A Study on the Efficiency of Sustainable Wine Grape Vineyard Management Strategies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Site
2.2. The “GreenGrapes” Strategies for the Vineyard Management
2.3. Plant Disease Monitoring Activity and Evaluation of Infections
2.4. Quality of Final Production and Vine Balance
2.5. Sustainability of the Production Processes Assessment
2.6. Stastistical Analysis
3. Results
3.1. Plant Diseases and Evaluation of Infection
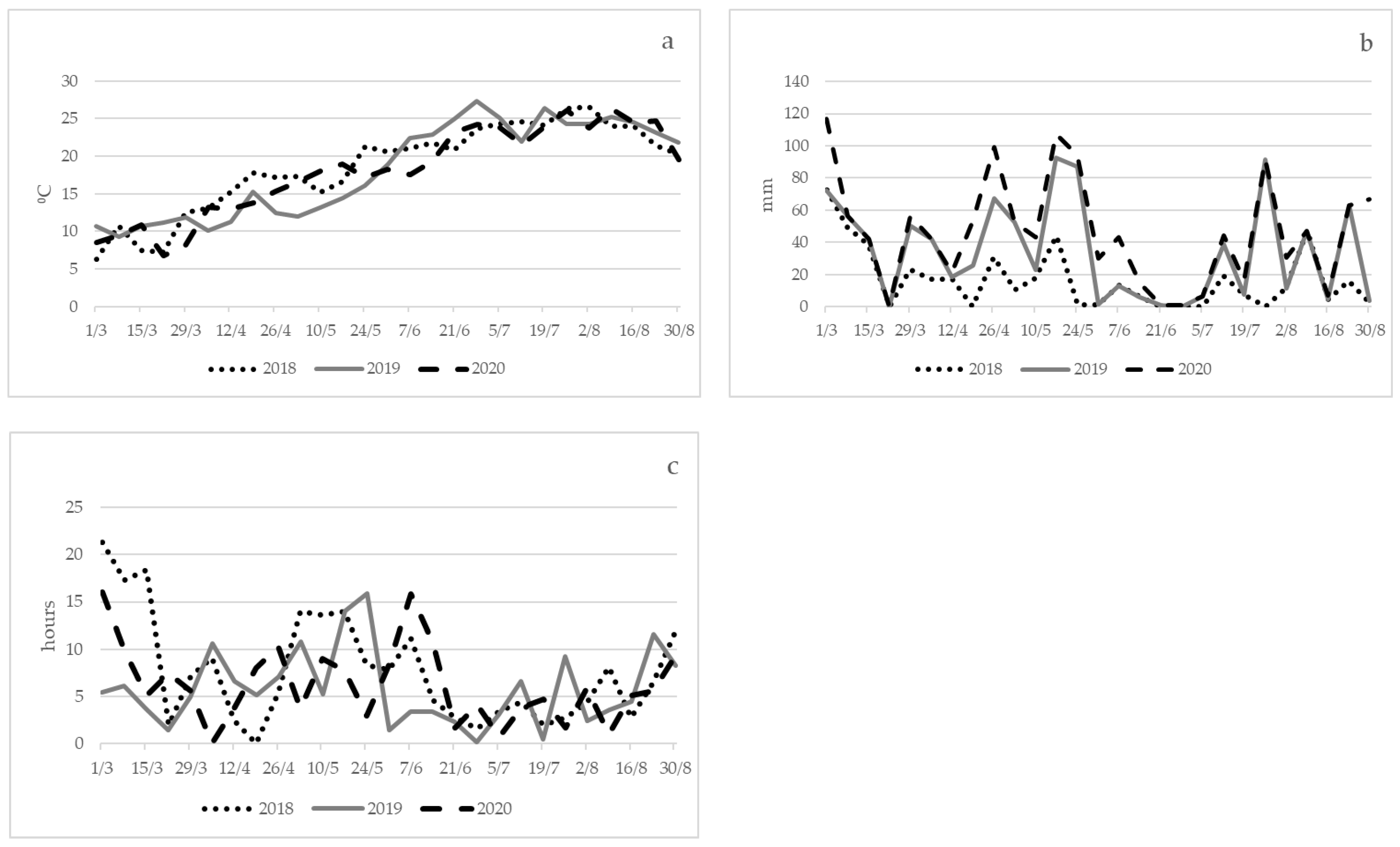
3.1.1. Climate Data: 2018
3.1.2. Climate Data: 2019
3.1.3. Climate Data: 2020
3.2. Quality of Final Production and Vine Balance
3.2.1. Soil Management
3.2.2. Disease Control Management
3.3. Sustainability of the Production Processes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization of the United Nations. 2020. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 3 December 2021).
- Cirillo, C.; Arena, C.; Rouphael, Y.; Caputo, R.; Amitrano, C.; Petracca, F.; De Francesco, S.; Vitale, E.; Erbaggio, A.; Bonfante, A.; et al. Counteracting the Negative Effects of Copper Limitations Through the Biostimulatory Action of a Tropical Plant Extract in Grapevine Under Pedo-Climatic Constraints. Front. Environ. Sci. 2021, 9, 76. [Google Scholar] [CrossRef]
- Damalas, C.A. Safe Food Production with Minimum and Judicious Use of Pesticides. In Food Safety; Selamat, J., Iqbal, S., Eds.; Springer: Cham, Switzerland, 2016; pp. 43–55. [Google Scholar]
- ISTAT—Istituto Nazionale di Statistica—Banca Dati. Available online: http://dati.istat.it/ (accessed on 3 December 2021).
