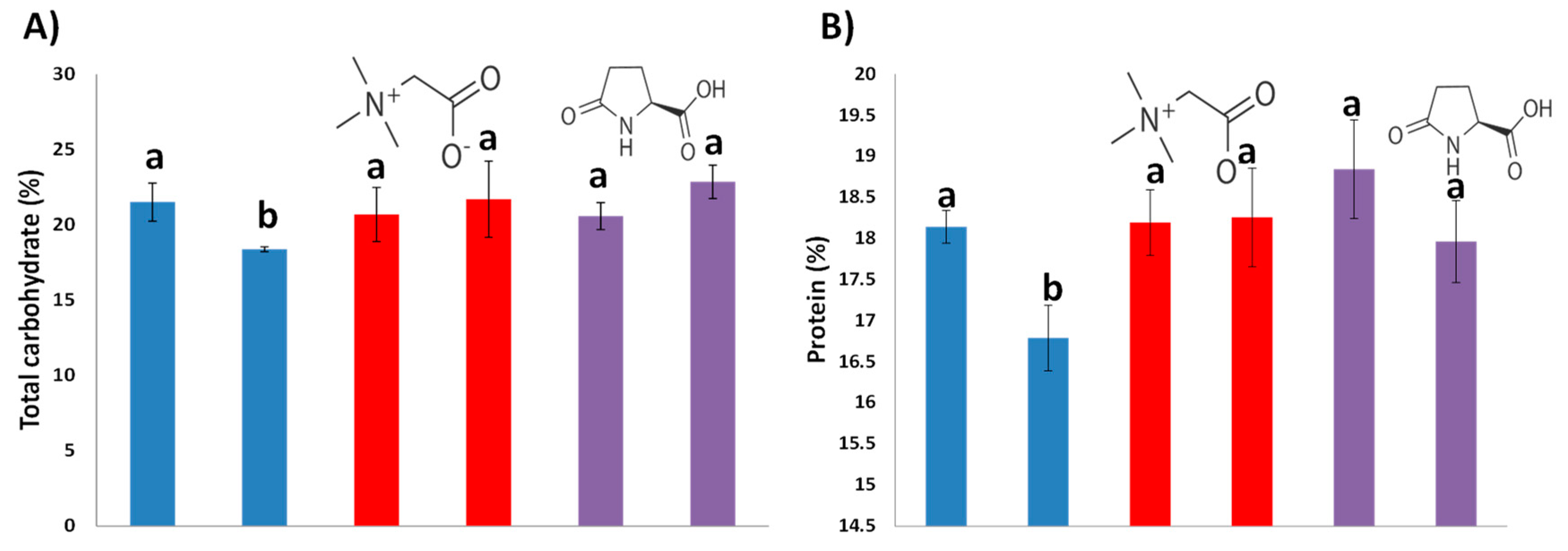
3.3. Nutritional Quality of Maize Flour Using Biostimulants
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- He, Y.; Fang, J.; Xu, W.; Shi, P. Substantial Increase of Compound Droughts and Heatwaves in Wheat Growing Seasons Worldwide. Int. J. Climatol. 2021, in press. [Google Scholar] [CrossRef]
- Lopez-Nicolas, A.; Pulido-Velazquez, M.; Macian-Sorribes, H. Economic Risk Assessment of Drought Impacts on Irrigated Agriculture. J. Hydrol. 2017, 550, 580–589. [Google Scholar] [CrossRef]
- Howitt, R.; MacEwan, D.; Medellín-Azuara, J.; Lund, J.; Sumner, D. Economic Analysis of the 2015 Drought for California Agriculture; University of California: Davis, CA, USA, 2015. [Google Scholar]
- GAR Special Report on Drought. 2021. Available online: https://www.undrr.org/publication/gar-special-report-drought-2021 (accessed on 26 June 2021).