- Provost, C.; Pedneault, K. The organic vineyard as a balanced ecosystem: Improved organic grape management and impacts on wine quality. Sci. Hortic. 2016, 208, 43–56. [Google Scholar] [CrossRef]
- Mackie, K.; Müller, T.; Kandeler, E. Remediation of copper in vineyards–A mini review. Environ. Pollut. 2012, 167, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Brunetto, G.; Miotto, A.; Ceretta, C.A.; Schmitt, D.E.; Heinzen, J.; Moraes, M.P.; Canton, L.; Tiecher, T.L.; Comin, J.J.; Girotto, E. Mobility of copper and zinc fractions in fungicide-amended vineyard sandy soils Arch. Agro. Soil Sci. 2014, 60, 609–624. [Google Scholar] [CrossRef]
- Tiecher, T.L.; Tiecher, T.; Ceretta, C.A.; Ferreira, P.A.A.; Nicoloso, F.T.; Soriani, H.H.; Tassinari, A.; Paranhos, J.T.; De Conti, L.; Brunetto, G. Plant Physiology and Biochemistry Physiological and nutritional status of black oat (Avena strigosa Schreb.) grown in soil with interaction of high doses of copper and zinc. Plant Physiol. Biochem. 2016, 106, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Tiecher, T.L.; Tiecher, T.; Ceretta, C.A.; Ferreira, P.A.; Nicoloso, F.T.; Soriani, H.H.; De Conti, L.; Kulmann, M.S.D.S.; Schneider, R.O.; Brunetto, G. Tolerance and translocation of heavy metals in young grapevine (Vitis vinifera) grown in sandy acidic soil with interaction of high doses of copper and zinc. Sci. Hortic. 2017, 222, 203–212. [Google Scholar] [CrossRef]
- Weng, L.; Temminghoff, E.J.M.; Lofts, S.; Tipping, E.; Van Riemsdijk, W.H. Complexation with Dissolved Organic Matter and Solubility Control of Heavy Metals in a Sandy Soil. Environ. Sci. Technol. 2002, 36, 4804–4810. [Google Scholar] [CrossRef]
- Michaud, A.M.; Chappellaz, C.; Hinsinger, P. Copper phytotoxicity affects root elongation and iron nutrition in durum wheat (Triticum turgidum durum L.). Plant Soil 2008, 310, 151–165. [Google Scholar] [CrossRef]
- Lequeux, H.; Hermans, C.; Lutts, S.; Verbruggen, N. Plant Physiology and Biochemistry Response to copper excess in Arabidopsis thaliana, impact on the root system architecture; hormone distribution; lignin accumulation and mineral profile. Plant Physiol. Biochem. 2010, 48, 673–682. [Google Scholar] [CrossRef]
- De Conti, L.; Ceretta, C.A.; Ferreira, P.A.; Lourenzi, C.R.; Girotto, E.; Lorensini, F.; Tiecher, T.L.; Marchezan, C.; Anchieta, M.G.; Brunetto, G. Soil solution concentrations and chemical species of copper and zinc in a soil with a history of pig slurry application and plant cultivation. Agric. Ecosyst. Environ. 2016, 216, 374–386. [Google Scholar] [CrossRef]
- Cataldo, E.; Fucile, M.; Mattii, G.B. Biostimulants in Viticulture: A Sustainable Approach against Biotic and Abiotic Stresses. Plants 2022, 11, 162. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, M.; Sommaggio, D.; Favretto, M.; Petruzzelli, G.; Pezzarossa, B.; Barbafieri, M. Earthworms as useful bioindicators of agroecosystem sustainability in orchards and vineyards with different inputs. Appl. Soil Ecol. 1998, 10, 137–150. [Google Scholar] [CrossRef]
- Pellegrini, A.; Prodorutti, D.; Frizzi, A.; Gessler, C.; Pertot, I. Development and evaluation of a warning model for the optimal use of copper in organic viticulture. J. Plant Pathol. 2010, 92, 43–55. [Google Scholar]
- Ge, C.R.; Zhang, Q.C. Microbial community structure and enzyme activities in a sequence of copper-polluted soils. Pedosphere 2011, 21, 164–169. [Google Scholar] [CrossRef]
- Mackie, K.A.; Muller, T.; Zikely, S.; Kandeler, E. Long-term copper application in an organic vineyard modifies spatial distribution of soil micro-organisms. Soil Biol. Biochem. 2013, 65, 245–253. [Google Scholar] [CrossRef]
- Fagnano, M.; Agrelli, D.; Pascale, A.; Adamo, P.; Fiorentino, N.; Rocco, C.; Pepe, O.; Ventorino, V. Copper accumulation in agricultural soils: Risks for the food chain and soil microbial populations. Sci. Total Environ. 2020, 734, 139434. [Google Scholar] [CrossRef]
- Karimi, B.; Cahurel, J.-Y.; Gontier, L.; Charlier, L.; Chovelon, M.; Mahé, H.; Ranjard, L. A meta-analysis of the ecotoxicological impact of viticultural practices on soil biodiversity. Environ. Chem. Lett. 2020, 18, 1947–1966. [Google Scholar] [CrossRef]
- Pertot, I.; Delaiti, M.; Mescalchin, E.; Zini, A.; Forti, D. The Activity of New Formulations of Copper Solutions against Downy Mildew; Reduced Doses and Alternative Products to Copper Used in Organic Viticulture; Atti delle Giornate Fitopatologiche: Trento, Italy, 2002; Volume 2, pp. 7–11. [Google Scholar]