- Dolan, F.; Lamontagne, J.; Link, R.; Hejazi, M.; Reed, P.; Edmonds, J. Evaluating the Economic Impact of Water Scarcity in a Changing World. Nat. Commun. 2021, 12, 1915. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Arias, D.; Morales-Sierra, S.; García-Machado, F.J.; García-García, A.L.; Luis, J.C.; Valdés, F.; Sandalio, L.M.; Hernández-Suárez, M.; Borges, A.A. Rejected Brine Recycling in Hydroponic and Thermo-Solar Evaporation Systems for Leisure and Tourist Facilities. Changing Waste into Raw Material. Desalination 2020, 496, 114443. [Google Scholar] [CrossRef]
- March, H.; Saurí, D.; Rico-Amorós, A.M. The End of Scarcity? Water Desalination as the New Cornucopia for Mediterranean Spain. J. Hydrol. 2014, 519, 2642–2651. [Google Scholar] [CrossRef] [Green Version]
- Carson, R.S. “Not in My Backyard” Is Not Sustainable. INCOSE Int. Symp. 2017, 27, 1749–1766. [Google Scholar] [CrossRef]
- López-Serrano, M.J.; Velasco-Muñoz, J.F.; Aznar-Sánchez, J.A.; Román-Sánchez, I.M. Economic Analysis of the Use of Reclaimed Water in Agriculture in Southeastern Spain, A Mediterranean Region. Agronomy 2021, 11, 2218. [Google Scholar] [CrossRef]
- García-García, A.L.; García-Machado, F.J.; Borges, A.A.; Morales-Sierra, S.; Boto, A.; Jiménez-Arias, D. Pure Organic Active Compounds Against Abiotic Stress: A Biostimulant Overview. Front. Plant Sci. 2020, 11, 1839. [Google Scholar] [CrossRef]
- Jiménez-Arias, D.; Morales-Sierra, S.; Borges, A.A.; Díaz, D.D. Biostimulant Nanoencapsulation: The New Keystone to Fight Hunger. J. Agric. Food Chem. 2020, 68, 7083–7085. [Google Scholar] [CrossRef]
- Jiménez-Arias, D.; García-Machado, F.J.; Morales-Sierra, S.; García-García, A.L.; Herrera, A.J.; Valdés, F.; Luis, J.C.; Borges, A.A. A Beginner’s Guide to Osmoprotection by Biostimulants. Plants 2021, 10, 363. [Google Scholar] [CrossRef]
- Jiménez-Arias, D.; García-Machado, F.J.; Morales-Sierra, S.; Luis, J.C.; Suarez, E.; Hernández, M.; Valdés, F.; Borges, A.A. Lettuce Plants Treated with L-Pyroglutamic Acid Increase Yield under Water Deficit Stress. Environ. Exp. Bot. 2019, 158, 215–222. [Google Scholar] [CrossRef] [Green Version]
- Binns, J. Farm to Fork Strategy. Available online: https://ec.europa.eu/food/farm2fork_en (accessed on 26 March 2021).
- du Jardin, P. Plant Biostimulants: Definition, Concept, Main Categories and Regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef] [Green Version]
- Sible, C.N.; Seebauer, J.R.; Below, F.E. Plant Biostimulants: A Categorical Review, Their Implications for Row Crop Production, and Relation to Soil Health Indicators. Agronomy 2021, 11, 1297. [Google Scholar] [CrossRef]
- Madende, M.; Hayes, M. Fish By-Product Use as Biostimulants: An Overview of the Current State of the Art, Including Relevant Legislation and Regulations within the EU and USA. Molecules 2020, 25, 1122. [Google Scholar] [CrossRef] [Green Version]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The Role of Biostimulants and Bioeffectors as Alleviators of Abiotic Stress in Crop Plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef] [Green Version]
- Sofy, M.R.; Elhawat, N. Tarek Alshaal Glycine Betaine Counters Salinity Stress by Maintaining High K+/Na+ Ratio and Antioxidant Defense via Limiting Na+ Uptake in Common Bean (Phaseolus vulgaris L.). Ecotoxicol. Environ. Saf. 2020, 200, 110732. [Google Scholar] [CrossRef]