- Matasci, C.L.; Gobbin, D.; Schärer, H.-J.; Tamm, L.; Gessler, C. Selection for fungicide resistance throughout a growing season in populations of Plasmopara Viticola. Eur. J. Plant Pathol. 2007, 120, 79–83. [Google Scholar] [CrossRef] [Green Version]
- Dagostin, S.; Schärer, H.-J.; Pertot, I.; Tamm, L. Dagostin, S.; Schärer, H.J.; Pertot, I.; Tamm, L. Are there alternatives to copper for controlling grapevine downy mildew in organic viticulture? Crop Prot. 2011, 30, 776–788. [Google Scholar] [CrossRef]
- Kullaj, E.; Shahini, S.; Varaku, S.; Çakalli, M. Evaluation of the efficacy for reducing copper use against downy mildew control in organic Mediterranean viticulture. Int. J. Pest Manag. 2016, 63, 3–9. [Google Scholar] [CrossRef]
- Pertot, I.; Caffi, T.; Rossi, V.; Mugnai, L.; Hoffmann, C.; Grando, M.; Gary, C.; Lafond, D.; Duso, C.; Thiery, D.; et al. A critical review of plant protection tools for reducing pesticide use on grapevine and new perspectives for the implementation of IPM in viticulture. Crop Prot. 2017, 97, 70–84. [Google Scholar] [CrossRef]
- Hildebrandt, A.; Guillamón, M.; Lacorte, S.; Tauler, R.; Barceló, D. Impact of pesticides used in agriculture and vineyards to surface and groundwater quality (North Spain). Water Res. 2008, 42, 3315–3326. [Google Scholar] [CrossRef]
- Komárek, M.; Čadková, E.; Chrastný, V.; Bordas, F.; Bollinger, J.-C. Contamination of vineyard soils with fungicides: A review of environmental and toxicological aspects. Environ. Int. 2010, 36, 138–151. [Google Scholar] [CrossRef]
- Nash, M.A.; Hoffmann, A.A.; Thomson, L.J. Identifying signature of chemical applications on indigenous and invasive nontarget arthropod communities in vineyards. Ecol. Appl. 2010, 20, 1693–1703. [Google Scholar] [CrossRef]
- Marinho, M.D.C.; Diogo, B.S.; Lage, O.M.; Antunes, S.C. Ecotoxicological evaluation of fungicides used in viticulture in non-target organisms. Environ. Sci. Pollut. Res. 2020, 27, 43958–43969. [Google Scholar] [CrossRef]
- Leroux, P.; Fritz, R.; Debieu, D.; Albertini, C.; Lanen, C.; Bach, J.; Gredt, M.; Chapeland, F. Mechanisms of resistance to fun-gicides in field strains of Botrytis cinerea. Pest Manag. Sci. 2002, 58, 876–888. [Google Scholar] [CrossRef] [PubMed]
- Savocchia, S.; Stummer, B.E.; Wicks, T.J.; Van Heeswijck, R.; Scott, E.S. Reduced sensitivity of Uncinula necator to sterol demethylation inhibiting fungicides in southern Australian vineyards. Aust. Plant Pathol. 2004, 33, 465–473. [Google Scholar] [CrossRef]
- D’Amico, M.; Di Vita, G.; Monaco, L. Exploring environmental consciousness and consumer preferences for organic wines without sulphites. J. Clean. Prod. 2016, 120, 64–71. [Google Scholar] [CrossRef]
- Viana, R.E.; López-Alfaro, I.; López, R.; Santamaría, P.; Gutiérrez, A.R.; González-Arenzana, L. Impact of Chemical and Biological Fungicides Applied to Grapevine on Grape Biofilm, Must, and Wine Microbial Diversity. Front. Microbiol. 2018, 9, 59. [Google Scholar] [CrossRef]
- Jacometti, M.A.; Wratten, S.D.; Walter, M.; Aust, J. Review, Alternatives to synthetic fungicides for Botrytis cinerea management in vineyards. Grape Wine Res. 2010, 16, 154–172. [Google Scholar] [CrossRef]
- Gadoury, D.M.; Pearson, R.C. Ascocarp dehiscence and ascospore discharge in Uncinula necator. Phytopathology 1990, 80, 93–401. [Google Scholar] [CrossRef]
- La Torre, A.; Righi, L.; Iovino, V.; Battaglia, V. Evaluation of copper alternative products to control grape downy mildew in organic farming. J. Plant Pathol. 2019, 101, 1005–1012. [Google Scholar] [CrossRef]
- Benìtez, T.; Rincòn, A.M.; Codòn, A.C. Biocontrol mechanisms of Trichoderma strains. International Microbiology 2004, 7, 249–260. [Google Scholar]
- Kauss, H.; Theisinger-Hinkel, E.; Mindermann, R.; Conrath, U. Dichloroisonicotinic and salicylic acid, inducers of systemic acquired resistance, enhance fungal elicitor responses in parsley cells. Plant J. 1992, 2, 655–660. [Google Scholar] [CrossRef]
- Walters, D.; Walsh, D.; Newton, A.; Lyon, G. Induced Resistance for Plant Disease Control: Maximizing the Efficacy of Resistance Elicitors. Phytopathology 2005, 95, 1368–1373. [Google Scholar] [CrossRef] [Green Version]
- Burketova, L.; Trda, L.; Otto, P.G.; Valentova, O. Bio-based resistance inducers for sustainable plant protection against pathogens. Biotechn. Adv. 2015, 33, 994–1004. [Google Scholar] [CrossRef]
- Pertot, I.; Giovannini, O.; Benanchi, M.; Caffi, T.; Rossi, V.; Mugnai, L. Combining biocontrol agents with different mechanisms of action in a strategy to control Botrytis cinerea on grapevine. Crop Prot. 2017, 97, 85–93. [Google Scholar] [CrossRef]
- Kraeva, E.; Tesniere, C.; Terrier, N.; Romieu, C.; Sauvage, F.X.; Bierne, J.; Deloire, A. Transcription of a β1,3-glucanase gene in grape berries in a late developmental period, or earlier after wounding treatments. Vitis 1998, 37, 107–111. [Google Scholar]
- Renault, A.S.; Deloire, A.; Bierne, J. Pathogenesis-Related proteins induced in grapevine by salicylic acid and Botrytis cinerea. Vitis 1996, 35, 49–52. [Google Scholar]
- Schweikert, C.; Mildner, M.; Vollrath, C.; Kassemeyer Hanns-Heinz, A.H. Systems for testing the efficacy of biofungicides and resistance inducers against downy mildew (REPCO Project). In Organic Eprint; Joint Organic Congress: Odense, Denmark, 2006. [Google Scholar]
- Harm, A.; Kassemeyer, H.-H.; Seibicke, T.; Regner, F. Evaluation of Chemical and Natural Resistance Inducers against Downy Mildew (Plasmopara viticola) in Grapevine. Am. J. Enol. Vitic. 2011, 62, 184–192. [Google Scholar] [CrossRef]
- Reuveni, M.; Zahavi, T.; Cohen, Y. Controlling downy mildew (Plasmopara viticola) in field-grown grapevine with β-aminobutyric acid (BABA). Phytoparasitica 2001, 29, 125–133. [Google Scholar] [CrossRef]
- Pinto, K.M.S.; Nascimento, L.C.D.; Gomes, E.C.D.S.; Da Silva, H.F.; Miranda, J.D.R. Efficiency of resistance elicitors in the management of grapevine downy mildew Plasmopara viticola: Epidemiological, biochemical and economic aspects. Eur. J. Plant Pathol. 2012, 134, 745–754. [Google Scholar] [CrossRef]
- Mancuso, S.; Azzarello, E.; Mugnai, S.; Briand, X. Marine Bioactive Substances (IPA Extract) Improve Foliar Ion Uptake and Water Stress Tolerance in Potted «Vitis vinifera» Plants. Adv. Hort. Sci. 2006, 20, 156–161. [Google Scholar]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef] [Green Version]
- Brown, P.D.; Morra, M.J. Control of soilborne plant pests using glucosinolate-containing plants. Adv. Agron. 1997, 61, 167–231. [Google Scholar]
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef] [Green Version]
- Kuflik, T.; Prodorutti, D.; Frizzi, A.; Gafni, Y.; Simon, S.; Pertot, I. Optimization of copper treatments in organic viticulture by using a web-based decision support system. Comput. Electron. Agric. 2009, 68, 36–43. [Google Scholar] [CrossRef]
- Rossi, V.; Caffi, T.; Legler, S.E.; Carotenuto, E.; Bigot, G. Large-scale application of a web-based Decision Support System for sustainable viticulture. In Proceedings of the IOBC/WPRS Working Group “Integrated protection and production in Viticulture”, Ascona, Switzerland, 13–17 October 2013; Volume 105, pp. 129–136. [Google Scholar]
- Pérez-Expósito, J.P.; Fernández-Caramés, T.M.; Fraga-Lamas, P.; Castedo, L. VineSens: An Eco-Smart Decision-Support Viticulture System. Sensors 2017, 17, 465. [Google Scholar] [CrossRef]
- Landis, D.A.; Wratten, S.D.; Gurr, G.M. Habitat Management to Conserve Natural Enemies of Arthropod Pests in Agriculture. Annu. Rev. Entomol. 2000, 45, 175–201. [Google Scholar] [CrossRef]
- Dane, K.M.; Hogg, B.N.; Wilson, H.; Yokota, G. Native grass ground cover provides multiple ecological services in Californian vineyards. J. Appl. Ecol. 2018, 55, 2473–2483. [Google Scholar] [CrossRef] [Green Version]
- Cherr, C.M.; Scholberg, J.M.S.; McSorley, R. Green Manure Approaches to Crop Production, a synthesis. Agron. J. 2006, 98, 302–319. [Google Scholar] [CrossRef] [Green Version]
- Coll, P.; Le Cadre, E.; Blanchart, E.; Hinsinger, P.; Villenave, C. Organic viticulture and soil quality: A long-term study in Southern France. Appl. Soil Ecol. 2011, 50, 37–44. [Google Scholar] [CrossRef] [Green Version]
- Rotaru, L.; Stoleru, V.; Mustea, M. Fertilization with green manure on Chasselas Doré grape vine as an alternative for sustainable viticulture. J. Food Agric. Environ. 2011, 9, 236–243. [Google Scholar]
- Longa, C.; Nicola, L.; Antonielli, L.; Mescalchin, E.; Zanzotti, R.; Turco, E.; Pertot, I. Soil microbiota respond to green manure in organic vineyards. J. Appl. Microbiol. 2017, 123, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- Zanzotti, R.; Mescalchin, E. Green manure effects on inorganic nitrogen dynamics in soil and its accumulation in grape must. BIO Web Conf. 2019, 13, 04010. [Google Scholar] [CrossRef]
- European and Mediterranean Plant Protection Organisation. EPPO Standards for the Efficacy Evaluation of Plant Protection Products. Available online: https://www.eppo.int/RESOURCES/eppo_standards/pp1_list;PP1/31(1) (accessed on 30 September 2017).