- Rasheed, R.; Iqbal, M.; Ashraf, M.A.; Hussain, I.; Shafiq, F.; Yousaf, A.; Zaheer, A. Glycine Betaine Counteracts the Inhibitory Effects of Waterlogging on Growth, Photosynthetic Pigments, Oxidative Defence System, Nutrient Composition, and Fruit Quality in Tomato. J. Hortic. Sci. Biotechnol. 2018, 93, 385–391. [Google Scholar] [CrossRef]
- Hamani, A.K.M.; Wang, G.; Soothar, M.K.; Shen, X.; Gao, Y.; Qiu, R.; Mehmood, F. Responses of Leaf Gas Exchange Attributes, Photosynthetic Pigments and Antioxidant Enzymes in NaCl-Stressed Cotton (Gossypium hirsutum L.) Seedlings to Exogenous Glycine Betaine and Salicylic Acid. BMC Plant Biol. 2020, 20, 434. [Google Scholar] [CrossRef]
- de Oliveira Maia Júnior, S.; de Andrade, J.R.; dos Santos, C.M.; Santos, J.V.; dos Santos Silva, L.K.; Aprígio Clemente, P.R.; Ferreira, V.M.; Silva, J.V.; Endres, L. Foliar-Applied Glycine Betaine Minimizes Drought Stress-Related Impact to Gas Exchange and the Photochemical Efficiency of PSII in Sugarcane. Theor. Exp. Plant Physiol. 2020, 32, 315–329. [Google Scholar] [CrossRef]
- Shemi, R.; Wang, R.; Gheith, E.-S.M.S.; Hussain, H.A.; Hussain, S.; Irfan, M.; Cholidah, L.; Zhang, K.; Zhang, S.; Wang, L. Effects of Salicylic Acid, Zinc and Glycine Betaine on Morpho-Physiological Growth and Yield of Maize under Drought Stress. Sci. Rep. 2021, 11, 3195. [Google Scholar] [CrossRef]
- Jander, G.; Kolukisaoglu, U.; Stahl, M.; Yoon, G.M. Editorial: Physiological Aspects of Non-Proteinogenic Amino Acids in Plants. Front. Plant Sci. 2020, 11, 2057. [Google Scholar] [CrossRef]
- Zhao, X.; Tong, C.; Pang, X.M.; Wang, Z.; Guo, Y.; Du, F.; Wu, R. Functional Mapping of Ontogeny in Flowering Plants. Brief. Bioinform. 2011, 13, 317–328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez-Arias, D.; García-Machado, F.J.; Morales-Sierra, S.; Suárez, E.; Pérez, J.A.; Luis, J.C.; Garrido-Orduña, C.; Herrera, A.J.; Valdés, F.; Sandalio, L.M.; et al. Menadione Sodium Bisulphite (MSB): Beyond Seed-Soaking. Root Pretreatment with MSB Primes Salt Stress Tolerance in Tomato Plants. Environ. Exp. Bot. 2019, 157, 161–170. [Google Scholar] [CrossRef]
- HOFFMANN, W.A.; POORTER, H. Avoiding Bias in Calculations of Relative Growth Rate. Ann. Bot. 2002, 90, 37–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Medrano, H.; Tomás, M.; Martorell, S.; Flexas, J.; Hernández, E.; Rosselló, J.; Pou, A.; Escalona, J.-M.; Bota, J. From Leaf to Whole-Plant Water Use Efficiency (WUE) in Complex Canopies: Limitations of Leaf WUE as a Selection Target. Crop J. 2015, 3, 220–228. [Google Scholar] [CrossRef] [Green Version]
- Allen, R.G.; Food and Agriculture Organization of the United Nations (Eds.) Crop Evapotranspiration: Guidelines for Computing Crop Water Requirements; FAO Irrigation and Drainage Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998; ISBN 978-92-5-104219-9. [Google Scholar]
- Leakey, A.D.B.; Ferguson, J.N.; Pignon, C.P.; Wu, A.; Jin, Z.; Hammer, G.L.; Lobell, D.B. Water Use Efficiency as a Constraint and Target for Improving the Resilience and Productivity of C3 and C4 Crops. Annu. Rev. Plant Biol. 2019, 70, 781–808. [Google Scholar] [CrossRef]
- Barrs, H.D.; Weatherley, P.E. A Re-Examination of the Relative Turgidity Technique for Estimating Water Deficits in Leaves. Aust. J. Biol. Sci. 1962, 15, 413–428. [Google Scholar] [CrossRef] [Green Version]
- Kirk, P.L. Kjeldahl Method for Total Nitrogen. Available online: https://pubs.acs.org/doi/pdf/10.1021/ac60038a038 (accessed on 24 November 2021).