- Saint-Cricq de Gaulejac, N.; Vivas, N.; Glories, Y. Maturation phénolique des raisins rouges. Relation avec la qualité des vins. Comparaison des cépages merlot et tempranillo. Prog. Agric. Et Viticole 1998, 115, 306–318. [Google Scholar]
- Caffi, T.; Rossi, V. Fungicide models are key components of multiple modelling approaches for decision-making in crop protection. Phytopathol. Mediterr. 2018, 57, 153–169. [Google Scholar]
- Krzyzaniak, Y.; Trouvelot, S.; Negrel, J.; Cluzet, S.; Valls, J.; Richard, T.; Bougaud, A.; Jacquens, L.; Klinguer, A.; Chiltz, A.; et al. A plant extract acts both as a resistance inducer and an oomycide against grapevine downy mildew. Front. Plant Sci. 2018, 9, 1085. [Google Scholar] [CrossRef]
- Delaunois, B.; Farace, G.; Jeandet, P.; Clément, C.; Baillieul, F.; Dorey, S.; Cordelier, S. Elicitors as alternative strategy to pesticides in grapevine? Current knowledge on their mode of action from controlled conditions to vineyard. Environ. Sci. Pollut. Res. 2013, 21, 4837–4846. [Google Scholar] [CrossRef]
- Bleyer, G.; Huber, B.; Steinmetz, V.; Kassemeyer, H.H. Growth-models, a tool to define spray intervals against downy mildew (Plasmopara viticola). IOBC WPRS BULLETIN 2003, 26, 7–12. [Google Scholar]
- Bleyer, G.; Lösch, F.; Schumacher, S.; Fuchs, R. Together for the Better: Improvement of a Model Based Strategy for Grapevine Downy Mildew Control by Addition of Potassium Phosphonates. Plants 2020, 9, 710. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Garde-Cerdán, T.; Costa, B.S.-D.; Moreno-Simunovic, Y. Strategies for the improvement of fruit set in Vitis vinifera L. cv. ‘Carménère’ through different foliar biostimulants in two different locations. Ciência E Técnica Vitivinícola 2018, 33, 177–183. [Google Scholar] [CrossRef] [Green Version]
- Frioni, T.; Sabbatini, P.; Tombesi, S.; Norrie, J.; Poni, S.; Gatti, M.; Palliotti, A. Effects of a biostimulant derived from the brown seaweed Ascophyllum nodosum on ripening dynamics and fruit quality of grapevines. Sci. Hortic. 2018, 232, 97–106. [Google Scholar] [CrossRef]
- Salvi, L.; Brunetti, C.; Cataldo, E.; Niccolai, A.; Centritto, M.; Ferrini, F.; Mattii, G.B. Effects of Aschophyllum nodosum extracts on Vitis vinifera: Consequences on plant physiology, grape quality and secondary metabolism. Plant Physiol. Biochem. 2019, 139, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Lisek, J.; Sas-Paszt, L.; Derkowska, E.; Mrowicki, T.; Przybył, M.; Frąc, M. Growth, yelding and healthiness of grapevine cultivars “Solaris” and “Regent” in response to fertilizers and biostimulants. J. Hortic. Res. 2016, 24, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Sabir, A.; Yazar, K.; Sabir, F.; Kara, Z.; Yazici, M.A.; Goksu, N. Vine growth, yield, berry quality attributes and leaf nutrient content of grapevines as influenced by seaweed extract (Ascophyllum nodosum) and nanosize fertilizer pulverizations. Sci. Hortic. 2014, 175, 1–8. [Google Scholar] [CrossRef]
- Taskos, D.; Stamatiadis, S.; Yvin, J.-C.; Jamois, F. Effects of an Ascophyllum nodosum (L.) Le Jol. extract on grapevine yield and berry composition of a Merlot vineyard. Sci. Hortic. 2019, 250, 27–32. [Google Scholar] [CrossRef]
- Basile, B.; Rouphael, Y.; Colla, G.; Soppelsa, S.; Andreotti, C. Appraisal of emerging crop management opportunities in fruit trees, grapevines and berry crops facilitated by the application of biostimulants. Sci. Hortic. 2020, 267, 109330. [Google Scholar] [CrossRef]
- Boselli, M.; Bahouaoui, M.A.; Lachhab, N.; Sanzani, S.M.; Ferrara, G.; Ippolito, A. Protein hydrolysates effects on grapevine (Vitis vinifera L., cv. Corvina) performance and water stress tolerance. Sci. Hortic. 2019, 258, 108784. [Google Scholar] [CrossRef]
- Soppelsa, S.; Kelder, M.; Casera, C.; Bassi, M.; Robatscher, P.; Matteazzi, A.; Andreotti, C. Foliar application of biostimulants promote growth, yeld and fruit quality of strawberry plants grown under nutrient limitation. Agronomy 2019, 9, 483. [Google Scholar] [CrossRef] [Green Version]
- Colla, G.; Nardi, S.; Cardarelli, M.; Ertani, A.; Lucini, L.; Canaguier, R.; Rouphael, Y. Protein hydrolysates as biostimulants in horticulture. Sci. Hortic. 2015, 196, 28–38. [Google Scholar] [CrossRef]
- La Torre, A.; Talocci, S.; Spera, G.; Valori, R. Control of downy mildew on grapes in organic viticulture. Commun. Agric. Appl. Biol. Sci. 2008, 73, 169–178. [Google Scholar] [PubMed]
- Feliziani, E.; Santini, M.; Landi, L.; Romanazzi, G. Pre- and postharvest treatment with alternatives to synthetic fungicides to control postharvest decay of sweet cherry. Postharvest Biol. Technol. 2013, 78, 133–138. [Google Scholar] [CrossRef]

| Active Ingredient | Commercial Product | Supplier | Years | Number of Applications for Treatment | Dose | |||
|---|---|---|---|---|---|---|---|---|
| IPM | IPM-GG | ORG | ORG-GG | Kg-l ha−1 | ||||
| Dimetomorf, Metiram | Forum top | Basf Italia S.p.a. | 2018 | 1 | 1 | 2.5 | ||
| 2019 | 3 | 3 | 2.5 | |||||
| 2020 | 3 | 1 | 2.5 | |||||
| Fluopicolide, Fosetyl Al | R6 Erresei Albis | Bayer | 2018 | 1 | 1 | 3 | ||
| 2019 | 2 | 1 | 3 | |||||
| 2020 | 2 | 2 | 3 | |||||
| Cymoxanil. Fosetyl Al, Copper | Vitene triplo | Sipcam Italia | 2018 | 1 | 1 | 4 | ||
| 2019 | 1 | 4 | ||||||
| 2020 | 1 | 4 | ||||||
| Metalaxil, Copper | Planet C | Upl Europe Ltd. | 2018 | 1 | 1 | |||
| 2019 | 1 | 1 | 4 | |||||
| 2020 | 4.5 | |||||||
| Mandipropamid, Zoxamide | Ampexio | Syngenta Italia | 2018 | 3 | 1 | 0.5 | ||
| 2019 | 1 | 0.5 | ||||||
| 2020 | 1 | 0.5 | ||||||
| Copper (Bordeaux mixture) | Bordoflow new | Manica S.p.a. | 2018 | 2 | 2 | 4 | 1 | 3.5 |
| 2019 | 3 | 4 | ||||||
| 2020 | 1 | 4 | ||||||
| Tribasic copper sulphate | Cuprofix ultra dispress | Upl Europe Ltd. | 2018 | 1 (2.1) * | 10 (1.7) | 10 (1.3) | 1 | |
| 2019 | 7 | 5 | ||||||
| 2020 | 2 (1.5) | 1 (1.5) | 9 (1) | 6 (1) | ||||
| Original blend of bioavailable nutrients (Mn. Zn). Combination of bacterial metabolites and enzymatic compounds | Pur’apres | Tailor’d Wine Design | 2018 | 5 | 1.5 | |||
| 2019 | 2 | 1.5 | ||||||
| 2020 | 3 | 1 | 1.5 | |||||
| Original blend of bioavailable nutrients (Mn. Zn). Combination of bacterial metabolites and enzymatic compounds | Pur’avant | Tailor’d Wine Design | 2018 | 1 | 1 | |||
| 2019 | ||||||||
| 2020 | 2 | 1 | ||||||
| Solid extract of alfalfa, algae and molasses | K&A Oomisine 2.0 | Kalos | 2018 | 1 | 2.5 | |||
| 2019 | 1 | 2.5 | ||||||
| 2020 | 1 | 2.5 | ||||||
| Solid extract of alfalfa, algae and molasses | K&A Evidence 2.0 | Kalos | 2018 | 2 | 2 | |||
| 2019 | 1 | 2 | ||||||
| 2020 | 2 | 2 | ||||||
| Fluid yeast extract containing brown algae | K&A Frontiere 2.0 | Kalos | 2018 | 2 | 2 | 0.75 | ||
| 2019 | 1 | 0.75 | ||||||