- Spinoni, J.; Vogt, J.V.; Naumann, G.; Barbosa, P.; Dosio, A. Will Drought Events Become More Frequent and Severe in Europe? Int. J. Climatol. 2018, 38, 1718–1736. [Google Scholar] [CrossRef] [Green Version]
- Veldkamp, T.I.E.; Wada, Y.; Aerts, J.C.J.H.; Döll, P.; Gosling, S.N.; Liu, J.; Masaki, Y.; Oki, T.; Ostberg, S.; Pokhrel, Y.; et al. Water Scarcity Hotspots Travel Downstream Due to Human Interventions in the 20th and 21st Century. Nat. Commun. 2017, 8, 15697. [Google Scholar] [CrossRef]
- Boretti, A.; Rosa, L. Reassessing the Projections of the World Water Development Report. NPJ Clean Water 2019, 2, 15. [Google Scholar] [CrossRef]
- Martínez-Alvarez, V.; Maestre-Valero, J.F.; González-Ortega, M.J.; Gallego-Elvira, B.; Martin-Gorriz, B. Characterization of the Agricultural Supply of Desalinated Seawater in Southeastern Spain. Water 2019, 11, 1233. [Google Scholar] [CrossRef] [Green Version]
- Christ, K.L.; Burritt, R.L. Water Management Accounting: A Framework for Corporate Practice. J. Clean. Prod. 2017, 152, 379–386. [Google Scholar] [CrossRef]
- Rabêlo, V.M.; Magalhães, P.C.; Bressanin, L.A.; Carvalho, D.T.; dos Reis, C.O.; Karam, D.; Doriguetto, A.C.; dos Santos, M.H.; Santos Filho, P.R.d.S.; de Souza, T.C. The Foliar Application of a Mixture of Semisynthetic Chitosan Derivatives Induces Tolerance to Water Deficit in Maize, Improving the Antioxidant System and Increasing Photosynthesis and Grain Yield. Sci. Rep. 2019, 9, 8164. [Google Scholar] [CrossRef] [PubMed]
- Blum, A. Drought Resistance, Water-Use Efficiency, and Yield Potential—Are They Compatible, Dissonant, or Mutually Exclusive? Aust. J. Agric. Res. 2005, 56, 1159–1168. [Google Scholar] [CrossRef]
- Passioura, J. Increasing Crop Productivity When Water Is Scarce—From Breeding to Field Management. Agric. Water Manag. 2006, 80, 176–196. [Google Scholar] [CrossRef] [Green Version]
- El-Hendawy, S.E.; Kotab, M.A.; Al-Suhaibani, N.A.; Schmidhalter, U. Optimal Coupling Combinations between the Irrigation Rate and Glycinebetaine Levels for Improving Yield and Water Use Efficiency of Drip-Irrigated Maize Grown under Arid Conditions. Agric. Water Manag. 2014, 140, 69–78. [Google Scholar] [CrossRef]
- Goodarzian Ghahfarokhi, M.; Mansurifar, S.; Taghizadeh-Mehrjardi, R.; Saeidi, M.; Jamshidi, A.M.; Ghasemi, E. Effects of Drought Stress and Rewatering on Antioxidant Systems and Relative Water Content in Different Growth Stages of Maize (Zea mays L.) Hybrids. Arch. Agron. Soil Sci. 2015, 61, 493–506. [Google Scholar] [CrossRef]
- Blum, A. Effective Use of Water (EUW) and Not Water-Use Efficiency (WUE) Is the Target of Crop Yield Improvement under Drought Stress. Field Crops Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Da Ge, T.; Sui, F.G.; Nie, S.; Sun, N.B.; Xiao, H.; Tong, C.L. Differential Responses of Yield and Selected Nutritional Compositions to Drought Stress in Summer Maize Grains. J. Plant Nutr. 2010, 33, 1811–1818. [Google Scholar] [CrossRef]