| 2020 | 2 | 1 | 0.75 | |||||
| Plant extracts | Dinamico | Fertenia | 2018 | 1 | 2 | 2.5 | ||
| 2019 | ||||||||
| 2020 | 2 | 2 | 2.5 | |||||
| Zeolite | Zeolite Fertenia | Fertenia | 2018 | 1 (2.5) | 5 (3) | 1 (2.5) | 1 (3) | |
| 2019 | 2 (4.5) | 4 (5.25) | 2 (4.5) | 3 (4) | ||||
| 2020 | 4 | 1 | 6 | 6 | ||||
| Sweet orange oil | Prev’am | Nufarm Italy | 2018 | 1 | 1 | 1.6 | ||
| 2019 | 2 | 1.6 | ||||||
| 2020 | 1 | 2 | 1.6 | |||||
| Botanical Family | Species |
|---|---|
| Fabaceae (55%) | Vicia faba L. (30%) |
| Vicia sativa L.(10%) | |
| Pisum sativum L. (10%) | |
| Trifolium incarnatum L. (5%) | |
| Graminaceae (38%) | Avena sativa L. (12–18%) |
| Hordeum vulgare L. (16–20%) | |
| Brassicaceae (5%) | Sinapis alba L. (3%) |
| Eruca sativa M. (2%) | |
| Hydrophyllaceae (2%) | Phacelia spp (2%) |
| Phenological State | Organ | Number of Organs Monitored | 2018 | 2019 | 2020 |
|---|---|---|---|---|---|
| BBCH 107-109 | Leaves | 100 | 6-May | 7-May | 5-May |
| BBCH 110-113 | Leaves | 100 | 18-May | 15-may | 13-May |
| BBCH 57-69 | Leaves/Cluster | 100/100 | 31-May | 30-May | 28-May |
| BBCH 65-73 | Leaves/Clusters | 100/100 | 6-June | 7-June | 3-June |
| BBCH 72-77 | Leaves/Clusters | 100/100 | 20-June | 19-June | 19-June |
| BBCH 79 | Leaves/Clusters | 100/100 | 9-July | 5-July | 3-July |
| BBCH 85-89 | Leaves/Clusters | 100/100 | 24-July | 28-July | 27-July |
| Year | Treatment | Leaf Incidence % | Leaf Severity % | Cluster Incidence % | Cluster Severity % |
|---|---|---|---|---|---|
| 2018 | IPM | 19.5 ± 4.55 b | 1.79 ± 0.98 b | 4.0 ± 3.16 c | 0.22 ± 0.23 b |
| ORG | 20.56 ± 5.50 b | 2.32 ± 0.47 b | 12.5 ± 6.69 c | 0.91 ± 0.29 b | |
| IPM-GG | 43.47 ± 8.5 a | 3.74 ± 0.99 ab | 32.0 ± 6.32 b | 4.05 ± 1.97 a | |
| ORG-GG | 45.50 ± 10.23 a | 4.94 ± 1.83 a | 53.5 ± 20.15 a | 5.20 ± 2.26 a | |
| 2019 | IPM | 3.0 ± 1.0 a | 0.075 ± 0.02 b | 0.0 | 0.0 |
| ORG | 2.0 ± 2.0 a | 0.325 ± 0.33 b | 0.0 | 0.0 | |
| IPM-GG | 2.5 ± 0.87 a | 0.150 ± 0.59 b | 0.0 | 0.0 | |
| ORG-GG | 3.5 ± 0.93 b | 1.0 ± 0.37 a | 0.0 | 0.0 | |
| 2020 | IPM | 1.88 ± 0.65 c | 0.10 ± 0.02 c | 0.38 ± 0.09 c | 0.02 ± 0.004 c |
| ORG | 24.75 ± 1.80 b | 3.38 ± 0.45 b | 6.88 ± 1.24 b | 1.49 ± 0.10 ab | |
| IPM-GG | 22.75 ± 4.87 b | 3.03 ± 0.9 b | 5.75 ± 1.30 b | 1.30 ± 0.18 b | |
| ORG-GG | 61.75 ± 2.50 a | 9.13 ± 1.65 a | 41.40 ± 5.63 a | 17.98 ± 1.44 a |
| Protection Protocol | Soil Management | Yield (Kg/vine) | Bunch Weight (g) | Bunch (n/Plant) | Wood Production (g/Plant) | Ravaz Index |
|---|---|---|---|---|---|---|
| IPM | g | 1.3 ± 0.5 a,b | 111.1 ± 36.9 a | 12 ± 5.2 e | 276.7 ± 57.9 a | 5.1 ± 1.6 a |
| m | 1.6 ± 0.7 c | 154 ± 37.3 b,c | 10.9 ± 4.7 d | 295.5 ± 100.9 a | 5.8 ± 1.6 a | |
| IPM-GG | g | 1.1 ± 0.5 a | 130.7 ± 36.8 a,b | 8.7 ± 3.3 a,b | 282.5 ± 112.6 a | 5.8 ± 19 a |
| m | 1.7 ± 1 c | 165.3 ± 55.2 c,d | 10.1 ± 4 c,d | 419.6 ± 47.3 b | 4.1 ± 2.3 a | |
| ORG | g | 1.5 ± 0.6 b,c | 183.1 ± 57 d,e | 8.5 ± 2.8 a,b,c | 277.3 ± 33.6 a | 4.4 ± 17 a |
| m | 1.5 ± 0.8 b,c | 225.7 ± 60.8 f | 6.9 ± 2.9 a | 332.5 ± 69.9 a | 5.5 ± 2.1 a | |
| ORG-GG | g | 1.2 ± 0.5 a | 155.4 ± 37.1 b,c | 7.2 ± 2 a,b | 275 ± 51.7 a | 4.1 ± 1.2 a |
| m | 1.7 ± 0.6 c | 195.9 ± 61.7 e | 8.9 ± 2.8 b,c | 317.3 ± 33.6 a | 5.6 ± 1.7 a |
| Defense Protocol | Soil Conduction | TSS (°Brix) | TA (g/L) | Total Anthocyanins (mg/Kg (+)-Malvidin) | Extractable Anthocyanins (mg/Kg Catechin 3 Glucoside) |
|---|---|---|---|---|---|
| ORG-GG | g | 24.2 ± 1.5 b | 5,6 ± 0.5 a,b | 1542 ± 271 a,b,c | 754 ± 158 a,b,c |
| m | 23.7 ± 1.6 a,b | 5,5 ± 0.4 a,b | 1430 ± 262 a,b | 727 ± 128 a,b | |
| ORG | g | 23.9 ± 0.9 a,b | 5,6 ± 0.5 a,b | 1681 ± 242 c | 792 ± 103 a |
| m | 23.7 ± 0.8 a,b | 5,6 ± 0.5 a | 1386 ± 225 a | 707 ± 141 a,b,c | |
| IPM-GG | g | 23.9 ± 1.6 a,b | 5.9 ± 0.7 a,b | 1655 ± 243 b,c | 847 ± 167 c |
| m | 23.6 ± 1.4 a,b | 5.8 ± 0.5 a,b | 1576 ± 285 a,b,c | 761 ± 172 a,b,c | |
| IPM | g | 23.9 ± 1.3 a,b | 5.5 ± 0.5 b | 1641 ± 291 b,c | 827 ± 132 b,c |
| m | 23.0 ± 2.2 a | 5.7 ± 0.6 a,b | 1532 ± 247 a,b,c | 793 ± 168 a,b,c |
| Disease Control Management | Active Molecule | Farm (kg/ha) | GG (kg/ha) | Reduction (%) |
|---|---|---|---|---|
| Ametoctradin | 0.09 | 0.00 | 100 | |
| Boscalid | 0.20 | 0.00 | 100 | |
| Cymoxanil | 0.14 | 0.07 | 47 | |
| Dimethomorph | 0.45 | 0.23 | 48 | |
| IPM | Fluopicolide | 0.22 | 0.13 | 39 |
| Fosetyl-Al | 4.54 | 3.07 | 32 | |
| Mancozeb | 0.33 | 0.00 | 100 | |
| Mandipropamide | 0.12 | 0.04 | 64 | |
| Metiram | 2.55 | 1.83 | 28 | |
| Metrafenone | 0.20 | 0.11 | 46 | |
| Myclobutanil | 0.08 | 0.00 | 100 | |
| Copper oxychloride | 0.79 | 0.39 | 50 | |
| Quinoxyfen | 0.09 | 0.03 | 61 | |
| Copper sulphate | 2.22 | 0.88 | 60 | |
| Zoxamide | 0.12 | 0.04 | 66 | |
| ORGANIC | Copper | 5.2 | 3.9 | 25.1 |
| Sulphur | 21.89 | 17.55 | 20 |
| Disease Control Management | Soil Management | Human Tox Score | Treatment Frequency Index | Eco Tox Score | Carbon Footprint (t CO2 eq/t of Production) | Carbon Sequestration (t of C/ha) | Water footprint (m3 of Water/t Production) |
|---|---|---|---|---|---|---|---|
| IPM | g | 141.5 | 22.46 | 169.4 | 0.3 | 1.36 | 1419.67 |
| m | 141.57 | 22.46 | 169.43 | 0.28 | 1.54 | 1348.00 | |
| IPM-GG | g | 107.83 | 16 | 114.73 | 0.34 | 1.25 | 1181.33 |
| m | 107.83 | 16 | 114.73 | 0.27 | 1.76 | 1021.00 | |
| ORG | g | 63.83 | 21.8 | 39.83 | 0.31 | 1.45 | 1844.00 |
| m | 63.83 | 21.8 | 39.93 | 0.33 | 1.59 | 1839.33 | |
| ORG-GG | g | 60.77 | 21.26 | 38.83 | 0.37 | 1.26 | 1769.33 |
| m | 61 | 21 | 38.83 | 0.28 | 1.61 | 1570.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perria, R.; Ciofini, A.; Petrucci, W.A.; D’Arcangelo, M.E.M.; Valentini, P.; Storchi, P.; Carella, G.; Pacetti, A.; Mugnai, L. A Study on the Efficiency of Sustainable Wine Grape Vineyard Management Strategies. Agronomy 2022, 12, 392. https://doi.org/10.3390/agronomy12020392
Perria R, Ciofini A, Petrucci WA, D’Arcangelo MEM, Valentini P, Storchi P, Carella G, Pacetti A, Mugnai L. A Study on the Efficiency of Sustainable Wine Grape Vineyard Management Strategies. Agronomy. 2022; 12(2):392. https://doi.org/10.3390/agronomy12020392
Chicago/Turabian StylePerria, Rita, Alice Ciofini, William Antonio Petrucci, Mauro Eugenio Maria D’Arcangelo, Paolo Valentini, Paolo Storchi, Giuseppe Carella, Andrea Pacetti, and Laura Mugnai. 2022. "A Study on the Efficiency of Sustainable Wine Grape Vineyard Management Strategies" Agronomy 12, no. 2: 392. https://doi.org/10.3390/agronomy12020392
APA StylePerria, R., Ciofini, A., Petrucci, W. A., D’Arcangelo, M. E. M., Valentini, P., Storchi, P., Carella, G., Pacetti, A., & Mugnai, L. (2022). A Study on the Efficiency of Sustainable Wine Grape Vineyard Management Strategies. Agronomy, 12(2), 392. https://doi.org/10.3390/agronomy12020392