- Rosanoff, A.; Weaver, C.M.; Rude, R.K. Suboptimal Magnesium Status in the United States: Are the Health Consequences Underestimated? Nutr. Rev. 2012, 70, 153–164. [Google Scholar] [CrossRef]
- Hermans, C.; Conn, S.J.; Chen, J.; Xiao, Q.; Verbruggen, N. An Update on Magnesium Homeostasis Mechanisms in Plants. Metallomics 2013, 5, 1170–1183. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Nazim, H.; Liang, Z.; Yang, D. Magnesium Deficiency in Plants: An Urgent Problem. Crop J. 2016, 4, 83–91. [Google Scholar] [CrossRef] [Green Version]
- Ciríaco da Silva, E.; Nogueira, R.J.; Silva, M.; Albuquerque, M. Drought Stress and Plant Nutrition. Plant Stress 2010, 5, 32–41. [Google Scholar]
- Rakszegi, M.; Darkó, É.; Lovegrove, A.; Molnár, I.; Láng, L.; Bedő, Z.; Molnár-Láng, M.; Shewry, P. Drought Stress Affects the Protein and Dietary Fiber Content of Wholemeal Wheat Flour in Wheat/Aegilops Addition Lines. PLoS ONE 2019, 14, e0211892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, D.; Cai, X.; Zhao, J.; Shen, X.; Lu, W. Effects of Drought after Pollination on Grain Yield and Quality of Fresh Waxy Maize. J. Sci. Food Agric. 2015, 95, 210–215. [Google Scholar] [CrossRef]
- Ignjatovic-Micic, D.; Vancetovic, J.; Trbovic, D.; Dumanovic, Z.; Kostadinovic, M.; Bozinovic, S. Grain Nutrient Composition of Maize (Zea mays L.) Drought-Tolerant Populations. J. Agric. Food Chem. 2015, 63, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Qu, Z.; Ma, G.; Wang, W.; Dai, J.; Zhang, M.; Wei, Z.; Liu, Z. Humic Acid Modulates Growth, Photosynthesis, Hormone and Osmolytes System of Maize under Drought Conditions. Agric. Water Manag. 2022, 263, 107447. [Google Scholar] [CrossRef]
- Sandhu, H.; Scialabba, N.E.-H.; Warner, C.; Behzadnejad, F.; Keohane, K.; Houston, R.; Fujiwara, D. Evaluating the Holistic Costs and Benefits of Corn Production Systems in Minnesota, US. Sci. Rep. 2020, 10, 3922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Treatment | Water Regime | |
|---|---|---|
| 100% | 80% | |
| None | WW | WD |
| L-pyroglutamic acid 1 mM | WW-PG | WD-PG |
| Glycine betaine 0.1 mM | WW-GB | WD-GB |
| Treatment | Leaf Lenght (cm) | Leaf Width (cm) | Grain Number | 100 Grain Weight (g) | All Grain Weight (g) |
|---|---|---|---|---|---|
| WW | 77.6 ± 19.6 a | 7.8 ± 1.1 a | 1.2 ± 0.4 a | 10.3 ± 0.5 a | 37.9 ± 5.5 a |
| WD | 66.4 ± 17.3 b | 6.9 ± 1.2 b | 0.8 ± 0.6 b | 8.3 ± 1.4 b | 29.2 ± 8.7 b |
| WW-GB | 73.1 ± 13.1 ab | 7.9 ± 1.2 a | 1.2 ± 0.3 a | 11.2 ± 1.3 c | 42.2 ± 8.5 a |
| WD-GB | 69.2 ± 11.8 ab | 7.5 ± 1 a | 1.1 ± 0.1 a | 10.5 ± 1.1 ac | 37.9 ± 11.2 a |
| WW-PG | 70.2 ± 13.5 ab | 7.5 ± 1.3 a | 1.1 ± 0.3 a | 9.8 ± 1 a | 35.2 ± 2.1 a |
| WD-PG | 69.6 ± 13.3 ab | 7.2 ± 1.1 ab | 1.1 ± 0.1 a | 9.7 ± 1 a | 35.1 ± 2.7 a |
| Treatment | Grain Mass (kg/ha) | WUEg (kg ha/m3) |
|---|---|---|
| WW | 2274 | 0.82 |
| WD | 1168 | 0.52 |
| WW-GB | 2321 | 0.91 |
| WD-GB | 2084 | 0.94 |
| WW-PG | 1936 | 0.70 |
| WD-PG | 1930 | 0.87 |
| Treatment | Mineralogical Compositionin mg/100g Flour | ||||||
|---|---|---|---|---|---|---|---|
| Ca | P | Mg | K | Fe | Cu | Zn | |
| WW | 6.1 ± 0.3 a | 143.9 ± 13 a | 54.7 ± 8.6 a | 309.6 ± 60.7 a | 0.6 ± 0.2 a | 0.3 ± 0.1 a | 0.3 ± 0.1 a |
| WD | 5.0 ± 0.3 b | 195.5 ± 11.7 b | 86.9 ± 9.7 b | 356.4 ± 43.2 a | 0.5 ± 0.1 a | 0.4 ± 0.4 a | 0.4 ± 0.1 a |
| WW-GB | 9.5 ± 1.4 c | 147.7 ± 20.4 a | 71.1 ± 4.7 b | 325.7 ± 40.7 a | 0.8 ± 0.2 ab | 0.2 ± 0.1 a | 0.5 ± 0.1 ab |
| WD-GB | 12.8 ± 1.7 c | 185.7 ± 6.3 b | 71.5 ± 88.4 b | 335.5 ± 41.3 a | 1.1 ± 0.1 b | 0.3 ± 0.1 a | 0.8 ± 0.1 b |
| WW-PG | 10.5 ± 2 c | 131.8 ± 5.8 a | 71.4 ± 5.4 b | 317.0 ± 64.3 a | 0.8 ± 0.1 ab | 1.2 ± 0.4 b | 0.5 ± 0.1 ab |
| WD-PG | 12.4 ± 1.7 c | 189.8 ± 15.1 b | 100.7 ± 4.6 c | 482.1 ± 37.4 b | 1.2 ± 0.2 b | 1.4 ± 0.1 b | 1.4 ± 0.1 c |
| Treatment | Grain Mass (kg/ha) | Water Consumption m3/ha | Profit Using Ground Water €/ha | Profit Using Desalinated Water €/ha |
|---|---|---|---|---|
| WW | 2274 | 967.9 | 389.5 | 99.2 |
| WW-GB * | 2321 | 462.4 | 172.8 | |
| WW-PG+ | 1936 | 117.4 | −172.9 | |
| WD | 1168 | 774.3 | 116.9 | −115.4 |
| WD-GB * | 2084 | 386.7 | 154.4 | |
| WD-PG+ | 1930 | 173.9 | −58.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiménez-Arias, D.; Hernándiz, A.E.; Morales-Sierra, S.; García-García, A.L.; García-Machado, F.J.; Luis, J.C.; Borges, A.A. Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate. Agronomy 2022, 12, 571. https://doi.org/10.3390/agronomy12030571
Jiménez-Arias D, Hernándiz AE, Morales-Sierra S, García-García AL, García-Machado FJ, Luis JC, Borges AA. Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate. Agronomy. 2022; 12(3):571. https://doi.org/10.3390/agronomy12030571
Chicago/Turabian StyleJiménez-Arias, David, Alba E. Hernándiz, Sarai Morales-Sierra, Ana L. García-García, Francisco J. García-Machado, Juan C. Luis, and Andrés A. Borges. 2022. "Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate" Agronomy 12, no. 3: 571. https://doi.org/10.3390/agronomy12030571
APA StyleJiménez-Arias, D., Hernándiz, A. E., Morales-Sierra, S., García-García, A. L., García-Machado, F. J., Luis, J. C., & Borges, A. A. (2022). Applying Biostimulants to Combat Water Deficit in Crop Plants: Research and Debate. Agronomy, 12(3), 571. https://doi.org/10.3390/agronomy12030571